Abstract
Cerebral ischemia models using mice have drawn increasing attention, particularly because of the availability of transgenic animals. However, the variability of intracranial vasculature at the circle of Willis in mice can influence the degree of ischemia in both the bilateral common carotid artery (CCA) occlusion and intraluminal suture occlusion models. We have developed a method to predict the extent of the anastomosis between carotid and vertebrobasilar circulation in three mouse strains (C57BL/6, CBA, and DBA/2) by measuring cortical microperfusion with laser Doppler flowmetry during a 1-minute occlusion of both CCA. When animals showed residual cortical microperfusion of less than 12% during bilateral CCA occlusion, the mice showed absence of functional anastomosis, developed ATP depletion in the frontal cortex during occlusion, and had ischemic neuronal death in the hippocampus and caudoputamen after occlusion for 15 minutes and recirculation for 7 days. Furthermore, those mice exhibited decreased local cerebral blood flow and associated ischemic neuronal death in the hippocampus, within the territory supplied by the posterior cerebral artery, with the intraluminal suture occlusion model. The current study demonstrates the need for assessment of intracranial vasculature in each animal by measuring cortical microperfusion during temporary occlusion of both CCA, no matter whether cerebral ischemia is produced by bilateral CCA occlusion or intraluminal suture occlusion in transgenic mice.
Keywords
The patency of the posterior communicating artery (PComA) has been regarded as a crucial factor in the development of forebrain ischemia after bilateral common carotid artery (CCA) occlusion in gerbils (Levine and Sohn, 1969; Kahn, 1972; Berry et al., 1975). In mice, strain differences have been observed regarding the patency of the PComA at the circle of Willis (Barone et al., 1993; Yang et al., 1997). Although the territory supplied by the posterior cerebral artery (PCA), including the hippocampus and thalamus, is expected to experience ischemia using the intraluminal suture occlusion model in the absence of collateral circulation through a patent PComA (Fig. 1), the importance of the PComA has not been critically evaluated in the context of an intraluminal suture occlusion model to produce occlusion of the middle cerebral artery (MCA). In the current study, we provide evidence that patency of the PComA is a crucial determinant of the development of bilateral forebrain ischemia after bilateral CCA occlusion as well as ischemia in the PCA territory using the intraluminal suture occlusion model in three murine strains, including C57BL/6, CBA, and DBA/2. We therefore developed a procedure to predict the patency of the PComA and found that this procedure would be useful for the reproducible induction of cerebral ischemia in transgenic mice.
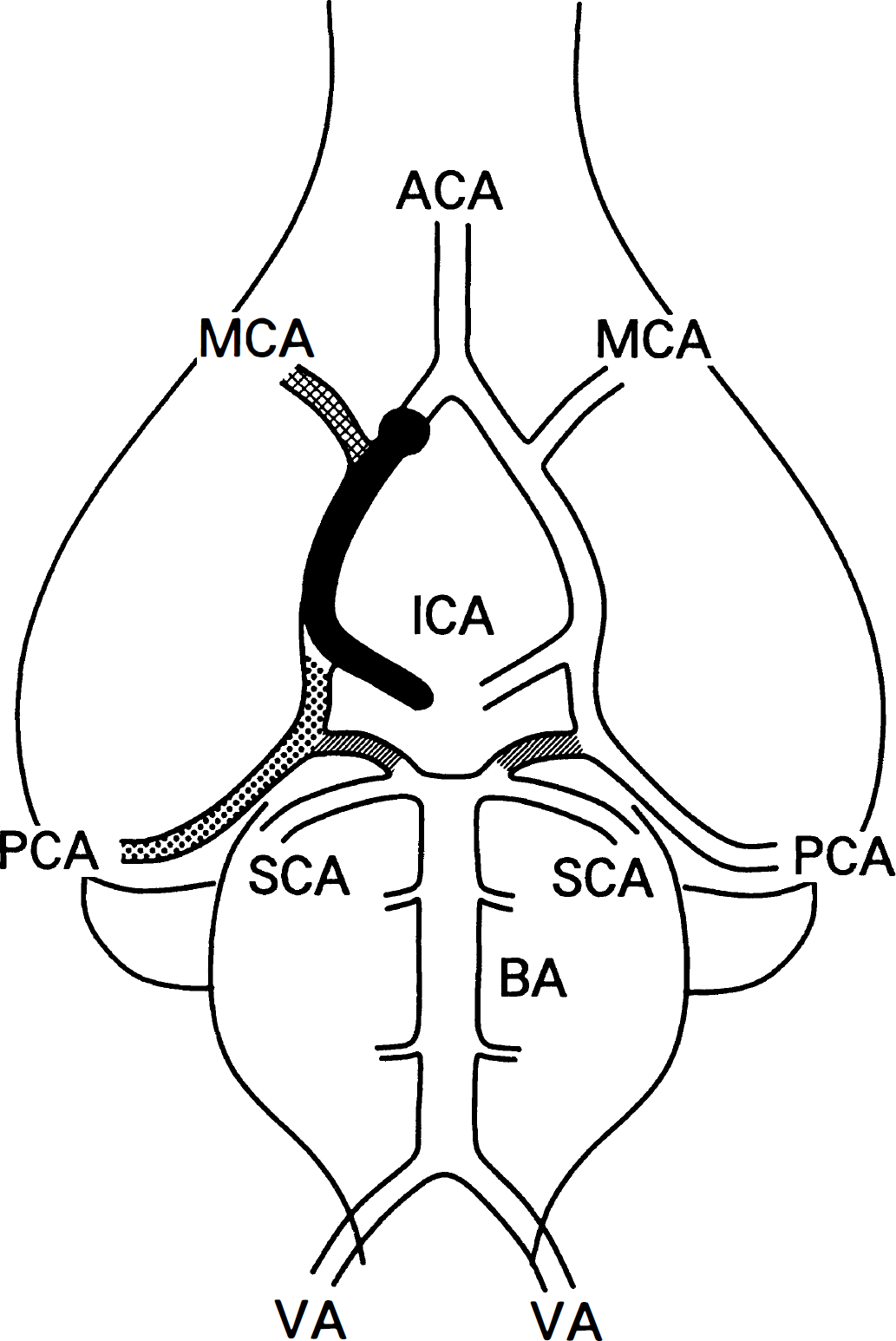
Diagram of the vasculature at the circle of Willis when a nylon suture is advanced from the internal carotid artery (ICA) into the anterior cerebral artery (ACA). Posterior communicating arteries (PComA) are shown by shading. The middle cerebral artery (MCA; crosshatch) territory ipsilateral to the inserted suture experiences ischemia. In addition, the posterior cerebral artery (PCA; dots) is supplied through the PComA during suture insertion, and the PCA territory theoretically experiences ischemia when the PComA is absent on the same side. BA, basilar artery; SCA, superior cerebellar artery; VA, vertebral artery.
MATERIALS AND METHODS
Three mouse strains were obtained from Charles River, Inc. (Yokohama, Kanagawa, Japan): C57BL/6NCrj (C57BL/6), CBA/jNCrj (CBA), and DBA/2NCrj (DBA/2). All mice were mature males aged 8 to 16 weeks, weighing 22.5 ± 0.4 g (C57BL/6, n=46), 21.8 ± 0.6 g (CBA, n=27), and 22.8 ± 0.4 g (DBA/2, n = 31). Mice were given free access to food and water before surgery. The procedures involving laboratory animals have been approved by the Institutional Animal Care and Use Committee of the Osaka University Medical School. General anesthesia was induced with 2.0% halothane and maintained with 0.5% halothane by means of an open-face mask. A polyacrylamide column with an inner diameter of 0.8 mm for measurement of cortical microperfusion by laser Doppler flowmetry (LDF; Advance Laser Flowmetry, model ALF-21, Advance Co., Ltd., Tokyo, Japan) was attached to the intact skull, 3.5 mm right to the bregma with dental cement. A metal plate type thermometer with a diameter of 3.0 mm also was attached to the skull over the right cerebral cortex to record skull temperature. To monitor blood pressure during ischemia and reperfusion, the right femoral artery was cannulated with a PE-10 polyethylene tube and connected to a pressure transducer attached to a calibrated polygraph. Then, both CCA were exposed in the neck and occluded with microaneurysm clips for 1 minute and reperfused. Cortical microperfusion was monitored continuously by LDF and expressed as a percentage of the average of three baseline values. After the measurement of the cortical microperfusion during 1 minute of bilateral CCA occlusion, the mice underwent the experimental manipulations outlined later. Body and skull temperatures were monitored and maintained at 36.0° to 37.5°C using a heat lamp. The heat lamp and the surgical microscope lamp were turned off when cortical microperfusion was measured with LDF.
