Abstract
Extracellular potassium ion activity ([K+]o) increases precipitously during brain ischemia when blood flow falls below threshold values less than approximately 15 mL/100 g/min. This flow threshold for increase of [K+]o occurs also in focal ischemia producing gradient from ischemic core to adjacent normally perfused brain. In this study we investigated the spatial and temporal stability of extracellular potassium ion and blood flow gradients after permanent middle cerebral artery occlusion (MCAO) in rats. [K+]o and regional CBF were measured, respectively, with K+-sensitive and polarographic hydrogen-sensitive microelectrodes at different cortical locations in the middle cerebral artery distribution region. Spatial assessment of [K+]o and regional CBF was conducted at 30, 90, and 180 minutes after MCAO. [K+]o in the more lateral cortex (core) increased from near 3 mmol/L before MCAO to greater than 50 mmol/L and was associated with flow values less than 25% of pre-ischemic levels. Measurements medial to the core (penumbra) indicated progressively decreasing levels of [K+]o and improvement of CBF. There was a tendency for [K+]o in penumbral zones to decrease toward normal levels with time, but there was little dissipation of [K+]o in core regions. In contrast, the spatial CBF profile remained remarkably constant for the entire recording period. Thus, unlike infarction which has been reported to expand with time after focal ischemia, the spatial [K+]o disturbance tends to contract primarily due to decreasing [K+]o with time in the penumbra. Thus, steady state levels of [K+]o after focal ischemia may not be a valuable predictor of cell viability.
Energy failure associated with brain ischemia produces severe changes in the distributions of most ions including the cations sodium, potassium, calcium, and hydrogen (Hossmann et al., 1977; Hansen et al., 1980; Siemkowicz and Hansen, 1981; Mutch and Hansen, 1984; Mayevsky et al., 1985; Hossmann and Grosse Ophoff, 1986; Hansen and Nedergaard, 1988; Hossmann et al., 1988; Raffin et al., 1991). The direction of ionic movement is down the electrochemical gradient for each ion species. For example, potassium ion increases in the extracellular compartment (Hossmann et al., 1977; Hansen et al., 1980; Raffin et al., 1991) while both sodium ion and calcium ion decrease in the extracellular compartment (Hossmann et al., 1977; Siemkowicz and Hansen, 1981) and increase intracellularly (Uematsu et al., 1988). Moreover, the changes in brain ionic concentrations during severe ischemia are not gradual but explosive in nature. Extracellular potassium ion increases suddenly from approximately 3 mmol/L to more than 50 mmol/L in a matter of seconds after the onset of ischemia while extracellular sodium and calcium ion concentrations decrease suddenly at approximately the same time.
Similar ionic changes have been observed in focal ischemia except that the region of major ionic disturbance is confined to the ischemic core (Branston et al., 1977; Watanabe et al., 1977; Strong et al., 1983; Young et al., 1987; Schielke et al., 1991; Rickels et al., 1993; Betz et al., 1994; Gido et al., 1997), with smaller changes in the penumbra surrounding the core (Branston et al., 1977). Major ionic impairment occurs when cerebral blood flow decreases to a threshold level (Branston et al., 1977; Mies et al., 1984; Schielke et al., 1991). Similar flow thresholds have been linked to edema formation, brain infarction, and irreversible damage (Schuier and Hossmann, 1980). In focal ischemia, flow deficits are graded from the ischemic core to penumbra to peripenumbral tissue resulting in large ionic concentration gradients over distances of a few millimeters. For example, extracellular potassium ion concentration increases to greater than 50 mmol/L in the ischemic core but remains at near 3 mmol/L in adjacent normal cortex (Strong et al., 1983; Schielke et al., 1991). The penumbra represents a region of tissue at the interface between severely depolarized brain with high extracellular potassium ion concentration and brain with normal ion distributions. Because of the extreme potassium ion concentration difference between core and normal tissue, it might be expected that the penumbra is unstable over time. Previous investigations have described expansion of the region of infarction over time after permanent focal ischemia (Busch et al., 1996). This expansion has been related to the occurrence of episodes of spreading depression-like depolarization waves that emanate from the ischemic core (Mies et al., 1993; Busch et al., 1996; Takano et al., 1996), but occurs also to some extent in the absence of such events (Busch et al., 1996).
In this study we monitored changes in the spatial profile of extracellular potassium ion in cerebral cortex for up to 3 hours after permanent middle cerebral artery occlusion (MCAO) in the rat. Our hypothesis was that the spatial potassium ion concentration gradient from core to normal cortex would promote an increase in the level of penumbral and peripenumbral extracellular potassium. Such a mechanism might explain the expansion of infarct volume over time reported by other investigators (Busch et al., 1996).
