Abstract
This study sought to establish the photothrombotic ‘ring’ stroke model with late spontaneous reperfusion in adult mice. By applying a 3.0-mm diameter laser ring-beam (514 nm, 0.21 mm thick, 0.65 W/cm2) onto the exposed skull for 60 secs with concurrent erythrosin B (4.25 mg/kg) intravenous infusion for 15 secs, the centrally located cortical region within the ring locus was progressively encroached by an annular ring-shaped perfusion deficit. In this region, local cerebral blood flow (ICBF) measured by 1aser-Doppler flowmetry declined promptly after irradiation to 43% of the baseline value at 30 mins poststroke. Using double tracer autoradiography, quantitative ICBF measured with [14C]iodoantipyrine was 46 to 17 to 58 ml/100 g/mins at 4 h to 48 h to 7 days postischemia in this area. Cerebral protein synthesis (CPS), as detected by [3H]leucine incorporation into protein, transiently decreased to 57% to 38% to 112% at 4 h to 48 h to 7 days postischemia in the center region. Morphologically, some neurons in the center region appeared swollen at 4 h. At 48 h, the majority of neurons were severely swollen with eosinophilia and pyknosis, whereas at 7 days poststroke’ the tissue morphology became partly restored. The center within the mouse photothrombotic ring lesion thus exhibits reversible alterations of local CBF, CPS and tissue morphology that are reminiscent of the cortical penumbra in other models of focal cerebral ischemia.
Keywords
Introduction
The evolution and final outcome of stroke depends largely on the severity and the duration of the ischemic impact. After focal vascular occlusion, the ischemic region can be differentiated into two parts: a central core of densely ischemic tissue in which ischemic injury is irreversible, and a peripheral penumbra, which is functionally impaired but still viable (Astrup et al, 1981; Symon et al, 1977). The ischemic penumbra is, therefore, the main target for therapeutic interventions.
In experimental stroke research, mutant mice are increasingly used to dissect the complex molecular interactions, which contribute to the progression of penumbral injury (Hossmann, 2004). To induce experimental stroke, mechanical occlusion of the middle cerebral artery (MCA) is the most commonly used method. However, in mice of different strains, genetic variations in the size of the vascular territory supplied by the MCA result in differences in infarct volume (Maeda et al, 1998), which may mask the effect of targeted gene mutation (Gerlai, 1996).
An alternative way to induce focal cerebral ischemia is by means of cortical photothrombosis (Watson, 1998; Watson et al, 1985). A modification of this approach is the photothrombotic ‘ring’ stroke model, which has been established in adult rats. In this model a ring-shaped ischemic lesion is formed, surrounding a penumbra-like area of hypoperfusion (Wester et al, 1995). The ischemic lesion inevitably transforms into pan-necrosis (Wester et al, 1995), but with proper adjustment of the intensity of laser beam irradiation, the penumbra-like ‘region-at-risk’ can undergo progressive hypoperfusion and transient critical tissue damage, followed by spontaneous reperfusion and tissue repair (Gu et al, 1999a, b; Hilger et al, 2004). In this stroke model, apoptosis and necrosis (Hu et al, 2002, 2004), upregulation of immediate early genes (Johansson et al, 2000), early occurring cortical neurogenesis (Gu et al, 2000) and angiogenesis (Gu et al, 2001) have been observed, all of which reflect the suitability of this approach for the elucidation of novel pathophysiologic and molecular mechanisms. An additional advantage of the model is the high reproducibility of the size of the photothrombotic lesion, which is due to invariance of the density of the cortical microvasculature (Watson, 1998), thus rendering irrelevant the inevitable variations in arterial blood supply to this area. This may be of particular importance when studying conditions with variations in the genetic background of the arterial brain vasculature.
Compared with other models of focal ischemia, the photothrombotic ‘ring’ stroke model is, therefore, of particular interest for application in genetically modified animals. As transgenic and knockout mutants are mainly produced in mice, we sought to establish this model in the adult C57BL mouse.
Materials and methods
Induction of Photothrombotic ‘Ring’ Stroke
The animal care and experimental procedures were performed in accordance with the European Communities Council Directive (86/609/EEC), and the experimental protocol was approved by the Ethics Committee for Animal Research at the University of Umeå. Adult male C57BL mice (B&K Sollentuna, Sweden) weighing 25 to 35 g were used. The mice were anesthetized for 3 mins in a chamber ventilated with 3% halothane in a mixture of 30% oxygen (O2) and 70% nitrous oxide (N2O) and then maintained at 2% halothane using a face mask. Body temperature was controlled at 37°C by means of a rectal thermometer connected to a heating pad system (CMA 150, CMA/Microanalysis, Sweden). Polyethylene PE10 catheters were inserted into a femoral artery and vein for monitoring arterial blood gases, glucose, arterial blood pressure and intravenous erythrosin B infusion. The head temperature was kept at 37.0°C ± 0.5°C, with a needle thermometer probe in the temporal muscle connected to a heating lamp system (Omega 6000, Stamford, CN, USA). The physiologic data were measured by sampling 100 μl of arterial blood from the right femoral artery and then supplemented with 100 μl saline at 2 mins before and 25 mins after irradiation (Belayev et al, 1999). Mice were mounted on a stereotaxic frame (David Kopf, Tujunga, CA, USA) and anesthesia was continued with 1% halothane in a 30/70 mixture of O2 /N2O delivered by a face mask. The skull bone was exposed and an argon ion laser (Innova 70-4, Coherent, Palo Alto, CA, USA) tuned to the 514.5-nm transition was used to produce a photothrombotic ring-shaped cortical lesion with a centrally located cortical region-at-risk (Gu et al, 1999a; Wester et al, 1995). Briefly, the output of the laser beam was focused with a planoconvex glass lens (focal length = 30 mm) into a 400 μm optical fiber (ST400E-FVwith SL sleeves, Mitsubishi Cable America, Inc., New York, USA) at an input angle of 29° to the fiber axis, resulting in a ring beam output. The outside diameter of the ring beam was set at 3.0 mm and the laser beam thickness on the skull was 0.21 mm to fit the anatomy of the adult mouse, as compared with the photothrombotic ‘ring’ stroke model in adult rats where the outside diameter is 5.0 mm and the laser beam thickness is 0.35 mm (Gu et al, 1999a). The ring beam was placed symmetrically within the coronal, sagittal and lambdoid sutures and the parietal bone crest. The exposed skull bone was irradiated by the laser ring beam with concurrent intravenous infusion of the photosensitizing dye erythrosin B (Sigma, #330000) by an infusion pump (KDS 100, Stoelting, III, USA). The mice were kept on the stereotaxic frame for 30 mins after irradiation with the physiologic parameters controlled, and then moved to a chamber with circulating fresh air for wakeup.
