Abstract
The effect of interleukin-1β (IL-1β) on a cerebral artery was investigated in anesthetized dogs. Intracisternal administration of IL-1β (0.03 and 0.3 μg) dilated the canine basilar artery in a dose-dependent manner, without affecting systemic blood pressure or heart rate. The increase in diameter induced by 0.3 μg of IL-1β was 28.4% ± 13.4% of control at 2 hours and was inhibited by 30 μg of the IL-1β receptor antagonist, zinc protoporphyrin (4.5% ± 13.5%, P < 0.05). Interleukin-1β did not affect the concentration of nitric oxide metabolites in CSF. However, there was an increase in the concentration of eicosanoids in CSF, and the elevation of 6-keto-PGF1α paralleled the vasodilation. Pretreatment with 30 μg of the selective inducible cyclooxygenase (COX-2) inhibitor NS-398 also inhibited the IL-1β-induced vasodilation significantly (5.9% ± 9.4% at 2 hours, P < 0.01). Western blot analysis revealed the expression of a 68-kD COX-2-like protein in basilar artery extracts. These findings suggest that the IL-1β-induced vasodilator effect is linked to the prostaglandin cascade, predominantly to prostaglandin I2, by induction of COX-2, but not to the stimulation of nitric oxide metabolism.
Interleukin-1β (IL-1β) is a proinflammatory cytokine capable of exhibiting a wide spectrum of biologic activities, although its constitutive expression in the nervous system is limited. Injection of IL-1β into the cerebral ventricles or specific brain regions has been reported to cause numerous pathologic processes, including fever (Palmi et al., 1995), slow-wave sleep (Krueger et al., 1984), pituitary adrenal activation (Berkenbosch et al., 1987; Katsuura et al., 1988), hypophagia (Uehara et al., 1989), hyperinsulinemia (Cornell and Schwartz, 1989), behavioral changes (Berkenbosch et al., 1987), glial proliferation (Giuilian et al., 1988) and neovascularization (Giuilian et al., 1988). Recent studies have shown that IL-1β inhibits long-term potentiation (Katsuki et al., 1990) and γ-aminobutyric acid receptors (Miller et al., 1991) in hippocampal and cortical slices. Interleukin-1β seems to affect many intercellular messages through specific IL-1 receptors in brain and peripheral tissues.
In cerebral diseases, including ischemia, trauma, and hemorrhage, IL-1β mRNA expression has been demonstrated to be acutely elevated, probably to mediate acute inflammatory steps (Liu et al., 1993; McClain et al., 1987; Minami et al., 1992). Expression of IL-1β mRNA has been observed in various cells of the central nervous system, including microglia, astrocytes, and vascular endothelial cells (Liu et al., 1993). Interleukin-1β has been shown to enhance blood flow and exacerbate ischemic brain injury caused by an increase in vascular permeability in a stroke model (Kadoya et al., 1995; Liu et al., 1993; Relton and Rothwell, 1992; Yamasaki et al., 1995), and IL-1α dilates pial arterioles through enhanced production of prostanoids in piglets in vivo (Shibata et al., 1990, 1996).
To evaluate a direct role of IL-1β on the cerebral circulation, we studied the vasoactive effect of IL-1β on the diameter of the basilar artery on angiograms after intracisternal administration of IL-1β in dogs in vivo. The vasodilator mechanism of IL-1β was investigated using pharmacologic and biochemical analysis of cerebrospinal fluid (CSF) and the cerebral arterial wall.
MATERIALS AND METHODS
Animal preparation
Experiments were carried out in accordance with the guidelines for the care and use of animals in the physiologic sciences as approved by the Physiological Society of Japan.
Mature mongrel dogs of either gender, weighing 8 to 14 kg, were anesthetized with ketamine hydrochloride (10 mg/kg, intramuscularly) and pentobarbital sodium (20 mg/kg, intravenously, followed by 5 mg/kg per hour). Slow intravenous infusion of saline was maintained during the procedure to prevent dehydration. Ventilation was controlled through an endotracheal tube, with room air delivered by a respirator (model B2, Igarashi Ika Kogyo Co., Tokyo, Japan). The respiratory rate (approximately 12 cycles/min) and tidal volume (20 mL/kg) were adjusted to maintain arterial blood gas levels and pH within physiologic limits. Body temperature was maintained at 37° to 38°C with a heating pad. The systemic mean arterial blood pressure (MABP) and heart rate (HR) were monitored with a catheter placed in the right femoral artery using a pressure transducer (model AP-601G, Nihon Kohden Co., Tokyo, Japan) in conjunction with a heart rate coupler (model AT-600G, Nihon Koden Co.).
Angiographic measurement of vascular diameter
The vasodilator effects of IL-1β on cerebral arteries were determined by means of angiography. A 5F catheter was inserted under fluoroscopic control into the vertebral artery via the left femoral artery. Angiography was performed at a fixed magnification with 3 mL of 65% iothalamate meglumine. Angiograms were obtained before, and at 0.5, 1, 2, 3, 4.5, and 6 hours after the injection of 1.5 mL of the control or test solutions into the cisterna magna. The internal diameters of the arteries were measured with a computerized image analysis system (Power Macintosh 7600 computer, Apple Computer, Cupertino, CA, U.S.A.; Image 1.59 software supplied by the National Technical Information Service, Springfield, VA, U.S.A.).
Effect of intracisternal IL-1β on MABP and HR
The effect of IL-1β (0.03 or 0.3 μg doses) on MABP and HR was determined after injection of the test solution into the cisterna magna. In some animals, 1.5 mL of normal saline containing 75 μL of phosphate-buffered saline was used as a control. All test substances were dissolved in the same amount of normal saline just before use. The pH of all test solutions was close to that of the control solution. Solutions were injected gently (1 mL/min) through a 22-gauge spinal needle after withdrawal of an equal amount of cerebrospinal fluid to maintain a constant intracranial pressure.
Effect of central IL-1β receptor blockade and inducible cyclooxygenase (COX-2) inhibitor on canine basilar artery response to intracisternal IL-1β
To determine whether the effects were mediated by the IL-1β receptor, the IL-1β receptor antagonist, zinc protoporphyrin (ZnPP, 30 μg), was administered into the cisterna magna simultaneously with the administration of 0.3 μg of IL-1β. The ZnPP was dissolved in a small amount of 0.2 N NaOH and diluted with phosphate-buffered saline. The pH of the solution was adjusted to 7.4 by titration of 0.2 N HCl and diluted in 1.5 mL of normal saline. Likewise, the selective inhibitor of COX-2, N-(2-cyclohexyloxy-4-nitrophenyl)-methanesulfonamide (NS-398), was injected into the cisterna magna 15 minutes before the administration of 0.3 μg of IL-1β to determine whether the effect was mediated by COX-2. The NS-398 was dissolved in ethanol and diluted in 1.5 mL normal saline (final concentration was 0.5%) and injected as described above.