Investigation of intracranial vasculature
Five mice from each of the three strains were used. Each mouse was deeply anesthetized with sodium pentobarbital and a complete sternotomy was performed to expose the heart. An incision was made in the right atrium, and the mouse was perfused with a short flush of saline solution and then with 10% formalin, followed by a mixture of gelatin and India ink, prepared according to Berry et al. (1975). The brains were carefully removed and placed in 10% formalin. The circle of Willis then was examined under a dissecting microscope, and the PComA was identified as the connection between carotid and vertebrobasilar circulation. The degree of anastomosis was assessed by comparing the diameter of the PComA on each side with the diameter of the basilar artery, and graded as 0 or 1. A degree of 0 was assigned to specimens having no anastomosis or less than one third of the diameter of the basilar artery, and a grade of 1 was assigned when the artery diameter was more than one third. The sum of the scores from both sides was determined as 0, 1, or 2. Three adult Mongolian gerbils and three Wistar rats also were used for comparison of intracranial vasculature.
Measurement of the tissue ATP content after bilateral common carotid artery occlusion
Five mice from each of the three strains were used. Both CCA were occluded for 5 minutes, and the mice were decapitated and each head was frozen in liquid nitrogen instantaneously. Another group of five mice from each strain were used as unoperated control and processed in the same way. To determine whether 1 minute of bilateral CCA occlusion caused the tissue ATP depletion, another five C57BL/6 mice were decapitated after 1 minute of bilateral CCA occlusion and processed in the same way. The frozen brains were chiseled out at -25°C, and the samples were dissected from the right frontal cortex and stored at -80°C until analysis. Extraction and measurement of ATP was done using a modification of the luciferase method of Lust and others (1981). Each frozen brain sample was placed in 3 mL of ice-cold 0.5 mol/L perchloric acid and immediately homogenized. The homogenate was centrifuged at 10,000 g for 30 minutes, and the pellet was dissolved in 3 mL of 0.1 mol/L sodium hydroperoxide. The protein content was determined with bovine serum albumin as control (Bio-Rad protein assay kit; Bio-Rad, Hercules, CA). After neutralization of the supernatant with 10 mol/L potassium hydroxide, the resultant precipitate was removed by centrifugation and the supernatant was used as the test sample. The ATP concentration in the solution was measured with luciferase (ATP bioluminescence CLS; Boehringer Mannheim, Indianapolis, IN, U.S.A.) and a Lumat LB9501 (Berthold Company, Wildbad, Germany). Integrated luminescent emission was counted for 10 seconds, and the emission count was converted to the ATP concentration in each sample using a logarithmic plot of the count versus the control ATP concentration (Stanley and Williams, 1969).
Ischemic neuronal death after transient bilateral common carotid artery occlusion
Six mice from each strain were subjected to occlusion of both CCA for 15 minutes. If a mouse died during ischemia or reperfusion, a replacement mouse was operated on to maintain six mice in each strain. Body and skull temperatures were monitored and maintained at 36.0° to 37.5°C during occlusion and until recirculation for 15 minutes. Cortical microperfusion was continuously monitored by LDF and recorded before occlusion, as well as 1, 5, 10, and 15 minutes after occlusion, and again at 5 and 15 minutes after recirculation. Mean arterial blood pressure also was continuously monitored and kept above 70 mm Hg by changing the concentration of halothane. Each mouse was allowed to recover and was kept for a day in a chamber where the ambient temperature was maintained at 35°C to prevent hypothermia. To determine whether 1 minute of bilateral CCA occlusion caused neuronal damage, another five C57BL/6 mice were subjected to occlusion of both CCA for 1 minute and allowed to recover for 7 days. On day 7, the mice were decapitated under ether anesthesia and their brains were removed, fixed by immersion into the alcohol–5% acetic acid solution for 5 hours at 4°C, dehydrated, and embedded in paraffin as described previously (Yanagihara et al., 1985). Tissue sections (5 μm) encompassing the central caudoputamen, 3-mm caudal to the frontal pole, and the hippocampus, 5-mm caudal to the frontal pole, were examined using conventional staining with hematoxylin–eosin and cresyl violet, as well as an immunohistochemical staining technique for microtubule-associated protein 2 (MAP2) (Kitagawa et al., 1989).
Intraluminal occlusion of the intracranial artery by nylon suture
Intraluminal occlusion of the intracranial artery including internal carotid artery and MCA by suture was accomplished using the method described previously for rats (Koizumi et al., 1986; Zea-Longa et al., 1989) and maintained under continuous monitoring of cortical microperfusion by LDF. The right CCA was exposed and ligated, and the external carotid artery was isolated and ligated. A 6–0 monofilament nylon suture, blunted at the tip, was introduced through the carotid bifurcation into the internal carotid artery and advanced until LDF revealed a sudden drop in cortical microperfusion over the MCA territory. The distance from the tip of the suture to the bifurcation of the CCA was 9.0 ± 1.0 mm in each mouse. Body and skull temperature as well as blood pressure were monitored and maintained during occlusion. After occlusion for 30 minutes (C57BL/6, n=6) or 60 minutes (CBA and DBA/2, n = 6 each), blood flow was restored through the anterior cerebral artery (ACA) and the PComA by removal of the nylon suture. If a mouse died, a replacement mouse was operated on to maintain six mice in each strain. We used a 30-minute occlusion time in the C57BL/6 mice because intraluminal suture occlusion for 60 minutes caused more than 80% mortality in the C57BL/6 mice in a preliminary study. In another five C57BL/6 mice, the right CCA was exposed and occluded with a microaneurysm clip during suture insertion. A nylon suture was inserted through the external carotid artery stump. After occlusion for 30 minutes, the nylon suture was removed, and cerebral blood flow was restored through the ipsilateral CCA after a 30-minute occlusion. Two days after the occlusion procedure, each mouse was decapitated and its brain was processed for histologic examination as described earlier.
The autoradiographic technique described by Sakurada and associates (1979) was used to examine the distribution pattern of the local cerebral blood flow during suture occlusion in the brains of the C57BL/6 and DBA/2 (n = 4 each) mice. A femoral artery and a saphenous vein were cannulated with PE-10 polyethylene tubes filled with saline containing 50 IU heparin/mL in each mouse. The nylon suture was introduced into the ACA through the carotid bifurcation under halothane anesthesia. After occlusion for 30 minutes, measurement of the mean arterial pressure and blood sampling (150 μL) for gas analysis were carried out through the arterial cannula. Ten microcuries [14C]iodoantipyrine (Amersham, Arlington Heights, IL, U.S.A.) in 0.1 mL of physiologic saline then was infused through the saphenous vein for 1 minute. Blood pH, Pa
Statistics
All values reported here are expressed as means ± SD. Overall statistical significance of differences among groups was tested by a one-way analysis of variance. P<0.05 was considered significant.
RESULTS
Vascular anatomy at the circle of Willis
The patency of the PComA on each side, as visualized by India ink perfusion (Fig. 2), and the change in cortical microperfusion, as detected by LDF, during bilateral CCA occlusion for 1 minute are summarized in Fig. 3. Bilateral CCA occlusion resulted in less than 12% of baseline cortical microperfusion in four of five of the C57BL/6 mice and one CBA mouse. In those five mice, no patent PComA was identified on either side (Fig. 2A and Fig. 3), as was also the case in the gerbils (Fig. 2D). In contrast, all five of the DBA/2 mice maintained more than 23% of baseline cortical microperfusion during bilateral CCA occlusion and the patent PComA was identified on each side (Fig. 2C and Fig. 3), as was the case in the Wistar rats (Fig. 2E). In the CBA mice, the percentage of cortical microperfusion during bilateral CCA occlusion varied from 7% to 52% of baseline, and the patency of the PComA was highly variable (Fig. 2B and Fig. 3).
