Abstract
To elucidate the mechanism of ischemia-induced signal transduction in vivo, we investigated the effect of the targeted disruption of the α and Δ isoforms of the cAMP-responsive element-binding protein (CREB) on c-fos and heat-shock protein (hsp) 72 gene induction, Permanent focal ischemia was induced by occlusion of the middle cerebral artery of the CREB mutant mice (CREB(−/–), n = 5) and the wild-type mice (n = 6). Three hours after onset of ischemia, the neurologic score was assessed and pictorial measurements of ATP and cerebral protein synthesis (CPS) were carried out to differentiate between the ischemic core (where ATP is depleted), the ischemic penumbra (where ATP is preserved but CPS is inhibited), and the intact tissue (where both ATP and CPS are preserved). There were no significant differences in neurologic score or in ATP, pH, and CPS between the two groups, suggesting that the sensitivity of both strains to ischemia is the same. Targeted disruption of the CREB gene significantly attenuated c-fos gene induction in the periischemic ipsilateral hemisphere but had no effect on either c-fos or hsp72 mRNA expression in the penumbra. The observations demonstrate that CREB expression, despite its differential effect on c-fos, does not modulate acute focal ischemic injury.
According to current concepts, cerebral ischemia induces a multitude of cellular degenerative reactions, including the release of excitatory amino acids, formation of oxygen free radicals, Ca2+ overload, activation of multiple cellular enzyme systems such as Ca2+-dependent proteases, and initiation of genomic responses that can affect the tissue outside the area of reduced blood flow (for review see Hossmann, 1994; Kristian and Siesjö, 1996; Paschen, 1996; Small and Buchan, 1996). Activation of proteases can cause destruction of cellular structural proteins (Matesic and Lin, 1994), and pathologic accumulation of cytosolic Ca2+ induced either by influx from the extracellular space or by release from intracellular stores activates, among others, the cAMP-responsive element-binding protein (CREB) by means of a Ca2+/calmodulin-dependent protein kinase (Kiessling and Gass, 1994).
cAMP-responsive element-binding protein is an important transcription factor in the control of many genes that are induced by an elevation in cytoplasmic cAMP or Ca2+ concentration. When CREB is phosphorylated on Ser-133 by either cAMP- or Ca2+-dependent protein kinases (Dash et al., 1991; Sheng et al., 1991), it promotes transcription from the cAMP-responsive element (CRE) (Chrivia et al., 1993; Hunter and Karin, 1992). cAMP-responsive elements lie upstream of a number of genes, including the immediate-early genes c-fos (Sassone Corsi et al., 1988) and zif/268 (Sakamoto et al., 1991), as well as genes encoding the K+ channel (Mori et al., 1993) and the a subunit of Ca2+/calmodulin-dependent kinase II (Olson et al., 1995). Although binding activity has not been demonstrated, CRE also lie upstream of heat-shock protein (hsp) 70 (Williams et al., 1989). It is well known that cerebral ischemia induces the immediate-early gene, c-fos (An et al., 1993; Kiessling and Gass, 1994; Kinouchi et al., 1994; Neumann et al., 1994), and hsp70 (Kinouchi et al., 1993; Massa et al., 1996; Nowak and Jacewicz, 1994). Both genes are thought to have protective effects against cerebral ischemia, c-fos because it induces biosynthesis of neurotrophic factors by increasing AP-1 binding activity (An et al., 1993; Colangelo et al., 1996) and hsp70 because it is one of the molecular chaperons that prevent the aggregation of denatured proteins and promote the refolding of damaged polypeptides (Becker and Craig, 1994; Hartl et al., 1994; McMillan et al. 1994). For these reasons, CREB should play an important role in the concert of genomic responses induced by cerebral ischemia, although only a few reports have been published so far (Salminen et al., 1995; Yoneda et al., 1994).
The recent development of knockout/transgenic technology in mice has made it easy to manipulate the expression of genes and their particular translational products (Aguzzi et al., 1996; Chan, 1996; Ryffel, 1996). It is now possible to analyze the effect of targeted disruption or overexpression of genes in vivo. Previously, a mouse mutant with a targeted disruption of the α and Δ isoforms of CREB has been reported (Blendy et al., 1996; Hummler et al., 1994). In the present study we investigated the effect of this disruption on ischemia-induced c-fos and hsp72 mRNA induction. For evaluating gene induction, we investigated 3 hours of permanent ischemia because both genes are strongly expressed at this time (Welsh et al., 1992). Moreover, to elucidate the pathophysiologic significance of c-fos and hsp72 mRNA induction by ischemia, we correlated the genomic responses with the severity of ischemic injury as assessed by neurologic scoring and pictorial evaluations of tissue pH, ATP, and cerebral protein synthesis (CPS).
MATERIALS AND METHODS
Homozygous CREB mutant mice (CREB(−/–) group, n = 5; 3 males and 2 females) and their wild-type littermates (CREB(+/+) group, n = 6; 4 males and 2 females), weighing 24 to 32 g, were used (Blendy et al., 1996). Mice were F1 hybrids from two strains that represented backcrosses of the mutation into the C57black/6J (F5) and the FVBN (F5) background (both strains were obtained from Charles River, Germany).
Animals were housed under diurnal lighting conditions and allowed access to food and water ad libitum until the day of the experiment. Anesthesia was induced by 1.5% halothane and maintained with 1% halothane in 70% N2O and 30% O2.
Animal preparation
Focal cerebral ischemia was induced by occlusion of the middle cerebral artery (MCA) using the intraluminal filament technique (Hara et al., 1996) as modified by Hata et al (Hata et al., 1998a). After a midline neck incision, the left common and external carotid arteries were isolated and ligated. A microvascular clip (FE691; Aesclap, Tuttlingen, Germany) was temporarily placed on the internal carotid artery. An 8-0 nylon monofilament (Ethilon; Ethicon, Norderstedt, Germany) coated with silicon resin (Xantopren; Bayer Dental, Osaka, Japan) was introduced through a small incision into the common carotid artery and advanced 9 mm distal to the carotid bifurcation for occlusion of MCA.
Two hours and 15 minutes after MCA occlusion,
Experiments were terminated by in situ freezing (Mies et al., 1991). Brains were removed in a cold temperature cabinet (−20°C) and cut into 20-µm-thick coronal cryostat sections at -20°C. Sections were mounted on coverslips for ATP-bioluminescence, on object holders for CPS autoradiography, and on 3-aminopropyl triethoxysilane—treated slides for in situ hybridization (see below)
Regional measurement of ATP, tissue pH, and protein synthesis
Pictorial measurements of ATP and tissue pH were carried out using ATP-specific bioluminescence (Kogure and Alonso, 1978; Paschen et al., 1992) and umbelliferone fluorescence (Csiba et al., 1985), respectively. The pH images were calibrated with graded pH standards and ATP images were calibrated by taking small tissue samples from the ipsilateral ischemic and the contralateral nonischemic parietal cortex for quantitative enzymatic analysis.
For CPS autoradiography, cryostat sections were first exposed together with 3H standards to an X-ray film (Hyperfilm 3H, Amersham) for 14 days to assess total 3H radioactivity. Brain slices were then incubated in 10% trichloroacetic acid to remove labeled free leucine and metabolites other than proteins, and subsequently reexposed for the same duration to perform autoradiography of 3H-labeled proteins (Mies et al., 1991).