Assay of IL-1β, nitric oxide metabolites, and eicosanoids in CSF
Assays for IL-1β, nitric oxide metabolites, and eicosanoids were performed using 1.5 mL CSF samples obtained before and at 0.5, 1, 2, 3, 4.5, and 6 hours after the injection of 1.5 mL of the control or test solution into the cisterna magna. Samples were stored at −1280°C until assayed. Interleukin-1β was measured using enzyme-amplified sensitivity immunoassay methods (Medgenix, Fleurus, Belgium). The nitric oxide end products, nitrite and nitrate, were assayed by the Griess reagents method (Cayman Chemical, Ann Arbor, MI, U.S.A.). 6-Ketoprostaglandin F1α (6-keto-PGF1α), thromboxane B2 (TXB2), and prostaglandin E2 (PGE2) were measured using commercially available enzyme immunoassay kits (Amersham, Buckinghamshire, U.K.). In each assay, the sample was assayed in duplicate, and the mean level was used. The limits of detection of these assays were 3.0 pg/mL for 6-keto-PGF1α, 3.6 pg/mL for TXB2, 40 pg/mL for PGE2, 2 pg/mL for IL-1β, and 2.5 μmol/L for nitrite and nitrate.
Canine basilar artery preparations
Mongrel dogs were exsanguinated through the carotid and femoral arteries 6 hours after injection of IL-1β (0.3 μg) and were perfused with 500 mL normal saline. The basilar artery was removed carefully with the entire brain. After removal of the arachnoid membrane and connective tissue, the basilar artery was quickly frozen in liquid nitrogen and stored at −80°C until further use. Dogs were killed by exsanguination under full anesthesia without injection of IL-1β, and the basilar arteries were used as control samples.
Western blot analysis
The basilar arteries were minced and homogenized in 30 μL/mg tissue electrophoresis sample buffer (2% sodium dodecyl sulfate (SDS), 10% glycerol, 0.1% bromophenol blue, 2% 2-mercaptoethanol, and 50 mmol/L Tris-HCl, pH 7.2). After sonication, solubilized proteins (10 μg/lane) were subjected to SDS-polyacrylamide gel electrophoresis (7.5% acrylamide, 1.0-mm-thick slab gels). Proteins were then transferred to a polyvinylidene difluoride membrane by semidry electroblotting. The membrane was blocked for 1 hour at room temperature with 1 % skim milk and incubated with rabbit polyclonal COX-2 antibody (1:500 dilution) for 45 minutes. After washing, the membrane was incubated for 30 minutes with donkey anti–rabbit IgG conjugated to horseradish peroxidase (1:1,000). Peroxidase activity was visualized with an enhanced chemiluminescence Western blotting detection system (Amersham).
