Abstract
The authors investigated the gene expression of the NR2A and NR2B subunits of N-methyl-D-aspartate (NMDA) receptor and the functional electrophysiologic activity of NMDA receptor complexes in the vulnerable CA1 and less vulnerable dentate gyrus subfields of the rat hippocampus at different times after transient cerebral ischemia. Decreased expression for both subtypes was observed in both the CA1 subfield and dentate granule cell layer at early times after challenge; however, the decreased expression in the dentate granule cell layer was reversible because mRNA levels for both the NR2A and NR2B subtypes recovered to, or surpassed, sham-operated mRNA levels by 3 days postchallenge. No recovery of expression for either subtype was observed in the CA1 subfield. The functional activity of NMDA receptor complexes, as assessed by slow field excitatory postsynaptic potentiations (slow f-EPSP) in CA1 pyramidal neurons, was maintained at 6 hours postchallenge; however, this activity was diminished greatly by 24 hours postchallenge, and absent at 7 days postchallenge. A similar pattern was observed for the non-NMDA receptor-mediated fast f-EPSP. In dentate granule neurons, however, no significant change in NMDA receptor-mediated slow f-EPSP from sham control was observed at any time after insult. The non-NMDA receptor-generated fast f-EPSPs also were maintained at all times postinsult in the dentate gyrus. These results illustrate that the activity of NMDA receptors remains functional in dentate granule neurons, but not in the pyramidal neurons of the CA1 subfield, at early and intermediate times after transient cerebral ischemia, and suggest that there is a differential effect of ischemia on the glutamatergic transmission systems in these two hippocampal subfields.
Severe transient cerebral ischemia leads to the insidious delayed death of selective neurons in the brain. The hippocampal formation, a brain structure that is integral to the acquisition and processing of learning and memory (Zola-Morgan et al., 1986; 1992), houses one of the most sensitive—as well as one of the more resistant—populations of neurons to global ischemia. Pyramidal neurons of the CA1 subfield of the hippocampus are a highly vulnerable population of neurons, whereas the granule cells of the dentate gyrus are neurons that primarily are resistant to global ischemia challenge (Kirino, 1982; Pulsinelli et al., 1982; Petito et al., 1987). Although the biochemical events leading to the expiration of the CA1 neurons after challenge remain unclear at present, considerable evidence suggests that misregulated calcium (Ca2+) ions may provide a crucial signal (Benveniste et al., 1988; Scharfman and Schwartzkroin, 1989; Choi, 1994). One site by which Ca2+ ions are known to enter neurons is through the complex of proteins that comprise the N-methyl-D-aspartate (NMDA) subtype of glutamate receptor (Hollmann and Heinemann, 1994, Monyer et al., 1992; Nakanishi, 1992), a system in which overstimulation has been shown to promote neuronal death in several model systems (see Olney, 1978; Choi, 1988; 1994; Mody and MacDonald, 1995).
In our previous investigations of NMDA receptor activity and expression responses to transient cerebral ischemia, we observed a differential pattern of gene expression response between the vulnerable CA1 subfield and the resistant dentate granule cell layer at early times after reperfusion (Zhang et al., 1997). We further demonstrated that in the dentate granule cells, but not in the CA1 subfield, a functional recovery of fast glutamatergic transmission (mediated by non-NMDA receptors) is present at delayed times after reperfusion (Shinno et al., 1997). However, we have not investigated how global ischemia affects the NMDA receptor-mediated component of the excitatory postsynaptic potential in the less vulnerable neurons of the dentate gyrus. Because of the potential involvement of NMDA receptors in the delayed neuronal death of the CA1 neurons, we questioned whether NMDA receptor activity and expression would respond similarly or differently in the less vulnerable dentate granule cells and in the vulnerable CA1 subfield after ischemia challenge. Because the dentate granule cells primarily are resistant to ischemia challenge, any altered response in NMDA, as well as non-NMDA, receptor activity between these subfields could shed light on mechanisms that result in the preservation of the dentate gyrus—but not the CA1 subfield—after cerebral ischemia.
Therefore, the purpose of these investigations was to examine whether NMDA receptors in the vulnerable CA1 subfield respond differently to cerebral ischemia versus those NMDA receptors in the less vulnerable dentate gyrus subfield when compared with equivalent hippocampal subfields in sham-operated control animals. Little currently is known about the functionality of glutamate receptors in dentate granule neurons after ischemia challenge or whether any differences—should they exist—in this less vulnerable hippocampal subfield could account for the resistant nature of these neurons to transient cerebral ischemia. We previously demonstrated that changes in NR2A and NR2B receptor expression occur at early times after challenge, and that in the CA1 subfield, these changes are paralleled by decreases in the functionality of the NMDA receptor complex (Zhang et al., 1997). In the present study, we examined the expression levels of the NMDA receptor subunits NR2A and NR2B, both at the early times previously reported (Zhang et al., 1997) and at later times after reperfusion, and compared the functionality of the NMDA receptor complexes of the CA1 and dentate granule cell subfields at these times after transient cerebral ischemia.
MATERIALS AND METHODS
Animal model
The four-vessel occlusion ischemia model was performed using a modified version of the method of Pulsinelli et al. (1982) and Kirino (1991), as described in detail previously (Hsu et al., 1996; Shinno et al., 1997; Zhang et al., 1997). Briefly, male Wistar rats were anesthetized, and both vertebral arteries were occluded permanently by electrocauterization on day 1. On the second day, both common carotid arteries were re-exposed and occluded with aneurysm clips for 15 minutes. Animals were required to meet the following criteria to be included in the study: 1) complete flat bitemporal electroencephalogram during the duration of the carotid occlusion, 2) maintenance of a dilated pupil and an absence of a cornea reflex due to light stimulation, and 3) consistent body temperature recordings. Animals that did not meet these criteria were excluded. Sham control animals received the same 2-day procedure, except no electrocauterization of the vertebral arteries or carotid artery occlusion occurred.
