Abstract
Recent evidence has shown crucial roles for cell-adhesion molecules in inflammation-induced rolling, adhesion, and accumulation of neutrophils in tissue. Intercellular adhesion molecule-1 (ICAM-1) is one of these adhesion molecules. Previous studies have shown marked reduction in the size of infarction after focal cerebral ischemia by depletion of granulocytes and administration of the antibody against ICAM-1. In the present study we investigated the role of ICAM-1 in the size of ischemic lesions, accumulation of granulocytes, and microcirculatory compromise in focal cerebral ischemia by using ICAM-1–knockout mice. Ischemic lesions were significantly mitigated in knockout mice after permanent and transient focal ischemia, even though the number of granulocytes in the infarcted tissue was almost the same between knockout and wild-type mice. Depletion of granulocytes further decreased the size of ischemic lesions after transient focal ischemia in ICAM-1–knockout mice. Microcirculation was reduced after focal ischemia, but it was better preserved in the cerebral cortex of knockout mice than that of wild-type mice. The present study demonstrated that ICAM-1 played a role in microcirculatory failure and subsequent development and expansion of infarction after focal cerebral ischemia. However, it is highly unlikely that ICAM-1 played a key role in accumulation of granulocytes after focal cerebral ischemia.
Keywords
Accumulation and transmigration of leukocytes, especially granulocytes, have drawn considerable attention as an aggravating factor in expansion of cerebral infarction (Hallenbeck et al., 1986; Barone et al., 1991; Garcia et al., 1994). Granulocytes can adhere to the capillary endothelium to cause microvascular occlusion (Schmidt-Schonbein, 1987; del Zoppo et al., 1991), and they can release free radicals and damage surviving neurons around the infarction (Matsuo et al., 1995). Depletion of granulocytes reduced the size of infarction after focal cerebral ischemia (Chen et al., 1992; Matsuo et al., 1994).
Accumulation of granulocytes on the endothelial surface and transmigration into inflammatory tissue may need interaction of several adhesion molecules and cytokines. It is believed that the selectin family, including P-selectin, mediates rolling or slowing of neutrophils on the endothelial surface (Lasky, 1992), and then intercellular adhesion molecule 1 (ICAM-1) executes firm adhesion and emigration of neutrophils (Smith et al., 1988). Expression of P-selectin and ICAM-1 has been detected in the endothelium several hours after the onset of ischemia in experimental animals (Okada et al., 1994; Zhang et al., 1995a). Synthetic peptides for lectin-binding domain of P-selectin (Morikawa et al., 1996) and the antibody against ICAM-1 (Zhang et al., 1994, 1995b) have been reported to diminish the size of infarction after focal cerebral ischemia with reperfusion.
Recent studies using ICAM-1–knockout mice showed reduction of ischemic lesions after transient focal cerebral ischemia with suture occlusion model (Connolly et al., 1996; Soriano et al., 1996). However, these groups did not consider variation in the patency of posterior communicating artery (PcomA) in each mouse, which has proved to be a crucial factor in determining ischemic severity and extent in the suture occlusion model (Kitagawa et al., 1998). Furthermore, it still remains unclear how ICAM-1 contributes to tissue damage, granulocyte accumulation, and microcirculatory disturbance after focal cerebral ischemia.
In this study, we tried to find answers to the following four questions by using an improved suture occlusion model. The first question is whether deficiency of ICAM-1 is protective not only after transient focal cerebral ischemia but also in the periphery of permanent focal ischemia. There is no information available regarding the latter type of cerebral ischemia. The second is whether deficiency of ICAM-1 diminishes accumulation of granulocytes in cerebral infarction. The third is whether deficiency of ICAM-1 is sufficient to suppress the pathogenic role of granulocytes in cerebral ischemia. The last question is whether deficiency of ICAM-1 reduces microcirculatory compromise during expansion of infarction after focal ischemia.
MATERIALS AND METHODS
Selection of ICAM-1–knockout mice
Intercellular adhesion molecule 1–knockout mice, originally produced by Sligh et al. (1993), were purchased from the Jackson Laboratory (Bar Harbor, Maine, U.S.A.). We first back-crossed them with the C57BL/6 mice (Charles River Inc., Yokohama, Japan) to have genetic backgrounds of the homozygote and wild-type mice, and used them in the subsequent experiments. The C57BL/6 strain was chosen because mice of this strain show little variation in intracranial collaterals and lack the patent PcomA on both sides (Yang et al., 1997; Kitagawa et al., 1998). After mating heterozygotes, we selected the homozygous and wild-type mice by polymerase chain reaction (PCR) amplification of genomic DNA extracted from the tail. The PCR primers used were oIMR017, 5′-CTGAGCCAGCTGGAGGTCTCG; oIMR018, 5′-GAGCGGCAGAGCAAAAGAAGC; and oIMR019, 5′-AGGACAGCAAGGGGGAGGATT. The sequence for oIMR017 and oIMR018 will amplify a 178-bp product from the wild-type allele. The sequence for oIMR018 and oIMR019 will amplify a 150-bp product from the mutant allele. The genotype was again confirmed after killing homozygous and wild-type mice by immunoblotting of the lung homogenate using an antibody against murine ICAM-1 (Pharmingen, San Diego, U.S.A.). All homozygous and wild-type mice used in the present experiments were mature males aged 8 to 16 weeks, weighing 25.6 ±1.5 g (homozygous, n = 64) and 25.5 ±1.4 g (wild-type, n = 52), respectively. Mice were given free access to food and water before surgery. The experimental procedures involving laboratory animals have been approved by the Institutional Animal Care and Use Committee of the Osaka University Medical School.
Surgery
General anesthesia was induced with 2.0% halothane and maintained with 0.5% halothane by means of an open face mask. Two polyacrylamide columns with an inner diameter of 0.8 mm for measurement of cortical microperfusion by laser—Doppler flowmetry (Advance Laser Flowmetry, Model ALF-21, Advance Co., Ltd., Tokyo, Japan) were attached to the intact skull, 3.5 mm and 1.0 mm lateral to the bregma with dental cement. A metal plate-type thermometer with a diameter of 3.0 mm was also attached to the skull over the parietal cortex to record skull temperature. A femoral artery was cannulated with a PE-10 polyethylene tube connected to a pressure transducer attached to a calibrated polygraph, and body temperature was monitored with a rectal thermometer. Body and skull temperatures were monitored and maintained at 36.0° to 37.5°C and 35.0° to 36.5°C, respectively, using a heat lamp. Blood pH, Pa
Evaluation and prediction of the patency of PcomA. To eliminate the influence of variation in the patency of PcomA in the severity of transient focal ischemia produced by intraluminal suture occlusion (Huang et. al., 1994; Yang et al., 1994), we first evaluated the relationship between reduction of cortical microperfusion during a 1-minute bilateral common carotid artery (CCA) occlusion and the patency of the PcomA (Kitagawa et al., 1998). Five homozygous and five wild-type mice were used. After blood sampling (150 µL) for gas analysis through the arterial cannula, each mouse was deeply anesthetized with sodium pentobarbital, and a sternotomy was performed to expose the heart. An incision was made in the right atrium, and the mouse was perfused with a short flush of physiologic saline, then with 10% formalin, and finally with a mixture of gelatin and India ink prepared according to Berry and colleagues (1975). The circle of Willis was then examined under a dissecting microscope and the PcomA was identified as the connection between the carotid and vertebrobasilar circulations. The degree of anastomosis was assessed by comparing the diameter of the PcomA on each side with the diameter of the basilar artery, and graded as 0 or 1. Grade 0 was assigned to specimens having no anastomosis or less than one third of the diameter of the basilar artery, and grade 1 was assigned when the arterial diameter was more than one third of the diameter of the basilar artery. Thus, the sum of the scores from both sides was 0, 1, or 2.
