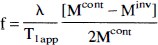Abstract
Thrombolysis of embolic stroke in the rat was measured using diffusion (DWI)-, T2 (T2WI)-, and perfusion (PWI)-weighted magnetic resonance imaging (MRI). An embolus was placed at the origin of the middle cerebral artery (MCA) by injection of an autologous single blood clot via an intraluminal catheter placed in the intracranial segment of internal carotid artery. Rats were treated with a recombinant tissue plasminogen activator (rt-PA) 1 hour after embolization (n = 9) or were not treated (n = 15). Diffusion-weighted imaging, T2WI, and PWI were performed before, during, and after embolization from 1 hour to 7 days. After embolization in both rt-PA-treated and control animals, the apparent diffusion coefficient of water (ADCw) and cerebral blood flow (CBF) in the ischemic region significantly declined from the preischemic control values (P < 0.001). However, mean CBF and ADCw in the rt-PA—treated group was elevated early after administration of rt-PA compared with the untreated control group, and significant differences between the two groups were detected in CBF (24 hours after embolization, P < 0.05) and ADCw (3, 4, and 24 hours after embolization, P < 0.05). T2 values maximized at 24 (control group, P < 0.001) or 48 hours (treated group, P < 0.01) after embolization. The increase in T2 in the control group was significantly higher at 24 hours and 168 hours than in the rt-PA—treated group (P < 0.05). Significant correlations (r ≥ 0.80, P < 0.05) were found between lesion volume measured 1 week after embolization and CBF and ADCw obtained 1 hour after injection of rt-PA. Within a coronal section of brain, MRI cluster analysis, which combines ADCw and T2 data maps, indicated a significant reduction (P < 0.05) in the lesion 24 hours after thrombolysis compared with nontreated animals. These data demonstrate that the values for CBF and ADCw obtained 1 hour after injection of rt-PA correlate with histologic outcome in the tissue, and that the beneficial effect of thrombolysis of an intracranial embolus by means of rt-PA is reflected in an increase of CBF and ADCw, a reduction in the increase of T2, and a reduction of the ischemic lesion size measured using MRI cluster analysis.
Keywords
Cerebral artery thromboembolism is a major cause of human stroke (Mohr et at., 1978; Sloan 1987). Thrombolytic canalization of occluded arteries may reduce ischemic cell damage if it is instituted before tissue injury becomes irreversible. Patients exhibit significant improvement of neurologic outcome after ischemic stroke when recombinant tissue plasminogen activator (rt-PA) is administered within 3 hours after onset of ictus (NINDS Stroke Study Group, 1995). Thus, the development of diagnostic tools to evaluate the efficacy and safety of thrombolysis is important for the management of human stroke. In experimental stroke, diffusion- and perfusion-weighted magnetic resonance imaging (DWI and PWI, respectively) have been used to monitor cerebral venous embolization (Röther et al., 1996). In three rats subjected to cerebral venous embolization and treated with rt-PA, DWI hyperintensity was reduced within 5 hours of treatment compared with untreated animals. However, the dynamic changes of cerebral blood flow (CBF) and the diffusion coefficient of water after thromboembolism with or without rt-PA intervention and the relationship between the early and delayed changes in CBF and ADCw with histopathologic outcome require further study.
Previous studies using thrombolytic agents to treat stroke have employed cerebrovascular thromboembolic models induced by autologous or heterologous blood clot embolization in rabbits and rats (Papadopoulos et al., 1987; Zivin et al., 1988; Overgaard et al., 1993b; Hamilton et al., 1994). A limitation of these models is the lack of uniformity in the size and location of the infarcts because the ultimate lodgement of the emboli is not well controlled (Hill et al., 1955; Penar and Greer, 1987; Phillips et al., 1988; Overgaard et al., 1992). We recently developed a model of embolic focal cerebral ischemia in the rat relevant to human thromboembolic stroke (Zhang et al., 1997a). This model provides a reproducible and predictable lesion localized to the territory supplied by the middle cerebral artery (MCA).
The timing of recanalization of an occluded intracerebral artery modifies the size of infarction. Autolytic recanalization of embolic occlusion in patients occurs within the first days after ictus (Sindermann et al., 1969; Irino et al., 1977; Ito et al., 1983), and 89% of patients show patent intracerebral vessels after 2 to 3 weeks (Fieschi and Bozzao, 1969). The timing and process of recanalization after administration of a thrombolytic agent likely varies with the site of embolic lodgement, composition and volume of the clot, and other factors.
There have been no systematic investigations performed on the temporal evolution of CBF, ADCw, and T2 with ischemic cell damage after administration of a thrombolytic agent. Therefore, in an effort to elucidate hemodynamic and biophysical changes in ischemic tissue after thrombolysis, we investigated by means of magnetic resonance imaging (MRI) and histopathology the effect of thrombolytic treatment with rt-PA of a reproducible rat model of embolic stroke.
MATERIAL AND METHODS
All experimental procedures have been approved by the Care of Experimental Animals Committee of Henry Ford Hospital.
Male Wistar rats weighing 300 to 450 g (n = 24) were used in the experiments. Animals were anesthetized with 3.5% halothane, and anesthesia was maintained with 1% halothane in 70% nitrous oxide (N2O) and 30% oxygen (O2) with a face mask. Rectal temperature was maintained at 37 ± 0.5°C throughout the surgical procedure using a feedback-regulated water-heating system. The right femoral artery and vein were cannulated with a PE-50 catheter for monitoring of blood pressure and gases (pH, partial pressure of oxygen [PO2], partial pressure of carbon dioxide [PCO2]), and for drug administration, respectively.
The method used to prepare a white embolus was adapted and modified (Zhang et al., 1997a) from Overgaard et al (1992). Briefly, femoral arterial blood from a donor rat was withdrawn into 20 cm of PE-50 tubing and retained in the tube for 2 hours to clot at room temperature, and subsequently retained for 22 hours at 4°C. Five centimeters of the PE-50 tube containing clot was cut and attached at each end to a 20-cm PE 10 tube interconnected by a syringe filled with saline. The clot was shifted by continuous alternating movement from one syringe to the other for 5 minutes. A single clot (25 mm × 3.14 × 0.01 mm2 = 0.8 μL) was transferred to a modified PE-50 catheter with a 0.3-mm outer diameter filled with saline.
Under the operating microscope (Carl Zeiss, Inc., Thornwood, NY, U.S.A.), the right common carotid artery, the right external carotid artery (ECA), and the internal carotid artery were isolated via a midline incision. A 5-0 silk suture was tied loosely at the origin of the ECA and ligated at the distal end of the ECA. The right common carotid artery and internal carotid artery were clamped temporarily using a curved microvascular clip (Codman & Shurtleff, Inc., Randolf, MA, U.S.A.). A modified PE-50 catheter with a 0.3-mm outer diameter filled with a 25-mm long clot, which was attached to a 100-μL Hamilton syringe, was introduced into the ECA lumen through a small puncture. The suture around the origin of the ECA was tightened around the intraluminal catheter to prevent bleeding, and the microvascular clip was removed. A 15-mm length of catheter was advanced gently from the ECA into the lumen of the internal carotid artery. At this point, the intraluminal catheter was 2 to 3 mm from the origin of the MCA. The clot plus 5 μL of saline in the catheter was injected into the internal carotid artery over 10 seconds. The catheter was withdrawn from the right ECA 5 minutes after injection. The right ECA was ligated. The duration of the entire surgical procedure was approximately 25 minutes. Heparin was not administered to any animal.