Materials
Interleukin-1β (recombinant human) was purchased from Genzyme (Cambridge, MA, U.S.A.). Zinc protoporphyrin IX (ZnPP), an IL-1 blocker, was purchased from Aldrich (Milwaukee, WI, U.S.A.). NS-398 was obtained from Biomol Research Laboratories (Plymouth Meeting, PA, U.S.A.). The rabbit polyclonal antibody against the C-terminal fragment of human COX-2 was obtained from Oxford Biomedical Research (Oxford, MI, U.S.A.); it does not cross-react with constitutive cyclooxygenase (COX-1). All other chemicals were reagent grade or the best commercially available grade.
Statistical analysis
Data are expressed as mean ± SD. Changes in vessel diameter, concentrations of prostanoids, and MABP and HR were evaluated statistically using an analysis of variance for repeated measures. Significant changes were subsequently delineated using the Bonferroni–Dunn method. A probability of less than 0.05 was considered statistically significant. Serial changes in nitric oxide metabolites were assessed by a one-way analysis of variance.
RESULTS
Angiography and concentration of IL-1β
Intracisternal injection of IL-1β (0.03 μg to 0.3 μg) induced a dose-dependent increase in the internal diameter of the canine basilar artery, as determined by angiography (Fig. 1). The middle portion of the basilar artery was chosen for the quantitative assessment of the change in blood vessel diameter. The maximal dilation induced by 0.3 μg of IL-1β was 28.4% ± 13.4% of the baseline at 2 hours (Fig. 2). No significant changes in MABP or HR were detected after administration of IL-1β, at either dose, during the 6 hours after injection (Table 1).
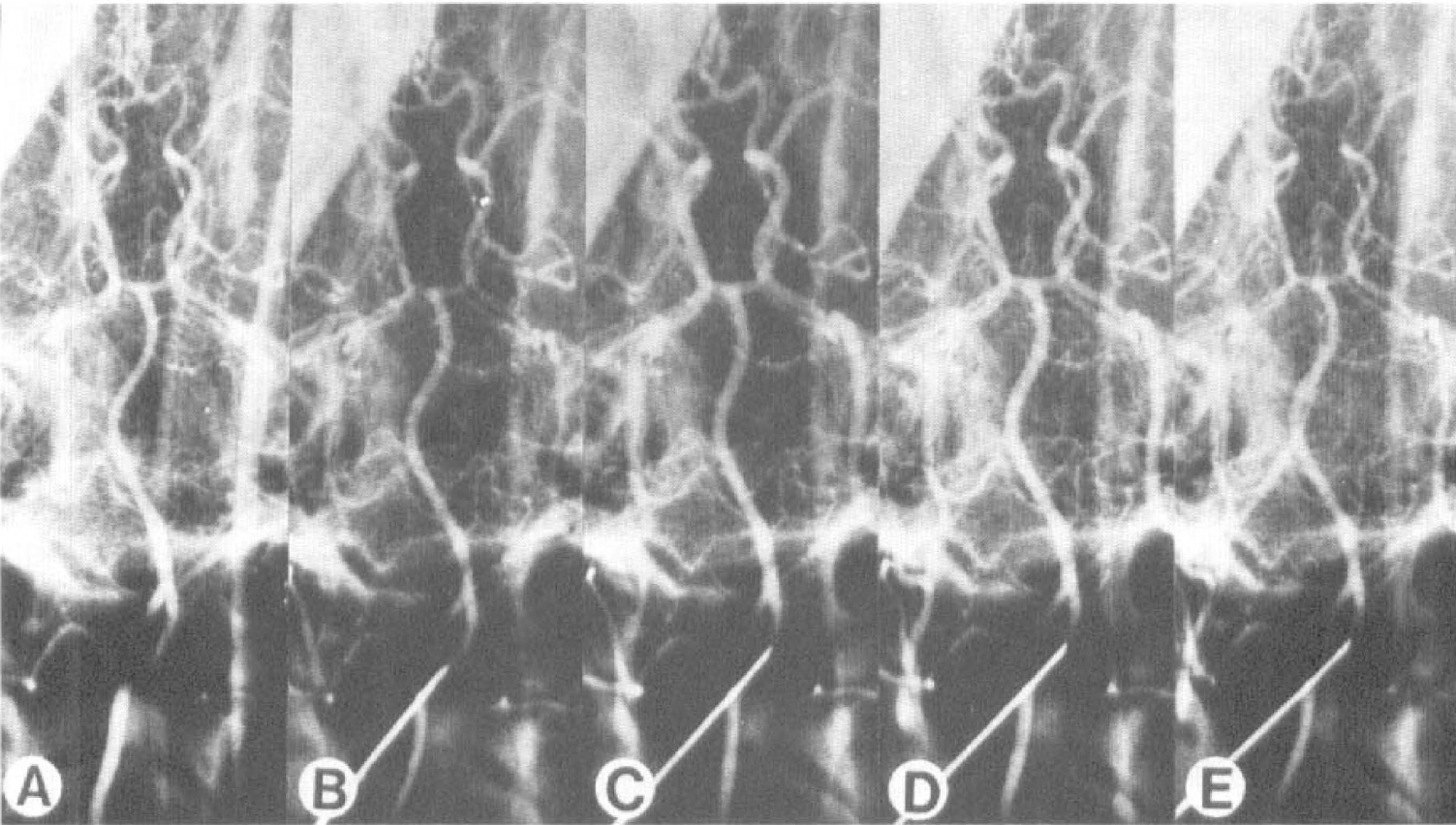
Representative vertebral angiograms showing the vasodilator effect of interleukin-1β (IL-1β) on the canine basilar
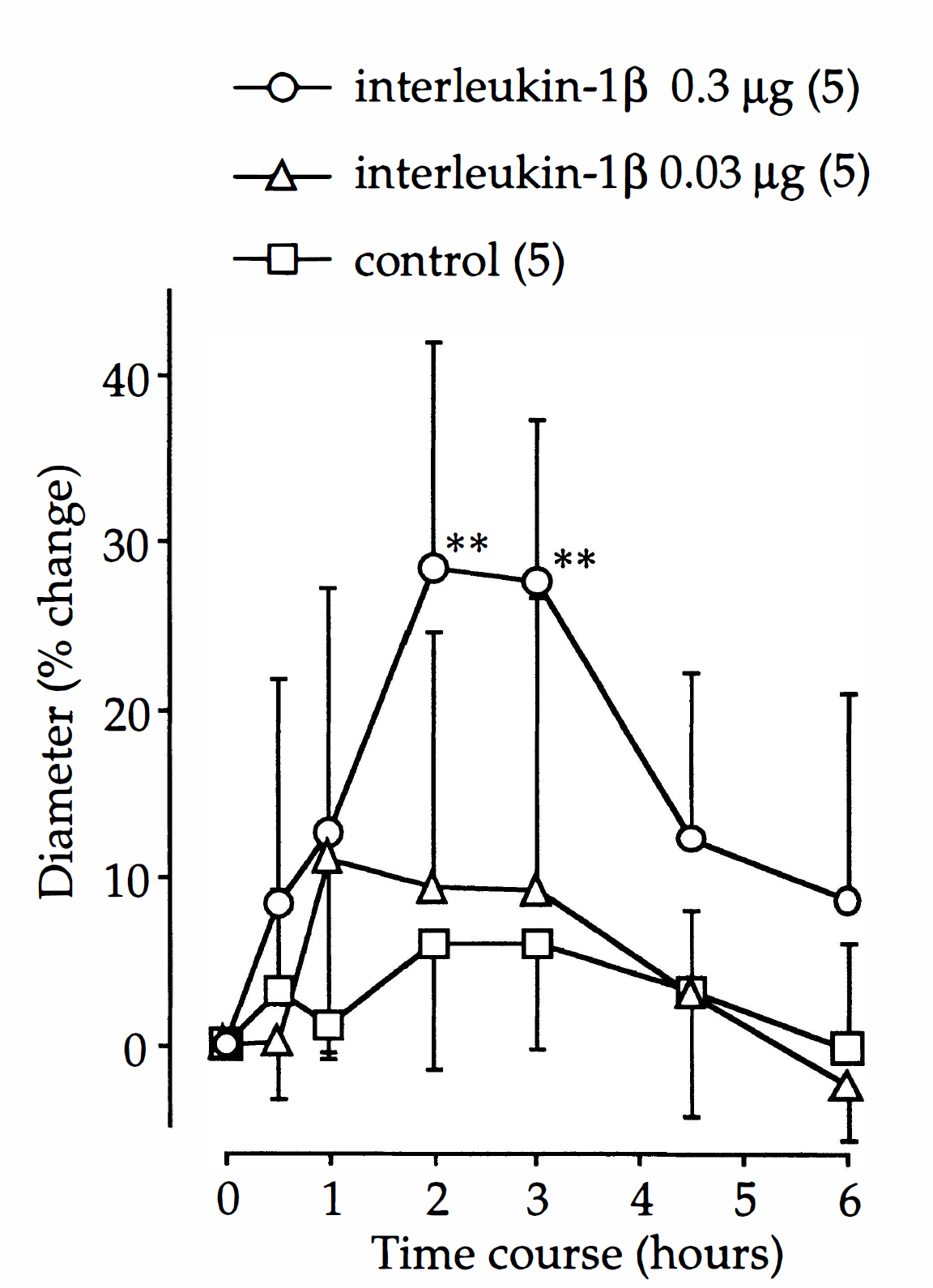
Time course of the vasodilator response of the canine basilar artery to intracisternal administration of interleukin 1β (IL-1β). The actual baseline diameter in control, IL-1β 0.03 μg, and IL-1β 0.3 μg groups are 1.11 ± 0.15 mm, 1.19 ± 0.08 mm, and 1.03 ± 0.06 mm, respectively. Values are mean ± SD; number of animals in parentheses. **Significantly different from before control group, P < 0.01.
Change in mean arterial blood pressure and heart rate after intracisternal administration of interleukin-1β
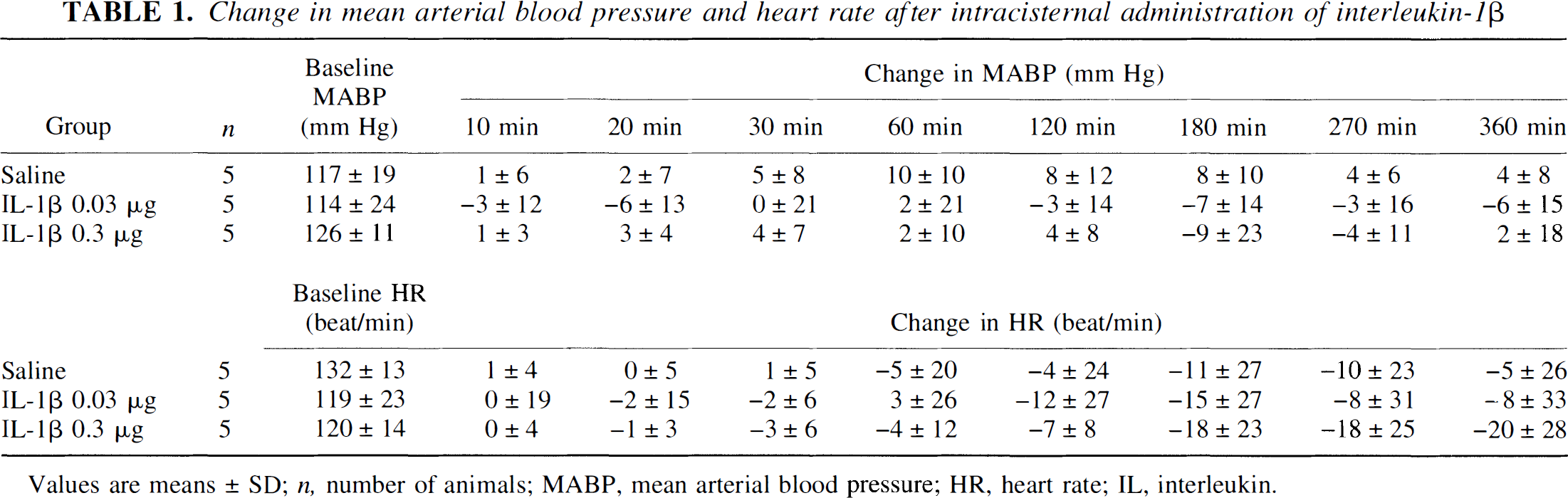
Values are means ± SD; n, number of animals; MABP, mean arterial blood pressure; HR, heart rate; IL, interleukin.
