Abstract
Both nitric oxide synthase (NOS) inhibitors and free radical scavengers have been shown to protect brain tissue in ischemia-reperfusion injury. Nitric oxide and superoxide anion act via distinct mechanisms and react together to form the highly deleterious peroxynitrite. Therefore the authors examined the effects and the interaction between the NOS inhibitor, NG nitro-L-arginine (LNA) and the antioxidant/superoxide scavenger, di-tert-butyl-hydroxybenzoic acid (DtBHB) in the rat submitted to 2 hours of middle cerebral artery occlusion. Posttreatment was initiated 4 hours after the onset of ischemia and infarct volume was measured at 48 hours. The dose-related effect of LNA resulted in a bell-shaped curve: 15, 56, 65, and 33% reduction of total infarct for 0.03, 0.1, 0.3, and 1 mg/kg (intravenously [IV]) respectively and 11% increase in infarct volume for 3 mg/kg (IV). Whereas DtBHB (20 mg/kg; intraperitoneally [IP]) was ineffective, the dose of 60 mg/kg produced 65% protection in infarct volume. The combination of a subthreshold dose of LNA (0.03 mg/kg; IV) and DtBHB (20 mg/kg; IP) resulted in significant reduction (49%) in infarct volume. These results show that LNA and DtBHB act synergistically to provide a consistent neuroprotection against ischemic injury when administered 4 hours after ischemia. This suggests that nitric oxide and free radicals are involved and interact in synergy in ischemia-reperfusion injury.
Free radicals are thought to be major contributors to reperfusion injury after cerebral ischemia. The oxygen free radicals species generated during reperfusion that may cause brain injury include superoxide anion, hydroxyl radical, hydrogen peroxide, and nitric oxide (Hall, 1989; Malinski et al., 1993; Kumura et al., 1995; Chan, 1996; Iadecola, 1997; Samdani et al., 1997). These deleterious species act independently but also interact together like nitric oxide (NO) which avidly reacts with superoxide anion to form the highly reactive oxidant agent peroxynitrite (Beckman et al., 1990) that is produced during cerebral ischemia (Forman et al., 1998; Fukuyama et al., 1998). For these reasons, scavenging free radicals by antioxidants (Xue et al., 1992; Clemens et al., 1993) or free radical spin traps (Cao and Phillis, 1994; Zhao et al., 1994; Kuroda and Siesjö, 1997) as well as inhibiting NO synthesis (Nowicki et al., 1991; Nagafuji et al., 1992; Margaill et al., 1997) have been used experimentally to seek neuroprotection and subsequently proposed as neuroprotective strategies to enhance neuronal survival after cerebral ischemia.
Nitric oxide is synthesized from L-Arginine by at least three isoforms of nitric oxide synthase (NOS): endothelial, neuronal, and inducible (Moncada et al., 1991). These enzymes are nonselectively inhibited by L-arginine derivative such as NG nitro-L-arginine (LNA). During ischemia, NO evokes contradictory effects depending on the cellular source and its time of production. Nitric oxide generated from endothelial NOS is a potent vasodilator and a platelet aggregation and leukocyte adhesion inhibitor and consequently may improve postischemic blood flow in the early phase. However, during ischemia, excessive production of NO from neuronal NOS when stimulated by glutamate release or from inducible NOS may in contrast exacerbate ischemic injury by promoting oxidative damage and energy failure (for review see Iadecola, 1997). These contrasting effects probably explain the conflicting observations showing that inhibition by nonselective NOS inhibitors such as LNA was found to reduce, enhance, or not affect cerebral ischemic damage (Demerle-Pallardy and Chabrier, 1995; Verrecchia et al., 1995; Iadecola, 1997; Samdani et al., 1997).
In the present study we have investigated the effects of LNA and the antioxidant di-tert-butyl-hydroxybenzoic acid (DtBHB) in transient focal cerebral ischemia in the rat. Because the two compounds protect brain tissue against ischemic injury by distinct mechanisms and interfere with the production of peroxynitrite via independent ways either by inhibiting NO formation or by trapping superoxide anion (Beckman et al., 1990), we consequently examined for a potential therapeutic interaction between the two drugs administered at subthreshold doses. A therapeutic window as wide as possible is necessary for any neuroprotective treatment in humans, we thus used a delayed posttreatment starting at 2 hours after recirculation, a paradigm more closely related to a clinical situation. This was also based on preliminary results (Spinnewyn et al., 1997) showing a significant reduction of infarct size afforded by delayed treatment with NOS inhibitors in transient focal ischemia.
