Abstract
To investigate the effects of bilateral subthalamic nucleus (STN) stimulation on patterns of brain activation during random number generation (RNG), a task that requires suppression of habitual counting and response selection under competition. We used
Keywords
Introduction
Random number generation (RNG) is a cognitive task that involves response selection by suppression of habitual responses (counting in series) (Baddeley et al, 1998; Jahanshahi et al, 1998, 2000b; Dirnberger et al, 2005). Random number generation is attention demanding and engages several related executive processes including strategic response selection through suppression of habitual responses, monitoring the responses generated (i.e. working memory), and altering generation strategies to avoid stereotyped output (Baddeley et al, 1998; Jahanshahi et al, 1998). The advantage of RNG over the Stroop or verbal fluency is that RNG performance remains constant within and across trials and a direct quantitative measure of the subject's ability to suppress habitual responses is available through the calculation of an index, Count score 1 (CS1) (Jahanshahi et al, 2000b). The CS1 score measures the tendency to count in ascending or descending series in steps of 1, for example, 1-2-3 or 8-7-6. According to the ʻnetwork modulation model' of RNG, to generate numbers in a random fashion subjects have to suppress habitual counting in series which represents spreading activation of strongly associated representations in a ʻnumber associative network' distributed in the temporal cortex. This suppression is achieved through the negative modulatory influence of the dorsolateral prefrontal cortex (DLPFC) over the temporal cortex, to prevent spreading activation and to allow strategic response selection (Jahanshahi et al, 1998, 2000b; Jahanshahi and Dirnberger, 1999). In support of the network modulation model of RNG, in young and elderly healthy subjects, compared with counting in series, RNG is associated with activation of the DLPFC and anterior cingulate cortex (ACC) and deactivation of the temporal cortex (Jahanshahi et al, 2000b; Daniels et al, 2003; Dirnberger et al, 2005). Furthermore, as proposed by the network modulation model, the primacy of suppression of habitual responses to performance of RNG was demonstrated by the significant negative correlation between regional cerebral blood flow (rCBF) in the prefrontal cortex and CS1 scores and conversely a positive correlation between rCBF in the temporal cortex and CS1 scores (Jahanshahi et al, 2000b; Dirnberger et al, 2005). In addition, studies using repetitive transcranial magnetic stimulation rTMS over the prefrontal cortex have established a significant increase in CS1 or stereotyped/habitual responses, suggesting that when the modulatory influence of the prefrontal cortex is disrupted, it is the suppression of habitual responses which is specifically altered, without any major effects on other measures of randomness (Jahanshahi et al, 1998; Jahanshahi and Dirnberger, 1999). Patients with Parkinson's disease (PD) show higher CS1 than matched controls and differentially greater increase in CS1 at faster rates of RNG (Brown et al, 1998; Dirnberger et al, 2005). Compared with elderly controls, this increased nonrandomness of responses during RNG in PD has been shown to be associated with (i) no activation of medial frontal areas (supplementary motor area (SMA) and ACC) (ii) a failure to modulate prefrontal and temporal activation as a function of task (RNG versus serial counting) and rate, and (iii) a deficit which was associated with altered rCBF in the internal segment of the globus pallidus (GPi), the final basal ganglia output pathway to the frontal cortex (Dirnberger et al, 2005).
As the early 1990s, deep brain stimulation (DBS) of the subthalamic nucleus (STN) has been shown to significantly improve the motor symptoms of PD (Limousin et al, 1995). In carefully selected PD patients, DBS of the STN does not produce any major adverse effects on intellectual ability or general cognitive functioning (Ardouin et al, 1999). However, detailed neuropsychological assessment has revealed consistent deficits on two cognitive tasks with DBS of the STN: verbal fluency and Stroop (Ardouin et al, 1999; Saint-Cyr et al, 2000; Jahanshahi et al, 2000a; Schroeder et al, 2002, 2003; Witt et al, 2004), which imaging has shown to be associated with reduced prefrontal or ACC activation compared with when the stimulators are off (Schroeder et al, 2002, 2003). In 16 PD patients with DBS of the STN, we (Jahanshahi et al, in preparation) recently found that at a fast rate of 1 Hz, STN stimulation decreased the ability of patients to generate numbers randomly and resulted in a significant increase of CS1 scores by 25.7% (mean 110.2 ± 9.5) compared with when the stimulators were off (mean 81.9 ± 7.2).
The aim of the present study was to identify the neural correlates of the PD patients' reduced ability to select responses randomly and to investigate the changes in brain activation induced by DBS of the STN during RNG performed at a fast rate of 1 Hz. Based on the commonalities of the processes involved, we predicted that STN stimulation would be associated, as for verbal fluency and Stroop, with lower prefrontal and ACC activation during performance of the RNG task compared with when the stimulators are off.
Methods
Patients
Six right-handed PD patients (mean age: 59.5 ± 4.6 years; mean disease duration: 15.2 ± 4.9 years; three males and three females) participated. The PD patients fulfilled the UK Parkinson's disease Brain Bank criteria for idiopathic PD. They suffered from akinetic-rigid symptoms and were treated by bilateral STN stimulation for at least 3 months (range 3 to 60 months). To ensure the absence of dementia and clinical depression, the patients were screened on the Mini Mental State Examination and the Beck Depression Inventory. Two patients reported mild or moderate depression on the Beck Depression Inventory, but none were clinically depressed or received antidepressant treatment. All patients received a combination of dopamine agonists and levodopa. The implantation of the electrodes (Model 3389; Medtronic, Minneapolis, MN, USA) was performed under local anaesthesia and guided by stereotactic magnetic resonance imaging (MRI). The absence of surgical damage and accurate placement of the electrodes were confirmed on the postoperative MRI. The electrodes were connected to a telemetrically controllable pulse generator (Kinetra or Itrell II, Medtronic). On the day of the positron emission tomography (PET) scan, the patients were off antiparkinsonian drugs for at least 12 h. The motor section of the Unified Parkinson's Disease Rating Scale (UPDRS), was completed just before the scan, both with bilateral stimulation on and off. The significant improvement of parkinsonian motor symptoms with DBS in every patient was a further (although not absolute) indication of the accurate positioning of the electrodes (see ‘Results’). The main demographic and clinical characteristics of the patients, the electrical stimulation parameters used, and the effects of STN stimulation on the UPDRS motor scores are presented in Table 1.
