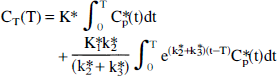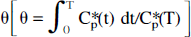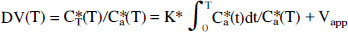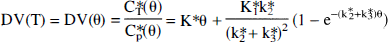Abstract
We tested in normal human subjects a less invasive method to obtain plasma input function required in the calculation of the brain serotonin synthesis rate measured with positron emission tomography (PET) and α-[11C]methyl-tryptophan (α-MTrp). The synthesis rates derived with the arterial input function were compared to those derived from venous plasma and venous sinus time-radioactivity curves obtained from dynamic PET images. Dynamic PET images were obtained for the lengths up to 90 minutes after an injection of α-MTrp (400 to 800 MBq). Input functions were generated from both artery and vein in three subjects, and from artery only in two subjects. Net unidirectional uptake constants of α-MTrp (K*; mL/g/min) were calculated in several brain regions graphically using data between 20 and 60 minutes after injection with different input functions. In the five subjects with arterial sampling, we tested two methods for correcting the input functions from the venous samples: (1) normalization to the mean exposure time at 20 minutes from arterial curve; and (2) the use of the venous sinus curve for the first 20 minutes. Venous curves coincided with the arterial ones after about 20 minutes. When the venous curves were used, there was an underestimation of the area under the curves up to 20 minutes, resulting in a 5% to 30% overestimation of K* values. Combined use of the sinus curve up to 20 minutes and venous curve from 20 to 60 minutes as an input function resulted in the K* (mL/g/min) values larger by 7.1 ± 3.8% than the K* values estimated with the arterial input function. Normalization of the venous curve to the exposure time at 20 minutes obtained from the arterial plasma curve resulted in a bias in the K* of about −0.34 ± 3.32%. The bias from the K* values was propagated to the serotonin synthesis rates. The use of a combination of the venous blood samples and venous sinus as the input function resulted in an acceptable bias in the serotonin synthesis rates from the tissue time-radioactivity curves generated by PET.
5-hydroxytryptamine (5-HT) is a neurotransmitter/modulator widely distributed in the brain, and believed to be involved in the regulation of impulse control, mood, eating, and sleep (van Praag, 1994; Heninger, 1995; Brewerton, 1995). Alterations in serotonergic neurotransmission are thought to play an important role in the pathophysiology of several psychiatric disorders, e.g., depression, suicide, anxiety, and eating disorders. Therefore, a method measuring 5-HT biosynthesis in the living human brain should improve our understanding of the neurobiology of these conditions. Until recently, the only available method to study 5-HT metabolism in humans was based on the peripheral measurements in CSF of the main metabolite of 5-HT, 5-hydroxyindole acetic acid. This measure provides a global index of 5-HT metabolism but is not a measure of 5-HT metabolism in brain per se because the concentration in CSF is confounded by release of the metabolite from the spine (Young et al., 1974). Furthermore, CSF concentrations of 5-hydroxyindole acetic acid reflect multiple processes other than 5-HT synthesis, including 5-HT catabolism and metabolite removal.
To measure rates of brain 5-HT synthesis more directly, a method using labeled α-methyl-L-tryptophan (α-MTrp) was recently developed successfully tested in rat (Diksic et al., 1990; Nagahiro et al., 1990), dog (Diksic et al., 1991) and human (Nishizawa et al., 1997; Muzik et al., 1997, Chugani et al., 1998). However, the α-MTrp method with positron emission tomography (PET), as originally described, requires arterial blood sampling to obtain an input function. To decrease the invasiveness of the α-MTrp method, we have tested two alternative methods in healthy volunteers. The methods described and tested use the venous blood samples corrected with the time-radioactivity curves from venous sinus obtained from dynamic PET images during the first 20 minutes of scanning, as an input function, or normalized venous plasma with a correction factor calculated from subjects with an arterial and venous sampling. Results are compared to those obtained using arterial input function.
MATERIALS AND METHODS
Subjects
We studied five healthy subjects (three male and two female; between 21 and 28 years old) recruited through newspaper advertisement. Because this study evaluated normalization and correction methods for the input function, no special attention in the selection of subjects used in this study was given to their gender. Psychiatric evaluations were performed using the Structured Clinical Interview for the Diagnostic & Statistical Manual (edition 3) (DSM-III-R), nonpatient version. Exclusion criteria included a past or present axis-I or axis-II DSM-III-R diagnosis in the subjects or in their first-degree relatives, or any significant medical illness. The study was approved by the Research and Ethics Committee of the Montreal Neurological Institute and subjects provided written informed consent before their enrollment in the study.
PET procedures
The radiopharmaceutical α-[11C]MTrp was prepared by the method reported by Mzengeza et al. (1995). Subjects were asked to eat precooked low-protein food packaged and delivered to the subject's home on the day before the study, and were asked to fast overnight to reduce variability of plasma amino acids levels, especially tryptophan, during scanning. All studies were performed in the morning. After the intravenous injection (contralateral to the venous sampling) of 400 to 800 MBq (amounts not normalized to the weight of subjects) of α-[11C]MTrp over 2 minutes, dynamic PET scans were performed on a Scanditronix 2048 15B scanner in one subject for 80 minutes and in three subjects for 90 minutes on an ECAT HR+ scanner (Siemens, Canada). Up to 16 blood samples were drawn at progressively longer intervals from both artery (radial artery) and vein (basilic vein) in three and only artery (radial artery) in two subjects to obtain a time-radioactivity course in plasma (input function). Five samples were also used for the high pressure liquid chromatography analysis to determine free and total tryptophan levels in plasma as generally used in the procedure (Nishizawa et al., 1997). All dynamic scans were preceded by a transmission scan for attenuation correction using 68Ga. All subjects underwent magnetic resonance imaging (MRI) (T1-weighted images with 2-mm slice thickness). These images were coregistered on PET images (10 to 20 anatomical landmarks used in coregistration are described in Nishizawa et al., 1997), and used for identification of detailed anatomical structures of the brain. The alignment of images, after selection of the landmarks as described in Nishizawa et al. (1997), was done by the method described by Evans et al. (1991). Regions of interest were drawn manually on the MRI and applied to the dynamic PET images to obtain regional radioactivity time-courses. The PET images were acquired for 0.5 minutes for the first 3 minutes, 1 minute between 3 and 10 minutes, 2 minutes between 10 and 20 minutes, and 5 minutes after 20 minutes to the end of scanning. The PET images were reconstructed with a 8.1 × 8.1 × 8.7 mm resolution, and as such were used for the generation of the tissue time-radioactivity curves.