Carbon Black Perfusion
At various times after stroke onset, carbon black was transcardially perfused and tissue morphology was investigated to identify the optimal experimental conditions for inducing a hypoperfused ‘region-at-risk’ in the center of the ring lesion. The mice were deeply anesthetized and thoracotomized. The descending aorta was clipped. A PE50 catheter was inserted into the left ventricle through the apex and the right atrium was incised. An infusion pump (Becton Dickinson) was connected through a pressure transducer (Ohmeda AB, Helsingborg, Sweden) to a blood pressure monitor (78353A Hewlett Packard) and a pulsation device (Medicinsk Teknik Umeå, Sweden). A measure of 10 ml 0.9% saline was infused at 5 ml/mins for washout of the blood, followed by the infusion of 2 ml filtered carbon black at 3 ml/mins (Design Higgins, no 4418, Faber-Castell, Lewisburg, TN, USA). The brain was taken from the skull and photographed under a microscope.
Laser-Doppler Measurement of Local Cerebral Blood Flow
Changes of 1CBF after stroke induction were measured semiquantitatively by laser-Doppler flowmetry (lCBFLDF) (Gu et al, 1999a). Animals underwent the same initial experimental procedure as described above. After the skull was exposed, an Ø = 1 mm burr hole was drilled into the center of the nonirradiated region. A very thin layer of skull bone was left intact. The hole was cleaned with 37.0°C saline so that the vessels on the brain surface could easily be identified under a Zeiss operating microscope. The LDF probe was held by a stereotaxic micromanipulator (David Kopf) and was lowered onto the bottom of the burr hole under the operating microscope. Probe placement on large branches of arteries or veins was avoided. The 1CBF was recorded for 30 mins to measure the baseline values. After registration of the probe coordinates, the LDF probe held by the stereotaxic micromanipulator was switched away and the skull was irradiated as described above. Immediately after the irradiation and erythrosin B infusion, the LDF probe was switched back to the same stereotaxic coordinates as during local CBF baseline recordings. The local CBF was continuously recorded for another 30 mins after irradiation. In the sham-operated group (n = 4), the mice were treated in the same way as in the ischemic group, but without erythrosin B administration. Local baseline CBF data are presented as average perfusion unit values during the 20 mins before irradiation and then reported at 3 mins intervals for another 30 mins after irradiation.
Double Tracer Autoradiography of Blood Flow (lCBFIAP) and Protein Synthesis (Cerebral Protein Synthesis)
Cerebral blood flow: Quantitative maps of lCBFIAP were obtained using the [14C]iodoantipyrine autoradiography method (Kennedy et al, 1972). This method requires the determination of tissue [14C]radioactivity at the end of the experiment (Reivich et al, 1969; Sakurada et al, 1978) where the [14C]-radioactivity in various regions of the brain can be measured three-dimensionally at high resolution by quantitative autoradiography (Hosokawa et al, 1977; Kennedy et al, 1972; Reivich et al, 1969; Sakurada et al, 1978). This method has been modified and validated for mice (Maeda et al, 2000). In brief, 15 μCi [14C]iodoantipyrine (specific activity: 5.5 mCi/mmol; concentration: 0.1 mCi/ml) was injected intraperitoneally at 2 mins before killing. The mice were frozen in situ in liquid nitrogen and the brains were removed at −20°C in a glove box (Termo Kyl, Helsinborg, Sweden). A blood sample from the heart was melted at ambient temperature on a filter paper and the [14C]-radioactivity was measured by a liquid scintillation system (Maeda et al, 2000). Cryostat coronal brain sections (20 μm thick) were collected throughout the ischemic lesion and mounted on object holders. To measure local CBF, [14C]iodoantipyrine labeled brain sections were exposed directly to a film (Hyperfilm-β max 18 × 24 cm2, high performance, Amersham, Sweden) with standard high-performance microscales (RPA511V Amersham, Sweden) for 7 days. The films were scanned with a black & white camera (Sony 388, Sweden) and a Northern Light illuminator (Imaging Inc., Canada). Autoradiograms were calibrated with the standard microscales and local blood flow was calculated with the NIH Image MG program, using the [14C]-radioactivity from the heart blood sample for the estimation of the tracer input function (Maeda et al, 2000).
Cerebral protein synthesis: Semiquantitative maps of cerebral protein synthesis (CPS) were prepared from [3H]leucine autoradiograms of the same tissue section used for CBF measurements (Paschen et al, 1985; Swanson et al, 1990). Briefly, 300 μCi [3H]leucine (specific activity: 171 Ci/mmol; concentration: 5.0 mCi/ml) was injected intraperitoneally 45 mins before the end of the experiments. The mice were frozen in situ and coronal slices were prepared as described above (Mies et al, 1991). After [14C]-autoradiography, labeled free leucine and metabolites other than proteins as well as [14C]iodoantipyrine were removed by 0.6 mol/L PCA (perchloric acid 70%) and 2 mol/L EGTA (ethylene Glycol-bis (β-aminoethyl ether)-NNN−-1N−-1-tetraacetic acid) (Mies et al, 1997; Paschen et al, 1985). The sections were then exposed to a [3H]-sensitive film (BioMax MS 18 × 24 cm2 Kodak, Sigma, Sweden) for 28 days with standard high-performance microscales (RPA 506, Amersham, Sweden) for autoradiographic analysis of [3H] proteins (Mies et al, 1991). The films were scanned with a black & white CCD camera (Sony 388, Sweden) and a Northern Light Illuminator (Imaging Inc., Canada). Data were calibrated with the microscales standards using the NIH Image MG program (Mies et al, 1997).
Tissue Morphology and Measurement of Lesion Volumes
Histopathology: Brains were fixed with FAM (formaldehyde, glacial acetic acid, methanol at the ratio 1:1:8) and embedded in paraffin. A measure of 10-μm-thick coronal brain sections were cut at 300 μm intervals throughout the ischemic lesion and stained with hematoxylin and eosin. Planimetry of the lesion was performed with an Olympus BX50 microscope equipped with a U-DA camera lucida attachment projecting the image onto a digitizing table (Summa Sketch III Series, Summagraphics Corporation, Connecticut, USA). The camera was connected to a PC with morphometric software (Morfometri-PC Version 2.0 J-O Dahlberg. Skellefteå, Sweden) as described previously (Gu et al, 1999b). The ischemic area was defined as a region in which most neurons exhibited ischemic morphologic changes (Garcia and Anderson, 1997; Kalimo et al, 1997) with at least one of the following criteria: cytoplasmatic microvacuolation, cytoplasmatic eosinophilia, cell body shrinkage, nuclear pyknosis, or hyperchromasia. The ischemic volume was calculated by integrating the ischemic areas of the sequential coronal sections and expressed as percentage of the ipsilateral hemispheric volume (Longa et al, 1989).
Immunohistochemistry: The ischemic region was investigated at 48 h after stroke by the neuronal marker Neu N and at 7 days by Neu N and the glial marker glial fibrillary acidic protein (GFAP). Paraffin-embedded coronal sections (10 μm thick) from the mid-part of the lesion were deparaffinized with xylene and hydrated through graded ethanol series. The slides were quenched with 3% H2O2 in methanol and blocked by 5% normal goat serum for 30 mins. The sections were incubated with the primary antibody mouse anti-Neu N, 1:750 overnight (Chemicon, Temicula, CA, USA) followed by incubation with the secondary antibody goat anti-mouse immunoglobulin G (IgG) 1:200 for 60 mins (Vector laboratories, Burlingame, CA, USA) and subsequent incubation with the avidin-biotin complex (ABC-peroxidase kit) for 60 mins (Vector). The horseradish peroxidase reaction was detected by incubation with the chromogen diaminobenzidine (DAB) according to the manufacturer's recommendations (Vector DAB substrate kit, Vector). The slides were counterstained by hematoxylin.
