Abstract
Subarachnoid hemorrhage (SAH) often leads to a long-term narrowing of cerebral artery called vasospasm. To understand the molecular mechanisms in vasospasm, signal transduction of tyrosine kinase pathway and phosphorylation of myosin light chain (MLC) and calponin (CaP) in the basilar artery were studied. Vasospasm was produced in the canine basilar artery by a two-hemorrhage method, and vasocontraction was induced by a local application of KCl or serotonin to the basilar artery after a transclival exposure. Intracellular substrates of tyrosine kinase pathway, including Shc, Raf1, and extracellular-regulated kinases in the basilar artery, were activated after SAH, and the activation of Shc suggests stimulation of signal transductions from tyrosine kinase receptors, G-coupled receptors, or both. The activation of tyrosine kinase pathway in vasospasm also was supported by dose-dependent dilation of the spastic basilar artery on days 0 and 7 by topical application of genistein, a tyrosine kinase inhibitor, and associated marked inhibition of tyrosine phosphorylation of intracellular substrates, including Shc. In addition, the generation of protein kinase M, catalytic fragment of protein kinase Cα (PKCα), in vasospasm on days 0 and 7 was inhibited in response to genistein, indicating an inactivation of μ-calpain. It is suggested, therefore, that the reversal of vasospasm by genistein is closely associated with the restoration of intracellular Ca2+ levels. However, the increased activities of Raf1 and extracellular-regulated kinases in vasospasm were declined on day 7 compared with those on day 0 or 2, suggesting that the activation of tyrosine kinase pathway is more closely associated with the early stage of vasospasm than with the late stage of vasospasm. The analysis by pyrophosphate polyacrylamide gel electrophoresis (PPi-PAGE) demonstrated three MLC bands in vasospasm on days 2 and 7, as well as in KCl- and serotonin-induced vasocontraction. Since PPi-PAGE resolves smooth muscle MLC into three bands in the MLC kinase (MLCK)-mediated phosphorylation and into a single band in the PKC-mediated phosphorylation based on the phosphorylation state, the current results suggest that MLC in vasospasm is phosphorylated by MLCK but not by PKC. In basilar artery, CaP was significantly down-regulated, and in addition, significantly phosphorylated on serine and threonine residues only in vasospasm on days 2 and 7. Although the significance of CaP phosphorylations in vivo still is controversial, CaP down-regulation and phosphorylation may attenuate the inhibition of Mg2+-ATPase activity by CaP and induce a potential enhancement of smooth muscle contractility in delayed vasospasm. Since CaP is phosphorylated vivo by PKC, activated PKC in vasospasm may phosphorylate CaP. Thus, SAH stimulates tyrosine kinase pathway to increase intracellular Ca2+ and activate PKC, and the former activates MLCK to phosphorylate MLC, whereas the latter phosphorylates CaP but not MLC.
Keywords
The detailed molecular mechanisms contributing to long-term narrowing of the cerebral artery in vasospasm are a topic of intense experimental study. In experimental vasospasm, μ-calpain-but not m-calpain-was continuously activated in the canine basilar artery (Yamaura et al., 1993; Sato et al., 1997), suggestive of a continuous increase in intracellular Ca2+ levels in vasospasm. On the other hand, μ-calpain was transiently activated only in the beginning of vasocontraction induced by KCl or serotonin. Increased intracellular Ca2+ levels may activate Ca2+/calmodulin-dependent myosin light chain kinase (MLCK) in smooth muscle to phosphorylate myosin light chain (MLC). This regulatory pathway of Ca2+-calmodulin-MLCK is the major potential feature of contractile system regulation in smooth muscle. Recently, vasospasm of canine anterior spinal artery has been reported to be associated with an increased phosphorylation of MLC (Butler, et al., 1996). The canine vasospasm was significantly reversed by a topical application of 1-(5-chloronaphthalenesulfonyl)-1H-hexa-hydro-1,4-diazepine (Kokubu et al., 1989), a selective MLCK inhibitor, suggestive of the involvement of MLCK in development of vasospasm. In addition, smooth muscle PP1, which dephosphorylates MLC (Alessi et al., 1992), was decreased in activity in vasospasm (Fukami et al., 1995). Much attention has been paid to phosphorylation-dephosphorylation of thin-filament proteins calponin (CaP) and caldesmon as the second potential feature of contractile system regulation. Calponin inhibits the actin-activated Mg2+-ATPase activity of smooth muscle through its binding to actin in vitro (Takahashi et al., 1986; Winder and Walsh, 1990; Naka et al., 1990), and the inhibitory effect of CaP is alleviated by phosphorylation catalyzed by protein kinase C (PKC) and calmodulin kinase II in vitro (Winder and Walsh, 1990; Naka et al., 1990; Horowitz et al., 1996b). The mechanism of involvement of PKC in vasospasm remains controversial, but we have demonstrated the activation of PKCα and the formation of protein kinase M (PKM), catalytic fragment of PKCα, in vasospasm (Minami et al., 1992; Sato et al., 1997). Smooth muscle PP1 and PP2A, which dephosphorylate CaP (Winder and Walsh, 1990; Fraser and Walsh, 1995), had decreased activity in vasospasm (Fukami et al., 1995). Thus, the phosphorylation-dephosphorylation mechanisms of MLC and CaP may play an important role in controlling smooth muscle contractility in vasospasm. The current study examines the following questions: whether activation of tyrosine kinase pathway in the canine basilar artery contributes to the development of vasospasm; which protein kinase is involved in MLC phosphorylation in vasospasm; and how CaP is involved in the regulation of smooth muscle contractility in vasospasm compared with vasocontraction induced by KCl or serotonin.
MATERIALS AND METHODS
Animal preparation
The care of the animals in this study complied with the United States Public Health Service standards. Adult mongrel dogs, each weighing 10 to 14 kg, were anesthetized with an intramuscular ketamine hydrochloride (10 mg/kg), then with intravenous pentobarbital sodium (15 mg/kg), and maintained with a 70% nitrous oxide/30% oxygen mixture. Muscle relaxation was assured with an intravenous injection of pancuronium bromide at half-hour intervals, and arterial carbon dioxide tension was kept at a mean of 32 ± 3 mm Hg by adjusting the respiratory pump or by adding carbon dioxide to the inspired gas. Body temperature was kept at 37°C with a heating blanket, and mean arterial blood pressure and pulse rate monitored continuously in the femoral artery were not changed during the procedure. Vertebral angiography was carried out through the femoral artery.
