Abstract
Postnatal rats at 7 and 21 days of age were subjected to unilateral hypoxia—ischemia (H/I) by right carotid artery ligation followed by 1.5 to 2 hours of hypoxia (8% oxygen). Brains were frozen at specific intervals of recovery from 0 to 24 hours. Western blots of samples of right and left forebrain were immunodeveloped with a monoclonal antibody specific for ubiquitin, RHUb 1. An elevation of ubiquitin conjugate levels in the right compared with the left forebrain of 7-day-old animals was detectable immediately following H/I and increased by close to 60% of control level within 1 hour of recovery. The conjugate immunoreactivity remained at this level for 6 hours but had declined to control levels by 24 hours of recovery. No such increase was observed in response to hypoxia alone. Similar changes were observed in samples from the 21-day-old rat brain. However, the elevation of ubiquitin conjugate levels was of slower onset and persisted longer than observed for the 7-day-old animals. Immunocytochemical studies of brain fixed by immersion in formaldehyde/acetone/methanol showed that ubiquitin-like immunoreactivity was increased in the right, but not left, cerebral cortex and hippocampus of animals subjected to H/I. The data suggest that elevated ubiquitination may represent a neuroprotective response to H/I.
Brain damage resulting from cerebral hypoxia—ischemia (H/I) in the perinatal period is a major cause of cerebral palsy, mental retardation, and epilepsy in the survivors (Larroche, 1977; Volpe, 1987). The damage results from a cascade of cerebral metabolic events that begins during the insult and proceeds well into the recovery period. In addition to a critical depletion of high-energy reserves, ATP, and phosphocreatine (Vannucci et al., 1989, 1994), the early recovery period is also characterized by an increased expression of immediate early and heat shock genes (Blumenfeld et al., 1992; Gubits et al., 1993; Aden et al., 1994). In the present study we have investigated the effect of unilateral cerebral H/I on protein ubiquitination and ubiquitin immunoreactivity in the immature rat.
Ubiquitin is a low molecular mass (8.5 kDa), highly conserved protein found in all eukaryotic cells (for reviews see Ciechanover, 1994; Wilkinson, 1994; Jenninsen, 1995; Muller and Schwartz, 1995; Weissman, 1997). All known functions of ubiquitin are mediated by its covalent conjugation to a wide range of target proteins through an isopeptide bond between the C-terminal glycine of ubiquitin and ε-NH2 groups of lysines present on the target proteins. A major function of ubiquitin is to target proteins for rapid degradation via a nonlysosomal pathway. The signal for this pathway is attachment of a polyubiquitin chain to the target protein, which results in its uptake by the 26S proteasome, a multicatalytic degradative complex. Ubiquitin is also suggested to target proteins for degradation via the lysosomal compartment (Laszlo et al., 1990; Landon et al., 1994). However, as some conjugates, notably histones 2A and 2B, are rapidly and reversibly ubiquitinated, ubiquitin must have other functions unrelated to protein degradation (Hunt and Dayhoff, 1977; Mueller et al., 1985; Dawson et al., 1991).
Ubiquitin, in part, functions as a heat shock protein. One of the three classes of ubiquitin genes encodes head-to-tail ubiquitin repeats and is under the control of a heat shock promoter (see Wilkinson, 1994). Increases in ubiquitin mRNA and conjugate levels have been observed in brain and in cultured neuron-like PC12 and C1300 neuroblastoma cell lines, and lens epithelia in response to several stresses including ischemia, iron loading, and exposure to hydrogen peroxide (Hayashi et al., 1991, 1992a; Dewar et al., 1993; Uney et al., 1993; Shang and Taylor, 1995). Previous studies show that ubiquitin conjugate and polyubiquitin mRNA levels are increased in the adult rat hippocampus following transient global ischemia, and elevated ubiquitin immunoreactivity is reported in several brain regions following permanent focal ischemia (Hayashi et al., 1991a; Noga and Hayashi, 1996). Both transient and permanent ischemias result in neuronal death and permanent tissue damage in the adult. The neonatal brain is more resistant to the effects of H/I and is able to tolerate longer periods of H/I without permanent brain damage, but this resistance decreases with age (Rice et al., 1981). The effects of H/I on ubiquitin conjugate levels and ubiquitin immunoreactivity in immature rat brain have not been reported. Here we show that unilateral carotid artery ligation combined with 1.5 to 2 hours of hypoxia (8% oxygen), in both 7- and 21-day-old rats, gives rise to rapid and transient increases in the levels of ubiquitin conjugates and immunoreactivity in the affected right forebrain regions.
METHODS
Induction of hypoxia—ischemia
Pregnant Wistar rats (Charles River, Wilmington, DE, U.S.A.) were housed in individual cages and fed standard laboratory chow ad libitum. Offspring were delivered vaginally and litter size adjusted to 10 pups/litter on the day of delivery. Cerebral H/I was induced as previously described (Rice et al., 1981). Briefly individual 7- and 21-day-old rat pups were anesthetized with halothane (4% induction, 1% maintenance)/30% oxygen/balance nitrous oxide, and the right common carotid artery was exposed through a midline neck incision, separated from contiguous structures, and ligated (3.0 surgical silk). The rat pups were allowed to recover from anesthesia and returned to their dams for 2 to 3 hours. Subsequently they were placed in 500-mL jars partially submerged in a 37°C water bath for 20 minutes to allow for temperature equilibration before being exposed to hypoxia (8% oxygen/balance nitrogen) for either 120 or 90 minutes (7- and 21-day-old animals, respectively), temperature being maintained at 37°C throughout. Pups were subsequently returned to their dam for the appropriate recovery period. The combination of unilateral common carotid artery ligation and hypoxia produces a spectrum of brain damage predominantly in the territory of the middle cerebral artery of the ipsilateral cerebral hemisphere. Damage rarely occurs in the cerebral hemisphere contralateral to the ligation. Hypoxia alone and ligation alone do not result in permanent damage. Therefore, some nonligated animals were exposed to hypoxia for the appropriate intervals as hypoxic controls. Other nonhypoxic control animals were maintained in open jars at 37°C for the appropriate intervals.
