Abstract
A reproducible model of thread occlusion of the middle cerebral artery (MCA) was established in C57 Black/6J mice by matching the diameter of the thread to the weight of the animals. For this purpose, threads of different diameter (80 to 260 μm) were inserted into the MCA of animals of different weights (18 to 33 g), and the success of vascular occlusion was evaluated by imaging the ischemic territory on serial brain sections with carbon black. Successful occlusion of the MCA resulted in a linear relationship between body weight and thread diameter (r = 0.46, P < 0.01), allowing precise selection of the appropriate thread size. Laser-Doppler measurements of CBF, neurological scoring, and 2,3,5-triphenyltetrazolium chloride staining confirmed that matching of animal weight and suture diameter produced consistent cerebral infarction. Three hours after MCA occlusion, imaging of ATP, tissue pH, and cerebral protein synthesis allowed differentiation between the central infarct core, in which ATP was depleted, and a peripheral penumbra with reduced protein synthesis and tissue acidosis but preserved ATP content. Perfusion deficits and ischemic tissue alterations could also be detected by perfusion- and diffusion-weighted magnetic resonance imaging, demonstrating the feasibility of dynamic evaluations of infarct evolution. The use of multiparametric imaging techniques in this improved MCA occlusion model opens the way for advanced pathophysiological studies of stroke in gene-manipulated animals.
During the last decade, the genomic response associated with the pathophysiology of cerebral ischemia has been intensively studied. Ischemia induces numerous genes such as the immediate-early genes c-fos, c-jun, and junB (An et al., 1993; Kiessling and Gass, 1994; Kinouchi et al., 1994; Neumann et al., 1994) and genes encoding the heat shock protein hsp72 (Kinouchi et al., 1993; Nowak and Jacewicz, 1994; Massa et al., 1996) and the cytokines interleukin-1, interleukin-6, and tumor necrosis factor-α (Wiessner et al., 1993; Buttini et al., 1996). The main interest in investigating ischemia-induced genomic responses in the brain is to clarify the mechanisms of inherent degenerative or protective processes and to define targets for specific modulations of these cellular responses. The development of knockout/transgenic technology in mice allows easy manipulations of gene expressions and their particular translational products in this species (Aguzzi et al., 1996; Chan, 1996; Ryffel, 1996). It is now possible to analyze the effect of targeted disruption or overexpression of genes, and there is an increasing number of reports about the effect of focal cerebral ischemia in such mutants (Chan et al., 1993; Crumrine et al., 1994; Huang et al., 1994; Kamii et al., 1994; Yang et al., 1994; Bruce et al., 1996; Connolly et al., 1996a; Hara et al., 1996).
Although murine models of middle cerebral artery (MCA) occlusion have been described, few of these have been well characterized (Connolly et al., 1996b). Moreover, little is known about the ischemic “penumbra” (Astrup et al., 1981), although this part of the evolving infarct is the main target of all molecular interventions. In the present study, we first established a reproducible focal ischemia model in mice and then imaged the ischemic penumbra by recording the regional alterations of metabolic parameters such as ATP, pH, and cerebral protein synthesis (CPS). In addition, we explored the feasibility of using magnetic resonance (MR) imaging to monitor the evolution of hemodynamics and cellular injury in this model. Our observations demonstrate that the here-described mouse focal ischemia model produces consistent infarction and is suited for advanced pathophysiological studies including functional MR imaging.
MATERIALS AND METHODS
Experiments were carried out in adult male C57 Black/6J mice weighing 18.0 to 33.0 g (Harlan Winkelmann, Borchen, Germany). Animals were housed under controlled diurnal lighting conditions and allowed access to food and water ad libitum until the day of the experiment. Anesthesia was induced by 1.5% halothane and maintained with 1% halothane in 70% N2O and 30% O2.
Animal preparation
Focal cerebral ischemia was induced by occlusion of the MCA using the intraluminal filament technique (Hara et al., 1996; Huang et al., 1996). After midline neck incision, the left common and external carotid arteries were isolated and ligated. A microvascular clip (FE691; Aesculap, Tuttlingen, Germany) was temporarily placed on the internal carotid artery. An 8-0 nylon monofilament (Ethilon; Ethicon, Norderstedt, Germany) coated with silicon resin (Xantopren; Bayer Dental, Osaka, Japan) was introduced through a small incision into the common carotid artery and was advanced to a position 9 mm distal from the carotid bifurcation for occlusion of the MCA.
Thread diameter and carbon black staining
To ensure successful induction of focal cerebral ischemia, the relationship between animal body weight and the required thread diameter was determined. Fifty-two animals with body weight between 18 and 33 g were selected. The distal 4 mm of the sutures was coated with silicon resin to various diameters (from 0.08 to 0.26 mm). Fifteen minutes after MCA occlusion, carbon black (1.0 mg/100 g; Bokusai; Fueki, Tokyo, Japan) was injected transcardially (Hata et al., 1994) at a rate of ~0.2 mL/min. After carbon black was allowed to circulate for a few minutes, animals were decapitated. Brains were then removed for macroscopic examination of perfusion deficits.
Laser-Doppler flowmetry
In separate mice (n = 10), CBF was determined in the territory of the MCA by laser-Doppler flowmetry using a flexible 0.5-mm fiberoptic extension to the master probe (PF2B; Perimed, Stockholm, Sweden). The tip of the probe was fixed to the intact skull over the supplying territory of the proximal part of the MCA (2 mm posterior and 6 mm lateral from the bregma) using a tissue adhesive (Aron Alpha; Toa, Tokyo, Japan). Changes in cortical perfusion after MCA occlusion were expressed as percentage of the baseline value of laser-Doppler flowmetry.
Neurological score and 2,3,5-triphenyltetrazolium chloride staining
In the animals selected for CBF measurement (n = 10), the severity of the neurological impact was evaluated 24 hours after MCA occlusion using a five-point deficit score (0 = normal motor function; 1 = flexion of torso and of contralateral forelimb upon lifting of the animal by the tail; 2 = circling to the contralateral side but normal posture at rest; 3 = leaning to contralateral side at rest; and 4 = no spontaneous motor activity) (Huang et al., 1994).
Then animals were killed with an overdose of pentobarbital and decapitated. The brains were removed and sectioned coronally into five 2-mm slices using a mouse brain matrix (BRM-2000C; Activational System, MI, U.S.A.). Slices were immediately stained with 2% 2,3,5-triphenyltetrazolium chloride (TTC) as described elsewhere (Bederson et al., 1986). The border between infarcted and noninfarcted tissue was outlined with an image analysis system, and the area of infarction was measured by subtracting the area of the nonlesioned ipsilateral hemisphere from that of the contralateral hemisphere (Swanson et al., 1990). The volume of infarction was calculated by integration of the lesion areas at five equidistant levels of forebrain.
Regional measurement of ATP, tissue pH, and protein synthesis
In randomly selected animals (n = 14), the right femoral arteries were cannulated with polyethylene catheters (PE-10; Clay Adams, Parsippany, NJ, U.S.A.) for sampling of arterial blood and monitoring of blood pressure. Seven mice were subjected to 3 hours of ischemia, and seven other mice were sham-operated controls, in which the thread was introduced into the common carotid artery but not advanced to occlude the MCA.
