Abstract
Little is known about how ischemia affects hemodynamic responses to neural activation in the brain. We compare the effects of a motor activation task and a cerebral vasodilating agent, acetazolamide (ACZ), on regional cerebral blood flow (rCBF) in primary sensorimotor cortex (PSM) in six patients with major cerebral artery steno-occlusive lesions without paresis of the upper extremities. Quantitative rCBF was measured in all patients using H215 O autoradiographic method and positron emission tomography. The CBF was determined at rest, during a bimanual motor activation task, and 10 minutes after ACZ administration. With bimanual motor activation, rCBF increased significantly in both PSM compared with at rest (P < 0.01 on lesion side, and P < 0.02 on contralateral side). However, rCBF did not increase after ACZ injection in the PSM on the lesion side, whereas rCBF increased significantly in the contralateral PSM after ACZ injection compared with the level at rest. This result suggests that despite a decreased hemodynamic reserve, there is a nearly normal flow response to neural activation, indicating that the mechanism of vasodilation responsible for perfusion change is different for acetazolamide and neural activation. The relations among neural activation, hemodynamic status, and cerebral metabolism in the ischemic stroke patients are discussed.
Keywords
In chronic ischemic stroke patients with major cerebral arterial steno-occlusive lesion, decreased hemodynamic reserve often is present with only minimal clinical symptoms (Baron et al., 1981; Gibbs et al., 1984; Yamauchi et al., 1990). In these patients, computed tomography or magnetic resonance (MR) imaging reveals only small or no cerebral lesions, but the CBF studies using positron emission tomography(PET), single-photon emission computed tomography, and stable xenonenhanced computed tomography with acetazolamide (ACZ) challenge demonstrate wider areas of hemodynamic compromise beyond the limits of the visible lesions on computed tomography or MR imaging (Vorstrup et al., 1986; Hirano et al., 1994; Nariai et al., 1995).
Many functional brain mapping studies have been performed in normal volunteers and patients with various cerebral diseases by using these modalities. In functional studies with PET, the principle is based on evidence that regional blood flow in the brain is tightly coupled to the local metabolic demand and hence is sensitive to the regional neuronal activity (Kuschinsky and Wahl, 1978; Siesjö, 1984). Although coupling between CBF and cerebral oxygen metabolism has been questioned under conditions of activation by Fox and Raichle (1986), the linkage of activation-related increase of CBF to cerebral metabolism of glucose is maintained (Fox et al., 1988b; Ginsberg et al., 1988). Little is known about the characteristics of neural activation in the brains of patients with impaired autoregulatory mechanisms who nevertheless have no neurologic symptom. In this study, we compare the effects of a motor activation task and the cerebral vasodilating agent acetazolamide (ACZ) on CBF in primary sensorimotor cortex (PSM) in patients with chronic ischemic stroke to clarify whether vascular responses to cerebral activation occur when the hemodynamic reserve is impaired.
METHODS
Subjects
We studied six patients (five men and one woman, all right-handed), aged 52 to 75 years(mean, 63.2 years). All had unilateral internal carotid artery (ICA) or middle cerebral artery (MCA) steno-occlusive lesion confirmed by angiography (Table 1). The T1- and T2-weighted MR imaging was performed in all patients within 2 weeks of performing this study. All MR images were 5-mm thick parallel to the orbitomeatal (OM) line. Four of the patients (Cases 1, 2, 4, and 6) had a transient ischemic attack with minimal hemiparesis about 2 months previously, and MR imaging revealed small anterior watershed zone infarction or subcortical lacunae. Angiography confirmed unilateral ICA occlusions. Another patient (Case 3) had a minor stroke and an infarct in the anterior cerebral artery (ACA) area about 10 years previously with right lower extremity weakness. He then had a transient ischemic attack (numbness of the right upper extremity) 3 months before entry into the study without new lesions on MR imaging. Angiography demonstrated severe stenosis (greater than 80%) of the right MCA trunk and a branch occlusion (pericallosal artery) of the ACA. This branch occlusion was deemed responsible for the old infarct in the ACA territory on MR imaging. Case 5 presented with left-sided amaurosis fugax without infarction on MR imaging, and was found to have a left ICA occlusion. Thus, transient ischemic attack in all patients occurred at least 2 months before their PET study. None of the patients had weakness in the upper extremity when the study was carried out. Written informed consent was obtained from all patients.
Subject profiles

F, female; M, male; Lt, left; Rt, right; MRI, magnetic resonance imaging; TIA, transient ischemic attack; AW, anterior watershed; ICA, internal carotid artery; MCA, middle cerebral artery; ACA; anterior cerebral artery.
Protocol
Quantitative rCBF was measured in all patients using the oxygen-15-labeled water (H215O) autoradiographic method and PET (Kanno et al., 1987). The CBF was determined at rest, during a motor activation task using both hands, and 10 minutes after ACZ administration.
The motor activation task consisted of repetitive opening and closing of both hands at a frequency of two times per second, paced with a metronome. The metronome, which was placed over the patient's head 1 m from the ears, began to beat 30 seconds before injection of the isotope. The motor task began 15 seconds before isotope injection, continuing for 2 minutes during scanning. Patients were instructed about the task and were allowed to briefly practice before scanning. Thirty minutes after the motor activation scan, ACZ (1 g in 10 mL distilled water) was injected intravenously over 1 minute. A post-ACZ scan was started 10 minutes after ACZ administration. Arterial blood gases were measured at rest, immediately after motor activation scan, and immediately before ACZ scan (10 minutes after ACZ injection). Systemic blood pressure was monitored on the lower leg during each scan throughout the study and expressed as mean arterial blood pressure.