Preparation of tissue samples
At specific intervals of recovery, animals were lightly anesthetized with halothane and decapitated. The brains were removed, and a sample was dissected from each hemisphere to include the area of distribution of the middle cerebral artery and snap-frozen on dry ice. Samples were stored at −70°C prior to homogenization in phosphate-buffered saline (PBS). Samples were prepared in the presence of the following protease inhibitors: leupeptin, pepstatin, chymostatin, and antipain (all 1 mg/mL final concentration) and phenylmethylsulfonyl fluoride (0.2 mM final concentration). The protein content of samples was determined using the Lowry et al. assay (1951).
Gel electrophoresis and western blotting
Sodium dodecyl sulfate (SDS) polyacrylamide gel electrophoresis was carried out according to the method of Laemmli (1970). Routinely aliquots containing 20 μg protein were separated on 8% gels. Gels were stained with Coomassie blue for protein visualization. Western blotting was essentially as described by Towbin et al. (1979). Electrophoretic transfer was carried out using a Bio-Rad mini-transblot apparatus at 100 V for 1 hour. Blotting efficiency was checked by staining blotted gels for residual protein with Coomassie blue. Protein was visualized on the blots by staining with Ponceau S prior to immunodevelopment.
Development of western blots
The detection of ubiquitin conjugates was enhanced by autoclaving the blots for 30 minutes prior to blocking, as we have previously described (Chapman et al., 1994). Blots were blocked by incubation in 5% (wt/vol) dried milk in PBS for 30 minutes. Blots were washed in PBS (1 × 15 minutes and 2 × 5 minutes) before incubation (1 hour) in RHUb1 anti-ubiquitin monoclonal antibody (Mab) added as neat hybridoma supernatant. Blots were washed as previously and incubated in secondary antibody, anti-mouse horseradish peroxidase-conjugated IgG (Amersham), diluted 1:2,000 in blocking solution. Blots were washed in PBS (1 × 15 minutes, 4×5 minutes) and immunoreactivity visualized using the enhanced chemiluminescence method (Amersham). Densitometric analysis of blots utilized a UVP gel documentation and analysis system.
Immunocytochemistry
Immersion-fixed tissue was prepared by decapitation of animals and opening of the skull to expose the brain. The heads were immersed in 100 mL of formaldehyde/acetone/methanol (1:1:8 by vol) for 24 hours at room temperature. Subsequently brains were carefully removed from the skulls and transferred to fresh formaldehyde/acetone/methanol prior to paraffin embedding and sectioning. For some studies tissue was fixed by perfusion. Animals were anesthetized with intraperitoneal injections of sodium pentobarbital (60 mg/kg) prior to transcardiac perfusion with normal saline followed by Bouin's fixative. The perfused brains were stored overnight in cold Bouin's fixative.
Material was dehydrated and paraffin embedded. Sections were cut at 15 μm, mounted on gelatin-coated slides, and immunoperoxidase stained on the slide by incubation in 10% normal horse serum in PBS for 30 minutes followed by overnight incubation in primary antibody, RHUb1 hybridoma supernatant diluted (1:20) in blocking solution. After three 5-minute washes in PBS, the sections were incubated for 2 hours in rabbit anti-mouse IgG conjugated to horseradish peroxidase, diluted 1:100 in blocking solution. Sections were washed in PBS (3 × 5 minutes) before incubation in diaminobenzidine to produce a brown reaction product. After further washing, the sections were mounted on glass slides and coverslips were applied with Permount.
Production of anti-ubiquitin monoclonal antibody
The anti-ubiquitin Mab, RHUb1, used in this study is secreted by a hybridoma line produced by fusion of a mouse immunized with SDS-denatured bovine ubiquitin-keyhole limpet hemocyanin conjugate. It has previously been shown that although antibodies raised against ubiquitin using this protocol recognize both free and conjugated ubiquitin, they exhibit stronger immunoreactivity for the latter (Hershko et al., 1982). Several observations confirm that the RHUb1 Mab is specific for ubiquitin (Chapman et al., 1994; Flann et al., 1997). First, the antibody exhibits strong immunoreactivity on enzyme-linked immunosorbent assay and Western blot assays against commercial preparations of bovine and yeast ubiquitin, both as free ubiquitin and when covalently cross-linked to keyhole limpet hemocyanin or ovalbumin. The antibody shows no cross-reactivity with carrier protein alone. Second, and more important, the immunoreactivity of all bands detected on Western blots of cultured cerebellar granule cell homogenates is very markedly reduced if the cells are incubated in the presence of 1 μg/mL oligomycin and 10 mmol/L 2-deoxyglucose for 30 minutes prior to harvesting and sample preparation, although no change in protein profile is detected on silver-stained gels (Chapman et al., 1994). This marked loss of immunoreactivity demonstrates that the RHUb1 Mab is highly specific for conjugated ubiquitin. The conjugates are depleted by rapid hydrolysis without further conjugation following the decrease in ATP level resulting from the 2-deoxyglucose/oligomycin treatment. A similar decrease in conjugate level is observed in experiments using synaptosomes. Third, essentially the same pattern of immunoreactivity is observed on Western blots developed with RHUb1 and several affinity-purified anti-ubiquitin polyclonal antisera raised in this laboratory (C. C. Rider and P. W. Beesley, unpublished observations).