For double-labeling experiments with Neu N and GFAP at 7 days after stroke, Neu N was first detected by ABC-peroxidase (Vector) for 60 mins and stained with the Vector Nova Red substrate kit (Vector). These sections were incubated with the second primary antibody rabbit anti-GFAP, 1:1000 at room temperature overnight (Dako A/S, Denmark) followed by incubation with the secondary antibody goat anti-IgG, 1:200 for 60 mins (Vector) and subsequent incubation by the ABC-peroxidase kit (Vector) for 60 mins. These were visualized by the Vector SG substrate kit (Vector), according to the recommended protocol. As controls, sham-operated and naïve mice were used as well as omission of the primary or secondary antibody, which abolished the immunoreactivity.
Statistics
For group comparisons, analysis of variance (ANOVA) and post hoc effects were performed by use of the system for statistics (SYSTAT; Wilkinson, 1969, version 10) where a P-value of less than 0.05 was regarded as significant.
Results
A total of 11 mice were excluded from the formal experiment because of bleeding from the catheters (n = 3), damage to the brain tissue during dissection (n = 2), unsuccessful intraperitoneal 14C-labeled iodoantipyrine injection (n = 2), irregular labeling of the blood sample tubes (n = 3), and missing brain slices (n = 1).
Physiologic Data
No statistically significant changes of the physiologic parameters were observed after ischemia induction. The physiologic data (mean ± s.d.) were as follows: pO2 (mm Hg) 122 ± 21 before and 117 ± 15 after irradiation; pCO2 (mm Hg) 43.2 ± 5.3 before and 48.1 ± 3.9 after irradiation; pH 7.28 ± 0.04 before and 7.30 ± 0.08 after irradiation; blood pressure (mm Hg) 104 ± 9.0 before and 95 ± 7.4 after irradiation; head temperature (°C) 36.8 ± 0.7 before and 37.1 ± 0.5 after irradiation; rectal temperature (°C) 36.1 ± 0.4 before and 36.2 ± 0.2 after irradiation; plasma glucose 11.5 ± 1.7 mmol/L.
Establishment of the Photothrombotic Irradiation Protocol
To induce a progressive perfusion deficit followed by spontaneous reperfusion in the nonirradiated central cortical region, stepwise titrations of the laser beam intensity and the dose of erythrosin B were performed (n = 54): At first, the erythrosin B dose was kept constant at 17 mg/kg body weight whereas the laser intensity was set at 1.09, 0.86, 0.65, 0.54, 0.43 and 0.27 W/cm2. With this setup, the central cortical region was severely swollen after as long as 72 h poststroke and reperfusion was absent. Then the dose of erythrosin B was decreased to 8.5 mg/kg and further to 4.25 mg/kg. Finally, the laser irradiation duration was tested at 60 and 30 secs. The optimal experimental condition, that is, a gradually progressing hypoperfusion followed by late spontaneous reperfusion in the central nonirradiated region at risk, was achieved when erythrosin B was infused intravenously for 15 secs at a dose of 4.25 mg/kg, and the laser beam intensity was set at 0.65 W/cm2 (power 12 mW) for 60 secs.
Carbon Black Perfusion
With this experimental setting, carbon black perfusion at 4 h after stroke induction revealed a ring-shaped perfusion deficit (paleness) of the cortical surface corresponding to the ring-shaped irradiation beam (ring lesion region), whereas staining of the nonirradiated central cortical region (region-at-risk) was only slightly reduced (see Figure 1). At 10 and 24 h after irradiation, the centrally located cortical region was progressively encroached by the inward expansion of the annular ring lesion, reflecting the progressive deterioration of the region-at-risk. At 48 h postischemia, the whole region-at-risk appeared pale. However, at 72 h and up to 7 days postischemia, the vessels in the region-at-risk refilled with carbon black. In sham-operated mice, the cortical surface was homogeneously perfused (see Figure 1).
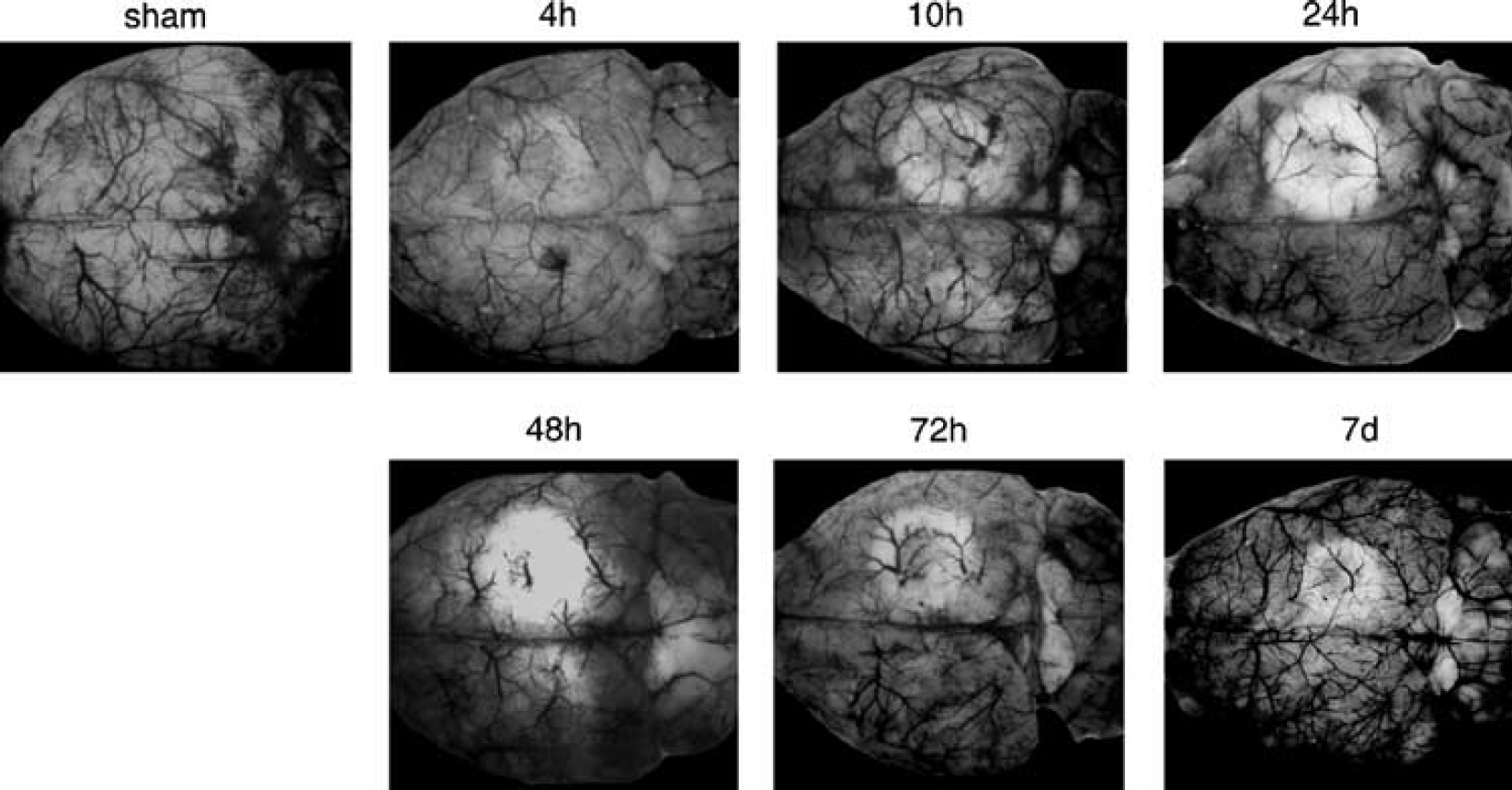
Representative cases of carbon-black perfusion in sham-operated mice and in animals at 4 h, 10 h, 24 h, 48 h, 72 h and 7 days after irradiation (n = 4 animals in each group). A 3.0-mm diameter laser ring-beam (514 nm, 0.21 mm thick, 0.65 W/cm2) was directed onto the exposed skull for 60 secs with concurrent erythrosin B (4.25 mg/kg) intravenous infusion for 15 secs.