Calculation of unidirectional uptake constant K* (mL/g/min)
Calculation is based on the unidirectional uptake of α-[11C]MTrp, which is transported into the brain and converted, in part, to α-methyl 5-HT through hydroxylation and decarboxylation (Diksic et al., 1990). The metabolite and/or tracer itself (see discussion in Vanier et al., 1995; Cohen et al., 1995; and Diksic et al., 1990, 1991, 1995) is trapped in serotonergic neurons because α-methyl 5-HT is not a substrate for monoamine oxidase and does not cross the blood-brain barrier back to plasma. The operational equation derived on the bases of known behavior of the tracer is (Diksic et al., 1990, 1991):
Here k2* and k3* are the first order rate constants for the loss of tracer from the precursor pool and entry of tracer into an irreversible pool, respectively. Cp*(t) and CT*(T) are the plasma tracer concentration as a function of time (t; minutes) and the tissue tracer concentration at time T (minutes) after injection. After reaching an apparent steady state, the net uptake (K*; mL/g/min) of α-MTrp in the brain is given by the linear portion of the uptake curve (Patlak et al., 1983; Diksic et al., 1990). The relationship between
and the volume of distribution DV(T) [mL/g] is:
where CT*(T) (nCi/g) and Ca*(T) (nCi/mL) are radioactivity in the brain and the arterial plasma at time T, respectively, and Vapp is a distribution space of the tracer in the brain often known as the precursor pool which includes plasma contribution. The K* values were determined by linear regression (Eq. 2) using a linear portion of the plot after an apparent steady state had been achieved, from 20 to 60 minutes (Nishizawa et al., 1997; Muzik et al., 1997), or by fitting full operational equation (Eq. 1) using a nonlinear least squares and arterial plasma as the input function (Diksic et al., 1991).
An alternative equation can be derived for the relationship between DV(θ) (mL/g) [note that DV(T) (Eq. 2) becomes DV(θ) (mL/g) in the θ-space] and θ (minutes). In the θ-space the plasma input function is by definition equal to unity because by definition θ is the time to which tissue needs to be expressed to a unit concentration in order to achieve the same volume of distribution. (Note that the transfer of a tracer from the plasma into a tissue is proportional to the area under the plasma curve.) In the θ space the plasma has value of one [nCi/mL] for the duration of θ min. This derivation is performed by inserting into the full operation equation (Eq. 1) Cp*(t)≡1 [nCi/mL] (unity), integrating it from zero to θ [minutes], transforming the integrated equation for the tissue tracer concentration into the volume of distribution by division with Cp*(θ) = 1 [nCi/mL], and rearranging. An advantage of fitting this equation [Eq. 3, below] to the experimental tissue time-radioactivity curves is that the entire tissue time-radioactivity curve in the form of the volume of distribution in the θ space is used, and in addition of obtaining K* values, direct estimates of the tissue precursor pool half-life (τ1/2 = ln(2)/(k2*+k3*); min) can be obtained. Because the entire tissue curve is used and the fitting is done to a closed form of the integral equation, the fitting procedure is more stable.
Venous blood curve corrections
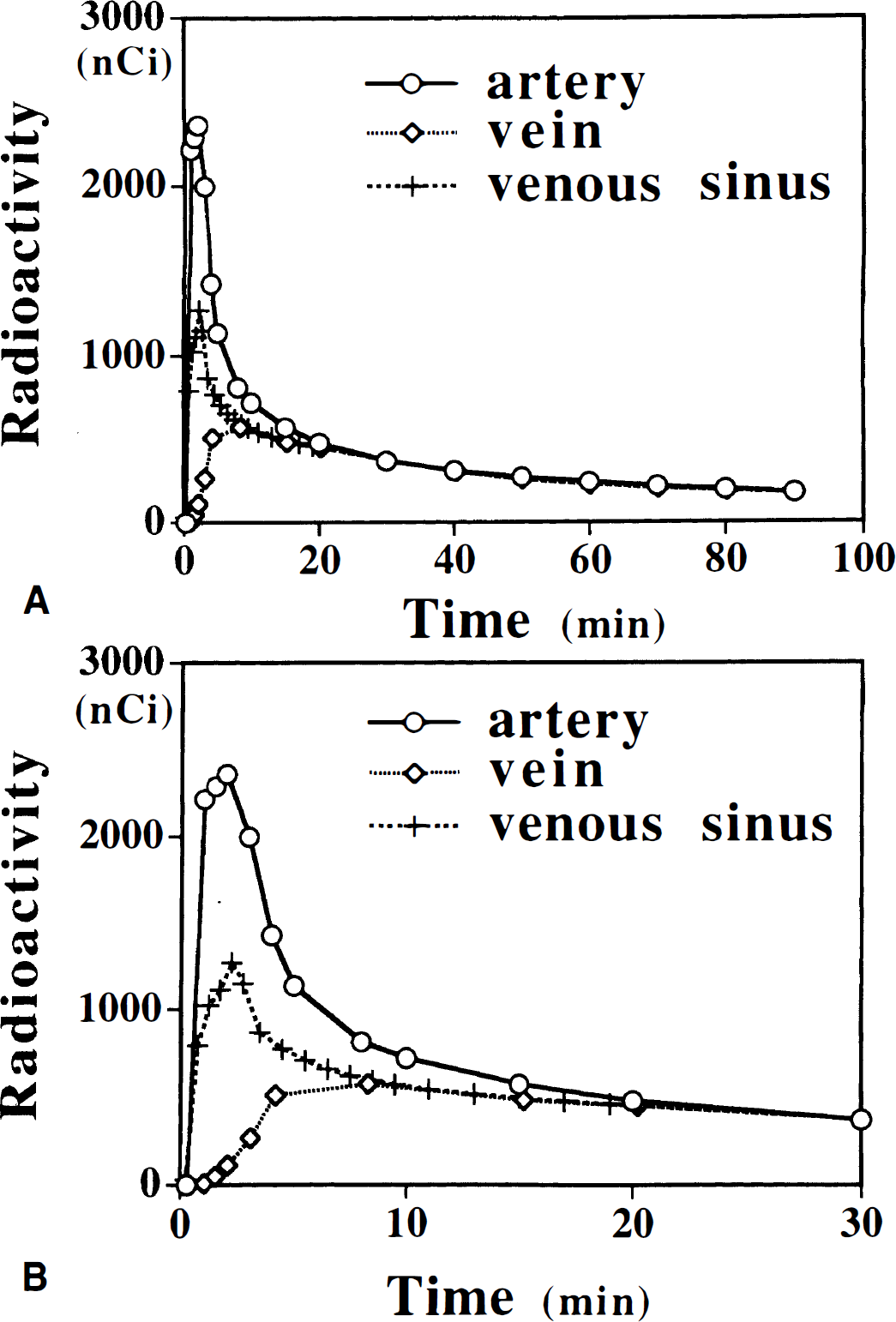
Representative time-radioactivity curves measured in the arterial (C * [over] a(t)) and venous plasma (C * [over[v(t)) from the same subject are shown in Fig. 1. The venous and arterial curves almost coincide from about 20 minutes after injection of tracer to the end of study (Table 1; Fig. 1). Also included in Fig. 1 is the time-radioactivity curve in the venous sinus (C * [over[s(t)) from the dynamic PET images normalized to the venous radioactivity at 20 minutes. Although the sinus curve still underestimates the arterial curve in an early phase, the peak and shape of the sinus curves are similar to those of the arterial curves. Because of the partial volume effect from the surrounding tissue showing gradual increase of radioactivity, the sinus curve does not represent well the arterial curve after 20 minutes.