The basal concentration of IL-1β in CSF before injection of IL-1β was 2.8 ± 3.8 pg/mL (n = 10). The concentrations of IL-1β in CSF after injection revealed a logarithmic disappearance. At 6 hours after injection of 0.3 μg and 0.03 μg of IL-1β, the concentrations of IL-1β in CSF were 1.35 ± 0.71 ng/mL and 0.13 ± 0.08 ng/mL, respectively (Fig. 3).
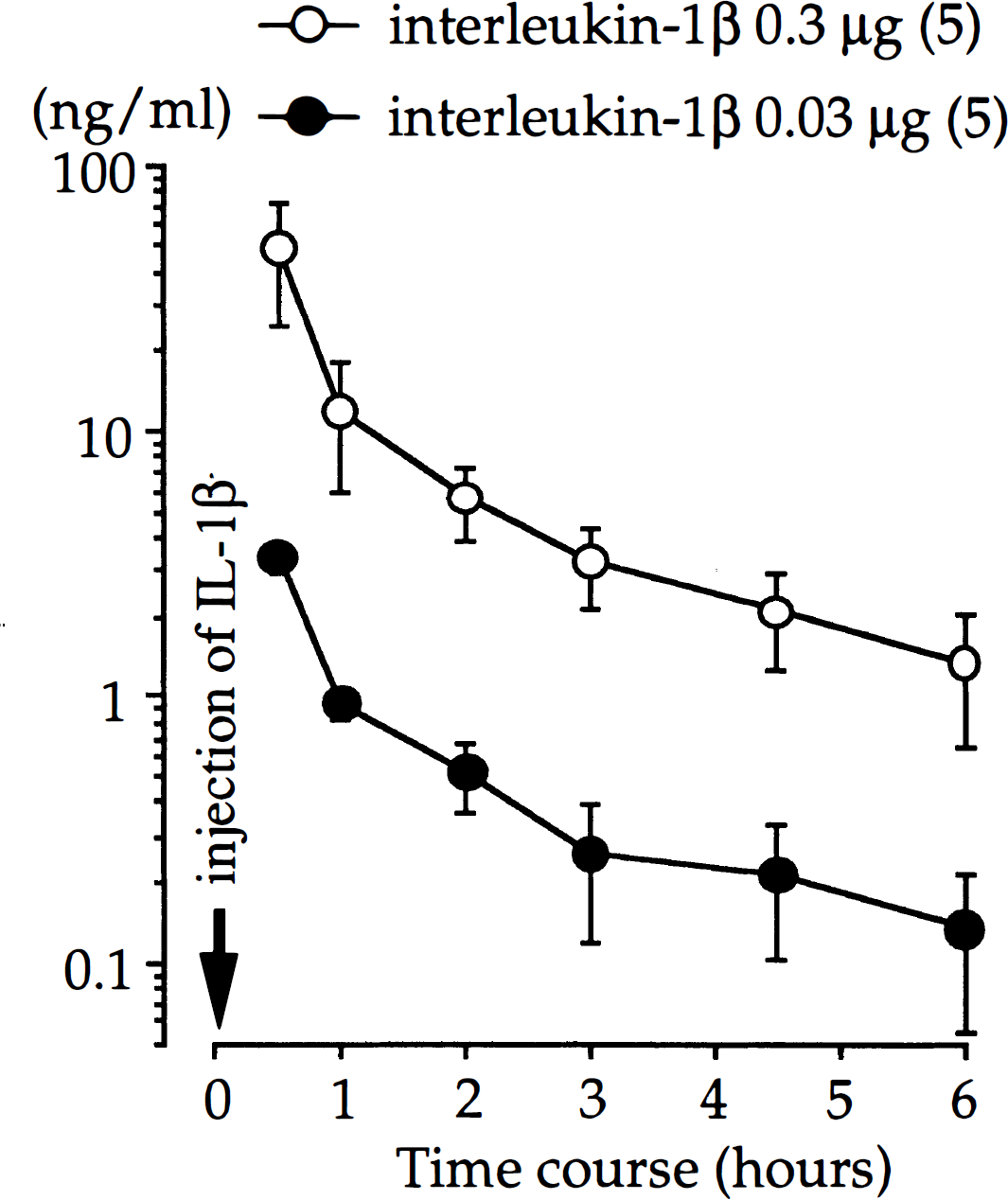
Serial changes in concentrations of interleukin-1β (IL-1β) in cerebrospinal fluid after intracisternal administration of IL-1β at time 0. Values are mean ± SD; number of animals in parentheses.
Effect of IL-1β receptor antagonist
Intracisternal injection of the IL-1β receptor antagonist, ZnPP (30 μg), alone did not significantly affect the diameter of the canine basilar artery. Simultaneous injection of ZnPP and IL-1β significantly inhibited the IL-1β-induced vasodilation (P < 0.05 at 2 and 3 hours; Fig. 4).