Main characteristics of the parkinsonian patients
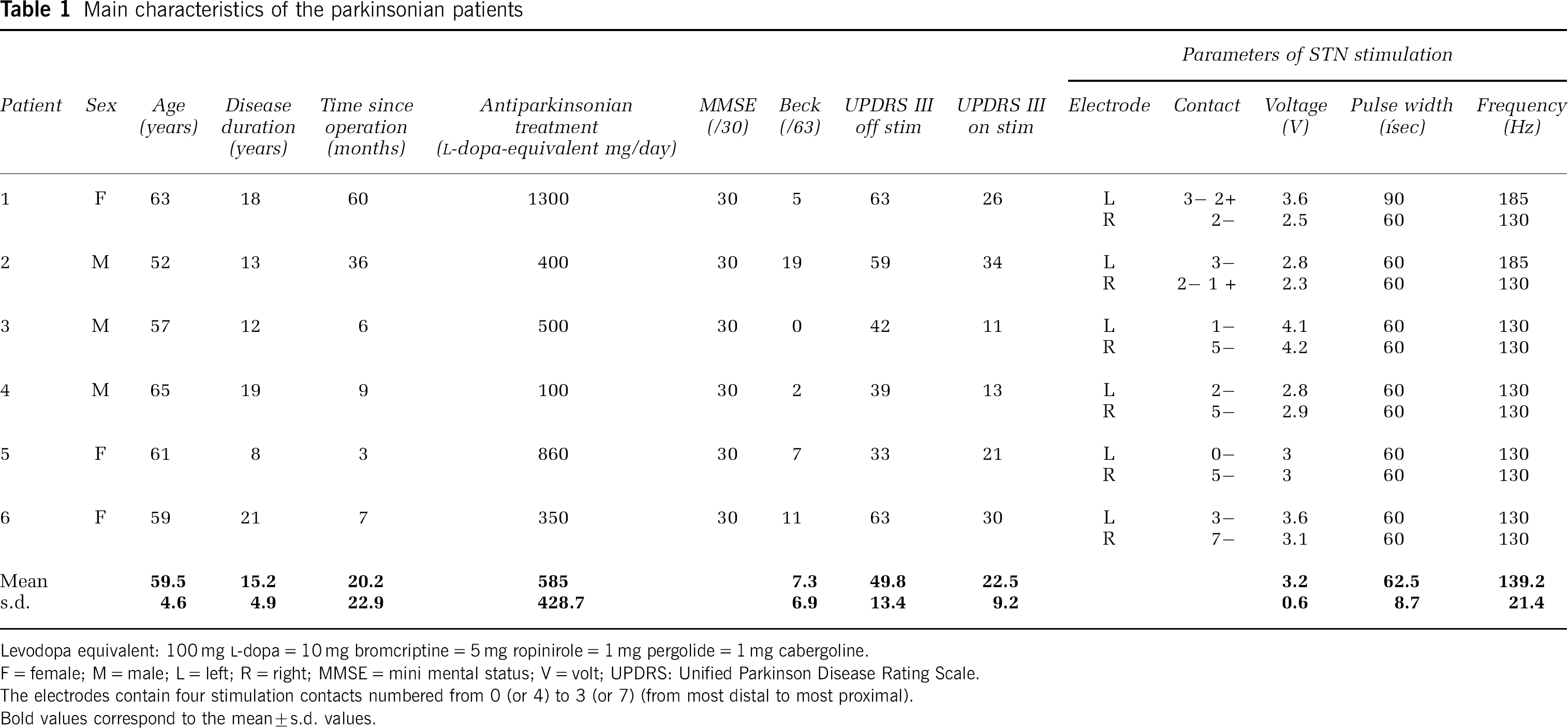
Levodopa equivalent: 100 mg
F = female; M = male; L = left; R = right; MMSE = mini mental status; V = volt; UPDRS: Unified Parkinson Disease Rating Scale.
The electrodes contain four stimulation contacts numbered from 0 (or 4) to 3 (or 7) (from most distal to most proximal).
Bold values correspond to the mean ± s.d. values.
The study was approved by the Joint Ethics Committee of the Institute of Neurology & the National Hospital for Neurology & Neurosurgery, as well as the Ethics Committee of the Hammersmith Hospital. All patients participated after the aims of the study and the nature of the procedures had been fully explained. They signed an informed consent form according to the Declaration of Helsinki.
Design
We used a 2 × 2 factorial parametric design with Task (RNG or counting (COUNT)) and STN Stimulation condition (On or Off) as the two factors.
Cognitive Tasks
Procedure: Each task was performed at 1 Hz, thus requiring a response every second. During the PET scan performance of both RNG and COUNT was paced by a tone presented at 1 Hz through a tape recorder. The loudness was adjusted to be comfortable for each patient. Patients were instructed to synchronize their responses with the tone as accurately as possible. With the counting task, patients were told to repeatedly count from 1 to 9 for 2.5 mins. During the RNG task, patients were instructed to generate numbers between 1 and 9 in a random fashion for 2.5 mins. The concept of randomness was explained using the analogy of picking out numbers out of a ʻhat’ (Jahanshahi et al, 1998). The order of tasks (RNG versus Count) and conditions (DBS On versus Off) was randomized across scans for each patient. The following four conditions were studied: (1) RNG-On: RNG with bilateral STN stimulation On; (2) RNG-Off: RNG with bilateral STN stimulation Off; (3) COUNT-On: counting task with bilateral STN stimulation On; (4) COUNT-Off: counting task with bilateral STN stimulation Off. Each condition was repeated three times in a randomized order leading to a total number of 12 scans per subject except for one subject who performed each condition only twice because of pain. The delay between the change of stimulation condition and the following scan was 8 mins (interval between the scans).
Measures of Randomness: We obtained measures of seriation, count scores, which have previously been found to show differential change as a function of rate of performing RNG (Jahanshahi et al, 1998). The CS1 measures the tendency to count in ascending or descending series in steps of 1. For example, 1-2-3 or 8-7-6. CS1 takes the length of the series into account and in calculating CS1, the sequence length is squared to give higher weights to longer runs of counting. Individuals may have a CS1 that is lower than predicted from a random series if they are avoiding particular counting tendencies or they may have a CS1, which is too high if they cannot suppress counting tendencies.
PET Scans
Patients were positioned supine on the scanner bed, with the eyes closed. The head was maintained in a fixed position using a moulded ʻhelmet' worn on the head. Head position was checked before and after each scan using a laser alignment together with reference points on the Reid's line. Positron emission tomography scans were obtained using a Siemens CTI HR + + /966 tomograph (CTI/Siemens, Knoxville, TN, USA) in a three-dimensional mode. The camera has an axial field of view of 23.4 cm and provides 95 transaxial planes. A correction for tissue attenuation of 511 keV γ-radiation was measured with a 5 mins three-dimensional transmission scan performed before tracer injection and acquired using a retractable 137Cs source. Images were reconstructed using a ramp filter yielding a full-width half-maximum spatial resolution of 5.1 mm (x) × 5.1 mm (y) × 5.9 mm (z).
Measurements of rCBF were estimated for each activation scan by recording the distribution of radioactivity after an intravenous injection more than 20 secs of 6 mCi (222 MBq) of [15O]H2O through a forearm catheter placed into the left brachial vein. The integrated counts were collected for 90 secs, starting 30 secs after the injection. An interscan interval of 8 mins was necessary between experimental conditions to achieve adequate decay of radioactivity.