In situ hybridization
Nonhomologous regions of the N-terminal domain of the NR2A and the NR2B NMDA receptor subunits were isolated as described using the polymerase chain reaction (PCR) (Zhang et al., 1997). The following primers were used in the reactions:
NR2A-sense 5′-GCGCGCAGCACGCCCCATTGCATCC-3′;
NR2A-antisense 5′-GGGCCACAGCCTCCTGGTCCGTGTCA-3′;
NR2B-sense 5′-CCCAGCATCGGCATCGCTGTGATCCTC-3′; and
NR2B-antisense 5′-CATGATGTTGAGCATGACGGAAGCTTG-3′.
Amplified products were subcloned into the PCR-2 cloning vector (Invitrogen), and subclones with an insert orientation that allowed the use of the T7 polymerase for producing an antisense RNA were identified. Antisense riboprobes were generated from gel-purified, polylinker linearized subclones to avoid vector sequence inclusion in the probe, purified over a diethyl pyrocarbonate-treated G-50 column, and used at a final concentration of 5 × 106 to 1 × 107 cpm/mL of hybridization solution, as described (Zhang et al., 1997; Simmons et al., 1989). This concentration was found to be in excess of target sequences and did not produce highly nonspecific background levels.
Animals were anesthetized at the appropriate time after insult and killed by transcardiac perfusion with a 4% paraformaldehyde solution using the method of Simmons et al. (1989), which has been used previously in our studies (Hsu et al., 1996; Zhang et al., 1997). After perfusion, the brain was removed and postfixed at 4°C overnight in 4% paraformaldehyde plus 15% sucrose. Brain sections of 20-μm were obtained by cryostat sectioning and mounted onto subbed poly-L lysine-coated microscope slides. Each individual slide contained a section from a sham-operated and one from an experimental animal that were matched closely in the plane of the dorsal hippocampal area. This allowed direct evaluation within the same hybridization reaction of a control and test section and minimized potential interhybridization errors. Control hybridizations consisted of “sense” probes, sections that were pretreated with RNAse before antisense hybridization, and sections that received only hybridization solution with no probe. None of these conditions generated specific hybridization.
Processed slides were mounted and exposed to XAR-5 film, and analyzed by quantitative densitometry using MCID-3 (St. Catherines, Ontario, Canada). A minimum of eight sections per animal time point in sham control and experimental pairs were used for each evaluation number. A minimum of three animal pairs per time point were investigated. Pixel densities were recorded for each region of the hippocampus, and film background was subtracted. The resulting values did not exceed the linear range of the film, as determined by included 14C standard strips. Densitometric values of experimental animals were normalized to those of matching sham control animals from the same slide and then averaged. Statistical evaluation was done using the Student's paired two-tailed t test across total averaged data pools for each individual time after challenge, as described (Hsu et al., 1996; Zhang et al., 1997).
Electrophysiologic analysis
Control slices were obtained from rats (n = 3) that had recovered from sham surgery, and postischemic slices were collected from animals receiving in vivo reperfusion of 6 (n = 3) and 24 (n = 4) hours and 6 to 7 days (n = 2). Briefly, rats were anesthetized with halothane and decapitated. The brain was removed quickly and maintained in an ice-cold artificial cerebrospinal fluid (ACSF) for 3 to 5 minutes before slicing, as described (Zhang et al., 1991; Shinno et al., 1997; Zhang et al., 1997). Slices were maintained in oxygenated ACSF at room temperature for at least 1 hour before recording and not maintained for more than 6 hours after dissection. The components of ACSF are: 125 mmol sodium chloride (NaCl), 2.5 mmol potassium chloride (KCl), 1.25 mmol monosodium phosphate4, 2 mmol calcium chloride (CaCl2), 1.3 mmol magnesium chloride (MgCl2), 26 mmol sodium bicarbonate (NaHCO3), and 10 mmol glucose. When aerated with 5% carbon dioxide (CO2)/95% oxygen (O2), the pH value of the ACSF is 7.4. Osmolarity of the ACSF is 300 ± 5 mOsmol (Zhang et al., 1991).
For electrophysiologic recordings, slices were transferred to a fully submerged chamber and perfused with the oxygenated ACSF continuously (Zhang et al., 1991) at a perfusion rate of 4 to 5 mL per minute. Extracellular recordings of synaptic field potentials were performed at slice temperatures between 32 and 33°C. Synaptic field potentials were recorded extracellularly using glass pipettes filled with 150 mmol NaCl. The recording pipette was placed in the dendritic field of either the CA1 subfield or the dentate gyrus, and either the Schaffer collateral afferents or the perforant pathway afferents were stimulated electrically using a tungsten bipolar electrode. Electrical stimulation was done through a Grass stimulator (S88, Grass Medical Instruments, Quincy, MA, U.S.A.), and constant current pulses were delivered every 20 seconds via a photoelectric stimulation isolation unit (Grass Medical Instruments). Stimuli at the maximum intensity (duration of 0.1–0.3 ms, 600–800 microAmps) were used to activate fully the synaptic responses (Lynch and Schubert, 1980). Data were collected only if stable field potentials were recorded, and averages were obtained from three to four slices per animal, and each time point included two to four animals.