MATERIALS AND METHODS
Data were obtained from 15 male Wistar rats weighing from 300 to 400 g. Rats were anesthetized with sodium pentobarbital (35 mg/kg) and catheters were inserted into a femoral artery and vein for monitoring blood pressure, sampling blood for blood gas analysis, and for administering additional anesthetic as required. A tracheal cannula was also inserted and the animals were placed on positive-pressure ventilation. The rate and stroke/volume of respiration were adjusted to maintain blood gas and pH values in the physiological range (PaO2 > 100 mm Hg, PaCO2 35 to 40 mm Hg, pH 7.35 to 7.45). Maintenance of adequate blood oxygenation without decreasing arterial PaCO2 was facilitated by respiring animals with 30% O2 – balance nitrogen.
Animals were prepared for proximal MCAO by exposing the artery through a left subtemporal incision, exposure and partial removal of the zygomatic arch, and craniotomy. The middle cerebral artery (MCA) was dissected from connective tissue and loosely held by surgical suture. A larger left temporalparietal craniotomy was performed to expose the cerebral cortex in the distribution zone of the MCA from the midsagittal suture, extending approximately 10 mm laterally over the temporal lobe. The dura was removed over this area to facilitate insertion of microelectrodes and the brain was kept moist with warmed saline. The exposed area was subdivided under microscopic control into eight 1 mm segments in the lateral to medial direction beginning near the first major branch of the MCA. These segments were used to facilitate reproducible placement of potassium-sensitive and hydrogen sensitive microelectrodes for measuring extracellular potassium ion activity [K+]o and blood flow, respectively.
[K+]o was measured with double-barreled glass microelectrodes (tip diameter 2 to 6 μm) as described previously (Raffin et al., 1991). One barrel of the microelectrode pair was filled with liquid ion exchanger resin for potassium (Fluka Potassium Cocktail 60031) and backfilled with 100 mmol/L KCl, while the other barrel was filled with 150 mmol/L NaCl. Electrical signals from the two barrels were monitored with a high impedance differential electrometer amplifier. The potential difference between the two barrels was logarithmically proportional to potassium ion activity at the microelectrode tip. Before and after each experiment, the microelectrode pair was calibrated in solutions containing known concentrations of potassium.
Local cortical microflow was measured with glass-insulated platinum polarographic microelectrodes (10 μm diameter tapering to 2 μm at the tip) as described previously (Feng et al., 1988). The microelectrodes were inserted approximately 500 μm into the cortical surface and were polarized to +250 mV with respect to a Ag-AgCl reference electrode placed in a nearby muscle. Blood flow was estimated from hydrogen clearance curves generated by respiring animals with 10% H2 − 30% O2 – balance nitrogen. Blood flow was calculated using the following equation: f = c(0.693/t)*100, where f is blood flow in mL/100 g/min; c is the blood-brain partition coefficient for H2, which was assumed to be unity, and t is the time (min) required for brain H2 to decay halfway from peak to baseline.
K+-sensitive and hydrogen clearance microelectrodes were initially placed in the lateral aspect of the MCA distribution zone which was determine from preliminary studies to always undergo depolarization after MCAO. The MCA was permanently occluded by gently lifting the MCA from the brain surface and applying electrocautery. Complete cessation of MCA blood flow was confirmed visually and by a sustained increase of [K+]o in the lateral zone. Animals in which [K+]o increase in lateral cortex was not sustained after electrocautery were not included for analysis.
Spatial measurements of [K+]o were conducted two separate ways. In a limited number of experiments, [K+]o was measured in six cortical zones with six separate K+-sensitive microelectrodes. Because of the difficulty of this approach and the high failure rate of individual measurements, subsequent spatial measurements were conducted by moving a single microelectrode to different recording locations. Both methods produced essentially identical results. Only data from the single electrode experiments were included for statistical analysis. In another group of animals [K+]o and CBF were measured simultaneously at three separate sites with three K+-sensitive and three hydrogen-sensitive microelectrodes. In these experiments the K+-sensitive and hydrogen-sensitive microelectrodes at each site were positioned within 100 μm of each other.
Spatial measurements of [K+]o and local CBF were acquired immediately, 30, 90, and 180 minutes after MCAO. Data were analyzed statistically by repeated measures analysis of variance, and individual mean comparisons were made by orthogonal contrasts. For statistical comparisons, CBF was expressed as a percentage of pre-ischemic control values.
RESULTS
Figure 1 is a diagrammatic representation of the MCA distribution in the rat cortex showing the regional scale and approximate position of recording microelectrodes. In subsequent figures, recording zones refer to the distance from midline with zone 8 being most medial and zone 1 being most lateral. Figure 2 shows sample records from an experiment in which six K+-sensitive microelectrodes simultaneously measured changes in [K+]o in zones 2–8 after permanent MCAO. [K+]o in zones 2, 4, and 5 showed sustained increased from control levels near 3 mmol/L to greater than 50 mmol/L with little dissipation in the entire 3-hour recording period. These regions were subsequently referred to as the ischemic core. [K+]o in more medial zones showed transient increase at the onset of ischemia which subsequently returned to or toward control levels. The duration of [K+]o increase decreased with distance from core regions. The transient increase of [K+]o in zone 8 likely represented cortical spreading depression as described by others (Nedergaard and Hansen, 1993) and did not reflect ischemia. Typically, eight to 14 episodes of spreading depression occurred in penumbral zones during 3 hours of permanent MCAO. Because the intent of the present study was to evaluate changes in steady state [K+]o across the MCAO distribution zone, further analysis of spreading depression-like events was not conducted. Zones 6 and perhaps 7 likely represent ischemic penumbra. Figure 3 shows similar data recorded from a single K+ – sensitive microelectrode that was moved from site-to-site after permanent MCAO. Also shown in this figure are actual values of [K+]o recorded in each location.