Cerebral blood flow measurements
A HEADTOME IV (Shimadzu Co., Kyoto, Japan) PET scanner was used for CBF measurement (Iida et al., 1989; Gambhir et al., 1997). The scanner consists of four rings to provide seven tomographic images (four direct slices and three cross-slices). The gantry z-motion is available to allow the axial interpolation scan, so that a total of 14 images are obtained. The scanner had a center-to-center slice distance of 6.5 mm with z-motion and effective resolution of 4.5 mm in a full width at half maximum in the center of the field of view and 5.0 mm in a full width at half maximum at 10 cm from the center. The patient's head was fixed and both eyes were closed during the experiment. The right radial artery and left cubital vein were catheterized for arterial blood sampling and isotope tracer injection, respectively. The CBF was calculated by an autoradiographic method (Kanno et al., 1987) using a lookup table procedure over a 2-minute accumulation period after the intravenous injection of 1480 MBq of H215O at rest, during motor activation task, and after ACZ injection.
Data analysis
We analyzed images in four tomographic planes 42.5, 62, 81.5, and 88 mm above and parallel to the OM line (i.e., the level of the basal ganglia, the level of centrum semiovale, and two levels of the cortical surface including the primary sensorimotor cortex, respectively). The regions of interest (ROI) in the levels of basal ganglia and the centrum semiovale included the territories of the ACA, MCA, posterior cerebral artery, and the watershed areas between the ACA and MCA (anterior watershed) and between the MCA and posterior cerebral artery (posterior watershed). Circular ROI, numbering 18 to 20 and containing 30 pixels each, were drawn over the gray matter of each cerebral hemisphere for each arterial territory. Irregular ROI of the PSM were drawn on the two superior planes containing 36 to 52 (mean 42) pixels each. The MR images parallel to the OM line, 5-mm thick with a 1-mm gap, were obtained. The PET images 13 mm (direct slice) or 11 mm (cross slice) in thickness also were obtained parallel to the OM line, and the center-to-center slice distance was 6.5 mm in our study. Thus, the distance from the OM line was calculated in each image. Because the image thickness differed in PET and MR imaging, the closest MR image was chosen as a reference MR image. The anatomical localization was identified by the atlas of Talairach and Tournoux (1988) and Kretschmann and Weinrich (1986). The ROI of each arterial territory were placed on axial PET images at rest using the atlas and reference MR images. The ROI of the PSM were placed on PET images of the motor task. The same ROI were automatically superimposed at the same loci on the other trial images. The patients' head movement was minimal in all of the studies, so no correction for displacement was performed. In three patients (Cases 2, 3, and 4) with infarcts in cerebral cortex, ROI corresponding to the infarct area were excluded from analysis by comparing PET images with MR images. The rCBF was calculated as the average of the ROI in each area.
For global normalization of rCBF in PSM during motor activation (Ramsay et al., 1993; Shimosegawa et al., 1995), the following equation was used:
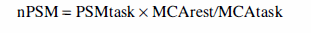
where nPSM is rCBF in PSM during motor task after global normalization; PSMtask is rCBF during motor task in PSM; MCArest is rCBF at rest in MCA; and MCAtask is rCBF during motor task in MCA.
In this equation for global normalization, we chose rCBF in the MCA area as an index of global CBF.
Statistical analysis
The rCBF values of each condition were compared in each brain area by two-factor factorial analysis of variance, and the systemic parameters were compared by one-factor factorial analysis of variance. The rCBF in PSM during motor task after global normalization(nPSM) was compared with rCBF in PSM at rest by paired t test on each lesion side and the contralateral side, respectively.
RESULTS
Illustrative cases
The reference T1 MR imaging and PET-CBF images of the triple measurements(rest, motor activation task, and post-ACZ injection) are illustrated (Figs. 1, 2 and 3). In Case 1 with right ICA occlusion (Fig. 1), a small white matter infarct in the right frontal lobe (anterior watershed area) was revealed on T1 MR imaging. In this case, CBF was slightly decreased in almost the entire right hemisphere (lesion side) at rest. The rCBF in both of the PSM was increased by bilateral motor activation. However, CBF in the lesioned hemisphere did not increase after intravenous injection of ACZ, whereas CBF in the normal hemisphere increased dramatically. In Case 2 with left ICA occlusion (Fig. 2), a small cortical infarct in the left frontal anterior watershed zone was demonstrated on MR imaging. The resting CBF image demonstrated mild hypoperfusion in most of the lesioned hemisphere. Motor-induced activation of both PSM occurred. After ACZ injection, however, CBF in the lesioned hemisphere did not increase. In Case 3 (Fig. 3), an old right ACA area infarct was demonstrated on T1 MR imaging. In the resting CBF image, this infarct was shown as a defect of isotope uptake. The lesioned hemisphere also showed mild hypoperfusion at rest. Both PSM were activated by the motor task. However, CBF in the lesioned hemisphere did not increase significantly after ACZ. The same findings were seen in the other three cases.