RESULTS
Western blot analysis of effects of hypoxia—ischemia on ubiquitin conjugates
Western blots of right and left forebrain samples (see Methods) prepared from 7-day-old animals subjected to H/I, animals subjected to hypoxia alone, and control animals were immunodeveloped with the RHUb1 anti-ubiquitin Mab. Samples from H/I animals were prepared immediately following the 2-hour hypoxia and at recovery times of 1, 2, 6, and 24 hours. Data from representative samples at each time point are shown in Fig. 1, and data for several individual animals at 0, 2, and 6 hours following hypoxia are shown in Fig. 2. As we have previously reported (Chapman et al., 1994; Flann et al., 1997), control brain (Fig. 1A, lanes 1 and 10) exhibits a complex pattern of immunoreactive bands ranging in molecular mass from 20 to >200 kDa. The previously demonstrated specificity of the RHUb1 antibody for ubiquitin (Chapman et al., 1994) confirms that these bands are ubiquitin-conjugated polypeptides. In addition, a low molecular mass band corresponding to free ubiquitin is detected close to the dye front. Data for RFB H/I compared with control samples (Fig. 1A, lanes 3 and 1, respectively) show an elevation in conjugate levels immediately following the 2-hour hypoxia. The conjugate level rises sharply by 1-hour recovery (Fig. 1A, lane 4) and remains similarly elevated at 2 and 6 hours of recovery (Fig. 1A, lanes 5 and 6, respectively). However, by 24 hours of recovery (Fig. 1 A, lane 7), the conjugate levels are not distinguishable from those observed in control animals killed at the same time (Fig. 1A, lane 10). Densitometric analysis of the data for individual animals (Fig. 2) shows that the conjugate level is increased by 55.5 ± 2.3 (SEM) and 57.9 ± 11.0%, respectively, at 2 and 6 hours of recovery in right compared with left forebrain. These data are significant at the P < 0.01 level (n = 3 for each group). This increase in conjugate level is specific to the right forebrain and is not detected in left forebrain samples for the same H/I animals (Fig. 1B, cf. lanes 3 to 7 with lanes 1 and 10). Although the data suggest that an increase in conjugate level in the right forebrain of H/I animals is accompanied by a decrease in the level of free ubiquitin (Fig. 1A, lanes 3 to 7), the blotting conditions have been optimized for the transfer of conjugates rather than free ubiquitin, thus excluding it from the quantitative interpretation of these data.
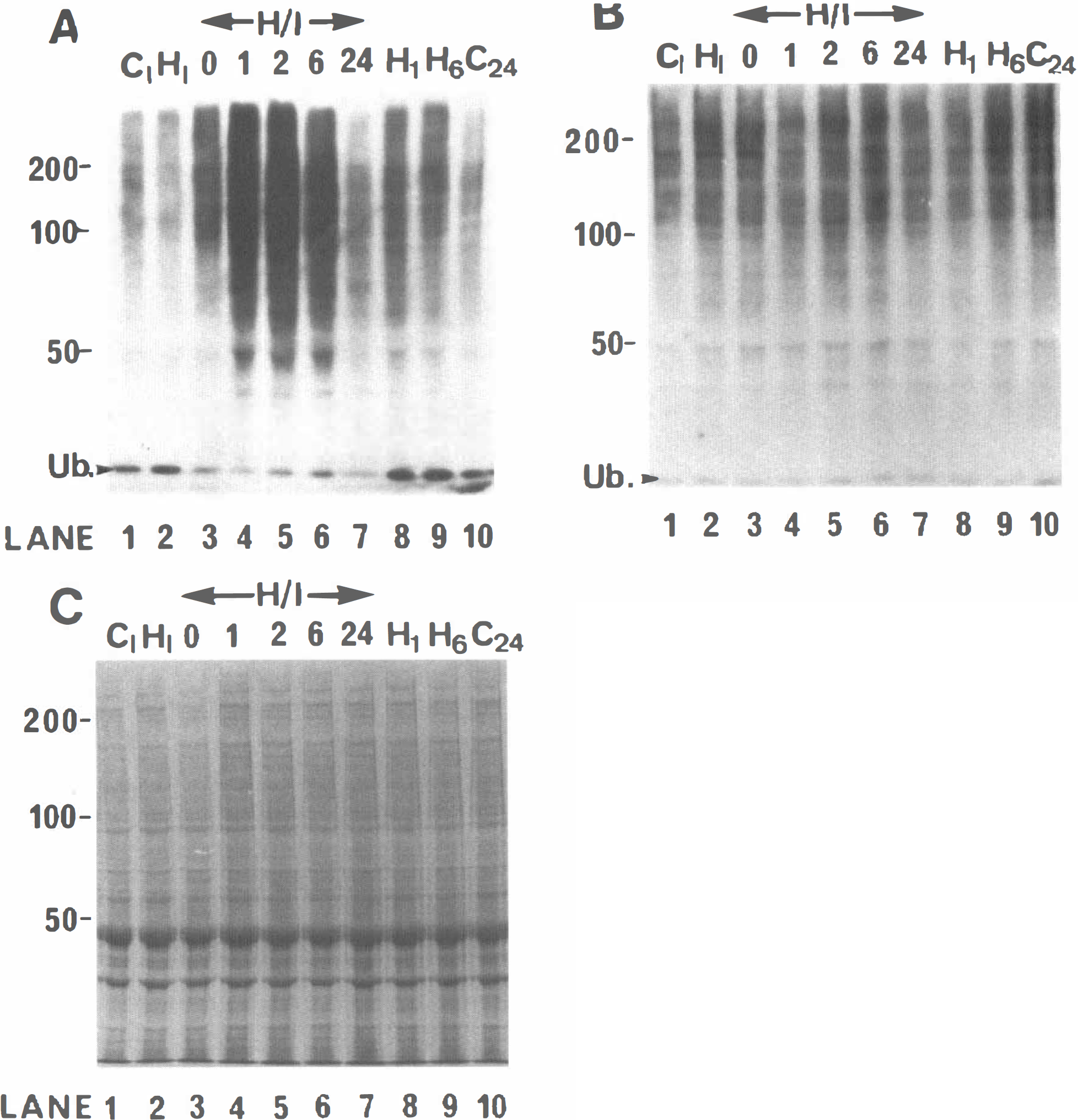
Time course of changes in ubiquitin conjugates in the right (ipsilateral) and left (contralateral) cerebral hemisphere of 7-day-old rats treated with hypoxia—ischemia (H/I). (
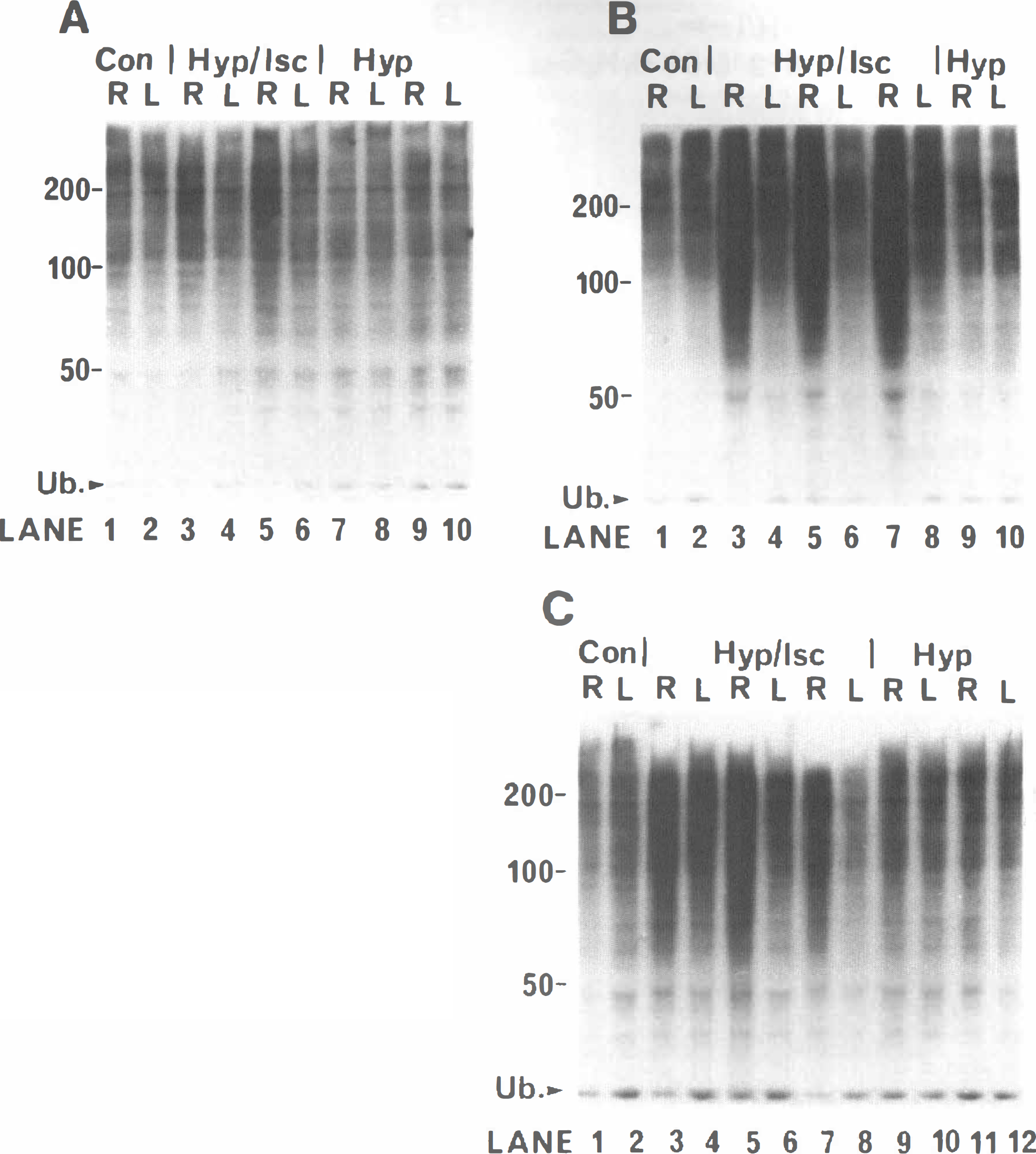