Permanent focal ischemia. The left middle cerebral artery (MCA) was permanently occluded by electrocoagulation (Tamura et al., 1981; Welsh et al., 1987; Kinouchi et al., 1991) with slight modification. Mice were placed in the recumbent position, and a curved vertical skin incision was made in the midpoint between the left orbit and the external auditory canal. The masseter muscles were incised at the inferior edge of the zygoma, and then the mandible was pulled downward to expose the skull base. Using a dental drill, a small burr hole was made into the skull over the left MCA. The dura was opened with a fine needle and the MCA was picked up. By using a microbipolar electrocoagulator at minimal power, the MCA was permanently occluded just proximal to the point where the olfactory branch came off. Cortical microperfusion, blood pressure, and body and skull temperatures were monitored and maintained under halothane anesthesia for 30 minutes after MCA occlusion. After discontinuation of halothane anesthesia, each mouse was allowed to recover for 2 hours in a chamber where the ambient temperature was maintained at 35°C to prevent hypothermia, and then each mouse was kept at room temperature.
Transient focal ischemia. Only mice that showed less than 10% of baseline cortical microperfusion as measured by laser—Doppler flowmetry during a 1-minute bilateral CCA occlusion were used. Intraluminal occlusion of the intracranial artery, including the internal carotid artery and MCA, by a surgical suture (Huang et al., 1994; Yang et al., 1994) was accomplished with slight modification under continuous monitoring of cortical perfusion by laser—Doppler flowmetry. The right CCA was exposed and ligated, and the right external carotid artery was isolated and ligated. A 6-0 monofilament nylon suture, blunted at the tip, was introduced through the carotid bifurcation into the right internal carotid artery and advanced until cortical microperfusion over the MCA territory dropped suddenly. After occlusion for 30 minutes or 45 minutes, blood flow was restored through the anterior cerebral artery by removal of the nylon suture.
Evaluation of ischemic damage
Forty-eight hours after permanent focal ischemia, animals were killed with an overdose of pentobarbital. Brains were removed and sectioned coronally into four 2-mm slices by using a mouse brain matrix (Muromachi Kikai Co., Ltd. Tokyo, Japan). Slices were placed in 2% 2,3,5-triphenyltetrazolium chloride (TTC) solution for 30 minutes at 37°C and fixed with 10% formalin overnight. The ischemic lesions without reaction to TTC were measured on the posterior surface of each slice. Forty-eight hours after permanent or transient focal ischemia, the whole brains were fixed for histologic and immunohistochemical examination of ischemic lesions by immersion into alcohol-5% acetic acid solution for 5 hours at 4°C, dehydrated, and embedded in paraffin as described previously (Yanagihara et al., 1985). Tissue sections (5 µm) were obtained every 1 mm beginning at the frontal pole and examined after conventional staining with hematoxylin-eosin and cresyl violet, as well as after immunohistochemical reaction for microtubule-associated protein 2 (MAP2) (Kitagawa et al., 1989). The volume of ischemic lesions was measured by using an MCID image analysis system (Imaging Research, Inc., St. Catharines, Ontario, Canada). The area with ischemic lesions and the area of both hemispheres (mm2) were calculated on TTC-stained and hematoxylin-eosin-stained coronal sections by tracing these areas on the computer screen. The volume (mm3) was determined by integrating the appropriate area and the section thickness. To reduce errors associated with processing of tissue for histologic evaluation, the lesion area and volume were corrected using the method of Swanson et al. (1990).
Ischemic damage in the caudoputamen and hippocampus after transient focal ischemia was examined with semiquantitative analysis. In the caudoputamen, ischemic lesion was expressed as the percentage of the region from the whole caudoputamen that lost the immunoreaction for MAP2. In the hippocampus, the degree of damage was assessed in the CA1 to CA3 sector and divided into the following four grades based on the percentage of damaged cells: grade O, no cell is damaged; grade 1, less than 10% of cells are damaged; grade 2, 10% to 50% of cells are damaged; and grade 3, more than 50% of cells are damaged. The distance of the CA1 to CA3 sector with each grading was measured and the mean histologic score was obtained for each mouse by dividing the integration of each grading and its length by the total length of the CA1 to CA3 sector as follows:
Assessment for accumulation of granulocytes
With the same paraffin sections as used for histologic evaluation of ischemic damage, accumulation of granulocytes was evaluated with the immunohistochemical reaction with an anti-myeloperoxidase (MPO) antibody (Dako, Glostrup, Denmark) as described previously (Chopp et al., 1996). The number of cells with positive immunoreaction for MPO was counted in the center of the MCA territory of the frontoparietal cortex and in the dorsolateral part of the caudoputamen by using the sections 3 mm from the frontal pole, and in the hippocampus by using the sections 5 mm from the frontal pole.
Depletion of granulocytes in ICAM-1–knockout mice
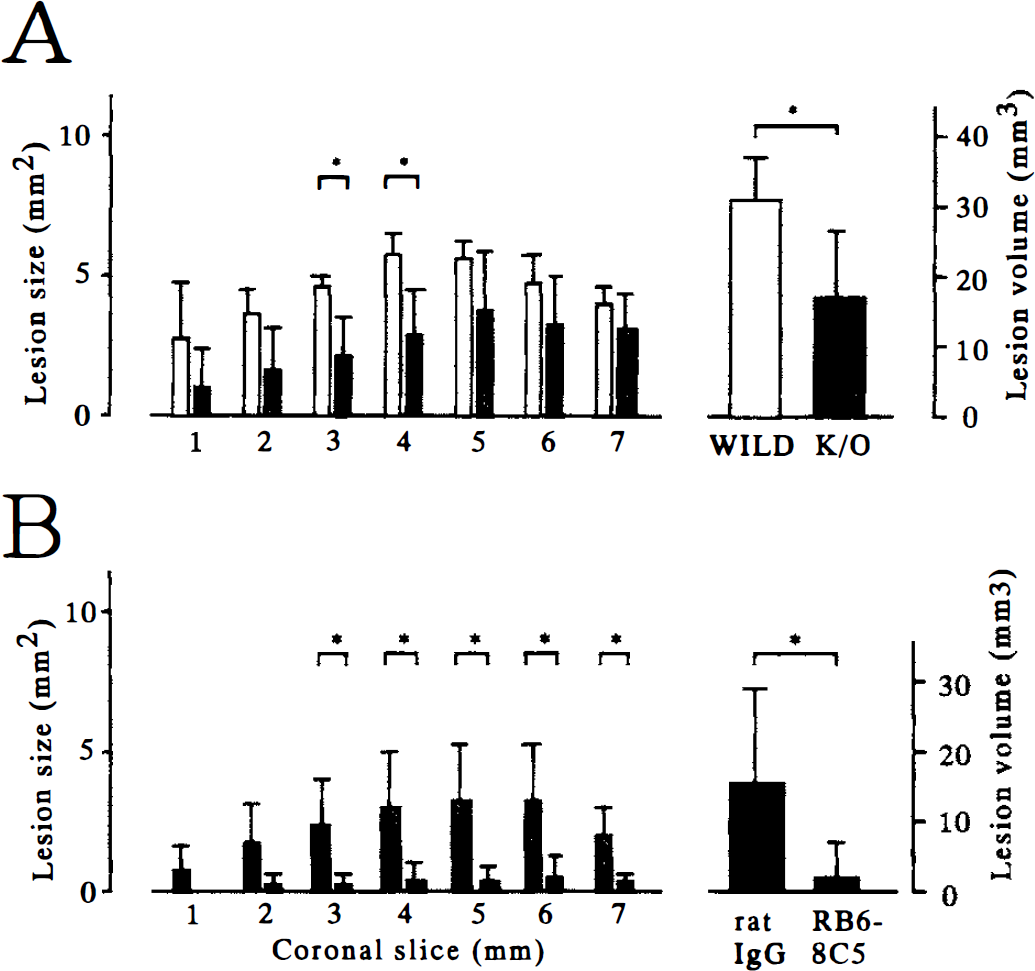
A monoclonal antibody RB6-8C5 (Pharmingen, San Diego, U.S.A.) was used to deplete granulocytes in ICAM-1–knockout mice as described previously (Stoppacciaro et al., 1993). Two hundred micrograms of the antibody was injected intraperitoneally in six ICAM-1–knockout mice 24 hours before induction of ischemia, and these mice were subjected to transient focal cerebral ischemia for 45 minutes. Before induction of cerebral ischemia, agranulocytosis was confirmed by Wright-Giemsa-stained peripheral blood smears. Six control ICAM-1–knockout mice received rat IgG before being subjected to transient focal cerebral ischemia for 45 minutes. Two days after intraluminal suture occlusion for 45 minutes, each mouse was decapitated and their brains were examined histologically.
