Abstract
This study investigates how the neuronal and inducible nitric oxide synthase (NOS) pathways contribute to the cerebrovascular changes in the early phase of experimental pneumococcal meningitis in rats. Using a closed cranial window preparation, the diameters of pial arterioles were measured during 4 hours after intracisternal injection of heat-killed pneumococci and compared with controls (n = 6). Injection of pneumococci (n = 7) caused a significant increase in pial arteriolar diameter (157 ± 22% after 4 hours; P < 0.05, compared with 104 ± 11% in controls), intracranial pressure, CSF white blood cell counts, and brain water content. Treatment with the neuronal NOS inhibitor 7-nitroindazole (50 mg/kg given intraperitoneally, n = 5) prevented pneumococci-induced vasodilation (107 ± 20% at 4 hours), whereas S-methylisothiourea (SMT; 0.1 mg/kg given intraperitoneally, n = 5), which predominantly inhibits the inducible NOS, did not influence pneumococci-induced vasodilation (154 ± 38% at 4 hours). S-methylisothiourea at a dose of 1.0 mg/kg (n = 5), attenuated the vasodilation (124 ± 18% at 4 hours). However, the increase in mean arterial blood pressure after SMT at 1.0 mg/kg, but not at 0.1 mg/kg, suggests that the higher dose of SMT influenced the constitutive NOS activity, causing inhibition of the pneumococci-induced vasodilation. Neither SMT (at both doses) nor 7-nitroindazole influenced the increase in brain water content, intracranial pressure, and CSF white blood cell counts in pneumococci-challenged rats. Our study suggests that pial arteriolar vasodilation in the early phase of experimental pneumococcal meningitis is mediated by the neuronal NOS pathway.
Bacterial meningitis in adults still is a serious disease. Mortality rates for pneumococcal meningitis are still as high as 20% to 30% (Berkowitz, 1993; Durand et al, 1993; Pfister et al., 1993). Major complications during the course of the disease include brain edema, increased intracranial pressure (ICP), and cerebrovascular alterations (e.g., focal cortical hyperperfusion, arterial stenosis and spasms), as well as septic sinus venous thrombosis (Pfister et al., 1992). Pial arteriolar vasodilation and an increase in cerebral blood flow are found in early stages of experimental bacterial meningitis (Pfister et al., 1990; Berkowitz et al., 1993), whereas a reduced cerebral blood flow may be observed in advanced phases of the disease (Smith et al., 1982; Tureen et al., 1990). Recent experimental studies show that nitric oxide (NO) is involved in the pathophysiologic alterations in bacterial meningitis (Habert et al., 1994; Boje, 1995; Buster et al., 1995; Koedel et al., 1995). Nitric oxide is produced from
MATERIALS AND METHODS
Rat model of pneumococcal meningitis
The protocols used in this study were approved by the government of Upper Bavaria. A well-characterized rat model of pneumococcal meningitis was used in this study, which was previously described in detail (Pfister et al., 1990). Briefly, 34 adult male Wistar rats (250 to 350 g) were anesthetized with thiopental (100 mg/kg given intraperitoneally), tracheotomized, and artificially ventilated with a small animal ventilator (model 683, Harvard Apparatus Co., South Natick, MA, U.S.A.). Supplemental anesthetic doses were administered to maintain anesthesia. A catheter was inserted into the left femoral artery for continuous monitoring of mean arterial blood pressure and for hourly analysis of Pa
Preparation of a closed cranial window