Five animals subjected to 3 hours of ischemia were used for regional evaluation of ATP, tissue pH, and CPS. Forty-five minutes before death,
The volumes of ATP depletion, tissue acidosis, and CPS inhibition were estimated using a modification of the semiautomated method described by Swanson et al. (1990). Briefly, tissue acidosis was defined as the fall of pH to <6.4 and ATP depletion as the decline to <1.0 μmol/g. The threshold for CPS inhibition was set to the lowest CPS value of the opposite, nonischemic hemisphere excluding the fiber tracts. Volumes of ATP depletion, tissue acidosis, and CPS inhibition were calculated as described for TTC staining (see above).
Nuclear magnetic resonance measurements
To explore the feasibility of diffusion- and perfusion-weighted MR imaging in the mouse model of focal ischemia, MR measurements were carried out using a 4.7-T Biospec system (Bruker Medizintechnik, Karlsruhe, Germany) with a 30-cm-bore horizontal magnet. The system was equipped with actively shielded gradient coils (maximal gradient strength: 100 mT/m; gradient rise time: 2,250 μs). Radiofrequency pulses were transmitted using a 12-cm-diameter Helmholtz coil, and MR signals were acquired with a 16-mm-diameter surface coil placed over the skull.
Sagittal multislice MR images (repetition/echo time 800/8 ms) were taken to ensure correct head position. Diffusion weighting was achieved using a Stejskal-Tanner type pulsed gradient spin echo sequence. Diffusion-weighted images were recorded 2.5 hours after MCA occlusion. Seven slices of 0.8-mm thickness, separated by 1 mm, were measured. The field of view was 1.8 cm with a resolution of 128 × 128 pixels (repetition/echo time 2325/35.2 ms). Three b-factors (30 to 1,500 s/mm2) were used for the measurements. From these images, apparent diffusion coefficient maps were reconstructed, as described before (Hoehn-Berlage et al., 1995).
For perfusion-weighted image measurements, a snapshot fast low-angle shot arterial spin-tagging procedure was used (Kerskens et al., 1996). One slice with a field of view of 3 cm (image resolution 128 × 64 pixels) was scanned at the level of the caudate putamen at 3.5 hours after MCA occlusion, using signal-averaging techniques to improve image contrast. Repetition and echo times were 7.4 and 3.9 ms, respectively.
Temperature control
Rectal temperature was maintained between 36.5 and 37.0°C. In the MR study, temperature was stabilized with a feedback-controlled heated water jacket wrapped around the body of the animal and in all other experiments with an infrared lamp (YSI, Yellow Spring, OH, U.S.A.).
Statistics
All values are presented as means ± SD. Differences in physiological parameters were evaluated for statistical significance using the Mann-Whitney U test. Differences in metabolic parameters were compared using one-way (ATP, CPS) or two-way (pH) analysis of variance followed by Bonferroni's multiple comparison test. Changes of mean arterial blood pressure and heart rate during ischemia were evaluated by one-way analysis of variance with repeated measures. A P value of <0.05 was considered to indicate statistical significance.
RESULTS
Selection of thread diameter for vascular occlusion
The relationship between thread size (0.08 to 0.26 mm) and successful MCA occlusion was evaluated in C57 Black/6J mice of different weights (18.0 to 33.0 g) by intravenous carbon black infusion. In 6 of 52 animals, no staining deficits were observed, indicating incomplete vascular occlusion. In 35 animals, staining deficits were present in the territory of the MCA, confirming successful obstruction of the artery. In the remaining 11 animals, the thread was too thick and could not be inserted. When the size of the thread was plotted against the body weight of successfully occluded animals, a linear relationship (r = 0.46, P < 0.01) was detected (Fig. 1). All mice with incomplete MCA occlusion exhibited higher weights, and those in which threads could not be inserted had lower weights than the mice with successful occlusion. Correlation analysis between suture thickness and the body weight of the successfully occluded animal revealed a numerical relationship that could be used to select the appropriate thread size (Fig. 1).
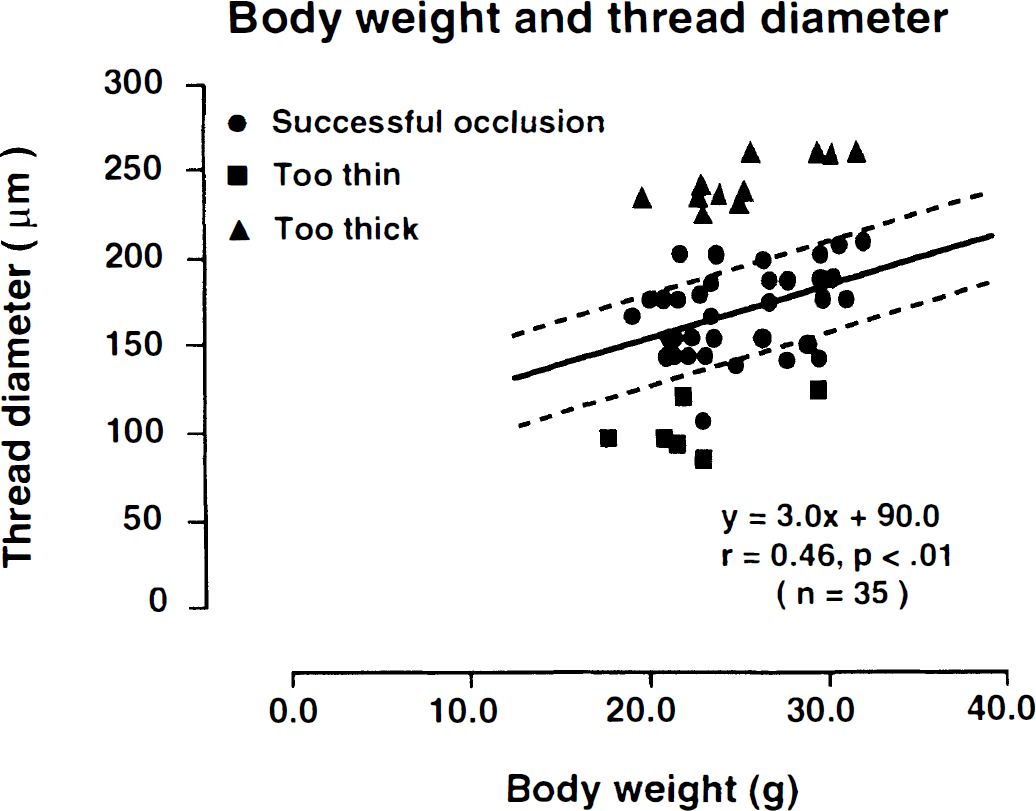
Relationship between body weight (g) and the diameter of thread (μm) required to occlude the middle cerebral artery in mice. Each symbol represents one experiment. Successful occlusion (•) was documented by the absence of carbon black staining of the tissue in the territory of the middle cerebral artery. When thread was too thin (▪), staining deficits were absent. Threads that were too thick (▴) could not be inserted. In successfully occluded animals (•), thread diameter correlated linearly with body weight (r = 0.46, P < 0.01).