Two groups of animals were investigated after embolization; 1) untreated animals (n = 15) and 2) treated animals in which rt-PA was administered 1 hour after embolization (n = 9). In the treated group, rt-PA (Genentech, Inc., South San Francisco, CA, U.S.A.) was administered at a dose of 10 mg/kg as a 10% intravenous bolus and the remainder continuously infused (intravenously) over a 30-minute interval using a Harvard pump (Harvard Apparatus, South Natick, MA). In these two groups, 12 animals (6 in the untreated control group and 6 in the treated group) were killed at 1 week. Four of 15 animals in the control group and 1 of 9 animals in the rt-PA—treated group died during the 4- to 24-hour period, and the remaining animals were killed 5 hours after embolization. Severe cerebral edema appeared to be the cause of death. Hemorrhage was not evident.
Magnetic resonance imaging measurements were performed using a 7 T, 20-cm bore superconducting magnet (Magnex Scientific, Abingdon, U.K.) interfaced to a console (SMIS, Surrey, England). A 12-cm bore actively shielded gradient coil set, capable of producing magnetic field gradients up to 20 gauss/cm, and a birdcage radiofrequency coil were used. Stereotaxic ear bars were used to minimize movement during the imaging procedure. During MRI measurements, anesthesia was maintained using a gas mixture of N2O (69%), O2 (30%), and halothane (0.75–1%). Rectal temperature was kept constant using a feedback-controlled water bath. A modified fast low angle shot (Frahm et al., 1986) imaging sequence was used for reproducible positioning of the animal in the magnet at each MRI session (Knight et al., 1991; Jiang et al., 1993). Diffusion-weighted imaging, T2WI, and PWI were performed preischemia, repeatedly for 4 to 5 hours after onset of embolization and 24, 48, and 168 hours after onset of embolization for both groups.
The ADCw was measured using the method described by LeBihan et al. (1986, 1989a). A two-dimensional Fourier transform multislice spin-echo sequence (7 slices, 32-mm field of view, 128 × 64 matrix, pulse repetition time (TR) = 1.5 seconds; echo time (TE) = 40 msec, two signal average) was modified to include two 10.5-msec diffusion-weighting gradient pulses, one on either side of the refocusing 180° radiofrequency pulse. The diffusion-weighting gradient was increased, in a nonlinear manner, from 0 to approximately 8.36 gauss/cm to obtain a series of five images with gradient b-values of 0, 200, 400, 600, and 800 seconds/mm2. Each image required a scan time of 3.2 minutes for completion. The total time for the entire sequence was approximately 16 minutes.
T2 was measured using standard two-dimensional Fourier transform multislice (7 slice) multiecho (4 echo) MRI. A series of four sets of images (7 slices for each set) were obtained using TEs of 30, 60, 90, and 120 msec and a TR of 3 seconds. Images were produced using a 32-mm field of view, 2-mm slice thickness, 128 × 64 image matrix. The total time for the entire sequence was approximately 3.2 minutes.
Perfusion measurements were performed before each diffusion measurement using a technique described by Williams et al. (1992). This method of CBF measurement is based on the selective inversion of blood water protons at the level of the carotid arteries before 1H MRI measurement in the brain. Two images were obtained for perfusion measurement with the following parameters: TR = 1.055 seconds, TE = 30 msec, 64 × 64 image matrix, 3-mm slice thickness, and a 32-mm field of view. In each animal, PWI was measured within a coronal section, with coordinates centered at interaural 8.2 mm and bregma 0.8 mm (Paxinos and Watson, 1986). The duration of the inversion pulse was 1 second at a B1 amplitude of 0.3 KHz.
After the final nuclear magnetic resonance measurement, animals were anesthetized with ketamine (44 mg/kg) and xylazine (13 mg/kg) and transcardially perfused with heparinized saline and 10% neutral buffered formalin. The head was immersed in formalin solution for 1 hour, after which the brain was cut into 2-mm thick coronal blocks, for a total of seven blocks. Brain tissue was processed and embedded, and 6-μm thick paraffin sections from each block were cut and stained with hematoxylin and eosin (H&E) for evaluation of ischemic cell damage. The lesion volume was measured using Global Lab Image analysis software (Data Translation, Malboro, MA, U.S.A.). Each H&E section was evaluated at 2.5× magnification. The area of infarction and the area of the ipsilateral hemisphere (mm2) were calculated by tracing the areas on the computer monitor screen, and the volume (mm3) was determined by multiplying the appropriate area by the section interval thickness (Zhang et al., 1994). To reduce errors associated with processing of tissue for histologic analysis, an indirect method for calculating lesion area was used in which the intact area of the ipsilateral hemisphere was subtracted from the area of the contralateral hemisphere (Swanson et al., 1990). The percentage of lesion to the area of the contralateral hemisphere and the percentage of lesion to the volume of the contralateral hemisphere were calculated.
Nuclear magnetic resonance image analyses of ADCw, T2, and CBF map sections were performed using Eigentool (Windham et al., 1988; Peck et al., 1992). All images were reconstructed using a 128 × 128 matrix. The ADCW and T2 maps were produced on a pixel-by-pixel basis using a least-squares fit to a straight line plot of the natural logarithm of normalized image intensity versus gradient b-value (ADCw) or TE (T2) values, respectively. Cerebral blood flow maps (f) were calculated according the following equation (Williams et al., 1992):
where λ is blood:brain partition coefficient, T1app is apparent T1 relaxation time of brain, and Mcont and Minv are the control and inversion image intensities, respectively. A value of 0.9 g mL−1 for λ and 1.7 seconds for T1app (Williams et al., 1992) were used for CBF calculation. Relative CBF values, which represent CBF of the lesion hemisphere after embolization normalized to preischemic CBF, were calculated. In addition, the relative area containing low CBF values, i.e., the number of pixels with CBF below 50 mL/100 g per minute normalized to the total numbers of pixels in the ipsilateral hemisphere, was calculated. Region of interest (5 × 5 pixels) measurements of the lesions also were obtained from the center of the DWI abnormality during acute ischemia. The relative ADCw values, which represent the ADCw values in the lesion region of interest after embolization normalized to preischemic ADCw values, and the T2 difference (ΔT2, T2-ischemia-T2-contralateral control) within the same regions of interest as used in the ADCw measurement were calculated.