Evaluation of cerebral microcirculation
To visualize the plasma circulation, we labeled plasma with dichlorotriazinyl amino fluorescein (DTAF; excitation 489 nm, emission 515 nm, Sigma-Aldrich, Tokyo, Japan) as described previously (Yoshimine et al., 1985). Forty milligrams of DTAF was dissolved in 4 mL of 0.5 mol/L Tris-HCl, pH 9.0, to conjugate DTAF with serum proteins at room temperature for 1 hour while maintaining pH at 9.0 by adding 1 N NaOH. At the end of the reaction, pH was readjusted to 7.4 by adding 0.3 N HCl, and the solution was stored in aliquots at -20°C. After permanent MCA occlusion for 6 hours or transient internal carotid artery occlusion with nylon suture for 30 minutes and reperfusion for 6 hours, 50 µL of DTAF conjugated to serum was injected for 10 seconds into the saphenous vein. Thirty seconds after completion of the injection, each mouse was decapitated and the brain was fixed in 80% ethanol for 24 hours. Brain slices, 50 µm in thickness, were prepared with a vibratome and mounted in glycerol-phosphate-buffered saline and examined under a fluorescence microscope. Photographs were taken in the core and the peripheral area of the MCA territory in the cerebral cortex and in the dorsolateral part of the caudoputamen. Each photograph was analyzed for the total length of microvessels filled with fluorescence by using the MCID image analysis system.
Statistics
All values reported here are expressed as mean ± standard deviation (SD). Statistical significance of differences among groups was tested by one-way analysis of variance. P < 0.05 was considered significant.
RESULTS
Confirmation of genotype
Polymerase chain reaction analysis of genomic DNA from ICAM-1–/–, ICAM-1+/–, and ICAM-1+/+ mice with three primers (oIMR017, oIMR018, and oIMR019) showed a 150-bp band, 150-bp and 178-bp bands, and a 178-bp band, respectively (Fig. 1A). Western blot analysis of total lung protein from wild-type (+/+) and homozygous mutant (−/–) mice showed no ICAM-1 protein in the lung of homozygous mutant mice (Fig. 1B).
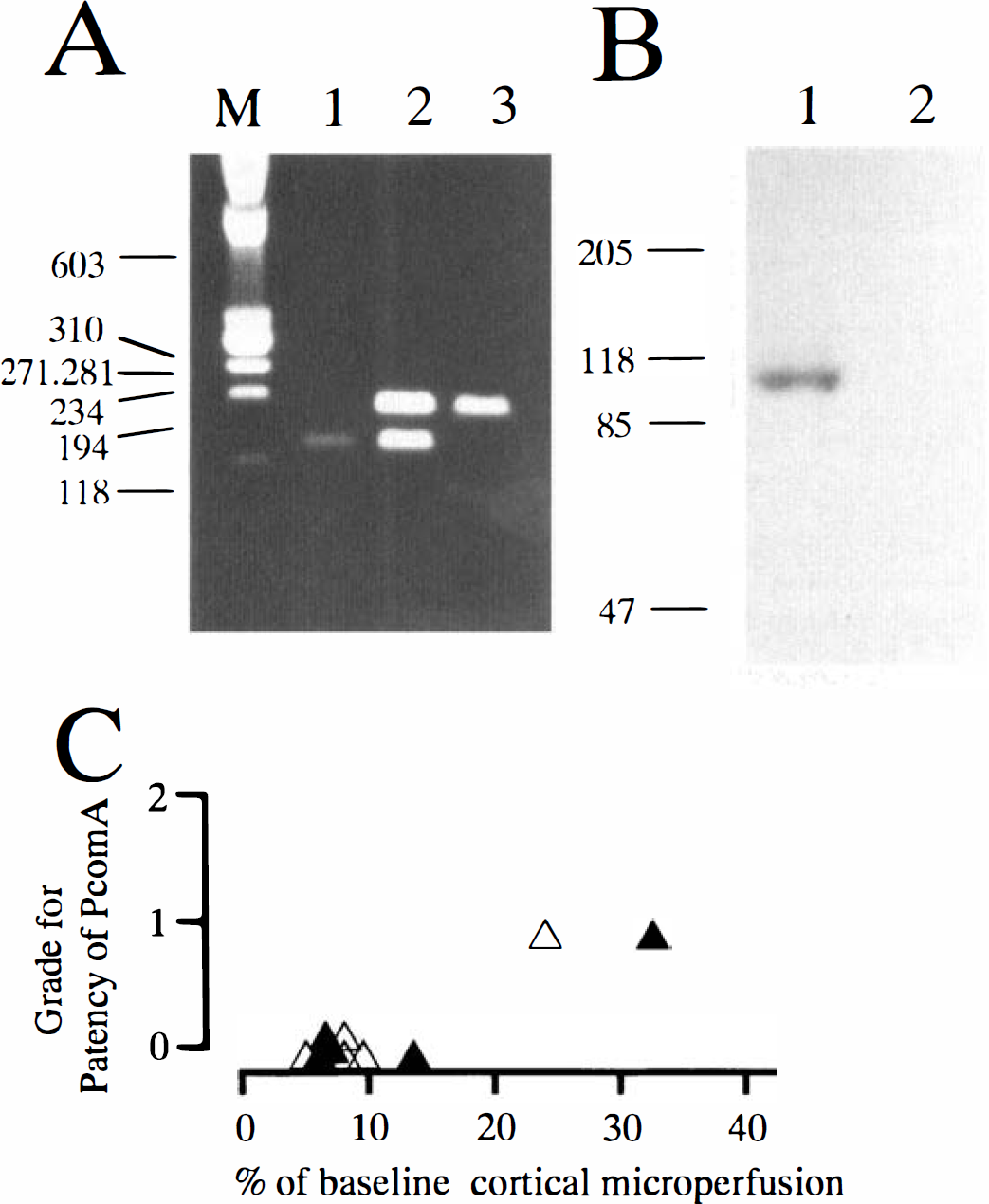
Genotyping of intercellular adhesion molecule 1 (ICAM-1)–knockout mice with polymerase chain reaction (PCR) and immunoblotting, and prediction of the intracranial collateral circulation and the extent of ischemia after bilateral common carotid artery occlusion.