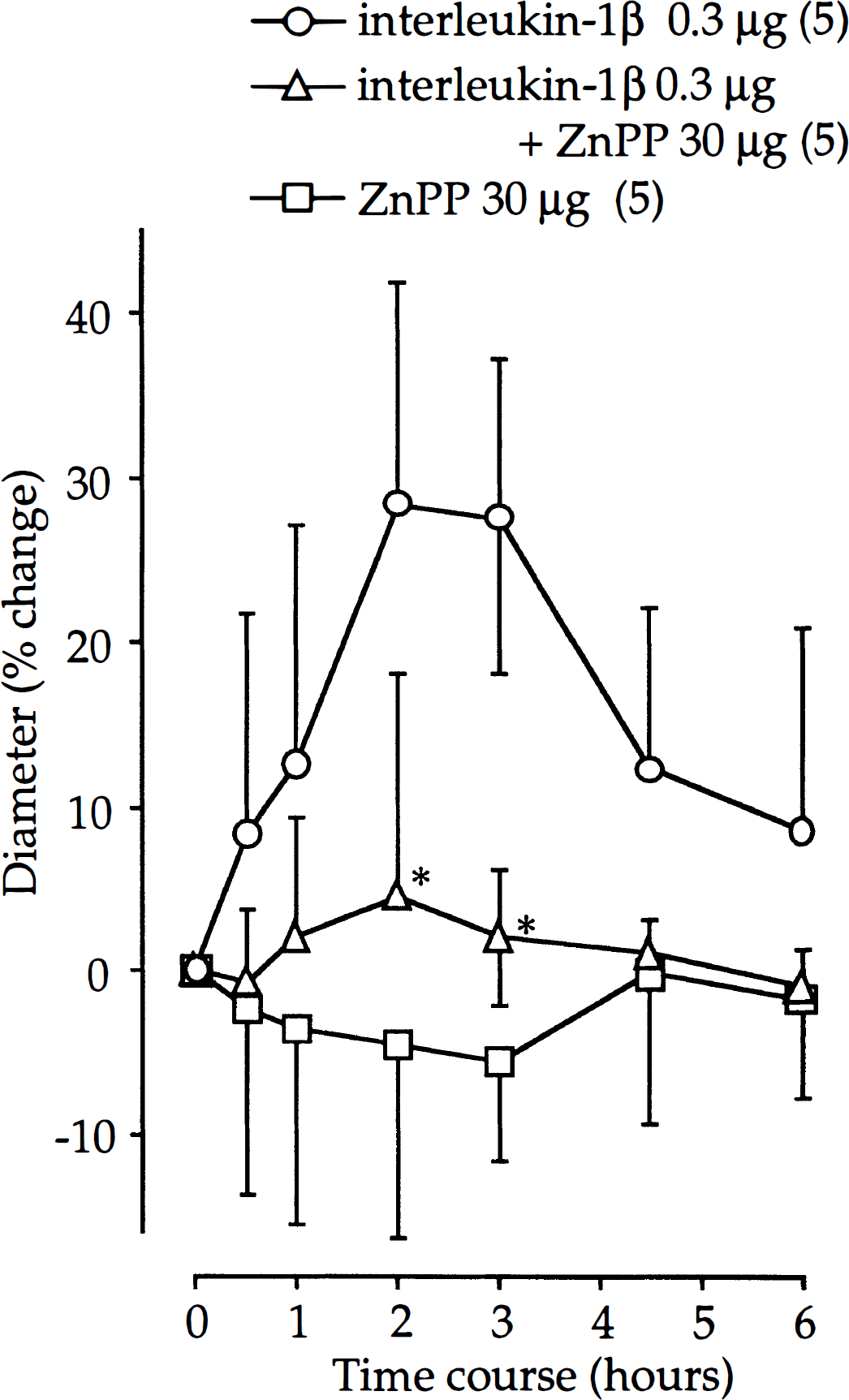
Effect of simultaneous administration of interleukin-1 receptor antagonist (ZnPP, 30 μg) on vasodilator response produced by intracisternal administration of interleukin-1β (IL-1β). The actual baseline diameter in ZnPP, ZnPP + IL-1β 0.3 μg, and ZnPP + IL-1β 0.3 μg groups are 1.28 ± 0.08 mm, 1.36 ± 0.18 mm, and 1.03 ± 0.06 mm, respectively. Values are mean ± SD; number of animals in parentheses. *Significant difference from IL-1β 0.3 μg group, P < 0.05.
Concentrations of nitric oxide metabolites and eicosanoids in CSF
The concentration of the nitric oxide metabolites, nitrite and nitrate, in CSF showed no significant increase from control after injection of IL-1β. The maximum concentration (1.40 ± 0.41 μmol/L) was observed 6 hours after injection of IL-1β (Fig. 5).
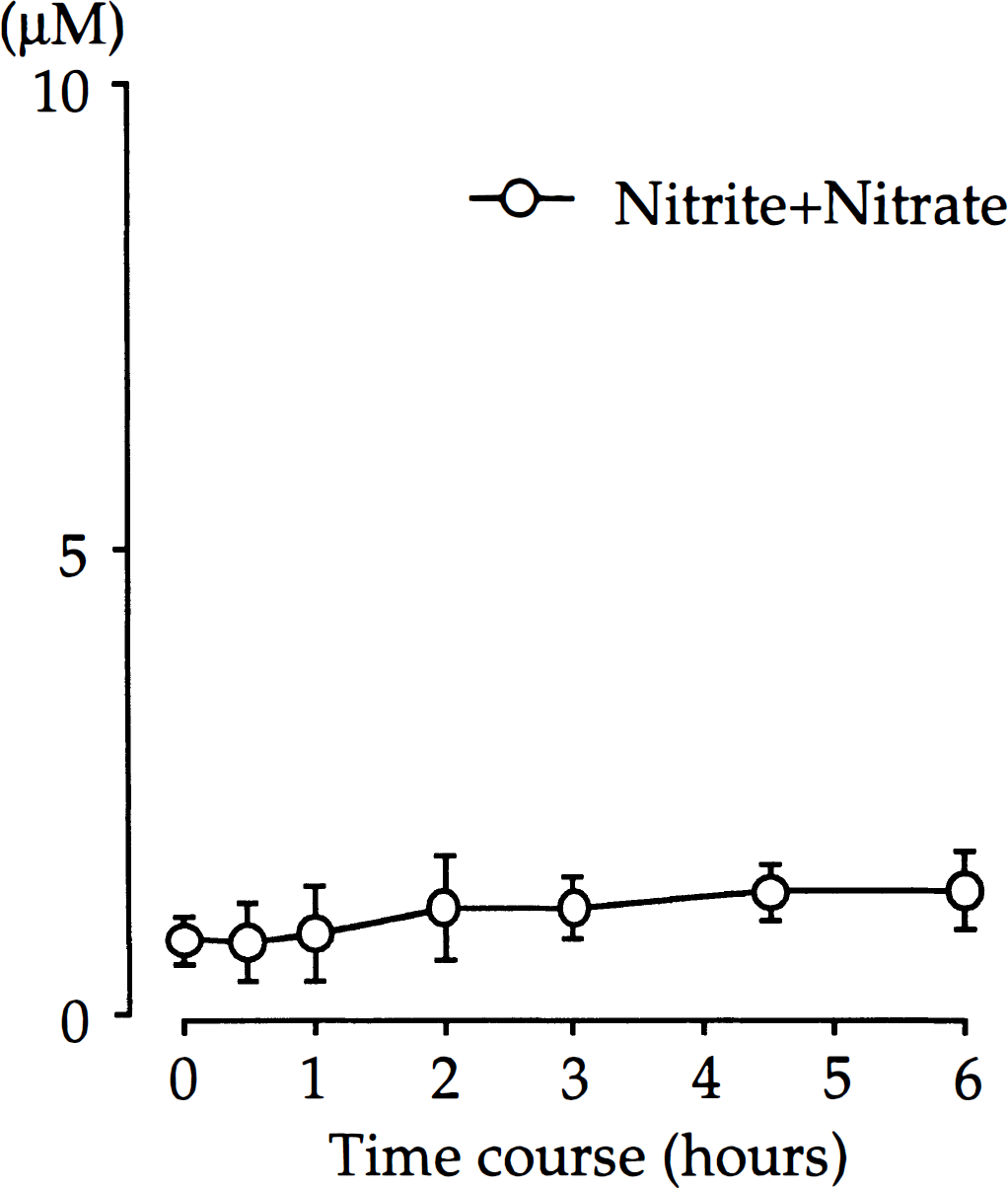
Serial changes in concentrations of nitric oxide metabolites, nitrite and nitrate, after intracisternal administration of 0.3 μg IL-1β. Values are mean ± SD. n = 5.