Cluster image analyses of ADCw and T2 map sections were performed using Eigentool software (Windham et al., 1988; Peck et al., 1992; Jiang et al., 1997). The ADCw versus T2 values from the contralateral hemisphere were plotted to generate a two-dimensional cluster scatterplot (Jiang et al., 1997). The cluster associated with cerebrospinal fluid with its elevated ADCw values is distinct from parenchymal tissue, and was excluded from analysis. The mean and standard deviation (SD) of ADCw and T2 values of the parenchymal tissue cluster (not cerebrospinal fluid) were calculated, and the normal pixels were defined using mean ADCw ± 1.5 SD and mean T2 ± 2 SD (Jiang et al., 1997). Once the normal tissue cluster was defined, the two-dimensional cluster scatterplot from the coronal section of brain then was generated, and the abnormal pixels were defined as pixels outside the normal cluster. The numbers of normal and abnormal pixels were calculated within the ipsilateral hemisphere. To reduce the effect of variation of the lesion size and also to capitalize on the experimental protocol that the ischemic tissue is not treated until 1 hour after onset ischemia, the number of abnormal pixels at time points beyond 1 hour were normalized to those obtained at the 1-hour time point ([abnormal pixels]> 1 hour/[abnormal pixels]1 hour). This analysis uses each animal as its own control and is a sensitive way to detect MRI differences between the treated and untreated groups.
Statistics
Observations are summarized as mean ± SDs. The early death of subsets of animals makes repeated measures analysis of variance impractical, so the data analysis was performed by use of paired Student's t tests for time effects and by two sample Student's t tests for rt-PA effects. Because the benefit of rt-PA treatment of thromboembolic stroke is most evident at 24 and 48 hours (Jiang et al., 1996; Zhang et al., 1997a) and because we have the largest sample size at 24 hours, multiple-comparisons issues with respect to rt-PA effects were addressed by considering the 24-hour comparisons as the primary test of an rt-PA effect. Unadjusted P values for the secondary tests at other times also are presented.
For tests of time effects (changes from baseline for CBF and ADCw, and for normal vs. ischemic hemisphere for T2), Bonferonni adjusted P values were used. There were eight comparisons for CBF and T2 and seven for ADCw. P values of 0.05/8 = 0.0065 and 0.05/7 = 0.0071, respectively, were considered significant for these tests.
Correlations were computed among the MRI parameter values postembolization and between these parameters and the histologically determined lesion volume observed at 7 days.
RESULTS
The physiologic parameters after embolism are summarized in Table 1. All physiologic parameters were within the normal range of values.
General physiologic parameters
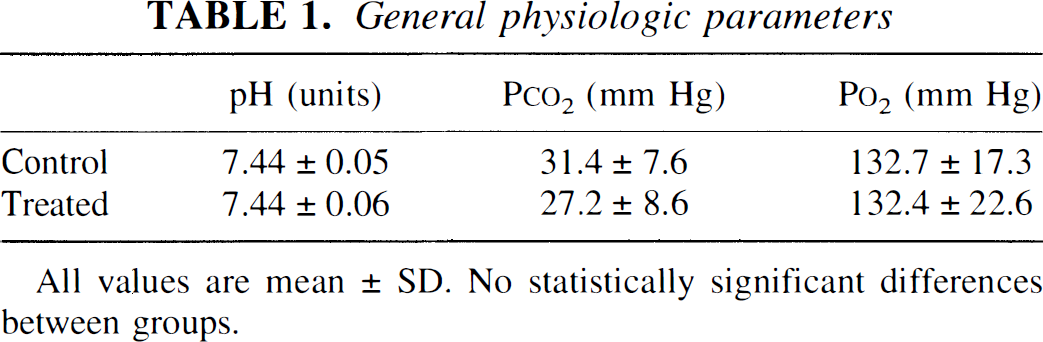
All values are mean ± SD. No statistically significant differences between groups.
Figures 1A and 1B present CBF maps from a representative untreated and an rt-PA—treated animal obtained at various times before and 1 to 48 hours after embolization. Cerebral blood flow maps exhibited large areas of reduced CBF in the ipsilateral hemisphere 1 hour after injection of embolus in both untreated and treated animals. Cerebral blood flow in the ipsilateral hemisphere of the untreated animal remained low up to 24 hours after embolization, whereas the treated animal exhibited a more rapid return toward preischemic CBF values. At 48 hours of embolization in the untreated animal, CBF increased to preischemic values in the ipsilateral cortex. However, subcortical CBF values remained low. In the treated animal, restoration of CBF was evident at 24 hours, and hyperemia was detected at 48 hours.
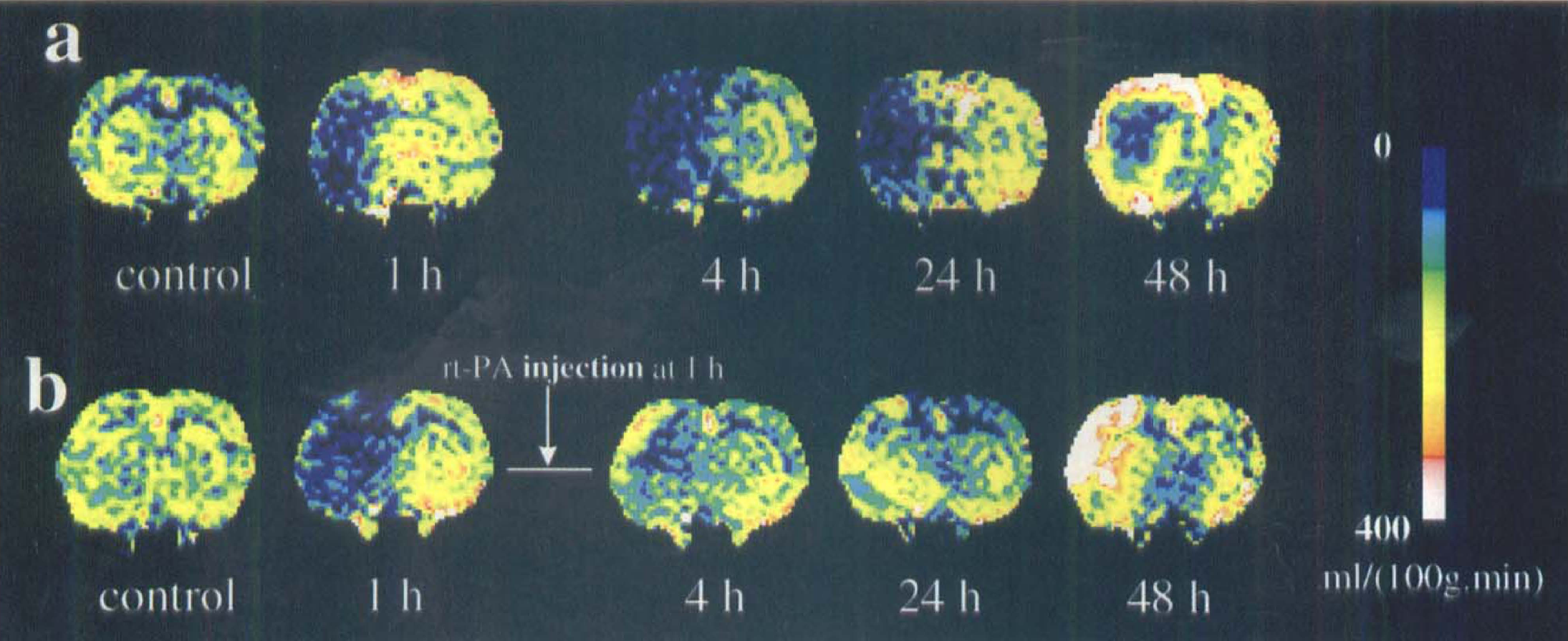
Cerebral blood flow (CBF) maps of coronal sections of an untreated (
Figure 2 shows temporal profiles of relative CBF changes of the ipsilateral hemisphere before and after embolization. As a result of embolization, CBF values in the ipsilateral hemisphere decreased (P < 0.001) from preischemic control values in both groups. In the control group, CBF in the ischemic hemisphere remained low up to 24 hours and returned to preischemic values at 48 hours. The mean CBF in the treated group gradually increased after injection of rt-PA. The difference in CBF between the groups was significant (P = 0.046) 24 hours after embolization.