Local Cerebral Blood Flow
At 30 mins after induction of photothrombosis lCBF, measured semiquantitatively by laser-Doppler flowmetry (lCBFLDF), declined in the center of the nonirradiated region to 43% ± 4% of the baseline value (n = 6) (Figure 2); in the sham-operated control group (n = 4) lCBFLDF did not change. Blood flow was also measured quantitatively with [14C]iodoantipyrine (lCBFIAP) (Figures 3 and 4). In the sham-operated control group (n = 3), lCBFIAP did not differ between the contralateral cortex (88 ± 46 ml/100 g/mins) and the ipsilateral cortex (90 ± 47 ml/100 g/mins). In the ischemic animals (n = 4 in each group), the lCBFIAP in the ring lesion and the nonirradiated region-at-risk decreased at 4 h postischemia to 23 ± 10 and 46 ± 11 ml/100 g/mins (mean ± s.d.), respectively. At 48 h, postischemia blood flow reached its minimum values of 5 ± 2 and 17 ± 3 ml/100 g/mins in the ring lesion and the nonirradiated central region but these values recovered at 7 days postischemia to 37 ± 19 and 57 ± 25 ml/100 g/mins, respectively.
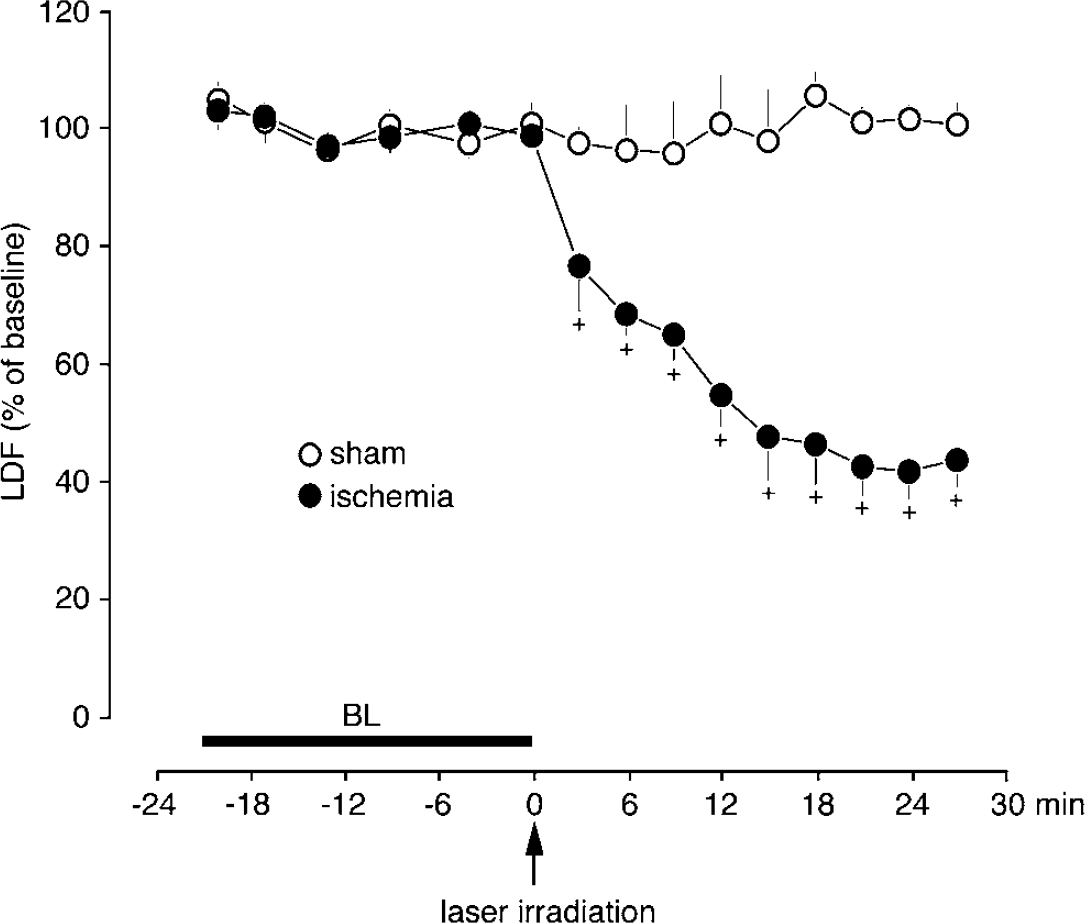
Local cerebral blood flow measured by laser-Doppler flowmetry (lCBFLDF) in the center of the nonirradiated cortical region (region at risk). Values are presented as mean ± s.d. + Statistically significant reduction compared with baseline (BL) values (P < 0.05 by ANOVA and post hoc tests, n = 6). Sham-operated animals (n = 4) showed no significant difference in lCBFLDF.