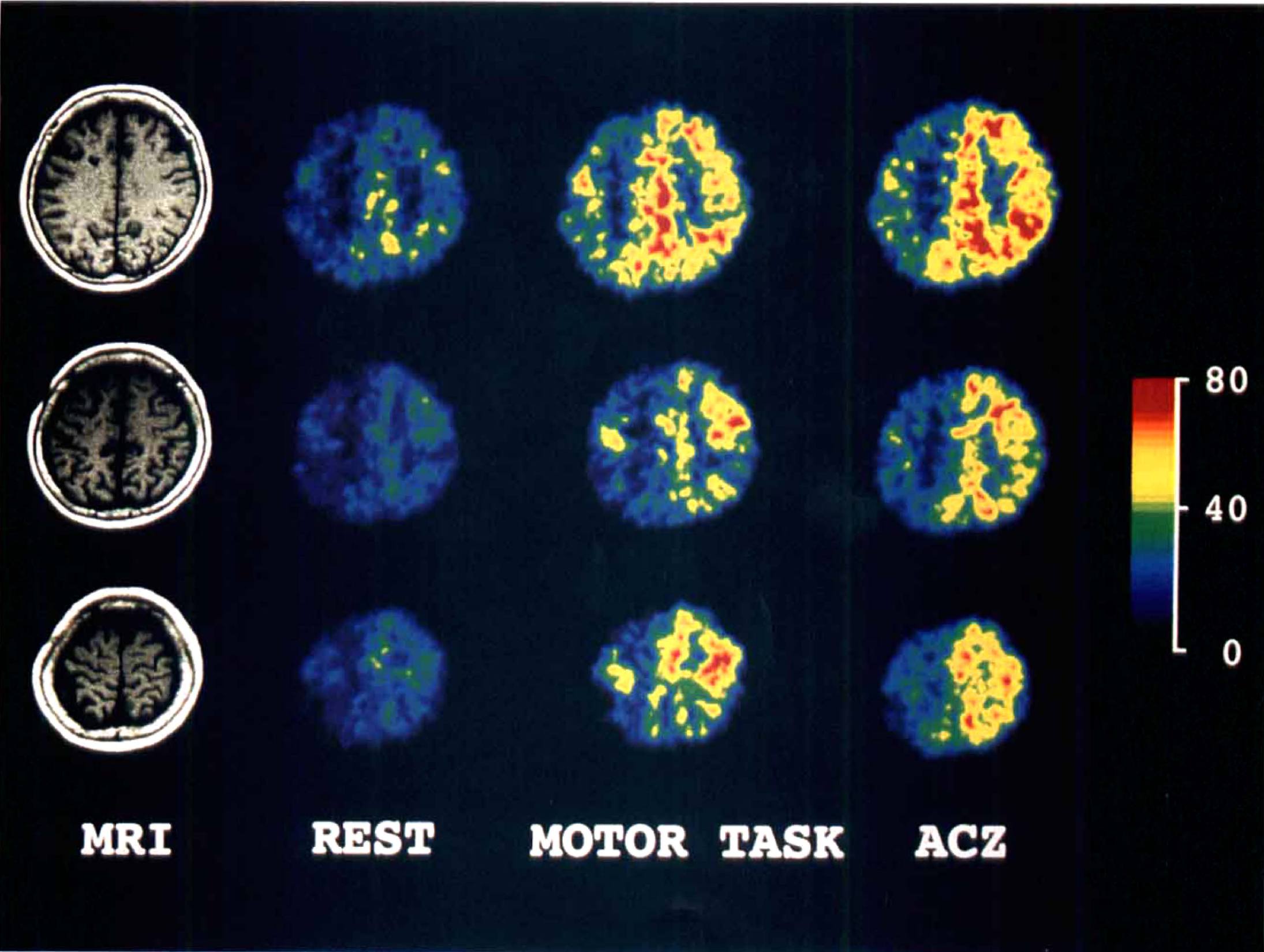
Reference T1 MR imaging and PET-CBF images of the triple measurements (rest, motor activation task, and post-acetazolamide [ACZ] injection) are illustrated. In Case 1 with right ICA occlusion, a small white matter infarct in the right frontal lobe is revealed on T1 MR imaging. The CBF was slightly decreased in almost the entire right hemisphere at rest. Regional CBF values in both primary sensorimotor cortices (PSM) were increased by the bilateral motor activation. However, CBF in the lesioned hemisphere did not increase after intravenous injection of ACZ, whereas CBF in the normal hemisphere increased dramatically. The scale of flow values indicates mL/100 g/min.