Probe for c-fos and hsp72 mRNAs
The sequence and specificities of oligonucleotide probes for c-fos and hsp72 have been described elsewhere (Kamii et al., 1994; Mikawa et al., 1995). The probe sequences of c-fos (45 mer) and hsp72 (30 mer) corresponded to amino acid residues 2 through 16 of rat c-fos and residues 122 through 129 of human hsp70, respectively. Each probe was 3' -end-labeled using terminal deoxynucleotidyl transferase (Gibco BRL, Eggenstein, Germany) and a 30:1 molar ratio of [35S]-dATP (1,200 Ci/mmol). Specific activity was greater than 0.5 × 109 dpm/µg.
In situ hybridization
In situ hybridization was performed as described previously (Wiessner et al., 1996). Briefly, coronal brain sections (20 µm) were fixed for 15 minutes in 4% paraformaldehyde/phosphate-buffered saline, pH 7.4. Sections were then treated with 0.25% acetic anhydride/triethanolamine for 10 minutes. After dehydration, 10 µL hybridization buffer containing 35S-labeled oligonucleotide probe (10 pg/µL), 2× standard—sodium citrate, 50% formamide, 10% dextran sulfate, 100 µg/mL poly(A), 120 µg/mL heparin, 1 mg/mL herring sperm DNA, 5 mmol/L dithiothreitol, and 1 mg/mL bovine serum albumin were added to the sections, and they were covered with a coverslip. After overnight hybridization at 42°C, the sections were washed twice at 42°C in 2× saline—sodium citrate/50% formamide for 30 minutes. Hybridized radioactivity was visualized by film autoradiography (Hyperfilm β-max, Amersham) with an exposure time of 1 week.
Morphometric analysis of ischemia-induced metabolic disturbance and gene expression
Bioluminescence and autoradiographic images were digitized with a charge-coupled device camera system connected to an image analyzer (NIH Image Software 1.58, National Institutes of Health, Bethesda, MD, U.S.A.). The volumes of ATP depletion, tissue acidosis, and CPS inhibition were estimated using the semiautomated method described by Swanson et al. (1990) with modification. Briefly, ATP depletion was defined as the decline to less than 1.0 µmol/g, and tissue acidosis as the fall of pH to less than 6.4. The threshold for CPS inhibition was set to the lowest CPS value of the nonischemic hemisphere excluding fiber tracts. The areas of ATP depletion and CPS inhibition were measured on each section by subtracting the area of the nonlesioned ipsilateral hemisphere from that of the contralateral hemisphere. The volumes of ATP depletion, tissue acidosis, and CPS inhibition were calculated by integration of the lesion areas at five levels of forebrain. The areas of preserved ATP and protein synthesis were outlined and superimposed to demarcate penumbral tissue in which protein synthesis was suppressed but ATP was preserved. At the level of the caudate-putamen, the signal intensities for c-fos and hsp72 mRNA were measured on film autoradiograms in cortical regions of interest, located in the core, the metabolic penumbra, and normal brain tissue, as defined by ATP and CPS imaging.
Optical densities of in situ hybridization signals of c-fos and hsp72 mRNA were measured at the level of caudate-putamen in cortical regions of interest, located in the core, the metabolic penumbra, and in normal brain tissue, as defined by ATP and CPS imaging. Optical densities were calibrated using Kodak Step Tablet No. 810ST606 standards according to the instructions of the NIH Image program. The autoradiographic procedures were standardized by using the same film (Hyperfilm β-max), exposure time, and film development for all experiments. Furthermore, sections of wild-type and CREB knockouts were exposed on the same film, and measurements from penumbra, periinfarct, and normal cortex were taken from the same tissue section.
Temperature control
Rectal temperature was maintained between 36.5° and 37.0°C by a heating lamp and a heating pad connected to a thermistor (YSI, Yellow Spring, OH, U.S.A.).
Statistics
All values are given as means ± SD. Differences in neurologic scores and areas of cortical c-fos and hsp72 mRNA expression were evaluated using the Mann-Whitney U-test. Differences in metabolic parameters and optical densities of mRNA were compared using two-way analysis of variance followed by Bonferroni's multiple comparison test. A P value of less than 0.05 was considered to indicate statistical significance.
RESULTS
Neurologic score
Focal ischemia resulted in marked neurologic disturbances 3 hours after MCA occlusion. All animals had developed at least grade 1 deficits, but there was no difference between the two groups (CREB(+/+) group, 2.33 ± 0.47 [mean ± SD]; CREB(−/–) group, 2.20 ± 0.98).
Regional brain tissue ATP, pH, and CPS
Representative images of the tissue content of ATP, tissue pH, and CPS in the wild-type mouse are shown in Fig. 1. ATP was depleted in the frontoparietal cortex, the lateral part of caudate-putamen, and the piriform cortex. Acidosis and suppression of CPS were present in the same areas but the changes extended distinctly more into the frontal, medial, and baso-occipital parts of the MCA territory. Quantitative measurements revealed significant reductions of ATP in the piriform cortex and the somatosensory area of the frontoparietal cortex at the level of striatum, in the temporal and piriform cortex at the level of the dorsal hippocampus, and in the lateral caudate-putamen. Changes of CPS and tissue pH extended further into the globus pallidus at the level of the striatum, into the parietal cortex at the level of the dorsal hippocampus, and into the temporal and entorhinal cortex at the level of the substantia nigra (Table 1). These changes were the same as in the CREB mutant mouse (Fig. 2 and Table 2).
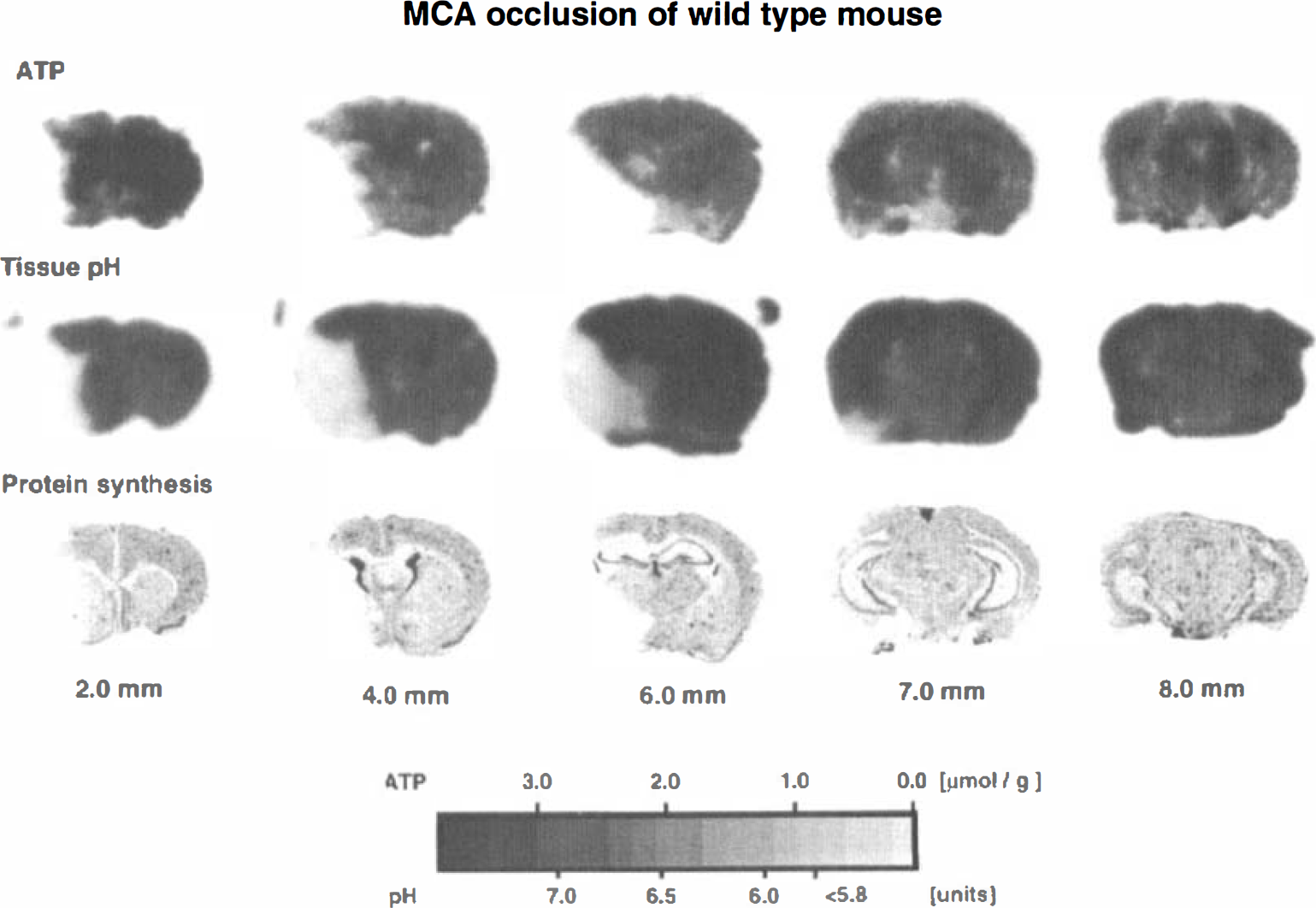
Representative ATP images, pH images, and 3H-leucine autoradiograms of brain sections of the wild-type mouse subjected to focal ischemia by middle cerebral artery occlusion for 3 hours. Numbers under each column indicate the distance in millimeters caudal to the frontal pole.