PET Images Transformation and Statistical Analysis: Image analysis was performed in MATLAB 5.3 (Math Works, Natick, MA, USA) using SPM99 software (Wellcome Department of Cognitive Neurology, London, UK) for statistical parametric mapping (Friston et al, 1994). Individual PET scans were oriented parallel along the intercommisural line using an averaged image from each patient, and then normalized into a standard stereotactic space (Talairach and Tournoux, 1988). Images were smoothed using an isotropic 14-mm kernel to account for variation in gyral anatomy and individual variability in structure—function relationships, and to improve the signal/noise ratio. Variations in global flow across subjects and scans were removed by proportionally scaling each image to have an arbitrary level of 50 mL/100 mL/min.
For analysing the effect of stimulation on RNG and COUNT, a multisubjects × conditions and covariates model was used. The following contrasts were performed:
Main Effect of STN Stimulation: The contrast (RNG-On + COUNT-On)–(RNG-Off + COUNT-Off) assessed the increase of rCBF related to the stimulation On compared with the stimulation Off state, independently of the cognitive task. The contrast (RNG-Off + COUNT-Off) – (RNG-On + COUNT-On) assessed the decrease of rCBF related to the stimulation On compared with the stimulation Off state independently of the cognitive task.
Main Effect of Task: The contrast (RNG-On + RNG-Off)–(COUNT-On + COUNT-Off) assessed the increase of rCBF during RNG compared with COUNT, irrespective of stimulation condition. The contrast (COUNT-On +COUNT-Off)–(RNG-On +RNG-Off) assessed the decrease of rCBF during the RNG compared with COUNT, across the two stimulation conditions.
rCBF Changes Induced by STN Stimulation During RNG
Simple Effect of Stimulation: The RNG-On—RNG-Off contrast assessed the increase of rCBF during the RNG task with DBS On compared with DBS Off. The contrast RNG-Off—RNG-On assessed the decrease of rCBF during RNG task with DBS On compared with DBS Off. These contrasts do not take into account the effects of DBS during the control task (COUNT) and, thus cannot distinguish these effects from those which are specific of the cognitive task (RNG).
Interaction: The contrast (RNG-On—COUNT-On)–(RNG-Off—COUNT-Off) assessed the increase of rCBF during the RNG task relative to the COUNT task with DBS On compared with DBS Off. The contrast (RNG-Off—COUNT-Off)–(RNG-On—COUNT-On) assessed the decrease of rCBF during the RNG task relative to the COUNT task with DBS On compared with DBS Off stimulation.
rCBF Changes Induced by STN Stimulation During COUNT
Simple Effect of Stimulation: The COUNT-On—COUNT-Off contrast assessed the increase of rCBF during the COUNT task with DBS On compared with DBS Off. The contrast COUNT-Off—COUNT-On assessed the decrease of rCBF during the COUNT task with DBS On compared with DBS Off.
Interaction: the contrast (COUNT-On—RNG-On)–(COUNT-Off—RNG-Off) assessed the increase of rCBF during the COUNT task relative to the RNG task with DBS On compared with DBS Off. The contrast (COUNT-Off—RNG-Off)–(COUNT-On—RNG-On) assessed the decrease of rCBF during the COUNT task relative to the RNG task with DBS On compared with DBS Off stimulation.
Psychophysiological Interaction: As STN DBS modifies cortical activation through its influence on the GPi, the main output structure from the basal ganglia, we determined the interaction between the GPi and other brain regions during RNG depending on the stimulation condition using a psychophysiological interaction analysis (Friston et al, 1997; Lee et al, 2003). More precisely, the psychophysiological interaction analysis assesses how the effective connectivity (slope of the regression line) between an index area (here the GPi) and other areas changes with a specific experimental manipulation or context (here the stimulation condition) (Friston et al, 1997; Lee et al, 2003). In SPM, having included the effects of the physiological component (rCBF in the right GPi) and the experimental or ʻpsychological' component (STN stimulation On versus STN stimulation Off during RNG) in the same model, a new covariate was generated. Then we determined the positive and negative correlations with this covariate, which allowed us to identify areas in which the degree of effective connectivity with the GPi is modulated by STN stimulation during RNG. In other words, a significant increase in the regression slope (positive interaction) would mean that during RNG, STN stimulation increases the effective connectivity between the GPi as the index area with other brain regions. A significant decrease of the regression slope would suggest the opposite.
Global differences in cerebral blood flow were covaried out for all voxels, comparisons across conditions were made using t-statistics with appropriate linear contrasts. Only voxels that exceeded a threshold of uncorrected P-value ≤0.001 were considered as significant in a region comprising at least 15 voxels. For regions with a strong a priori hypothesis (i.e. cingulate cortex, internal pallidum, DLPFC, temporal cortex), small volume corrections were applied irrespective of the voxel uncorrected P-value, and a voxel-based corrected P-value <0.05 considered as significant. All coordinates reported are based on the Talairach atlas and were derived from procedures developed by M Brett (http://www.mrc-cbu.cam.ac.uk/Imaging).
Results
Effect of STN Stimulation on the Motor Symptoms of PD
Off medication, the mean scores on the UPDRS motor scale were 49. 8 (s.d. = 13.4) with the stimulators off and 22.5 (s.d. = 9.2) with the stimulators on. A paired t-test was conducted to compare UPDRS motor scores in the two conditions (DBS On versus Off). Subthalamic nucleus stimulation induced a significant improvement of motor symptoms with a mean 54.8% reduction of the UPDRS motor scores (P < 0.001). This notable improvement of the motor symptoms of PD was present in every case (see Table 1).
Effect of STN Stimulation on Measures of Synchronization and Randomness during RNG
During the PET scan, the total time to generate 100 random numbers was not different for stimulation on (125.75 ± 8.38) or off (124.03 ± 9.71) (P > 0.05). During the scan, for paced RNG at 1 Hz, the mean CS1 score was 65.2 ± 26.7 with DBS Off and 73.1 ± 39.7 with DBS On. This 10.8% increase of the CS1 scores (i.e. reduced randomness) with stimulation was not significant at P < 0.05 (P = 0.08), but this was largely due to the small sample size. Indeed, the significance of this tendency was confirmed, as mentioned in the introduction, in a larger group of 16 PD patients including all six patients from the current PET study.