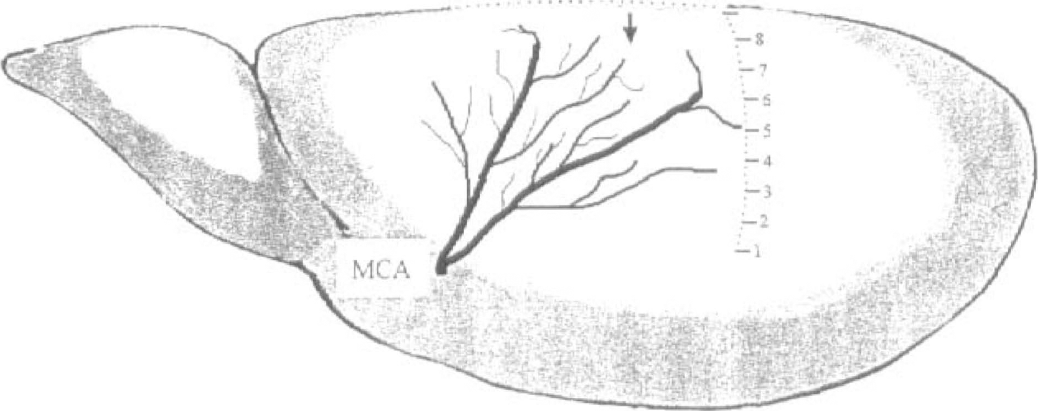
Diagram of the rat brain showing the distribution of the middle cerebral artery (MCA) and the medial-lateral zones for recordings of extracellular potassium ion activity and CBF. The arrow indicates the approximate anterior-posterior positioning of microelectrodes.
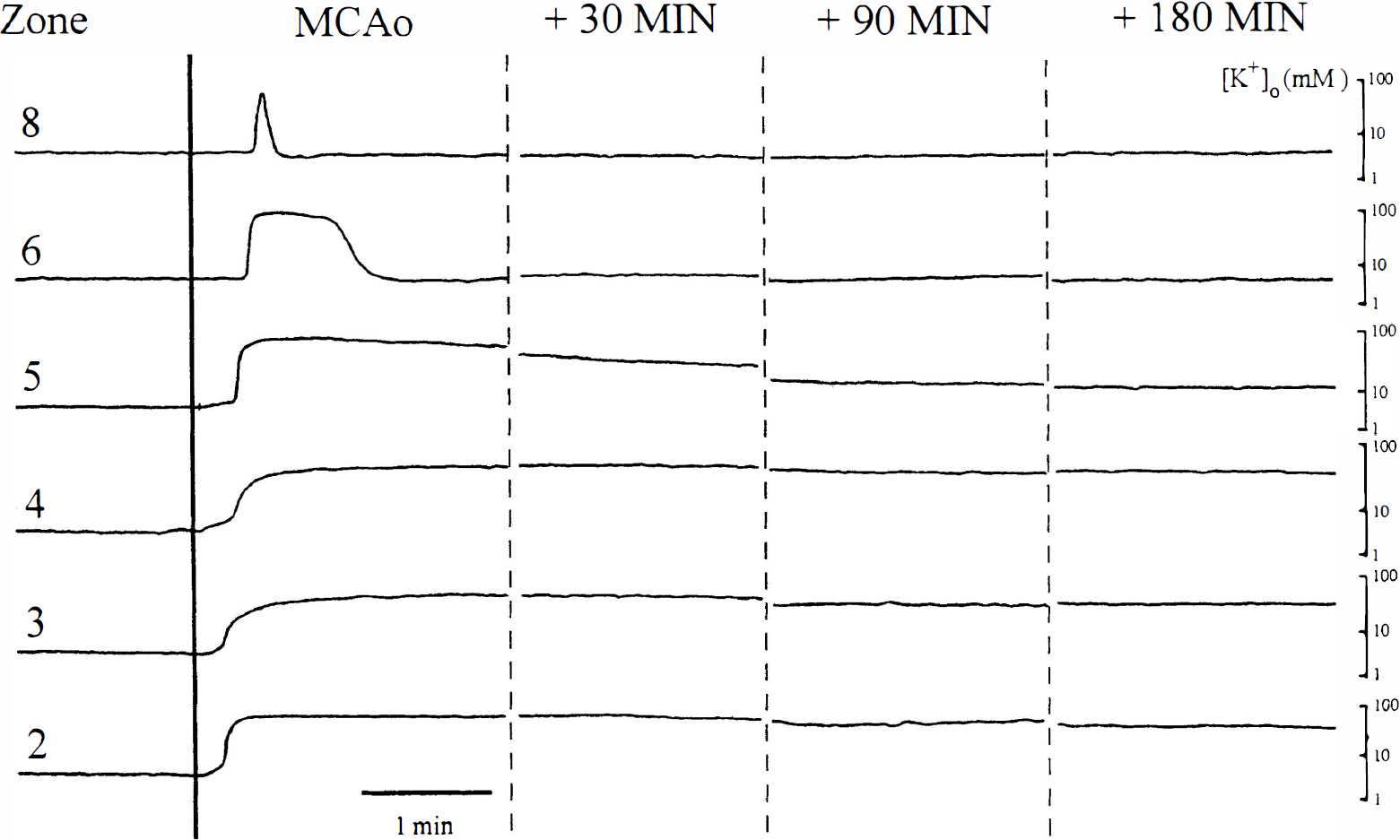
Six simultaneous recordings of extracellular potassium ion activity ([K+]o) after permanent middle cerebral artery occlusion (MCAo). Exerpts of records are shown before occlusion, during occlusion, and 30, 90, and 180 minutes after occlusion. Each record was selected to show typical baseline [K+]o levels and was selected to avoid changes associated with episodes of spreading depression (see text). Zones refer to the medial-lateral position of K+-sensitive microelectrodes as described in Fig. 1.