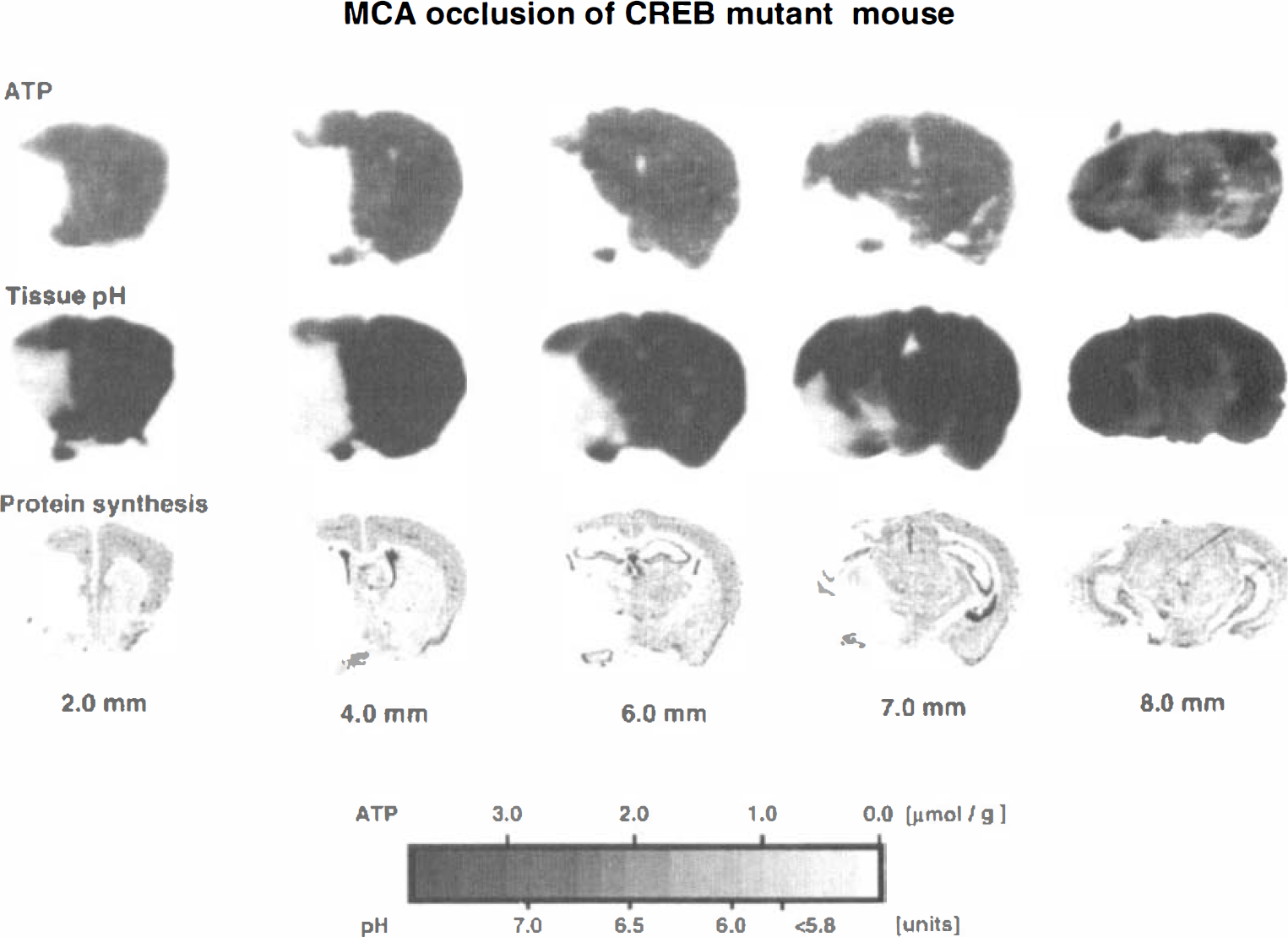
Representative ATP images, pH images, and 3H-leucine autoradiograms of brain sections of the CREB mutant mouse subjected to focal ischemia by middle cerebral artery occlusion for 3 hours. Numbers under each column indicate the distance in millimeters caudal to the frontal pole.