Based on these findings, the following procedure was used for all subsequent MCA occlusion experiments. After calculating the individual target thickness, a slightly thicker thread was selected to compensate for the statistical scatter of the regression analysis. In those cases in which the thread could not be introduced, insertions were reattempted with stepwise thinner threads until the target diameter was reached.
Physiologic parameters
The influence of MCA occlusion on physiological parameters was investigated by comparing sham-operated animals (n = 7) with animals subjected to 3-hour MCA occlusion (n = 7; Table 1). There were no significant differences of Pa
Physiologic variables before and at 3 hours after middle cerebral artery occlusion in mice
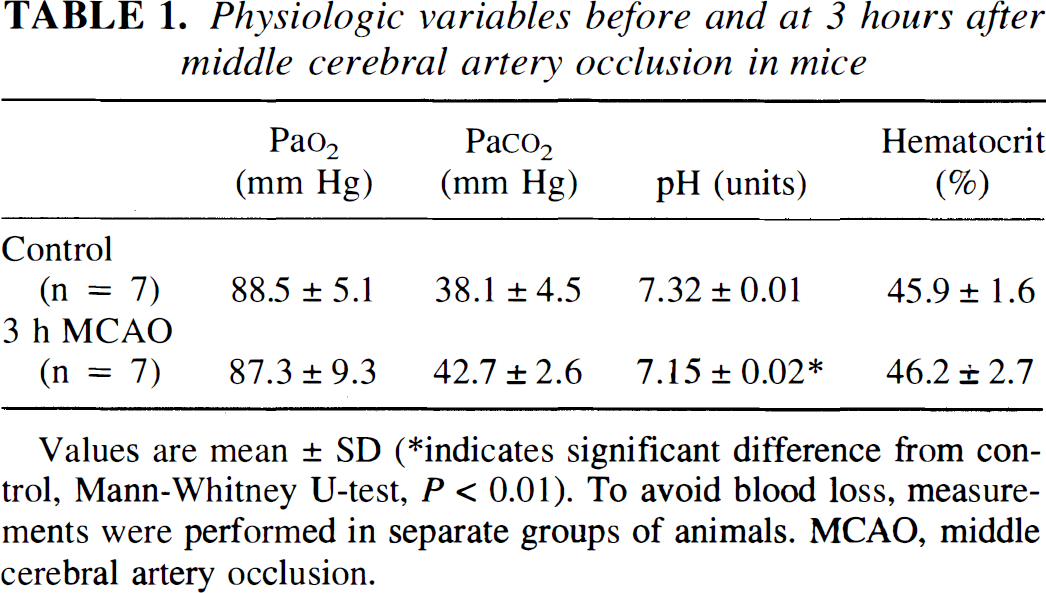
Values are mean ± SD
indicates significant difference from control, Mann-Whitney U-test, P < 0.01). To avoid blood loss, measurements were performed in separate groups of animals. MCAO, middle cerebral artery occlusion.
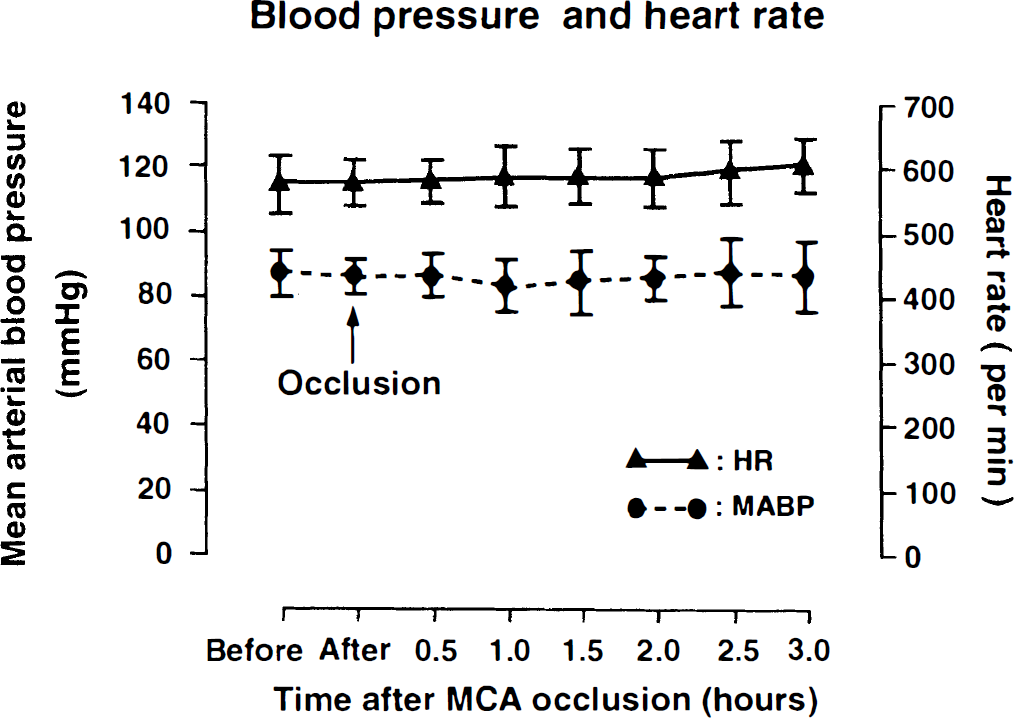
Recording of mean arterial blood pressure (means ± SD) and heart rate (means ± SD) before and after middle cerebral artery occlusion in mice. Note absence of significant cardiovascular changes.
CBF, TTC staining, and neurologic score
After thread insertion, laser-Doppler flow measured in temporoparietal cortex abruptly declined to 21.8 ± 3.7% (mean ± SD, n = 10) of baseline value. Imaging of the ischemic territory by TTC staining showed infarcts in temporoparietal cortex and in the laterocaudal part of the caudate putamen in all animals. The probability of thalamic infarction was 50 to 75% and that of more peripheral parts of the MCA territory was 25 to 50% (Figs. 3 and 4).