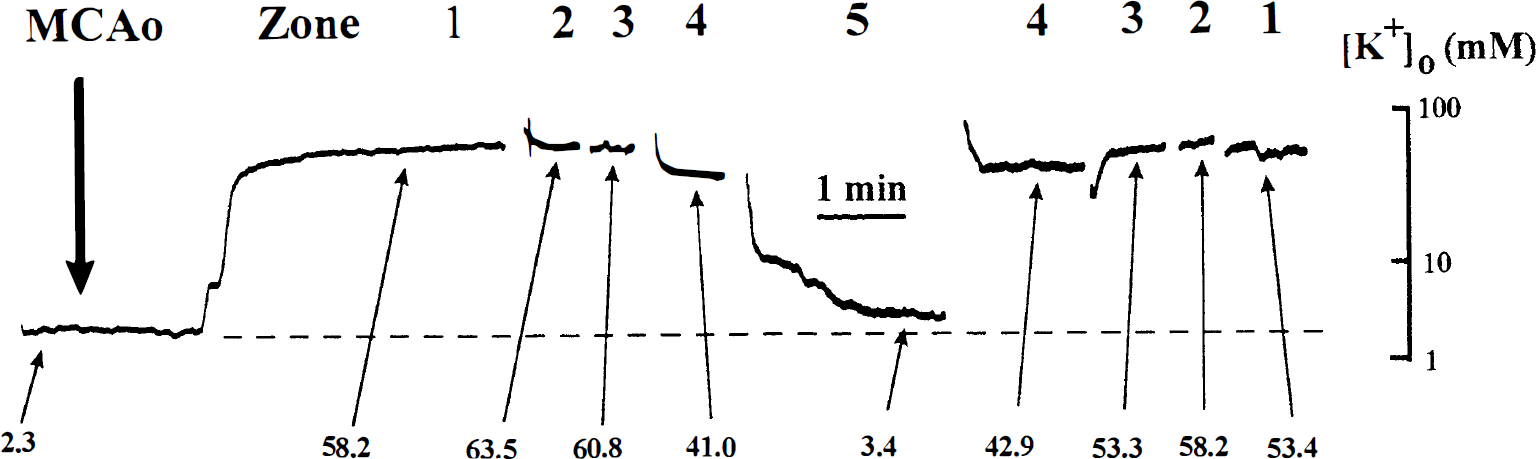
Sample record showing values of extracellular potassium ion activity recorded in different middle cerebral artery zones with a single K+-sensitive microelectrode after middle cerebral artery occlusion (MCAo). Zones refer to medial-lateral position of the microelectrode as shown in Fig. 1. Values of extracellular potassium ion activity are shown at the times indicated.
Figure 4 summarizes the spatial distributions of [K+]o and the changes in these distributions 30, 90, and 180 minutes after MCAO. As expected, there was a significant difference in [K+]o values recorded from lateral to medial cortex (from zone 1 to 8) after MCAO (F = 31.3, P < .001). Thirty minutes after MCAO, [K+]o in zones 1–3, respectively, averaged 56.6 ± 7.6 mmol/L, 57.2 ± 7.9 mmol/L, and 58.3 ± 11.4 mmol/L. In zones 4–8, [K+]o averaged 45.4 ± 21.9 mmol/L, 38.3 ± 17.2 mmol/L, 26.9 ± 21.5 mmol/L, 12.4 ± 9.5 mmol/L, and 2.7 ± 0.5 mmol/L. Thus, the steepest [K+]o gradient occurred between zones 4–6. This region likely represents ischemic penumbra.