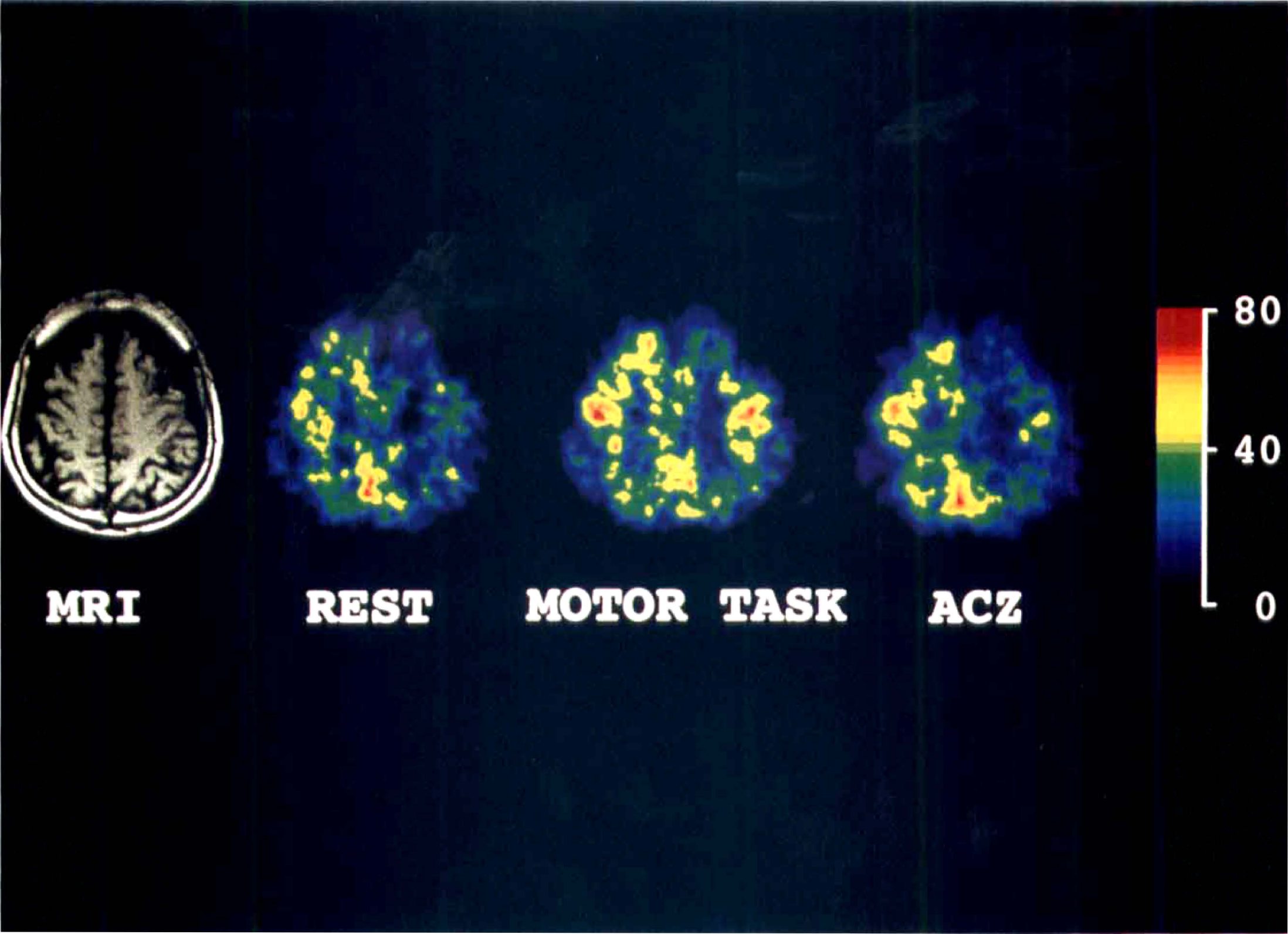
In Case 2 with left ICA occlusion, a small cortical infarct in the left frontal anterior watershed zone was demonstrated on MR imaging. The resting CBF image demonstrated mild hypoperfusion in almost the entire lesioned hemisphere. Motor-induced activation of both PSM occurred. After ACZ injection, however, CBF in lesioned hemisphere did not increase. The scale of flow values indicates mL/100 g/min.
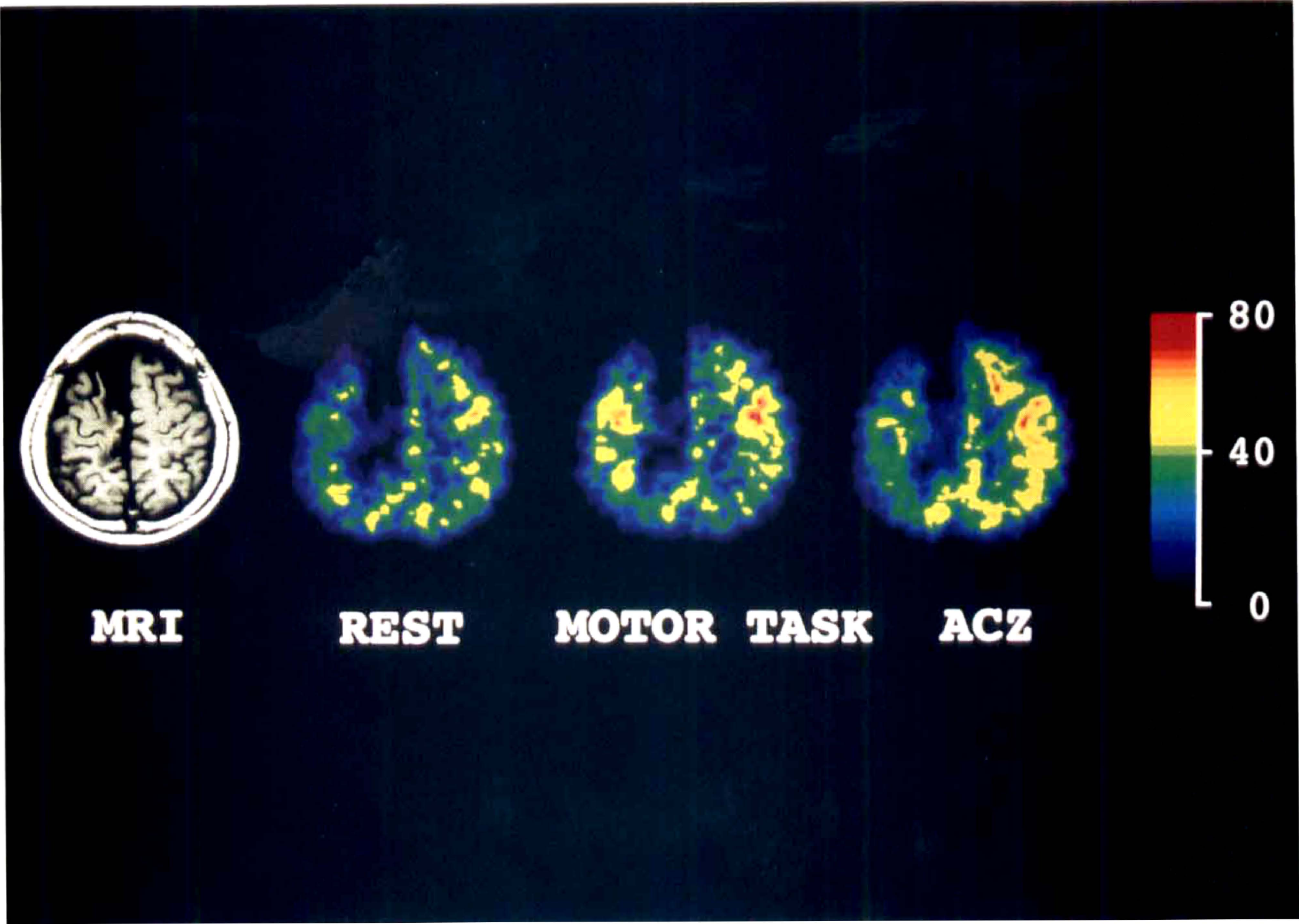
In Case 3, an old right ACA area infarct was demonstrated on T1 MR imaging. In the resting CBF image, this infarct area was shown as a defect of isotope uptake. The lesioned hemisphere also showed mild hypoperfusion at rest. Both PSM were activated by the motor task. However, CBF in the lesioned hemisphere did not increase significantly after ACZ. The scale of flow values indicates mL/100 g/min.
Changes in regional cerebral blood flow and systemic parameters
Systemic blood pressure did not significantly vary throughout the study. Arterial blood gas data did not change significantly between the resting state, immediately after the motor task, and 10 minutes after ACZ injection (Table 2).
Physiologic parameters