The basal concentrations of 6-keto-PGF1α, TXB2, and PGE2 in CSF were 0.60 ± 0.20 ng/mL (n = 6), 0.44 ± 0.35 ng/mL (n = 6), and 0.57 ± 0.58 ng/mL (n = 5), respectively. Interleukin-1β induced a dose-dependent increase in the concentrations of these eicosanoids. The maximum concentrations of 6-keto-PGF1α, TXB2, and PGE2 induced by IL-1β (0.3 μg) were 29.6 ± 21.3 ng/mL at 3 hours, 18.2 ± 5.8 ng/mL at 1 hour, and 15.4 ± 4.1 ng/mL at 2 hours, respectively (Fig. 6).
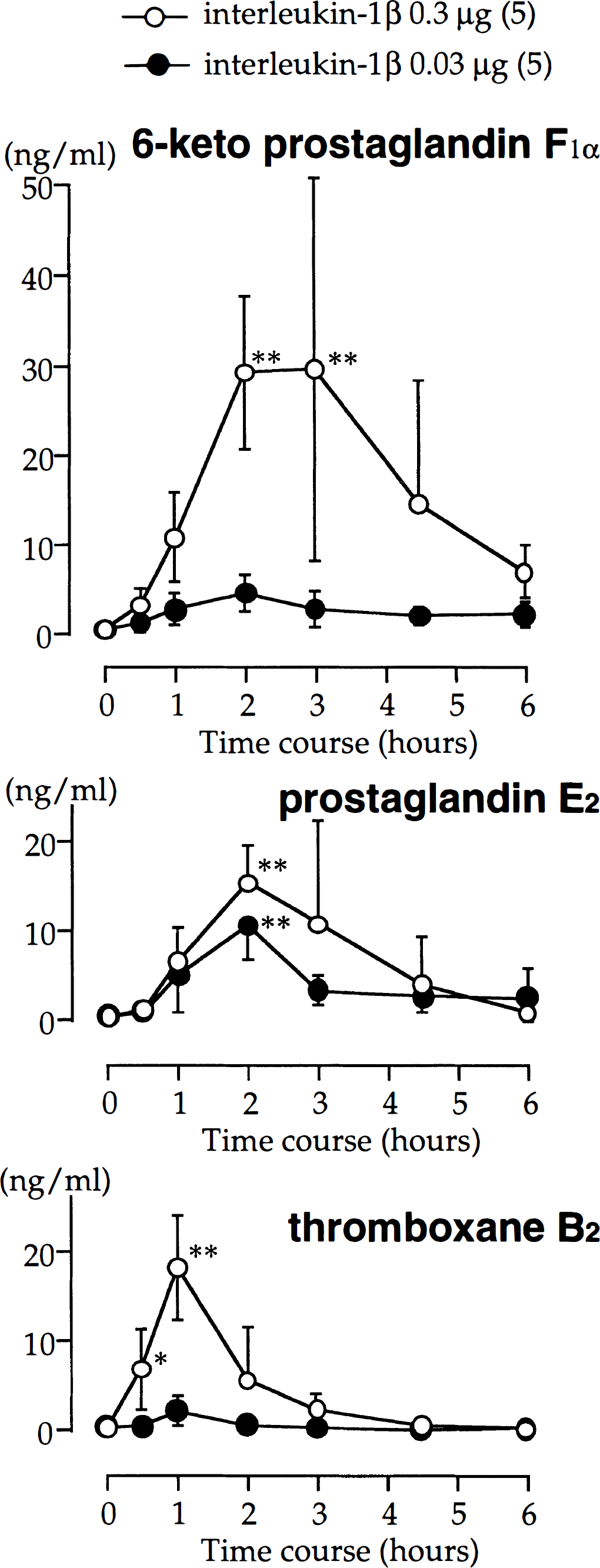
Serial changes in concentrations of 6-keto-prostaglandin F1α, thromboxane B2, and prostaglandin E2 after intracisternal administration of 0.3 μg (○) or 0.03 μg (•) interleukin-1β (IL-1β). Values are mean ± SD; number of animals in parentheses. Significantly different from before control group, **P < 0.01, *P < 0.05.
Effect of COX-2 inhibitor on basilar artery dilation
Pretreatment with NS-398 (30 μg) for 15 minutes before IL-1β injection inhibited the IL-1β-induced vasodilation (P < 0.01 at 2 and 3 hours; Fig. 7). This pretreatment alone caused no significant change in the diameter of the basilar artery.
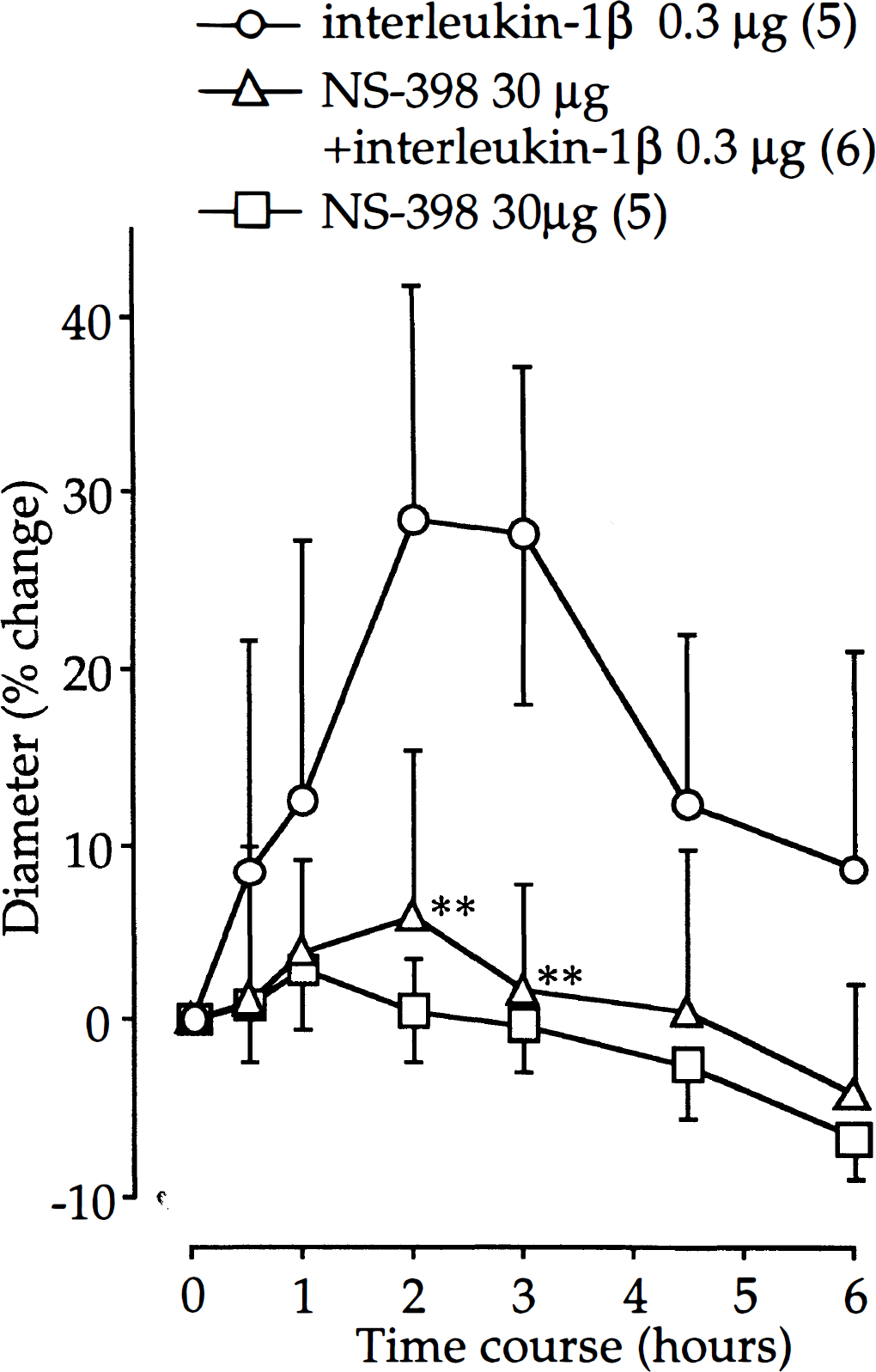
Effect of pretreatment with inducible cyclooxygenase inhibitor NS-398 on vasodilator response produced by intracisternal administration of interleukin-1β (IL-1β). The actual baseline diameters in NS-398, NS-398 + IL-1β 0.3 μg, and NS-398 + IL-1β 0.3 μg groups are 1.28 ± 0.23 mm, 1.36 ± 0.13 mm, and 1.03 ± 0.06 mm, respectively. Values are mean ± SD; number of animals in parentheses. **Significant difference from IL-1β 0.3 μg group, P < 0.01.