Changes of rCBF
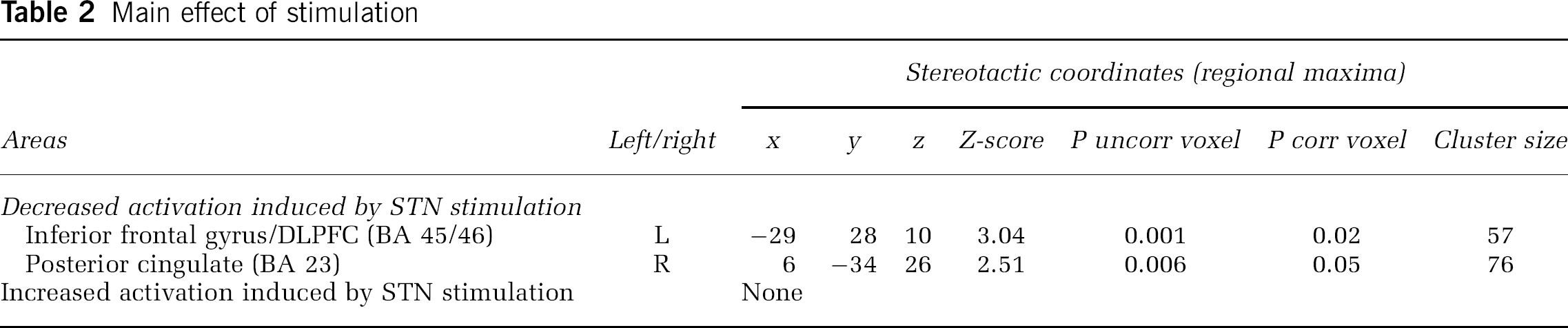
Main Effect of STN Stimulation: The contrast (RNG-Off + COUNT-Off)–(RNG-On + COUNT-On) showed that across the two tasks, DBS of the STN induced a significant reduction of rCBF in the left inferior frontal gyrus/DLPFC (BA 46/45) and in the right posterior cingulate cortex (PCC) (BA 23).
As shown by the contrast (RNG-On + COUNT-On)–(RNG-Off+COUNT-Off), no significant rCBF increase was observed with STN stimulation. These results are presented in Table 2.
Main effect of stimulation
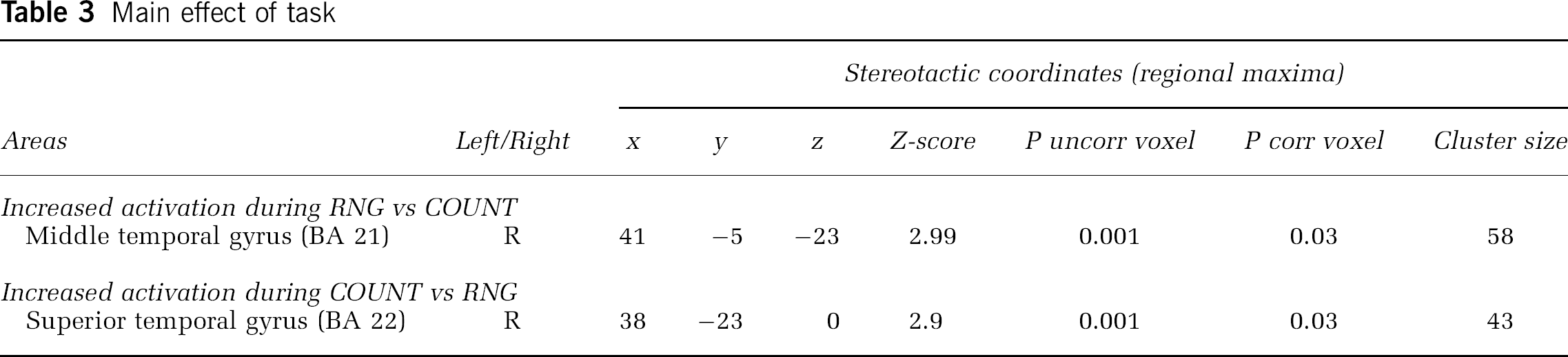
Main Effect of Task: The contrast (RNG-On + RNG-Off)–(COUNT-On + COUNT-Off), showed that irrespective of stimulation condition, during the RNG task there was a significant rCBF increase in the right middle temporal gyrus (BA 21). The contrast (COUNT-On+COUNT-Off)–(RNG-On+RNG-Off) showed that during the COUNT task, a significant rCBF increase was found in the right superior temporal gyrus (BA 22). These results are presented in Table 3.
Main effect of task
rCBF Changes Induced by STN Stimulation during RNG
Simple Effect of Stimulation: These results are presented in Table 4. The contrast RNG-Off—RNG-On showed that during the RNG task, STN stimulation induced a significant decrease of rCBF in the left inferior frontal gyrus/DLPFC (BA 46), left PCC (BA 23) and right ACC (BA 24) (Figure 1A). The contrast RNG-On—RNG-Off established that during RNG, STN stimulation induced a significant rCBF increase in the right GPi (Figure 1B). The differences in thresholds adopted for the two sets of analyses was responsible for the apparent discrepancy between the main effect of stimulation and the effect of stimulation during RNG. In fact, for the main effect of stimulation there was a trend for increased rCBF in the right GPi (coordinates: x = 18; y = 0; z = 2; Z-score = 2.03; P uncorrected = 0.021) when an uncorrected P was considered.