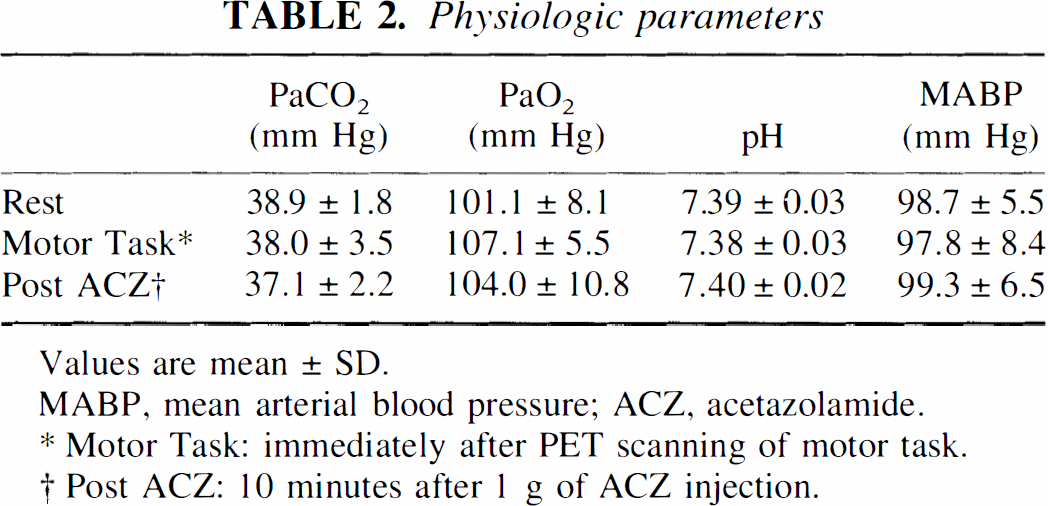
Values are mean ± SD.
MABP, mean arterial blood pressure; ACZ, acetazolamide.
Motor Task: immediately after PET scanning of motor task.
Post ACZ: 10 minutes after 1 g of ACZ injection.
Table 3 summarizes the mean rCBF for various ROI in the three different conditions. The values of rCBF in PSM on motor activation after normalization (nPSM) also are shown. The rCBF values in all areas of the lesioned hemispheres at rest are lower than those in the contralateral hemisphere. But these differences are not significant except in ACA, in which periinfarct lesions were included. Although rCBF increased in all areas with the motor activation task both on the lesioned and the contralateral sides, only the rCBF in both PSM increased significantly from rest by the motor task. After ACZ injection, rCBF values in ACA, anterior watershed, MCA, and PSM of the lesioned hemisphere did not increase significantly compared with rest or the motor activation values. In the normal hemisphere after ACZ injection, the rCBF increased significantly in all areas compared with at rest. Normalized rCBF increased significantly from rest in both PSM with the motortask.