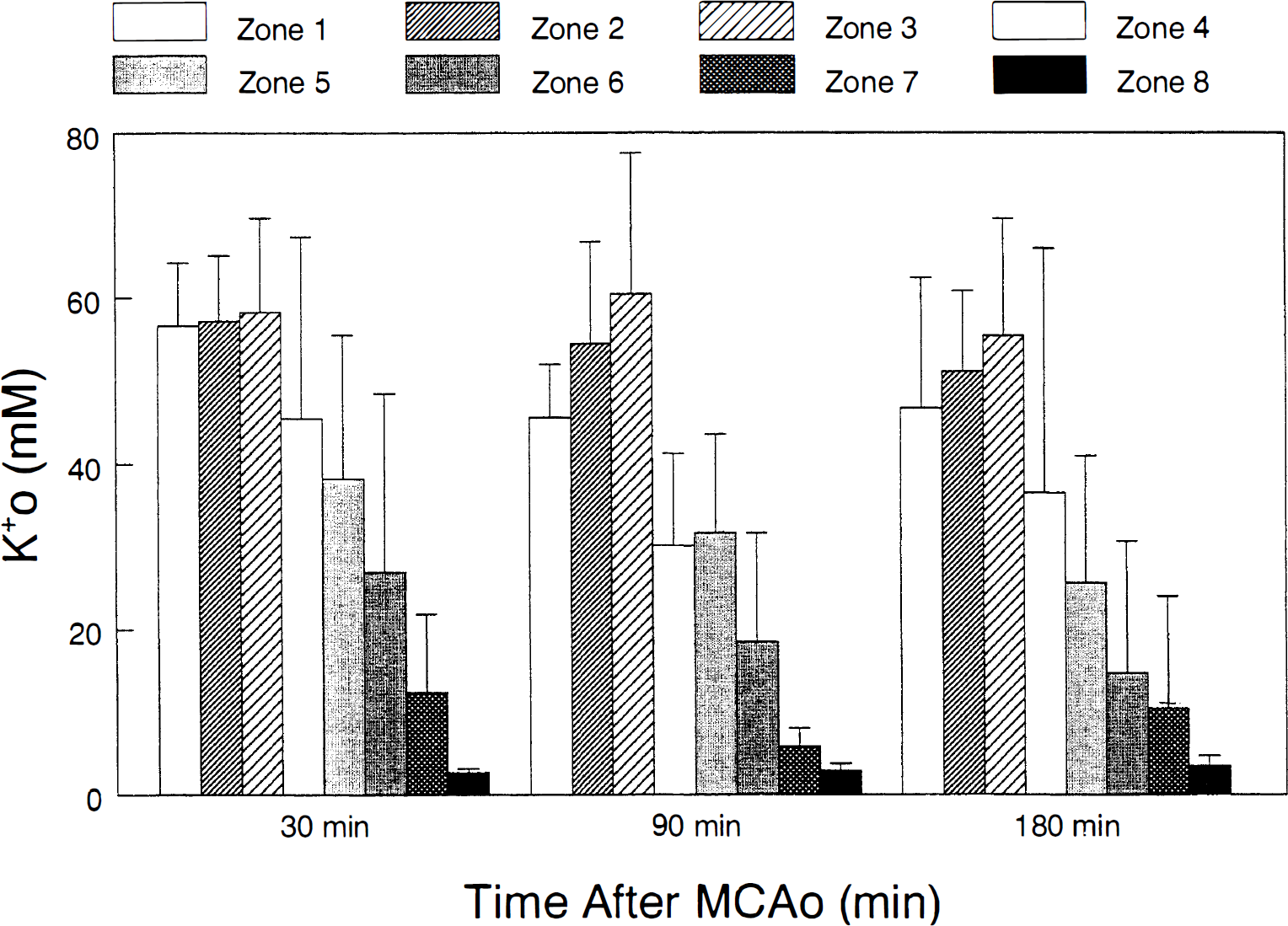
Average values of extracellular potassium ion activity (mean ± SD, n = 7) recorded in medial-lateral zones 1–8, 30, 90, and 180 minutes after middle cerebral artery occlusion (MCAo). All zones were significantly increased compared to zone 8 (P < .01.
The distribution of [K+]o across cortical zones 1–8 also changed significantly with time after MCAO (F = 5.3, P < .01). There was a tendency for [K+]o in zones 4–6 to decrease with time after MCAO, especially between 30 and 90 minutes. For example, [K+]o in zone 4 decreased from 45.4 ± 21.9 mmol/L to 30.1 ± 11.1 mmol/L. The significant decrease in [K+]o in penumbral zones was also observed in a second group of animals (see below) in which [K+]o and CBF were measured simlutaneously. However, there was little change in [K+]o over time in core zones (1–3), and in the most medial zones (7 and 8).