The pial microcirculation was visualized through a closed cranial window prepared as previously described (Morii et al., 1986; Koedel et al., 1995). A rectangular craniotomy (8 × 5 mm) was performed in the mediocaudal portion of the right parietal bone. Bone wax was placed around the bone gap, embedding the end of two polyethylene tubes subsequently used as an inlet and outlet. The dura was incised and stripped, exposing the transparent arachnoid and the underlying pial vessels on the brain surface. The cranial window was closed with a glass coverslip and sealed with dental acrylic (Paladur, Heraeus, Wehrheim, Germany). The tubes and the space under the window were filled with artificial CSF composed as follows: 150 mEq/L of Na+, 3 mEq/L of K+, 2.5 mEq/L of Ca2+, 1.3 mEq/L of Mg2+, 140 mEq/L of Cl−, 3.7 mEq/L of glucose, 6 mmol/L of urea, and 25 mEq/L of HCO3- (Morii et al., 1986). This fluid was equilibrated with 6.6% oxygen, 5.9% carbon dioxide, and the balance of nitrogen, which gave a pH ≈ 7.4 at 37°C. Pial arterioles ranging from 18 to 112 μm (median 47 μm) were observed with a trinocular microscope (Leitz GmbH, Munich, Germany) and recorded by using a low light level video camera (Panasonic WV 1550/G) connected with a video recorder (Panasonic AG 6200-EG) and a video monitor (Barco CD 233, Kortijk, Belgium). Pial arteriolar diameters were measured with a image analysis system for a personal computer (Stemmer Elektronik GmbH, Puchheim, Germany). We investigated the diameter of pial arterioles at different sites (three per animal) every 30 minutes for a period of 4 hours after intracisternal injection of pneumococci. Changes of diameter were expressed as percentage from a baseline of 100%. Carbon dioxide reactivity of pial arterioles was investigated at baseline conditions to eliminate disturbed vasoreactivity caused the surgical procedure. After hypercapnia, an increase in diameter of more than 1 % per 1 mm Hg increase in Pa
Intracisternal inoculum
Heat-killed (60°C for 4 hours) unencapsulated pneumococci, which stem from an isogenic mutant of the encapsulated strain Streptococcus pneumoniae type 3 (no. 17260), were used for intracisternal challenge (Pfister et al., 1990; Koedel et al., 1995). Intracisternal injection of heat-killed pneumococci induces meningeal inflammation with changes in regional cerebral blood flow, brain water content, ICP, and CSF-WBC counts similar to those seen after injection of live pneumococci, as previously reported (Koedel et al., 1995).
Induction of meningitis
When carbon dioxide reactivity of pial vessels was documented, 100 μL CSF was removed using the intracisternal catheter. The rats were challenged intracisternally with 100 μL of 108 colony forming units per milliliter of heat-killed pneumococci.
Experimental groups
Seven experimental groups were investigated:
Statistics
Data on vessel diameter, ΔICP, brain water content, CSF-WBC counts, and physiologic parameters for groups 1 and 2 were compared hourly after intracisternal injection of PBS/pneumococci using the unpaired Student's t-test. Data on vessel diameter, ΔICP, brain water content, CSF-WBC counts, and physiologic parameters from groups 2 through 7 were compared hourly after intracisternal injection of PBS/pneumococci using one-way analysis of variance and Scheffe's test. P values were corrected for repeated measurements using the Bonferroni Holm procedure. Data on increase in mean arterial blood pressure 30 minutes after administration of SMT (both doses) (groups 6 and 7) were compared with group 2 using one-way analysis of variance and Scheffe's test. Data on increase in mean arterial blood pressure 30 minutes after administration of 7-NI (group 3) were compared with group 2 using the unpaired Student's t test. Differences were considered significant at P < 0.05. Data are expressed as means ± standard deviation.