RESULTS
Gene expression changes after cerebral ischemia for NR2A and NR2B
CA1 subfield. The expression levels for both the NR2A and NR2B receptor subtypes were altered dramatically in the CA1 subfield in a time-dependent manner after challenge (Figs. 1 and 2). Consistent with our previous report, no significant change in either NR2A or NR2B expression was observed in the CA1 subfield at 6 hours after challenge (P > 0.05); however, expression of both subunits was decreased significantly to levels near 50% of sham-operated controls (P < 0.01) at 24 hours postchallenge (Zhang et al., 1997). By day 3 postchallenge, only approximately 15% of the sham control expression remained for NR2A, whereas NR2B expression had changed only marginally from the values observed at 24 hours. By day 7 postchallenge, NR2A expression barely was observed and NR2B expression was found to be only approximately 20% that of sham control levels.
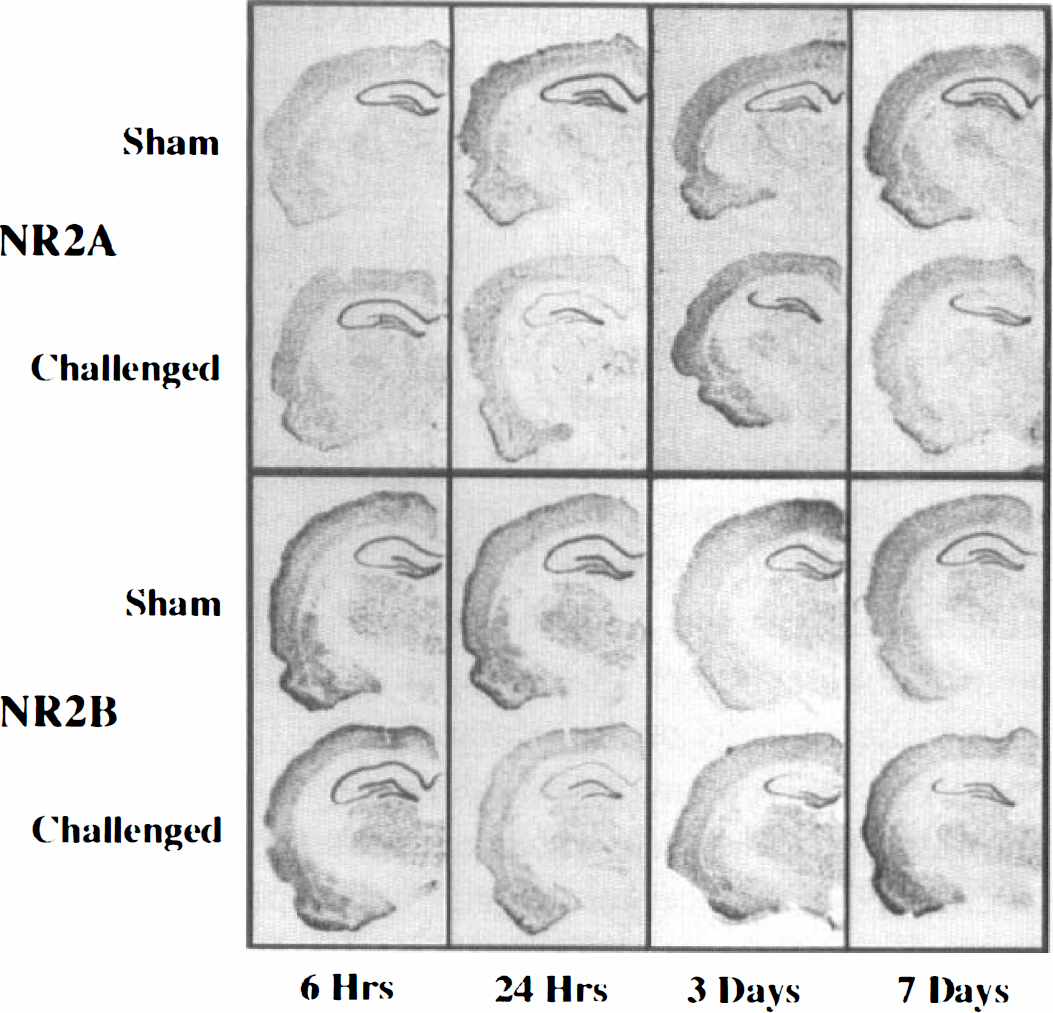
Representative examples of NR2A expression (top panel) or NR2B expression (bottom panel) at different times after transient cerebral ischemia. The top section of each pair originates from a time matched sham control animal, whereas the bottom section is obtained from the global ischemic animal.