Spastic group
Cerebral vasospasm was produced in dogs by an injection of 5 mL of fresh autogenous arterial blood into the cisterna magna, followed by another injection 2 days later. Vertebral angiography was repeated 30 minutes (day 0), 2 days (day 2), and 7 days (day 7) after the first intracisternal injection of blood. The caliber of the basilar artery was measured at three predetermined levels on the magnified angiogram and was expressed as a percentage of the prespastic caliber. Thus, the angiogram on days 0 and 2 was obtained after the animal had received a single injection of blood, whereas that on day 7 was obtained after two injections of blood had been administered. As control of the spastic group, 5 mL of saline was injected into the cisterna magna instead of fresh blood, and the angiographic caliber of the basilar artery was examined 2 days after the injection.
Potassium chloride and serotonin groups
The normal basilar artery was exposed by gently removing the clivus and carefully incising the dura and arachnoid under a surgical microscope. The basilar artery then was contracted by a local application of 80 mmol/L KCl or 0.5 μmol/L serotonin for 15 minutes (KCl-15 and serotonin-15 groups) or 90 minutes (KCl-90 and serotonin-90 groups). The concentrations of KCl and serotonin used in this study induced the maximum tonic contraction of the canine basilar artery in vitro. The KCl-induced contraction was obtained in the presence of the α-adrenergic blocker phentolamine (1 μmol/L) to abolish the effects of norepinephrine released by high K+ depolarization. The reduced caliber of the basilar artery was photographed for measurement and expressed as a percentage of the untreated control caliber.
Tissue preparation for analysis
The animals were killed after perfusion at 120 mm Hg with 500 mL of ice cold physiologic salt solution containing 140 mmol/L NaCl, 4.7 mmol/L KCl, 1.2 mmol/L Na2HPO4, 1.2 mmol/L MgSO4, 1.6 mmol/L CaCl2, 0.02 mmol/L ethylenediamine tetraacetic acid (EDTA), 5.6 mmol/L glucose, 2 mmol/L 3-morpholinopropanesulfonic acid, and 1 mmol/L sodium orthovanadate, pH 7.4, and then the basilar artery was removed together with the entire brain. In the spastic group, the blood clot around the basilar artery and its branches was carefully removed on an ice bath without any mechanical stimulation given to the arteries. After a brief washing, the basilar artery was quickly frozen in liquid nitrogen until used. The basilar arteries in the control, KCl, and serotonin groups underwent a similar procedure without the removal of the blood clot.
Immunoprecipitation and immunoblotting analysis
The basilar artery was pulverized in liquid nitrogen and lysed on ice in lysis buffer (20 mmol/L N-2-hydroxypiperazine-2-ethanesulfonic acid [HEPES] pH 7.5, 1% Triton X-100, 50 mmol/L NaCl, 1 mmol/L ethylene-glycol-bis(β-amino-ethylether)N,N′-tetraacetic acid [EGTA], 5 mmol/L β-glycerophosphate, 30 mmol/L sodium pyrophosphate, 100 μmol/L Na3VO4, 1 mmol/L phenylmethylsulfonyl fluoride [PMSF], 10 μg/mL aprotinin, and 10 μg/mL leupeptin). For immunoprecipitation of Shc, lysates were absorbed with Affi-Gel protein A (Bio-Rad, Hercules, CA, U.S.A.)-normal rabbit serum complex for 90 minutes at 4°C, and then incubated with Affi-Gel protein A-polyclonal anti-Shc antibody (UBI, Lake Placid, NY, U.S.A.) complex, which had been treated in advance with 20 mmol/L dimethyl pimelimidate in 0.2 mol/L sodium borate for 30 minutes at room temperature for cross-linking. The immunoprecipitates and the lysates were separately boiled for 5 minutes in a 2 × sample buffer, subjected to 10% sodium dodecylsulfate polyacrylamide gel electrophoresis (SDS-PAGE), except for immunoblotting of Raf1 in which 6% SDS-polyacrylamide gel was used, and transferred to separate polyvinylidene difluoride membranes, which were blocked overnight in Tris-buffered saline containing 0.1% Tween 20 supplemented with 1% gelatin. The membrane of analysis of Shc activation was probed for 1 hour with mouse monoclonal anti-phosphotyrosine-HRP conjugate (Amersham International, Buckinghamshire, U.K.), and immunologically reprobed with anti-human Shc antibody after stripping each of primary antibody off the membranes. The membranes for immunoblotting were incubated for 1 hour with anti-phosphotyrosine antibody (Upstate Biotechnology, Waltham, MA, U.S.A.) for analysis of intracellular substrates of tyrosine kinases, with anti-CFα4 antibody (Sato et al., 1997: antibody against human PKCα sequence produced at the cleavage site by μ-calpain) for analysis of PKM, or with rabbit polyclonal anti-Raf1 antibody (Santa Cruz, Santa Cruz, CA, U.S.A.) for study of mobility shift by Raf1 immunoblot. The secondary antibody used was HRP-conjugated antibodies (Dako, Glostrup, Denmark). Immunoblots were detected by the ECL system (Dupont/NEN, Boston, MA, U.S.A.).
In gel myelin basic protein kinase assay for extracellular signal-regulated kinase
The pulverized basilar artery was lysed on ice in lysis buffer (20 mmol/L Tris-HCl pH 7.5, 5 mmol/L EGTA, 1 mmol/L Na3VO4, 20 mmol/L β-glycerophosphate, 10 mmol/L NaF, 1 mmol/L dithiothreitol [DTT], 1 μg/mL aprotinin, 0.1 mmol/L PMSF, 0.1 mmol/L N-tosyl-L-phenylalanine chloromethyl ketone, and 0.1 mmol/L N α-p-tosyl-L-lysine chloromethyl ketone). Lysates were boiled in 2 × sample buffer for 5 minutes and electrophoresed in 11% SDS-polyacrylamide gel containing 0.5 mg/mL myelin basic protein fragment. After SDS was removed from the gel using 20% propanol, the gel was washed in 50 mmol/L Tris-HCl at pH 8.0 and 5 mmol/L β-mercaptoethanol, thereafter denatured with 6 mol/L guanidine, renatured in 50 mmol/L Tris-HCl at pH 8.0/5 mmol/L β-mercaptoethanol containing 0.04% Tween 40, and incubated with kinase buffer (48 mmol/L HEPES pH 8.0, 25 μCi [γ-32P]ATP [Dupon/MEN], 30 μmol/L ATP, 6 mmol/L MgCl2, 2.4 mmol/L DTT, and 0.12 mmol/L EGTA) at room temperature for 1 hour. After washing with 5% trichloroacetic acid and 1% pyrophosphate solution, the gel was dried and kinase activity was visualized using FUJIX Bio-Imaging Analyzer BAS2000 (Fuji Photo Film, Tokyo, Japan). Lysates also were probed with anti-ERK1/ERK2 antibody for immunoblotting.