MATERIALS AND METHODS
Animal model
Male Sprague Dawley rats, weighing 300 to 330 g, (Charles River, France) were used in the experiments. Animals were anesthetized with isoflurane (2% in O2) and anesthesia was maintained with 1.0% isoflurane in O2 (vol/vol) using a face mask. Rectal temperature was maintained at 37 ± 0.5°C throughout the surgical procedure using a heating blanket (pad). The left common carotid artery was exposed, under an operating microscope, through a midline neck incision and was carefully dissected free from surrounding nerves and fascia from its bifurcation to the base of the skull. The common carotid artery, the external carotid artery, and the occipital artery branch of the external carotid artery were dissected and ligated. The internal carotid artery was isolated and carefully separated from the adjacent vagus nerve, and the pterygopalatine artery was ligated close to its origin with a 5-0 silk suture. Next, a 4-cm length of 4-0 monofilament nylon suture (ethilon, Ethicon, Neuilly-Sur-Seine, France) was inserted into the internal carotid artery and then into the circle of Willis, effectively occluding the middle cerebral artery. The silk suture around the external carotid artery stump was tightened around the intraluminal nylon suture to prevent bleeding. Before use, the tip of the suture was heat-blunted and a 10-mm distal segment was coated with poly-L-lysine (0.1% wt/vol; Sigma, St. Quentin, Fallavier, France), in deionized water and dried at room temperature overnight. The suture was inserted 18 to 20 mm from the bifurcation of the common carotid artery according to the animal's body weight. After the intraluminal suture was placed, the neck incision was closed with a silk suture. After 2 hours the filament was retracted to allow reperfusion of the ischemic region. The animals were allowed to recover from anesthesia. Core temperature was continuously monitored with a rectal probe (Harvard, South Natick, MA, U.S.A.) and maintained at 37 ± 0.5°C during 6 hours after middle cerebral artery occlusion (MCAO). Rats without circling or gait disturbance during ischemia and reperfusion or with subarachnoidal hemorrhage at the time of death were excluded from analysis.
Measurement of infarct volume
Forty-eight hours after MCAO, animals were deeply anesthetized with isoflurane and decapitated. Brains were removed quickly and coronally sectioned into six 2-mm-thick slices (from rostral to caudal, first to sixth, S1 to S6). The brain slices were incubated for 20 minutes in a 2% solution of 2,3,5-triphenyl-tetrazolium chloride (Sigma) at room temperature and fixed by immersion in a 10% buffered formalin solution (Sigma). Lesions (noted by pale staining) were evaluated by an indirect method for calculating infarct volume (Jiang et al., 1994) using an image analysis system (Biocom, Les Ulis, France).
Experimental design
Dose-dependent effect of LNAA and DtBHB. LNA (Novabiochem, Meudon, France) was dissolved in physiologic saline and administered at 0.03 to 3 mg/kg as an intravenous (IV) bolus (2 mL/kg) 4 and 24 hours after the onset of MCAO. Di-tert-butylphenols possess antioxidant properties and are beneficial in models of cerebral ischemia in rat (Clemens et al., 1993) or in gerbil (O'Neill et al., 1997). We used the di-tert-butylphenol analogue: 3'5-di-tert-butyl-4-hydroxybenzoic acid (DtBHB; Aldrich, St. Quentin, Fallavier, France), which inhibits superoxide anion generated from murine macrophage cell line J774A1 after activation by phorbol myristate acetate (IC50 = 45.2 µmol/L; data not shown). DtBHB was suspended in tween 80 with physiologic saline and administered at 20 and 60 mg/kg as an intraperitoneal (IP) bolus (10 mL/kg) 4 and 24 hours after the onset of MCAO.
Effect of combination treatment. For the combination experiment, a subthreshold dose of LNA (0.03 mg/kg; IV) and DtBHB (20 mg/kg; IP) were administered alone or in combination 4 and 24 hours after the onset of MCAO.
Statistics
All values are expressed as mean ± SD. Statistical analysis was performed with two-tailed Dunnett's test (dose effect experiments) or two-tailed Student's test (combination experiments). Statistical significance was presented at the P <0.05 level.