To assess the contribution of blood flow changes to the spatial distribution of [K+]o after MCAO, [K+]o and CBF were recorded simultaneously in three cortical regions 30, 90, and 190 minutes after ischemia from a second group of animals. Figure 5 shows sample records of hydrogen clearance traces and [K+]o recordings before and after permanent MCAO in zones 2, 6, and 8. In this animal, blood flow ranged from 37.1 to 57.7 mL/100 g/min and [K+]o was near 3 mmol/L in all three regions before MCAO. After MCAO, core flow (zone 2) decreased to less than 10 mL/100 g/min and [K+]o increased to greater than 50 mmol/L. The decrease in CBF and increase of [K+]o was sustained throughout the 180-minute recording period. In penumbral cortex, CBF decreased to less than 30 mL/100 g/min and [K+]o was increased to near 10 mmol/L. In zone 8, CBF remained normal or slightly hyperemic and [K+]o remained at control levels.
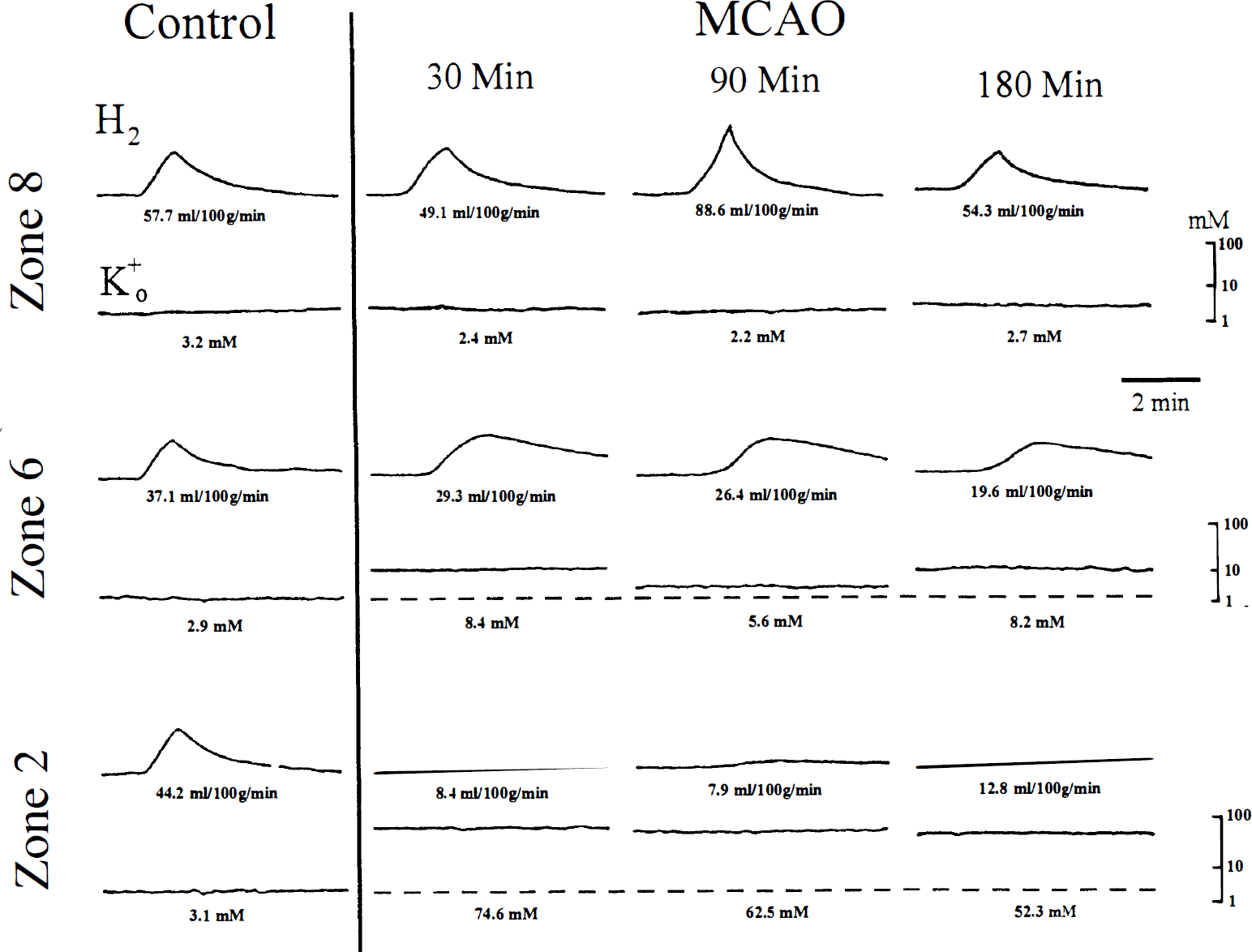
Examples of simultaneous hydrogen clearance traces and extracellular potassium ion activity recordings in three medial-lateral zones before and 30, 90, and 180 minutes after middle cerebral artery occlusion (MCAo). Numbers shown are local CBF in mL/100 g/min, and extracellular potassium ion activity in mmol/L.