Response of spastic artery to genistein
After the exposed spastic basilar artery was photographed, dose-response measurements were obtained after the topical application of successively increasing concentrations of genistein. At the end of each experiment, 4 mmol/L EGTA was added. The mean diameter of spastic basilar artery was calculated by measurement at three predetermined levels and expressed as a percentage of the relaxation induced by EGTA. The percentage relaxations (mean ± SD) induced by the increasing concentrations of genistein were charted on logarithmic dose-response curves. The animals' mean arterial blood pressure and pulse rate were not changed significantly during the application of genistein.
Pyrophosphate polyacrylamide gel electrophoresis
The pulverized basilar artery was put in buffer (100 mmol/L sodium pyrophosphate, 5 mmol/L EGTA, 1 mmol/L EDTA, 100 mmol/L NaF, 10% glycerol, 2.5 mmol/L DTT, 1 mmol/L PMSF, pH 8.8) and centrifuged at 10,000 g for 15 minutes. The supernatant was incubated in 0.6 mol/L KI for 30 minutes at 4°C, and then sucrose was added together with glycerol to adjust to final 20% sucrose solution and to pH 7.5. Polyacrylamide gel used was composed of 3.8% acrylamide, 0.101% methylenebisacrylamide, 40 mmol/L Na4P2O7, 10% glycerol, and 15% NNN′N′-tetramethylethylenediamine, pH 8.5, and polymerized by the addition of freshly prepared 12.5% ammonium persulphate after deaeration. Electrophoresis buffer contained 40 mmol/L Na4P2O7, 10% glycerol, and 2 mmol/L L-cysteine, pH 8.5. After a pre-electrophoresis at 11 voltage/cm for 30 minutes at 2°C, the basilar artery extract was loaded on the top of gels and electrophoresed overnight under a constant voltage gradient of 8 to 11 voltage/cm using a regulated power supply. Proteins on the gels were stained with 0.1% Coomassie brilliant blue R250.
Urea-glycerol polyacrylamide gel electrophoresis
The protein bands on PPi-PAGE were cut off separately, soaked with 9 mol/L urea containing 10 mmol/L Tris-HCl (pH 6.8) and 5% β-mercaptoethanol for 8 minutes, and then subjected to glycerol PAGE for 8 hours. Stacking gel used contained 0.125 mol/L Tris-HCl at pH 6.7, 5.7% acrylamide, 0.152% bisacrylamide, and 40% glycerol, whereas separation gel was composed of 0.37 mol/L Tris-HCl at pH 8.7, 14% acrylamide, 0.37% bisacrylamide, and 40% glycerol. The reservoir buffer contained 20 mmol/L Tris-glycine at pH 8.7. The MLC bands were visualized with a highly sensitive silver staining.
Phosphorylation of calponin
The pulverized basilar artery was boiled at 100°C for 5 minutes in lysis buffer (50 mmol/L imidazole-HCl pH 7.3, 190 mmol/L NaCl, 1 mmol/L EDTA, 5 mmol/L EGTA, 3 mmol/L DTT, 1.5% Triton X-100, 1 μmol/L p-amidinophenyl methanesulfonyl fluoride hydrochloride, 50 mmol/L NaF, 50 mmol/L sodium pyrophosphate, 1 μmol/L okadaic acid, 1 mmol/L Na3VO4, 50 μg/mL leupeptin, and 2% SDS). Lysates were absorbed with protein G-sepharose (Sigma, St. Louis, MO, U.S.A.) and normal mouse serum beads for 90 minutes at 4°C, and then incubated with protein G-sepharose and purified mouse monoclonal anti-CaP antibody (Sigma) beads. The immunoprecipitates were boiled at 100°C for 3 minutes in a 2 × sample buffer and subjected to 12% SDS-PAGE. Separated proteins were visualized with the silver staining for quantitation of CaP and then electrophoretically transferred onto polyvinylidene difluoride membrane, which was probed with mouse monoclonal antibody against phosphoserine (Sigma), phosphothreonine (Sigma), or phosphotyrosine (Upstate Biotechnology). The secondary antibody was HRP-conjugated rabbit anti-mouse IgG (Dako).
RESULTS
Caliber of the basilar artery
No significant angiographic narrowing of the basilar artery was shown before the animals in the control group were killed. The mean percent calibers in the spastic group were 78.0 ± 5.3% (P < 0.0007) on day 0, 70.2 ± 7.5% (P < 0.0009) on day 2, and 60.4 ± 6.8% (P < 0.0002) on day 7, whereas those in the KCl and serotonin groups decreased over a period of approximately 45 minutes after the treatment and then stabilized (87.4 ± 2.6% [P < 0.02] in the KCl group and 80.4 ± 1.8% (P < 0.003) in the serotonin group). Statistical significance of mean percent calibers was examined in five dogs in each group by two-tailed t test for uncorrelated pairs.
Activation of tyrosine kinase pathway
Phosphorylation of intracellular substrates of tyrosine kinase taken from the spastic basilar arteries apparently was increased in vasospasm on days 0, 2, and 7 and was markedly inhibited by a topical application of median effective dose (ED50) of genistein for reversal of vasospasm on day 7 to the spastic basilar artery on day 7, as shown in Fig. 1. The anti-Shc antibody specifically recognized the 46-, 52-, and 66-kd isoforms of Shc (Fig. 2). When Shc immunoprecipitates were immunoblotted with anti-phosphotyrosine antibody, only 46- and 52-kd isoforms were shown to be tyrosine phosphorylated before initiation of vasospasm (52-kd isoforms were more phosphorylated than 46-kd ones). In vasospasm on days 0, 2, and 7, the phosphorylation of Shc, including the 66-kd isoform, was increased, whereas the levels of individual immunoreactive Shc isoforms were not changed, indicating activation of Shc. The activities of Raf1 were determined by the presence of Raf1 with reduced gel electrophoretic mobility from phosphorylation. As shown in Fig. 3, a single immunoreactive 70-kd Raf1 band became doublets after subarachnoid hemorrhage, indicating increased phosphorylation of Raf1. Phosphorylation of extracellular signal-regulated kinases (ERK) was estimated by in gel myelin basic protein kinase assay. Both ERK1 and ERK2 were slightly phosphorylated to similar degree in the control group, and their phosphorylations, although ERK1 phosphorylation was less than ERK2 phosphorylation, were increased after subarachnoid hemorrhage (Fig. 4). In contrast, the immunoreactive ERK1 and ERK2 levels were not changed.