Ratio of the venous to the arterial radioactivities taken in three subjects having both venous and arterial blood sampling at different times after tracer injection

From the characteristics of the arterial, venous, and venous sinus curves, we designed and tested methods to correct underestimation of the area under the venous curve occurring up to 20 minutes. One correction was performed by using time-radioactivity curve from the venous sinus measured by the dynamic PET images for the first 20 minutes and normalizing it to the venous curve at 20 minutes. After this time, the venous plasma curve was used to the end of experiment (Method 1). Method 2 was designed by making correction to the θ at 20 minutes calculated with the venous plasma time-radioactivity curve using the mean value of the arterial θ at 20 minutes (θa(20)). The θa(20) used in normalization was calculated as an average from the time-radioactivity curves of five subjects with arterial samples. The θa(20) for each subject was calculated as:
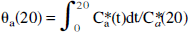
In Method 2 the venous curve was normalized in such a way that venous exposure time at 20 minutes (θv(20)) was made equal to the arterial exposure time (θv(20)≡θa(20)). From this equality and the fact that the concentration of the tracer at 20 minutes in both plasma was about the same, the corrected plasma integral for the venous plasma was calculated. This corrected plasma integral for the first 20 minutes was added to the venous plasma integrals after 20 minutes for the calculation of all subsequent venous plasma normalized exposure times. The slopes K* (mL/g/min) were then calculated using normalized exposure time points (Eq. 2). The rates of 5-HT synthesis (R; pmol/g/min) were calculated as: R = K* Cp/LC; where Cp represents plasma concentration of nonprotein bound tryptophan and the LC represents the lumped constant (Vanier et al., 1995). The LC of 0.42 measured in vivo in rat brain (Vanier et al., 1995) was used. The LC of 0.18 measured in vitro in monkey brain was recently reported (Shoaf and Schmall, 1996), but the calculation was not the same as that in Vanier et al. (1995). When the LC of Shoaf and Schmall (1995) is recalculated in the way of measurements of Vanier et al. (1995), one obtains a value of 1.5 (Diksic, 1997).
All statistical comparisons were performed by the NCSS 97 statistical software (Number Cruncher Statistical Systems, Kaysville, Utah, USA). The K* values obtained by using different input functions, Table 2, and those obtained by fitting of different parts of the tissue time-radioactivity curves in each subject to Eq. 1 given in Table 3, were compared by evaluating standardized z-scores for differences with SD of differences calculated as the square root of the sum of individual variances.
The values of the unidirectional constant K∗ (mL/g/min) for the frontal cortex
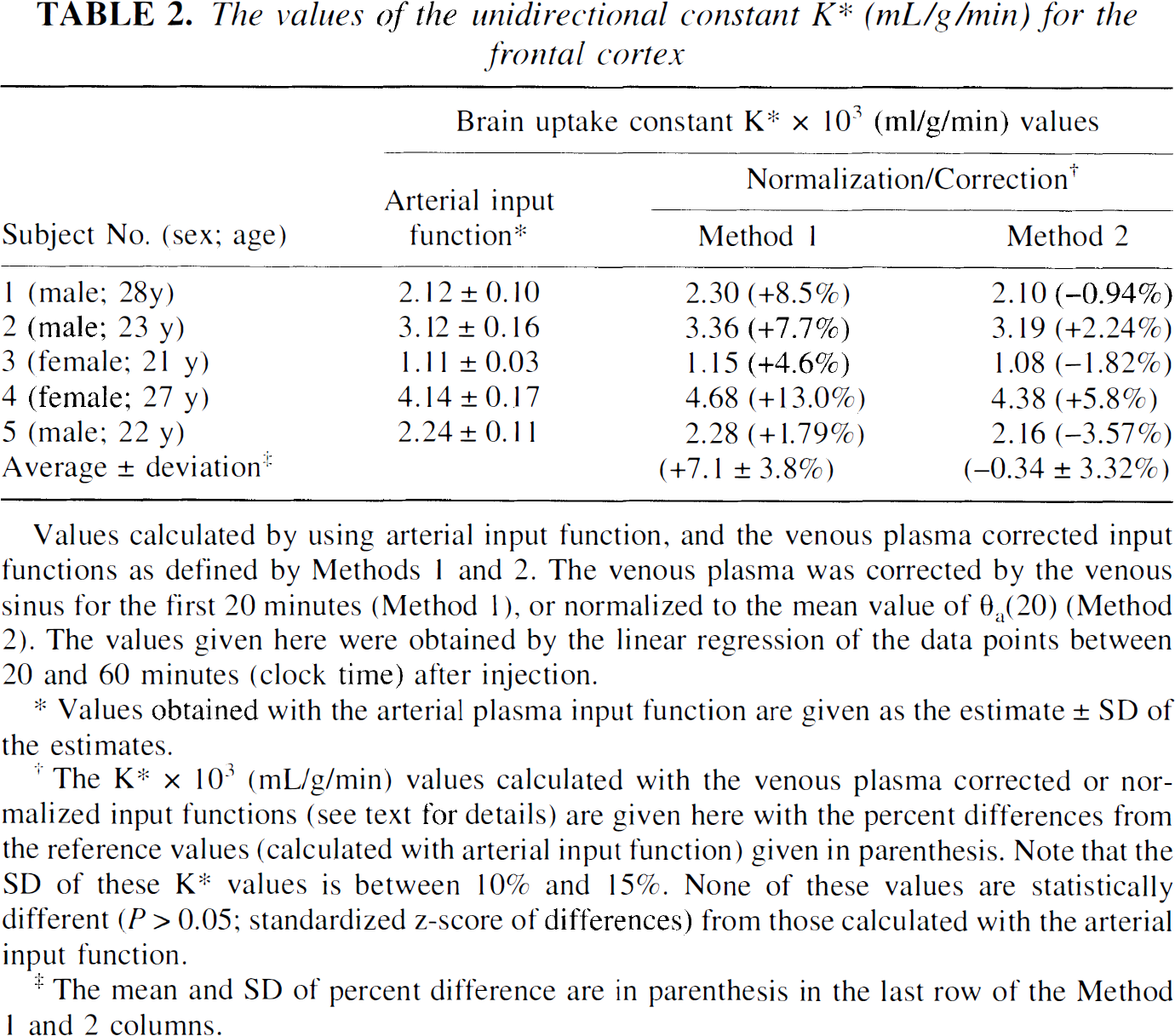
Values obtained with the arterial plasma input function are given as the estimate ± SD of the estimates.