The hypoxia—ischemia-induced rise in ubiquitin conjugate levels in right cerebral hemisphere is reproducible between animals. Homogenates prepared from corresponding right (ipsilateral) and left (contralateral) cerebral hemispheres were separated on 8% sodium dodecyl sulfate polyacrylamide gels and western blotted. The blots were immunodeveloped with RHUb1 monoclonal antibody. (
Exposure of animals to hypoxia alone does not result in any reproducible increase in conjugate level at any of the recovery times tested. Some samples do show an apparent slight increase in conjugate levels compared with controls. Thus, conjugate levels in right forebrain samples prepared from animals subjected to hypoxia alone and allowed to recover for 1 and 6 hours (Fig. 1A, lanes 8 and 9, respectively) are apparently elevated with respect to control samples (Fig. 1A, lanes 1 and 10). However, no elevation of conjugate levels is observed in the corresponding left forebrain hypoxia alone samples compared with control animals (Fig. 1B, cf. lanes 8 and 9 with lanes 1 and 10). Since hypoxia alone will affect both right and left forebrain equally, we conclude that these differences lie within the range of normal variation. Furthermore, ligation of the right carotid artery alone does not alter conjugate levels in right or left forebrain (results not shown).
Analysis of the protein composition of the right forebrain samples by SDS polyacrylamide gel electrophoresis shows no change in the pattern and relative level of protein bands in H/I animals at any of the recovery times tested (Fig. 1C, lanes 3 to 7). Nor do any of these samples differ from control (Fig. 1C, lanes 1 and 10) or hypoxia alone samples (Fig. 1C, lanes 2, 8, and 9). Thus, the increase in ubiquitin conjugate level in the right forebrain of H/I animals does not merely reflect changes in protein composition.
The large transient increase in conjugate level in the right compared with the corresponding left forebrain samples of individual H/I animals is entirely reproducible (Fig. 2). Conjugate levels are already increased in right forebrain samples prepared immediately following exposure of H/I animals to hypoxia (Fig. 2A, cf. lanes 3 and 5 with lanes 4 and 6). As expected, a much greater increase in conjugate level is observed in samples from the right forebrain of H/I animals following 2-hour recovery (Fig. 2B, cf. lanes 3,5, and 7 with lanes 4, 6, and 8). An almost identical increase is observed at 6 hours (Fig. 2C, cf. lanes 3, 5, and 7 with lanes 4, 6, and 8). The data also suggest a small elevation of conjugate levels in the left forebrain samples for H/I (Fig. 2C, lanes 4, 6, and 8) compared with control (Fig. 2C, lane 2) and hypoxia alone (Fig. 2C, lanes 10 and 12) animals. The same reproducibility of data is seen in samples from H/I animals allowed to recover for 1 and 24 hours following exposure to hypoxia (data not shown).