Western blot analysis
Western blot analysis using a specific antibody against the C-terminal fragment of human COX-2 showed that a 68-kD band corresponding to the size of the COX-2 protein appeared in basilar artery treated with IL-1β (0.3 μg); such a distinct band was not detectable in non-treated control basilar artery (Fig. 8).
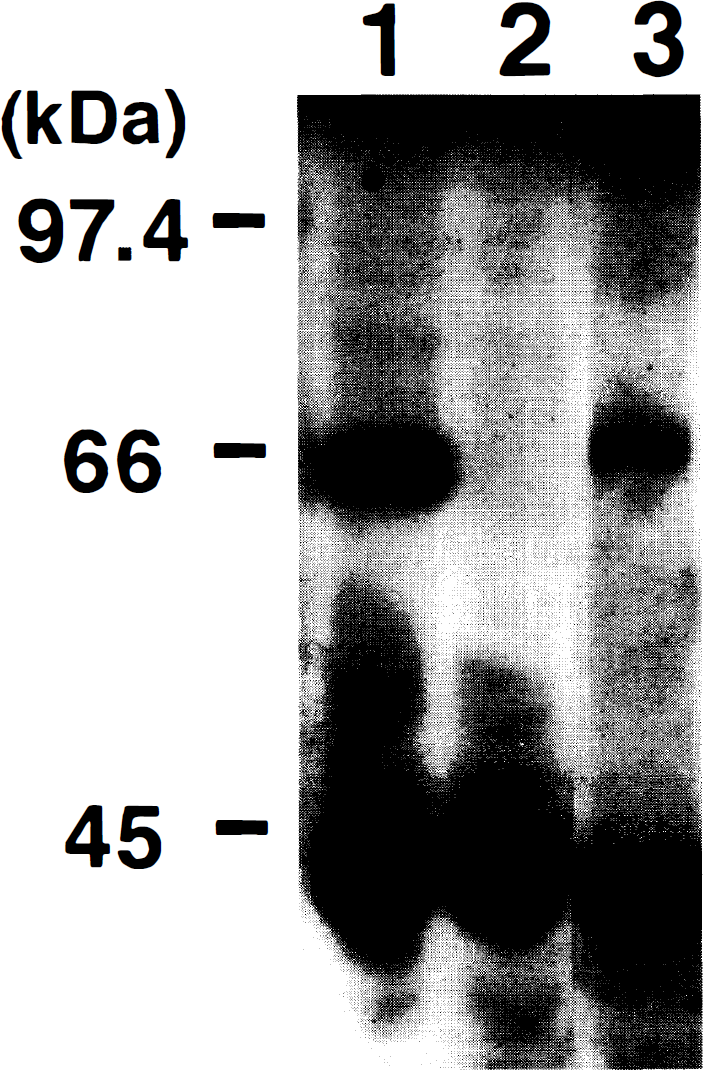
Western blot analysis of inducible cyclooxygenase (COX-2) -like protein induced by 0.3 μg of interleukin-1β (IL-1β) in canine basilar artery. Solubilized proteins from canine basilar arteries were separated by 7.5% sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred to a polyvinylidene difluoride membrane. The membrane was stained using anti–inducible cyclooxygenase polyclonal antibody. Molecular size markers in kilodaltons are shown on the left. Lane 1, positive control; lane 2, control sample; lane 3, treated with 0.3 μg IL-1β.
DISCUSSION
In the present study, intracisternal administration of IL-1β dilated the canine basilar artery in a dose-dependent manner in anesthetized dogs without affecting hemodynamics. Simultaneous injection of the IL-1β receptor antagonist ZnPP significantly inhibited the vasodilator effect of IL-1β. The vasodilation was associated with increases in the concentrations of eicosanoids in CSF; in particular, the elevation of 6-keto-PGF1α was parallel to the vasodilaton induced by IL-1β. No significant changes in the concentrations of nitric oxide metabolites in CSF were seen. Pretreatment with the selective inducible cyclooxygenase inhibitor NS-398 also significantly inhibited the IL-1β-induced vasodilation. A COX-2-like protein was detected in the extract of the basilar artery at 6 hours after injection of IL-1β using Western blot analysis. These findings suggest that IL-1β acts on specific receptors and the vasodilator effect is mediated through the prostaglandin cascade, especially prostacyclin, stimulated by the induced COX-2.
Hemodynamic changes produced by IL-1β, mediated via prostanoids, especially PGE2, have been well studied. An increase in blood pressure in awake rats after intraperitoneal injection of IL-1β (Morimoto et al., 1992) and in awake dogs after intravenous injection of IL-1β (Fukushima et al., 1991) have been documented. In contrast, intravenous injection of IL-1β decreased systemic arterial blood pressure in anesthetized rabbits (Okusawa et al., 1988). After intrathecal administration of IL-1β, there was no significant change in blood pressure in normal anesthetized rats (Plata-Salamán et al., 1995). Likewise our results showed no change in either mean arterial pressure or heart rate after intracisternal administration of IL-1β in anesthetized dogs. Doses and injected routes of IL-1β, species differences, and animal conditions may partly explain these disparities.