Changes of ATP and cerebral protein synthesis of the wild-type and CREB mutant mouse brain after 3 hours of middle cerebral artery occlusion
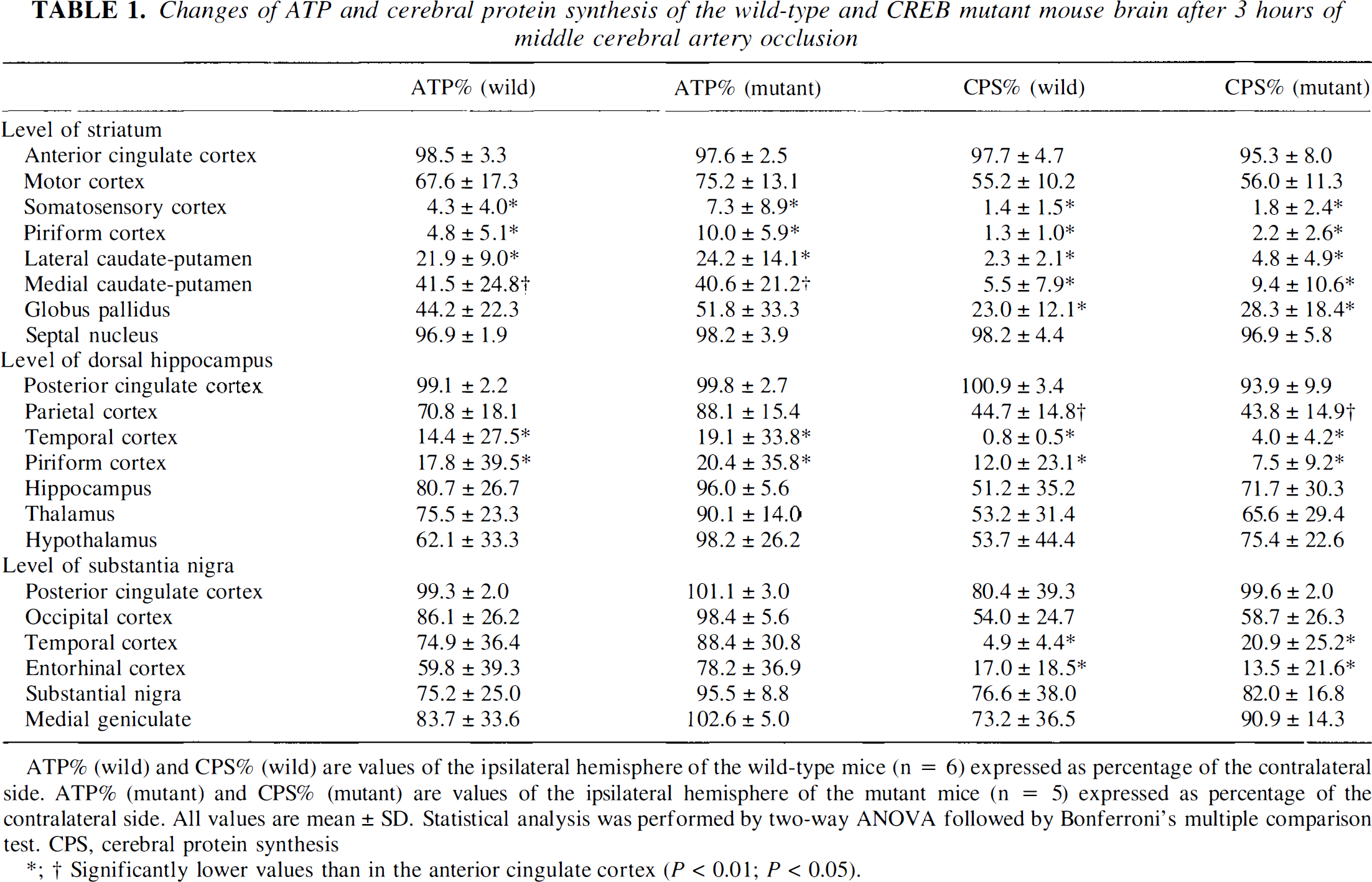
ATP% (wild) and CPS% (wild) are values of the ipsilateral hemisphere of the wild-type mice (n = 6) expressed as percentage of the contralateral side. ATP% (mutant) and CPS% (mutant) are values of the ipsilateral hemisphere of the mutant mice (n = 5) expressed as percentage of the contralateral side. All values are mean ± SD. Statistical analysis was performed by two-way ANOVA followed by Bonferroni's multiple comparison test. CPS, cerebral protein synthesis
Significantly lower values than in the anterior cingulate cortex (P < 0.01; P < 0.05).
Changes of tissue pH of the wild-type and CREB mutant mouse brain after 3 hours of middle cerebral artery occlusion
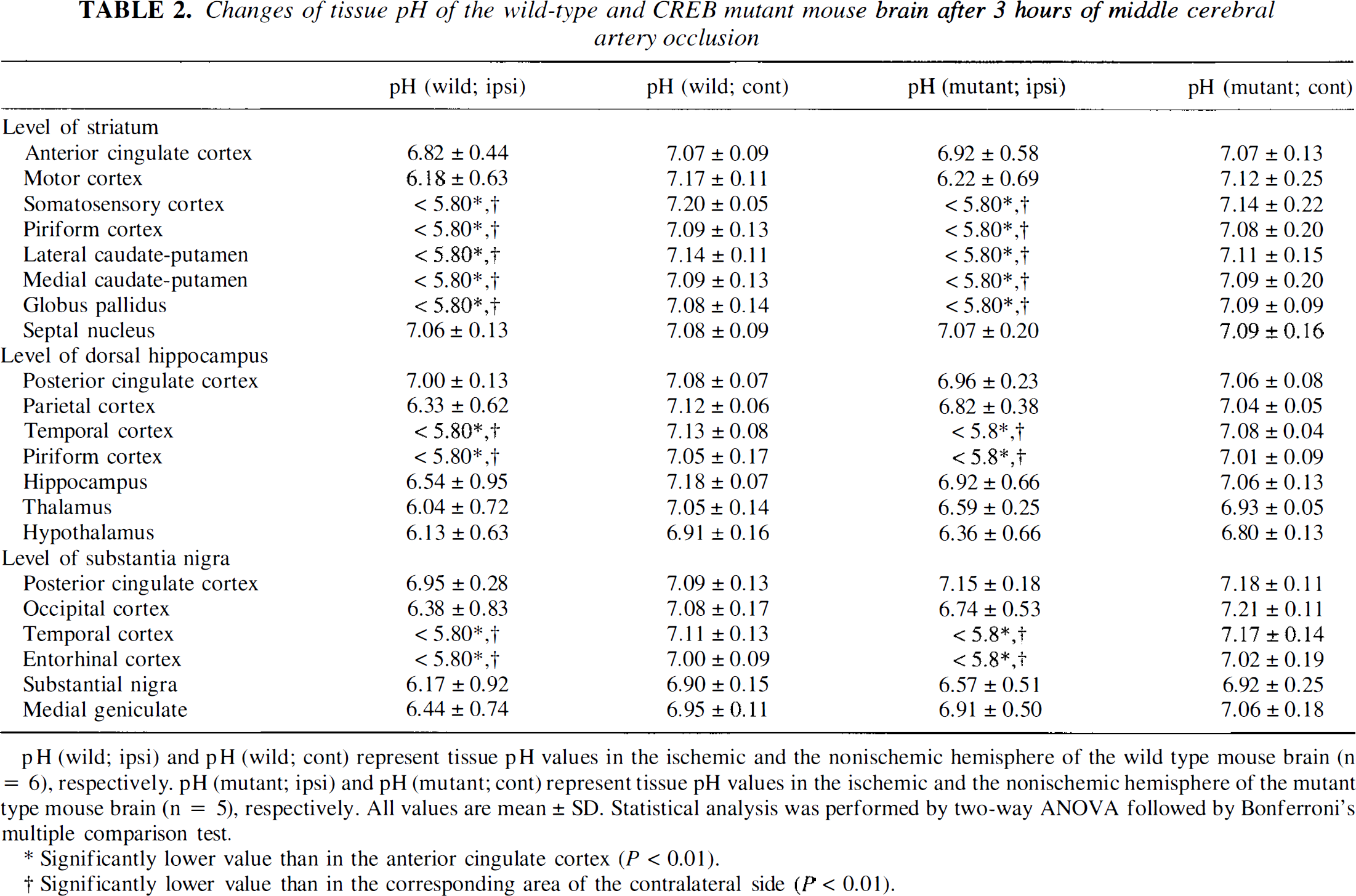
pH (wild; ipsi) and pH (wild; cont) represent tissue pH values in the ischemic and the nonischemic hemisphere of the wild type mouse brain (n = 6), respectively. pH (mutant; ipsi) and pH (mutant; cont) represent tissue pH values in the ischemic and the nonischemic hemisphere of the mutant type mouse brain (n = 5), respectively. All values are mean ± SD. Statistical analysis was performed by two-way ANOVA followed by Bonferroni's multiple comparison test.
Significantly lower value than in the anterior cingulate cortex (P < 0.01).
Significantly lower value than in the corresponding area of the contralateral side {P < 0.01).
The volume of metabolic disturbances, measured 3 hours after MCA occlusion, differed markedly depending on the chosen parameter in both CREB(−/–) and CREB(+/+) groups. In the CREB(+/+) group, ATP was depleted in 34.5% ± 11.7% of the contralateral hemisphere, tissue acidosis was detected in 43.4% ± 12.6%, and CPS inhibition in 52.7% ± 14.0%. In the CREB(−/–) mice the corresponding values were 27.3% ± 6.0% for ATP depletion, 36.3% ± 7.0% for tissue acidosis, and 48.2% ± 7.0% for inhibition of CPS. There were no significant differences in these three parameters between the two groups (Figs. 3, 4).