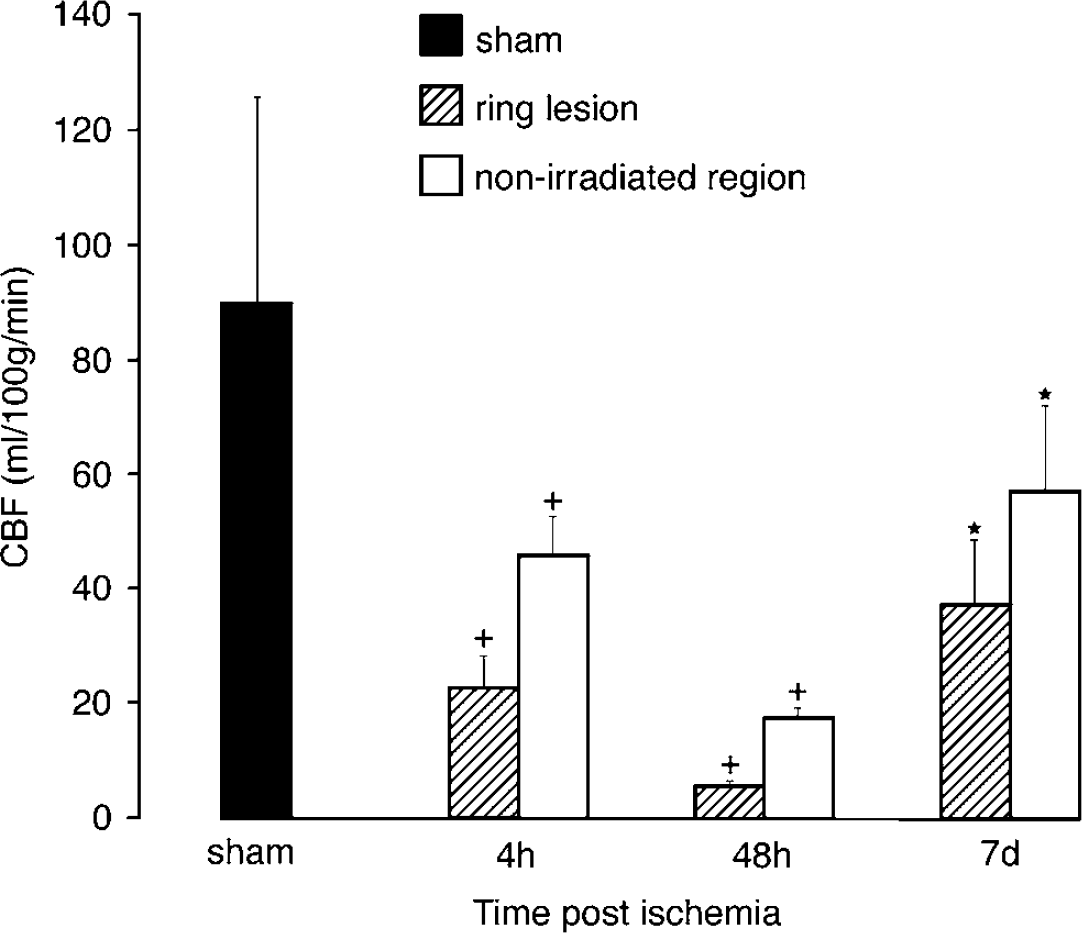
Local cerebral blood flow measured by [14C]iodoantipyrine (ICBFIAP). Values are presented as mean ± s.d. in ml/100 g/mins. Sham-operated controls (n = 3) and irradiated animals at 4 h, 48 h and 7 days postischemia (n = 4 in each group). + Statistically significant reduction compared with sham-operated animals (P < 0.05 by ANOVA and post hoc tests). *Statistically significant increase compared with values at 48 h after ischemia (P < 0.05 by ANOVA and post hoc tests).
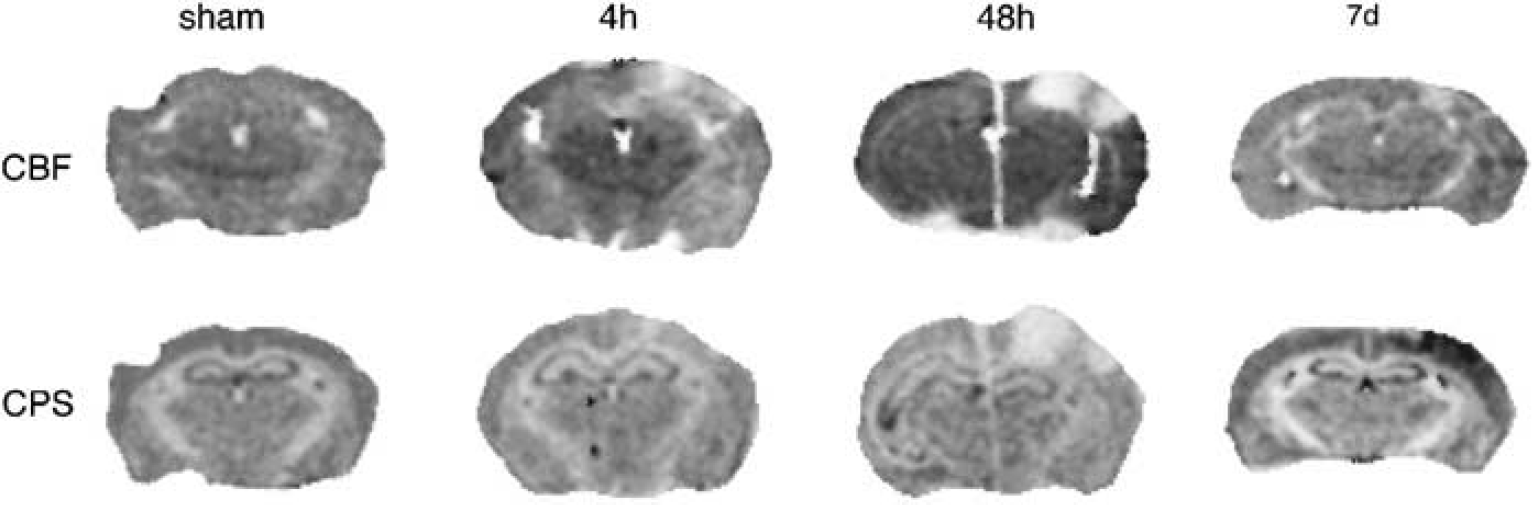
Representative images of cerebral blood flow (CBF) and protein synthesis (CPS) prepared by double tracer autoradiography of [14C]iodoantipyrine and [3H]leucine from the same coronal sections after photothrombotic ‘ring’ stroke induction. Sham-operated controls and irradiated animals at 4 h, 48 h and 7 days postischemia.
Cerebral Protein Synthesis
In sham-operated animals (n = 3) CPS, measured semiquantitatively by [3H]leucine incorporation into proteins, was not different between the two hemispheres. After photothrombosis (n = 4 in each group), CPS in the nonirradiated central region-at-risk decreased to 57% ± 23% (mean ± s.d. of contralateral side) at 4 h and further to 38% ± 14% at 48 h, followed by an overshoot to 115% ± 28% at 7 days postischemia (Figures 4 and 5). At 7 days postischemia, the protein synthesis was highest in the ischemic boundary zone surrounding the ring lesion region (Figure 5). In the ring lesion, CPS dropped to 18% ± 6% and 12% ± 5% at 4 and 48 h, and partly recovered to 51% ± 17% at 7 days postischemia.