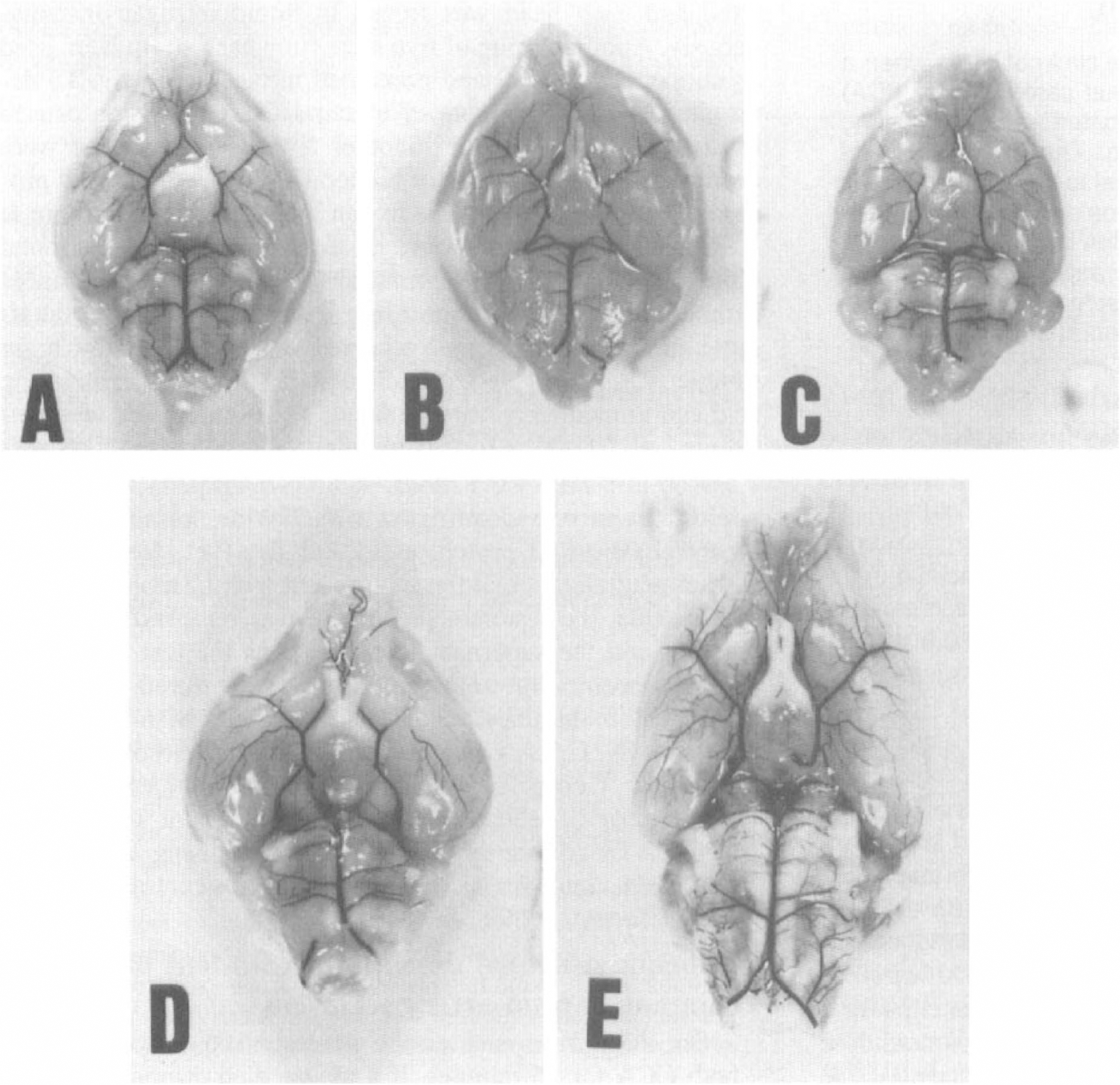
Brain vasculature at the circle of Willis in three mouse strains (
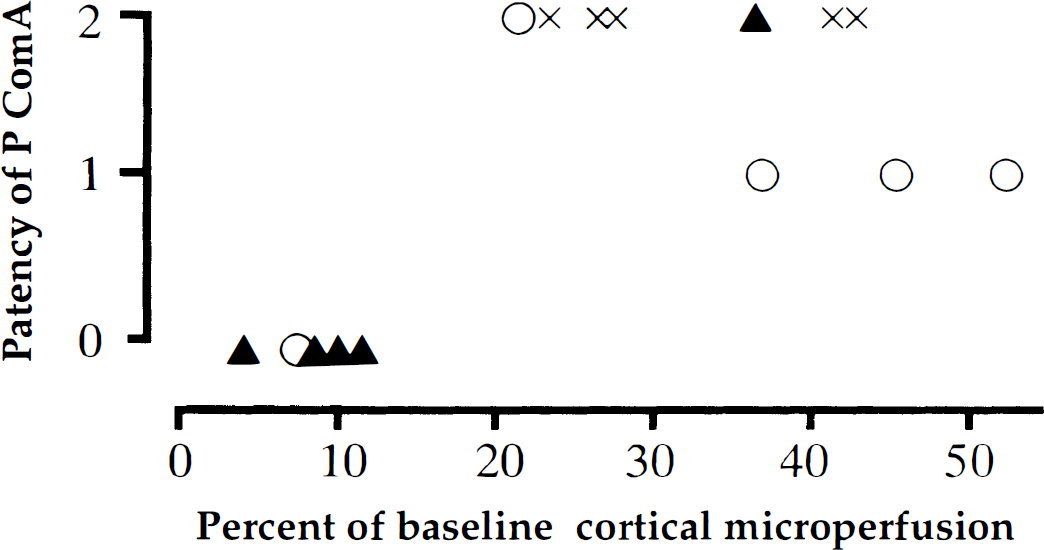
Relation between the patency of the PComA and the reduction in cortical microperfusion during temporary bilateral common carotid artery (CCA) occlusion for 1 minute. All mice with less than 12% of baseline cortical microperfusion during temporary bilateral CCA occlusion had no patent PComA on either side. A patent PComA was detected on at least one side when mice showed more than 20% of baseline cortical microperfusion during temporary bilateral CCA occlusion. ▴, C57BL/6; ○, CBA; ×, DBA/2.
Tissue ATP content after bilateral common carotid artery occlusion
The tissue ATP content of the frontal cortex was 28.5 ± 3.4, 27.8 ± 2.9, and 28.0 ± 3.3 n mol/mg protein in the control C57BL/6, CBA, and DBA/2 mice, respectively (Fig. 4), and they compared well with published values (Nowak et al., 1985). In all mice that showed less than 10% of baseline cortical microperfusion during temporary bilateral CCA occlusion, including four of the C57BL/6 mice and one CBA mouse, the tissue ATP levels fell to essentially zero after 5-minute bilateral CCA occlusion (Fig. 4). On the other hand, in mice that maintained greater than 20% of the baseline cortical microperfusion during bilateral CCA occlusion, including all of the DBA/2 mice, four of the CBA mice, and one C57BL/6 mouse, the ATP level was preserved in the normal range in the frontal cortex after 5-minute bilateral CCA occlusion (Fig. 4). The tissue ATP level was preserved after 1-minute bilateral CCA occlusion, even if the mice showed less than 10% of baseline cortical microperfusion.