RESULTS
Effects of LNA and DtBHB
To construct a dose-response curve with LNA, 88 rats were studied, two LNA-treated and four controls were excluded because they did not exhibit right circling after MCAO. Temperatures were unchanged in both groups before and after drug administration eliminating hypothermia as a cause of cerebral neuroprotection. Four out of 38 rats died in the control group whereas 4 rats out of 8 died in the LNA (3 mg/kg) treated group. This mortality was not caused by subarachnoid hemorrhage secondary to surgery as assessed by postmortem examination. Thus, LNA (3 mg/kg) increased the mortality by 38%. Two hours of transient cerebral ischemia resulted in a total infarct of 222 ± 96 mm3 in the vehicle-treated group. The administration of 0.03, 0.1, 0.3, and 1 mg/kg LNA reduced the total infarct volume measured at 48 hours by 15, 56, 65, and 33% respectively (Fig. 1). Reduction of the infarct volume was evident throughout the hemisphere, and a statistically significant decrease in the infarct area was detected in section 3–6; 2–6 and in section 1 for the doses of 0.1, 0.3, and 1 mg/kg, respectively. By contrast, the highest dose of LNA (3 mg/kg) increased infarct volume by 11% (P > 0.05). Thus, a dose-related effect of LNA for reduction of infarct size resulted in a bell-shaped curve. A 0.03 mg/kg dose was not effective to reduce significantly infarct volume and was considered as a subthreshold dose.
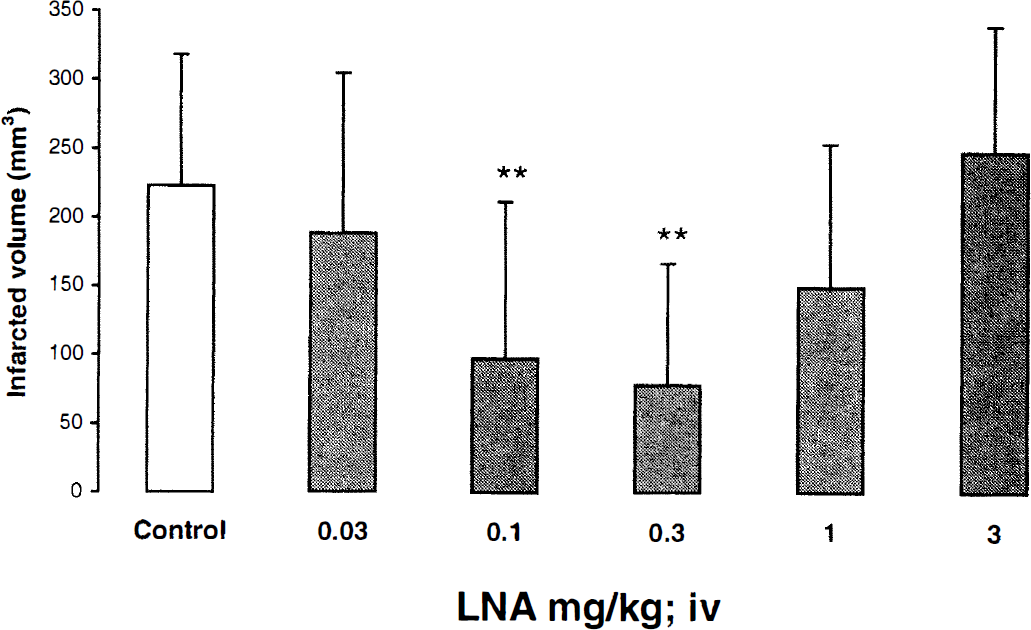
Dose-response of the effect of NG nitro-L-arginine (LNA) on total infarcted volumes induced by transient focal cerebral ischemia. Values are mean + SD. At 4 and 24 hours after onset of ischemia, rats were injected with bolus doses of LNA (0.03, 0.1, 0.3, 1, and 3 mg/kg; n = 13, 8, 8, 7, and 4, respectively or with saline (control; n = 34). LNA significantly reduces infarct area (**P < 0.01) as compared with saline treated-group. IV, intravenous.