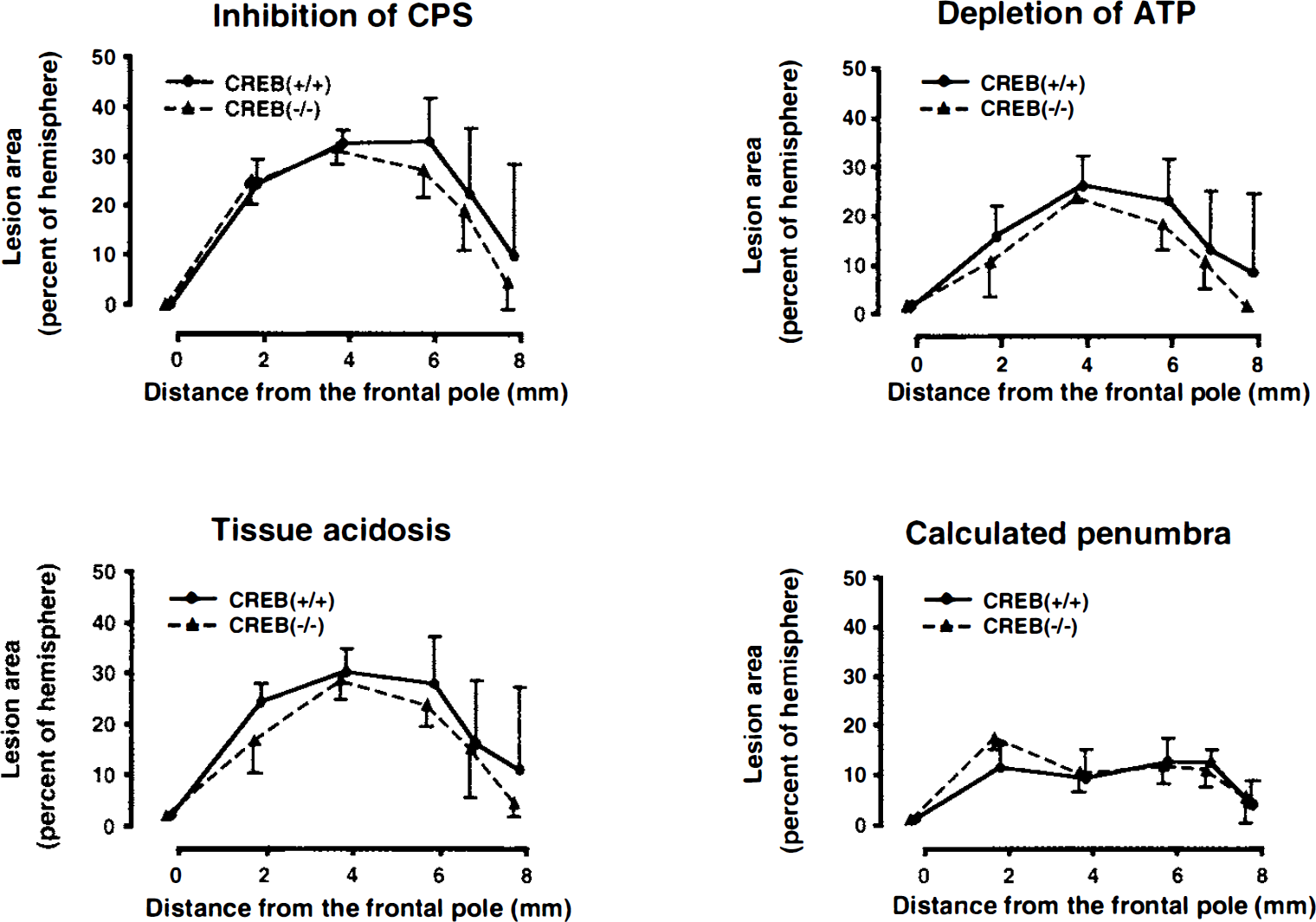
Areas of disturbed cerebral protein synthesis (CPS), acidosis, and ATP depletion in coronal brain sections of mice subjected to focal ischemia for 3 hours. The lesion areas (means ± SD) are plotted against the distance from the frontal pole and expressed as percentage of ipsilateral hemisphere. The area of penumbra was calculated by subtracting the percentage of ATP depletion from that of CPS inhibition. No significant differences were observed between the CREB mutant (CREB(−/–), n = 5) and the wild-type mice (CREB(+/+), n = 6).
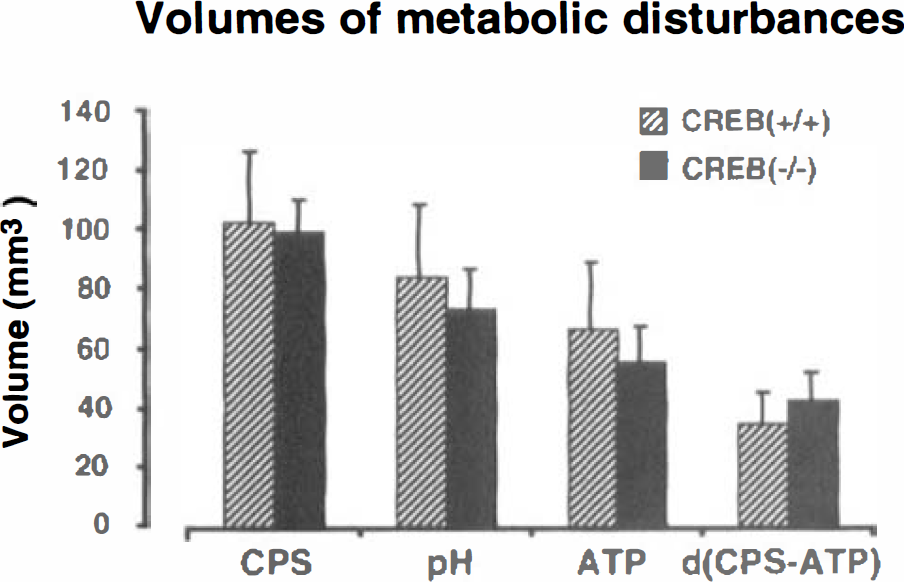
Lesion volumes of metabolic disturbances (inhibition of cerebral protein synthesis (CPS), acidosis, and ATP depletion) in mice subjected to focal ischemia for 3 hours. The volume of penumbra was calculated by subtracting the volume of ATP depletion from the volume of CPS inhibition. Values are means ± SD of 5 CREB mutant and 6 wild-type animals. Note absence of significant differences between the two groups.
c-fos and hsp72 mRNA expression
Three hours after MCA occlusion, the topical distribution of c-fos and hsp72 mRNA expression differed greatly. Expression of c-fos mRNA was prominent not only in the metabolic penumbra but also in more distant parts of the ipsilateral hemisphere of both CREB(−/–) and CREB(+/+) groups. Superimposition of the outlines of ATP- and CPS-preserved regions on the hybridization autoradiograms revealed that c-fos mRNA expression was present both in the penumbra and the normal nonischemic cortex, particularly in the nonischemic cingulate cortex of the ipsilateral hemisphere (Figs. 5, 6). There was no significant difference in the areas of the cortical c-fos mRNA expression (CREB(+/+), 4.11 ± 0.27 mm2; CREB(−/–), 4.24 ± 0.17 mm2) between the two groups. In the metabolic penumbra, there was also no difference in the mean optical densities of the c-fos mRNA hybridization signal between the two groups. However, in the nonischemic periinfarct cortex, the mean optical densities of the c-fos mRNA of the CREB(−/–) group was significantly (P < 0.05) reduced by 33% as compared with the CREB(+/+) group (Fig. 7).