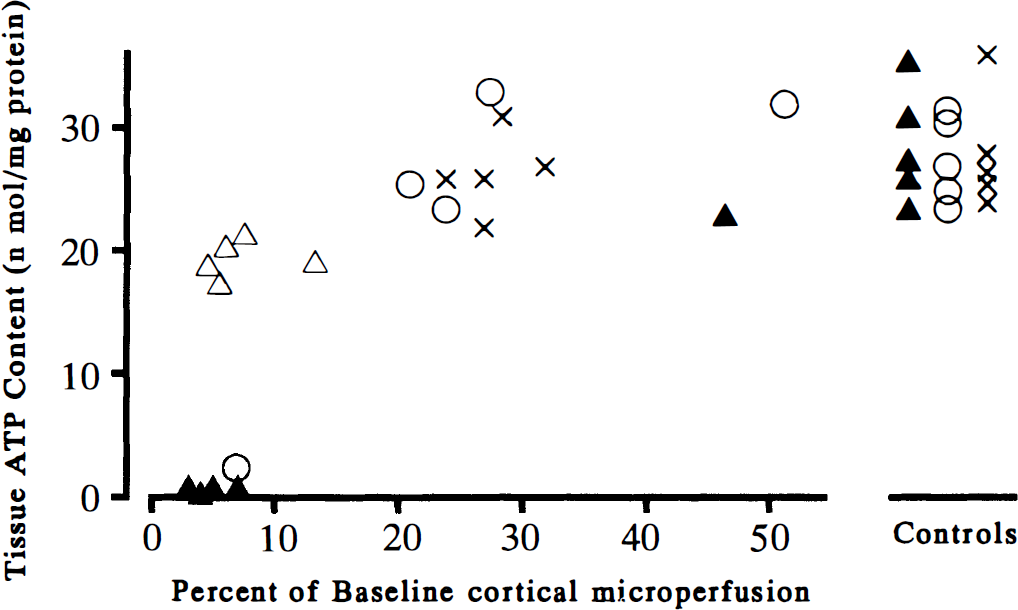
Relation between the tissue ATP content and the reduction in cortical microperfusion during bilateral CCA occlusion. All mice with less than 10% of baseline cortical microperfusion during bilateral CCA occlusion for 1 minute showed the tissue ATP level falling almost to zero after bilateral CCA occlusion for 5 minutes. However, all mice with more than 20% of baseline cortical microperfusion during bilateral CCA occlusion had the tissue ATP content preserved in normal range, as shown on the right side. The tissue ATP level was preserved after 1-minute bilateral CCA occlusion, even if the mice showed less than 10% of baseline cortical microperfusion. ▴, C57BL/6; △, C57BL/6 (1 minute); ○, CBA; ×, DBA/2.
Histologic examination after bilateral common carotid artery occlusion for 15 minutes and reperfusion
Changes in cortical perfusion at 1 minute after bilateral CCA occlusion was closely correlated with those at 15 minutes after bilateral CCA occlusion (Table 1). Most mice that showed about 10% of baseline cortical microperfusion at 1 minute after bilateral CCA occlusion developed further decreases to around 5% at 15 minutes after bilateral CCA occlusion. Blood pressure elevated soon after bilateral CCA occlusion and dropped suddenly after removal of the carotid clips (Table 1).
Physiologic conditions in the bilateral carotid occlusion model
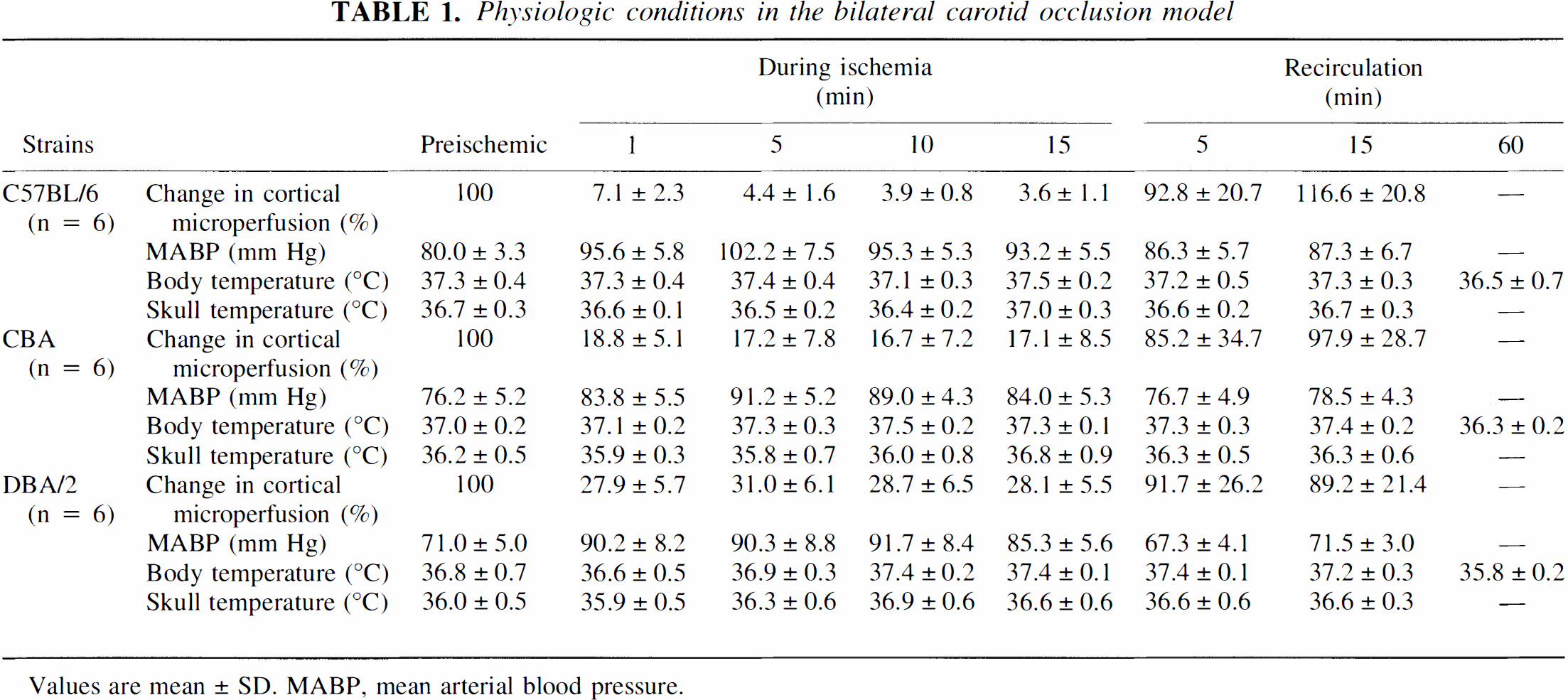
Values are mean ± SD. MABP, mean arterial blood pressure.
Two of eight C57BL/6 mice died during recirculation, and all CBA and DBA/2 mice survived for 7 days. All C57BL/6 mice with less than 12% of baseline cortical microperfusion during bilateral CCA occlusion for 1 minute developed ischemic neuronal death in the hippocampus or caudoputamen after bilateral CCA occlusion for 15 minutes and reperfusion for 7 days (Fig. 5 and Fig. 6). Four of six C57BL/6 mice that survived for 7 days had ischemic lesions bilaterally, and the two remaining mice showed them unilaterally (Fig. 6). Four of six CBA mice with less than 23% of baseline cortical microperfusion during bilateral CCA occlusion for 1 minute showed ischemic lesions in the hippocampus or caudoputamen unilaterally. Two of six CBA mice, with 18% and 26% of baseline cortical microperfusion, respectively, exhibited no neuronal death (Fig. 5 and Fig. 6). All of the DBA/2 mice with more than 20% of baseline cortical microperfusion exhibited no neuronal death 7 days later. No neuronal death was observed in the hippocampus and caudoputamen 7 days after 1 minute bilateral CCA occlusion, even if the mice showed less than 10% of baseline cortical microperfusion.