The K∗ × 103 (ml/g/min) values calculated with the venous plasma corrected or normalized input functions (see text for details) are given here with the percent differences from the reference values (calculated with arterial input function) given in parenthesis. Note that the SD of these K∗ values is between 10% and 15%. None of these values are statistically different (P > 0.05; standardized z-score of differences) from those calculated with the arterial input function.
The mean and SD of percent difference are in parenthesis in the last row of the Method 1 and 2 columns.
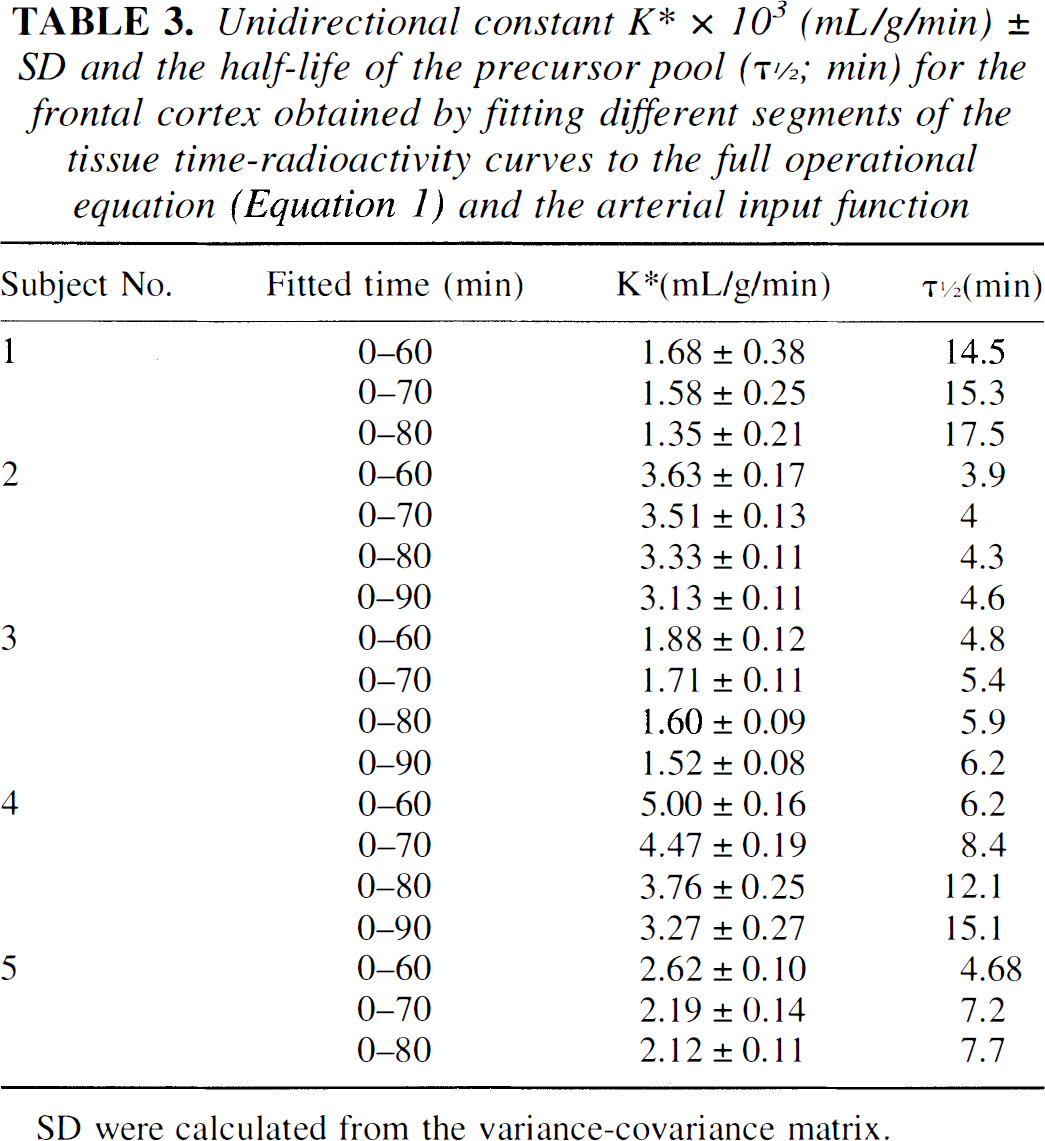
Unidirectional constant K∗ × 103 (mL/g/min) ± SD and the half-life of the precursor pool (τ1/2; min) for the frontal cortex obtained by fitting different segments of the tissue time-radioactivity curves to the full operational equation (Equation 1) and the arterial input function
RESULTS
Representative venous and arterial time-radioactivity curves are shown in Figs. 1 and 2. In Fig. 1A comparisons between arterial, venous, and venous sinus curves are shown. An enlarged portion of the first 30 minutes of the plasma (both arterial and venous) and venous sinus curves (Fig. 1B) are given to exemplify differences between different plasma time-radioactivity curves during first 20 minutes after injection, as well as similarities between them after approximately 20 minutes. From these graphs it is evident that the venous plasma radio-activity is substantially lower than that in the arterial plasma for approximately 20 minutes after injection. After 20 minutes there is no difference between arterial and venous plasma radioactivity concentration. A similar behavior was observed between plasma time-radioactivity curves in the venous sinus and the arterial plasma (Figs. 1A and 1B). Comparing the shapes of the venous plasma curves (Fig. 2), the reason for the underestimation of the plasma integral becomes obvious, mainly because of the underestimation in the plasma time-radioactivity curves during first 20 minutes. This underestimation eventually results in an underestimation of the exposure time θ which results in an overestimation of K* (see later), hence of 5-HT synthesis rates. Simulation studies showed that underestimation of an area under the curve for the first 20 minutes by 20% to 50% results in 8% to 22% overestimation of K* values when the graphical analysis (Patlak et al., 1983) in the α-MTrp method (Diksic et al., 1991) is used.