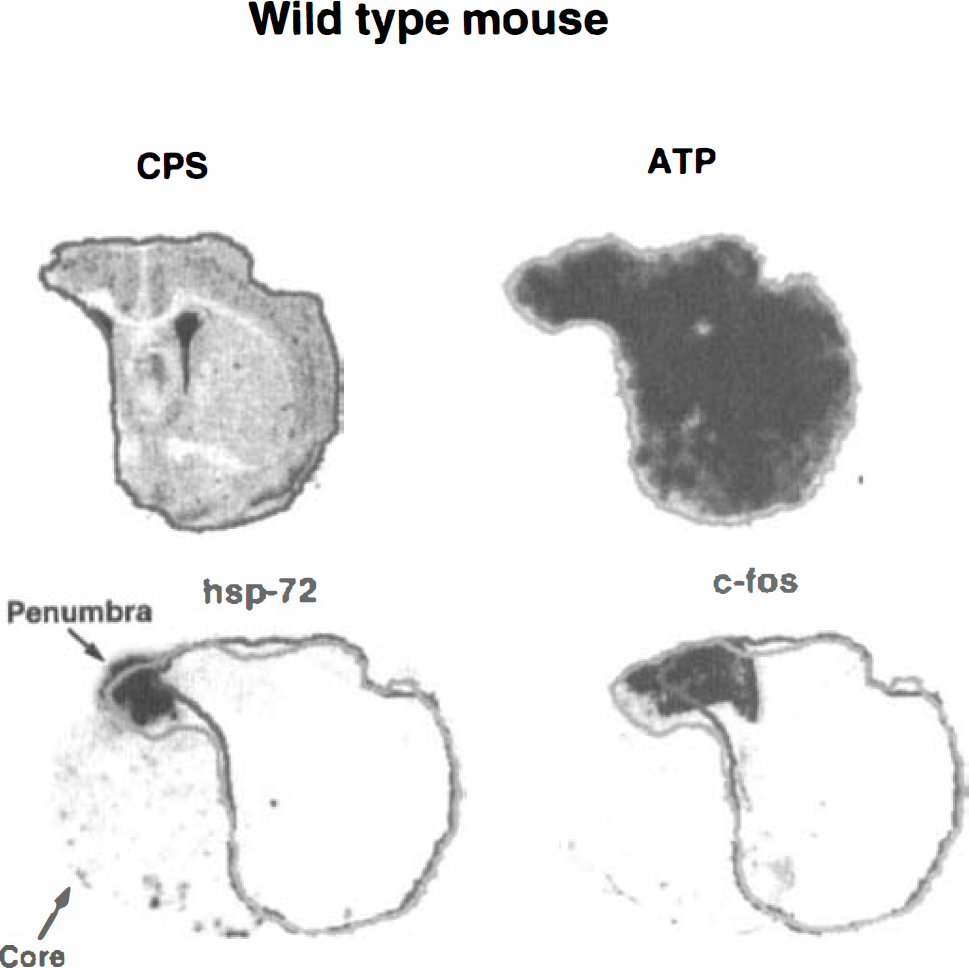
Representative images of cerebral protein synthesis (CPS), ATP, hsp72 mRNA, and c-fos mRNA of the wild-type mouse at the level of the caudate-putamen. The outlines of intact CPS (black) and preserved ATP (gray) were superimposed on the hsp72 and c-fos mRNA images for differentiation between core and penumbra (see text for definitions). Note selective expressions of hsp72 in the penumbra as compared with the more widespread expression of c-fos.
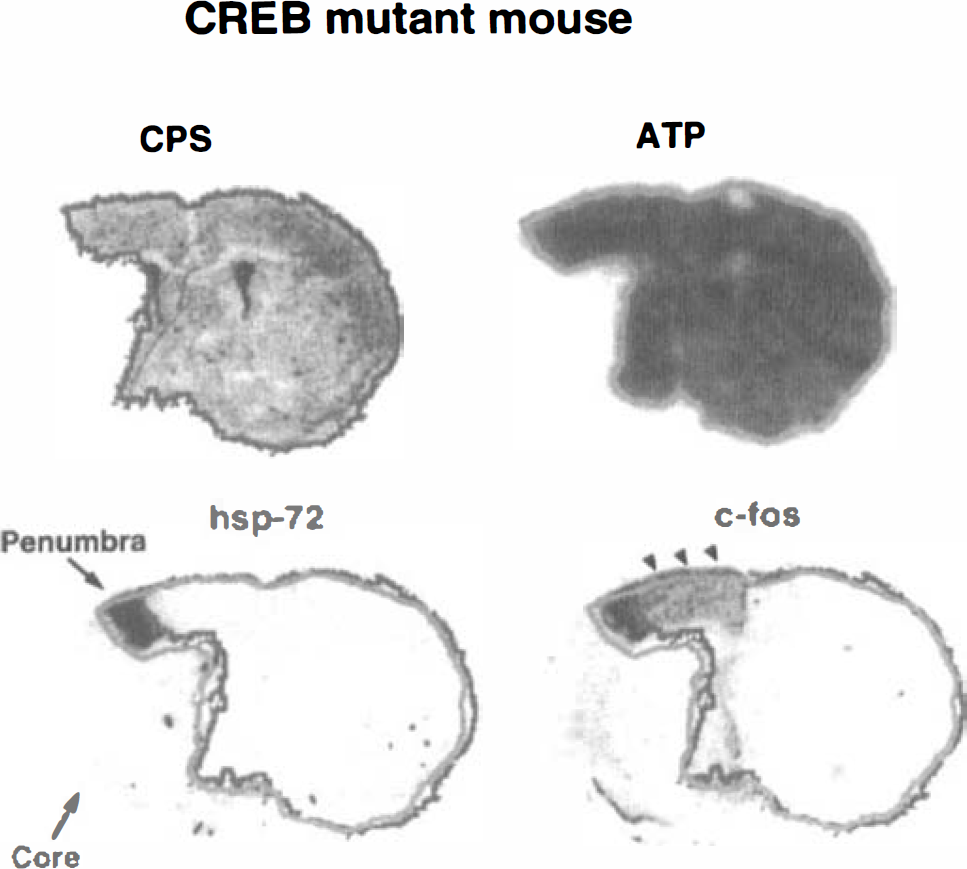
Representative images of cerebral protein synthesis (CPS), ATP, hsp72 mRNA, and c-fos mRNA of the CREB mutant mouse at the level of the caudate-putamen. The outlines of intact CPS (black) and normal ATP (gray) were superimposed on the hsp72 and c-fos mRNA images for differentiation between core and penumbra (see text for definitions). Note similar genomic expression pattern as in the wild-type mice, except for the less intense c-fos expression in the periischemic cortex (arrowheads).