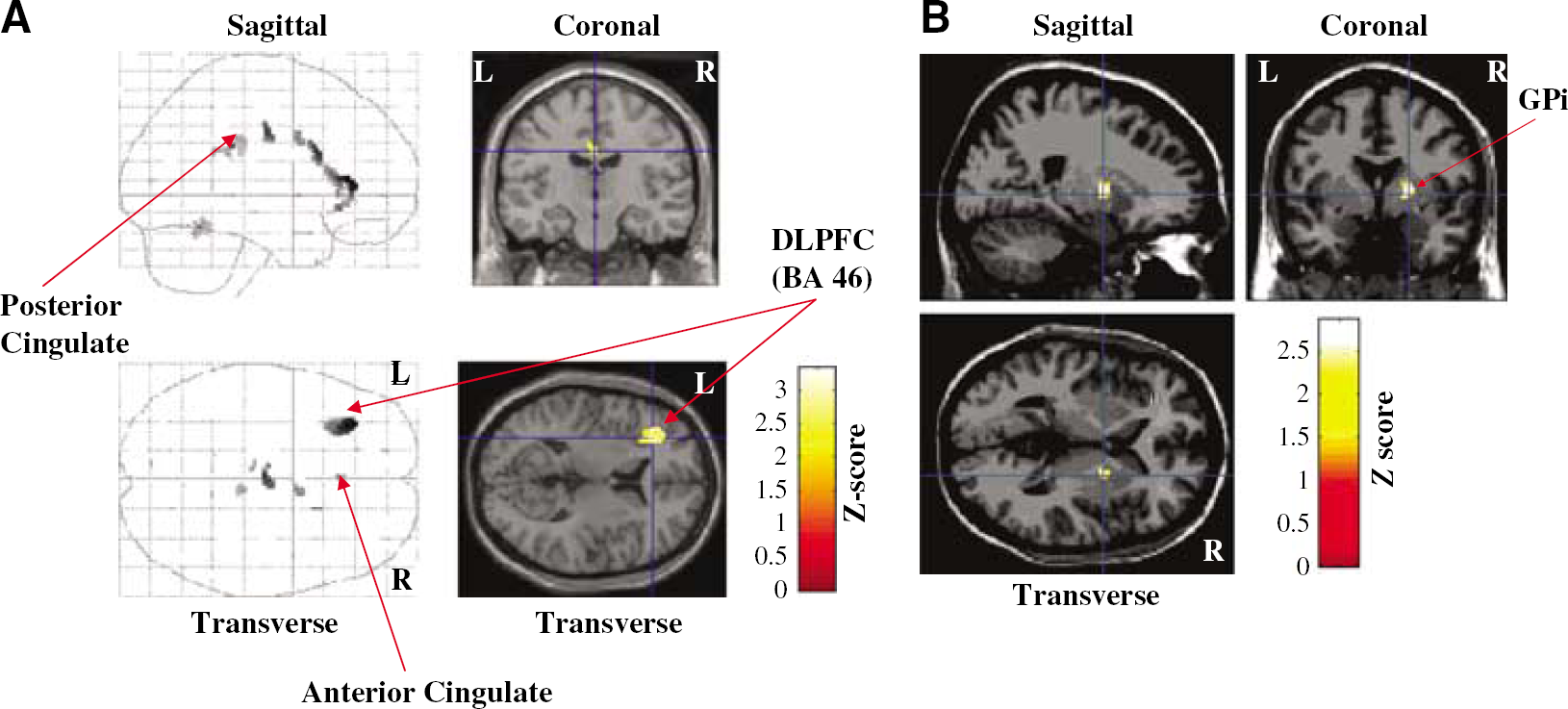
(
Modifications of rCBF induced by STN stimulation during RNG
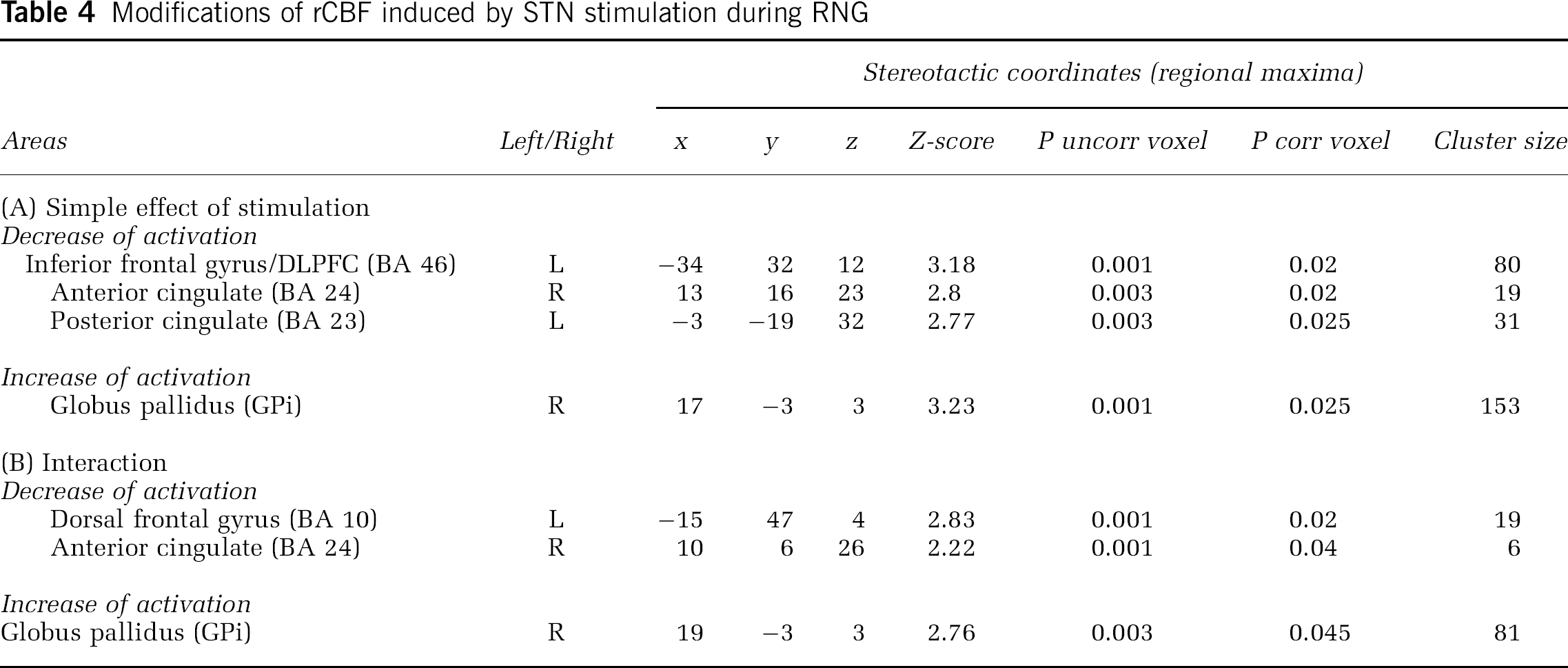
Interaction: The contrast (RNG-Off—COUNT-Off)–(RNG-On—COUNT-On) showed that during the RNG task, STN stimulation induced a significant decrease of rCBF in the left dorsal frontal gyrus (BA 10) and right ACC. The contrast (RNG-On—COUNT-On)–(RNG-Off—COUNT-Off) showed that during RNG, STN stimulation induced a significant rCBF increase in the right GPi. These results are presented in Table 4.
rCBF Changes Induced by STN Stimulation during COUNT: rCBF changes induced by STN stimulation during COUNT, was examined with the following contrasts: COUNT-On—COUNT-Off; COUNT-Off-COUNT-On; (COUNT-On—RNG-On)–(COUNT-Off—RNG-Off); (COUNT-Off—RNG-Off)–(COUNT-On—RNG-On). No significant rCBF increase or decrease was induced by STN stimulation during COUNT in either the simple effect or interaction.