These data are summarized in Fig. 6 which shows both local CBF and [K+]o recorded simultaneously in zones 2, 6, and 8 after MCAO. Thirty minutes after MCAO, CBF in the ischemic core (zone 2) averaged 22.1 ± 13.3% of pre-ischemia levels. In penumbra (zone 6), CBF averaged 37.8 ± 24.0% of pre-ischemia levels. As in the previous group of animals, [K+]o decreased significantly in penumbra (zone 6) 90 and 180 minutes after MCAO compared to levels observed at 30 minutes. Little change in [K+]o was observed over time in core (zone 2) or peripenumbra (zone 8). While there was an inverse relationship between [K+]o and CBF across cortical zones after MCAO, it did not appear that the decrease in [K+]o in penumbra between 30 and 90 minutes after MCAO was flow related. There was no concomitant increase in blood flow 90 or 180 minutes after MCAO in zone 6.
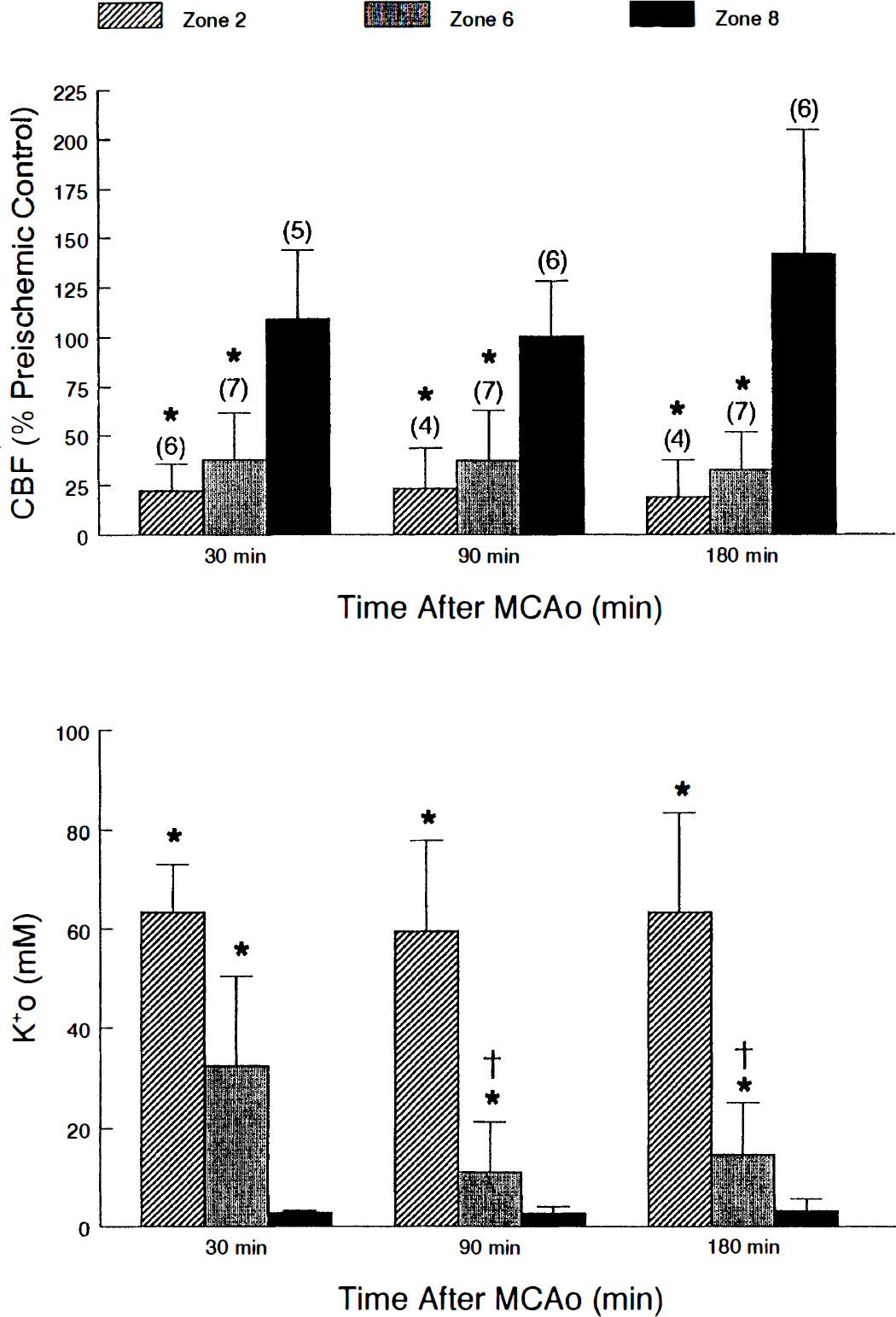
Average values (mean ± SD) of local CBF expressed as a percentage of pre-ischemic control levels, and extracellular potassium ion activity in mmol/L, taken 30, 90, and 180 minutes after permanent middle cerebral artery occlusion (MCAo). Numbers in parentheses indicate the number of animals. (* significantly different from zone 8, P < .01; significantly different from zone 6 at 30 minutes, P < .01).