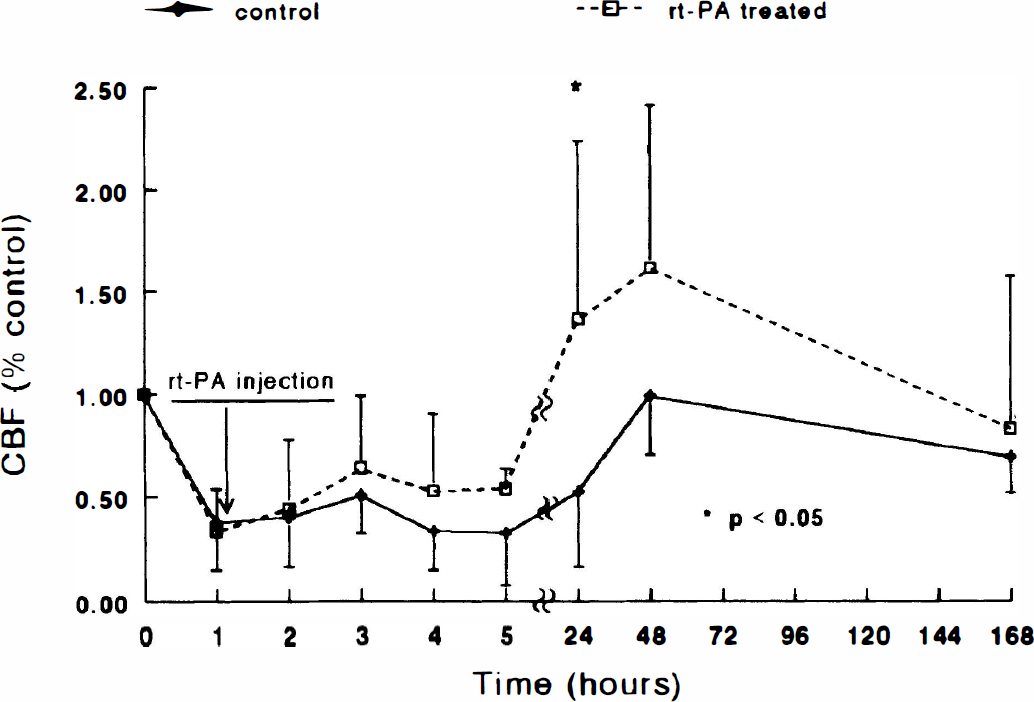
Average relative cerebral blood flow (CBF) values in the ischemic hemisphere of treated and untreated animals plotted as a function of time after embolization. As a result of embolization, CBF values in the ischemic hemisphere significantly decreased (P < 0.001) in both the treated and untreated groups. The increase in mean CBF was higher in the treated group than in the untreated group 2 hours after embolization (1 hour after injection of recombinant tissue plasminogen activator [rt-PA]), and statistical significance was detected 24 hours (P < 0.05) after embolization.
Figure 3 shows temporal profiles of the relative areas with low CBF (percent area containing CBF < 50 mL/100 g per minute to the area of the ipsilateral hemisphere within the reference coronal section) in the ipsilateral hemisphere before and after embolization. As a result of embolization, the relative areas with low CBF in the ipsilateral hemisphere increased (P < 0.01) from preischemic control values in both groups. In the control group, areas with low CBF in the ischemic hemisphere remained high up to 24 hours and decreased at 48 hours. The areas with low CBF in the treated group gradually decreased after injection of rt-PA. The difference in the areas with low CBF between the groups was significant at 4 hours (P = 0.008), 5 hours (P = 0.006), 24 hours (P < 0.001), and 48 hours (P = 0.025) after embolization.
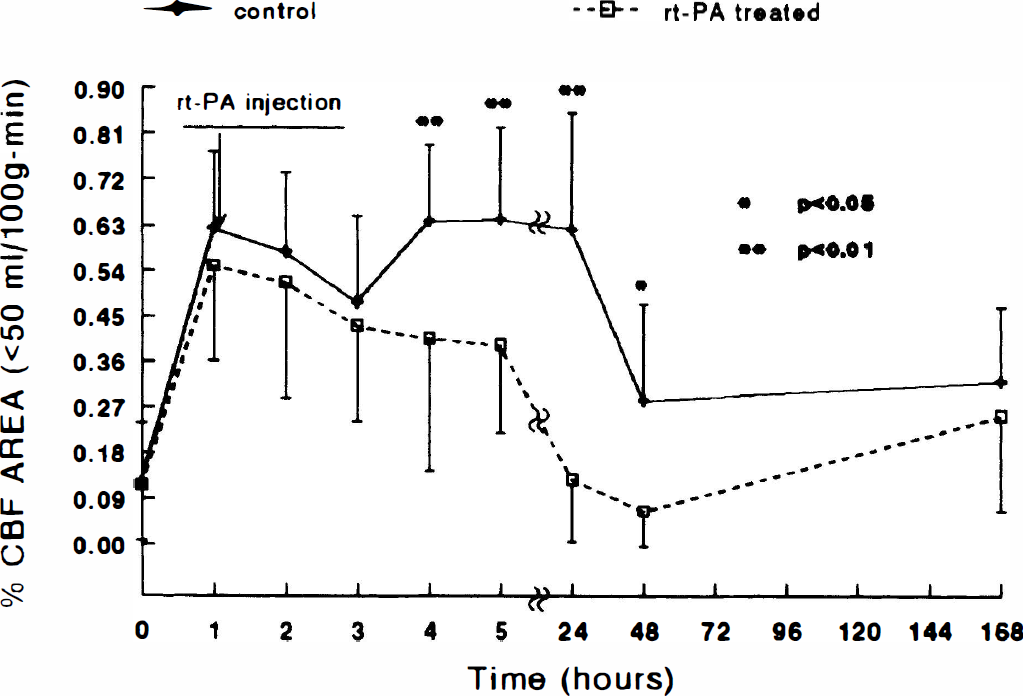
Average relative area with low cerebral blood flow (CBF; percent area containing CBF < 50 mL/100 g per minute to the area of the ipsilateral hemisphere) in the ipsilateral hemisphere of treated and untreated animals plotted as a function of time after embolization. As a result of embolization, the relative areas with low CBF in the ipsilateral hemisphere increased (P < 0.01) from preischemic control values in both groups. The decrease in the mean values of the relative areas with low CBF was greater in the treated group than that in the untreated group, and significant differences were detected at 4 hours (P = 0.008), 5 hours (P = 0.006), 24 hours (P < 0.001), and 48 hours (P = 0.025) after embolization. rt-PA, recombinant tissue plasminogen activator.