RESULTS
Physiologic variables
Data on Pa
Physiological parameters 0, 2, and 4 hours after intracisternal injection of heat-killed pneumococci (phosphate-buffered saline controls)
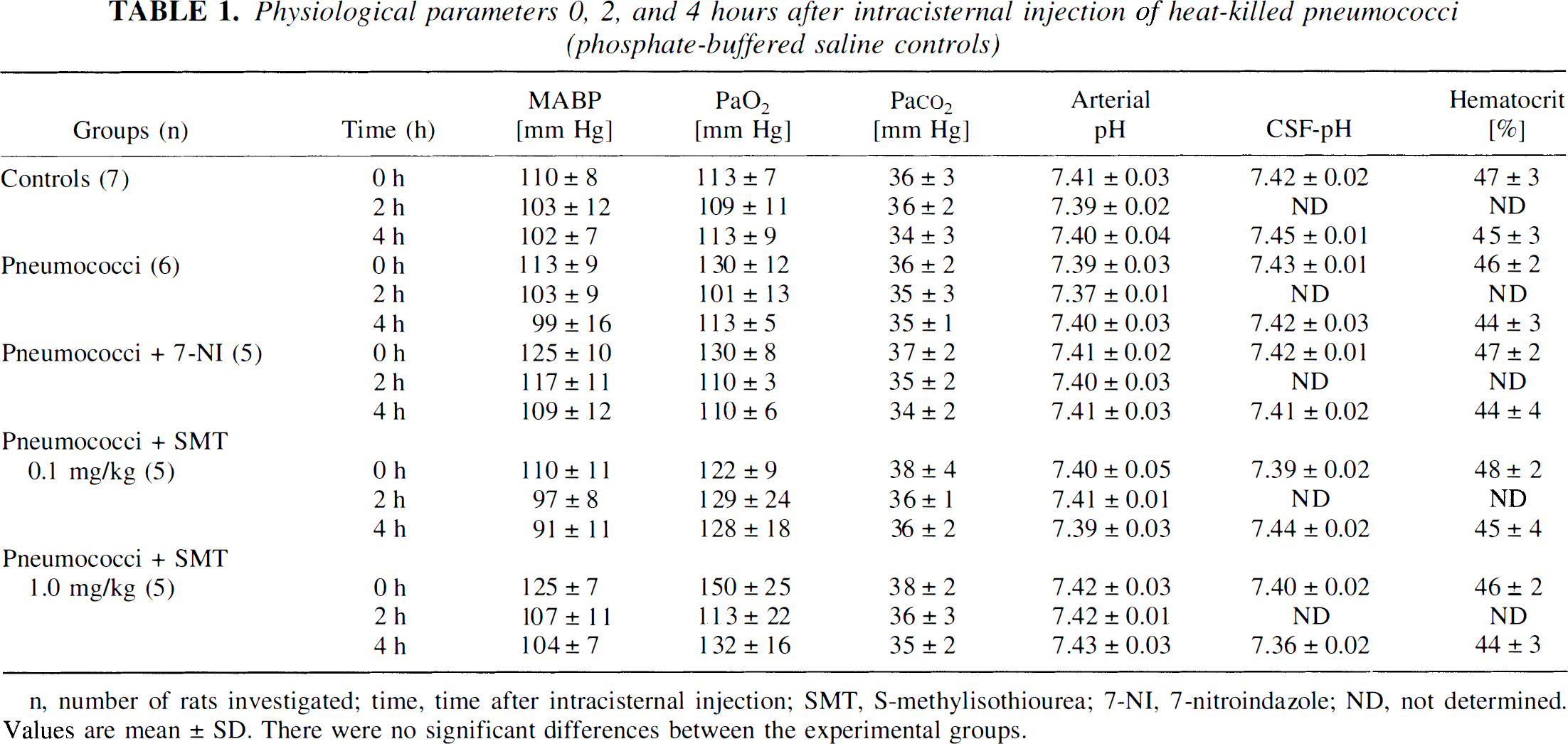
n, number of rats investigated; time, time after intracisternal injection; SMT, S-methylisothiourea; 7-NI, 7-nitroindazole; ND, not determined. Values are mean ± SD. There were no significant differences between the experimental groups.
Documentation of meningeal inflammation
There was a significant increase in CSF-WBC in rats challenged with pneumococci (group 2: 3124 ± 3437) compared with controls (group 1: 1 ± 2; P < 0.05). Treatment with 7-NI (group 3: 1280 ± 1015; ns) and SMT in both doses (group 6: 5973 ± 4327; group 7: 5081 ± 4390; ns) did not significantly alter CSF-WBC counts compared with untreated, pneumococci-injected rats (group 2). Treatment with peanut oil (vehicle) per se did not influence the CSF-WBC counts of rats intracisternally injected with PBS and pneumococci (group 4; 0 ± 0; group 5: 3776 ± 2448).
Effect of 7-nitroindazole and S-methylisothiourea on mean arterial blood pressure
7-Nitroindazole and SMT at 0.1 mg/kg had no significant effect on the change in mean arterial blood pressure (3 ± 9 mm Hg and 4 ± 5 mm Hg, respectively, compared with −1 ± 4 mm Hg in untreated rats) 30 minutes after administration. This suggests that there was no significant inhibition of eNOS activity. An increase in mean arterial blood pressure is regarded as an indicator of eNOS inhibition (Szabó et al., 1994). S-Methylisothiourea at 1.0 mg/kg caused a significant increase in mean arterial blood pressure by 8 ± 5 mm Hg (compared with rats treated with SMT at 0.1 mg/kg and untreated rats; P < 0.05) measured 30 minutes after administration, indicating an inhibitory effect on eNOS activity at this dose.