In a second series of experiments, DtBHB was administered by a IP route using the same treatment schedule. Administration of DtBHB (60 mg/Kg; IP) showed a significative diminution (65% reduction) of infarct size (control: 247 ± 99 mm3; DtBHB: 86 ± 160 mm3). However, deleterious neurologic signs (e.g., tremor) were observed at this dose, thus preventing the use of higher doses. The dose of 20 mg/kg (IP) did not reduce the infarct size nor was accompanied by increased neurologic behavior impairments and thus was used for further experiments as a subthreshold dose (Fig. 2).
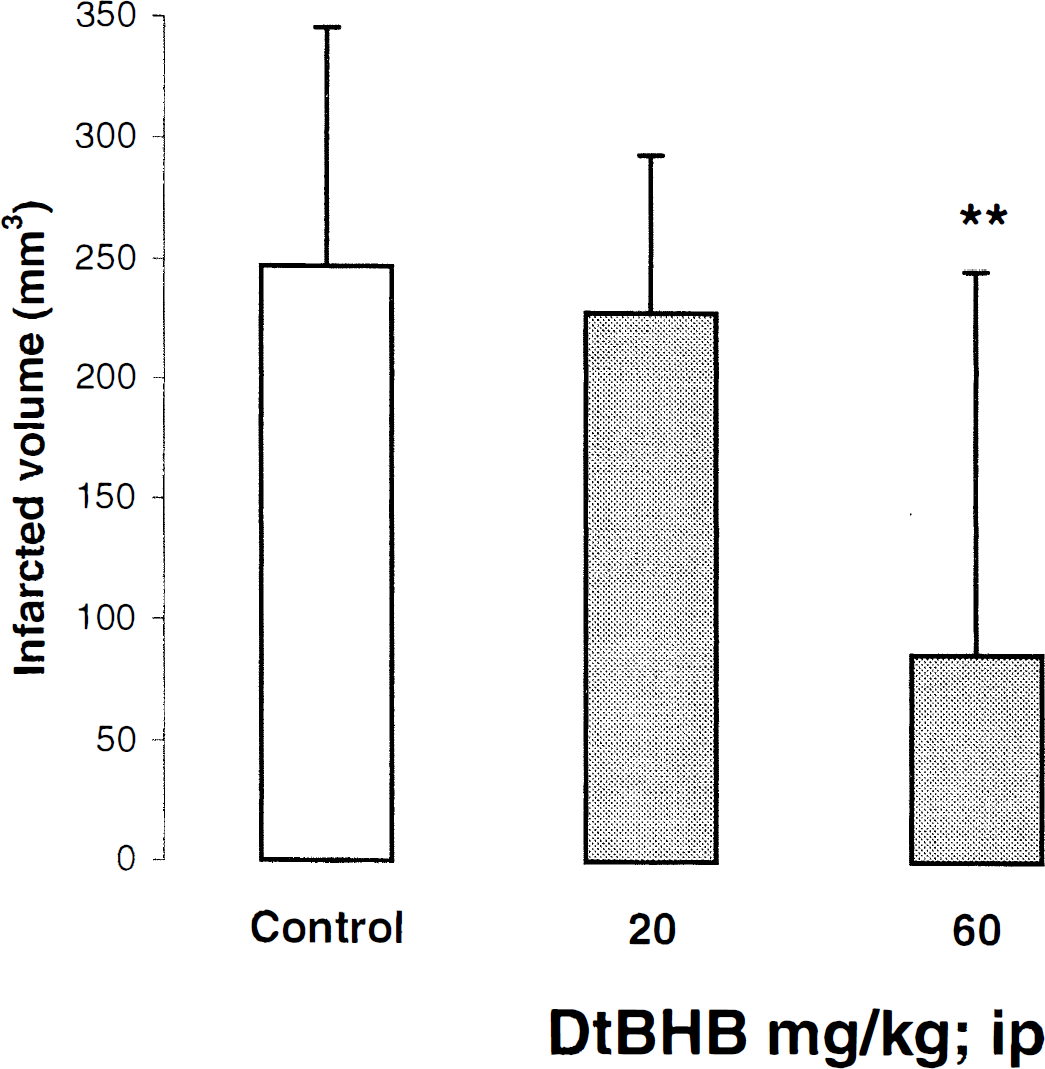
Effect of di-tert-butyl-hydroxybenzoic acid (DtBHB) on total infarcted volumes induced by transient focal cerebral ischemia. Values are mean + SD. At 4 and 24 hours after onset of ischemia, rats were injected (IP) with bolus doses of DtBHB (20 and 60 mg/kg; n = 8, and 6 respectively) or with solvent (control; n = 14). DtBHB (60 mg/kg) significantly reduces infarct area (**P < 0.01) as compared with saline treated-group.