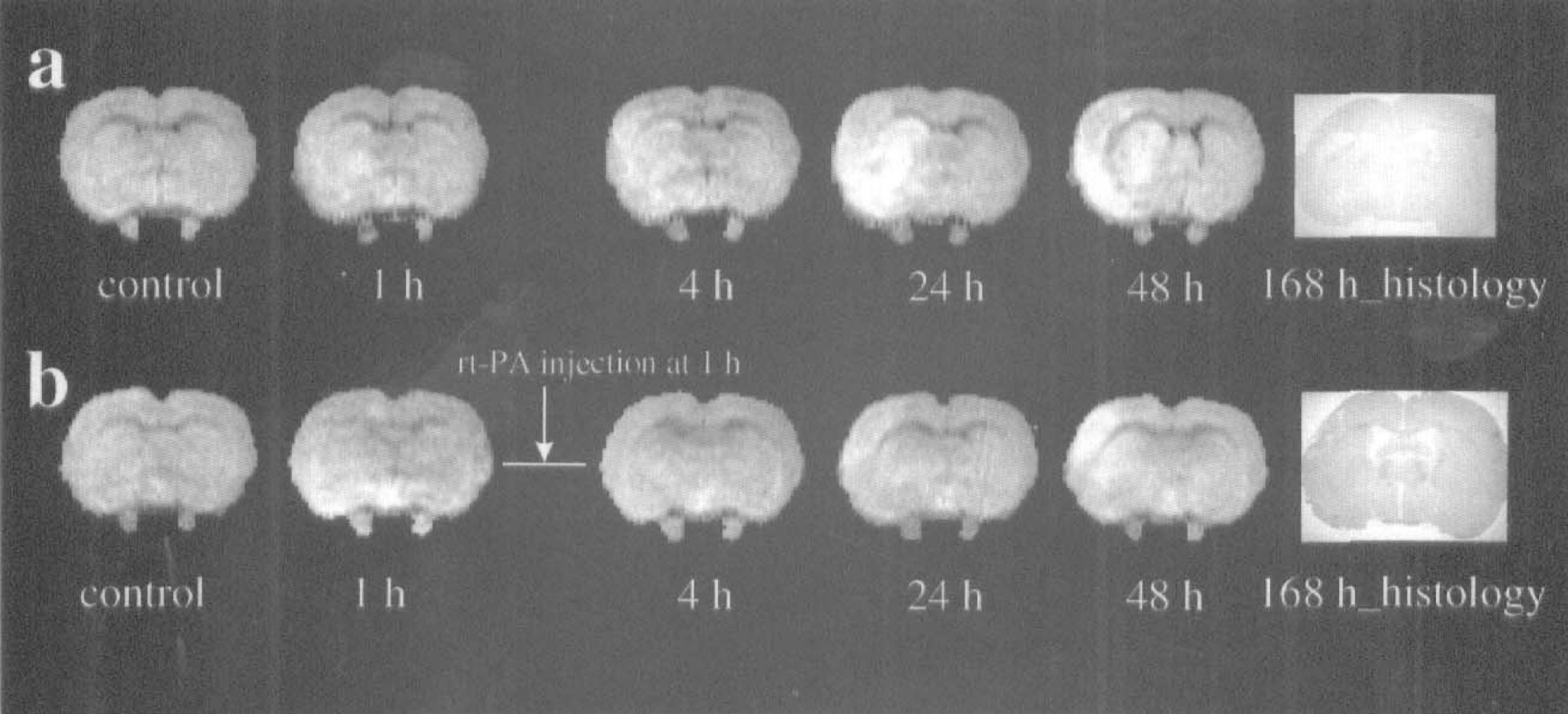
Figures 4A and 4B present DWI (b = 800 s/mm2) obtained from 1 to 48 hours after injection of embolus from the same animals shown in Figs. 1A and 1B. A hyperintensity is apparent in the DWI of the ipsilateral hemisphere in both animals after embolization. In the untreated animal (Fig. 4A), a DWI hyperintensity is apparent in the ipsilateral caudate putamen and insular cortex after the onset of ischemia; image contrast and the area of hyperintensity increased as ischemia progressed and maximized at 24 to 48 hours. In the treated animals (Fig. 4B), changes of DWI hyperintensity were variable after injection of rt-PA. Two treated animals exhibited a pattern and temporal profile of DWI hyperintensity similar to that for the control animals, and three treated animals exhibited an early disappearance of DWI hyperintensity (within 24 hours). The remaining treated animals exhibited a slower increase in image contrast and area of hyperintensity than the control animals.
Diffusion-weighted images (b = 800) of coronal sections from the same animal in Fig. 2 without (
The last images (marked 168 h_histology) in Fig. 4A and 4B show the H&E-stained coronal sections from the same animals shown in Fig. 4A and 4B. The damaged area in the H&E-stained section from untreated and treated animals corresponds closely with the hyperintense area in the diffusion-weighted image obtained 24 hours after embolization. The representative untreated animal exhibited a lesion area incorporating putamen and parietal and insular cortex, whereas the representative treated animal exhibited a small lesion area localized to the parietal cortex. The rt-PA-treated animals exhibited smaller normalized areas (P = 0.04) and volumes (P = 0.01) of ischemic damage (34 ± 9%, percent of the lesion area to the area of the contralateral hemisphere within the reference coronal section; 20 ± 11%, percent of the lesion volume to the volume of the contralateral hemisphere) than that in the untreated animals (65 ± 20%, percent of the lesion area to the area of the contralateral hemisphere within the reference coronal section; 43 ± 14%, percent of the lesion volume to the volume of the contralateral hemisphere).
Figure 5 shows the temporal profiles of normalized ADCw (ADCw values for ischemic damaged tissue normalized to preischemic control tissue) for a region of interest localized to the center of the DWI abnormality in the ipsilateral hemisphere in both groups. After embolization, the ADCw values in the ischemic striatum significantly (P < 0.001) decreased from preischemic values in both untreated and treated groups. In the untreated group, ADCw in the ischemic striatum remained below preischemic ADCw values up to 24 hours. In the treated group, ADCw gradually increased after injection of rt-PA with a transient decline 4 hours after embolization. Higher mean ADCw values were present from 1 to 23 hours after injection of rt-PA in the treated group compared with the untreated group. Statistically significant differences were detected in normalized ADCw values between the groups at 3 hours (P = 0.039), 4 hours (P = 0.017), and 24 hours (P = 0.023). Two rt-PA—treated animals with very low CBF (< 15% of preischemic values) 1 hour after administration of rt-PA exhibited a temporal profile of ADCw similar to that in the untreated animals, i.e., a low ADCw out to 24 hours. These two animals also exhibited the largest lesion volume measured by histopathology. In contrast, three treated animals with relatively high CBF 1 hour after administration of rt-PA (> 60% of preischemic values) showed a rapid return of ADCw.