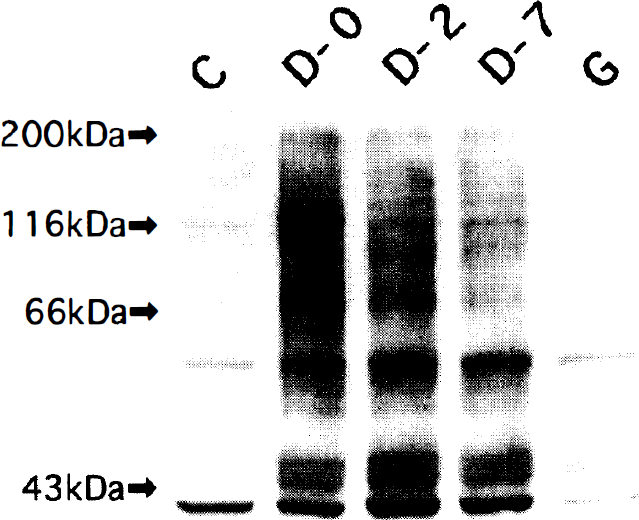
Immunoblot analysis by anti-phosphotyrosine antibody shows a marked increase in phosphorylation of several intracellular substrates of tyrosine kinase taken from the spastic basilar arteries on days 0, (D-0), 2 (D-2), and 7 (D-7). The increase in tyrosine phosphorylation observed in the spastic artery on day 7 is considerably inhibited by a topical application of 17.2 μmol/L genistein (median effective dose [ED50] for reversal of vasospasm on day 7). Similar results were obtained in three experiments. C, control; G, genistein.

Analysis of Shc activation showing a marked increase in tyrosine phosphorylation of 46-, 52-, and 66-kd isoforms of Shc in the spastic basilar arteries on days 0 (D-0), 2 (D-2), and 7 (D-7). The increase in tyrosine phosphorylation of Shc on day 7 is markedly inhibited by a topical application of ED50 of genistein. Tyrosine phosphorylation of the 66-kd isoform of Shc is scarcely detected before vasospasm and after treatment with genistein. Basilar artery lysates were immunoprecipitated with anti-Shc antibody, resolved by 10% sodium dodecylsulfate polyacrylamide gel electrophoresis (SDS-PAGE), transferred onto polyvinyl idene difluoride membranes, and immunologically probed with anti-phosphotyrosine-HRP conjugate (left). The membranes were reprobed with anti-Shc antibody, showing no significant changes in levels of 46-, 52-, and 66-kd isoforms of Shc after subarachnoid hemorrhage (SAH) and after treatment with genistein (right). The results are from a representative study performed three times with comparable outcomes. C, control; G, genistein.
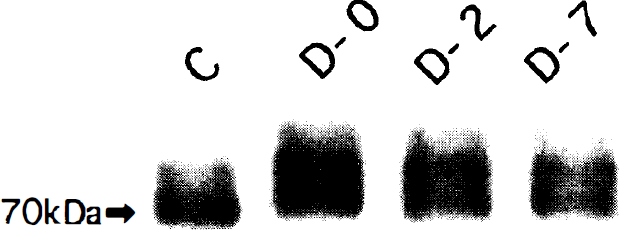
Analysis of Raf1 activation after SAH shows that immunoreactive Raf1 with reduced electrophoretic mobility becomes detectable in vasospasm on days 0 (D-0), 2 (D-2), and 7 (D-7), suggesting the increase in phosphorylation of Raf1. Basilar artery lysates were subjected to 6% SDS-PAGE, transferred onto PVDF membranes, and probed with anti-Raf1 antibody by immunoblotting. Similar results were achieved on three separate experiments with comparable outcomes. C, control.

Analysis of activities of extracellular signal-regulated kinases (ERK) by in gel MBP kinase assay showing a marked increase in phosphorylation of ERK1 and ERK2 in vasospasm on days 0 (D-0), 2 (D-2), and 7 (D-7). Basilar artery lysates were electrophoresed in 11% SDS-polyacrylamide gel containing 0.5 mg/mL myelin basic protein. The gel was incubated with a kinase buffer containing 25 μCi[γ-32P]ATP to measure kinase activity (left). Lysates also were immunologically probed with anti-ERK1/ERK2 antibody, showing no significant change in levels of ERK1 and ERK2 after SAH (right). Results are representative of three independent experiments. C, control.
Response of spastic artery to genistein
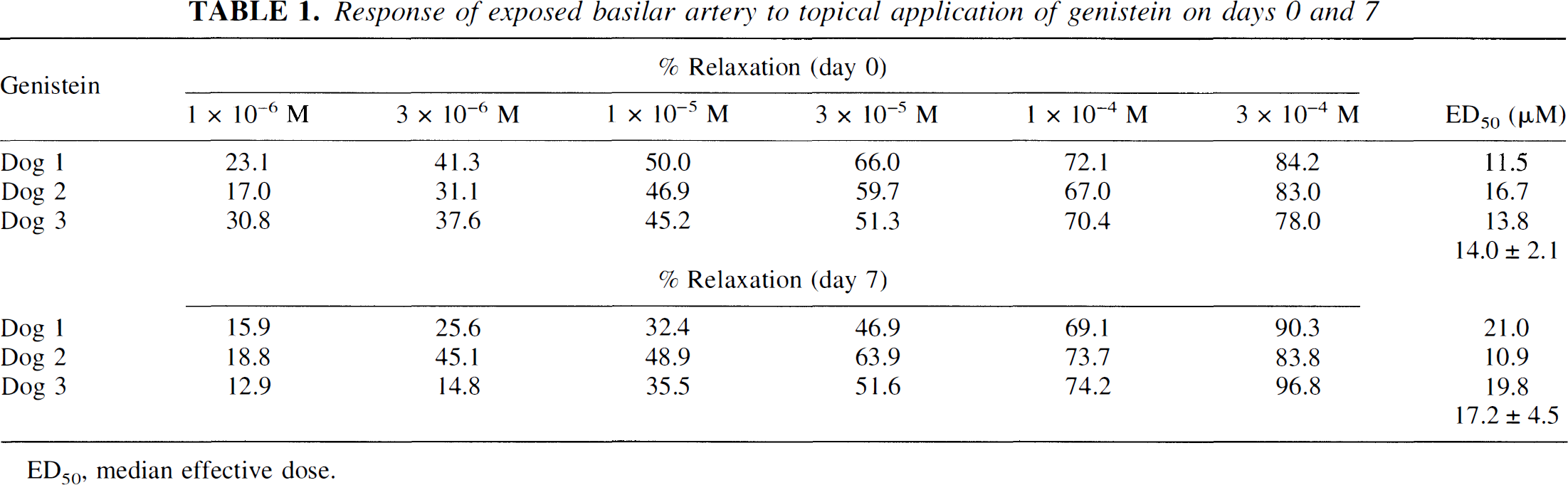
A brief washing with warm saline after careful removal of the blood clot around the spastic basilar artery on days 0 and 7 did not induce a significant change in the caliber of the spastic basilar artery. The exposed spastic basilar artery was dilated dose-dependently by a topical application of 1 × 10−6 to 3 × 10−4 mol/L genistein. Taking the relaxation induced by 4 mmol/L EGTA as 100%, the mean ± SD values of ED50 of genistein for relaxation of the spastic basilar artery in three dogs were 14.0 ± 2.1 μmol/L on day 0 and 17.2 ± 4.5 μmol/L on day 7 (Table 1). When the spastic basilar artery on day 7 was treated with the ED50 of genistein, the phosphorylation of intracellular substrates of tyrosine kinase (Fig. 1), including Shc isoforms (Fig. 2), was markedly decreased, but the immunoreactive Shc isoform levels remained constant (Fig. 2), indicating decrease in activities of Shc. The generation of PKM in vasospasm on days 0 and 7 detected by anti-CFα4 antibody (Sato et al., 1997) was inhibited in response to the topical treatment with respective ED50 of genistein (Fig. 5).