Physiologic parameters
The physiologic parameters while allowing spontaneous breathing under halothane anesthesia were determined for Pa
Evaluation of intracranial collateral circulation via the posterior communicating artery
Reduction of cortical microperfusion during a 1-minute bilateral CCA occlusion was closely related to the patency of the PcomA (Fig. 1C). Bilateral CCA occlusion reduced cortical microperfusion to less than 10% of the baseline in four of five homozygous and wild-type mice, respectively. In those mice, no patent PcomA was identified on either side (Fig. 1C). Therefore, in the following experiments using transient focal ischemia with a nylon suture, we used only mice showing less than 10% of basal cortical microperfusion during a 1-minute bilateral CCA occlusion.
Ischemic lesions in cerebral ischemia
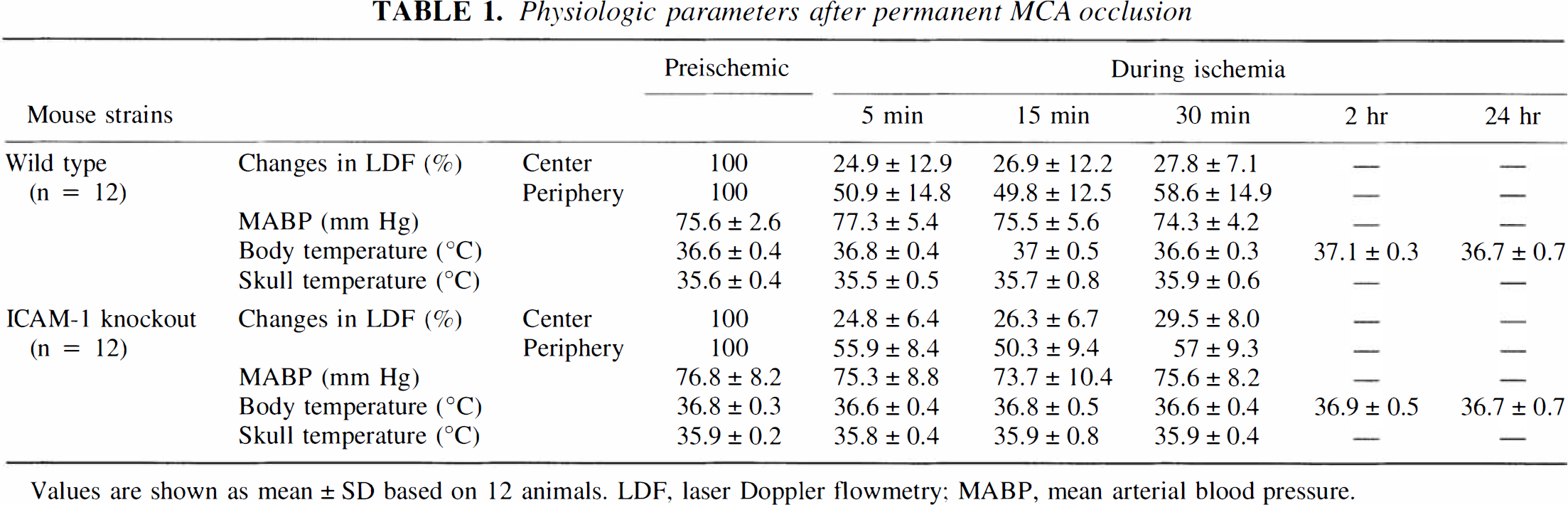
Permanent focal ischemia. Cortical microperfusion in the ischemic center and peripheral area after MCA occlusion was 25% to 30% and 50% to 60% of the baseline values, respectively, and the values were very similar between knockout and wild-type mice (Table 1). Other physiologic parameters, including blood pressure and body and skull temperatures, were maintained similarly between both types of mice (Table 1).
Physiologic parameters after permanent MCA occlusion
Values are shown as mean ± SD based on 12 animals. LDF, laser Doppler flowmetry; MABP, mean arterial blood pressure.
Physiologic parameters after transient focal cerebral ischemia
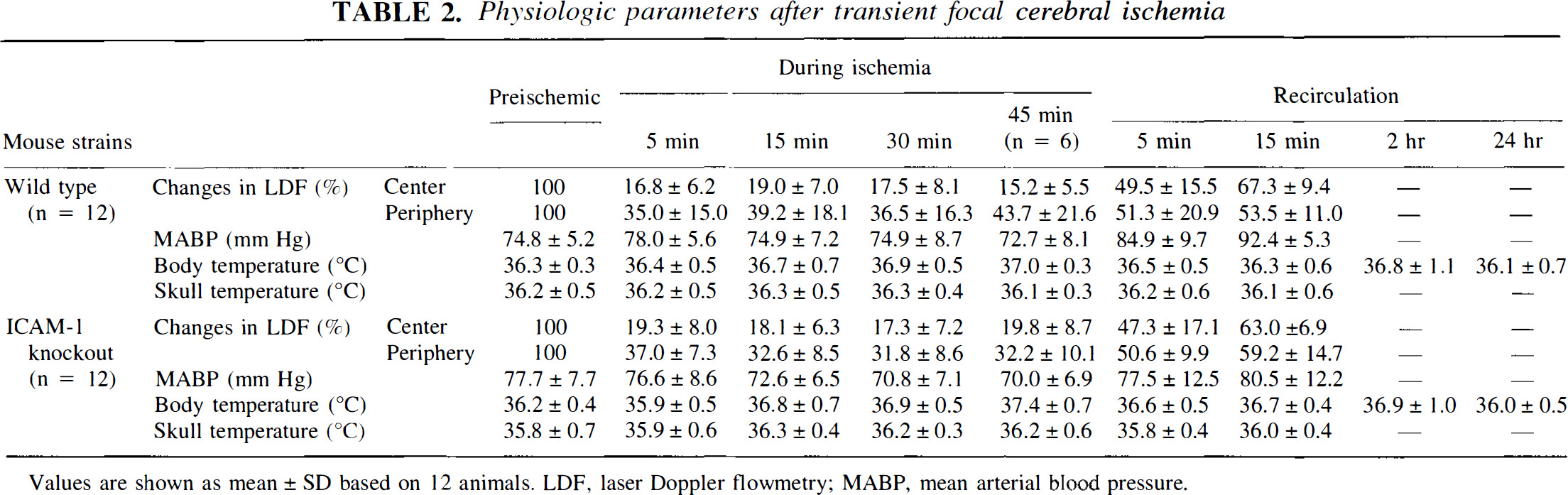
Values are shown as mean ± SD based on 12 animals. LDF, laser Doppler flowmetry; MABP, mean arterial blood pressure.