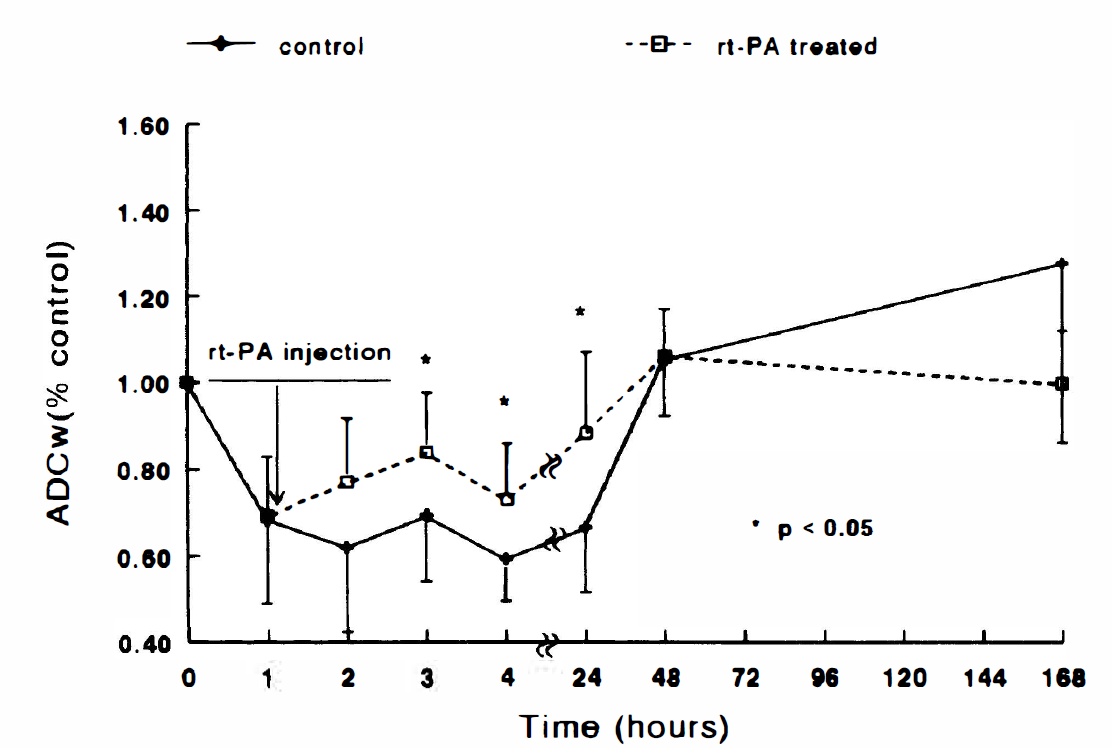
Relative apparent diffusion coefficient of water (ADCw) in the lesion under control and recombinant tissue plasminogen activator (rt-PA)–treated conditions plotted as a function of time after embolization. The relative ADCw values in the ischemic area significantly (P < 0.001) decreased from preischemic values after embolization in both groups. Significant differences (P < 0.05) were detected 3, 4, and 24 hours after embolization between the treated and untreated groups.
The correlations between CBF, ADCw, and lesion volume with and without rt-PA intervention were analyzed using the Pearson correlation method. In the rt-PA—treated group, significant correlations were detected between the relative hemispheric CBF (1 hour after rt-PA injection, r = 0.91, P = 0.031), the relative area with low CBF (at 1 hour, r = 0.95, P = 0.012; at 2 hours, r = 0.91, P = 0.033), and ADCw (at 1 hour, r = 0.94, P = 0.017) 1 or 2 hours after injection of rt-PA and lesion volumes measured 1 week after embolization in the rt-PA treated group. Furthermore, significant correlations between lesion area within the reference coronal section and the relative hemispheric CBF (1 hour after rt-PA injection, r = 0.92, P = 0.027), the relative area with low CBF (at 1 hour, r = 0.986, P = 0.002; at 2 hours, r = 0.945, P = 0.015), or ADCw (at 1 hour, r = 0.88, P = 0.046) were detected 1 or 2 hours after injection of rt-PA. Significant correlations in the rt-PA—treated animals also were detected between CBF at 1 hour with ADCw at 1 hour (r = 0.80, P = 0.017) and at 2 hours (r = 0.73, P = 0.040) after injection of rt-PA. However, at later time points, significant correlations between CBF and ADCw and lesion volume were not detected. In the control group, significant correlations between CBF, ADCw, and lesion volume were not detected.
Figure 6 shows the postembolic temporal profiles of ΔT2 in the lesion (the same region of interest as that in ADCw measurements) in both the rt-PA—treated group and the untreated group. A small increase in ΔT2 was detected within 4 hours, and ΔT2 maximized at 24 hours in the untreated group (P < 0.001) and at 48 hours in the treated group (P = 0.006), and then declined 1 week after MCA occlusion. However, ΔT2 at 1 week remained elevated relative to baseline in both groups. The rt-PA—treated group had a lower ΔT2 (P = 0.003) 24 hours after embolization than the control group.
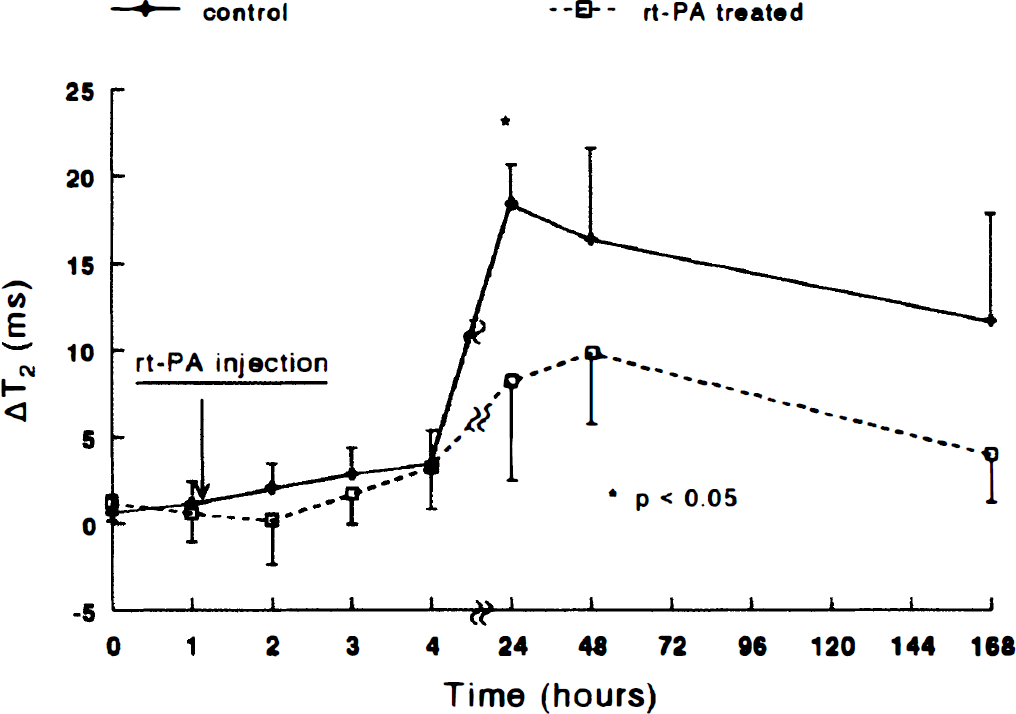
The temporal profiles of ΔT2 (the difference of T2 values in homologous regions of the ipsilateral and contralateral hemispheres) in the lesion during ischemia and after withdrawal of the suture in both the treated and untreated groups. A small increase in ΔT2 was detected within 4 hours, and ΔT2 maximized at 24 hours in the untreated group (P < 0.001) and at 48 hours in the treated group (P < 0.01) and then declined 1 week after embolization. The treated group had lower ΔT2 (P = 0.003) 24 hours after embolization than the control group. rt-PA, recombinant tissue plasminogen activator.