Response of exposed basilar artery to topical application of genistein on days 0 and 7
ED50 median effective dose
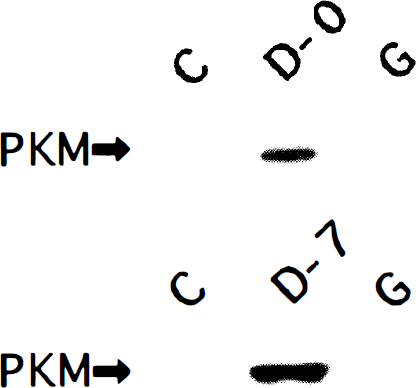
Immunoblot analysis of protein kinase M (PKM) probed with anti-CFα4 antibody showing that the generation of PKM in the spastic basilar artery on days 0 (D-0) and 7 (D-7) is not detected after a topical application of respective ED50 of genistein on days 0 and 7. Similar results were obtained in separate three experiments. C, control.
Phosphorylation of myosin light chain
The phosphorylation state of MLC in the basilar artery was examined by PPi-PAGE. Four bands-designated as F, AM, AMP1, and AMP2-were detected in the spastic group on days 2 and 7, as well as in the KCl-15, KCl-90, serotonin-15, and serotonin-90 groups (Fig. 6). Protein in F band showed the same mobility as did gizzard muscle filamin (Wang et al., 1975) (data not shown). Since PPi-PAGE in each group was performed separately, F, AM, AMP1, and AMP2 bands in each group in Fig. 6 were not arranged exactly along the same lines. To determine the extent of phosphorylation of MLC, each band on PPi-PAGE was subjected to urea-glycerol PAGE, as shown representatively in the spastic arteries on day 7 in Fig. 7. In addition to unphosphorylated 17-kd MLC, the AM band contained unphosphorylated 20-kd MLC, AMP1 monophosphorylated and unphosphorylated 20-kd MLC, and AMP2 monophosphorylated 20-kd MLC. The amounts of monophosphorylated and unphosphorylated 20-kd MLC in AMP1 are almost equivalent, indicating that AMP1 is composed of heterodimer with monophosphorylated and unphosphorylated 20-kd MLC. On the other hand, the unphosphorylated 20-kd MLC in the AM band and the monophosphorylated 20-kd MLC in the AMP2 band were, respectively, about twice as high as the unphosphorylated and the monophosphorylated MLC in AMP1, indicating that AM consists of a homodimer of unphosphorylated 20-kd MLC and AMP2 of homodimer of monophosphorylated 20-kd MLC. Similar phosphorylation states were shown in AM, AMP1, and AMP2 in other spastic group, as well as in the KCl and serotonin groups. The AM, AMP1, and AMP2 bands in the current study correspond to GM, GMP1, and GMP2, respectively, in gizzard myosin phosphorylated by MLCK (Takano-Ohmuro and Kohama, 1986a, b, and 1987).

Pyrophosphate polyacrylamide gel electrophoresis (PPi-PAGE) of basilar artery extract shows representatively four bands, designated F (line 1), AM (line 2), AMP1 (line 3), and AMP2 (line 4), similar to the spastic (lanes 1 and 2), KCl (lanes 3 and 4), and serotonin (lanes 5 and 6) groups. The results are from a representative study performed in five dogs in each group with comparable outcomes. Lane 1, day 2; lane 2, day 7; lane 3, 15 minutes after KCl treatment; lane 4, 90 minutes after KCl treatment; lane 5, 15 minutes after serotonin treatment; lane 6, 90 minutes after serotonin treatment.
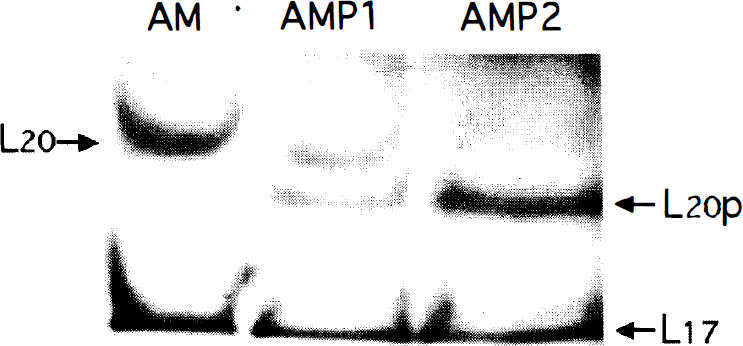
Urea-glycerol polyacrylamide gel electrophoresis. Three bands (AM, AMP1, and AMP2) on PPi-PAGE in the spastic basilar artery on day 7 were cut off separately and subjected to urea-glycerol PAGE. In addition to unphosphorylated 17-kd MLC (L17), AM is composed of unphosphorylated 20-kd MLC (L20), AMP1 of unphosphorylated (L20) and monophosphorylated (L20p) 20-kd MLC, and AMP2 of monophosphorylated 20-kd MLC (L20p). The densities of L20 in AM and L20p in AMP2 are about twice as high as L20 and L20p in AMP1, respectively, suggesting that AM consists of homodimer of L20 and AMP2 of homodimer of L20p. Similar results were achieved in five dogs in each of spastic, KCl, and serotonin groups with comparable outcomes.