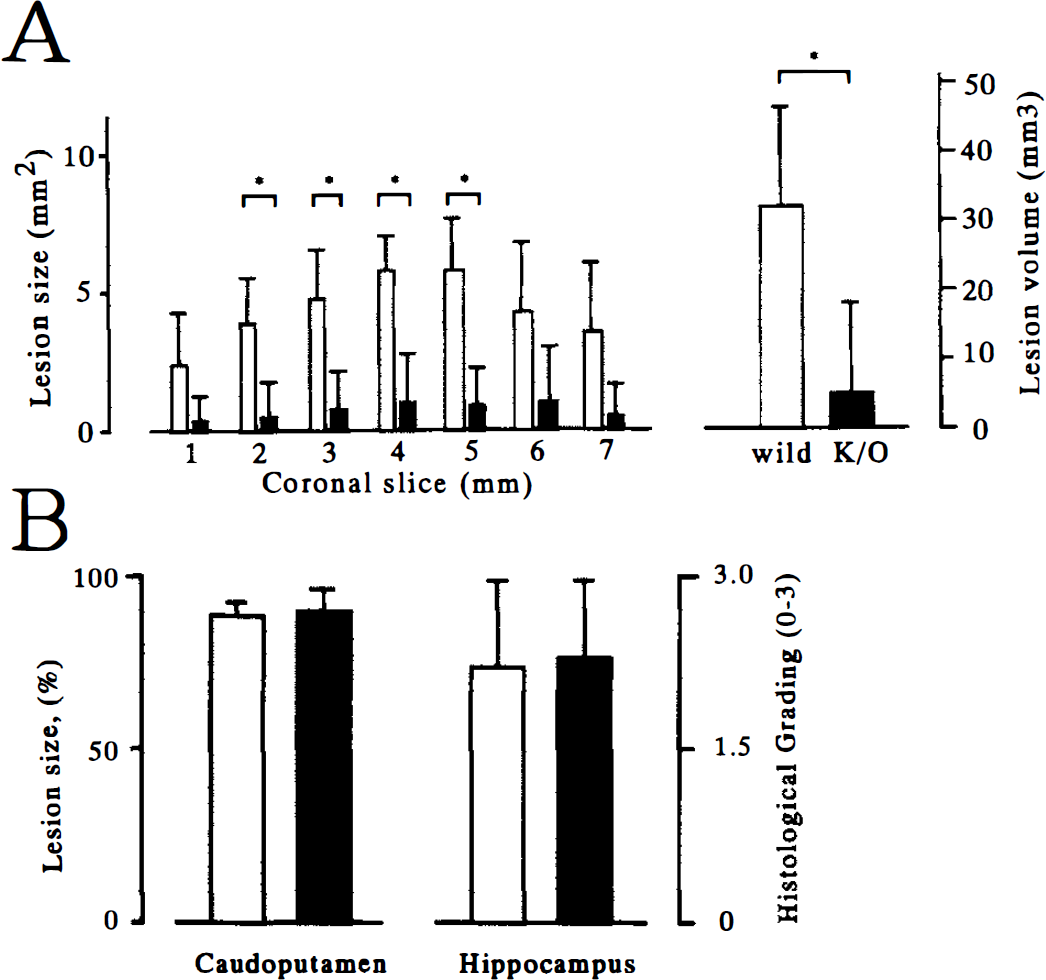
Lesion areas detected with TTC staining were significantly smaller in knockout mice than those in wild-type mice on the coronal slices 3 mm and 5 mm from the frontal pole (Fig. 2A). The total lesion volume was also smaller in knockout mice (Fig. 2A). The difference was mainly in the peripheral area of the cerebral cortex supplied by the MCA. Histologic examination also showed that ischemic damage was significantly smaller in knockout mice than that in wild-type mice on the coronal slices 5 mm and 6 mm from the frontal pole (Figs. 2B). The total lesion volume was also smaller in knockout mice (Fig. 2B).
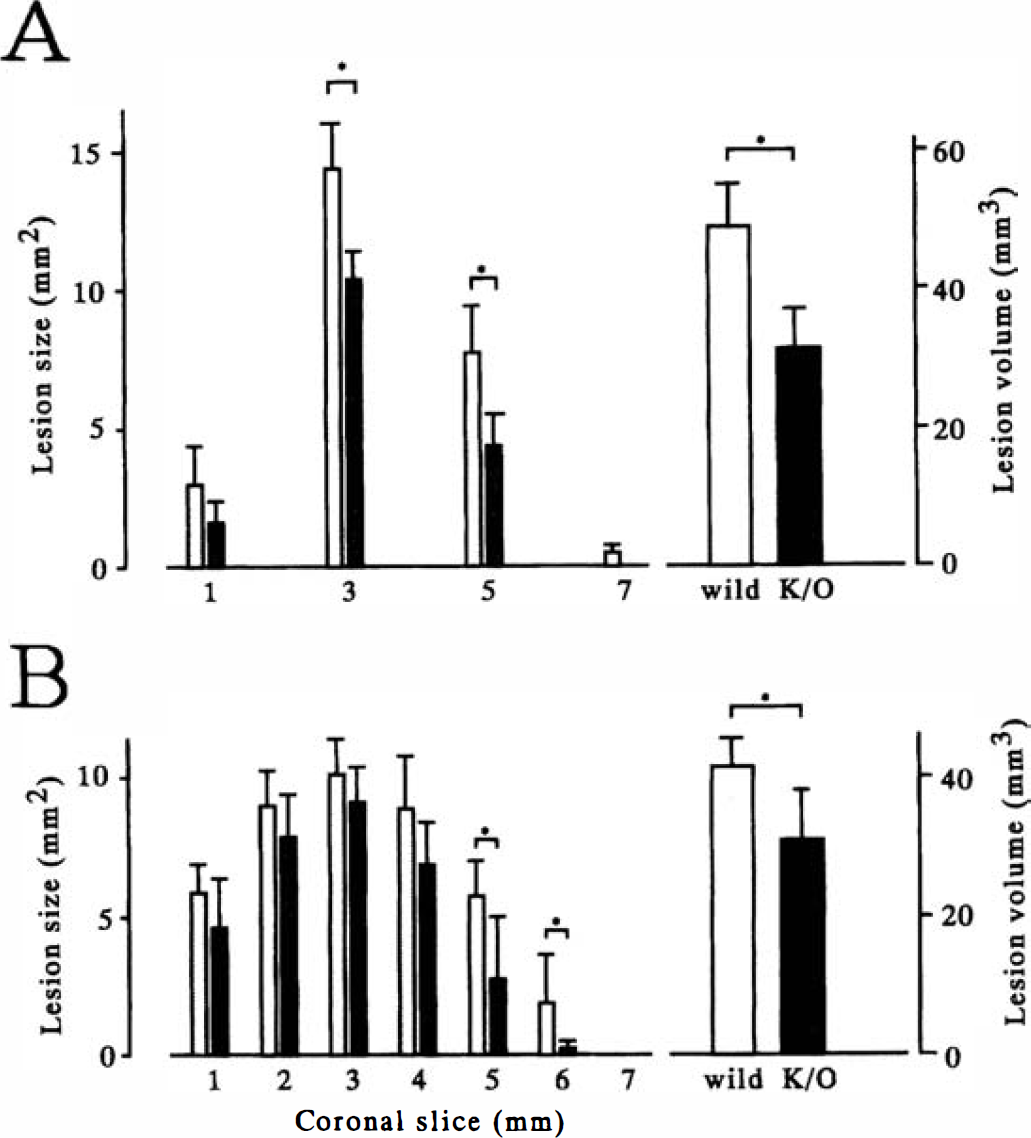
The area and the total volume of ischemic lesions after permanent middle cerebral artery (MCA) occlusion as detected with 2,3,5-triphenyltetrazolium chloride (TTC) staining
Transient focal ischemia. Ischemic lesions were evaluated only with the histologic method, including the immunoreaction for MAP2, because the areas showing neuronal death with viable astrocytes and endothelial cells were not detected with TTC staining. The cortical microperfusion in the ischemic core and the peripheral area was 15% to 20% and 30% to 45% of the baseline, respectively, during occlusion with a nylon suture, and recovery was 60% to 70% and 50% to 60%, respectively, 15 minutes after recirculation. The values were similar between knockout and wild-type mice (Table 2). Physiologic parameters were maintained similarly between the two types of mice (Table 2). After transient focal ischemia for 30 minutes and reperfusion for 48 hours, the cortical lesion in knockout mice was markedly reduced as compared with that in wild-type mice. The protection was evident widely in the cerebral cortex of the ischemic hemisphere (Fig. 3A), but ischemic neuronal damage in the caudoputamen and hippocampus was present in all mice and was not mitigated in knockout mice (Fig. 3B). After intraluminal suture occlusion for 45 minutes and reperfusion for 48 hours, significant reduction of cortical damage was still present in knockout mice, but four of six knockout mice developed infarction in the center of the MCA territory (Fig. 4A). In knockout mice, however, depletion of granulocytes by administration of the monoclonal antibody RB6-8C5 further reduced the size of ischemic lesion in the cerebral cortex even after ischemia for 45 minutes (Fig. 4B).