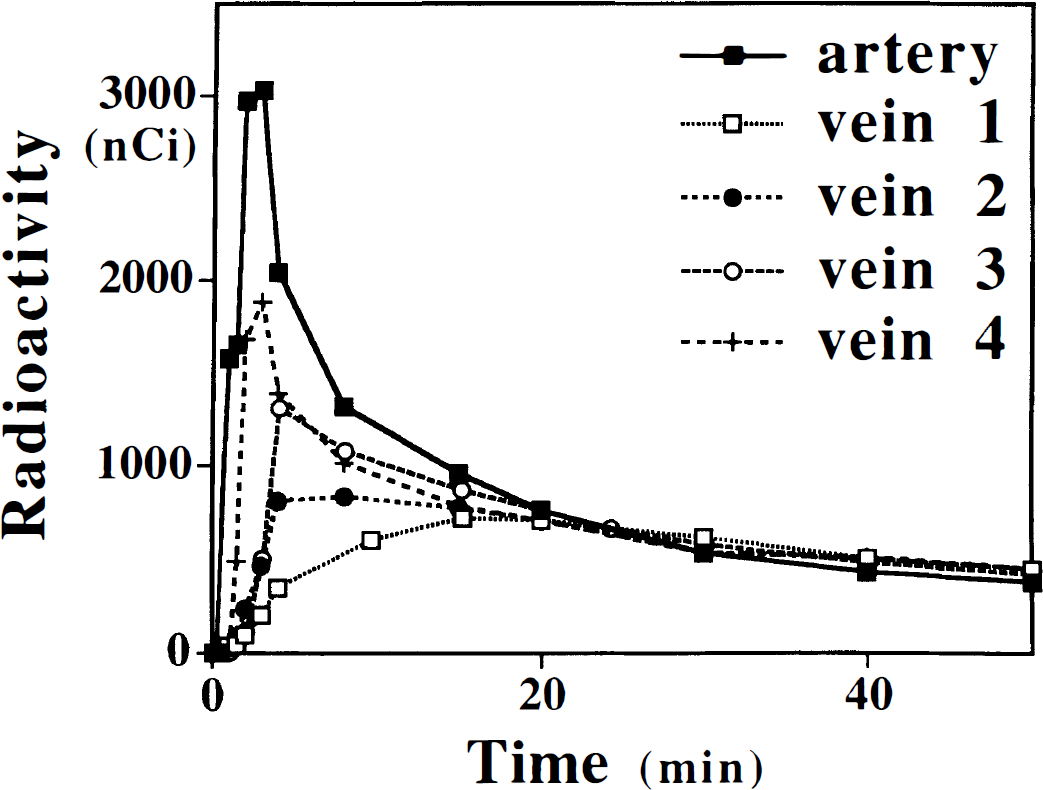
Representative venous curves from 4 subjects of 20 scanned so far are shown to exemplify extreme shapes of the venous curves. For comparison, an arterial curve from one subject is given. Venous curves were normalized to the mean value of the arterial plasma at 20 minutes. Because the venous curves normalization was performed with the mean value of the arterial curves (mean Ia(20)), there is no exact overlap of venous curves with the individual arterial curve. The shape of the venous curves varied among subjects for first 20 minutes. Area under the venous curve varied also from subject to subject and the area under the venous curve was 10% to more than 60% smaller than that under the arterial curve.
The venous curves almost coincide with the arterial curves from approximately 20 minutes after injection onward (Figs. 1A and 1B); however, they were on average 8.3% lower at 20 minutes in three subjects who underwent both arterial and venous sampling (Table 1). In 18 subjects with venous samples (unpublished data) the θv(20) was calculated by equation 4 and using venous plasma radioactivity in the place of the arterial plasma radioactivity. The θv(20) varied from 11.4 to 26.6 minutes (mean ± SD: 20.4 ± 3.4 minutes). In five subjects with arterial samples, the θa(20) calculated by equation 3 using the arterial time-radioactivity curve varied from 29.8 to 38.9 minutes (mean ± SD: 33.5 ± 3.3 minutes). If one uses uncorrected venous plasma time-radioactivity curves as the input function, the K* values are overestimated by 5% to 30%. This observation is in good agreement with the above-mentioned simulation.
In column 2 of Table 2, the K* values and their significant differences in the frontal cortex of five subjects calculated with the arterial input function are given. These values were used as the reference values and were considered to represent the best estimates of K*-s for a particular structure. For comparison, the K* values calculated using methods 1 and 2, and the percent differences between these values and those obtained using arterial input function are also given. There was no significant difference (P > .05; standardized z-score of differences) between K*-s calculated using arterial input function and the input functions defined by methods 1 and 2. The K* values calculated by the methods 1 and 2 had significant differences between 10% and 15%; normalization added additional variability.
When method 1 (venous sinus time-radioactivity curve for the first 20 minutes and after 20 minutes the venous curve normalized to the sinus value) was used, the K* values were overestimated by about 10% in comparison to the K* values calculated with the mean value of θa(20) from five subjects with arterial sampling. The K* values calculated with method 2 [normalizing venous plasma θv(20) to the average value of the arterial Ia(20)] agreed better (difference from reference values: −0.34 ± 3.32%, mean ± SD; Table 2) with the reference values than those calculated by using method 1 (7.1 ± 3.8% difference). These differences are not significantly different (P > .05; standardized z-score of differences). There was generally good agreement between rates in other brain structures calculated by these two methods as the one obtained from the K* values given in Table 2 for the frontal cortex. The agreement between 5-HT synthesis rates calculated using method 2 and those calculated with the arterial input function is shown by images of the 5-HT synthesis rates obtained in one subject shown in Fig. 3 for three different brain cross sections. The MRI images at the same level are shown in the bottom row.