Phosphorylation of calponin
Immunoprecipitated CaP (34 kd), visualized by silver staining, is shown representatively in Fig. 8A and B, and its mean percent levels are shown in Fig. 9. They were not changed significantly in the spastic group on day 0 or in the KCl-15, KCl-90, serotonin-15, and serotonin-90 groups, but decreased in the spastic group on days 2 and 7. The levels of phosphorylation of CaP on serine, threonine, and tyrosine residues are shown by immunoblotting of immunoprecipitated CaP with anti-phosphoserine (Fig. 8C and D), anti-phosphothreonine (Fig. 8E and F), and anti-phosphotyrosine antibodies (Fig. 8G and H), respectively. Their mean percent values of phosphorylation were calculated by dividing the densitometric values of phosphorylation levels of immunoprecipitated CaP on serine, threonine, and tyrosine residues by those of immunoprecipitated CaP levels visualized by silver staining, respectively. The mean percent levels of CaP phosphorylation on serine and threonine residues were not changed significantly in the spastic group on day 0 but increased on the spastic group on days 2 and 7 (Fig. 10). The mean percent levels of CaP phosphorylation on tyrosine residues also were enhanced significantly in the spastic group on days 0, 2, and 7 but were not changed significantly in the KCl and serotonin groups (Fig. 10).
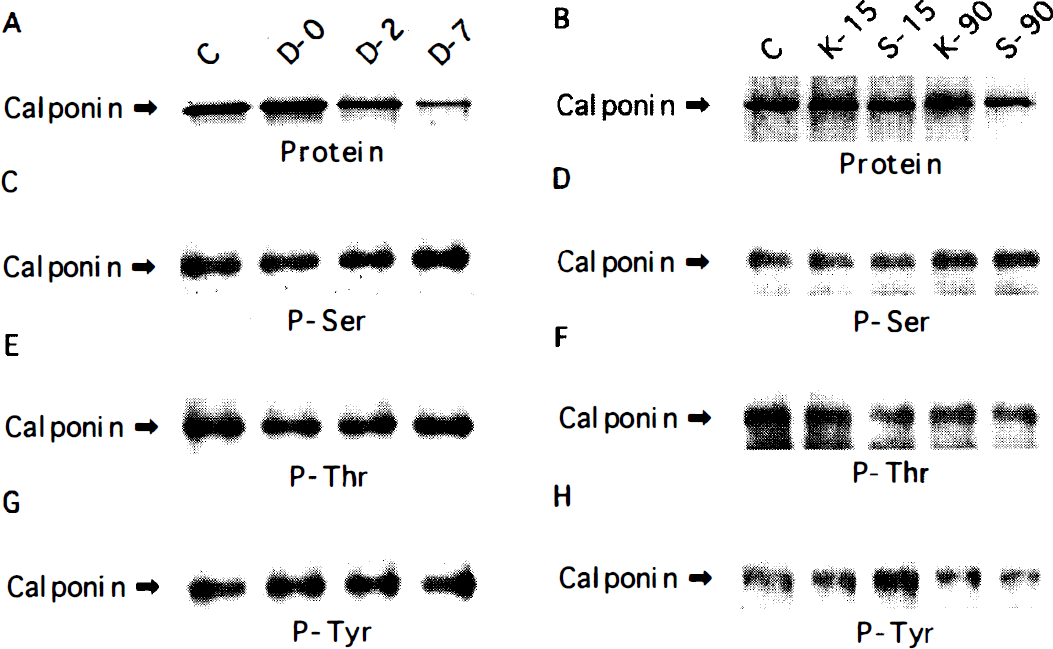
Phosphorylation of calponin. The basilar artery lysates were immunoprecipitated using anti-calponin antibody, resolved by 12% SDS-PAGE, and visualized with a highly sensitive silver staining, as shown in spastic group
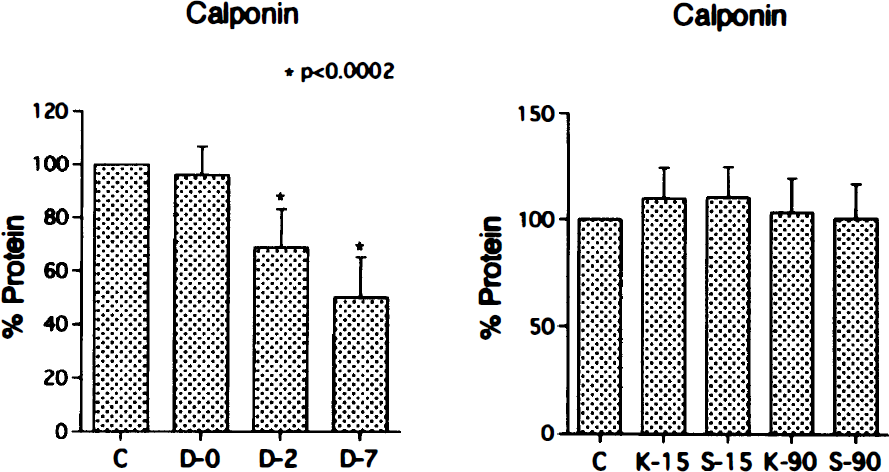
Bar graphs show mean percent values of immunoprecipitated calponin in five dogs in each group visualized by the silver staining and measured by densitometry. Vertical lines indicate SD. Significance was examined by two-tailed t test for uncorrelated pairs. C, control; D-0, vasospasm on day 0; D-2, vasospasm on day 2; D-7, vasospasm on day 7; K-15, vasocontraction 15 minutes after KCl treatment; K-90, vasocontraction 90 minutes after KCl treatment; S-15, vasocontraction 15 minutes after serotonin treatment; S-90, vasocontraction 90 minutes after serotonin treatment.