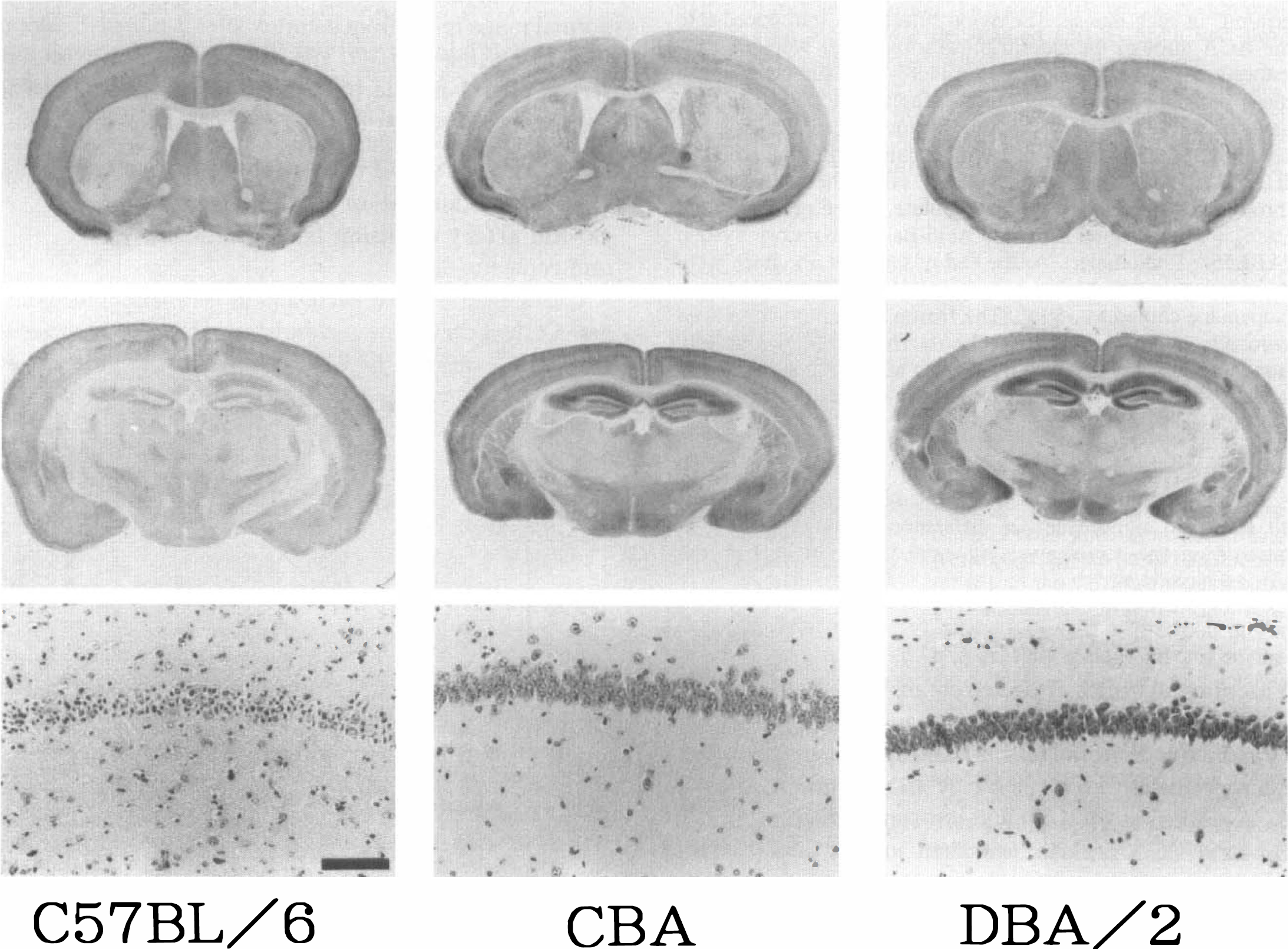
Histologic findings after bilateral CCA occlusion for 15 minutes and reperfusion for 7 days in C57BL/6, CBA, and DBA/2 mice. The top and middle panels show the immunoreactivity for microtubule-associated protein 2 (MAP2) in the coronal section at the level of the caudoputamen and the hippocampus, respectively. The bottom panel shows cresyl violet staining in the right hippocampal CA1 sector. In the C57BL/6 mouse brain, ischemic lesions with loss of MAP2 immunoreactivity are seen in the caudoputamen and hippocampus bilaterally. In the CBA mouse brain, the ischemic lesion is detected in the caudoputamen unilaterally. No ischemic lesion is visualized in the DBA/2 mouse brain. Bar = 0.1 mm.
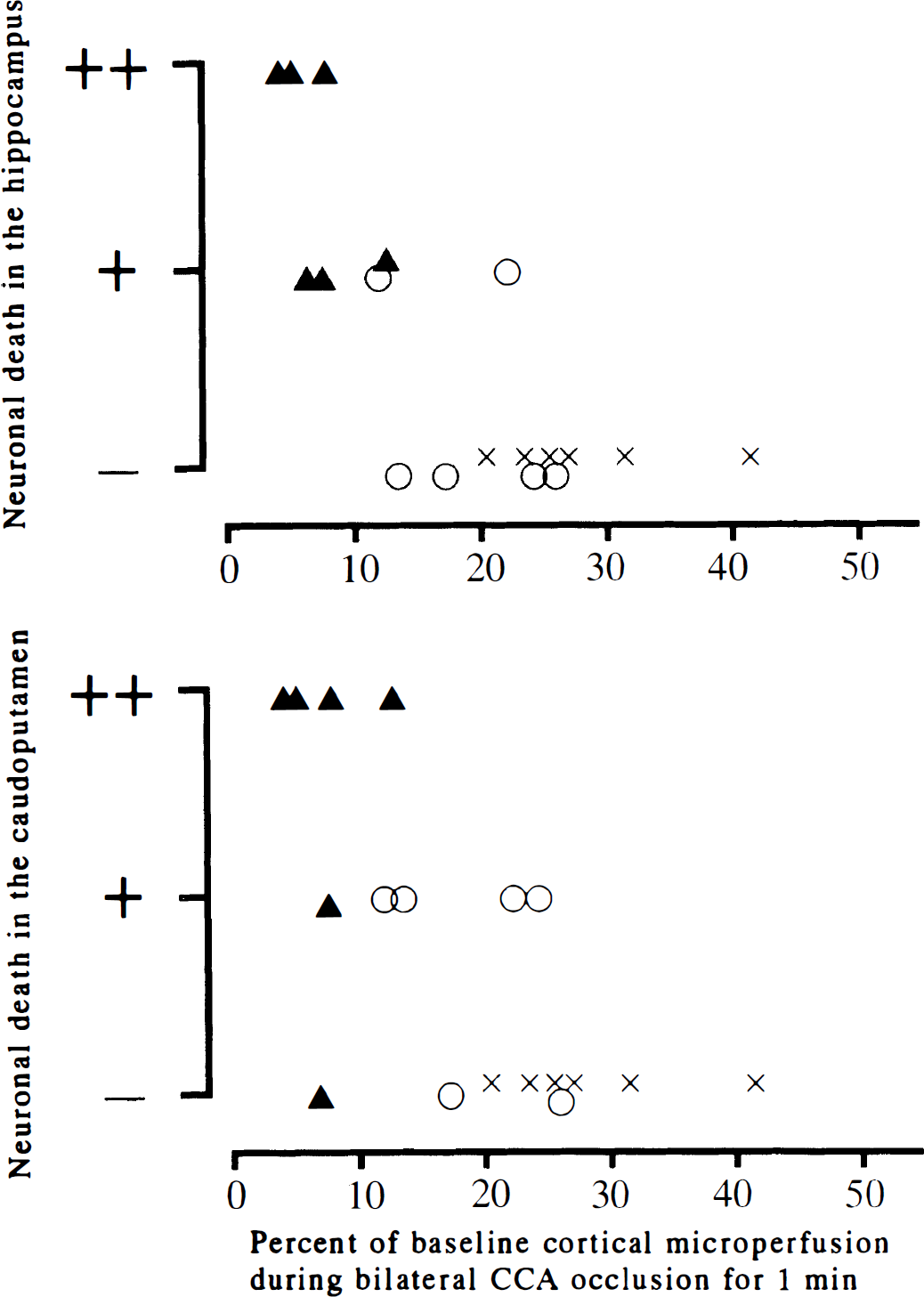
Relation between the histologic outcome and reduction in cortical microperfusion during bilateral CCA occlusion. The upper panel shows the histologic outcome in the hippocampus, and the lower panel shows that in the caudoputamen. All mice with less than 12% of baseline cortical microperfusion during bilateral CCA occlusion for 1 minute showed ischemic neuronal death on at least one side of the hippocampus or the caudoputamen after 15-minute bilateral CCA occlusion. ++, neuronal death found bilaterally; +, neuronal death found unilaterally; –, no neuronal death; ▴, C57BL/6; ○, CBA; ×, DBA/2.