Effect of 7-nitroindazole and S-methylisothiourea on pneumococci-induced pial arteriolar vasodilation
Intracisternal injection of pneumococci caused a progressive dilation of pial arterioles that became statistically significant as early as 1 hour after pneumococcal challenge (114 ± 13%; P < 0.05, compared with controls) reaching 157 ± 22% 4 hours after intracisternal injection (compared with 104 ± 11% in controls; P < 0.05) (Fig. 1). Treatment with 7-NI significantly inhibited pneumococci-induced pial arteriolar vasodilation (107 ± 20% 4 hours after intracisternal challenge; P < 0.05, compared with untreated, pneumococci-injected rats). Intraperitoneal injection of 10 mL/kg peanut oil (vehicle) had no significant effect on PBS-injected rats (104 ± 4% compared with 104 ± 11% in controls without vehicle 4 hours after intracisternal challenge) and pneumococci-injected rats (160 ± 29% compared with 157 ± 22% in pneumococci-injected rats without vehicle 4 hours after intracisternal challenge). Treatment with SMT at 0.1 mg/kg did not significantly alter pneumococci-induced vasodilation (154 ± 38% at 4 hours after pneumococcal challenge; ns, compared with 157 ± 22% after 4 hours in pneumococci-injected, untreated rats; P < 0.05, compared with 107 ± 20% after 4 hours in pneumococci-injected rats, treated with 7-NI). Treatment with SMT at 1.0 mg/kg significantly attenuated pneumococci-induced vasodilation (125 ± 18% 4 hours after intracisternal injection; P < 0.05, compared with untreated, pneumococci-injected rats as well as to rats treated with SMT at 0.1 mg/kg).
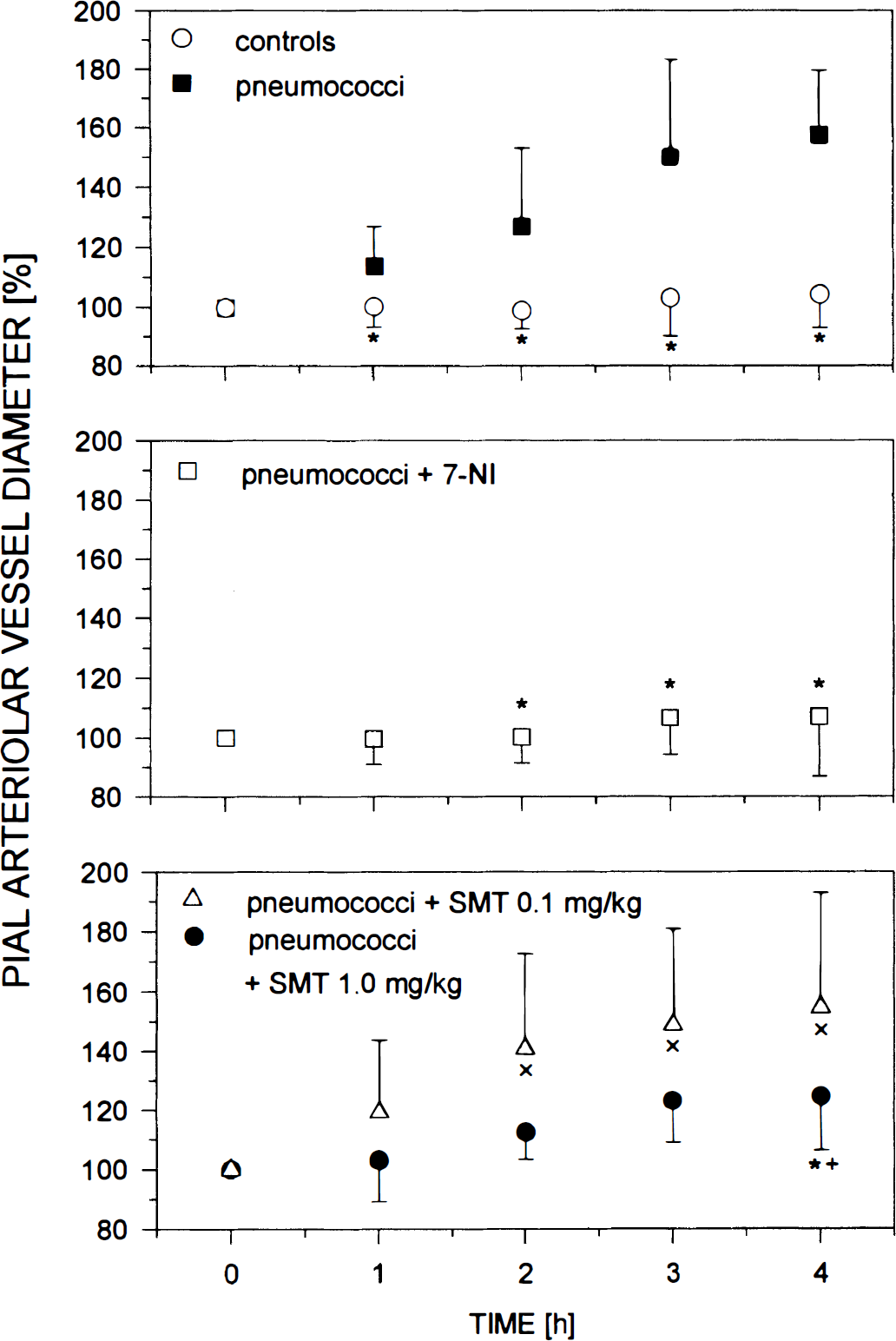