Ischemic lesions in the cerebral cortex
Ischemic lesions in the cerebral cortex after transient focal ischemia for 45 minutes and reperfusion for 48 hours
Accumulation of granulocytes in cerebral infarction
Granulocytes, identified as MPO-positive cells, were rarely observed in the brain sections of nonischemic control mice or in the contralateral hemisphere of ischemic mice. In the center of the MCA territory encompassing the cerebral cortex and caudoputamen where infarction evolved in both types of mice after permanent focal ischemia, MPO-positive cells accumulated not only in the wild-type mice but also in ICAM-1–knockout mice (Fig. 5). The number of granulocytes was almost the same between the two types of mice (Fig. 6). After transient focal ischemia for 30 minutes and reperfusion for 48 hours, MPO-positive cells were less frequent in the cerebral cortex of ICAM-1–knockout mice, where infarction was not evident, as compared with that of wild-type mice, in which infarction was evident. However, MPO-positive cells accumulated in the infarcted area of the cerebral cortex of ICAM-1–knockout mice when the ischemic period was extended to 45 minutes before reperfusion (Fig. 6). In the caudoputamen and hippocampus, where ischemic neuronal death occurred but the tissue integrity was fairly well preserved after ischemia for 45 minutes and reperfusion for 48 hours in both types of mice, there were many fewer MPO-positive cells in both types of mice as compared with those in the cerebral cortex (Fig. 6).
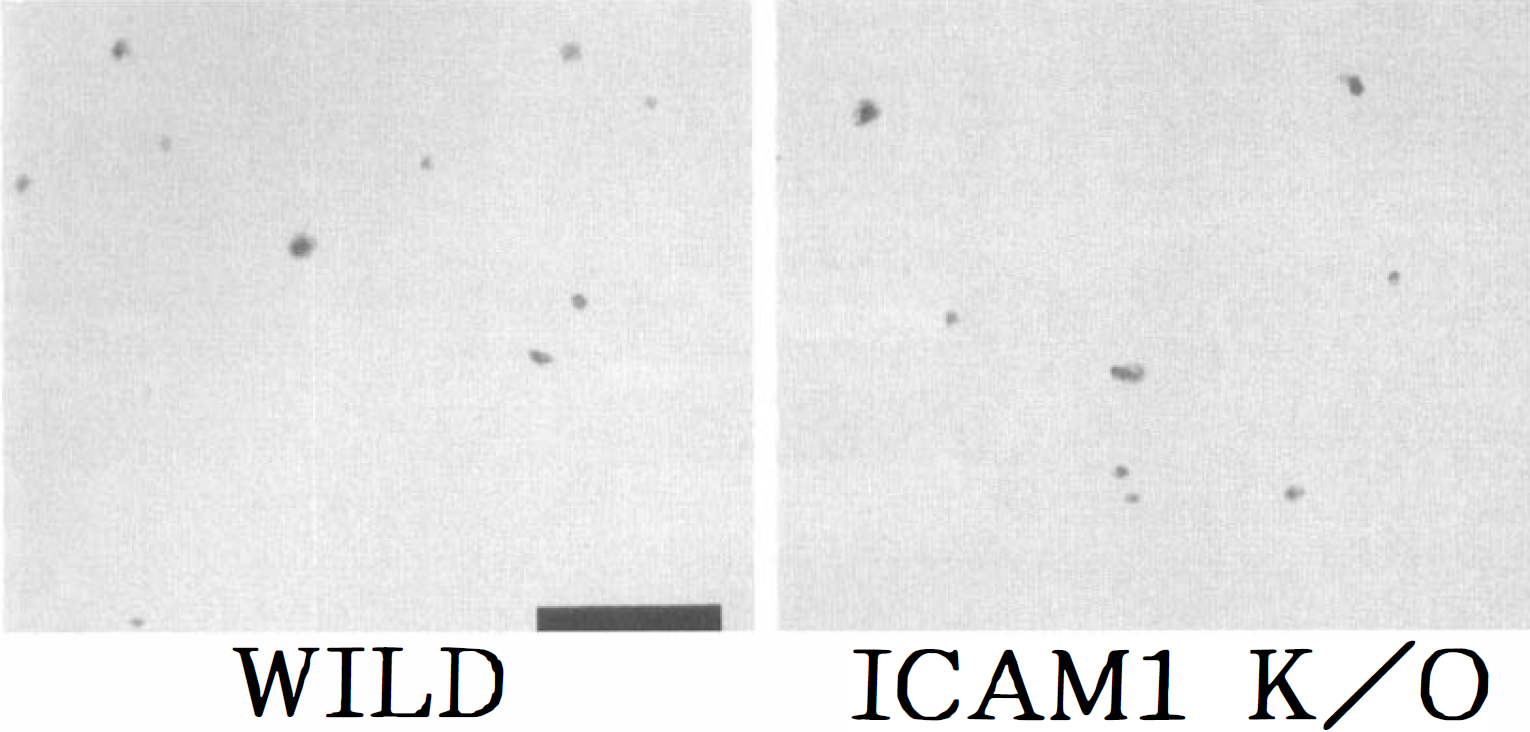
Cells positive for myeloperoxidase (MPO) in the infarcted area of the MCA territory in the cerebral cortex of wild-type and ICAM-1–knockout mice. Almost the same number of MPO-positive cells accumulated in the infarcted area of both types of mice. Bar = 40 µm.
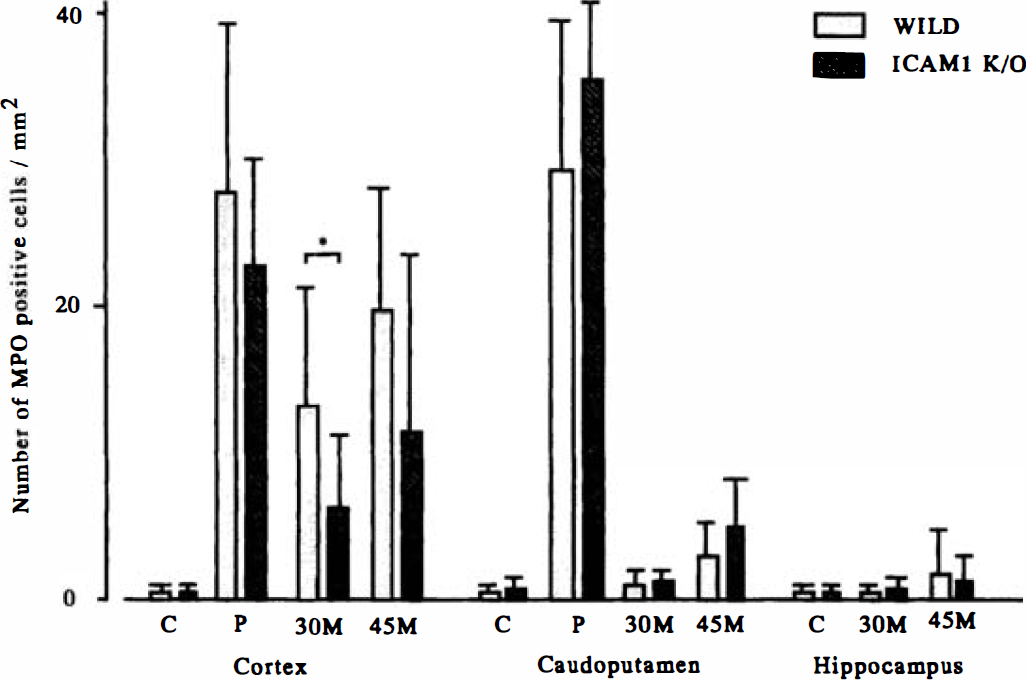
The number of MPO-positive cells in the cerebral cortex, caudoputamen, and hippocampus after permanent and transient focal cerebral ischemia. Almost the same number of MPO-positive cells accumulated in the cerebral cortex and caudoputamen after permanent focal ischemia in both types of mice. Myeloperoxidase-positive cells accumulated less in the cerebral cortex of ICAM-1–knockout mice after 30 minutes of transient focal ischemia; however, no significant difference was observed after 45 minutes of transient focal ischemia between the two types of mice. Accumulation of MPO-positive cells was much less in the caudoputamen and hippocampus of both types as compared with that in the cerebral cortex after transient focal ischemia. C, normal control; P, permanent focal cerebral ischemia; 30 M and 45 M, transient focal cerebral ischemia for 30 and 45 minutes with reperfusion for 48 hours, respectively.