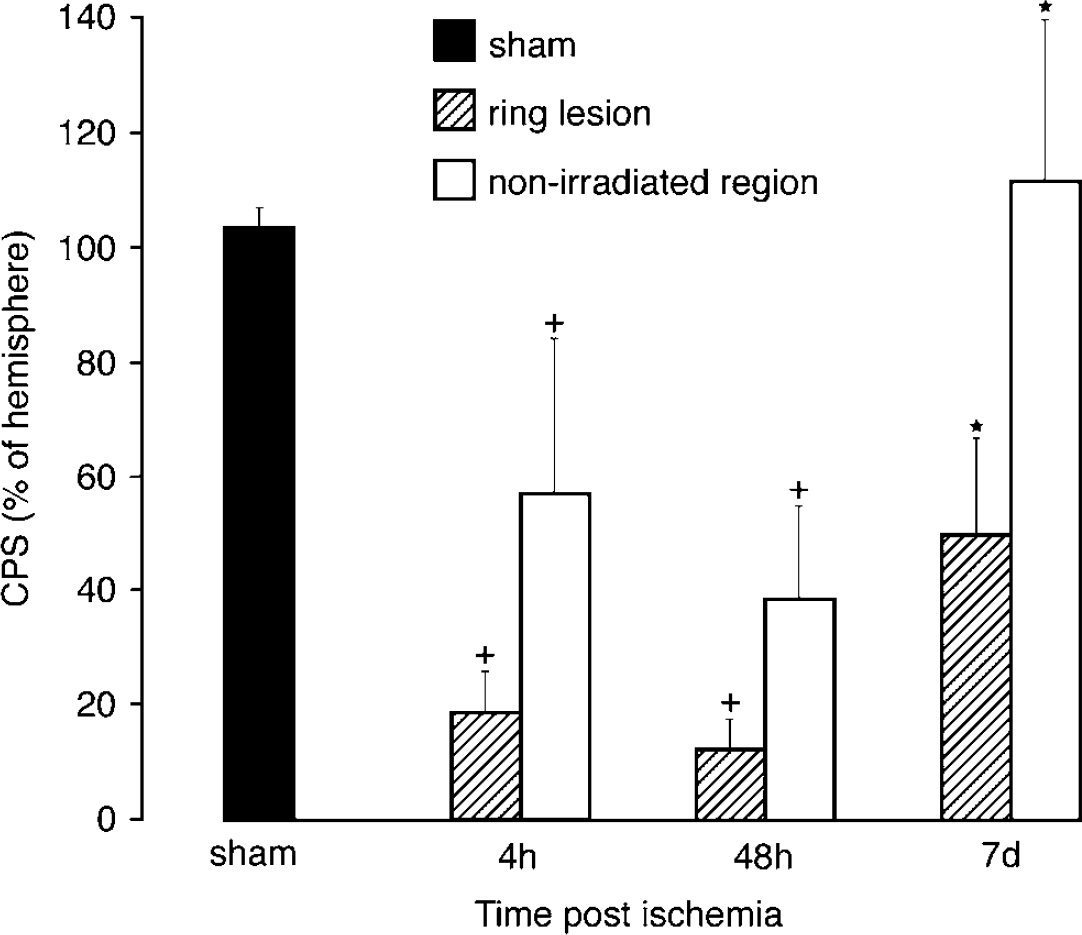
Cerebral protein synthesis (CPS) values measured by [3H]leucine incorporation are presented as percentage of homotopic areas of the contralateral hemisphere; sham-operated controls (n = 3) and irradiated animals at 4 h, 48 h and 7 days postischemia (n = 4 in each group). ± Statistically significant reduction compared with sham-operated animals (P < 0.05 by ANOVA and post hoc tests). *Statistically significant increase compared with values at 48 h postischemia (P < 0.05 by ANOVA and post hoc tests).
Tissue Morphology
Histopathology: Under low magnification, two wedge-shaped cortical lesions corresponding to the position of the ring-shaped laser irradiating beam (ring lesion region) were observed in the H&E stained coronal brain sections at 4 h postischemia (Figure 6B). The centrally located nonirradiated region was relatively spared at this time point. At 48 h after photothrombosis, the ring lesion expanded inward, and also outward, resulting in a progressive encroachment into the center region (Figure 6C). At 7 days, the ring lesion had transformed into a well-demarcated necrosis whereas the nonirradiated central region was only partially compromised (Figure 6D).
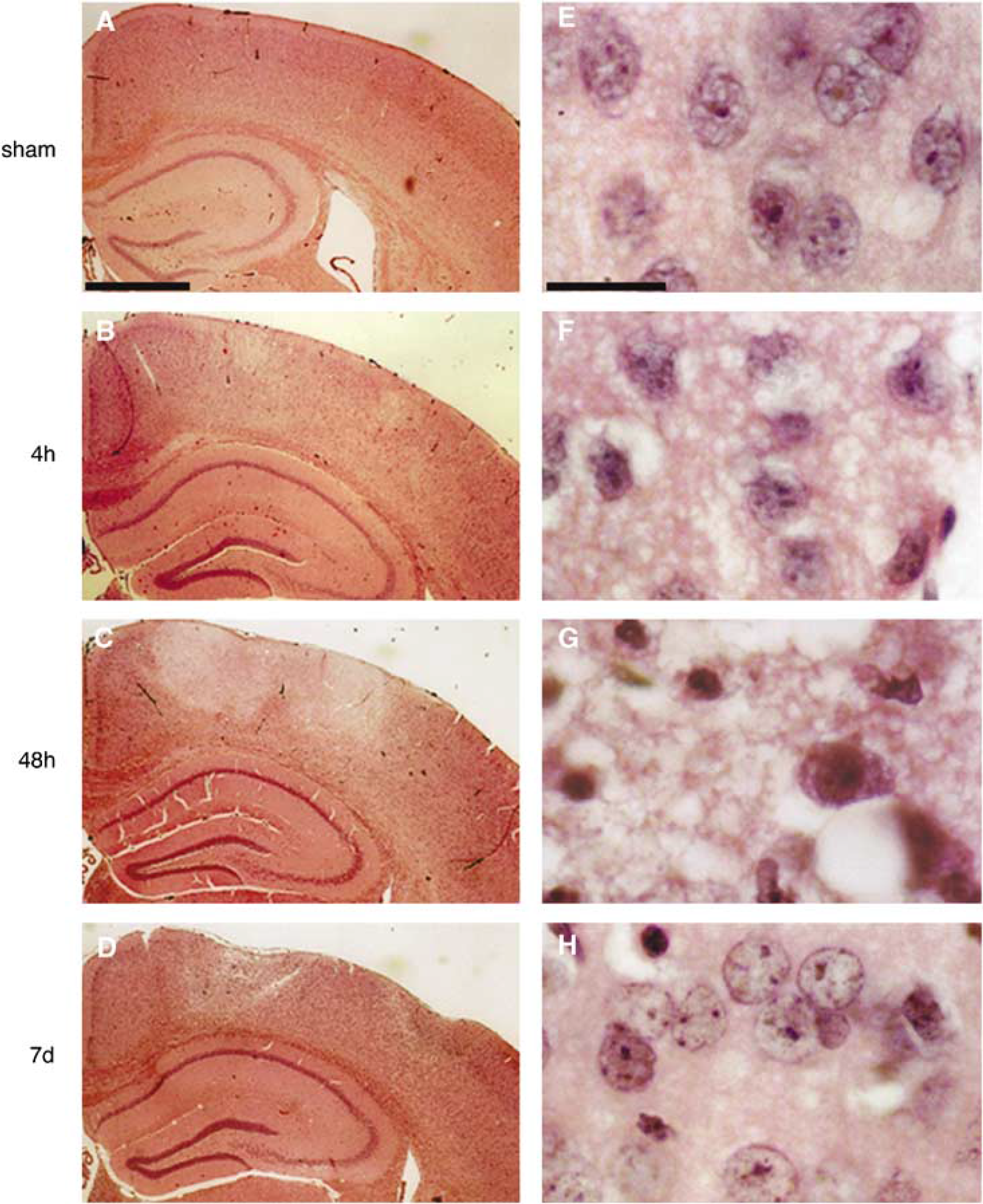
Photomicrographs of paraffin-embedded coronal sections through the epicenter of the ischemic lesion stained with hematoxylin and eosin. Sham-operated controls and irradiated animals at 4 h, 48 h and 7 days postischemia (n = 4 to 6 in each group). The left panel is at low magnification (scale bar = 390 μm) and the right panel at high magnification (scale bar= 10 μm) with tissue examined from the center part of the nonirradiated region-at-risk.