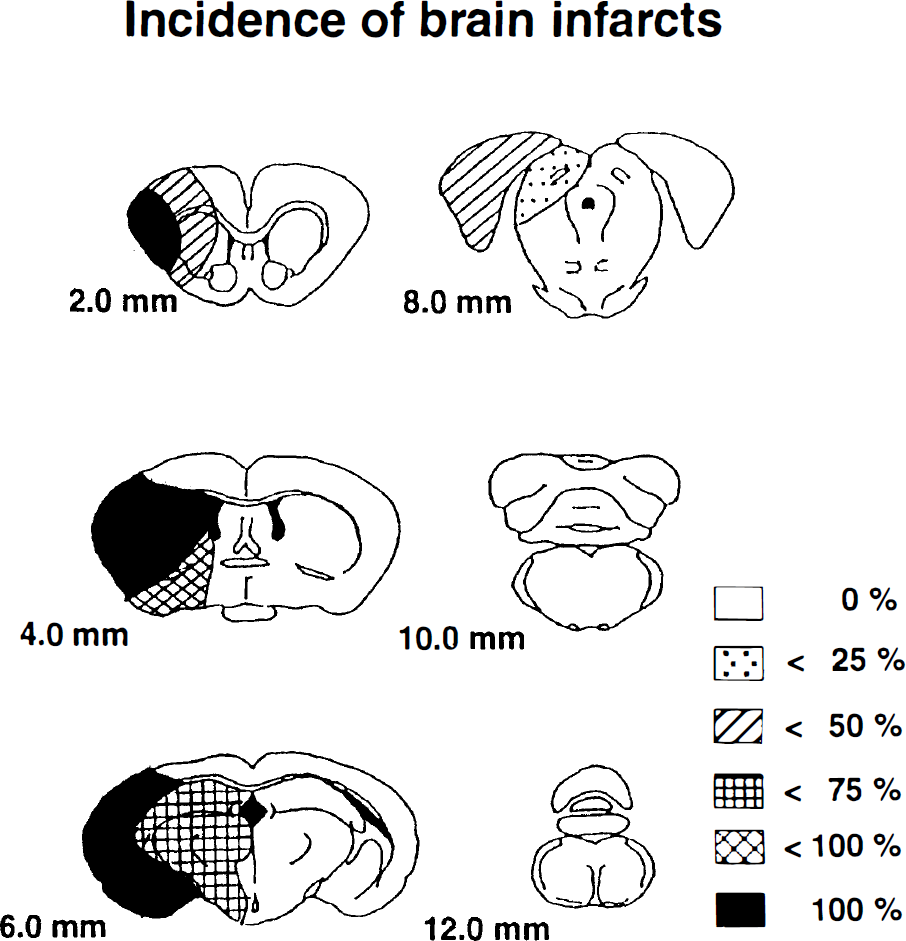
Schematic representation of the distribution of infarcts after thread occlusion of middle cerebral artery (MCA) in mice. Infarcts were visualized by 2,3,5-triphenyltetrazolium chloride staining 1 day after occlusion on coronal brain sections prepared at various distances from the frontal pole. The incidence of local ischemic alterations is expressed as percent of the total number of MCA-occluded animals (n = 10).
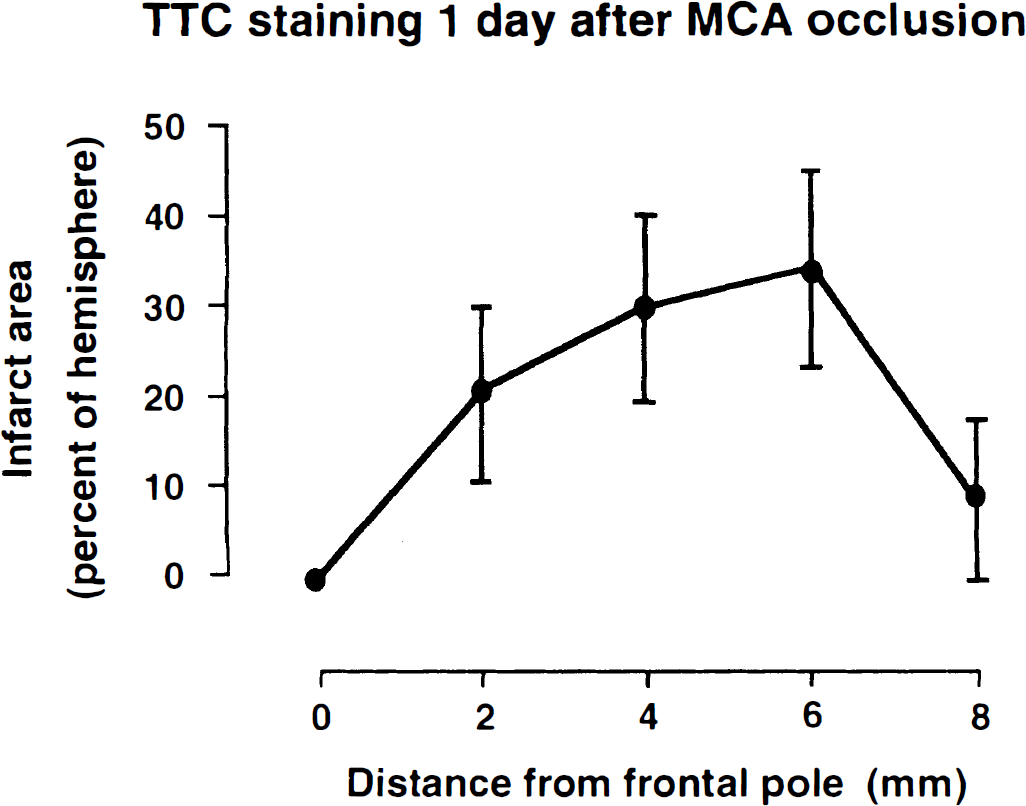
Mean brain infarct size following permanent middle cerebral artery occlusion in mice. Infarcts were visualized by 2,3,5-triphenyltetrazolium chloride staining on four coronal slices prepared at various distances from the frontal pole. Measurements were carried out 1 day after vascular occlusion and expressed as percent (means ± SD) of the ipsilateral hemispheric area (n = 10). Note maximal extension of infarcts on slices at a distance between 4 and 6 mm from frontal pole.
Focal ischemia resulted in marked neurological disturbances 1 day after MCA occlusion. All animals had developed at least grade 2 deficits, the average neurological deficit being 3.1 ± 0.7 (mean ± SD, n = 10).
Regional ATP, tissue pH, and protein synthesis rate
Representative images of the tissue content of ATP, of tissue pH, and of protein synthesis are shown in Fig. 5. ATP was depleted in frontoparietal cortex, in the lateral part of caudate putamen, and in the piriform cortex. Acidosis and suppression of protein synthesis were present in the same areas, but the changes extended distinctly more into the frontal, medial, and basooccipital parts of the MCA territory. Quantitative measurements revealed significant reductions of ATP in the somatosensory area of frontoparietal cortex at the level of striatum, in the temporal and piriform cortex at the level of dorsal hippocampus, and in the lateral caudate putamen. Changes of protein synthesis and tissue pH extended further into the piriform cortex and into the globus pallidus at the level of striatum, into the parietal cortex at the level of dorsal hippocampus, and into the temporal and entorhinal cortex at the level of substantia nigra (Table 2).