Microcirculation in focal cerebral ischemia
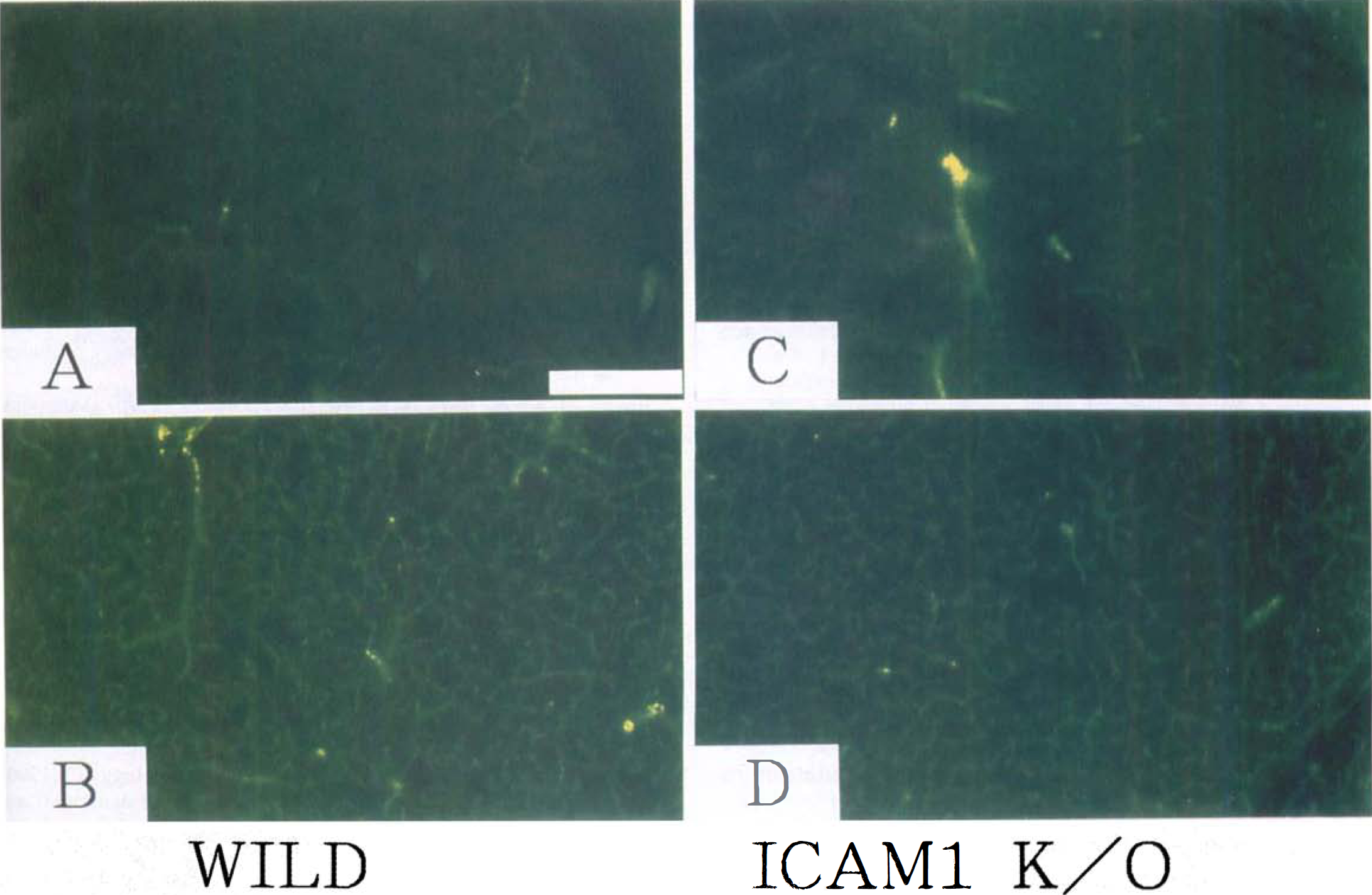
Fluorescence was observed within the vascular network in the entire brain of nonischemic wild-type and ICAM-1–knockout mice and in the contralateral hemisphere of ischemic mice (Fig. 7A and D). After permanent MCA occlusion for 6 hours, capillary fluorescence was hardly observed in the center of the MCA territory cortex or caudoputamen in both wild-type and knockout mice (Fig. 8A and C). In the peripheral area of the MCA territory, reduction of microcirculation was observed in both types of mice; however, capillary filling was better preserved in knockout mice than in wild-type mice. (Fig. 8B and E). After nylon suture occlusion for 30 minutes and reperfusion for 6 hours, the foci of nonfilling capillaries were frequently observed in the cerebral cortex of wild-type mice (Fig. 8C), but such reduction in microcirculation was not observed in the cerebral cortex of ICAM-1–knockout mice (Fig. 8F). In the caudoputamen, where ischemic neuronal death occurred later, reduction in microcirculation was not observed in either type of mice (Fig. 8B and D). Semiquantitative evaluation of the total length of the perfused capillary is summarized in Fig. 9.

Capillary perfusion in the cerebral cortex after focal cerebral ischemia.
Capillary perfusion in the caudoputamen after focal cerebral ischemia.