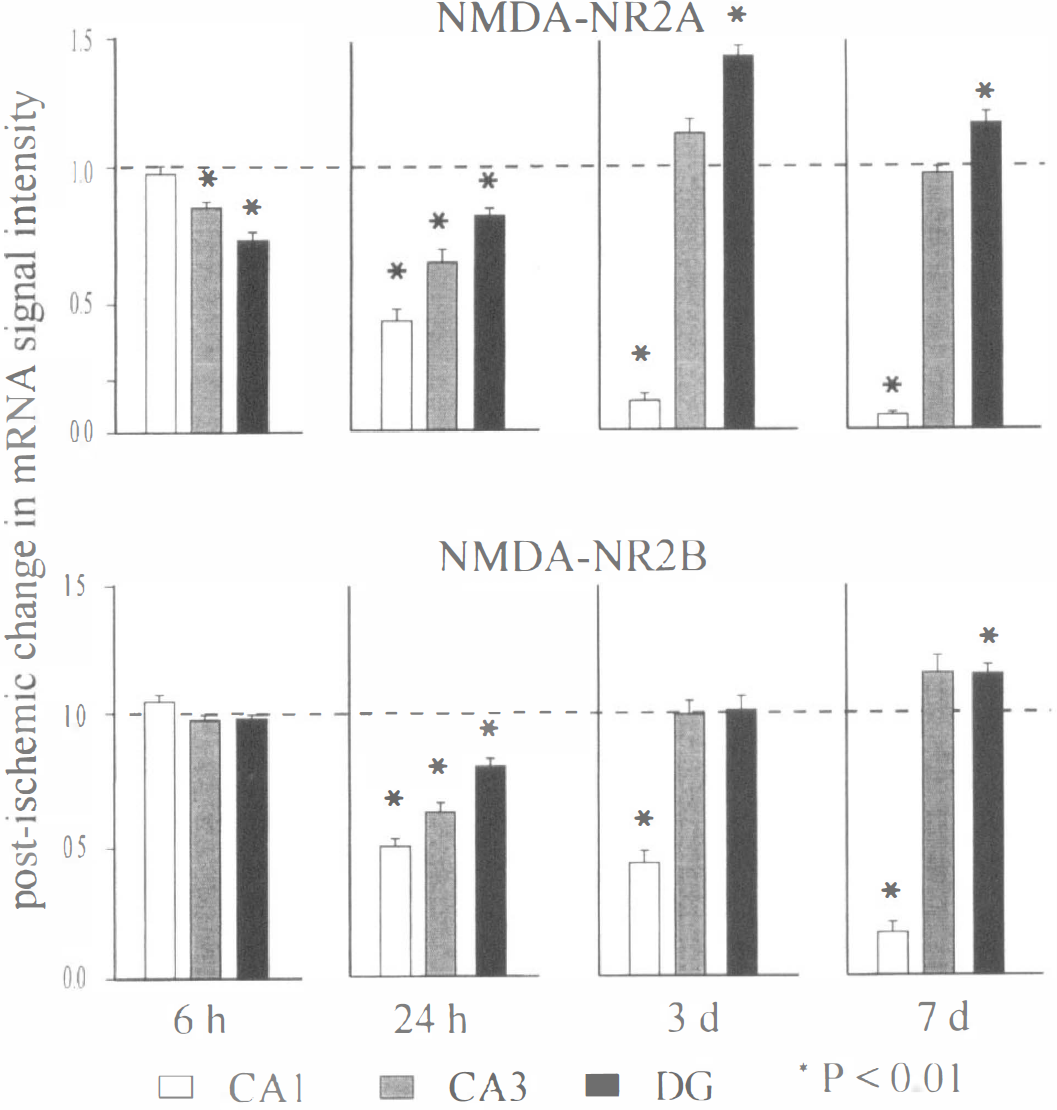
Histogram illustrating the mean and standard error of the densitometric analysis of the antisense in situ hybridization. Analysis consisted of a minimum of four animals for each time point, and at least four paired sections from each sham and ischemic animal time set were obtained. Control expression levels corresponding to sham control sections are represented as a value of 1 by a dashed line, and any changes in expression resulting from global ischemia are shown as a percentage from the control value. Statistical significance is denoted by an asterisk, at levels P < 0.01 (Students paired two-tailed t test). Hippocampal subfields and times postischemia are shown below the histograms.
Dentate granule cell subfield. NR2A and NR2B expression in the granule cell layer of the dentate gyrus displayed the same trend as that observed in the CA1 at early times postchallenge, however, expression levels recovered to—or exceeded—sham levels by 3 days postchallenge. As we previously reported (Zhang et al., 1997), at 6 hours postchallenge, NR2A expression was diminished in the dentate gyrus (P < 0.01); however, no change from sham control was evident for the NR2B subunit (P > 0.05). At 24 hours postchallenge, both NR2A and NR2B expression were slightly, but significantly, diminished from sham control. At days 3 and 7 postchallenge, however, NR2A and NR2B expression either had returned to, or exceeded, the expression levels observed in sham-operated animals (Figs. 1 and 2). NR2A expression at day 3 postinsult was nearly 140% of sham control, and this elevated expression was maintained at day 7 postchallenge at nearly 120%. Although NR2B expression was indistinguishable from sham control at 3 days postchallenge, there was an observed elevation in expression to approximately 120% sham control at day 7 postchallenge.
Postischemia synaptic responses in the CA1 and dentate gyrus
Slow and fast field excitatory postsynaptic potentiations in sham-operated animals. Sham-operated animals were examined 24 hours after surgery to establish the control levels of NMDA receptor activity in both the CA1 subfield and dentate granule neurons, as described previously (Zhang et al., 1997). First, stable fast f-EPSPs were elicited from both the CA1 and dentate granule cell layers in these slices (Table 1, Fig. 3). The slow NMDA receptor component of the f-EPSP then was identified by perfusing the slice with a modified ACSF solution in which MgCl2 was omitted (“Mg2+ free”) (Zhang et al., 1997). Both 6-cyano-7-nitroguinoxaline-2,3-dione and bicuculline were added to this Mg2+-free ACSF to block AMPA/kainate and gamma-aminobutyric acid A receptors. Under these conditions, stimulation of the Schaffer collateral pathway induced a slow dendritic CA1 f-EPSP that subsequently was blocked by the addition of 50 μmol 2-amino-5-phosphonopentanoic acid (AP-5) (Fig. 3). Stimulation of the perforant pathway also induced a slow dendritic f-EPSP in dentate granule neurons that was attenuated similarly by 50 μmol AP-5 (Fig. 3). The sensitivity to AP-5 of both the CA1 and dentate granule cell layer slow f-EPSP recorded in this manner suggests that these slow f-EPSPs primarily are mediated by AP-5–sensitive NMDA receptors (Farrant et al., 1994; Spruston et al., 1995). All values obtained from these recordings were consistent with those observed from the sham-operated animals employed in previous investigations (Shinno et al., 1997; Zhang et al., 1997).