Changes of ATP, cerebral protein synthesis and tissue pH of mouse brain after 3 hours of middle cerebral artery occlusion
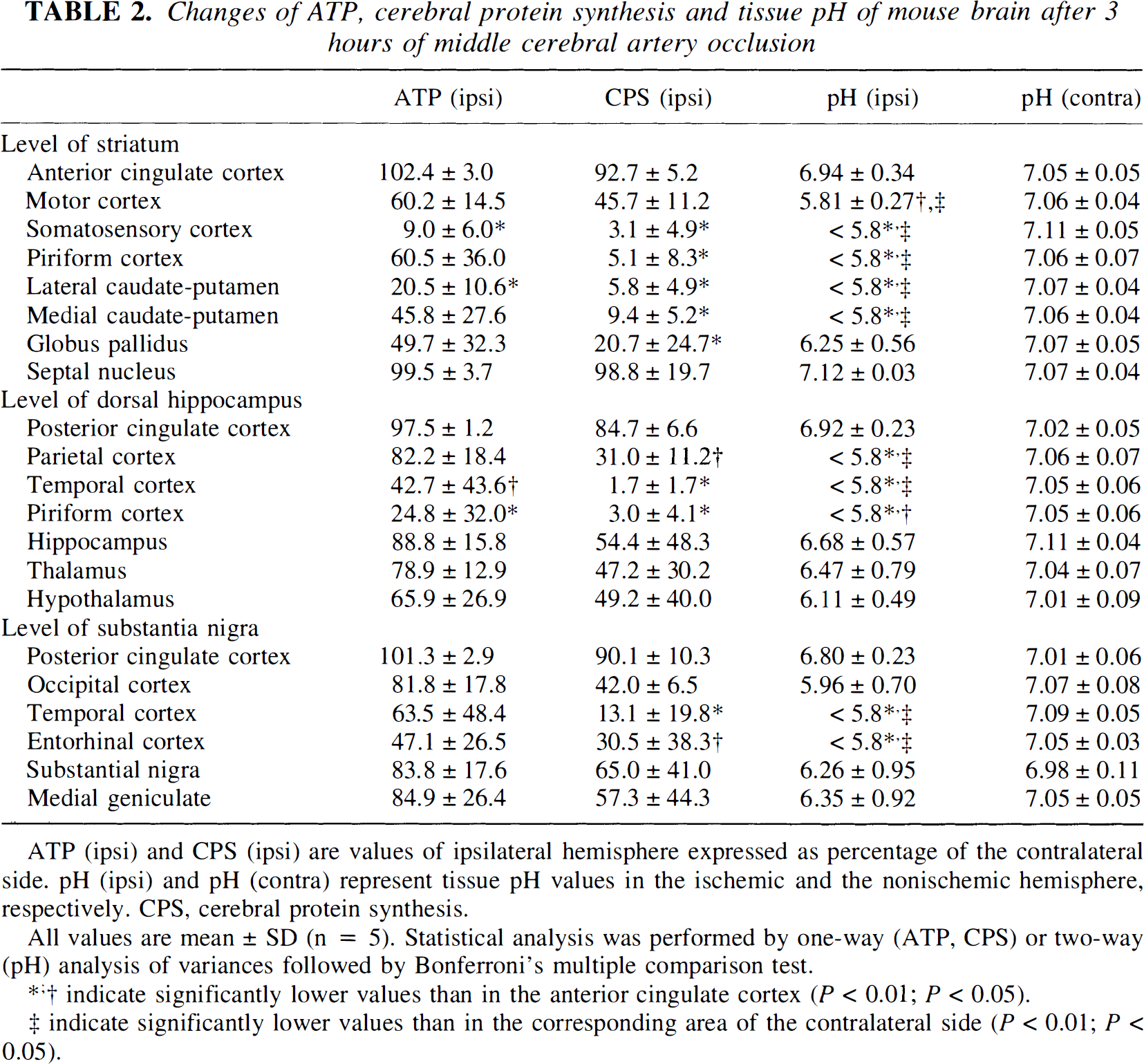
ATP (ipsi) and CPS (ipsi) are values of ipsilateral hemisphere expressed as percentage of the contralateral side. pH (ipsi) and pH (contra) represent tissue pH values in the ischemic and the nonischemic hemisphere, respectively. CPS, cerebral protein synthesis.
All values are mean ± SD (n = 5). Statistical analysis was performed by one-way (ATP, CPS) or two-way (pH) analysis of variances followed by Bonferroni's multiple comparison test.
indicate significantly lower values than in the anterior cingulate cortex (P < 0.01; P < 0.05).
indicate significantly lower values than in the corresponding area of the contralateral side (P < 0.01; P < 0.05).
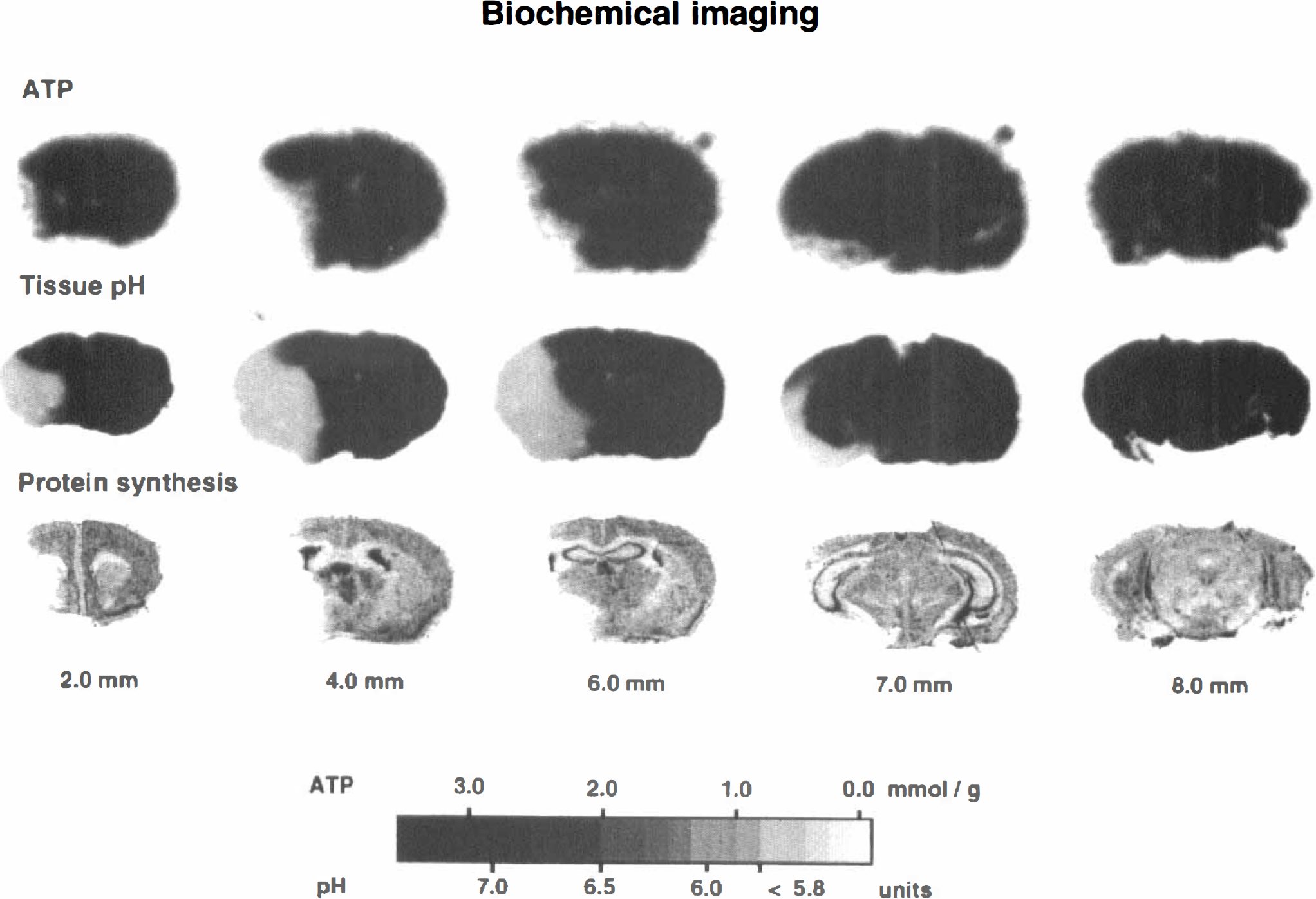
Pictorial evaluation of ATP concentration, of tissue pH, and of cerebral protein synthesis in coronal brain sections of mouse prepared at 3 hours after middle cerebral artery occlusion (for technical details, see Materials and Methods). Brain sections were prepared at various distances from the frontal pole. Note smaller areas of ATP depletion as compared to acidosis or inhibition of protein synthesis. For quantification of ATP and pH, see gray scale; inhibition of protein synthesis is visualized by the absence of radioactivity in autoradiograms following [3H]leucine incorporation.