Time course of pial arteriolar vessel diameters expressed as percentage of a baseline of 100% within 4 hours after intracisternal injection of phosphate-buffered saline in control rats (controls), rats challenged with heat-killed pneumococci, and pneumococci-injected rats treated with 50 mg/kg of 7-nitroindazole (7-NI), 0.1 mg/kg of S-methylisothiourea (SMT), and 1.0 mg/kg of SMT. Injection with pneumococci caused a progressive dilation of pial arterioles. Treatment with 7-NI prevented the vasodilation. Treatment with SMT at 0.1 mg/kg had no inhibitory effect on pneumococci-induced vasodilation. Treatment with SMT at 1.0 mg/kg significantly attenuated pneumococci-induced vasodilation. Data were compared every hour. Data are mean ± standard deviation. *P < 0.05 compared with pneumococci-injected, untreated rats. ×P < 0.05 compared with pneumococci-injected rats treated with 7-NI. +P < 0.05 compared with pneumococci-injected rats treated with SMT at 0.1 mg/kg.
Effect of 7-nitroindazole and S-methylisothiourea on brain water content and intracranial pressure
Intracisternal injection of pneumococci caused a significant increase in ICP (ΔICP was 6.6 ± 2.5 mm Hg at 4 hours after pneumococcal challenge) (Fig. 2) and brain water content (79.47 ± 0.46%), compared with controls (ΔICP was −0.2 ± 0.4 mm Hg at 4 hours after injection of PBS, brain water content was 78.92 ± 0.27%; P < 0.05). Treatment with 7-NI and SMT (0.1 mg/kg and 1.0 mg/kg, respectively) had no significant inhibitory effect on ΔICP (8.0 ± 3.9 mm Hg, 8.8 ± 6.8 mm Hg, and 8.4 ± 1.9 mm Hg 4 hours after intracisternal injection, respectively) (Fig. 2) and brain water content (79.73 ± 0.35%, 79.44 ± 0.23%, and 79.88 ± 0.51%, respectively), compared with untreated, pneumococci-injected rats. Treatment with peanut oil (vehicle) per se did not influence ΔICP and brain water content of rats intracisternally injected with PBS and pneumococci (0.3 ± 0.6 mm Hg and 7.2 ± 3.8 mm Hg after 4 hours, 78.81 ± 0.32% and 79.65 ± 0.36%, respectively).