Effect of combination treatment
In a third series of experiments 38 rats were used. The determined subthreshold doses of DtBHB (20 mg/kg, IP) or LNA (0.03mg/kg) when administered individually were unable to reduce the infarct size (Fig. 3) confirming our precedent experiments. However, the combination treatment of LNA and DtBHB produced a dramatic reduction (49%) in infarct volume. Reduction of the infarct was evident throughout the hemisphere, and a statistically significant decrease in area of infarct was detected in section 2-4.
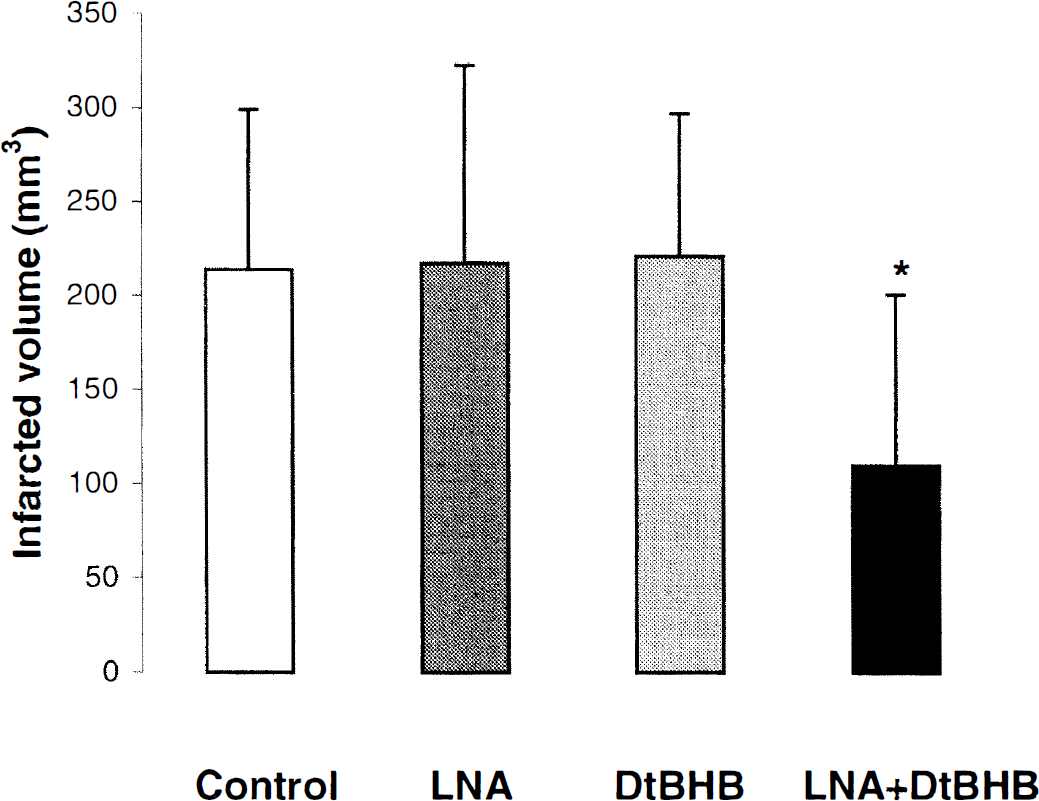
Effect of NG nitro-L-arginine (LNA) and di-tert-butyl-hydroxybenzoic acid (DtBHB) alone or in combination on total infarcted volumes induced by transient focal cerebral ischemia. Values are mean + SD. At 4 and 24 hours after onset of ischemia, rats were injected with bolus doses of LNA (0.03 mg/kg, IV; n = 8); DtBHB (20 mg/kg, IP; n = 8) or both (n = 8) or with saline (n = 14). Combination of LNA and DtBHB significantly reduced infarct area (*P < 0.05) as compared with saline treated-group.
DISCUSSION
The present experiments are the first studies to show synergistic protective effects between an inhibitor of NOS and an antioxidant in brain injury after transient focal ischemia. Using LNA as a prototype of NOS inhibitor and DtBHB as antioxidant superoxide scavenger, we showed that subthreshold doses of the two drugs, ineffective when administered individually, provide a dramatic reduction of infarct size (49% reduction) when combined even if the treatment is initiated 4 hours after onset of ischemia.