Under high magnification, some neurons in the center of the nonirradiated central region appeared slightly swollen at 4 h postischemia (Figure 6F). At 48 h postischemia, most cells were swollen and the majority of neuronal cells exhibited eosinophilic cytoplasm and pyknotic nuclei (Figure 6G). At 7 days postischemia, some neurons were slightly swollen, and a few were pyknotic but most of them exhibited a normal appearance (Figure 6H). In the exterior counterpart of the region-at-risk, that is, the boundary zone immediately outside the ring lesion region, similar cytologic changes were observed as in the boundary zone immediately inside the ring lesion region.
Immunohistochemistry: At 48 h after stroke, many Neu N immunopositive cells appeared swollen and others with pyknotic nuclei in the cortical layer II to VI of the center of the nonirradiated region-at-risk and also in the boundary zone outside the ring lesion (Figures 7A and 7B). In the cortical ring lesion, few cells were labeled with Neu N. In addition, many cells that were counterstained by hematoxylin were not labeled with Neu N, representing non-neuronal cells (Figure 7B). At 7 days postischemia, the cortical region at risk was heavily populated by Neu N immunpositive neurons (Figures 7C and 7D). At the same time, many GFAP immunolabeled cells were interspersed among these Neu N immunopositive cells (Figures 7C and 7D). In the ring lesion region and the surrounding boundary zone, GFAP immunolabeled cells representing glia was predominantly observed.
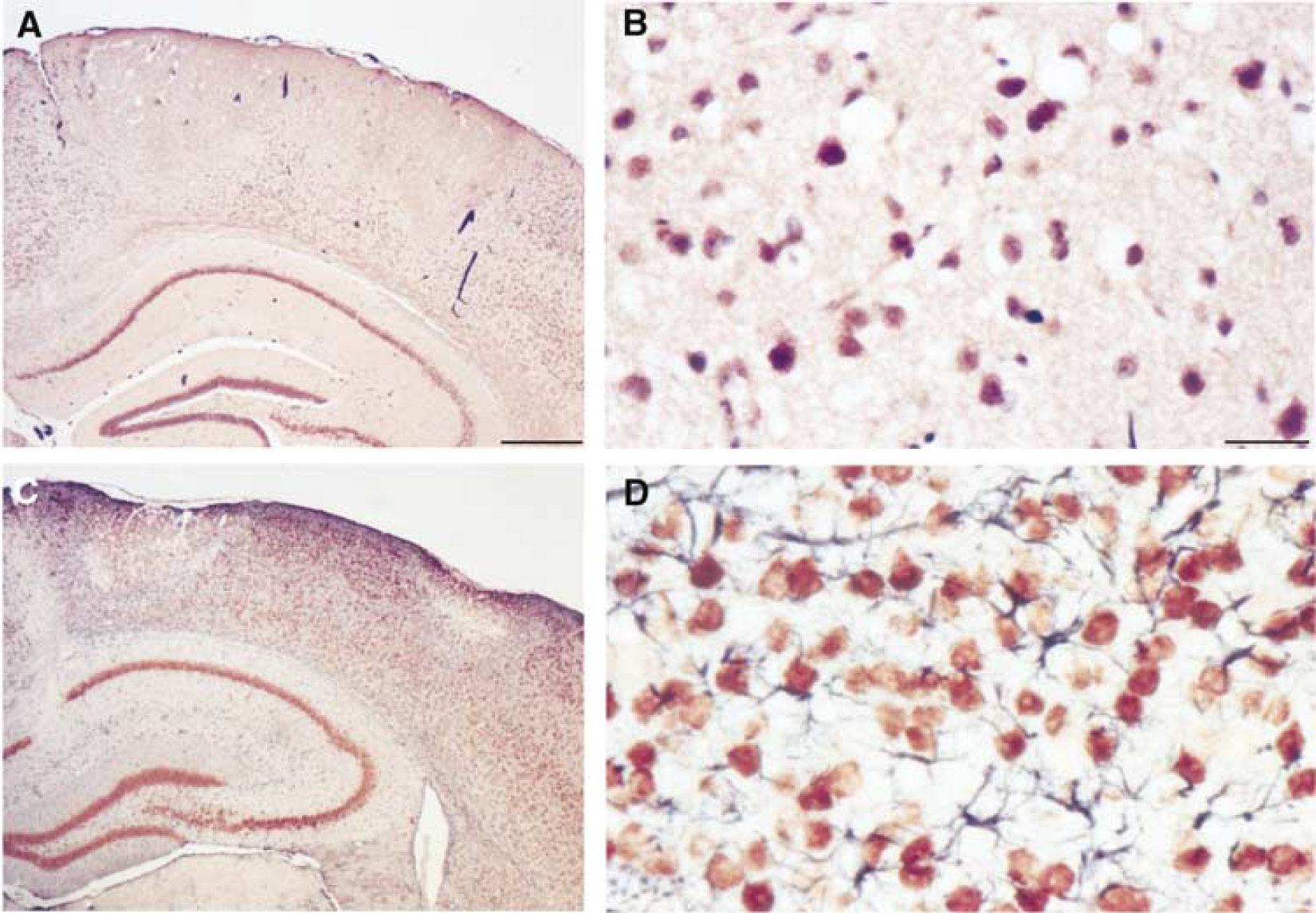
Immunohistochemical cell labeling of coronal brain sections through the epicenter of the ischemic lesion stained with the neuronal marker Neu N at 48 h (
Lesion Volume and Outer Border Expansion
The volume of the photothrombotic lesion amounted to 3.1% ± 0.9% of the ipsilateral hemisphere at 4 h postischemia (mean ± s.d.). With ongoing ischemia time, the lesion volume steadily increased and reached its maximum of 8.2% ± 1.2% at 48 h after photothrombosis (Figure 8). At 72 h postischemia, the lesion volume began again to decline until, after 7 days postischemia, it returned to 3.3% ± 1.0% (Figure 8). The diameter of the ring lesion region increased gradually from 2.0 ± 0.2 (mean ± s.d.)mm at 4 h to its maximum level of 3.3 ± 0.2 mm at 24 h after irradiation. These values remained increased at 48 h (3.2 ± 0.3 mm) and 72 h (3.2 ± 0.2 mm) and then partially returned at 7 days to 2.2 ± 0.5 mm.