The volume of metabolic disturbances, measured 3 hours after MCA occlusion, differed markedly depending on the chosen parameter (Fig. 6). ATP was depleted in 65.3 ± 20.9 mm3 or 33.8 ± 11.2% of the contralateral hemisphere, acidosis could be detected in 83 ± 26.1 mm3 or 43.2 ± 13.1% of hemisphere, and inhibition of protein synthesis in 98.8 ± 29.2 mm3 or 50.8 ± 15.1% of hemisphere. If the infarct core is defined as the area of depleted ATP and the penumbra as that of reduced protein synthesis without ATP depletion, the average volume of penumbra after 3 hours of MCA occlusion amounted to 33.5 mm3 or ~50% of the core volume.
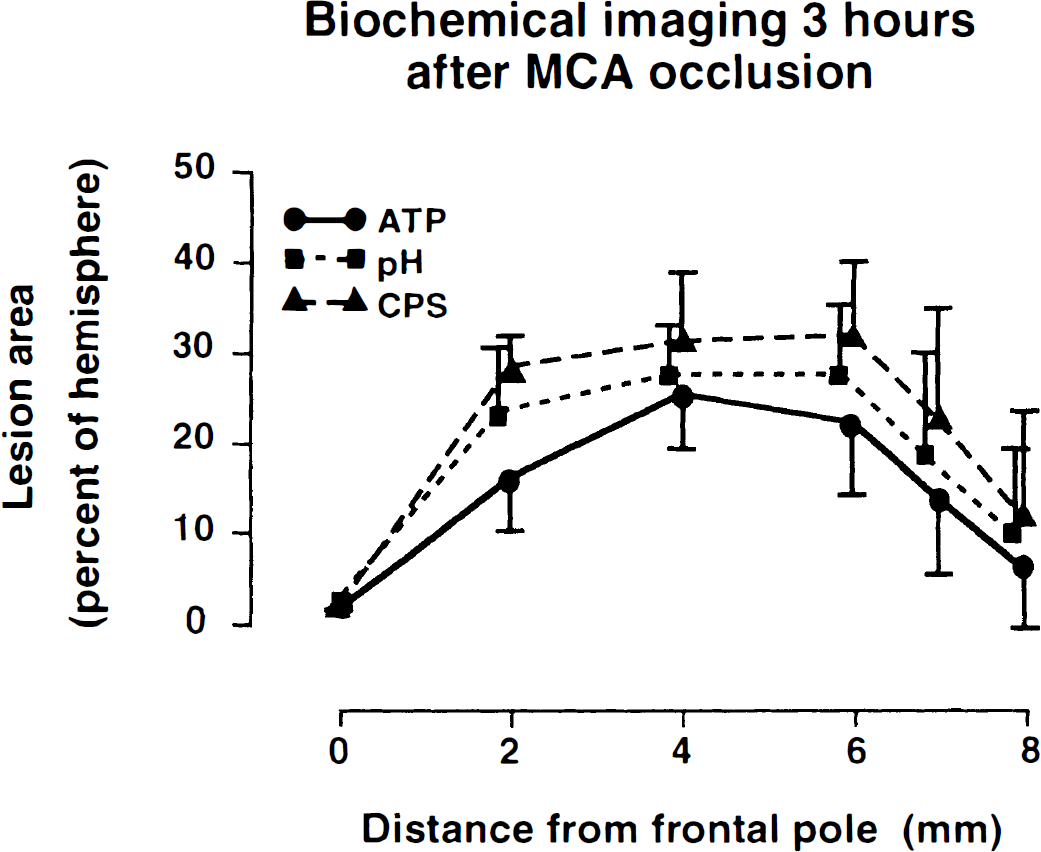
Comparison of areas of ATP depletion, tissue acidosis, and inhibition of cerebral protein synthesis on coronal brain slices prepared at various distances from the frontal pole. Note different sizes reflecting differences between the core region of infarct (in which all biochemical variables are impaired) and the penumbra (in which ATP is preserved). Lesion areas (means ± SD) are expressed as percentage of ipsilateral hemisphere (n = 5).
Magnetic resonance imaging
In all MCA-occluded mice investigated so far (n = 3), the ischemic territory could be clearly detected both by perfusion and by diffusion-weighted MR imaging. At 2.5 hours of vascular occlusion, the apparent diffusion coefficient of brain tissue in the center of the MCA territory was up to 60% lower than in the homotopic region of the opposite nonischemic hemisphere (Fig. 7). The volume of brain tissue in which the apparent diffusion coefficient declined to below 90% of control was 99 to 127 mm3, and that in which it declined to below 75% was 64 to 83 mm3. These volumes come close to the above-described volumes of protein synthesis inhibition and ATP depletion and confirm that apparent diffusion coefficient thresholding allows differentiation between core and penumbra of the evolving infarct.