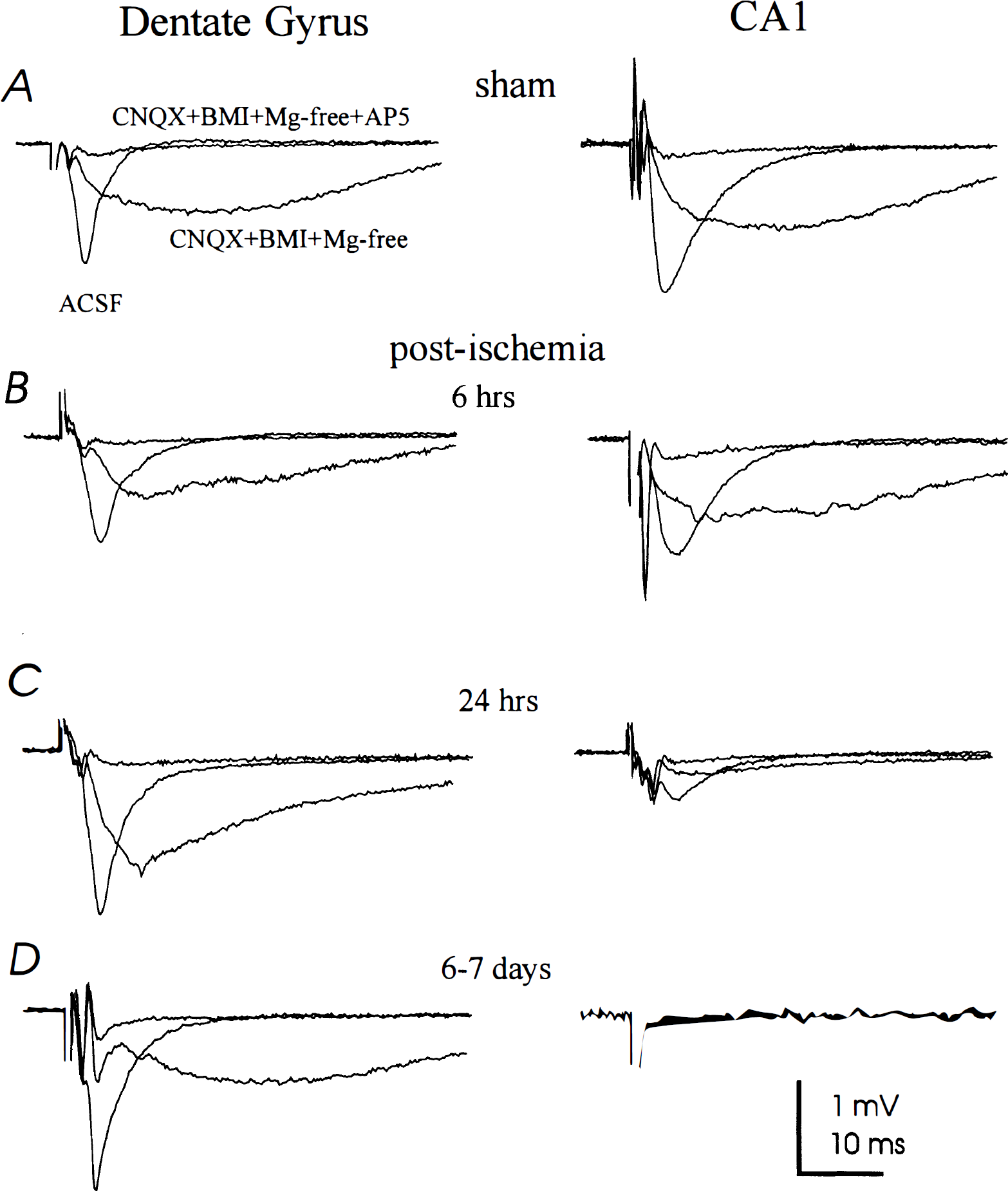
Synaptic field potentials in sham-operated and postischemia slices. Field responses recorded from either the dentate gyrus or the CA1 subfields were collected from slices prepared at 24 hours after sham surgery (
Excitatory postsynaptic potentials recorded in slices obtained from the proximal dendritic regions of the CA1 and dentate granule cell layers of sham-operated or postglobal ischemia animals
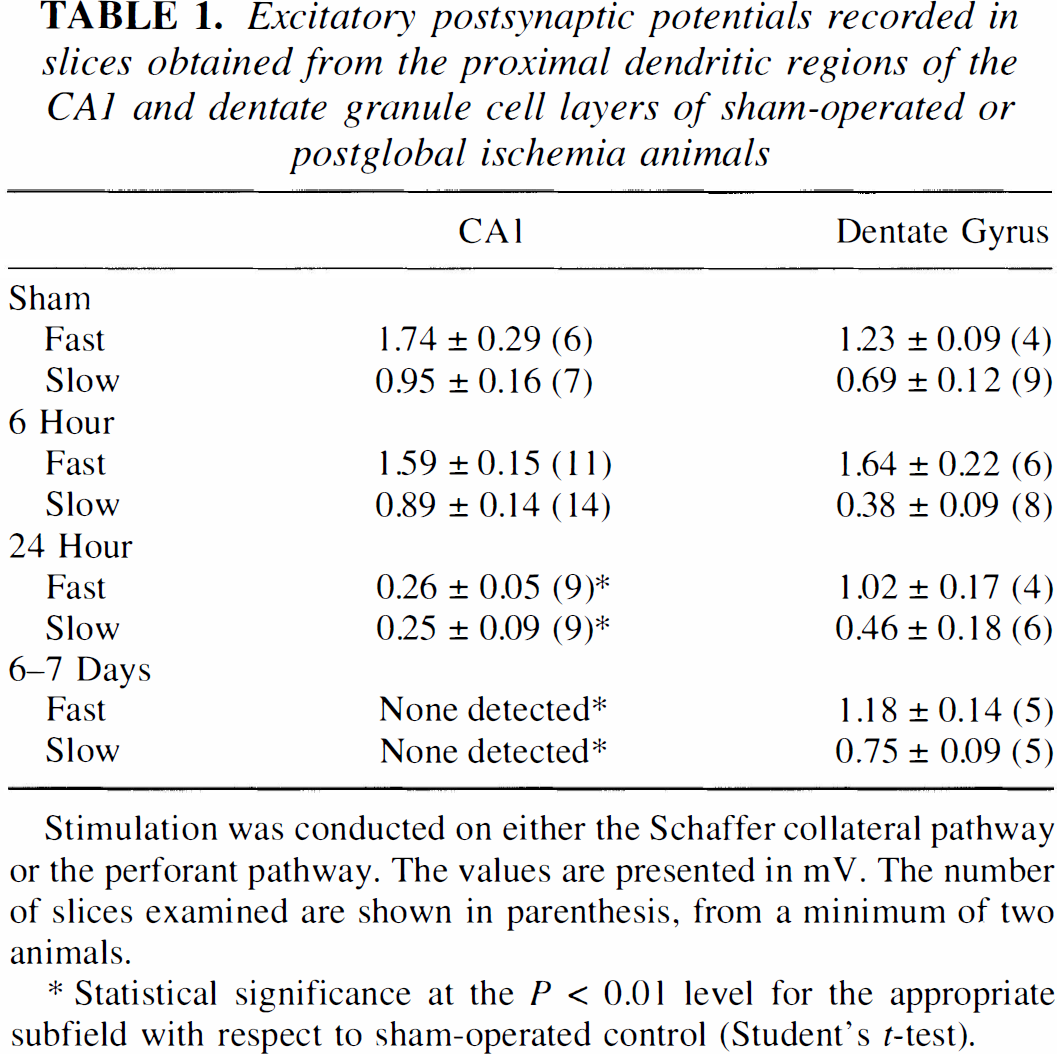
Stimulation was conducted on either the Schaffer collateral pathway or the perforant pathway. The values are presented in mV. The number of slices examined are shown in parenthesis, from a minimum of two animals.
Statistical significance at the P < 0.01 level for the appropriate subfield with respect to sham-operated control (Student's t-test).
Slow and fast field excitatory postsynaptic potentiations 6 hours postischemia. At 6 hours postischemia, maximal stimulation of the Schaffer collateral pathway promoted AP-5–sensitive slow f-EPSPs that did not differ significantly from those slow f-EPSPs recorded from sham controls in either CA1 dendrites or dentate granule cells (Figs. 3 and 4, Table 1). A trend toward a decreased slow f-EPSP level in the dentate granule cells was observed; however, this decrease failed to reach the P < 0.05 