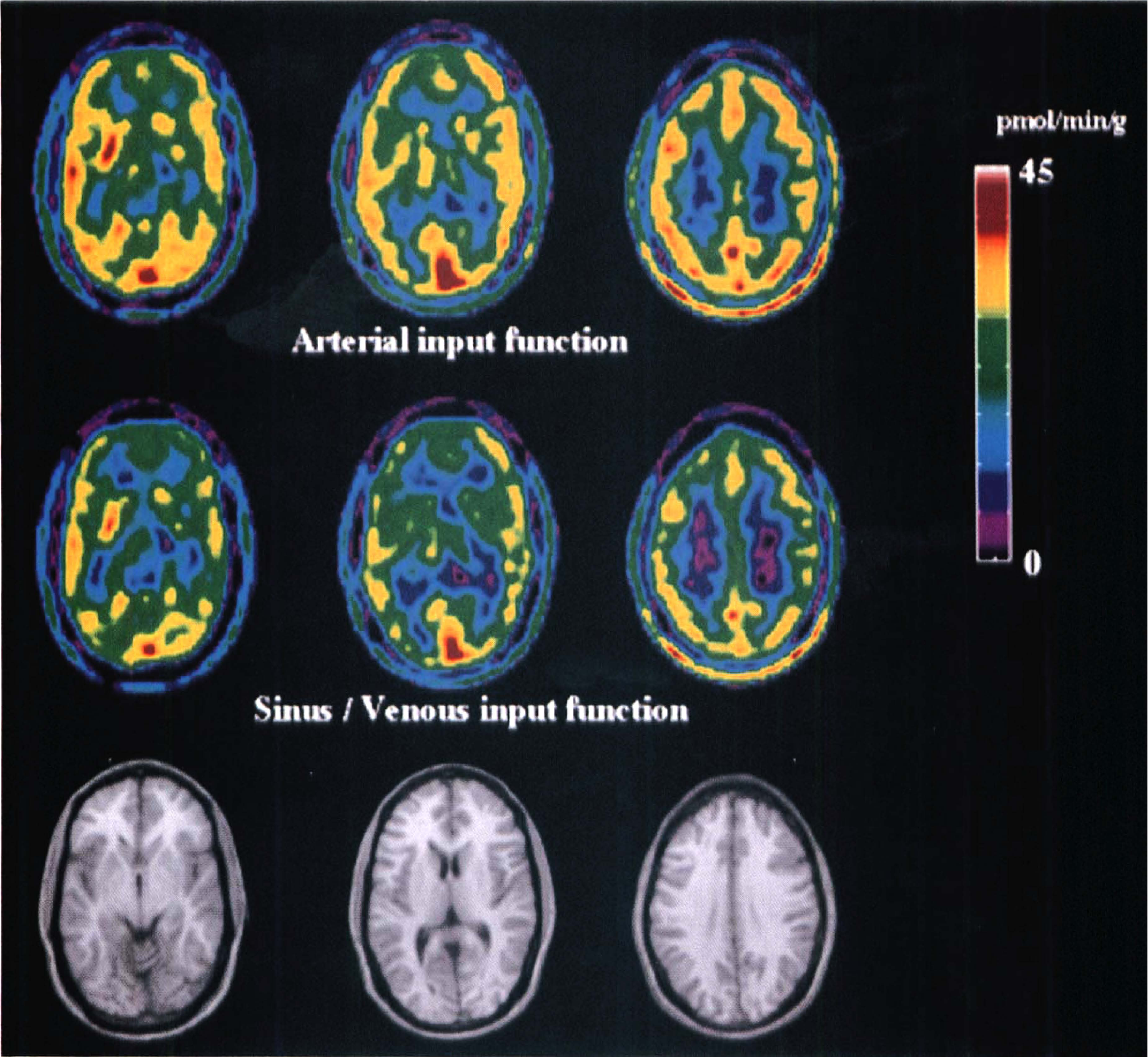
Representative set of positron emission tomographic images showing regional serotonin synthesis obtained by using arterial (top row) and venous sinus normalized venous (middle row; method 1) input function. In the bottom row, magnetic resonance images are shown to aid in the structure identification. The color bar at the side gives the range of the synthesis presented in the positron emission tomographic images.
In Fig. 4 a set of representative plots of the tissue volume of distribution (y-axis) as function of the exposure times (x-axis) is shown. Curves were generated by using venous, arterial, and venous sinus normalized by the venous plasma (method 1), and the venous plasma normalized by the mean value of the arterial Ia(20) (method 2) as the input functions. The measured points are identified by respective symbols (see legend of Fig. 4). The linear fits to the data points between clock times of 20 and 60 minutes are shown as lines. Curves obtained by the fitting of the experimental tissue time-radioactivity curves using arterial (solid circles) and the venous sinus normalized venous plasma (open triangles) as input functions are shown as dotted lines. In general, there was a good agreement between fitted curves and the experimental points.
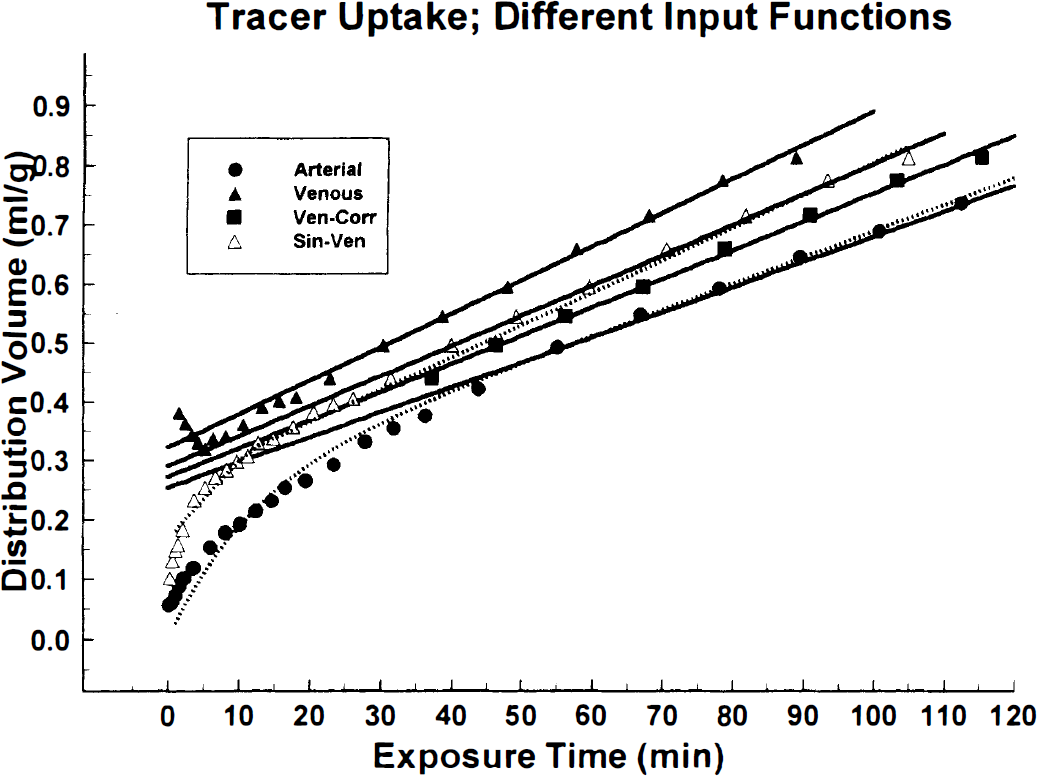
A set of representative plots exemplifying relationship between volume of distribution volume (mL/g) and exposure time (minutes) obtained with the arterial plasma (•); the venous plasma corrected by the venous sinus for the first 20 minutes (method 1); the venous plasma normalized to the mean arterial value of θa(20) (▪; method 2); and the venous plasma without any correction (▴). It can be seen from these curves and the slopes of linear portions that when the venous input function is used the slope is greatest, but it becomes not significantly different (P > .05; two-tailed t-test of slopes) from the one obtained with the arterial input function when the venous plasma is corrected by the venous sinus (method 1) or normalized to the arterial values of the exposure time at 20 minutes (method 2). The fitted curves to equation 3 for the arterial (•) and venous sinus normalized venous (▵; method 1) input functions are shown as dotted lines.