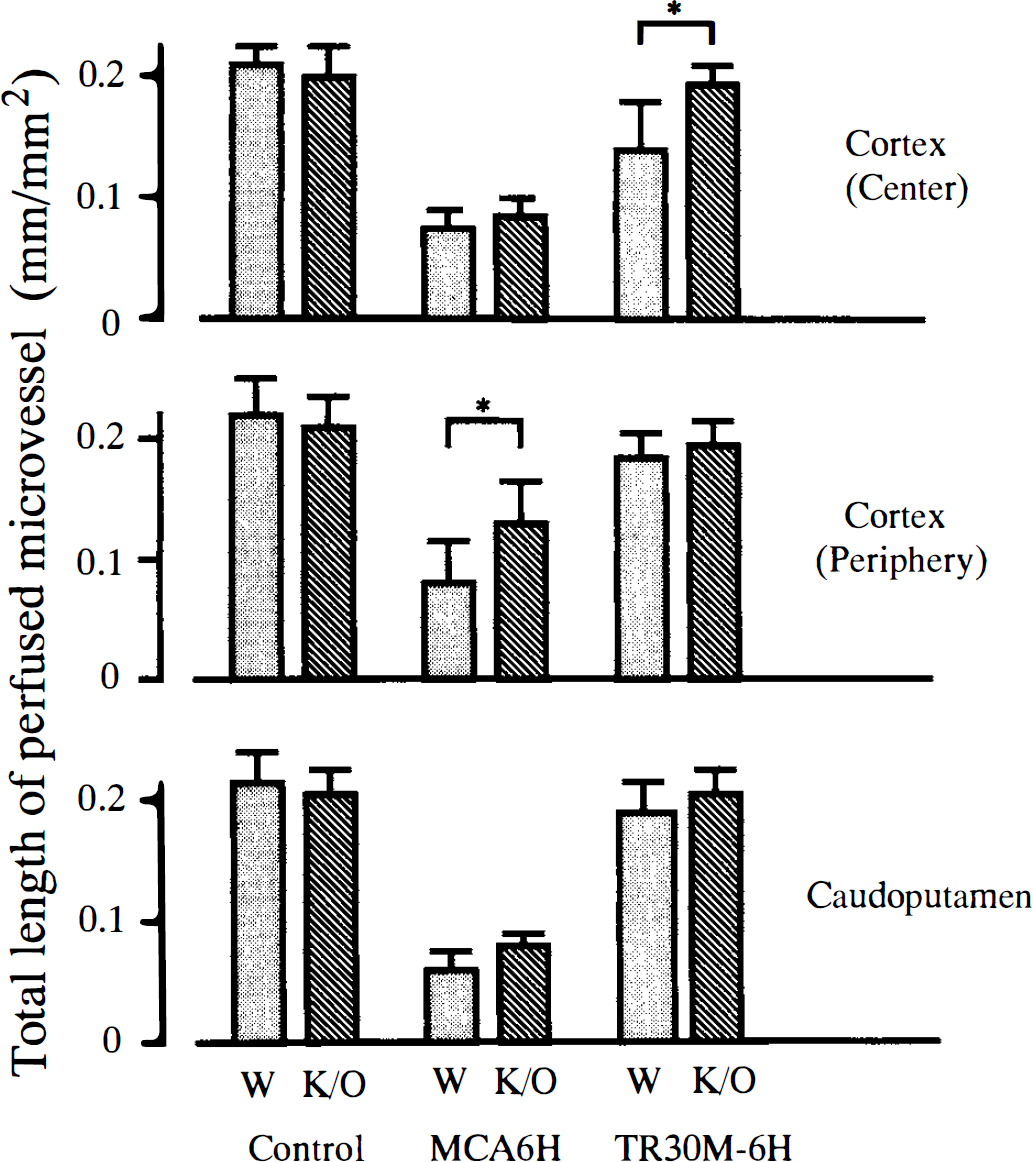
Semiquantitative evaluation of the cerebral microcirculation after focal cerebral ischemia in wild-type (stippled column) and ICAM-1–knockout mice (hatched column). Two areas of the cerebral cortex, the center (upper panel) and the peripheral area (middle panel) of the MCA territory, and the dorsolateral part of the caudoputamen (lower panel) were examined in the control, 6 hours after permanent MCA occlusion (MCA6H) and 6 hours after 30 minutes of transient focal ischemia (TR30M-6H). The degree of microperfusion was expressed as the total length (mm) of perfused microvessels per area (mm2). After permanent MCA occlusion, significant preservation of perfused microvessels was observed in the peripheral area of the MCA territory of ICAM-1–knockout mice as compared with the microperfusion in wild-type mice. In the ischemic core including the center of the MCA territory of the cerebral cortex and caudoputamen, however, a marked decrease of perfused microvessels was seen in both types of mice. After transient focal ischemia and reperfusion, the microcirculation was significantly more preserved in the center of the MCA territory of the cerebral cortex of ICAM-1–knockout mice as compared with that of wild-type mice.
DISCUSSION
Intercellular adhesion molecule 1 is believed to be involved in adhesion and accumulation of leukocytes in inflammation (Smith et al., 1988). However, the way leukocytes accumulate in inflammatory tissue seems to differ in different organs (Doerschuk et al., 1990). A study using ICAM-1–knockout mice has shown that ICAM-1 and P-selectin are strongly involved in leukocyte accumulation in peritonitis, but they are involved to a lesser extent in the pulmonary inflammation (Bullard et al., 1995). White blood cells, especially granulocytes have been shown to accumulate in cerebral infarction and are believed to aggravate ischemic brain injury by secreting cytotoxic factors or compromising the microcirculation (Kochanek and Hallenbeck, 1992). In addition to P-selectin and ICAM-1, hypoxic or ischemic insult has been shown to stimulate production of cytokine-induced neutrophil chemoattractant (Yamasaki et al., 1995), which could lead to accumulation of neutrophils in the infarcted tissue. Intercellular adhesion molecule 1 mediates leukocyte adhesion to the endothelial surface in the brain and has been considered to be an important factor for ischemic brain injury (del Zoppo, 1994). Recent studies using ICAM-1–knockout mice showed reduction of ischemic lesions after transient focal cerebral ischemia (Connolly et al., 1996; Soriano et al., 1996). There is no doubt that knockout mice are a valuable tool to determine the role of a single molecule in cerebral ischemia (Kinouchi et al., 1991; Huang et al., 1994). However, the strain difference in intracranial collateral circulation is an important factor in determining the severity and extent of ischemia, particularly with the nylon suture model (Kitagawa et al., 1998).
In the present study, we first demonstrated that the peripheral area of the MCA territory with the permanent MCA occlusion model could be salvaged by deficiency of ICAM-1. Although the protective effect in transient focal ischemia was consistent with the previous data using the anti-ICAM-1 antibody and ICAM-1–knockout mice, significant protection after permanent MCA occlusion is in contrast with the previous study showing no effect of the anti-ICAM-1 antibody after permanent focal ischemia (Zhang et al., 1995b). We believe that the deficiency of ICAM-1 in knockout mice was a more effective suppressor of ICAM-1 function than administration of the antibody.
We next showed that accumulation of granulocytes did occur in the infarcted tissue even in the absence of ICAM-1. Previous studies have shown reduction of granulocytes in ischemic brains of antibody-treated rats (Zhang et al., 1994) or ICAM-1–knockout mice (Connolly et al., 1996). However, it was uncertain in these reports whether the tissue for assessment of leukocyte accumulation had been infarcted or was intact. Furthermore, the present study clearly demonstrated that inhibition of ICAM-1 alone was not enough to suppress the aggravating role of granulocytes, because depletion of granulocytes in ICAM-1–knockout mice after transient focal ischemia further reduced the size of infarction. The protective effect of the peptide for lectin-binding domain (Morikawa et al., 1996) and laminin peptide (Yanaka et al., 1997) in focal cerebral ischemia also supports the presence of a pathway for leukocyte accumulation in ischemic tissue that is independent of ICAM-1.
Finally, we investigated the effect of ICAM-1 deficiency on microcirculation in focal ischemia. We chose permanent MCA occlusion for 6 hours or transient focal ischemia for 30 minutes with reperfusion for 6 hours, because infarction expanded peripherally at this period in permanent focal ischemia (Garcia et al., 1993) and because infarction was still not clear in transient focal ischemia after recirculation for 6 hours. The results indicated that the compromise in the microcirculation was minimized in ICAM-1–knockout mice before development of infarction, and supported the notion that this compromise played a major role in the development of infarction after transient focal ischemia (Hallenbeck et al., 1986; del Zoppo, 1994), and that ICAM-1 at least partially contributed to reduction of the microcirculation. Thus, inhibition of ICAM-1 function may have a potential therapeutic role in limiting the lesion size. In addition to improvement of the microcirculation and suppression of inflammation, however, other neuroprotective measures may be required to protect neurons themselves, especially in the vulnerable areas after successful reestablishment of circulation in stroke patients.
Footnotes
Acknowledgments
The authors thank Mr. Nobuo Katsube, Yoshifumi Takenobu, and Kigen Kondo of Ono Pharmaceutical Company for their technical assistance and careful management of animals, and Miss R. Manabe and Miss N. Hinokami for their secretarial assistance.