Changes in ubiquitin conjugate level in 21-day-old H/I rat brain have also been analyzed. By this age the rat brain is more comparable with the adult brain and more sensitive to H/I. The conjugate levels of representative samples prepared from H/I animals immediately following exposure to hypoxia and at 1, 2, 6, 24, and 72 hours of recovery are shown in Fig. 3. As for the 7-day-old pups, a striking increase in conjugate levels is observed in the right (Fig. 3A, lanes 3 to 8) but not the left (Fig. 3B, lanes 3 to 8) forebrain of the 21-day-old H/I animals. However, in contrast to the 7-day-old animals, no significant elevation of conjugate levels in the right forebrain is observed immediately following exposure of the H/I animals to hypoxia (Fig. 3A, lane 3). Conjugate levels are markedly elevated in right forebrain samples from H/I animals following 1, 2, 6, and 24 hours of recovery (Fig. 3A, lanes 4, 5, and 6, respectively). Densitometric analysis shows a 51.2 ± 2.8 and 95.0 ± 6.6% increase in conjugate levels in right compared with left forebrain at 2 and 24 hours of recovery, respectively (data significant at P < 0.01 level, n = 3 for each group). This latter observation is in contrast to the 7-day-old animals where conjugate levels return to control values by 24 hours of recovery. A decline in conjugate level in the right forebrain of the 21-day-old animals is detected only at the 72-hour recovery time point (Fig. 3A, lanes 7 and 8, respectively). Thus, a slower onset, but longer persistence, in elevation of conjugate levels is observed for the 21-day-old compared with the 7-day-old animals. As for the 7-day-old animals, no elevation in conjugate levels is observed in either right or left forebrain following exposure of animals to hypoxia alone in samples prepared immediately (Fig. 3A and B, lane 2) or after recoveries of 2 hours (Fig. 3A and B, lane 9) or 6 hours (Fig. 3A and B, lane 10).