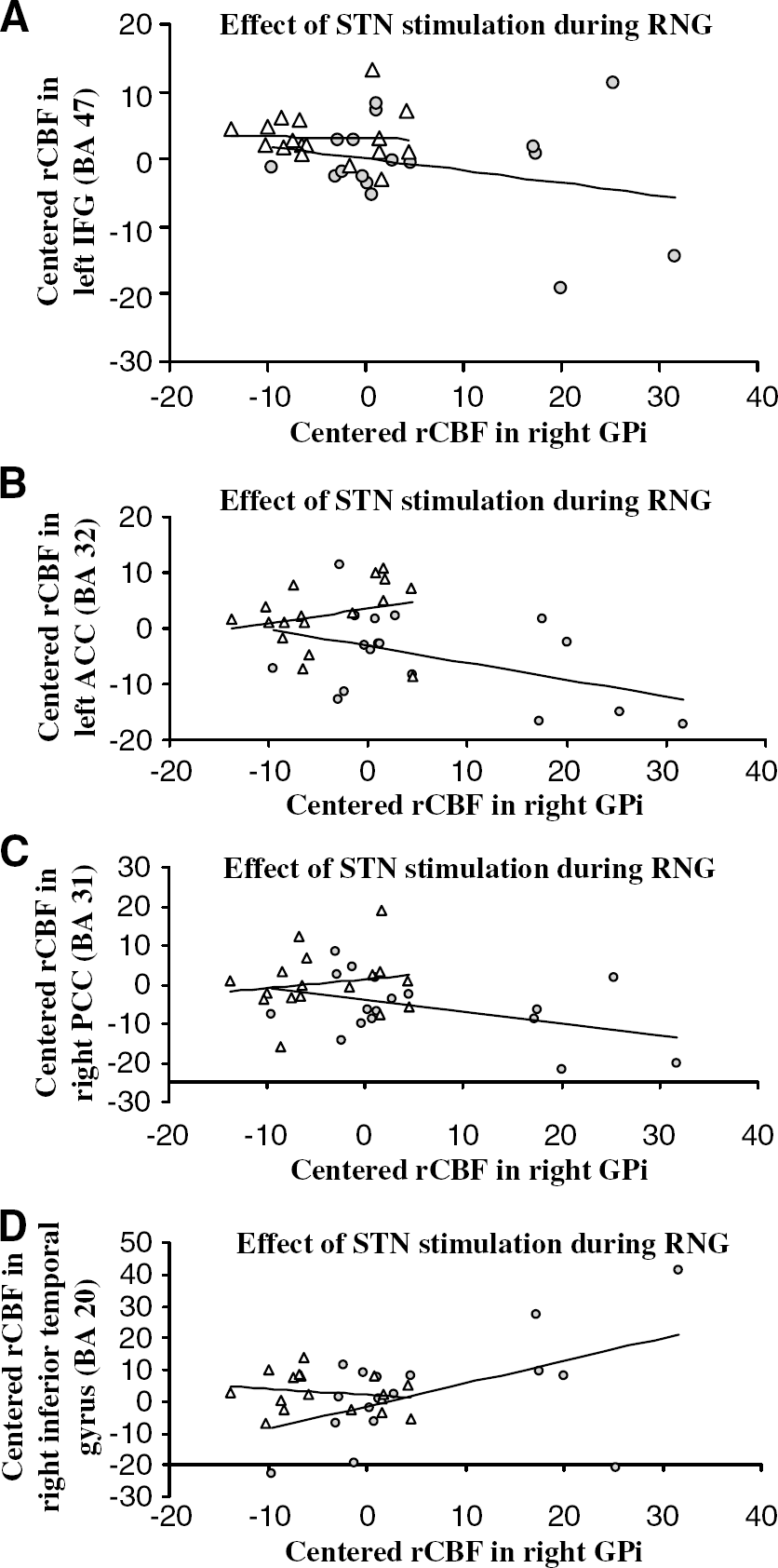
Psychophysiological Interactions: During RNG, compared with when the stimulators were off, there was a significant decrease in the regression slopes between the GPi and the left ACC, left inferior frontal gyrus, right PCC, and cerebellar vermis with stimulation of the STN. This indicated that STN stimulation reduced the effective connectivity or coupling between these regions and the GPi during RNG, relative to when the stimulators were off. This negative covariation means that greater activation of the GPi during RNG with STN stimulation is associated with lower activation in the left ACC, right PCC, left inferior frontal gyrus, and cerebellar vermis. In contrast, with DBS of the STN, a significant positive regression was noted between the right GPi and the left superior temporal gyrus (BA 38) and the right inferior temporal gyrus (BA 20). This indicates that STN stimulation increased the coupling between the GPi and the temporal gyrus during RNG compared with when the stimulators were off. These results are presented in Table 5 and Figure 2.
Psychophysiological interactions. (
Psychophysiological interactions
Discussion
DBS of the STN produced a significant 55% improvement in the motor symptoms of PD, an improvement which was evident in every case. With paced RNG at the fast rate of 1 Hz, STN stimulation had a negative effect on performance, leading to reduced randomness and increased habitual counting; although this did not reach significance for the sample of 6, the DBS On versus Off effect was significant for a larger sample. During RNG at the fast rate of 1 Hz, STN stimulation induced a reduction of rCBF in the left dorsal frontal gyrus, left inferior frontal gyrus, left DLPFC, left posterior, and right ACC and an increase of rCBF in the right GPi, compared with DBS Off. Psychophysiological interactions revealed that compared with when the stimulators were off, STN stimulation was associated with negative pallidal—prefrontal/cingulate coupling and positive pallidal—temporal cortex effective connectivity during RNG.
Changes in Brain Activation with DBS of the STN during RNG
During RNG, STN stimulation decreased rCBF in the prefrontal cortex (dorsolateral and inferior parts), ACC and PCC. Concomitantly rCBF increased in the right GPi.
Prefrontal Cortex: The negative impact of STN stimulation on fast paced RNG, resulting in reduced randomness of the numbers generated, was associated with a reduced activation of the inferior frontal gyrus/DLPFC (BA 46) (simple effect of stimulation on RNG) and the dorsal frontal gyrus (BA 10) (interaction analysis). These results are in line with the crucial role of the prefrontal cortex, particularly the dorsolateral and inferior sections, in response selection during RNG (Jahanshahi et al, 1998; Dirnberger et al, 2005). The DLPFC is involved in response selection through inhibition of competing and inappropriate responses (Jahanshahi et al, 2000b; Frith, 2000). There is evidence from functional imaging or behavioural studies of random response generation in patients with frontal lesions or frontal dysfunction for such a role for the DLPFC (Frith et al, 1991; Deiber et al, 1991; Daniels et al, 2003; Dirnberger et al, 2005). Further imaging studies have more clearly established that the specific role of the DLPFC is in response selection rather than in working memory processing (Rowe and Passingham, 2001). In addition, use of transcranial magnetic stimulation has established that disrupting processing in the left DLPFC with transcranial magnetic stimulation over this area interfered with random response selection and increased generation of habitual or stereotyped responses and slowed response times on a motor response selection task without any working memory demands (Jahanshahi et al, 1998; Jahanshahi and Dirnberger, 1999). This clearly establishes that the DLPFC is essential for response selection, which is also its role in RNG.