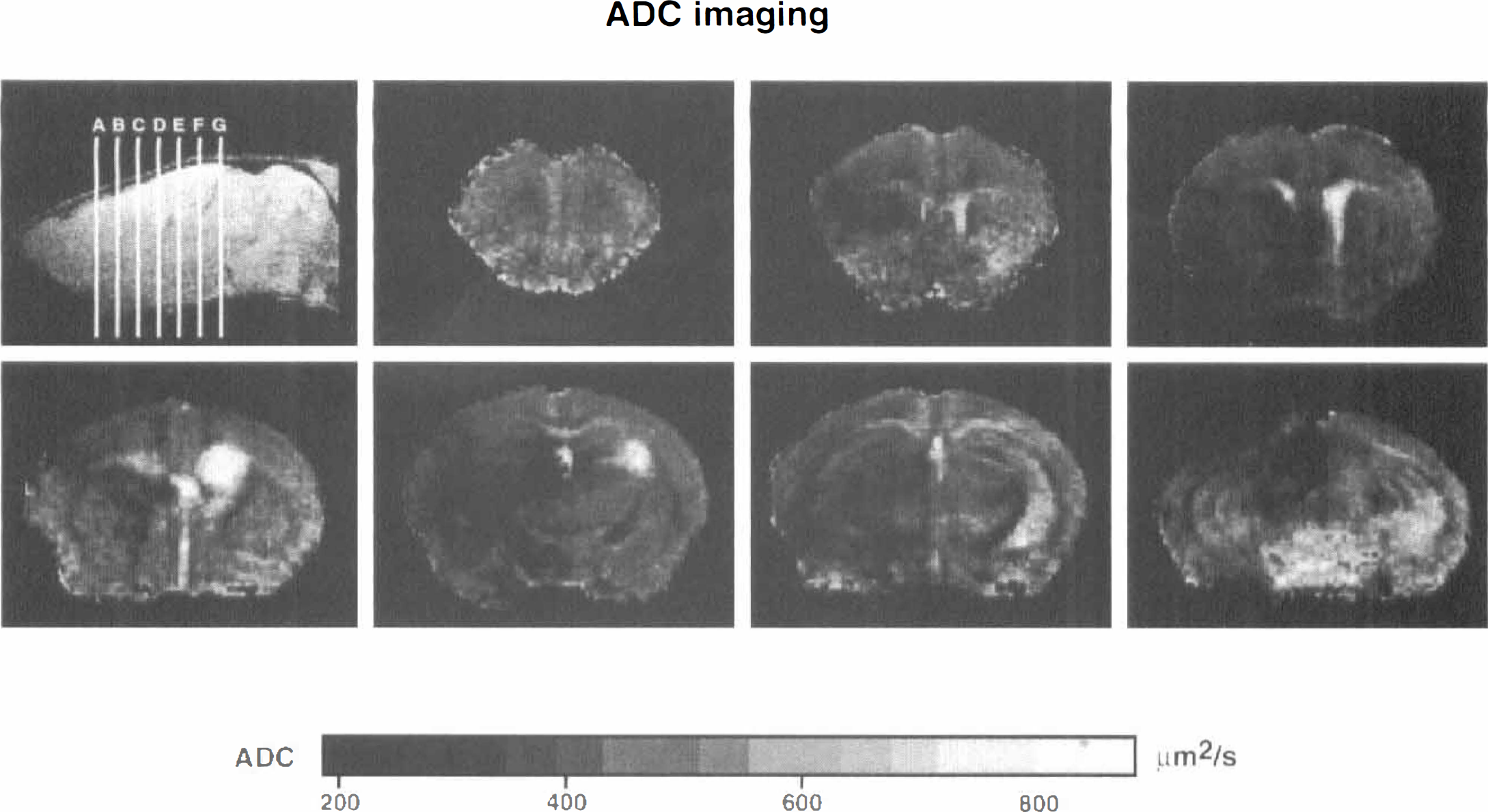
Maps of the apparent diffusion coefficient of mouse brain at 2.5 hours after middle cerebral artery occlusion. Apparent diffusion coefficient maps were reconstructed from three diffusion-weighted images with increasing b-factors. The localization of apparent diffusion coefficient maps (1.5 to 7.5 mm from the frontal pole) is indicated on the sagittal section. The ischemic lesion is visualized by the sharp decline of apparent diffusion coefficient (see gray scale for quantitative values).
Perfusion-weighted images of tissue slices passing through the center of the MCA territory revealed reduced signal intensity throughout the ipsilateral hemisphere (Fig. 8). Although perfusion-weighted images provide only qualitative information on blood flow, the decline of signal intensity in areas that are clearly outside the region in which energy metabolism or protein synthesis is impaired suggests that mice exhibit a gradient of gradually reduced blood flow that stretches far into the periinfarct surrounding.
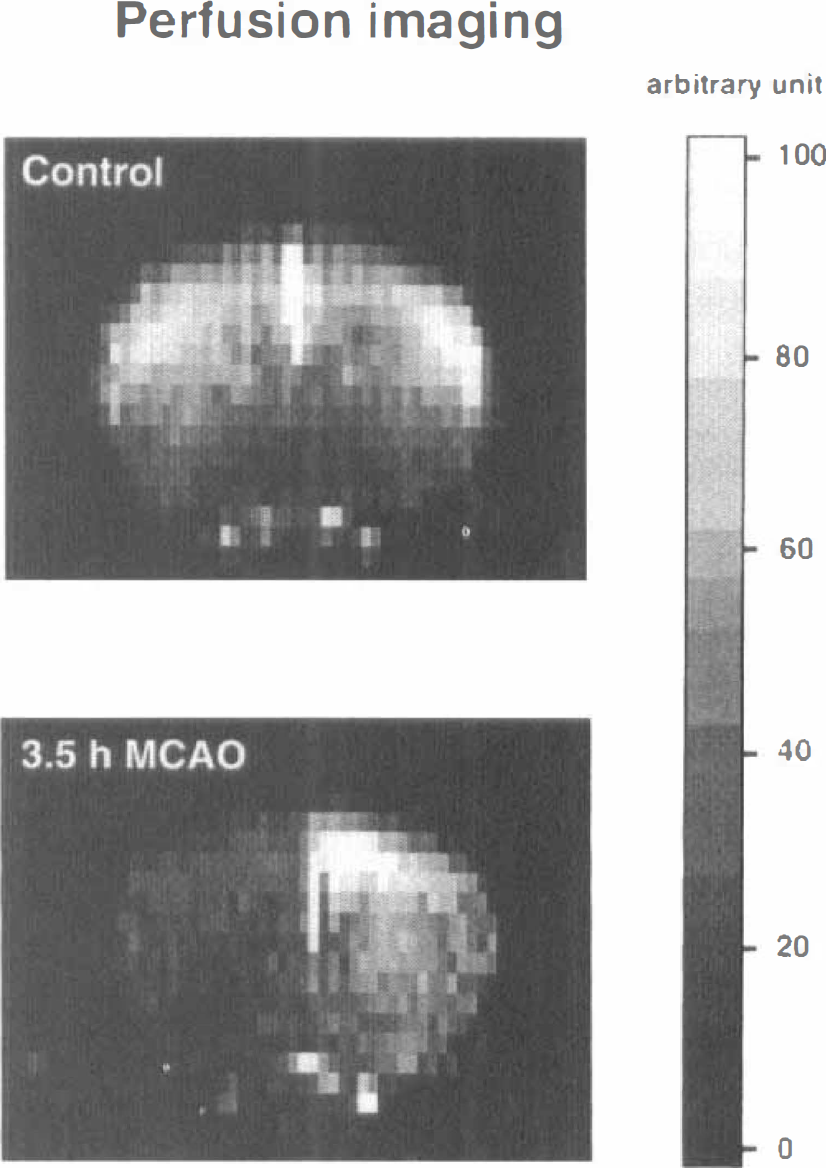
Perfusion-weighted magnetic resonance images in a coronal slice of mouse brain before (control) and 3.5 hours after middle cerebral artery occlusion. Regional perfusion changes were evaluated by magnetic spin tagging, reduction of blood flow being reflected by the decrease in signal intensity (see gray scale for arbitrary density units). Note marked reduction of regional blood flow in the left hemisphere.