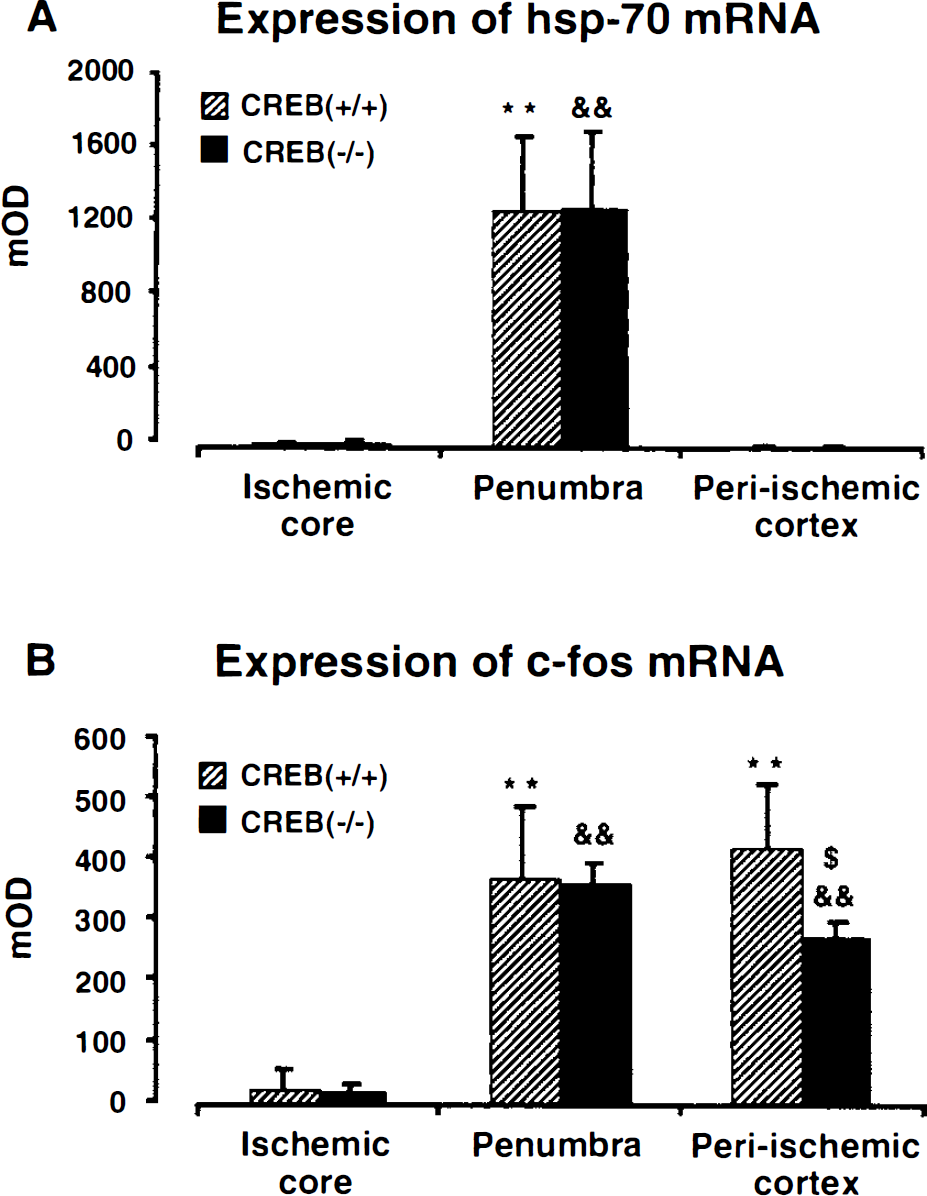
Semiquantitative measurement of hsp72 mRNA (A) and c-fos mRNA (B) in mice brain after 3 hours of middle cerebral artery occlusion. Mean optical densities (mOD) of in situ hybridization autoradiograms were measured in the ischemic core (temporoparietal cortex with suppression of both protein synthesis and ATP), penumbral cortex (parietal cortex with suppressed protein synthesis and preserved ATP), and intact cortex (ipsilateral cingulate cortex with preserved protein synthesis and ATP). Values are means ± SD of 5 CREB mutant and 6 wild-type animals. **, & indicate significant difference (P < 0.01) between core and penumbra; $ indicates significant difference (P < 0.05) between the CREB mutant and the wild-type mouse. Note differential expression of hsp72 and c-fos in penumbra and periinfarct cortex, and significant reduction of c-fos expression in the periischemic cortex of the CREB mutant mouse.
In contrast to c-fos mRNA, hsp72 mRNA was only expressed in the periinfarct rim of both CREB(−/–) and CREB(+/+) groups. Superimposition of the outlines of ATP- and CPS-preserved regions revealed that hsp72 mRNA expression was clearly restricted to the metabolic penumbra (Figs. 5, 6). Furthermore, there was no significant difference in the areas of the cortical hsp72 mRNA expression (CREB(+/+), 2.06 ±0.18 mm2; CREB(−/–), 2.14 ± 0.72 mm2) or the mean optical densities of the penumbral hsp72 mRNA hybridization signal between the two groups (Fig. 7).
DISCUSSION
In the present investigation permanent MCA occlusion in the CREB mutant and the wild-type mice led to similar tissue acidosis and disturbance of cerebral energy metabolism and protein synthesis, indicating that the sensitivity of both strains to ischemia is the same. However, in the periinfarct cortex of the CREB mutant mice, expression of the immediate-early gene c-fos was markedly attenuated as compared with the wild-type mice. This leads us to conclude that despite the different c-fos mRNA induction in the periinfarct area between the two strains, the expression of CREB does not modulate the susceptibility of brain tissue to acute ischemia.
Mechanism of c-fos induction
The major DNA elements responsible for c-fos induction are the sis-inducible element, the serum-response element (SRE), and the CRE (reviewed in Edwards, 1994; Karin et al., 1997). The sis-inducible element is recognized by the transcription factor, STAT (signal transducer and activator of transcription) (Darnell et al., 1994). These transcription factors are activated by Janus kinases through phosphorylation of tyrosine residues. They are translocated to the nucleus, bind to the sis-inducible element, and stimulate transcription of c-fos. These Janus kinase—STAT pathways can be activated by certain growth factors, cytokines, and interferons (Darnell, 1996). The serum-response element is recognized by a dimer of the serum-response factor that recruits the monomeric ternary complex factors (Treisman, 1992). A member of the E26-transformation specific family of nuclear proto-oncoproteins, Elk-1 (Hipskind et al., 1991), is phosphorylated by ERK (extracellular signal-regulated kinases), and the Elk-1–serum-response factor-SRE ternary complex is necessary for full SRE function (Edwards, 1994). As ERK are rapidly activated by most mitogens, this pathway may account for mitogen-induced c-fos transcription (Karin et al., 1997).
The CRE is recognized by CREB, which is phosphorylated on Ser-133 by protein kinase A or Ca2+/ calmodulin-dependent protein kinases II and IV in response to intracellular increases of cAMP or Ca2+. The CREB is also phosphorylated by the Ras—Raf—ERK—pp90 pathway. The phosphorylated form of CREB promotes transcription from CRE, which in turn contributes to c-fos induction (Finkbeiner et al., 1997; Ginty, 1997; Hagiwara et al., 1996).
It is reasonable to assume that induction of c-fos after MCA occlusion is mediated at least in part by CREB because ischemia and the associated periinfarct depolarization induces a sharp rise of cytosolic calcium. This interpretation is supported by the present finding that in the CREB mutant mice, c-fos induction was markedly attenuated. However, c-fos was not completely suppressed, indicating that other factors must also be involved. These could be either other transcriptional factors of the CREB family, such as CREM and ATF-1, or other pathways such as the SRE-dependent c-fos induction. In fact, both CRE and SRE are able to confer Ca2+-dependent induction of c-fos in vitro (Bading et al., 1993; Misra et al., 1994). Another explanation could be that the CREB splice variant, CREB-β, is upregulated in the CREB mutant mice, although this isoform is only present at low level in wild-type mice (Blendy et al., 1996). However, activator (τ) and inhibitor (α and β) forms of CREM are also upregulated, and these upregulations are thought to result in an 80% to 90% decrease in CREB activity in these mutant mice (Blendy et al., 1996; Maldonado et al., 1996). It is, therefore, likely that the observed attenuation of c-fos induction is caused by a decrease in CREB activity.