Regional cerebral blood flow (mL/100 g · min)
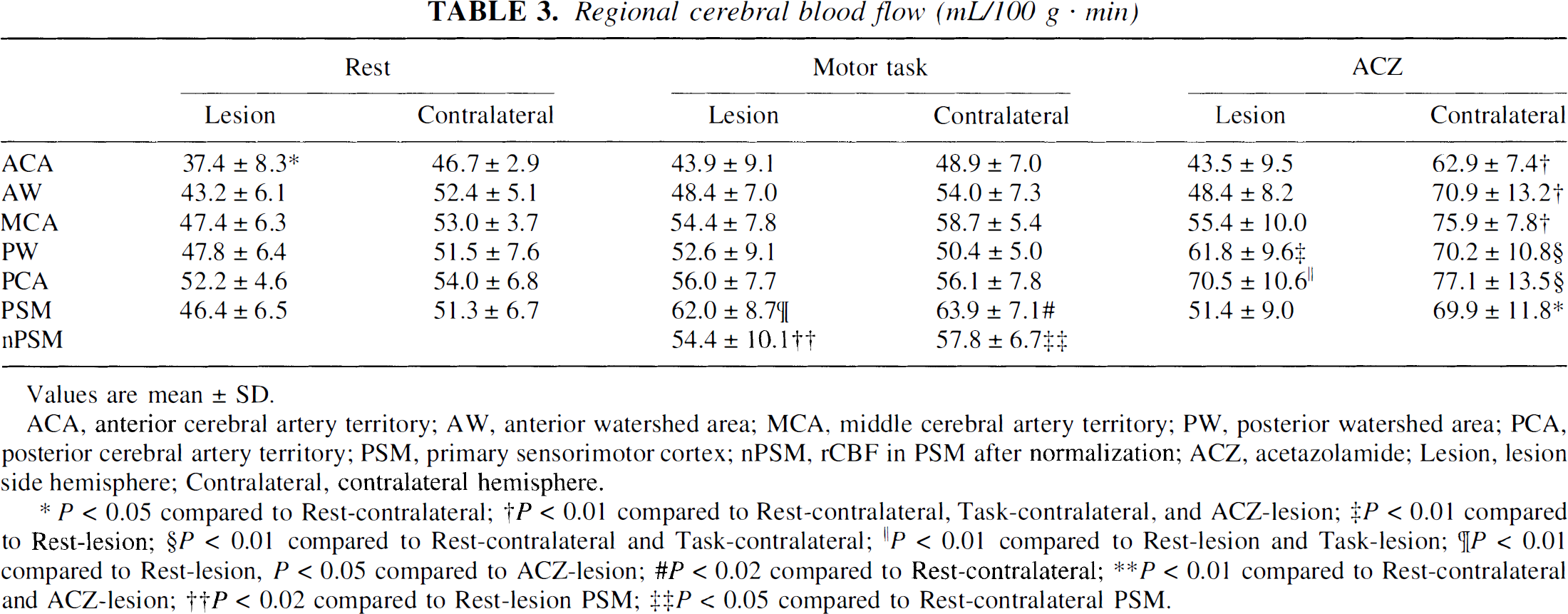
Values are mean ± SD.
ACA, anterior cerebral artery territory; AW, anterior watershed area; MCA, middle cerebral artery territory; PW, posterior watershed area; PCA, posterior cerebral artery territory; PSM, primary sensorimotor cortex; nPSM, rCBF in PSM after normalization; ACZ, acetazolamide; Lesion, lesion side hemisphere; Contralateral, contralateral hemisphere.
P < 0.05 compared to Rest-contralateral;
P < 0.01 compared to Rest-contralateral, Task-contralateral, and ACZ-lesion;
P < 0.01 compared to Rest-lesion;
P < 0.01 compared to Rest-contralateral and Task-contralateral;
P < 0.01 compared to Rest-lesion and Task-lesion;
P < 0.01 compared to Rest-lesion, P < 0.05 compared to ACZ-lesion;
P < 0.02 compared to Rest-contralateral;
P < 0.01 compared to Rest-contralateral and ACZ-lesion;
P < 0.02 compared to Rest-lesion PSM;
P < 0.05 compared to Rest-contralateral PSM.
In Fig. 4, absolute values of rCBF in both PSM are summarized for the three different conditions. Motor activation increased rCBF in both PSM significantly compared with at rest (P < 0.01 on lesioned side, and P< 0.02 on contralateral side). However, rCBF of the PSM on the lesioned side did not increase after ACZ injection, whereas rCBF in the contralateral normal PSM increased significantly after ACZ injection compared with rest.