Ischemic lesions using the intraluminal suture occlusion model
All mice subjected to the intraluminal suture occlusion method demonstrated a sudden drop in cortical microperfusion immediately after occlusion (Table 2) and developed ischemic lesions in the caudoputamen and cerebral cortex territory supplied by the MCA (Fig. 7). Percentage of baseline cortical microperfusion during suture occlusion was 20.4%, 18.1%, and 29.3% after 5 minutes in C57BL/6, CBA, and DBA/2 strains, respectively. Two of eight C57BL/6 mice and one of seven CBA mice died during recirculation. Notably, mice that showed less than 12% of baseline cortical microperfusion during bilateral CCA occlusion for 1 minute, including all of the C57BL/6 mice and one of six CBA mice, developed ischemic neuronal death in the ipsilateral hippocampus (Fig. 7 and Fig. 8). Another CBA mouse developed neuronal death in the hippocampus despite maintaining 21% of baseline cortical microperfusion during bilateral CCA occlusion for 1 minute. All of the DBA/2 mice and three of six CBA mice maintained more than 20% of baseline cortical microperfusion during bilateral CCA occlusion for 1 minute, and the ischemic lesions were confined to the caudoputamen and cerebral cortex (Fig. 8). A separate group of five C57BL/6 mice subjected to intraluminal suture occlusion for 30 minutes without CCA ligation showed neuronal death in the hippocampus, caudoputamen, and cerebral cortex after reperfusion through the ipsilateral CCA, ACA, and PcomA (Fig. 8).
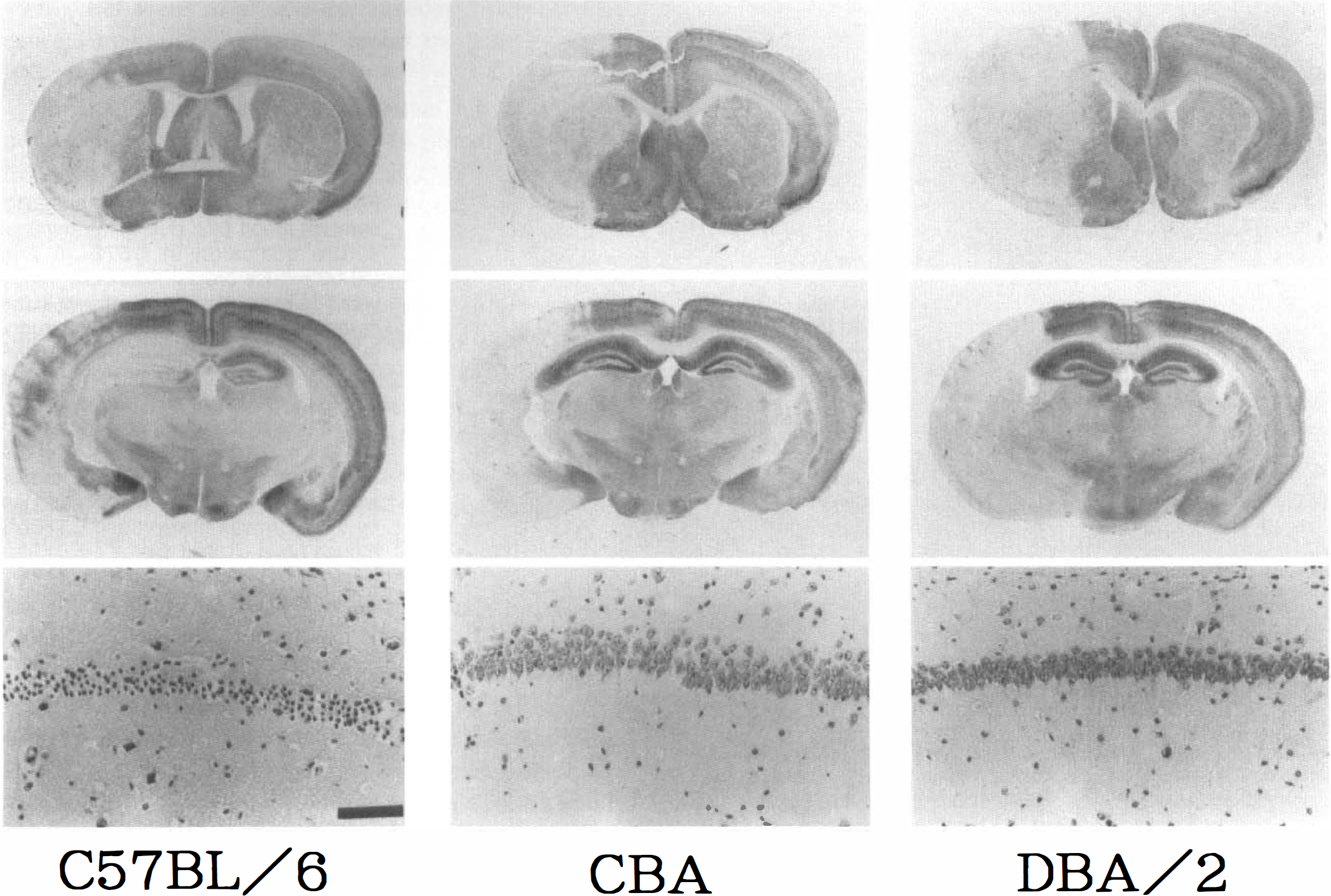
Histologic findings after intraluminal suture occlusion in C57BL/6, CBA, and DBA/2 mice. The top and middle panels show immunoreactivity to MAP2 in the coronal sections at the level of the caudoputamen and the hippocampus, respectively. The bottom panel shows cresyl violet staining in the right hippocampal CA1 sector. In the C57BL/6 mouse brain, ischemic lesions with loss of MAP2 immunoreactivity are seen not only in the caudoputamen and cerebral cortex but also in the hippocampus, where neurons are necrotic. In the CBA and DBA/2 mouse brains, ischemic lesions are confined to the caudoputamen and cerebral cortex and the hippocampal neurons remain intact. Bar = 0.1 mm.
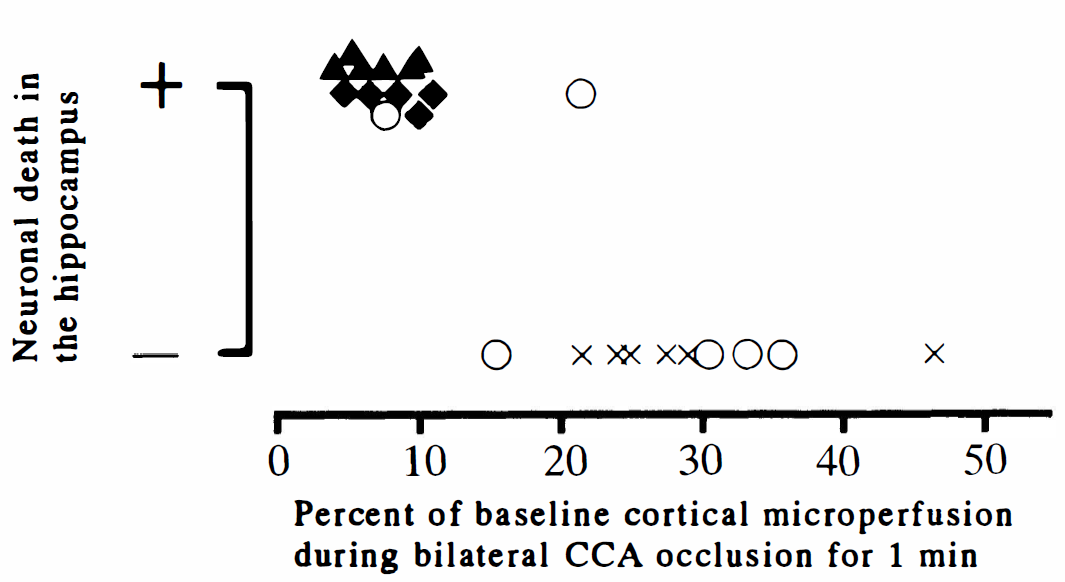
Relation between the histologic outcome in the hippocampus after intraluminal suture occlusion and the reduction in cortical microperfusion during temporary bilateral CCA occlusion for 1 minute. All mice with less than 12% of baseline cortical microperfusion during temporary bilateral CCA occlusion for 1 minute had ischemic neuronal death in the hippocampus in addition to the MCA territory after intraluminal suture occlusion. +, neuronal death in the hippocampus; –, no neuronal death in the hippocampus; ▴, C57BL/6; ⋄, C57BL/6 with insertion of nylon suture from the external carotid artery stump without CCA ligation and reperfusion through the ipsilateral CCA, ACA, and possibly PComA; ○, CBA; ×, DBA/2.