DISCUSSION
The loss of transmembrane ion gradients that accompanies focal cerebral ischemia is similar to that reported after global ischemia except that the changes are confined only to brain regions with severely limited blood flow. The data reported here confirm earlier reports that extracellular potassium ion levels increase to greater than 50 mmol/L in the ischemic core where blood flow is reduced to a threshold level of 10 to 15 mL/100 g/min (Branston et al., 1977; Strong, 1983 #9228]; Schielke et al., 1991). We also confirm that small (5 to 10 mmol/L) increases in extracellular potassium ion are associated with moderate decreases in blood flow in penumbral cortex (Branston et al., 1977; Strong et al., 1983; Schielke et al., 1991), and that there is little increase in extracellular potassium ion in cortex immediately adjacent to the ischemic penumbra (peripenumbra) with normal blood flow. Thus, the ionic disturbance in focal cerebral ischemia could be described as an island of severely depolarized tissue surrounded by relatively normal tissue within just a few millimeters of the ischemic core. We further report here that the extracellular ionic disturbance in the ischemic core is spatially stable, changing little after 3 hours of ischemia. However, extracellular potassium ion concentration in the ischemic penumbra decreased significantly over time. Thus, we conclude that expansion of the ischemic core over time reported by other investigators (Busch et al., 1996) does not simply result from a progressive elevation of extracellular potassium ion, driven by the concentration gradient of this ion from core to penumbra and peripenumbra.
Dynamic changes in the ionic composition of the ischemic core and penumbra are important because they may ultimately contribute to the extent of neuronal necrosis and neurologic impairment. High levels of extracellular potassium and perhaps also excitotoxic agents, such as glutamate, in close proximity to moderately impaired or normal tissue, could provide conditions for expansion of infarction. An extreme example described by many investigators is the propagation of waves of cortical spreading depression waves from core tissue into peripenumbral cortex (Mies et al., 1993; Iijima et al., 1992; Back et al., 1994; Hossmann, 1996). Prevention of post-ischemic cortical spreading depression decreased focal infarction, while triggering spreading depression from remote sites increased infarction volume (Busch et al., 1996; Takano et al., 1996). However, even in the absence of cortical spreading depression, infarction tends to expand over time (Busch et al., 1996). It was reasonable to speculate, then, that expansion of brain infarction could result from continued exposure of viable brain tissue to the high levels of potassium and excitotoxic agents (Matsumoto et al., 1996; Butcher et al., 1990).
In this study we found remarkable stability of increased extracellular potassium ion in the ischemic core. The data show that under conditions of low blood flow and compromised cellular metabolism, the brain is poorly equipped to dissipate high extracellular levels of this ion. Decreasing levels of [K+]o in the core to those found in normal cortex must take many hours and perhaps days. Diffusion of extracellular solutes, such as potassium, from regions of high concentration in the core to regions of lower concentration in the penumbra and peripenumbra may be hindered in part by the long diffusion distances involved (many millimeters). Also, diffusion through the extracellular compartment will be limited because of shrinkage of the extracellular space that occurs after ischemia (Hossmann et al., 1977; Hansen and Olsen, 1980). Other mechanisms for removal of extracellular potassium ion such as glial spatial buffering (Gardner-Medwin, 1983; Largo et al., 1996 a,1996 b) likely are not operative in the ischemic core where potassium ion concentrations are uniformly distributed above 50 mmol/L. In the penumbra, where we observed a significant decrease in extracellular potassium ion over time, both passive diffusion and glial spatial buffering could play a role. However, we observed no increase in extracellular potassium in penumbral or peripenumbral tissue, which might suggest extracellular potassium ion movement from the core. But it remains possible that over long periods such increases might have been too small to observe. Also, it is unlikely that cellular elements, either neurons or glia, in the penumbra could buffer the large extracellular potassium ion loads occurring in the adjacent core.
The tendency in the present study for extracellular potassium ion concentration in penumbra to decrease over time is consistent with the notion that this ion might be removed through the brain vasculature (Bradbury and Stulcova, 1970). Activation of the Na,K-ATPase by potassium in isolated microvessels occurs in the range from 3 to 14 mmol/L (Schielke et al., 1990), concentrations that clearly occur in the ischemic penumbra. Thus, marginal blood flow in the penumbra and normal blood flow in peripenumbra would tend to limit movement of potassium from ischemic core into normal cortex. Movement of extracellular potassium into the vasculature would also explain the loss of total brain potassium reported by others (Schuier and Hossmann, 1980; Watanabe et al., 1977; Bremer et al., 1978). It has been reported that an increase in extracellular potassium exacerbates transport of sodium from blood to brain (Schielke et al., 1991), thus facilitating cellular depolarization, edema, and infarction.
In conclusion, we originally suspected that [K+]o in penumbral and perhaps peripenumbral cortex might continue to increase with time after MCAO due to diffusion of potassium from the core or from further release of potassium from cellular elements influenced by high [K+]o in the nearby core. Rather, we observed a decreased in [K+]o in cortex adjacent to the core with time after MCAO. The data indicate that changes in penumbral steady state [K+]o may not predict well the evolution of tissue damage in focal ischemia.