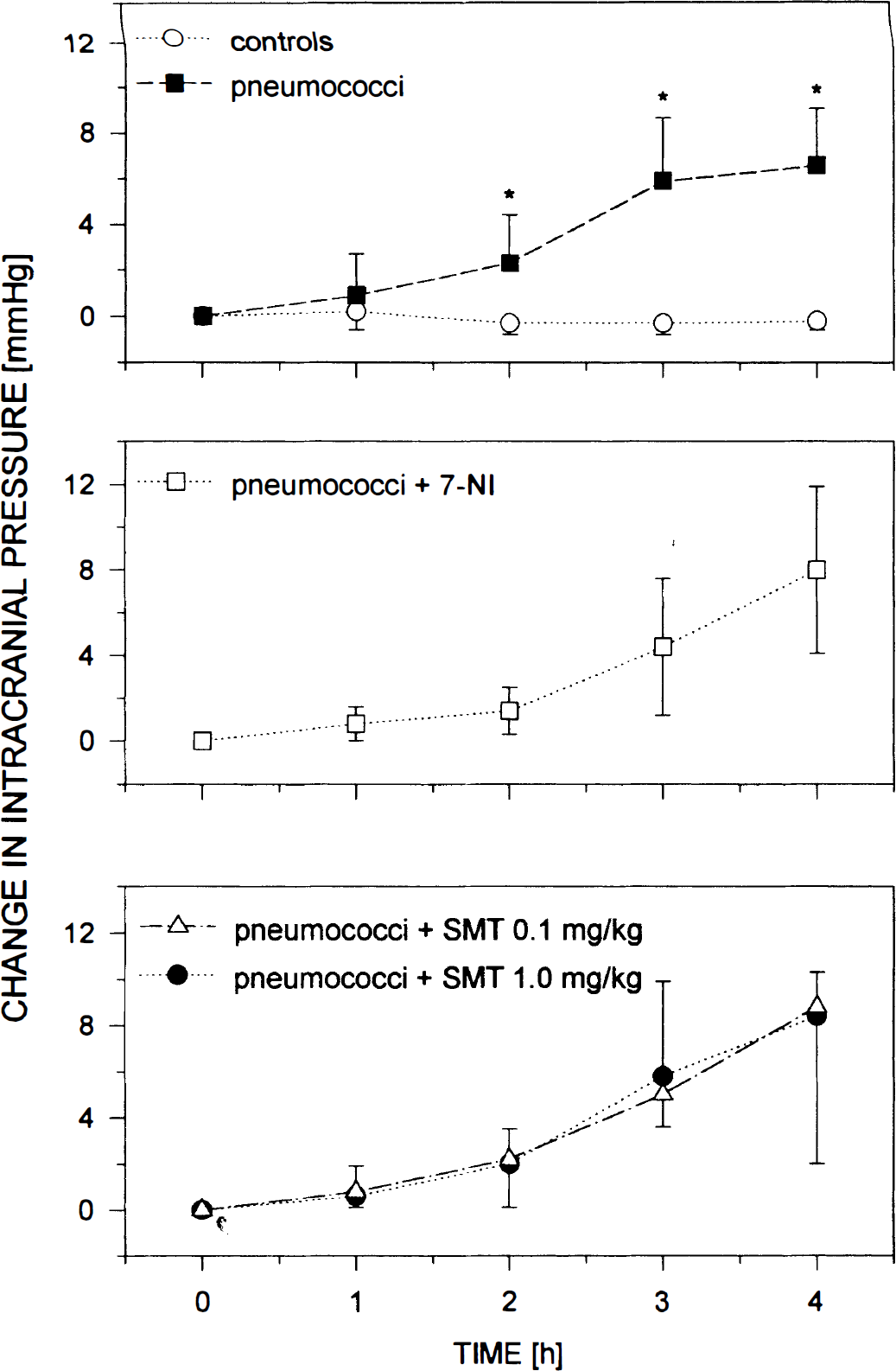
Time course of increase in intracranial pressure (ΔICP) within 4 hours after intracisternal injection of phosphate-buffered saline in control rats (controls), rats challenged with heat-killed pneumococci, and pneumococci-injected rats treated with 50 mg/kg of 7-nitroindazole (7-NI), 0.1 mg/kg of S-methylisothiourea (SMT), and 1.0 mg/kg of SMT. Injection with pneumococci caused a progressive increase in ICP. Treatment with SMT (at both doses) and 7-NI had no inhibitory effect on increase in ICP. Data were compared every hour. Data are means ± standard deviation. *P < 0.05 compared with controls.