DISCUSSION
Reproducibility of focal ischemia
The use of transgenic animals for the study of molecular or hemodynamic mechanisms of focal ischemic injury requires a highly reproducible experimental model that allows the detection of even small differences in infarct volume as compared with that of the wild type. In the present study, we describe a modified mouse thread occlusion model that is suited for this purpose. Middle cerebral artery thread occlusion for induction of focal ischemia was first used in rats by Koizumi et al. (1986) and was later refined by Kamii et al. (1994) and Hara et al. (1996) for application in mice. The main advantage as compared with other MCA occlusion models is the avoidance of craniotomy and the possibility of reperfusion. On the other hand, successful vascular occlusion requires careful selection of the thread diameter because too large threads cannot be advanced far enough to reach the origin of the MCA and too small threads will lead to incomplete obstruction of the vessel lumen. Since both variable thread diameter and variable vessel size will result in inconsistent vascular occlusion, reproducible ischemia requires precise standardization of the experimental conditions. To overcome this problem, we have therefore determined the numerical relationship between thread size and the weight of the animals. Our validation experiments clearly demonstrate that individual selection of thread size according to this relationship results in highly reproducible ischemia in all animals despite large variations in body weight. Obviously, the regression line of this relationship is valid only for the here-investigated C57 Black/6J strain. However, since many gene-manipulated mice are derived from this strain, a similar relationship may also hold for such mutants.
Ischemic penumbra
Another major requirement for the study of focal ischemia in mouse mutants is the presence of a clearly distinguishable periinfarct penumbra. In the infarct core, oxygen supply is too low to sustain cell viability irrespective of any other injury-promoting or -alleviating genomic responses. Specific modulations of genomic responses are therefore most likely detected in the penumbra.
The original concept of the ischemic penumbra goes back to Symon et al. (1977) who described two thresholds of blood flow reduction: a higher one below which electrical activity ceases and a lower one that results in membrane depolarization. The penumbra is the peripheral part of the ischemic territory with flow rates in between the two thresholds, representing an area that is functionally disturbed but structurally intact (Astrup et al., 1981). Later, core and penumbra were differentiated in other ways but with the general agreement that the core is the irreversibly damaged and the penumbra is the still viable part of the ischemic territory (for review see Hossmann, 1994). Since preservation of energy-producing metabolism is a basic requirement of cell viability, core and penumbra can be demarcated by the presence of energy metabolites: The core is the central part of the ischemic territory in which ATP is depleted and the penumbra the peripheral part in which the energy state is still preserved. However, imaging of ATP does not allow the demarcation between penumbra and normal tissue. For this reason, various propositions have been made to identify the penumbra by other means, such as the expression of stress proteins (Kinouchi et al., 1993) or the loss of calmodulin staining as an indicator of the increase in intracellular Ca2+ (DeGraba et al., 1993). Here we propose two markers of the beginning functional impairment: tissue acidosis and the suppression of global protein synthesis. The region of disturbed protein synthesis was slightly larger than that of tissue acidosis, which is in line with the previously described differences in flow thresholds (Hossmann, 1994). In fact, a beginning disturbance of protein synthesis has been described in the rat at flow values of below 50 mL 100 g−1 min−1 (Mies et al., 1991), whereas stimulation of anaerobic glycolysis and tissue acidosis occurs at flow values below 35 mL 100 g−1 min−1 (Paschen et al., 1992).
The suppression of global protein synthesis in the penumbra is not at variance with the increased expression of immediate-early or stress-related genes because the protein products of such inducible genes are quantitatively negligible as compared with the global protein synthesis rate. The present demonstration of a dissociation between energy and protein synthesis in the periphery of focal ischemia in the mouse underscores the usefulness of this model for the study of penumbra changes.
Magnetic resonance imaging
Another important result of our study is the demonstration of the feasibility of advanced MR imaging in the mouse focal ischemia model. Previously, diffusion- and perfusion-weighted imaging has been successfully used for noninvasive detection of acute cerebral ischemia in various species including humans (for reviews see Hossmann and Hoehn-Berlage, 1995; Moseley et al., 1995) but not in mice. The interest of this technique lies in the possibility to monitor, with high temporal and spatial resolution, both the manifestation and the regression of ischemic brain lesions. It is therefore possible to investigate the influence of pharmacological or genomic interventions on the dynamics of brain infarct evolution.
The signal intensity change visible in diffusion-weighted imaging depends on the volume and tortuosity of the extracellular space (Mintorovitch et al., 1994; Szafer et al., 1995), which are a function of the energy and ion homeostasis of the brain (Hossmann, 1994). Quantification of the apparent diffusion coefficient—by recording a series of diffusion-weighted images with increasing gradient strengths—allows the numerical correlation with thresholds of various metabolic disturbances and thus contributes to the differentiation between core and penumbra (Hoehn-Berlage et al., 1995).
Recent advances in MR technology also allow the noninvasive measurement of tissue perfusion. Here we demonstrate for the first time that the arterial spin-tagging technique proposed by Detre et al. (1992) can be used in mice. Arterial water spins that are labeled in the neck vessels by magnetic inversion flow into the brain and exchange with tissue water. The resulting decrease in tissue water magnetization can be detected by fast MR imaging and provides qualitative information on regional blood flow (Williams et al., 1992).
The perfusion-weighted images of the present study demonstrate a decline in signal intensity in the ipsilateral hemisphere that is in good spatial agreement with the region of low apparent diffusion coefficient values. This concordance is similar to that previously described in rats (Jiang et al., 1994) and confirms the feasibility of MR imaging techniques for the investigation of stroke in mice.
CONCLUSION
We propose an improved thread occlusion model for the production of focal ischemia in mice that can be applied to animals with greatly different weights. By matching the diameter of the thread to the weight of the animal, consistent cerebral infarction could be produced. Three hours after MCA occlusion, the metabolic penumbra could be demonstrated by imaging of ATP, tissue pH, and protein synthesis. Furthermore, we succeeded in diffusion- and perfusion-weighted imaging of the mouse brain and demonstrated that ischemic lesions are visible in the vascular territories of the occluded arteries. These data may be useful for the pathophysiological investigation of cerebral ischemia not only in normal mice but also in gene-manipulated animals.
Footnotes
Abbreviations used
Acknowledgments
The authors thank Ms. U. Uhlenküken, Ms. M. Nelles, Ms. S. Krause, and Ms. U. Beckmann for technical assistance, and Ms. D. Schewetzky for secretarial assistance.