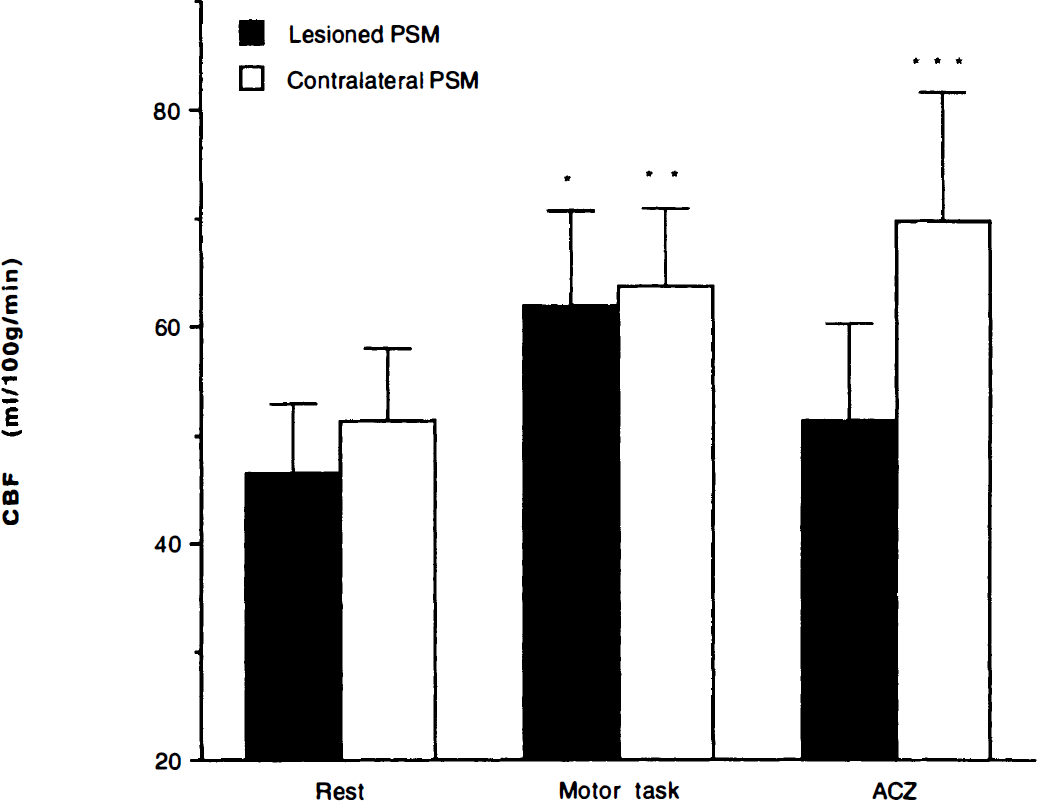
Absolute values of rCBF in both PSM are summarized for the three different conditions. Motor activation increased rCBF in both PSM significantly compared with at rest (*P < 0.01 in lesioned side and **P < 0.02 in contralateral side). However, rCBF of the PSM in the lesioned side did not increase after ACZ injection, whereas rCBF in the contralateral normal PSM increased significantly (***P< 0.01) after ACZ injection compared with at rest. Solid bar, lesioned PSM; hollow bar, contralateral PSM. ACZ, acetazolamide; PSM, primary sensorimotor cortex; rCBF, regional cerebral blood flow.
DISCUSSION
In brain tissue with an insufficiency of cerebral perfusion pressure associated with major cerebral artery steno-occlusive lesions, the vascular response to a vasodilator is expected to be reduced because of compensatory and preexisting autoregulatory vasodilation (Norrving et al., 1982). Thus, the vasodilatory capacity has been widely accepted as an essential parameter for evaluating hemodynamic insufficiency (Vorstrup et al., 1986). It might be thought that activation-related increases of rCBF in brains with such hemodynamic insufficiency also would be reduced because of the preexisting vascular dilation. However, our study clearly demonstrates that primary sensorimotor cortex in the hemodynamically compromised hemisphere, identified by acetazolamide challenge, can be activated to a nearly normal extent.
Hemodynamic status and neuronal function
Numerous reports describe the relation between hemodynamic function and oxygen metabolism in cerebral ischemia with no evidence of infarction. Frackowiak (1985) reports the relation between tissue blood supply and cerebral metabolic demand in ischemia. Powers and coworkers (1987, 1991) further explain that the cerebral oxygen metabolism was maintained in the face of decreased cerebral perfusion pressure by local compensatory mechanisms, including intraparenchymal vasodilation (stage I) and increased cerebral oxygen extraction fraction (OEF) (stage II). Herold and et al. (1988) and Kanno et al. (1988) report on the significant inverse linear relation between CO2 reactivity and OEF in ischemic brain, whereas Hirano and others (1994) showed that reduced ACZ reactivity corresponded to elevated OEF. These results indicate that reduced chemical vasodilatory capacity, as determined by CO2 or ACZ responsiveness, may represent stage II of hemodynamic compromise, in which decreased CBF, increased cerebral blood volume, and increased OEF (partial exhaustion of cerebral oxygen reserve) keep the cerebral oxygen metabolism constant. Although all of our cases had only mild reduction of resting CBF in both MCA territory and PSM in the lesioned hemisphere, ACZ responsiveness was significantly reduced. Thus, they might have had stage II hemodynamic failure, although the OEF was not measured in our cases.
Baron and colleagues (1981) called such a situation the misery-perfusion syndrome. They noticed that in the brain with misery-perfusion syndrome, the neuronal oxygenation, and hence function, could range from normal to inadequate, depending on the level of reduced CBF level and the extent of the compensation by increased OEF. In our cases, the activation-related increase of CBF was nearly normal, indicating that the neuronal function and thus the demand of blood flow were maintained within the nearly normal range. This was matched with clinical signs; that is, all of the patients had no paresis on the upper extremities. However, closer scrutiny of the present data indicates that the absolute values of rCBF on motor activation in both MCA territory and PSM in the lesioned hemisphere were lower than those of the contralateral hemisphere, although this difference was not significant. We consider that this lesser increase after neural activation in the affected hemisphere was partly caused by the decreased number of neurons resulting from ischemia. Recent reports (Hatazawa et al., 1995; Nakagawara et al., 1997) have identified lesions with hemodynamic insufficiency with central benzodiazepine receptor imaging. The results suggest the loss of a limited number of neurons in reduced perfusion areas that appear intact on computed tomography or MR imaging.