As noted below, the STN stimulation-induced reduction of rCBF was observed for the ACC as well as the prefrontal cortex. Interestingly, similar STN-stimulation induced reduction of ACC or inferior prefrontal activation has been observed respectively during the Stroop and phonemic verbal fluency (Schroeder et al, 2002, 2003). As all these tasks involve response selection through suppression of dominant/habitual responses, taken together these results suggest that in PD, DBS of the STN induces a deficit in response selection under competition, which is mediated by a reduction of prefrontal and ACC activation and altered pallidal—prefrontal coupling. The direct and indirect pathways from the striatum to the output nuclei of the basal ganglia, that is to the GPi and the substantia nigra pars reticulata, are considered to produce opposing effects to respectively facilitate or suppress cortically initiated activity (Alexander and Crutcher, 1990). Activation of the direct pathway between the putamen and GPi/substantia nigra pars reticulata results in disinhibition of thalamic nuclei, which in turn facilitate cortically initiated activity. In contrast, the net effect of activity in the indirect pathway from the STN to the GPi/substantia nigra pars reticulata via the external segment of the globus pallidus (GPe) and the STN is increased inhibition of thalamic targets and consequently reduced thalamic input to cortical areas. Within the fronto-striatal circuits, the direct and indirect pathways constitute an ideal system for response selection under competition, with the indirect pathway inhibiting inappropriate responses to allow selection of the appropriate response through the direct pathway (Chevalier and Deniau, 1990). Thus, a key mechanism for the response selection process would be the focusing of activation in the basal ganglia circuits, mediated through the D1 and D2 receptors, respectively, in the direct and indirect pathways. Therefore, one would predict that an inhibition of the direct pathway by D1 receptors antagonists would impair performance on task such as RNG, whereas D2 receptors antagonists would have the opposite effect. In fact, a role for the basal ganglia in action selection under competition has been proposed (Mink and Thach, 1993). For example, Mink and Thach (1993) proposed a ʻcentre-surround' inhibition model, based on the two pathways from the cortex to the basal ganglia output through the GPi namely via the STN or striatum, acting in parallel to inhibit potentially competing programmes and to enable the cortically selected programme. The present results and those of Schroeder et al (2002, 2003) suggest that for tasks such as RNG, phonemic verbal fluency, and the Stroop which involve response selection under competition, STN stimulation alters/interferes with suppression of habitual or prepotent responses necessary for selection of the appropriate response. This would account for the increased habitual counting during RNG observed in the present study, and worse performance on phonemic verbal fluency and reduced accuracy on the Stroop reported with STN stimulation compared with when the stimulators are switched off (Schroeder et al, 2002, 2003; Jahanshahi et al, 2000a).
Anterior and Posterior Cingulate Cortices: In the present study, STN stimulation induced a reduction of rCBF during RNG in the ACC and PCC. This is in agreement with previous studies showing activation of the ACC during RNG or other motor or cognitive tasks involving random response generation (Frith et al, 1991; Deiber et al, 1991; Jahanshahi et al, 2000b; Daniels et al, 2003; Dirnberger et al, 2005). The ACC is also frequently recruited in tandem with the DLPFC in the course of dual or effortful task performance or in tasks such as the Stroop, which require response selection (naming ink colour) through suppression of a dominant but inappropriate responses (reading the words) (Carter et al, 1998). In such tasks, the role of the ACC is considered to be one of maintaining ʻattention-to-action' or ʻconflict' monitoring (Frith et al, 1991; Deiber et al, 1991; Carter et al, 1998; Jahanshahi et al, 2000b; Daniels et al, 2003; Dirnberger et al, 2005). In a previous study, during RNG, PD patients failed to activate the ACC and the SMA, activations which were observed in the elderly controls (Dirnberger et al, 2005).
Subthalamic nucleus stimulation also induced a decrease of rCBF in the PCC during RNG, leading to a global deactivation of the cingulate. The PCC receives projections from the anterior nuclei of the thalamus and is involved in emotional processing as well as in episodic memory (Maddock et al, 2003). A functional MRI study has shown that this posterior part of the cingulate plays a role in the modulation of memory processes by emotions (Maddock et al, 2003). In addition, it has been showed that the PCC has a role in attentional processing, and is involved in monitoring errors in a GO-NO-GO task, similar to the ACC (Mesulam et al, 2001). Thus, the DBS-induced reduction of activation in the PCC could reflect disruption of attentional, mnemonic or error processing during RNG all of which are essential for performance during this task.
Temporal Cortex: Regardless of the stimulation condition, rCBF in the middle temporal gyrus increased during RNG and rCBF in the superior temporal gyrus increased during COUNT. These results confirm the importance of keeping activation in the middle and superior temporal gyri low during RNG and are in agreement with the proposed role of the temporal cortex in generating an automatic series of numbers through spreading activation in a number associative network (Jahanshahi et al, 2000b). In healthy subjects, performance of the control counting task at faster rates is associated with increased rCBF in the right temporal cortex) (Jahanshahi et al, 2000b). As previously noted, according to the network modulation model of RNG, in controls, at slow rates of RNG when the subjects can keep up with the pacing tone, strategic selection of numbers in a random fashion is achieved through the modulatory and inhibitory control of the DLPFC over the temporal cortex, the putative site of the number associative network so as to prevent spreading activation which would otherwise have resulted in habitual counting in series. At faster rates of paced RNG, when task demands exceed capacity, the modulatory prefrontal—temporal interaction breaks down and prefrontal cortex activation decreases while temporal cortex activation increases simultaneously, leading to a reduction of randomness and a tendency to count (Jahanshahi et al, 2000b). In the present study, the activation of the middle temporal gyrus during paced RNG at 1 Hz suggests that, at this fast rate, patients could not keep up with task demands and simply activated the temporal cortex without showing any significant differential activation of the prefrontal cortex relative to the counting task with the stimulator off. The present data are consistent with the network modulation of RNG, and establish that STN stimulation has opposite effects on rCBF in the prefrontal and temporal cortices during RNG as borne out by the opposite effect of STN stimulation on the patterns of pallidal—prefrontal (negative coupling with DBS On) and pallidal—temporal (positive coupling with DBS On) connectivity during RNG.
Internal Pallidum: During RNG, STN stimulation induced a rCBF increase in the right GPi. This increase is very specific, as it was only found for the RNG X Stimulation interaction. The increased rCBF in the right GPi is associated with a reduction of the randomness during the RNG task.
Why did bilateral STN stimulation only alter rCBF in the right GPi during RNG? Interestingly, in PD, Dirnberger et al (2005) also found an abnormal modulation as a function of rate of RNG for the right but not the left GPi. One explanation could be that RNG involves mental imagery of the numbers, which is associated with right-sided subcortical activation similar to right-sided cortical visuo-spatial representation (Owen et al, 1998; Dirnberger et al, 2005).