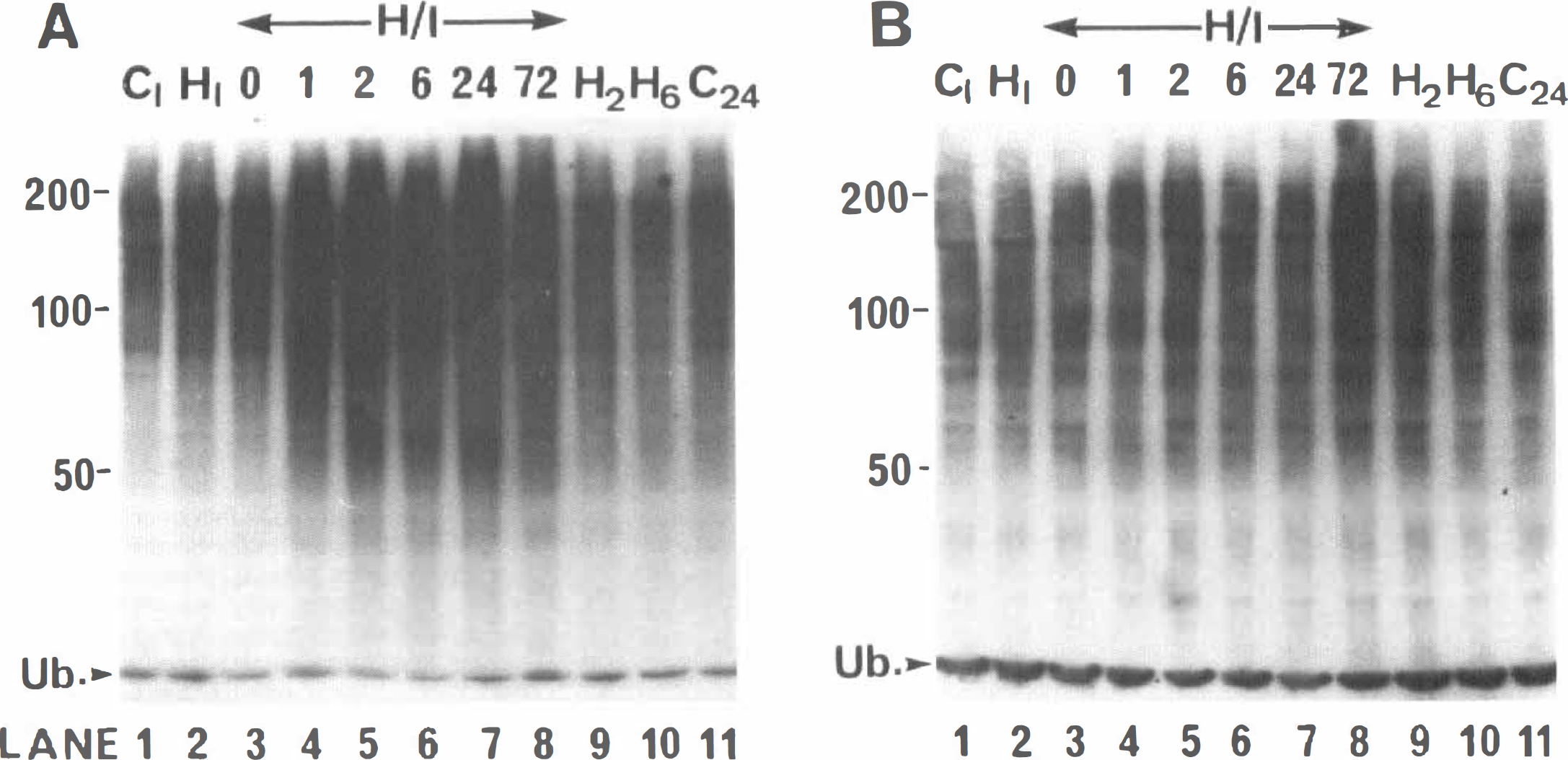
Time course of changes in ubiquitin conjugates in the right (ipsilateral) and left (contralateral) forebrain following hypoxia—ischemia (H/I) in 21-day-old rats. Western blots of a series of right (ipsilateral) and left (contralateral) cerebral hemisphere samples prepared from animals subjected to H/I or hypoxia alone (H) and control animals (C) that were allowed to recover for the stated times are shown in (
Immunocytochemical analysis of effects of hypoxia—ischemia on ubiquitin immunoreactivity
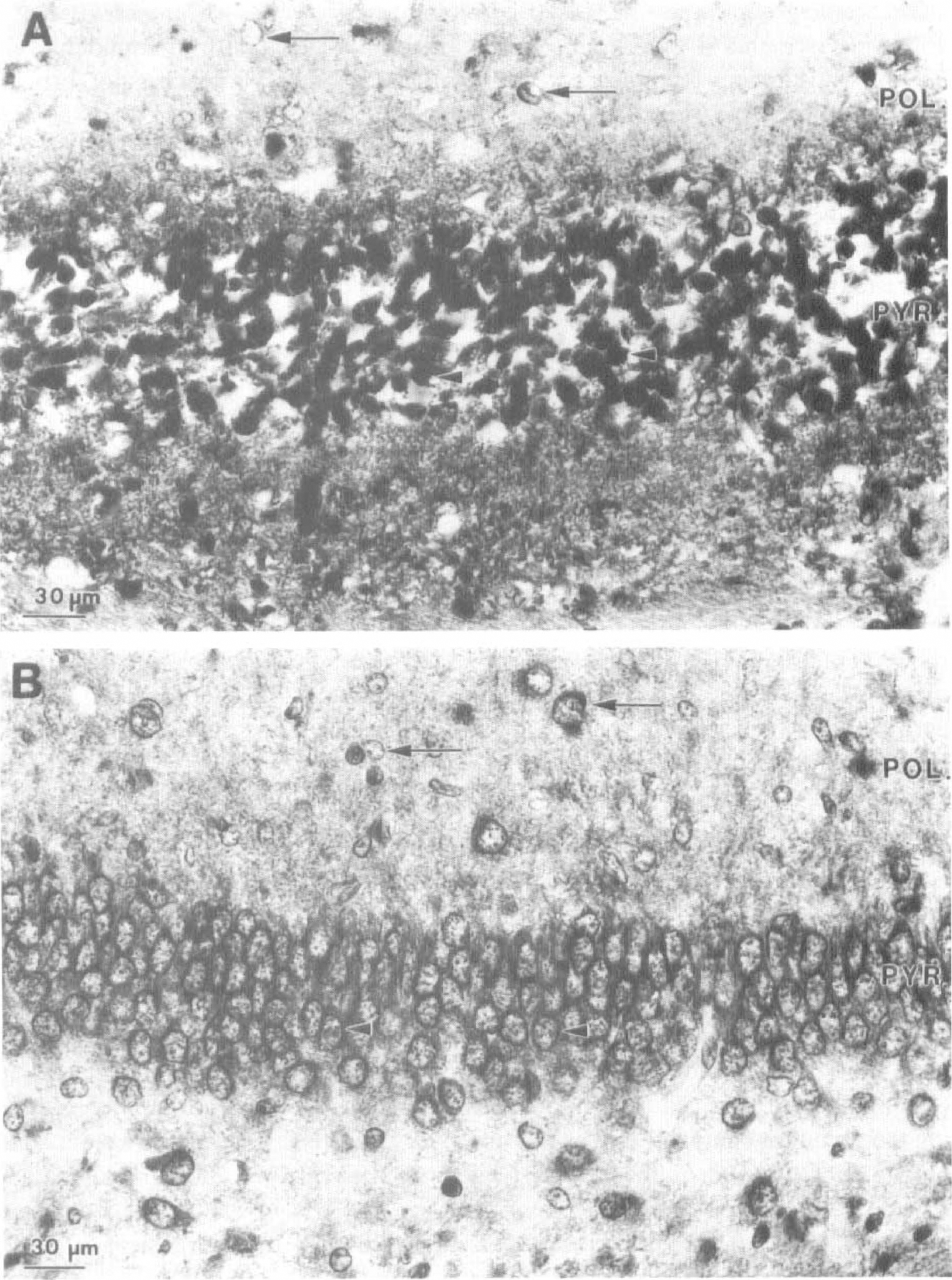
Immunocytochemical studies of 7-day-old brain were carried out using immersion-fixed tissue prepared from H/I animals following 2 hours of recovery from exposure to hypoxia. Use of immersion fixation avoids perfusion artifacts that arise from edematous swellings in the infarct area (unpublished observations). The distribution of ubiquitin immunoreactivity for the left piriform cortex (Fig. 4B) and isocortex (Fig. 4D) of control animals closely resembles that that we have described for perfusion-fixed tissue from normal developing brain (Flann et al., 1997) using both Mab RHUb1 and several polyclonal antisera specific for ubiquitin that were raised in our laboratory. Prominent staining of pyramidal cell perikarya and nuclei is observed (Fig. 4B and D, arrowheads) together with fainter staining of dendrites (Fig. 4D, arrows). The intranuclear reaction product is concentrated in a perinuclear ring, with granular deposits dispersed throughout the nucleoplasm.