The fact that knockout of CREB alone decreases c-fos mRNA is in line with earlier conclusions that in vivo stimulation of any given element on the c-fos promoter is not sufficient to produce the entire c-fos induction (Robertson et al., 1995; Bontempi and Sharp, 1997).
Mechanism of hsp70 induction
Although the human hsp70 gene promoter possesses CRE/ATF and SRE (Williams et al., 1989), the major regulatory factor for stress-induced activation of hsp70 transcription is the heat-shock response element and the cytoplasmic heat-shock factor-1 (Morimoto et al., 1996). Various kinds of stress such as heat shock result in the formation of heat-shock factor-1 trimer, which after phosphorylation migrates to the nucleus and binds to heat-shock response element, causing the transcription of hsp70 mRNA (Baler et al., 1993). Some studies suggest that calcium or cAMP are required for activation of hsp70 transcription and that the cAMP sensitivity is conferred by both CRE/ATF and heat-shock response element (Choi et al., 1991; Price and Calderwood, 1991). On the other hand, agents that increase cAMP failed to stimulate the expression of hsp70 gene in intact cells (Murakami et al., 1991; Pizurki and Polla, 1994). In our study the mRNA of hsp72, a member of the 70-kDa heat-shock protein family, was readily induced after MCA occlusion in both the CREB mutant and the wild-type mice, and their expression patterns were similar. This suggests that ischemia-induced expression of hsp72 mRNA in mice is not under the control of CREB.
Imaging ischemic penumbra
Focal cerebral ischemia induces a complex pattern of genomic responses that may interfere in different ways with cell survival, depending on the severity and duration of ischemic injury. The interpretation of such data depends crucially on the exact differentiation between core, penumbra (Astrup et al., 1981), and normal brain tissue because the same signal transduction pathway may result in different responses, depending on the metabolic status of the tissue. Such a differentiation can be obtained by the use of multiparametric imaging techniques (Mies, 1993). According to the threshold concept of ischemic brain injury, inhibition of CPS and tissue acidosis occur at higher flow values than the breakdown of the energy metabolism state (Hossmann, 1994). Imaging of these variables on adjacent cryostat sections allows the precise demarcation between the ischemic core in which CPS is suppressed but ATP is preserved, and the intact tissue in which brain metabolism is normal. This has been previously documented in the C57black/6J mice after MCA occlusion for the same duration as in the present study (Hata et al., 1998b). The comparison of these data with the present study revealed that there are no differences in the volumes of tissue affected by metabolic disturbances among the C57black/6J mice, the CREB mutant mice, and their wild-type littermates. This suggests that the genetic background of the three strains does not modulate the sensitivity of brain tissue to acute ischemia despite the recently reported differential susceptibility to excitotoxicity (Choi, 1997; Schauwecker and Steward, 1997).
Penumbra and c-fos induction
In accordance with our previous study in the C57black/6J mice, c-fos mRNA was expressed both in the penumbra and in the more distant periinfarct cortex of the ipsilateral hemisphere. It has been documented that mRNA synthesis of the c-fos gene does not require protein synthesis (Herrera and Robertson, 1996). This explains why c-fos mRNA was expressed in the penumbra although global protein synthesis was severely suppressed. The additional expression in the more distant parts of the periinfarct surrounding has been attributed to the transient increase in cytosolic calcium evoked by the passage of periinfarct spreading depressions (for review see Gass and Herdegen, 1995; Herrera and Robertson, 1996; Hossmann, 1996).
Interestingly, penumbral c-fos mRNA expression was similar in the wild-type and the CREB mutant mice, but in the more distant areas, in particular the ipsilateral cingulate cortex, c-fos mRNA induction was significantly attenuated in the mutant mice. This suggests that in focal ischemia c-fos mRNA is induced by at least two different mechanisms, depending on the region involved.
This interpretation is supported by pharmacologic studies. Treatment with the noncompetitive N-methyl-
The attenuated induction of c-fos mRNA in the distant area of the CREB mutant mice suggests that in this area CREB may contribute to c-fos induction in vivo, although the SRE (not the CRE) is an important regulatory element in the c-fos induction through calcium influx via NMDA receptor-operated ion channels in vitro (Bading et al., 1993). In the periinfarct penumbra, in contrast, c-fos is expressed in the absence of CREB, which suggests that in this part of the evolving infarct SRE may play the dominant role for c-fos induction after calcium influx through NMDA receptor-operated channels, voltage-sensitive calcium channels, or both (Santella and Carafoli, 1997).
An important deduction of this observation is the independence of infarct progression from CREB expression. In the CREB mutant and the wild-type mice, the derangements of energy metabolism and protein synthesis as well as tissue acidosis were quite similar. Our study, therefore, suggests that the expression of CREB is not an important factor in the concerted molecular interactions involved in the evolution of brain infarcts during acute ischemia.
Penumbra and hsp70 induction
In both the CREB mutant and the wild-type mice, hsp72 mRNA expression strictly matched a narrow rim in the periphery of the ischemic territory in which postischemic protein synthesis was inhibited but ATP was preserved. hsp72, an inducible member of the 70-kDa heat-shock protein family, acts as a molecular chaperon that prevents the aggregation of denatured proteins and promotes the refolding of damaged polypeptides (Becker and Craig, 1994; Hartl et al., 1994). Although the precise mechanism of hsp72 gene induction after ischemia remains unknown, an obvious activator of hsp72 transcription after ischemia is the presence of denatured proteins. There are also indications from in vivo studies that inhibition of protein synthesis promotes transcription (Nowak and Jacewicz, 1994; Planas et al., 1997). Induction of hsp72 gene does not respond to periinfarct spreading depression, and a treatment of infarcts with NMDA antagonists does not suppress its induction in the penumbra (Yamashita et al., 1996).
It has been repeatedly reported that heat-stress proteins are neuroprotective. In fact, hsp72 mRNA and its protein product have been demonstrated in neurons that survive transient global (Kanemitsu et al., 1994) or permanent focal ischemia (Kinouchi et al., 1993). However, the protective effect of the stress protein is abolished whenever other metabolic disturbances supervene. As an example, the metabolic workload induced by periinfarct spreading depression results in irreversible energy failure in the periinfarct border zone despite expression of stress proteins (Back et al., 1994). Imaging of stress proteins may, therefore, be useful to evaluate progression or regression of the ischemic injury. If, for instance, hsp72 mRNA expression is detected in a region in which ATP is depleted, this constellation would indicate that ischemic injury has progressed into the penumbra region. Conversely, the expression of hsp72 mRNA in a region with preserved CPS would indicate reversal of injury. In the present study, hsp72 expression was clearly restricted to the penumbra in both the CREB mutant and wild-type mice subjected to focal cerebral ischemia for 3 hours. This is in line with the observation that the severity of ischemia was similar in both groups and suggests that CREB does not modulate the progression of injury.
CONCLUSION
The comparison of c-fos and hsp72 mRNA expression after 3 hours of MCA occlusion in the CREB mutant and the wild-type mice revealed a significant reduction in c-fos mRNA in the distant periinfarct surrounding of the CREB mutant strain. However, hsp72 mRNA expression, and the associated disturbances of energy metabolism, tissue pH, and protein synthesis did not differ between the two strains. We therefore conclude that the expression of CREB and the periinfarct induction of c-fos mRNA are not major pathogenic factors for the evolution of brain infarcts during acute ischemia.
Footnotes
Acknowledgments
The authors thank Mrs. U. Beckmann, Mrs. M. Nelles, and S. Krause for excellent technical assistance, and Mrs. D. Schewetzky for the careful preparation of the manuscript.