Contribution of NO to the ischemic damage is well documented. However, the effectiveness of NOS inhibitors is more controversial and results from reduction to worsening of infarct have been reported depending on the dosage regimen (Nowicki et al., 1991; Dawson et al., 1992; Margaill et al., 1997; Nagafuji et al., 1992; Zhang et al., 1996). Using a model of transient focal ischemia, the present results illustrate the dose-related effect of LNA and confirm the limited therapeutic index of LNA observed in a model of global ischemia (Nagafuji et al., 1993) or permanent focal ischemia (Carreau et al., 1994). This was attributed to the nonselective action of LNA and to detrimental inhibition of endothelial NOS (3mg/kg LNA increased the mean blood pressure by approximately 20 mm Hg, data not shown). Nevertheless, our treatment paradigm agrees with the observation that NOS inhibitors are neuroprotective in posttreatment as already observed with the nonselective NOS inhibitors NG nitro-L-arginine methyl ester (Margaill et al., 1997), or the more selective neuronal NOS inhibitor TRIM (Escott et al., 1998).
Reactive oxygen species (e.g., superoxide anion, hydroperoxide, and hydroxyl radicals) have also been implicated in the pathophysiologic cascade leading to ischemic damage (Braughler and Hall, 1989; Kuroda and Siesjö, 1997). Superoxide anion might arise from infiltrating activated leukocytes, neurons, and astroglia during ischemia-reperfusion injury (Copin et al., 1992; Lafon-Cazal et al., 1993; Matsuo et al., 1995). Superoxide scavengers such as alpha-phenyl-N-tert-butyl nitrone and pterin-6-aldehyde (Mori et al., 1998);) afford neuroprotection in ischemia (Zhao et al., 1994; Mori et al., 1998) even when alpha-phenyl-N-tert-butyl nitrone treatment was delayed until 12 hours after ischemia in a model of permanent MCAO in rats (Cao and Phillis, 1994). Thus, both NO and superoxide anion participate in the pathophysiologic cascade leading to ischemic damage. In addition, NO and superoxide anion react together with a high rate constant to give peroxynitrite, which at physiological pH is a stronger oxidant than superoxide anion or NO. Interfering with superoxide anion or NO production may thus afford protection in ischemic-reperfusion injury. The present results show a synergistic reduction of infarct size by the combined treatment of LNA, a nonselective NOS inhibitor and di-tert-butylphenol, a superoxide scavenger (IC50 = 45.2 µmol/L) more potent than pterin-6-aldehyde and alpha-phenyl-N-tert-butyl nitrone (IC50 = 150 µmol/L and 22 mmol/L respectively; Mori et al., 1998). This indicates that controlling both superoxide anion and NO is more efficient than the inhibition of only one radical species. This also suggests a cooperative deleterious effect of superoxide anion (or other oxyradicals) and NO. These observations tail with the recent finding of Sampei et al., 1997 showing an increased resistance to ischemia of combined mutant mice lacking neuronal NOS and overexpressing superoxide dismutase in comparison to single mutant mice.
The second important feature of this study is the large therapeutic time window of intervention in inhibiting superoxide anion and NO since the treatment was initiated 4 hours after the onset of ischemia. This is consistent with previous results obtained with inhibitors of NOS (Margaill et al., 1997; Escott et al., 1998) or antioxidant (Cao and Phillis, 1994). In addition, if we consider that the neuroprotective effects of LNA are caused by inhibition of neuronal NOS and the deleterious effects observed at higher doses are associated with inhibition of endothelial NOS, the present results show that combination with an antioxidant allows a reduction in the dose of LNA leading to a significant neuroprotection.
In conclusion, we have shown that the combination of a NOS inhibitor and an antioxidant (e.g. superoxide scavenger) is synergistic toward the reduction of neuronal damage during ischemia-reperfusion supporting the idea that NO, superoxide anion, and peroxynitrite play important independent, as well as cooperative, roles in neuronal injury. This also suggests the potential beneficial use of a combination of a NOS inhibitor and an antioxidant in ischemia.
Footnotes
Acknowledgment
The authors thank Jeremiah Harnett for skillfully reading the manuscript.