Psychophysiological interactions using the GPi as the index area showed that during RNG STN stimulation modulated the interaction between this area and the prefrontal and temporal cortex, the ACC and the PCC. More precisely we observed 1) a ʻnegative' coupling between the GPi and the inferior frontal cortex (BA 47), ACC, PCC, and the cerebellar vermis and 2) a ʻpositive' coupling between the GPi and the left superior temporal gyrus and right inferior temporal gyrus. These interactions show changes in effective connectivity between the GPi and these cortical areas, depending on the stimulation condition. A change of coupling between the GPi and other brain areas can be interpreted either as a change in the influence of the GPi on other regions or as a change in the responsiveness of the GPi to inputs from these other regions (Friston et al, 1997; Lee et al, 2003). In the present study, the analysis of the regression slopes showed that with the stimulators on, an increase of rCBF in the GPi was associated with a decrease of rCBF in the left inferior frontal gyrus, ACC, and the right PCC. Conversely, increased rCBF in the GPi with stimulation was associated with an increased rCBF in the temporal cortex. In light of the organization of the striato-frontal circuitry and the direct effect of STN DBS on the GPi, it seems more likely that during RNG, STN DBS modulates the influence of the GPi, the main output structure from the basal ganglia, on cortical regions. These results suggest that during RNG, STN stimulation induced a switch from a GPi-prefrontal-cingulate network to a GPi-temporal pathway. These altered and opposite patterns of pallidal—prefronal and pallidal—temporal connectivity with stimulation compared with DBS Off are consistent with the changes in performance induced by stimulation and the predictions of the network modulation model of RNG.
During RNG, DBS of the STN also induced a negative pallidal coupling with the vermis. Activation of the vermis during paced RNG may relate to the synchronization demands of the task. In a previous study of RNG in young healthy subjects, activation of the dentate nucleus showed rate-dependent change suggesting a role in synhcronization (Jahanshahi et al, 2000b).
In the present study, during RNG the increase of rCBF in the GPi combined with a decrease of rCBF in prefrontal and cingulate cortices suggests an excitatory effect of STN stimulation on axonal projections from the STN to the GPi. This subcortically driven inhibition of a prefrontal-cingulate network was supported by the psychophysiological interaction showing that, during RNG, STN stimulation ʻdecreased' the functional coupling between the GPi and the prefrontal and cingulate cortices. It is tempting to link these results with microdialysis experiments showing that STN stimulation induced glutamate release in the ipsilateral GPi suggesting activation of STN efferent output (Stefani et al, 2005). However, despite evidence showing a tight coupling between rCBF and neuronal activity, there is no proof that an increased glutamate release would result in increased rCBF in the GPi (Shmuel et al, 2006).
Differential Effects of STN Stimulation on Motor Versus Cognitive Tasks: The principal evoked mechanism of action of STN stimulation is an inhibition of the STN itself. Electrophysiological studies have shown that high-frequency stimulation of the STN decreased activity of STN and substantia nigra cells, while it increased thalamic ventrolateral nucleus cell activity (e.g. Benazzouz et al, 2000). According to the current model of bradykinesia in PD this inhibition of the STN would reduce GPi activity leading to a release of its inhibitory influence on the thalamus which would in turn restore cortical activity (Dostrovsky and Lozano, 2002). These electrophysiological data fit well with the modifications of brain energy metabolism induced by STN DBS. Indeed, it has been showed using PET and 18F-FDG that STN stimulation induced a metabolic reduction in the GPi and midbrain and a metabolic increase in the thalamus, frontal, parietal, occipital and temporal cortex, and cerebellum (Hilker et al, 2004; Vafaee et al, 2004; Trost et al, 2006). This is also in line with the increase of rCBF induced by STN stimulation in the prefrontal, premotor, and cingulate cortices during a limb movement observed in numerous studies (Limousin et al, 1997; Ceballos-Baumann et al, 1999; Thobois et al, 2002). However, at rest, the consequences of STN stimulation are opposite, and an increase of GPi activation and a concomitant reduction of frontal activation are observed, which would suggest an inhibition of cortical projections (Ceballos-Baumann et al, 1999; Hershey et al, 2003). In addition, for a given motor task, the changes of cerebral activation depend on the electrical parameters of DBS. Indeed, unilateral low-frequency STN stimulation leads to increased rCBF in the ACC controlateral to hand movement and bilatearlly in the SMA. An additional increase of rCBF in the DLPFC is found with high-frequency STN stimulation. Finally, bilateral high-frequency STN stimulation induces an increase of rCBF in the same regions as well as in the GP bilaterally (Strafella et al, 2003). Therefore, the consequences of STN stimulation appear to be task and stimulation parameters-dependent. This conclusion is supported by the present results, showing that the rCBF decrease in the prefrontal and cingulate areas and the increased rCBF in GPi were specific to the RNG task and not observed for the COUNT task. Similarly, the rCBF decreases in prefrontal or cingulate cortices or the ventral striatum reported by Schroeder et al (2002, 2003) were specific to the verbal fluency and Stroop tasks and not the control tasks. The proposal that the effects of DBS STN are task-specific and depend on the particular networks engaged by specific tasks is in agreement with the results of animal studies suggesting that striato-frontal connectivity as well functional connectivity between different nuclei of the basal ganglia depend on brain state, such as whether the animal is at rest or active (Magill et al, 2004). In fact, based on a review of the evidence from a variety of sources, it has been proposed (McIntyre et al, 2004) that the mechanism of action of STN stimulation is not simply inhibitory or excitatory but depends on more complex modifications of activity in a pathological network. Furthermore, the effect of stimulation on the soma and on the axons of STN neurons is not the same, with STN stimulation having an inhibitory effect on the soma and an excitatory effect on the projections from the neurons and on fibres passing near the STN (McIntyre et al, 2004). It is possible that the balance between the inhibition of the soma and the excitation of the axons could be different for specific neural networks engaged by particular tasks, which could explain the differential effect of STN stimulation on the different fronto-striatal circuits during motor versus cognitive tasks.
Conclusion
The present study has established that STN stimulation has a detrimental effect on effortful tasks such as RNG, which require suppression of habitual or prepotent responses to allow strategic response selection. This effect is in contrast to the beneficial effect of DBS of the STN on limb movements and the associated increase of rCBF in prefrontal, premotor, and cingulate cortices (Limousin et al, 1997). The mechanisms for these task-specific and differential effects of STN stimulation, as suggested by the present and other functional imaging studies, appears to be, on the one hand, an STN stimulation induced increase of prefrontal, premotor, and cingulate cortex activation during a motor task, and, however, a decrease of prefrontal and cingulate cortex activation during effortful cognitive tasks requiring suppression of inappropriate responses and strategic response selection. Our study clarifies the mechanisms of cortical deactivation observed with STN stimulation during executive processing and shows that this STN stimulation-induced reduction of cortical recruitment is driven by an increased GPi activation which in turn has an inhibitory influence on prefrontal cortical areas. This is the first direct evidence that STN stimulation significantly alters pallidal coupling with prefrontal, cingulate, and temporal cortices during performance of an attention-demanding cognitive task that requires response selection under competition.
Footnotes
Acknowledgements
The authors thank Jane Warren, MD for helpful discussions and all the radiochemistry and cyclotron staff. The authors would like to thank all the patients for their participation.