Since scanning in three subjects was performed for 90 minutes, and in two for 80 minutes, the K* (unidirectional tissue uptake constant; mL/g/min) and the half-lives (τ1/2 = ln 2/(k2*+k3*)) of tissue precursor pool were calculated by using 60-, 70-, 80- and 90-minute data by fitting these data sets to the full operation equation (Eq. 1). The results are given in Table 3. The K* values and the resultant 5-HT synthesis rates calculated using 60-minute tissue data are in most cases significantly larger (P < .05; standardized z-score of differences) than those calculated from 90-minute tissue time-radioactivity curves. The half-lives of the precursor pool showed a reverse trend; as the time was longer the τ1/2 became larger (Table 3). The τ1/2 is variable in different subjects. A similar trend was observed when data were fitted to equation 3; however, the actual absolute values of K* were slightly larger (not significantly) and the values of τ1/2 were smaller for a factor varying between 2 and 2.5.
The influence of the time span in the assumed linear part of data on the estimation of K* is exemplified for two subjects in Figs. 5 and 6. For the values presented in the Fig. 5 the starting point of the data was always the same (20 minutes), but the end point varied. As the starting point became closer to the end point the K* values became smaller in one subject (general trend) while in the other there was no real influence on K*. Generally, the values of K* calculated with the venous sinus normalized venous plasma input function were larger than those calculated with the arterial plasma input function (Fig. 4). In Fig. 6 the influences of a 40-minute (the last segment was 30 minutes) data segment of the tissue time-radioactivity curves on the values of K* are shown. It can be seen that taking data at the later stage after injection decreased the difference between K* calculated with arterial and venous sinus normalized venous plasma input function, but at the same time the absolute values became smaller. This trend was not found in one subject in whom the difference and the absolute values were not effected by the time span (Fig. 5).
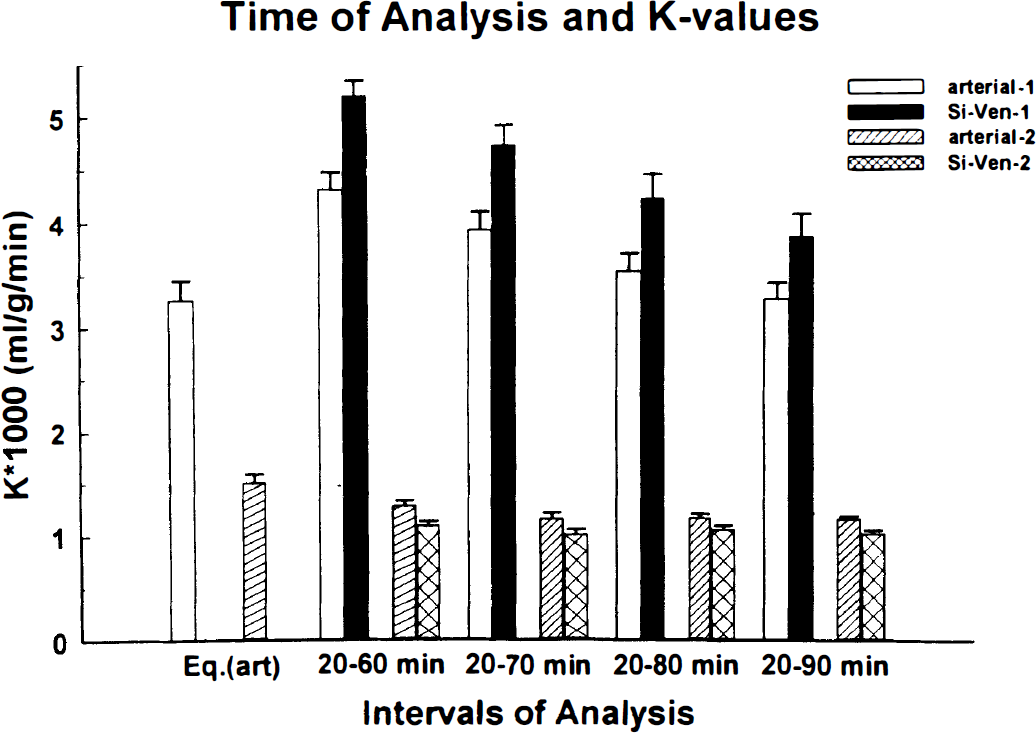
Influence of the end point on the tissue time-radioactivity curves on the values of K* (mL/g/min) calculated with the graphical method is exemplified on data from two subjects. In one subject, as was a general trend, the K* values obtained with the arterial input function (empty bars) were smaller than the K* values obtained by using venous sinus normalized venous plasma time-radioactivity curves (solid bars). The value of K* calculated with the full operational equation (Eq. 1) and using 90-minutes tissue time-radioactivity curve, identified in the figure by Eq. (art) (Eq. 1 with arterial input) is also given. To exemplify an exemption to the above, a subject in which there was no real influence of the time of the tissue time-radioactivity curves and the use of the arterial plasma (stripped bars) or the venous sinus normalized venous plasma (crossed bars) as input function on the values of K* is given. The error bars represent standard deviations of the calculated values and were obtained as square root of the diagonal elements of the variance-covariance matrix.
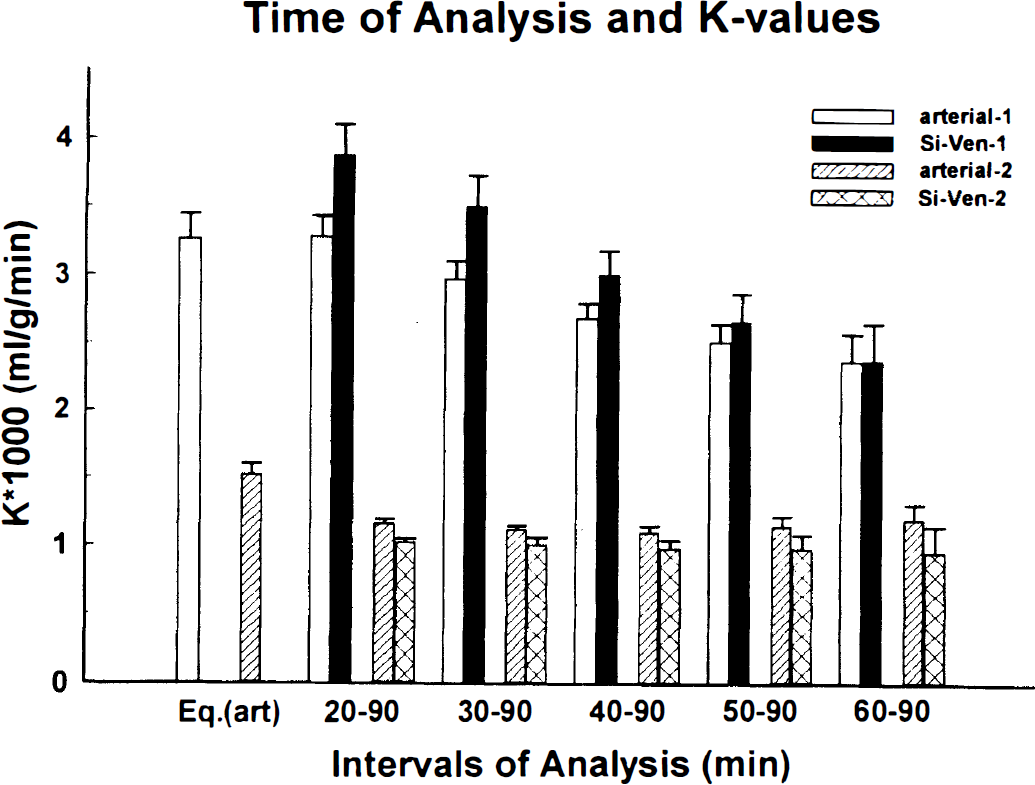
Examples showing the influence of the 40 minutes of the tissue time-radioactivity curves on the value of K* (mL/g/min), and the arterial and venous sinus normalized plasma input functions. Values are given for two subjects similarly as in Fig. 5. The error bars shown have the same meaning as those in Fig. 5. The values of K* calculated by nonlinear least squares fitting of the tissue time-radioactivity curves to equation 1 with arterial input function identified as Eq. (art) (Eq. 1 with arterial input) are given for comparison.