Physiologic conditions in the intraluminal suture occlusion model
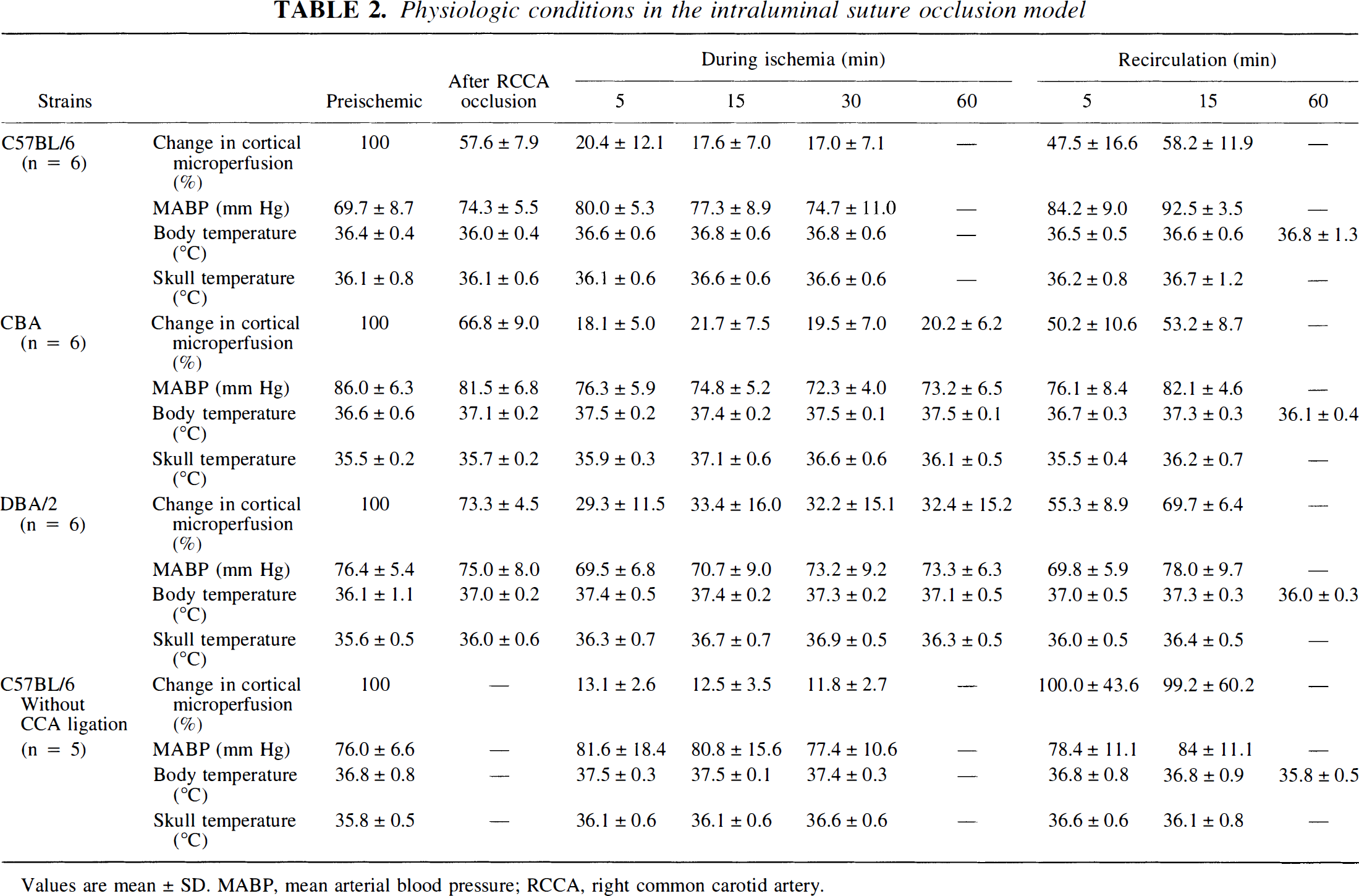
Values are mean ± SD. MABP, mean arterial blood pressure; RCCA, right common carotid artery.
The physiologic condition was determined before infusion of [14C]iodoantipyrine for mean arterial blood pressure (C57BL/6, 78.0 ± 2.5 mm Hg; DBA/2, 79.3 ± 1.8 mm Hg), P
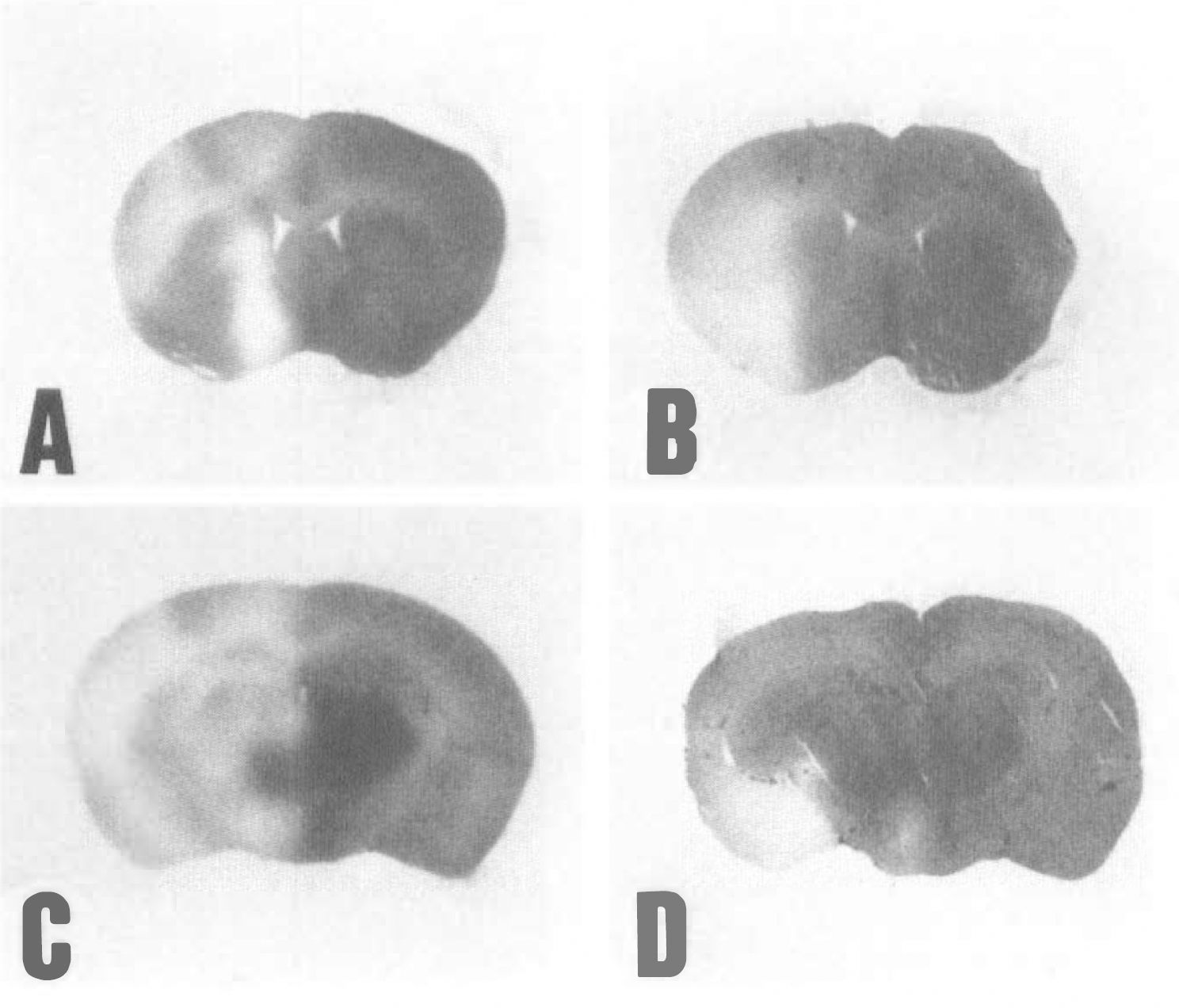
The distribution pattern of local cerebral blood flow during intraluminal suture occlusion in C57BL/6 and DBA/2 mouse brains. Autoradiography with [14C]iodoantipyrine is shown in the coronal slices at the level of the caudoputamen (
DISCUSSION
This study demonstrates that differences in vascular anatomy must be considered when cerebral ischemia is produced in a transgenic or knockout mouse using either bilateral CCA occlusion or the intraluminal suture occlusion. The degree of anastomosis between carotid and vertebrobasilar circulation at the circle of Willis is crucial to determine whether bilateral CCA occlusion produces global forebrain ischemia (Levine and Sohn, 1969; Kahn 1972; Berry et al., 1975). Although rats have this anastomosis (Fig. 2E) and are thus resistant to ischemia caused by bilateral CCA occlusion, gerbils lack such an anastomosis (Fig. 2D) and are highly susceptible to ischemia from bilateral CCA occlusion. In mice, the anastomosis is highly variable and strain-dependent, as shown by Barone et al. (1993) and Yang et al. (1997). The intraluminal suture occlusion model originally was developed by Koizumi et al. (1986) and subsequently modified by Zea-Longa et al. (1989) to produce ischemia only in the MCA territory in rats. In the current study, cortical microperfusion over the MCA territory during suture occlusion was 20.4%, 18.1%, and 29.3% after 5 minutes in C57BL/6, CBA, and DBA/2 strains, respectively. The first two values compared well with published values for ischemic core using LDF (Huang et al., 1994; Yang et al., 1994; Kamii et al., 1994). The last one for the DBA/2 strain was a little higher than the published values for ischemic core, but lower than that for periischemic penumbra area (Huang et al., 1994; Yang et al., 1994). When the suture enters into the ACA and shuts off the blood flow from the contralateral carotid circulation, the ipsilateral PCA is supplied mainly by the basilar artery through the PComA (Fig. 1). When the PComA is absent or poorly developed, the PCA territory (i.e., the hippocampus and thalamus) would experience ischemia by insertion of the suture material into the ACA. Gerbils that lack the connection between carotid and vertebrobasilar circulation, as well as that between right and left carotid circulation, undergo hemispheric ischemia, including damage to the hippocampus and thalamus, during unilateral carotid occlusion (Berry et al., 1975; Matsumoto et al., 1988; Kitagawa et al., 1989). This supports the notion that the patency of the PComA is crucial in determining the ischemic area after intraluminal suture occlusion. Therefore, evaluation of the patency of the PComA in each mouse is important when the intraluminal suture occlusion model is used and the extent of ischemic lesions is compared between transgenic and control wild mice. In addition to the tissue ATP assay, histologic examination, and autoradiographic technique, we established a method to predict vascular anatomy and the extent of ischemia after bilateral carotid occlusion or intraluminal suture occlusion in the current study, since the extent of reduction in cortical microperfusion during bilateral CCA occlusion for 1 minute was found to be a reliable predictor of the patency of the PComA. Bilateral CCA occlusion for 1 minute caused neither ATP depletion (Fig. 4), as reported previously in complete compression ischemia in the rat (Ljunggren et al., 1974), nor neuronal death, as shown in the results. It is also unlikely that 1-minute ischemia shows preconditioning effect because 1-minute ischemia never induced tolerance against subsequent ischemia in gerbils (Kitagawa et al., 1990). When mice of three strains show less than 10% to 15% of baseline cortical microperfusion during temporary bilateral CCA occlusion for 1 minute, they will show depletion of tissue ATP after 5-minute bilateral CCA occlusion and ischemic neuronal death 7 days after 15-minute bilateral CCA occlusion, and will show severe reduction of hemispheric cerebral blood flow during intraluminal suture occlusion and ischemic neuronal death in the caudoputamen, cerebral cortex, and hippocampus after reperfusion. However, the exact correlation between the extent of reduction in cortical microperfusion during temporary bilateral CCA occlusion and patency of PcomA or