Vascular response and acetazolamide
Although the vasculature is thought to be submaximally dilated in stage II hemodynamic failure, our data show that the vasculature has an ability to dilate to a large extent with neural activation. Kanno et al. (1988) estimated the theoretical point of exhaustion of the capacity for vasodilation in chronic ischemic patients. They indicated that the metabolic reserve was used before complete exhaustion of the perfusion reserve, since the elevation of OEF and decrease of the vascular response to CO2 occur in parallel. Therefore, in our case with stage II hemodynamic failure, some vasodilatory capacity remains. From the viewpoint of the microcirculation, Gjedde and colleagues (1990) report that in human brain study using PET, the functional capillary density was reduced after the ischemic stroke, and that this persisted even when the adequate tissue perfusion returned. The same research group (Kuwabara et al., 1992) also demonstrated that in the normal subject during the functional activation, the capillary density increased in proportion to the change of blood flow. These results might explain the current data that the mild hypoperfusion at rest as well as the lesser increase of CBF on the neural activation in our cases might be related to the reduction of the functional capillary density. Of course, the hypoperfusion at rest also is directly related to the occlusive lesion of the major arteries.
Although the true mechanism of the vasodilatory effect of ACZ is still unknown, it has been widely used to investigate cerebral vasodilatory capacity of the patients with ischemic stroke. Inhibition of carbonic anhydrase is the only well-known action of ACZ (Maren, 1967). Carbonic anhydrase is mainly located in the erythrocyte (Maren, 1967) and also is demonstrated in the glial cells (Giacobini, 1962) and the endothelium of the capillaries in the brain tissue (Ridderstrale et al., 1985). Inhibition of carbonic anhydrase in circulating red blood cells or the blood vessels walls interferes with CO2 clearance from the brain to circulating bicarbonate. Subsequently, increased local levels of CO2 around cerebral vessels might cause cerebral vasodilation. On the other hand, a direct action on vascular smooth muscle, independent of inhibition of carbonic anhydrase, also has been suggested (Hauge et al., 1983). Rapid vasodilatory action of ACZ within a few minutes supports a direct vasodilatory action. However, our previous experimental work denied the direct effects of ACZ on isolated cerebral arterioles (Mori et al., 1996). Although the extracellular pH was reduced after intravenous administration of ACZ, its effect on the cerebral vasculature may not be through a change of cerebral tissue metabolism because cerebral intracellular pH and CMRO2 never change after ACZ administration (Vorstrup et al., 1984; Bickler et al., 1988; Vorstrup et al., 1989; Inao et al., 1994). Acetazolamide might dilate cerebral vessels by increasing CO2 only locally around the vascular smooth muscle through inhibition of carbonic anhydrase. As ACZ is transported by the circulating blood to the local area on the effective site, the effect of ACZ might be flow dependent. We believe that the reduced cerebrovascular reactivity to ACZ in hemodynamic insufficiency is caused by a delayed or lesser delivery of ACZ to the affected area because of hypoperfusion (Inao et al., in press).
Methodologic issues
In the study protocol, we chose a frequency rate for hand movement of two per second, following Sadato and others (1996), who report that the maximum rCBF increase in PSM was achieved by a high-frequency motor task (more than two per second). We also estimated the ACZ effect on rCBF 10 minutes after ACZ injection. This time interval is known to result in a maximum effect of ACZ on CBF (Inao et al., 1997; Gambhir et al., 1997; Inao et al., in press). In the activation study, the normalization by global CBF is necessary to allow quantitative comparison of the relative regional increase in blood flow induced by selective stimulation (Fox et al., 1988a; Ramsay et al., 1993). In our study, rCBF in the MCA area was chosen as an index of global CBF because the MCA territory in all six patients under study was free of infarction. In our study, we placed the circular ROI containing 30 pixels on the gray matter of each cerebral hemisphere for each arterial territory. Actually, the diameter of this circle is about 12.5 mm. The pixel counts of ROI in the PSM (mean 42 pixels) were larger than that of each arterial territory. Because the size of ROI we placed is two times larger than that of full width at half maximum(4.5–5.0 mm), we believed that the spatial resolution of our scanner sufficed to detect the CBF changes after the neural activation. Moreover, each activated area on the PET image was identical to the anatomical localization of the PSM on the atlas.
CONCLUSION
In conclusion, the vasculature can be dilated to a nearly normal extent by the neural activation despite an impaired hemodynamic reserve, suggesting that the cerebral metabolism, function, and the local vasodilatory response are maintained within the nearly normal range. Our data also suggest that the mechanism of vasodilatory response differs for ACZ and neural activation. Reduced vasoreactivity to ACZ in the hemodynamically compromised brain might not be caused by the preexisting compensatory vasodilation, but less distribution of ACZ through the circulating blood from the hypoperfusion.
Footnotes
Acknowledgements
The authors thank Dr. R. S. J. Frackowiak for his kind advice and comments concerning this study, as well as Drs. K. Ichimi and M. Shibayama and the staff of the PET section for their technical assistance. The authors also thank Mr. T. Sato and Mr. S. Morimoto of Shimazdu Co. for their valuable assistance with the illustration.