In Figs. 5 and 6, the K* values calculated with the full operational equation (Eq. 1) and using 90-minute tissue time-radioactivity curves are given (identified Eq.[art] [Eq. 1 with arterial input]) for comparison. Comparing values of K* obtained with the full operational equation with those calculated using different parts of the tissue uptake curves and different input functions, one observes a reasonable agreement with the K* values calculated from 20- to 60-minute segments of the tissue time-radioactivity curves and linear fitting (Fig. 5).
DISCUSSION
Disturbances in brain serotonergic neurotransmission are thought to increase vulnerability to a wide range of psychiatric and neurologic disorders (Brewerton, 1995). Many of these disorders can be alleviated by treatments that are thought to affect brain 5-HT function. However, because adequate methods for studying brain serotonergic function (e.g., 5-HT synthesis) in living human brain are only now being developed, the above hypotheses remain largely untested. The method presented here should help the collection of data that would permit a better understanding of what constitutes normal and abnormal 5-HT synthesis. To reduce the invasiveness associated with arterial sampling which is usually required for in vivo methods, a simplified method using venous samples and a normalization procedure is proposed and tested here.
Alternatives to arterial sampling have been used successfully in other PET studies. Input functions derived from venous samples (Phelps et al., 1979) and a population-based standardized arterial input function with one or two point calibration (Takikawa et al., 1993) have been used in 18F-deoxyglucose PET studies. In cardiac studies, time-radioactivity curves from the left ventricle (Weinberg et al., 1988) or thoracic aorta (Gambhir et al., 1989) have been used as the input function to calculate cardiac blood flow with 15O-water or glucose use with 18F-fluorodeoxyglucose.
In the method using α-[11C]MTrp the most convenient way to calculate tissue uptake, K* values, is by graphical analysis (Diksic et al., 1991; Muzik et al., 1997) proposed by Patlak et al. (1983). In the graphical analysis, accurate time-course of the radioactivity is required for the linear part of the plot, but up to that time point, it is not the exact time-course of activity but the area under the plasma curve that affects the accuracy of the K* values (Kato et al. 1985).
We used a venous curve for from 20 to 60 minutes for the analysis of tissue time-radioactivity curves. At 20 minutes, the venous radioactivity was still on average 8% lower than the arterial one, which resulted in an error of estimated K* values (Table 1). On the basis of the arterial venous differences, one could overcome this bias by using the venous curve after 30 minutes. But for the cases in which only 60-minute data are available, this approach would result in a larger error of the estimated slope for a small structure with the graphical analysis (Muzik et al., 1997, present report).
In an attempt to correct underestimation and variability of the area under the venous curve (Fig. 2) up to 20 minutes, we tested the method using the standardized Ia(20) obtained from five subjects (method 2), and the time-radioactivity curves from the confluence of venous sinuses in the dynamic PET images (method 1). Sinus curves from PET images are affected by the loss caused by the limited spatial resolution, spillover from the surrounding tissues, and possible differences in the tracer concentration in the plasma versus whole blood. It was not possible to correct properly for these factors because the venous sinus on the transverse image is very small and varies in size. An attempt to find a common recovery coefficient and spillover fraction was not successful. Therefore, individual sinus curve was normalized to the venous curve at 20 minutes, which in addition to the venous curve after 20 minutes was used as the input function.
There is a negative correlation between the length of time after 20 minutes and the values of K* (K*-s calculated as the slope of a linear regression of data between 20 and 60, 20 and 70, 20 and 80, and 20 and 90 minutes) in majority of subjects but not in all (Fig. 5). The main reason for the great dependance of the K* values on the portion of the time-radioactivity curve used, when occurs (it was not present in all of our subjects), is probably the half-life of the precursor pool (Muzik et al., 1997) because the biological system has not reached an apparent steady state. However, it could be also related to an imperfection of the biological model. A large variability in the calculated half-lives are also, probably, in part a biological reality, and it could be related to the imperfection of the biological modeling. The biological model probably only grossly approximate biological reality, as is the case with all biological models.
The K* values calculated with the input function normalized according to methods 1 and 2 agreed well with the K* values calculated with the arterial input functions (Table 2). It seems that the agreement between K* values calculated with venous curves normalized with θa(20) and those calculated using arterial input function is somewhat better than that obtained between K*-s calculated with the venous sinus corrected venous plasma curve and those with the arterial plasma (Table 2; Fig. 4). However the difference between the two approaches is not significant (Table 2). From these comparisons one can conclude that the normalization of the venous plasma time-radioactivity curve with θa(20) would be the method of choice. Therefore, the venous plasma curve normalization by either approach described here can be clinically useful because 5-HT synthesis rates can be measured with a less invasive approach. The K* values reported in Table 2 are within a range (between 1 × 10−3 and 8.5 × 10−3 mL/g/min) observed by us in a group of 25 normal subjects scanned so far (to be published). In this larger group of normal subjects we did not observe a significant correlation between K* or R, and the plasma total or free tryptophan (to be published), which is in an agreement with the findings reported earlier by us (Nishizawa et al., 1997). The adequacy of either normalization of the venous input function is not surprising because only a small fraction of this tracer is extracted by the brain (less than 10%; Takada et al., 1993).
SUMMARY
We tested the validity of the method using venous samples as an input function for the estimation of the rate of the 5-HT synthesis with PET and α-[11C]MTrp. Combined use of the radioactivity-time courses from the venous samples, corrected by the venous sinus from dynamic PET images, or population mean Ia(20), provided reasonable estimates of K* values. This less invasive method without arterial blood sampling is more convenient to be applied to subjects with different disorders in which brain 5-HT may be involved.
Footnotes
Acknowledgements
The authors thank the staff of the Medical Cyclotron and PET Units of the McConnell Brain Imaging Centre, Montreal Neurological Institute.