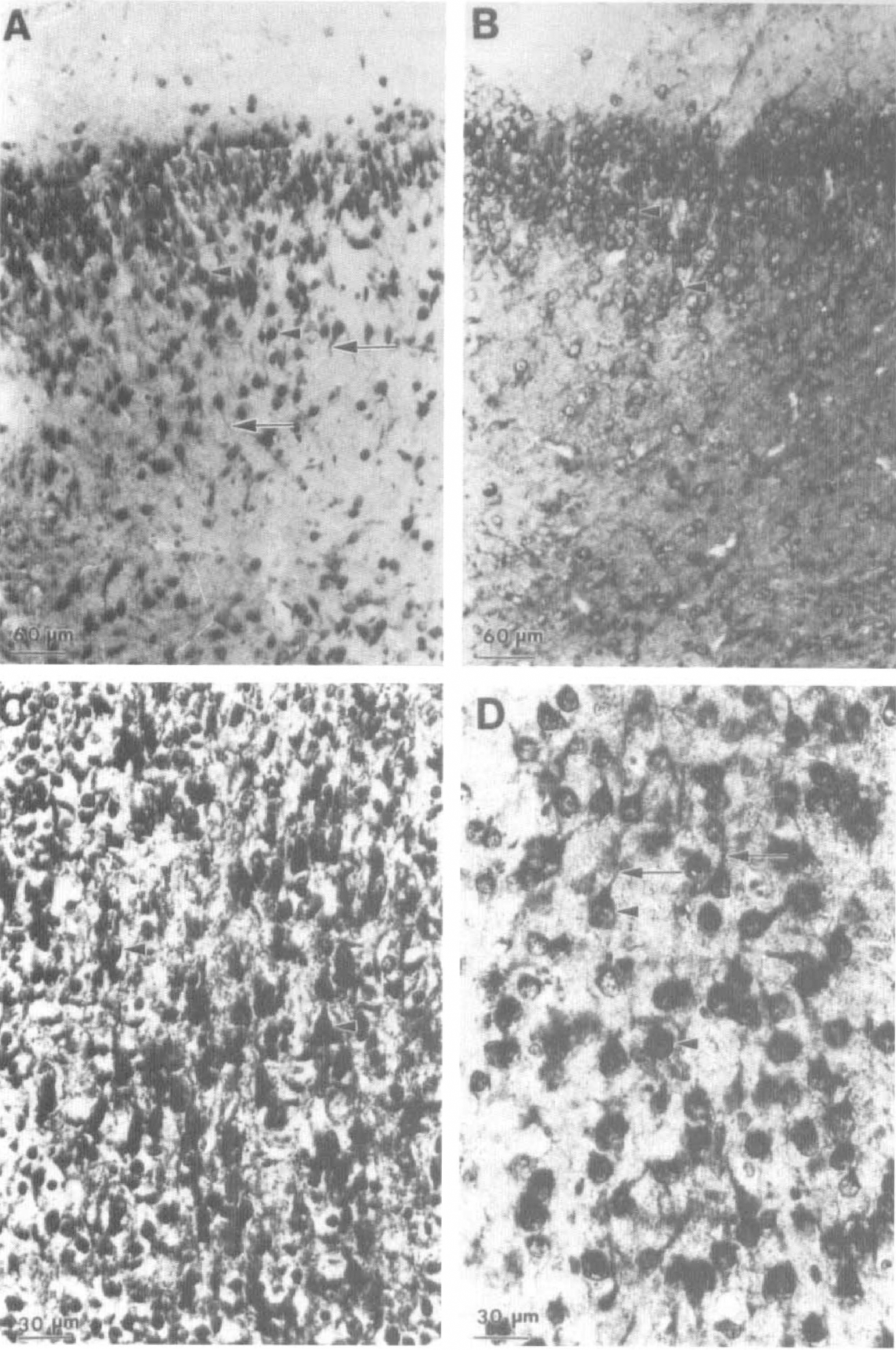
Light microscope immunocytochemical analysis of ubiquitin immunoreactivity in right and left cortex of 7-day-old rats following hypoxia—ischemia (H/I). Sections of right (ipsilateral;
A marked elevation in ubiquitin-like immunoreactivity is observed in the right piriform and isocortex of the H/I animals (Fig. 4A and C), consistent with the elevation of conjugate levels detected by western blotting. Again, immunoreactivity is predominantly associated with the pyramidal cell perikarya and dendrites (Fig. 4A and C, arrowheads and arrows, respectively). In addition, many pyramidal cell perikarya and nuclei (Fig. 4C, arrowheads) are stained in their entirety in contrast to the perinuclear staining observed for pyramidal cells in the left cortex. Furthermore, there is shrinkage of pyramidal cell perikarya in right compared with left isocortex, consistent with tissue damage and the onset of neuronal degeneration (Fig. 4C and D, respectively). Similar results are obtained for hippocampus (Fig. 5). Prominent staining of neuronal perikarya and nuclei is observed in the left hippocampus (Fig. 5B). This is particularly marked for the large neurons of the pyramidal cell layer (Fig. 5B, arrowheads) but is also observed in cells scattered throughout the polymorphic layer (Fig. 5B, arrows). As for the cortex, a dramatic increase in ubiquitin immunoreactivity is observed in the right hippocampus. The majority of pyramidal cell perikarya are stained in their entirety compared with the perinuclear staining observed in the left hippocampus (Fig. 5A and B, respectively). Fragmentation of the ordered pyramidal cell layer is clearly observed in the right hippocampus, together with a shrinkage of cell somata. Similar left compared with right side differences are observed in other brain regions, including the thalamus and caudate/putamen (results not shown).
DISCUSSION
The present Western blot and immunocytochemical data represent the first demonstration that exposure of perinatal rats to H/I causes a marked rapid and transient increase in the level of ubiquitin conjugates and ubiquitin immunoreactivity. These changes are specifically associated with the H/I insult and are restricted to the vulnerable brain regions, i.e., predominantly within the territory of the middle cerebral artery of the ipsilateral cerebral hemisphere. In 7-day-old animals, the increase in conjugate levels is detected immediately following H/I and has reached maximal elevation within 1 to 2 hours of recovery. Conjugate levels are still maximally elevated at 6 hours but are not distinguishable from control levels by 24 hours of recovery. Similar changes are observed in 21-day-old animals, but the increase in ubiquitin conjugate levels occurs more slowly and persists longer. Indeed, in the 21-day-old animals, conjugate levels are still markedly elevated after 24 but not 72 hours of recovery. There are several possible explanations for this difference. It may be associated with (a) a developmental decrease in the ubiquitin system in brain that occurs predominantly between postnatal days 14 and 21 (Flann et al., 1997); (b) the more delayed and selective neuronal necrosis observed in the more mature brain (Towfighi et al., 1997); or (c) the increased vulnerability of the more mature brain to H/I, even of shorter duration. A 21-day-old rat brain is energetically more comparable with an adult's and has a far greater energy requirement than a 7-day-old rat brain, which could lead to its increased sensitivity to substrate/oxygen limitation (Nehlig et al., 1988).