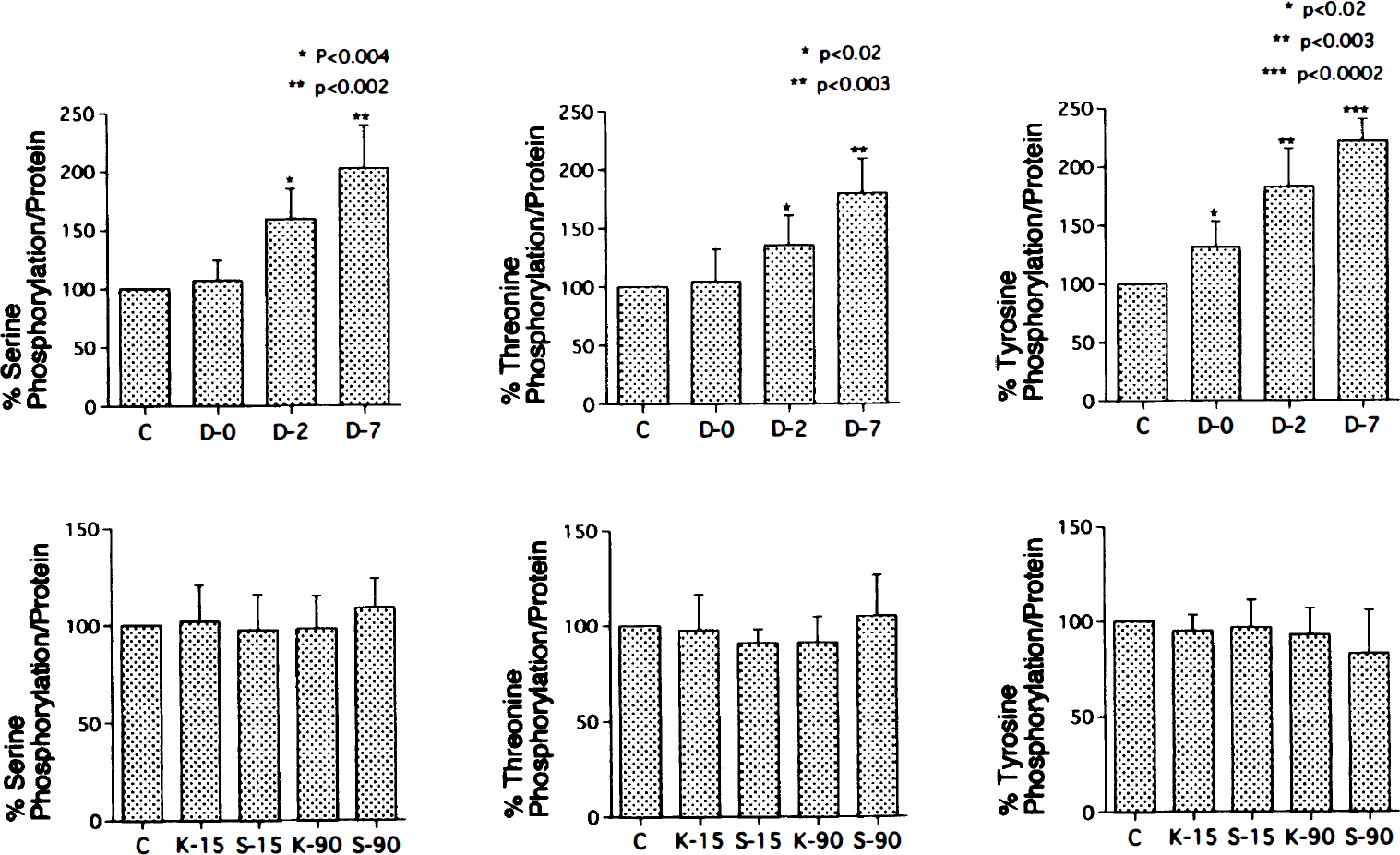
Bar graphs show mean percent levels of phosphorylation of immunoprecipitated calponin on serine, threonine, or tyrosine residues in five dogs in each group. The mean percent phosphorylation levels of calponin were calculated by dividing densitometric values of the phosphorylation levels of immunoprecipitated calponin by those of immunoprecipitated calponin levels visualized by silver staining. Vertical lines indicate SD. Significance was examined by two-tailed t test for uncorrelated pairs. C, control; D-0, vasospasm on day 0; D-2, vasospasm on day 2; D-7, vasospasm on day 7; K-15, vasocontraction 15 minutes after KCl treatment; K-90, vasocontraction 90 minutes after KCl treatment; S-15, vasocontraction 15 minutes after serotonin treatment; S-90, vasocontraction 90 minutes after serotonin treatment.
DISCUSSION
Activation of tyrosine kinase pathway in vasospasm
Increases in phosphorylation of intracellular substrates of tyrosine kinase, including Shc, Raf1, and ERK, in spastic basilar artery clearly indicate activation of the tyrosine kinase pathway. One established mechanism of receptor protein tyrosine kinase coupling involves activation of Ras by translocation of the Grb2-Sos complex to the plasma membrane as a consequence of its binding to an autophosphorylation site in the receptor protein tyrosine kinase itself or to substrate or adapter protein such as Shc, phosphorylated by the receptor protein kinase (van der Geer et al., 1994). In addition, Raf1 and ERK also are activated by both Ras-dependent (Crespo et al., 1994; van Biesen et al., 1995) and Ras-independent (van Biesen et al., 1996) signaling pathways in response to activation of G protein-coupled receptors. The release of the βγ subunits of heterotrimeric G proteins after ligand binding to Gi-coupled receptors led to tyrosine phosphorylation of Shc and increased functional association of Shc, Grb2, and Sos (van Biesen et al., 1995). In addition, Gq protein-coupled angiotensin II receptor activates the Fyn-Shc-Grb2-Sos pathway (Sadoshima and Izumo, 1996). These data suggest that Gβγ subunits can activate Ras using mechanisms that are similar to those used by receptor tyrosine kinase, and that Shc is implicated as a go-between that relays a message from Gβγ-coupled receptors to protein tyrosine kinase receptors. Thus, the current results of signal transduction of tyrosine kinase pathway in vasospasm indicate that tyrosine kinase is activated by signaling from tyrosine kinase receptors, G-coupled receptors, or both. Since Raf1 and ERK activation is mediated by PKC (Rapp, 1991; Thomas et al., 1992) in addition to activation of tyrosine kinase receptors or G-coupled receptors, activated PKCα and PKM in vasospasm (Sato et al., 1997) may contribute to the activation Raf1 and ERK.
In addition, the spastic basilar arteries on days 0 and 7 are dilated dose-dependently by the topical application of genistein with ED50 of 14.0 ± 2.1 μmol/L on day 0 and 17.2 ± 4.5 μmol/L on day 7. The ED50 values of genistein are 2.6 to 26 μmol/L for protein tyrosine kinase of epidermal growth factor receptor and pp60v-src (Akiyama and Ogawa, 1991). DiSalvo and others (1993) report that tyrosine kinase inhibitors such as genistein exhibit high specificity for inhibition of smooth muscle tyrosine kinases and do not affect the activity of vascular smooth muscle MLCK or cyclic AMP-dependent protein kinase. Thus, the dilation of the spastic basilar artery by genistein does not result from the inhibition of MLCK but is closely associated with the decrease in phosphorylation of intracellular substrates of tyrosine kinase, including Shc.
The activation of protein tyrosine kinase receptors and G protein-coupled receptors of vascular smooth muscles stimulates the hydrolysis of phosphatidylinositol 4,5-bisphosphate, yielding the two second messengers inositol 1,4,5-trisphosphate (IP3) and diacylglycerol. IP3 mobilizes Ca2+ from sequestered stores, whereas diacylglycerol stimulates PKC. Thus, the increase in intracellular Ca2+ and the activation of PKC mediated by tyrosine kinase pathway are important causal events to induce vasospasm. The mobilization of Ca2+ from sequestered stores appears to be limited, but since the topical treatment with EGTA induces the reversal of vasospasm, the continuously increased intracellular Ca2+ in vasospasm also may result from Ca2+ influx in addition to the intracellular mobilization of Ca2+. The absence of PKM in the spastic artery after genistein treatment indicates the inactivation of μ-calpain, which may be induced by the decline of intracellular Ca2+ levels through the inhibition of tyrosine kinase pathway. The activation of Raf1 and ERK in vasospasm are declined on day 7 compared with those on days 0 or 2, suggesting that the activation of tyrosine kinase pathway is more closely associated with the early than with the late stage of vasospasm.