level of significance. In regular Mg2+-containing ACSF, the fast f-EPSP was recorded in each hippocampal subfield to demonstrate the viability of the slice and provide a reference tracing for comparison of the slow f-EPSP currents (Fig. 3). Neither the fast f-EPSP in the CA1 nor the fast f-EPSP in the dentate gyrus were significantly different from those values obtained from the corresponding subfield in sham controls at a value of P < 0.05 (Table 1, Figs. 3 and 4).
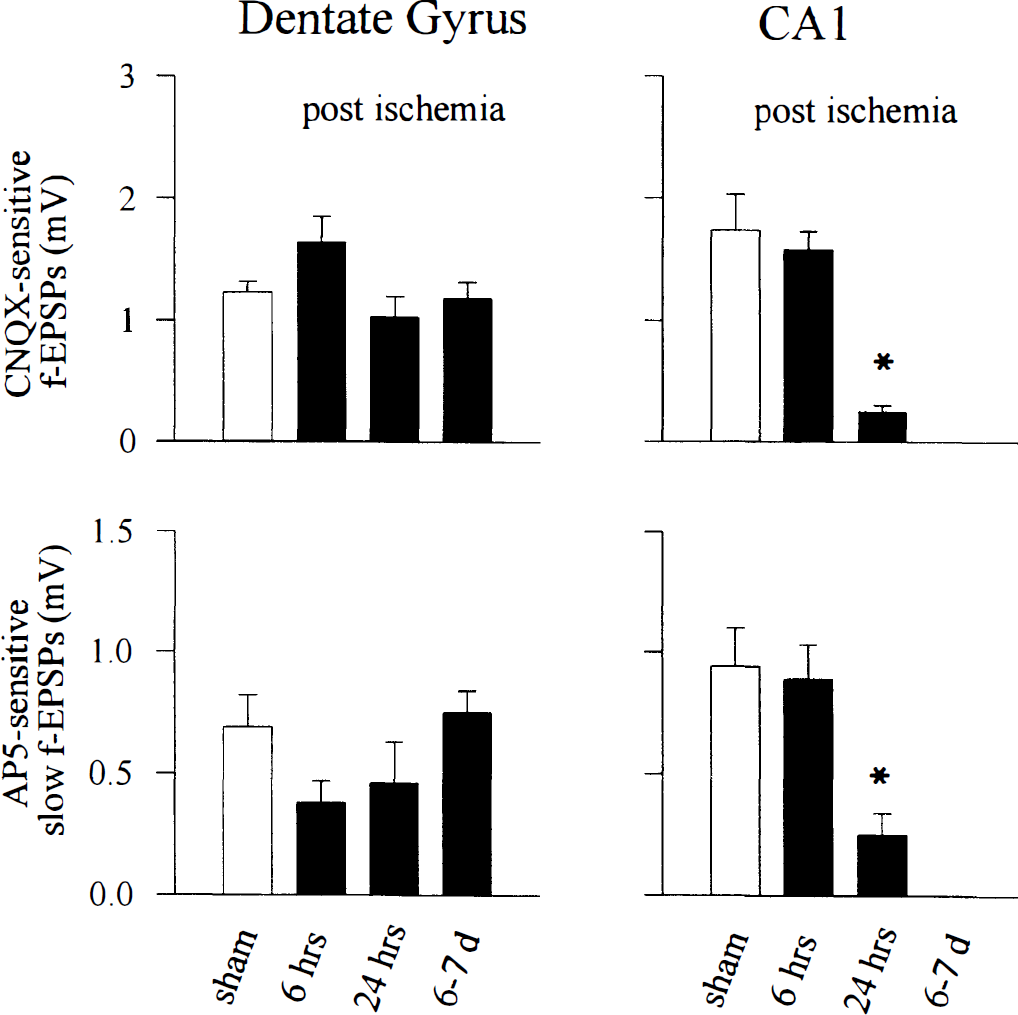
Histogram illustrating the mean and standard error of the elicited field potential responses for dentate gyrus (left) and CA1 subfield (right). CNQX-sensitive responses (top panels) denote fast field excitatory postsynaptic potentiations (f-EPSPs), and AP-5-sensitive responses represent slow f-EPSPs measured in conditions for determining N-methyl-D-aspartate (NMDA) receptor activity (see Methods section and Fig. 3 legend). Statistical significance is denoted by an asterisk, at levels of P < 0.01 for postglobal ischemia vs. sham-operated controls for the same hippocampal subfields (Student's unpaired two-tailed t test). Sham control values and values for postischemia times are shown below the histograms. The corresponding measured values are presented in Table 1. CNQX, 6-cyano-7-nitroquinoxaline-2,3-dione.