Light microscope immunocytochemical analysis of ubiquitin immunoreactivity in right and left hippocampus of 7-day-old rats following hypoxia—ischemia (H/I). Sections of right (ipsilateral;
The immunocytochemical studies of immersion-fixed tissue show that the increase in ubiquitin immunoreactivity is predominantly neuronal and is particularly associated with pyramidal cell perikarya and to a lesser extent dendrites. The increase in ubiquitin immunoreactivity is uniform throughout all sections examined and as such is similar to that seen for the immediate early gene, c-fos (Blumenfeld et al., 1992; Aden et al., 1994; Munell et al., 1994).
Changes in ubiquitin conjugate level, ubiquitin immunoreactivity, and ubiquitin gene expression in adult brain following ischemia and transient global H/I have been reported. Hayashi et al. (1991, 1992a) report an increase in ubiquitin conjugate level in the hippocampus following reperfusion in animals subject to transient global ischemia using the four-vessel occlusion model. This increase is rapid (Hayashi et al., 1992b) and occurs within 5 to 15 minutes of reperfusion following 15-minute ischemia (P. W. Beesley, C. C. Rider, J. Gurd, C. Wallace, unpublished observations). Several studies have used immunocytochemical methods to establish the effects of hypoxia and H/I on ubiquitin immunoreactivity in adult brain. Some groups report increases in immunoreactivity following ischemia (Dewar et al., 1993; Gubellini et al., 1997), while others report a decrease (Magnusson and Wieloch, 1989; Hayashi et al., 1991a). These differences may be attributable to the various model systems used but may also be associated with the use of different antibodies recognizing different epitopes on the ubiquitin molecule. Thus, the accessibility of some ubiquitin epitopes may be masked on some conjugate species or in some subcellular compartments in tissue sections. Indeed, Hayashi et al. (1991) attribute the discrepancy between their immunocytochemical and Western blot data to the presence of hidden epitopes on high molecular mass conjugates, making their detection in tissue sections difficult. Although western blotting is the sole available method for quantitating changes in ubiquitin conjugate levels, only the present study and that of Hayashi et al. (1991a) have used this approach to date. A striking feature of the present data is the rapid increase in the level of ubiquitin conjugates in response to H/I. Other events occurring in the ipsilateral hemisphere during the early recovery period include changes in energy metabolism, particularly a partial restitution, prior to secondary depletion of ATP and phosphocreatine levels and an uncoupling of oxidative phosphorylation (Palmer et al., 1990; Yager et al., 1992; Vannucci et al., 1994), accumulation of cytosolic calcium, and expression of several immediate early genes (Blumenfeld et al., 1992; Aden et al., 1994; Munell et al., 1994). Thus, one plausible mechanism for the increased protein ubiquitination is up-regulation of ubiquitin gene expression. In this context, one component of the ubiquitin system, ubiquitin C-terminal hydrolase, an enzyme responsible for cleaving the ubiquitin conjugate isopeptide bond, has recently been identified as an immediate early gene in Aplysia (Hedge et al., 1997).
Up-regulation of ubiquitin gene expression following transient global ischemia in both gerbil and rat has been reported (Caday et al., 1993; Noga and Hayashi, 1996). In the latter study, increased expression of the UbC ubiquitin gene is observed in the hippocampus and cortex of the adult rat following transient global ischemia. The UbC gene encodes a polyubiquitin sequence and contains a heat shock element in the 5′ upstream region. However, the increase in UbC expression is observed only after 4 and 6 hours of reperfusion, much later than the observed rise in conjugate level that is detected by 15 minutes of reperfusion. Smaller changes in the expression of the UbB and none in the expression of the UbS30 genes were detected. Surprisingly, a decrease in expression of all the classes of ubiquitin gene was observed during the early phase of reperfusion. The more delayed time course of ubiquitin gene expression is more comparable with that observed for hsp70 gene expression following cerebral H/I in 7-, 15-, and 23-day-old rats (Blumenfeld et al., 1992). Indeed, our own data obtained from immunodevelopment of Western blots with an Mab specific for hsp70 show that the hsp70 protein is not detected in the right forebrain of H/I animals until 2 hours of recovery, and its level increases very markedly between 2 and 6 hours of recovery (data not shown). In this regard, it is the rapidity of the increase in conjugate level in H/I animals in the present study that suggests that this effect is not due to increased ubiquitin gene expression, at least not at early recovery times. These observations raise the possibility of novel transcription-independent mechanisms for regulation of protein ubiquitination.
The functional consequences of the elevated protein ubiquitination in H/I animals are not yet clear. However, the rapid onset of the response suggests that it has a neuroprotective role, although the possibility that it is part of a pathway leading to cellular damage or death cannot be excluded. Indeed, the present immunocytochemical data confirm signs of cell damage and disintegration by 2 hours of recovery. In the immature brain, histological evidence of tissue damage can be observed very early into recovery, although it becomes much more pronounced at 24 to 72 hours of recovery (Towfighi et al., 1995). Studies on the effects of free radical-producing agents on neuron-like cells provide further evidence that elevated protein ubiquitination is a protective response rather than occurring merely as a consequence of tissue damage. Thus, exposure of nerve growth factor-differentiated PC12 cells to sublethal, but not lethal, concentrations of hydrogen peroxide causes a marked increase in ubiquitin conjugate levels (S. J. Flann et al., unpublished observations).
The present study indicates that increased protein ubiquitination is an early change resulting from H/I and is plausibly a protective response. Although the increase is transient, it persists for several hours and thus may be amenable to biochemical manipulation. The functions of the increased ubiquitination will have to await the development of suitable assays to establish the consequences of ubiquitination of individually identified proteins. However, elevation of ubiquitin levels in yeast is known to be protective and at least in part associated with increased protein degradation (Finley et al., 1987).
Footnotes
Abbreviations used
Acknowledgments
We thank Estrella Gonzales and Lisa Seaman for their technical assistance.