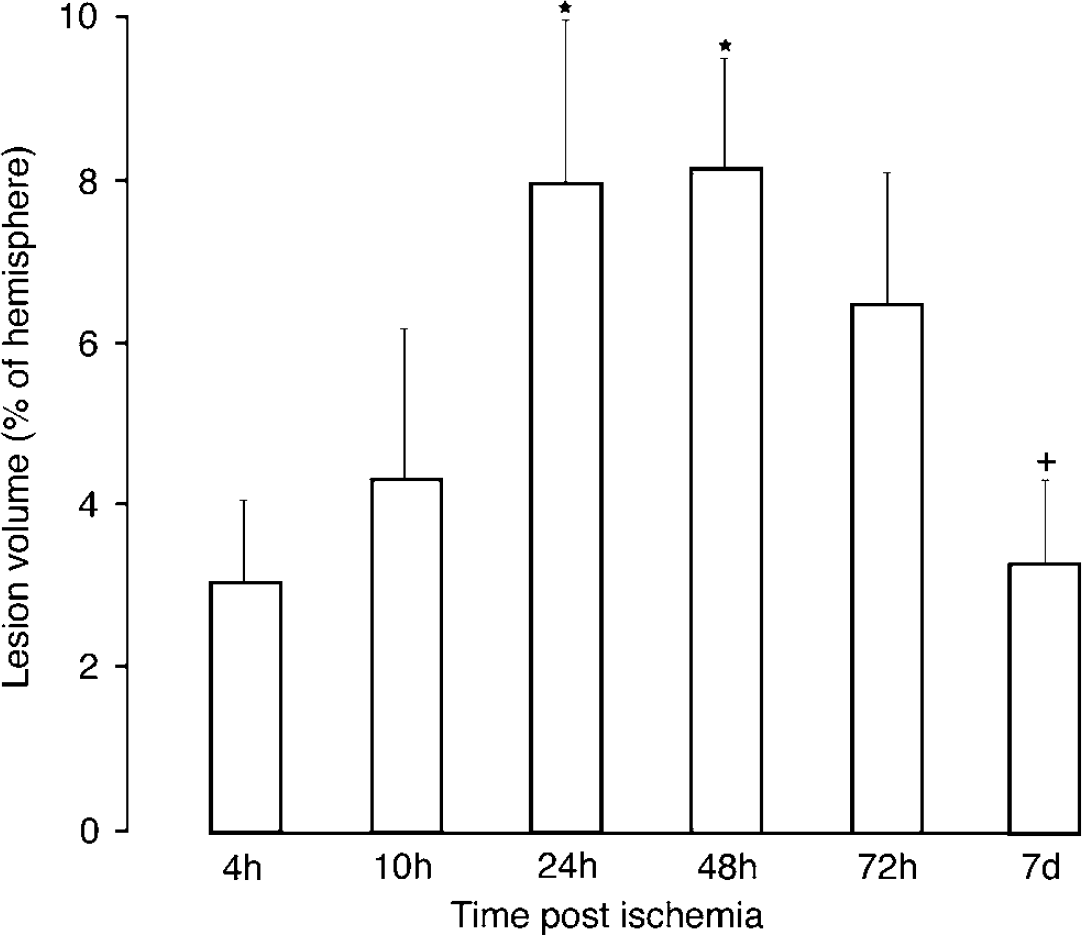
Ischemic lesion volume in percent of the ipsilateral hemisphere at various times after induction of photothrombosis. *Statistically significant increase as compared with 4 h (P < 0.05 by ANOVA and post hoc tests); + Statistically significant decrease as compared with 48 h (P < 0.05 by ANOVA and post hoc tests).
Discussion
The present study shows that the photothrombotic ‘ring’ stroke model with late spontaneous reperfusion could be established in the adult mouse with similar characteristics to those previously observed in adult rats (Gu et al, 1999a, b). To set up this model, the following strategy was used. First, the laser irradiation system was reconfigured so that the ring beam was located within the exposed skull bone of the mice. Second, the laser irradiation intensity was stepwise altered at different concentrations of erythrosin B and different durations of irradiation. The optimal experimental condition was an irradiation intensity of 0.65 W/cm2, an erythrosin B concentration of 4.25 mg/kg, and an irradiation duration of 60 secs. With these settings the nonirradiated cortical center inside the ring lesion underwent progressive hypoperfusion and then late spontaneous reperfusion as observed by transcardial carbon black perfusion (Gu et al, 1999b). lCBF measured by laser Doppler flowmetry (lCBFLDF) in the center of the ring lesion declined to 43% ± 4% of the baseline value at 30 mins after irradiation, which falls into the flow range of the penumbra (Astrup et al, 1981; Symon et al, 1977). The functional impact in this ‘region at risk’ was confirmed by the measurement of CPS, which declined during the phase of hypoperfusion and recovered when blood flow improved. This is similar to the peri-infarct penumbra after vascular occlusion, which also exhibits a suppression of CPS that improves when the tissue is reperfused (Hata et al, 2000; Kokubo et al, 2002). Future studies are required to elucidate whether the cells with unremarkable cytologic appearance at 7 days after stroke induction recovered from the pyknotic appearance seen at 48 h (Gu et al, 1999b) and/or if some of them reflect newly synthesized neurons as observed in the rat (Gu et al, 2000; Jiang et al, 2001). Other findings that replicate the previous observations in rat are the progressive transformation of the irradiated ring lesion into pan-necrosis, which contrasts with the remarkable tissue recovery in the nonirradiated central ‘region at risk'.
An important side effect of the photothrombotic ‘ring’ stroke model is the pronounced tissue swelling caused by the generation of ischemic brain edema. In fact, the lesion volume increased within 48 h by a factor of 2.6, and the ring lesion diameter increased by 65%, followed by the return to the initial size when edema was resolved. It is likely that edema-induced microvascular compression contributed to the progressive deterioration of blood flow in the center of the ring lesion and that the resolution of edema led to the subsequent flow improvement. Other likely factors contributing to the reversal of ischemia are postischemic angiogenesis and the spontaneous wash-out of thromboembolic material (Gu et al, 2001, 1999b; Hilger et al, 2004).
With the establishment of the photothrombotic ‘ring’ stroke model in the mouse, a convenient and technically simple method of focal ischemia has become available, which can be used for a wide range of applications in transgenic and knockout animals. In contrast to the common MCA occlusion models, the ischemic lesion volume is much more reproducible and it does not depend on variations of the genetic background of the arterial brain vasculature since the photochemical insult induces primarily microvascular endothelial damage. The previously documented stimulation of angiogenesis and neurogenesis (Gu et al, 2000, 2001) as well as the differential neuronal injury due to apoptosis and necrosis (Hu et al, 2002, 2004) provide the opportunity to explore the occurrence and interplay of fundamental factors of ischemic cell death or survival in great detail. The herein described murine photothrombotic ‘ring’ stroke model, therefore, opens a wide range of applications in small animals and may be of particular interest for mouse mutants that are suited for the dissection of the mechanisms of ischemic injury and its potential of spontaneous recovery.
Footnotes
Acknowledgements
We thank Professor Brant Watson for discussion and advice throughout this study. Thanks also to André Janz for the technical help on multiparametric imaging.