Slow and fast field excitatory postsynaptic potentiations 24 hour postischemia. At 24 hours postischemia, the slow f-EPSP recorded from the CA1 dendritic region in Mg2+-free ACSF after maximal stimulation of the Schaffer collateral pathway was decreased significantly from that of sham-operated controls (P < 0.01), consistent with our previous report using different animal subjects (Zhang et al., 1997). This is in contrast to the slow f-EPSP recorded in dentate granule neurons after perforant pathway stimulation, which was maintained at levels that did not significantly differ from sham-operated controls. The fast f-EPSP at this time postinsult was not altered significantly from sham controls in the dentate granule cell layer (P > 0.05), although a significant decrease in fast f-EPSP currents was observed in the CA1 subfield (Figs. 3 and 4), which was consistent with the observations from another study (Shinno et al., 1997).
Slow and fast field excitatory postsynaptic potentiations 6 to 7 days postischemia. Stimulation of the Schaffer collateral pathway of slices obtained from animals between 6 and 7 days postischemia failed to generate any detectable f-EPSP in the CA1 subfield (Figs. 3 and 4, Table 1). This is in contrast to the f-EPSPs recorded from dentate granule cells, where values comparable to those of sham-operated controls were obtained. In the dentate granule cell layer, neither the fast nor slow f-EPSP differed significantly from those values obtained from sham-operated controls (Figs. 3 and 4, Table 1).
DISCUSSION
We investigated the differential responsiveness of NMDA receptor components in two regions of the rat hippocampus after transient global ischemia. Two principal findings emerge from this work: 1) the gene expression levels for both the NR2A and NR2B subunits decrease at early times after reperfusion in both the CA1 and dentate gyrus, but the expression levels of both subunits later recover in the dentate gyrus subfield; and 2) transient cerebral ischemia affects the fast f-EPSP and the slow NMDA receptor components of the f-EPSP differently in the CA1 subfield than in the dentate granule cell layer. Specifically, both the fast and slow f-EPSPs are maintained at 6 hours postischemia and diminished at 24 hours and 7 days postischemia in the CA1 subfield, but neither f-EPSP is altered significantly in the dentate granule cell subfield at any time postinsult.
Delayed neuronal death in the CA1 subfield of the hippocampus is a hallmark of the events that follow a period of severe transient global ischemia. However, the mechanisms by which the loss of these CA1 neurons occur remain unclear. The potential involvement of Ca2+ ions in the progression toward cell death has been proposed, and much experimental evidence supports the involvement of Ca2+ ions in degenerative pathways (Benveniste et al., 1988; Auer and Coulter, 1994; Dienel, 1984; Desphande et al., 1987; Scharfman and Schwartzkroin, 1989; Tymianski et al., 1993). Within the hippocampus, the NMDA receptor complex is believed to be composed of combinations of different subunits, with the NR1 subunit ubiquitous to all complexes (Monyer et al., 1992; Nakanishi, 1992; Hollmann and Heinemann, 1994; Westbrook, 1994). Because the NR1 subunit is common to all complexes, because many specific properties of NMDA receptors are dependent on the NR2 subunits (Monyer et al., 1992; Hollmann and Heinemann, 1994; Krupp et al., 1996), and because we have not observed any change in the immunoreactive protein levels for the NR1 subunit after global ischemia (Zhang et al., 1997; and Gurd, unpublished), we concentrated our examination on the expression levels of the nonubiquitous NR2A and NR2B subunits of the highly Ca2+ ion permeant NMDA receptor system in the vulnerable and less vulnerable regions of the hippocampus of rats subjected to a period of severe transient global ischemia and correlated these observations to the functional activity of the NMDA receptor complexes present in these hippocampal subfields at various times postinsult. Our results indicate that cerebral ischemia promotes a profound alteration in the functionality of both the NMDA and non-NMDA receptor complexes in the CA1 subfield but does not significantly or irreversibly alter the functionality of either subtype in the dentate granule cell layer.