Figure 7 shows the temporal profiles of normalized number of abnormal pixels ([abnormal pixels]> 1 hour/[abnormal pixels]1 hour) after embolization in the ipsilateral hemispheres in the corresponding coronal section in both the rt-PA—treated group and the untreated group. The relative number of abnormal pixels in the untreated animals increased 4 hours after embolization and maximized 24 hours and 1 week after embolization. The relative number of abnormal pixels in the rt-PA—treated animals increased at 4 hours and maximized at 48 hours after embolization but declined afterward. The average relative number of abnormal pixels in the treated animals were lower than those in the untreated animals after injection of rt-PA. Statistically significant differences were detected 24 hours (P = 0.009) and 168 hours (P = 0.022) after embolization.
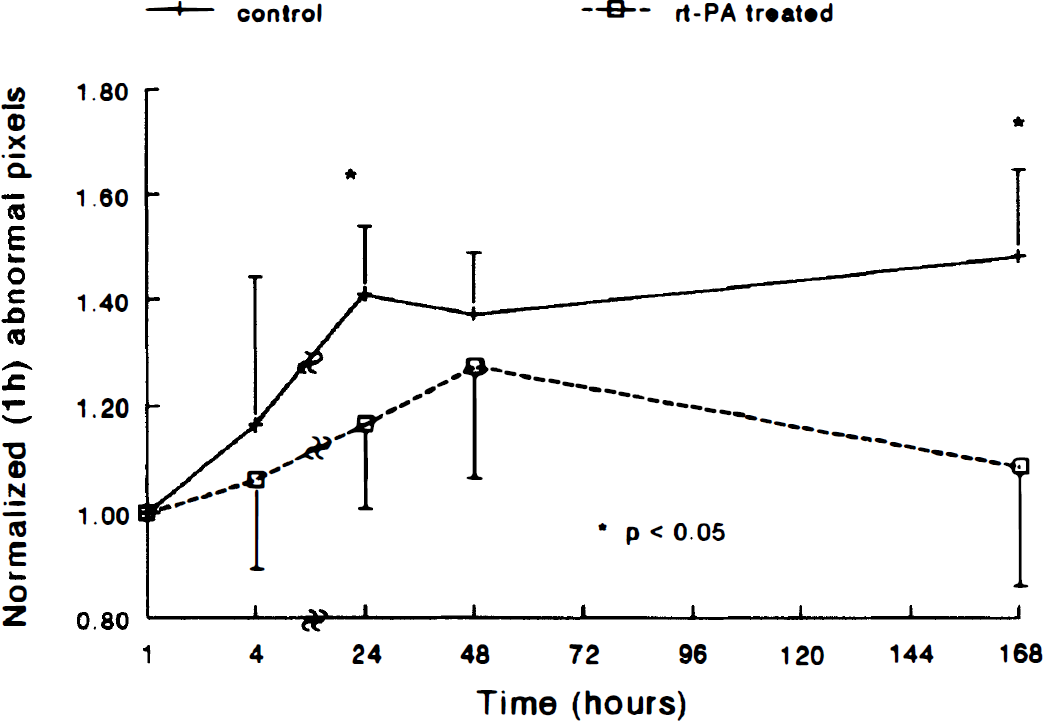
The normalized number of abnormal pixels in control and treated animals plotted as a function of time after embolization in the ipsilateral hemispheres. The normalized abnormal pixels in the control group increased compared with treated animals 4 hours after ischemia, and a significant difference (P < 0.05) was detected 24 and 168 hours after embolization. rt-PA, recombinant tissue plasminogen activator.
DISCUSSION
In this study, we employed MRI to monitor embolization and thrombolysis. Significant correlations were demonstrated between CBF, ADCw, and lesion volume after administration of rt-PA. Perfusion-weighted imaging provided a sensitive and continuous indication of embolization and thrombolysis. Cerebral blood flow in the ipsilateral hemisphere was reduced profoundly after injection of the embolus. It gradually increased after administration of rt-PA in the treated group and was elevated consistently compared with the control group. The ADCw in the ipsilateral hemisphere declined significantly after injection of embolus, and low ADCw persisted for at least 24 hours in the untreated group. Treatment with rt-PA elevated ADCw at acute times, with an early return at 24 hours. Small increases in mean T2 values were noted during the acute period and maximized at 24 to 48 hours after embolization in both the rt-PA—treated group and the control group, respectively. However, the increase in ΔT2 of the control group was significantly higher at 24 hours than in the rt-PA—treated group. Treatment of rats subjected to embolization with rt-PA reduced the volume of the ischemic infarct. This reduction was reflected in an increase in mean CBF and ADCw and a reduction of the T2 increase. Cluster analysis indicates that the rate of lesion growth is reduced.
In the treated animals, significant correlations were found between CBF 1 hour after administration of rt-PA and lesion volume and area measured 1 week after embolization in the treated animals. These data imply that rapid recanalization of embolically occluded brain arteries leads to reduced lesions. Studies by many investigators (Sundt et al., 1969; Crowell et al., 1970; Sundt and Waltz, 1971; Watanabe et al., 1977; Pulsinelli et al., 1982; Weinstein et al., 1986; Chen et al., 1993; Garcia et al., 1993) using a variety of animal models have identified a critical period of 2 to 7 hours after ischemia in which reversible cell damage transforms to irreversible damage. The time to salvage tissue probably is related to the degree and duration of CBF reduction, the specific tissue, and the animal species. Induction of focal cerebral ischemia models in the primate with reperfusion instituted within 4 hours of MCA occlusion preserves brain tissue and reduces infarction (Sundt et al., 1969; Crowell et al., 1970; Weinstein et al., 1986). The efficacy of reperfusion in reducing cell damage, however, strongly depends on its rapidity because survival of ischemic tissue decreases exponentially with time (Astrup et al., 1981; Jones et al., 1981; Heiss and Rosner, 1983). Thus, in our model of embolic stroke in the rat, CBF measured 1 hour after administration of rt-PA may be a good indicator of outcome.