the extent of ischemic neuronal death after intraluminal suture occlusion has to be evaluated in each experiment when this method is applied in the intraluminal suture model of transgenic and wild mice. Furthermore, this method was not sensitive enough to discriminate between mice with sufficient anastomosis on one side versus both sides (Fig. 3). The laterality of the anastomosis does not matter in bilateral CCA occlusion, but it can cause variability of ischemia in the intraluminal suture occlusion model because the ipsilateral PCA territory theoretically experiences ischemia when the suture is introduced into the ACA on the side lacking anastomosis. There are potentially two ways to resolve this variability. The first and reliable method is to examine the vascular anatomy of sufficient numbers of transgenic and wild mice and confirm that the PComA are patent on both sides in essentially all transgenic and wild mice. There are variabilities in the patency of the PComA at the circle of Willis among mouse strains, including BALB/c, ICR, CBA, and C3H strains, whose genetic backgrounds have been used to produce transgenic mice (Barone et al., 1993; Yang et al., 1997). The second and more practical approach is to examine the hippocampus histologically in all mice because hippocampal neurons are highly vulnerable to ischemia and they should degenerate when the PCA territory experiences ischemia (Fig. 7 and Fig. 8).
In the current study, we inserted the suture mainly through the carotid bifurcation instead of the external carotid artery stump, and ligated the ipsilateral CCA because it is an easier and more sterile approach. Unilateral CCA occlusion usually resulted in reduction of cortical microperfusion to 40% to 80% of the baseline in the ipsilateral cerebral cortex (Table 2). It is likely that the perfusion pressure during reperfusion through the ACA and PComA alone is lower than that through the ipsilateral CCA, ACA, and PComA together. However, ligation of the ipsilateral CCA did not alter ischemia in the PCA territory critically because all five additional C57BL/6 mice that were reperfused through the ipsilateral CCA also showed neuronal death in the hippocampus (Fig. 8). We recorded and maintained the skull temperature over the ischemic cortex to keep up the brain temperature in addition to the blood pressure and the body temperature in both models because the skull was exposed to room air for LDF. We again demonstrated that the C57BL/6 strain has the poorest patency of the PComA, as was shown in our recent study (Yang et al., 1997), but there may be some breeder differences because Panahian et al. (1996) report the patency of the PComA in C57BL/6 mice.
The occlusion period of intraluminal suture model for producing infarction varied from 45 minutes to 3 hours in published reports (Connolly et al., 1996; Bruce et al., 1996; Yang et al., 1994; Hara et al., 1996). In this experiment, 30-minute occlusion consistently caused ischemic lesion in the cerebral cortex and caudoputamen, and 60-minute occlusion showed the same ischemic lesion but also exhibited more than 80% mortality in the C57BL/6 mice. High mortality rate after prolonged ischemia in C57BL/6 mice was consistent with the published mortality rate using the intraluminal suture model with prolonged ischemia in rats (Zea-Longa et al., 1989; Nagasawa et al., 1989) and mouse (Connolly et al., 1996). In mice lacking patent PcomA like the C57BL/6 strain in the current study, the intraluminal suture model occludes not only MCA but also PCA and causes severe hemispheric ischemia, therefore 30-minute ischemia is enough to produce infarction like the gerbil model of hemispheric ischemia (Kitagawa et al., 1991). On the contrary, in mice having patent PcomA like CBA and DBA/2 strains, 60-minute occlusion showed ischemic lesion in the MCA territory with less than 15% mortality. As shown in Fig. 9, collateral blood supply through the ACA during suture occlusion also is an important factor to determine ischemic severity in this model, as reported in the rat (Nagasawa et al., 1989).
In conclusion, the vascular anatomy at the circle of Willis is highly variable in mice, and the patency of the PComA is a crucial determinant of the ischemic effects of either bilateral CCA occlusion or intraluminal suture occlusion models that are employed in transgenic mice. Measurement of cortical microperfusion during brief bilateral CCA occlusion is an easy and reliable method to predict forebrain ischemia after bilateral CCA occlusion and additional ischemia in the PCA territory after intraluminal suture occlusion.
Footnotes
Acknowledgments
The authors thank Miss R. Manabe and Miss N. Hinokami for secretarial assistance.