The expression levels in the hippocampus for both the NR2A and NR2B receptor subunits were found to change in a time-dependent manner after challenge. At 6 hours postischemia, no significant change in expression levels from controls were observed in the CA1 subfield for either NR2A or NR2B; however, a significant decrease in expression was observed for NR2A in dentate granule cell region. Expression levels for both NR2A and NR2B at 24 hours postinsult were found to be decreased significantly in all hippocampal subfields examined. At more delayed times postischemia, mRNA expression levels for the NR2A and NR2B subtypes suggest that the granule cells of the dentate gyrus have rebounded from the initial effects of the ischemia. At 3 days postischemia, a time postchallenge when greater than 90% of the CA1 pyramidal cells display pyknotic morphology and apparently have committed to cell death (Shinno et al., 1997), NR2A and NR2B expression levels in the CA1 subfield were reduced dramatically from controls. However, expression levels in the dentate granule cell layer were elevated from control for NR2A and equal to control for NR2B. At 7 days postinsult, expression levels for both subtypes barely were detected in the CA1 subfield, but both subunits displayed an elevated expression in the dentate granule cell layer. These results are in contrast to those of Heurteaux et al. (1994), who report that at similar times postreperfusion, an increased level of NR2A and NR2B expression is observed in the CA1 subfield. In the present study, these late times were the only times postchallenge when elevated expression levels for one or both of the NMDA receptor subunits occurred in any hippocampal subfield, and those elevations were restricted to the granule cell layer of the dentate gyrus. The nature regarding any increase in NMDA receptor mRNA within the hippocampus at these delayed times postchallenge must be addressed with caution, however, because Gottlieb and Matute (1997) recently demonstrated that an induction of NR2A/2B subunits occurs between 3 and 28 days postischemia in reactive glial cells present in the deteriorating CA1 subfield. The degree to which such cells contribute to the pool of cells present in the dentate granule cell layer, where we observed increased NR2A and NR2B mRNA at extended days postchallenge, is under investigation.
The functionality of the NMDA receptor complex was found to differ between the postischemic CA1 subfield and the postischemic dentate granule cell layer. Although the NMDA receptor f-EPSP was not altered significantly from control at 6 hours postchallenge in either subfield, the CA1 subfield displayed a significantly diminished NMDA receptor mediated f-EPSP at 24 hours postchallenge, whereas the dentate granule cell layer NMDA receptor activity primarily was maintained. A trend toward a decreased NMDA receptor mediated slow f-EPSP that did not reach a significance value of P < 0.05 was observed in the dentate granule cell layer at 6 and 24 hours postchallenge; however, these decreases in activity were minimal when compared with the dramatic lack of NMDA receptor generated slow f-EPSP in the CA1 at 24 hours postchallenge. Furthermore, the fast f-EPSP response, which is generated primarily by AMPA/kainate receptors, was maintained in the dentate granule cell layer at all times examined postchallenge, but not in the CA1 subfield. Previous investigations have demonstrated that the morphologic integrity of CA1 neurons remains preserved at 24 hours postchallenge (Shinno et al., 1997). This leads to the speculation that although morphologically intact, the vulnerable CA1 neurons may be entering a state of nonresponsiveness at this time, and without intervention, are unable to recover. This is in contrast to the less vulnerable dentate granule neurons, for which the degree of impaired NMDA receptor function—as well as AMPA/kainate receptor function—is minimal at or before 24 hours postinsult.
We find our observations of a decreased level of activity for both NMDA and non-NMDA glutamate receptors in the CA1 subfield, but not in the dentate gyrus subfield, at 24 hours postischemia intriguing, given that the vulnerable CA1 neurons expire at least in part through programmed cell death pathways (Heron et al., 1993; Okamoto et al., 1993; Kihara et al., 1994; Nitatori et al., 1995; Krajewski et al., 1995). In several models, programmed cell death systems may be activated—at least in part—as a result from a lack of trophic interactions (Schecterson and Bothwell, 1992; Thoenen, 1995; Blochl and Thoenen, 1995; Lindholm at al., 1996). Because the activity-dependent exchange of trophic signals is well established (Thoenen, 1995; Blochl and Thoenen, 1995; Lindholm et al., 1996), it seems likely that in a population of quiescent neurons, such impaired trophic interactions could result and subsequently contribute to the demise of the neuron. The infusion of certain trophic factors, such as brain-derived neurotrophic factor (Beck et al., 1994), platelet-derived growth factor B (Iihara et al., 1997), ciliary neurotrophic factor (Wen et al., 1995), perhaps nerve growth factor (Shigeno et al., 1991; Tanaka et al., 1994, however, see Beck et al., 1992); insulin, and insulin-like growth factors (Zhu and Auer, 1994) have been reported to improve neuronal outcome after ischemia challenge. Thus, the resistant nature of the dentate granule cell neurons may result—at least in part—from their ability to maintain synaptic activity during the critical time window that precedes neuronal expiration, and that the CA1 neurons may continue into a programmed cell death cycle as a result of their inability to maintain significant excitatory activity at or before the 24-hour postischemia time window. The elements that promote the quiescent state in the CA1 neurons remain unclear at this time, although an overactive inhibitory system has been observed in the CA1 subfield 12 hours postreperfusion (Shinno et al., 1997). Interestingly, excessive inhibition through gamma-aminobutyric acid A receptors has been reported to inhibit the expression of brain-derived neurotrophic factor and neuropeptide Y in cultured hippocampal interneurons (Marty et al., 1996), and glutamatergic receptor activity increases brain-derived neurotrophic factor and nerve growth factor mRNA levels in the hippocampus (Zafra et al., 1990; Gwag and Springer, 1993). These observations strengthen the possibility that the potentiated inhibitory state present in the CA1 by 12 hours postinsult could lead to decreased trophic support in the sensitive CA1 subfield, but not in the less vulnerable dentate gyrus.
Footnotes
Abbreviations used
Acknowledgments
The authors thank Lucy Teves and Richard Logan for technical assistance, Judy Trogadis for photography, and Dr. J. Francis for manuscript assistance and helpful discussion.