Phosphorylation of myosin light chain in vasospasm
It is generally accepted that the MLCK-catalyzed phosphorylation of MLC is a dominant regulation for the smooth muscle contraction (Kamm and Stull, 1985). The PPi-PAGE resolves smooth muscle MLC into three bands in the MLCK-mediated phosphorylation (Takano-Ohmuro et al., 1986a,b) and into a single band in the PKC-mediated phosphorylation (Trybus and Lowey, 1985) based on the phosphorylation state. These results are compatible with observations that MLCK and PKC phosphorylate smooth muscle MLC at different sites, with serine 19 and threonine 18 being the primary and the secondary residues, respectively, in MLCK-mediated phosphorylation (Ikebe and Hartshorne, 1985), and threonine9 and serine1 or serine2 the primary and the secondary residues, respectively, in PKC-mediated phosphorylation (Nishikawa et al., 1984; Ikebe et al., 1987). The difference in MLCK- and PKC-specific phosphorylation sites of MLC also is compatible with the fact that phosphorylation by MLCK activates smooth muscle actin-activated Mg2+-ATPase, but phosphorylation by PKC does not (Nishikawa et al., 1984; Kamm and Stull, 1985; Ikebe et al., 1987). When MLC is phosphorylated by MLCK and PKC, PPi-PAGE analysis shows a single band (Takano-Ohmuro and Kohama, 1987). The current study clearly demonstrates the resolution of MLC into three bands based on the phosphorylation state in the spastic, KCl, and serotonin groups, suggesting that MLC in the three groups is phosphorylated by MLCK but not by PKC.
Biochemical analysis reveals that PKC reduces the actin-activated Mg2+-ATPase activity of myosin prephosphorylated by MLCK by approximately 50% (Ikebe et al., 1987; Nishikawa et al., 1984). In addition, the PKC-catalyzed phosphorylation of MLCK decreases its activity (Ikebe and Hartshorne, 1985), suggesting that the in vitro effects of PKC-catalyzed phosphorylation of MLC and MLCK would be consistent with a relaxation rather than a contractile response. The only response of permeabilized gizzard smooth muscle in response to phosphorylation of MLC on PKC-specific residues is an attenuation of Ca2+-induced contraction (Sutton and Haeberle, 1990; Parente et al., 1992). In addition, phorbol ester-induced contractions of smooth muscles are not accompanied by significant phosphorylation of MLC on the PKC-specific sites (Singer, 1990; Sutton and Haeberle, 1990). Furthermore, contraction of intact smooth muscle fibers in response to physiologic agents that activate PKC is not accompanied by MLC phosphorylation on PKC-specific sites but is accompanied by MLC phosphorylation on MLCK-specific sites (Singer, 1990). Thus, much effort has been devoted to identifying the substrates of PKC that are involved in the contractile response, but the weight of evidence is against either MLC or MLCK being involved as substrates of PKC. The decrease in activity of smooth muscle PP1 in vasospasm (Fukami et al., 1995) probably results from activation of PKC (Masuo et al., 1994) and contributes to maintaining MLC phosphorylation.
Involvement of calponin in vasospasm
Several examples have been reported of an increase in CaP phosphorylation in intact smooth muscle in response to various contractile stimuli (Winder et al., 1993; Carmichael et al., 1994), and the CaP phosphorylation during contraction of porcine coronary artery induced by both endothelin-1 and phorbol 12,13-dibutyrate was first reported to be catalyzed in vivo predominantly by PKC (Mino et al., 1995). Other investigators, however, report that CaP is not phosphorylated in intact muscles in response to stimuli (Bárány and Bárány, 1993; Adam et al., 1995). There has been growing physiologic support for the role of CaP in inhibiting the actin-activated Mg2+-ATPase activity of smooth muscle through its binding to actin, the phosphorylation of CaP markedly lowers the affinity of CaP for actin to alleviate inhibition of actin-activated myosin Mg2+-ATPase activity (Ito et al., 1995; Horowitz et al., 1996a; Pohl et al., 1997), and the properties of CaP are restored after dephosphorylation (Winder et al., 1992; Fraser and Walsh, 1995). The mechanism of involvement of PKC in vasospasm remains controversial. Membrane PKC activity has been reported to increase with a reciprocal decrease in cytosolic PKC activity (Nishizawa et al., 1992), that is, the activation of intact PKC. Another report shows a decrease of 40% to 45% in cytosolic PKC activity without any significant changes in membrane PKC activity, indicating an absence of activation of intact PKC; levels of immunoreactive PKCα and PKCε but not PKCζ were decreased in spastic arteries (Takuwa et al., 1993). We have demonstrated the activation of PKCα and the generation of PKM in vasospasm (Minami et al., 1992; Sato et al., 1997). The activation of PKC may be induced by the diacylglycerol in the presence of the increased Ca2+ produced by the hydrolysis of phosphatidylinositol through the activation of tyrosine kinase pathway and phosphorylate CaP in vasospasm. The current study demonstrates that protein levels and phosphorylation levels on serine and threonine residues of CaP are not changed significantly in vasospasm on day 0 or in vasocontraction induced by KCl or serotonin. On the other hand, vasospasm on days 2 and 7 is associated with a significant decrease in CaP levels (Oka et al., 1996) and with a significant increase in CaP phosphorylation on serine and threonine residues. Both alterations are more marked in vasospasm on day 7 than on day 2 and are predicted to alleviate its inhibition of the actin-activated myosin Mg2+-ATPase to induce a potential increase of smooth muscle contractility in vasospasm. In addition, decreased activities of smooth muscle PP1 and PP2A in vasospasm (Fukami et al., 1995) may contribute further to the maintenance of CaP phosphorylation because PP1 and PP2A dephosphorylate CaP (Winder et al., 1992; Ichikawa et al., 1993). The phosphorylation of CaP on tyrosine residues, which was increased significantly in early and delayed vasospasm but not in vasocontraction induced by KCl or serotonin, may be associated with activation of tyrosine kinase pathway in vasospasm, but its significance is unknown.
Footnotes
Acknowledgements
The authors thank Michinari Hamaguchi, MD, PhD, Department of Molecular Pathogenesis, Nagoya University School of Medicine, and Katsuhide Mabuchi, PhD, Muscle Research Group, Boston Biomedical Research Institute, for their kind advice.