DISCUSSION
There is increasing evidence that NO acts as a mediator of inflammation during bacterial meningitis, since the NOS inhibitors NG-nitro-
7-Nitroindazole is a potent and competitive inhibitor of rat brain NOS in vitro and in vivo (Babbedge et al., 1993; Moore et al., 1993; Bland-Ward and Moore, 1995). 7-Nitroindazole does not influence the endothelium-dependent vasodilation of acetylcholine (Yoshida et al., 1994), nor does it cause an increase in arterial blood pressure in vivo (Moore et al., 1993), indicating that eNOS function is preserved. In agreement with these observations, we did not find an increase in mean arterial blood pressure in the current study 30 minutes after administration of 7-NI. The nNOS is present in neurons (Bredt and Snyder, 1994) and glial cells (Kugler and Drenckhahn, 1996). Moreover, it seems that nerve fibers that innervate large cerebral arteries and pial vessels contain nNOS (Bredt et al., 1990). There are studies indicating that the nNOS is at least partly responsible for the control of cerebral blood flow under normal (Kelly et al., 1995; Iadecola et al., 1996) and pathophysiologic conditions, such as hypercapnia (Wang et al., 1995) and hemorrhagic hypotension (Kovach et al., 1994). In addition, inhibition of nNOS by 7-NI was shown to reduce the infarction size in a model of focal cerebral ischemia (Yoshida et al., 1994), which suggests that brain-derived NO plays an important role in neurotoxicity. The nNOS pathway can be activated by various substances, including glutamate (Wood and Garthwaite, 1994), vasoactive intestinal polypeptide (Kummer et al., 1992), and substance P (Kajekar et al., 1995). In a rabbit model of pneumococcal meningitis, elevated concentrations of glutamate were detected in the CSF 22 hours after infection (Guerra-Romero et al., 1993), indicating an increased release of glutamate during the course of the inflammation. Substance P, which is released by perivascular nerves originating from the trigeminal ganglion (Edvinsson, 1991), induces dilation of pial arterioles (Edvinsson et al., 1981). A previous study using the same rat model as in the current study showed that the substance P antagonist spantide was able to prevent pial arteriolar vasodilation in the early phase of experimental pneumococcal meningitis (Pfister et al., 1995).
To investigate the contribution of the iNOS pathway to the early inflammatory process of experimental meningitis, we tested the possible effect of SMT, a potent NOS inhibitor, which mainly inhibits iNOS (Szabó et al., 1994; Southan et al., 1995). Intraperitoneal administration of 0.1 mg/kg of SMT was previously shown to improve the survival rate in a mice model of septic shock (Szabó et al., 1994). In a rat sepsis model, SMT at a dose of 0.01 to 3 mg/kg restored the lipopolysaccharide-induced decrease in mean arterial pressure to preshock values (Kengatharan et al., 1995). In the current study, 0.1 mg/kg of SMT did not alter pneumococci-induced pial arterial vasodilation. The higher dose of SMT attenuated pneumococci-induced vasodilation; however, this dose caused an increase in mean arterial blood pressure, suggesting that SMT at 1.0 mg/kg additionally inhibits the constitutive NOS pathway. Both doses of SMT did not modulate changes in brain water content, ICP, and CSF pleocytosis after pneumococcal challenge. Therefore, our data suggest that NO produced by iNOS may not be involved in the early phase of experimental pneumococcal meningitis.
Further arguments for this are the observations that (1) mRNA of iNOS was detected in meninges of rat brains during systemic inflammation 6 hours, but not 2 hours after systemic lipopolysaccharide administration (Wong et al., 1996); (2) iNOS activity in homogenates of different tissues was found 3 hours, but not 1 hour after systemic administration of lipopolysaccharide in rats (Szabó et al., 1993); and (3) elevated CSF nitrite concentrations were found 4 hours, but not 2 hours after intracisternal challenge in a rat model of meningitis (Buster et al., 1995). All of these observations suggest that the iNOS pathway is not the major source of NO during the first hours of experimental meningitis.
We recently found that the unselective NOS inhibitors NG-nitro-