Significant correlations were detected between CBF at 1 hour and ADCw at 1 and 2 hours after administration of rt-PA. The ADCw is sensitive to CBF (Moseley et al., 1990; Busza et al., 1992). Busza et al. (1992) reported that the decrease in ADCw is related to a threshold of CBF. However, ADCw is a function of a number of variables (Cooper et al., 1970; Tanner, 1978; LeBihan et al., 1989b; Moseley et al., 1990; Moonen et al., 1991; Benveniste et al., 1992; Helpern et al., 1992), not only CBF. Benveniste et al. (1992) demonstrated that ADCw declines after direct microdialysis treatment of rat brain with ouabain, a known inhibitor of Na+,K+-ATPase, which acts without changing CBF. Therefore, in the acute ischemic phase, decline in ADCw may be related to energy failure. Mintorovitch et al. (1994) demonstrated a significant decline in Na+,K+-ATPase activity at 30 and 60 minutes after MCA occlusion in the rat. After induction of embolic stroke, the relationship between CBF and ADCw with rt-PA intervention may be time dependent, with the changes in ADCw after rt-PA intervention being coupled tightly to CBF only at specific times.
In this study, we failed to detect a significant correlation between CBF and lesion volume in the control animals. In contrast, rt-PA—treated animals demonstrated a significant correlation between CBF within 1 hour of treatment and final infarct volume. This may be related to the therapeutic efficacy of rt-PA being dependent of the ability to increase CBF very early after treatment. In addition, acute and small increases in CBF may sensitize the tissue to the subsequent and beneficial effects of increased CBF. Subpopulations of animals that benefit from rt-PA treatment may be those that undergo rapid CBF perturbations caused by treatment.
Small changes in CBF may be the critical factors that determine the efficacy of thrombolytic treatment. The degree of reduction and duration of CBF after ischemia determine the transformation of reversible to irreversible ischemic injury (Dearden, 1985; Morawetz 1990). A 4-mL/100 g per minute CBF difference early in ictus can double the duration for reversible ischemic injury to transform to irreversible ischemic injury (Morawetz, 1990). Thus, the variation in CBF response and relatively small increase in CBF at early times after rt-PA treatment may be the reason that no significant difference in CBF between untreated and treated animals was detected early after injection of rt-PA. In addition, as noted in Fig. 2, the standard deviations of CBF in both groups of animals are large. This likely is due to variable biological events as well as to errors inherent to the spin tagging CBF methodology. Errors in the proton spin tag MRI CBF measurement may derive from the relatively low signal-to-noise ratio (approximately 5:1 for CBF map) and the assumptions of constant values of λ and T1app applied in this study (Herscovitch and Raichle, 1985; Williams et al., 1992). Overestimates of CBF will appear in the ischemic damaged areas because of the assumption of constant values of T1app. The relatively low signal-to-noise ratio and consequent large standard deviations may reduce the ability to detect small CBF differences between groups. Thus, the slight increase in CBF evoked by rt-PA treatment, which may dictate the survival of the tissue, may be obscured. Evidence of a rapid response of treated tissue to rt-PA intervention is more apparent when measuring the relative areas of decreased CBF (Fig. 3) than the mean hemispheric CBF (Fig. 2). Significant differences in areas of low CBF are visible 3 hours after treatment, although there is only a trend of difference between groups in Fig. 2.
The temporal profile of changes in ADCw after embolization is similar to that obtained in the intraluminal filament MCA occlusion model in which ADCw declines significantly after onset of MCA occlusion and remains depressed up to 24 hours after MCA occlusion (Jiang et al., 1993, 1994). The rt-PA intervention causes an increase in ADCw and a more rapid return in ADCw after embolization compared with untreated animals. Changes in ADCw after ischemia may be related to the permeability of cell membranes, restriction barriers of cellular microstructure, alterations of the distribution of volume between intracellular and extracellular compartments in the damaged tissue, energy depletion, temperature, and tortuosity (Cooper et al., 1970; Tanner 1978; LeBihan et al., 1989b; Moseley et al., 1990; Moonen et al., 1991; Benveniste et al., 1992; Busza et al., 1992; Helpern et al., 1992). Normalization of ADCw after an ischemic insult has been reported after a short duration of transient ischemia (≤ 30 minutes of MCA occlusion) with no ischemic damage (Minematsu et al., 1992). The decrease in ADCw during the acute ischemic period correlates to histopathologic outcome of the tissue: a large decrease in ADCw is predictive of more severe damage (Jiang et al., 1993). The increase in ADCw during the acute time period after injection of rt-PA and its early return, therefore, are most likely related to reduction of ischemic cell damage. A recent study in a rat model of cerebral venous thrombosis also exhibited a partial recovery in ADCw after rt-PA treatment (Röther et al., 1996).
Combination therapy with rt-PA and cytoprotective agents may be a promising therapeutic option. Although rt-PA has shown benefit in both clinical and experimental studies (Zivin et al., 1985, 1988; del Zoppo et al., 1986; Overgaard et al., 1993a; NINDS, 1995), not every patient responded to rt-PA treatment in the NINDS rt-PA stroke trial (NINDS, 1995). The efficacy of thrombolytic intervention not only depends on arterial recanalization but also on the secondary effects of thrombolysis, such as redistribution of clot material, effects of rt-PA on the fibrinolytic system, breakdown of the blood—brain barrier, and reperfusion injury (Meden et al., 1993; Chan, 1994; del Zoppo, 1994; Overgaard, 1994; Phillis, 1994; Jiang et al., 1995; Midori et al., 1997). Neuroprotectant drugs have enhanced the thrombolytic treatment either by improving recanalization or by reducing adverse secondary effects of thrombolysis (Donley and Sundt, 1973; Selman et al., 1982; Hubbard and Sundt, 1983; Meden et al., 1993; Overgaard et al., 1993b; Yenari et al., 1997).
Magnetic resonance imaging can characterize the response of ischemic brain to cytoprotective drugs. The beneficial effect of neuroprotective drugs are reflected in the early reduction of areas of abnormal DWI and infarction (Buchan et al., 1992; Lo et al., 1994, 1997; Sauer et al., 1994). Magnetic resonance imaging also is useful in detecting the beneficial effect of combined thrombolytic and neuroprotective therapies (Yenari et al., 1997). Our data are consistent with these studies in demonstrating the sensitivity of MRI to detect therapeutic efficacy early after treatment.
Our data demonstrate the application of MRI to a model of cerebral embolization and suggest that DWI, T2WI, and PWI may be beneficial in monitoring the efficacy of thrombolytic intervention.
Footnotes
Abbreviations used
Acknowledgments
The authors thank Ping Jiang, Upul Senaratne, Paul Wu, and Jonathan Prostak for data analysis, and Denice Janus for manuscript preparation.