Interleukin-1 receptors have been shown to be present throughout the brain, especially where neurons are grouped in tight clusters (Farrar et al., 1987). Zinc protoporphyrin is well known to have anti-inflammatory properties because it inhibits IL-1 receptors, and ZnPP has also been shown to reduce brain infarct size and edema formation after ischemia (Kadoya et al., 1995; Yamasaki et al., 1995). In our study, ZnPP alone slightly, but not significantly, decreased the diameter of the basilar artery for 3 hours, probably because of impairment of endothelial nitric oxide synthase or guanylate cyclase. Zinc protoporphyrin is known to block heme-dependent enzymes, such as guanylate cyclase, heme oxygenase, and nitric oxide synthase (Luo and Vincent, 1994). In fact, we demonstrated that ZnPP inhibits the vasodilation induced by
The concentrations of IL-1β in CSF after injection of IL-1β into the cisterna magna disappeared logarithmically. Even 6 hours after injection of 0.3 μg of IL-1β, the concentration of IL-1β in CSF was 1.35 ± 0.71 ng/mL, significantly higher than the basal concentration of IL-1β (2.8 ± 3.8 pg/mL). The vasodilator response was maximal 2 hours after injection and then gradually declined to 8.6% ± 12.3% of the baseline at 6 hours. This phenomenon seems to be consistent with results obtained by McKean et al. (1994) showing that only 20% binding of surface receptors with IL-1α results in the subsequent desensitization of all cell surface IL-1 type I receptors within 4 hours after stimulation of IL-1 type I receptor–transfected Jurkat T cells. These desensitized cells still have unbound IL-1 type I receptors on the cell surface, and the affinity of these receptors is normal. This novel pathway for IL-1 signaling as a self-regulation system plays an important role in the acute inflammatory response.
Cyclooxygenase has a strong resemblance to nitric oxide synthase (NOS). Both require heme as a cofactor and have constitutive and inducible forms. Interleukin-1β induces the inducible form of NOS (iNOS) in rat vascular endothelial cells and cardiocytes (Kanno et al., 1994; Tsujino et al., 1994), and it induces both iNOS and COX-2 in rat islets of Langerhans (Corbett et al., 1993). Nitric oxide itself stimulates the activity of cyclooxygenase (Corbett et al., 1993; Salvemini et al., 1993; Tetsuka et al., 1994). On the contrary, endogenous PGE2, a COX product, downregulates iNOS induction in rat mesangial cells (Tetsuka et al., 1994). Nitric oxide synthase and COX pathways interact with each other, and the cross talk between these two pathways may be important in inflammatory responses. In our study, IL-1β stimulated the formation of COX-2 in canine basilar artery, as evidenced by the finding that a selective inhibitor of COX-2 completely inhibited the vasodilation caused by IL-1β (Futaki et al., 1994; Masferrer et al., 1994). Western blot analysis using a specific polyclonal antibody against COX-2 also showed a 68-kD protein in basilar artery extracts 6 hours after IL-1β stimulation. However, IL-1β did not stimulate the formation of iNOS. A similar prostaglandin-dependent and nitric oxide-independent response has been demonstrated in human vascular smooth muscle cells (Beasley and McGuiggin, 1995) and in rabbit isolated mesenteric artery (Marceau et al., 1991) after IL-1β stimulation.
Interleukin-1 or endotoxin has been shown to induce prostacyclin predominantly compared with PGE2 in rabbit isolated mesenteric artery (Marceau et al., 1991) and in bovine aortic endothelial cells (Akarasereenont et al., 1995). Cerebromicrovascular endothelial cell lines produce prostacyclin and PGE2 in a ratio of 3:1 (Moore et al., 1988). Prostacyclin and TXB2 are thought to be produced from cerebral vessels, while PGE2 and PGF2α are considered products of nervous tissue. Our in vivo results are in agreement with those of Shibata et al. (1990). In our experiments, 6-keto-PGF1α was significantly increased and PGE2 and TXB2 were also increased in the CSF surrounding the canine basilar artery after IL-1β administration, and in their experiments, 6-keto-PGF1α and a similar concentration of PGE2 were induced in the CSF surrounding the pial arterioles in piglets.
The vasoactivity of PGE2 may be different between the major cerebral arteries and pial arterioles. It has been well known from in vivo studies that PGE2 produces vasoconstriction in the basilar artery in dogs and cats (Handa et al., 1974; Toda et al., 1991; White et al., 1975), whereas it produces vasodilation in the pial arterioles in cats and pigs (Armstead, 1995; Ellis et al., 1979). It seems that PGE2, in addition to TXB2, a well-known vasoconstrictor (Ellis et al., 1977), induced vasoconstriction in our study. Significant vasodilation induced by IL-1β was observed beginning 2 hours after administration of IL-1β. This vasodilation probably reflects the increase of the 6-keto-PGF1α concentration in CSF, because the increasing pattern of 6-keto-PGF1α concentration is parallel to the vasodilation.
The time course of IL-1-induced cyclooxygenase synthesis has been shown to be relatively fast (2 to 8 hours) in both in vitro and in vivo systems. The concentration of PGE2 in CSF appeared to be maximal at 100 minutes after intracerebroventricular administration of IL-1β in rabbits (Palmi et al., 1995). Albert et al. (1994) demonstrated an increase in the mRNA for COX-2 within 30 minutes and an increase in the COX-2 protein itself within 2 hours in amnion-derived WISH cells. In human umbilical vein endothelial cells in vitro, the COX-2 protein and its enzyme activity appeared at 6 to 8 hours and was correlated with an increase in prostacyclin synthesis (Maier et al., 1990). The time course of vasodilation induced by IL-1α (10.8 μg) or IL-1β (1.5 μg) was significantly faster than that observed in this study, within 30 minutes in pial arterioles in piglets (Shibata et al., 1990, 1996).
In piglet pial arterioles, the vasodilation induced by IL-1 was accompanied by a significant increase in prostanoids (6-keto-PGF1α, PGE2, and PGF2α) in CSF (Shibata et al., 1990, 1996). These prostanoids caused a vasodilation not only by accumulation of cyclic nucleotide in the smooth muscle cells, but also by stimulation of NOS activity (Armstead, 1995; Shibata et al., 1990, 1996). In our study, nitric oxide metabolites in CSF did not increase after IL-1β administration. Based on these findings, we suggest that major arteries and pial arterioles respond differently to the proinflammatory cytokine IL-1. It appears that prostaglandins in arterioles preferentially affect endothelial NOS and control the release of nitric oxide, but the prostaglandin cascade stimulated by IL-1β-induced COX-2 produces a direct vasoactive function without affecting NOS in major arteries.
Another rate-limiting step of prostaglandin formation is the release of arachidonic acid from membrane phospholipids, which is mediated by phospholipase A2 (PLA2). Two types of PLA2 exist: cytosolic PLA2 and secretory PLA2. Interleukin-1β has been found to induce not only COX-2 but also cytosolic PLA2 in human synovial cells (Angel et al., 1994; Hulkower et al., 1994) and rat mesangial cells (Schalkwijk et al., 1992). Production of large amounts of PGE2 is considered to be the cause of rheumatoid arthritis and some renal diseases. Interleukin-1 stimulates secretory PLA2 in rat cultured astrocytes (Oka and Arita, 1991). However, the role of PLA2 in the central nervous system in acute inflammation remains unknown.
In conclusion, intracisternal administration of IL-1β dilated the basilar artery in a dose-dependent manner in anesthetized dogs with no systemic hemodynamic effects. This vasodilator effect of IL-1β was mediated through specific receptors in the basilar artery. These receptors seem to be linked to the prostaglandin cascade and not to the nitric oxide pathways. The proinflammatory cytokine IL-1β exerts various effects through these prostanoids, and selective blockage of the induced enzyme may be another potential therapeutic method for patients with acute cerebral disease.
