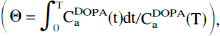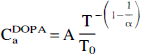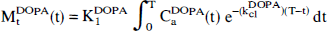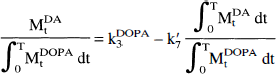Abstract
To test the hypothesis that dopamine (DA) receptors influence cerebral DOPA-decarboxylase (DDC) activity in vivo, we used HPLC to measure the kinetics of the cerebral uptake and metabolism of [3H]DOPA in carbidopa-treated rats, and in rats also treated acutely with a DA receptor antagonist (flupenthixol, 2 mg/kg, intraperitoneally) or a DA receptor agonist (apomorphine, 200 μg/g, subcutaneously). The unidirectional blood-brain clearance of [3H]DOPA (K1DOPA, 0.030 mL g−1 min−1) increased by 50% after flupenthixol. The magnitudes of the relative DDC activity (k3DOPA) in striatum (0.20 min−1), olfactory tubercle (0.11 min−1), and hypothalamus (0.15 min−1) of carbidopa-treated rats were doubled with flupenthixol, but cortical DDC activity was unaffected (0.02 min−1). Apomorphine reduced the magnitude of k3DOPA in striatum by 20%. The rate constant for catabolism of [3H]DA formed in brain (k7′, monoamine oxidase [MAO] activity), which ranged from 0.025 min−1 in striatum to 0.08 min−1 in hypothalamus of carbidopa-treated rats, globally increased 2- to 4-fold after flupenthixol, and decreased to 0.003 min−1 in striatum after apomorphine. These in vivo results confirm the claim that acute blockade of DA receptors with flupenthixol stimulates the synthesis of [3H]DA from [3H]DOPA, and that this [3H]DA is subject to accelerated catabolism. Conversely, activation of the DA receptors with apomorphine inhibits DDC activity and DA catabolism.
Keywords
It has been claimed recently that the activity of L-3,4-dihydroxypenylalanine (DOPA) decarboxylase (DDC) in living striatum determines the fraction of DOPA that is trapped as dopamine (DA) and not cleared from brain by facilitated diffusion or metabolism (Gjedde et al., 1993; Cumming et al., 1995a; Opacka-Juffry and Brooks, 1995). DOPA formed in brain is not committed solely to catecholamine synthesis (Cumming et al., submitted). By competing with other processes causing elimination of DOPA from brain, DDC activity may coregulate cerebral DA synthesis with tyrosine hydroxylase and may be rate-limiting for the synthesis of DA in patients with Parkinson's disease. It has been noted that DDC from rat striatum, its activity measured ex vivo, is activated after treatment with DA receptor antagonists such as flupenthixol and is partially inactivated after treatment with apomorphine, a DA receptor agonist (Zhu et al., 1992; Hadjiconstantinou et al., 1993; Zhu et al., 1994). These results suggest that an antagonist selective for DA autoreceptors, by stimulating DDC activity, might increase the rate of DA synthesis from exogenous DOPA and so potentiate the therapeutic action of DOPA in Parkinson's disease. Conversely, apomorphine would be expected to reduce the rate of DA synthesis from exogenous DOPA. However, the effects of drugs binding to dopamine autoreceptors on the uptake and metabolism of DOPA in brain of living rat are unknown.
We have formulated a compartmental model describing the uptake and cerebral metabolism of 6-[18F]fluoro-L-3,4-dihydroxyphenylalanine (FDOPA, Cumming et al., 1994) and [3H]DOPA (Cumming et al., 1995a) based on the temporal changes in the concentrations of labelled metabolites measured by HPLC fractionate of arterial plasma and brain extracts after bolus tracer injection. This model includes the fractional rate constant, also known as the relative rate constant, for the activity in vivo of DDC with respect to radiolabelled DOPA (k3DOPA, min−1) relative to the corresponding Michaelis constants (k3DOPA = Vmax/Km). The model also defines the fractional rate constant for the decomposition of labelled DA formed in brain (k7′ min−1), identified as the activity in vivo of monoamine oxidase (MAO) relative to the corresponding Michaelis constants of MAO for DA.
The reported effects of drugs such as flupenthixol and apomorphine on DDC activity measured ex vivo in striatal homogenates may be complicated by postmortem effects. Specifically, DDC activity measured in homogenized tissue may have been altered by disruption of the normal compartmentalization and regulation of the enzyme that prevailed in living brain. Therefore, we wished to confirm in vivo the regulation by flupenthixol and apomorphine of DDC activity reported on the basis of ex vivo assays under saturating conditions. The present model of [3H]DOPA metabolism in vivo also tests the effects of flupenthixol and apomorphine on the rate constants for the decomposition of labelled DA formed in living brain (k7′).
METHODS
The kinetic model for [3H]DOPA metabolism (Fig. 1) is discussed in detail elsewhere (Cumming et al., 1994; Cumming et al., 1995a,b). Briefly, in the periphery [3H]DOPA is metabolized by catechol-O-methyltransferase at the rate established by the rate constant k0DOPA, yielding O-methyl-[3H]DOPA (OMe-[3H]DOPA) which is cleared from circulation at the rate described by the rate constant k−1OMD (Cumming et al., 1993). [3H]DOPA is reversibly transferred across the blood-brain barrier by the facilitated diffusion of large neutral amino acids described by the blood-brain clearance (K1DOPA, mL g−1 min−1) and the rate constant of brain-blood clearance (k2DOPA, min−1). Within brain, [3H]DOPA is conceivably O-methylated by cerebral catechol-O-methyltransferase (k5DOPA, min−1), and is decarboxylated by DDC with a rate constant of k3DOPA (min−1) to yield [3H]DA. If not stored in vesicles (circle Fig. 1), this product is catabolized by MAO with a rate constant of k7′(min−1). [3H]DOPAC and its metabolite [3H]HVA are eliminated from brain at rates determined by the rate constants k9a and k9b respectively.
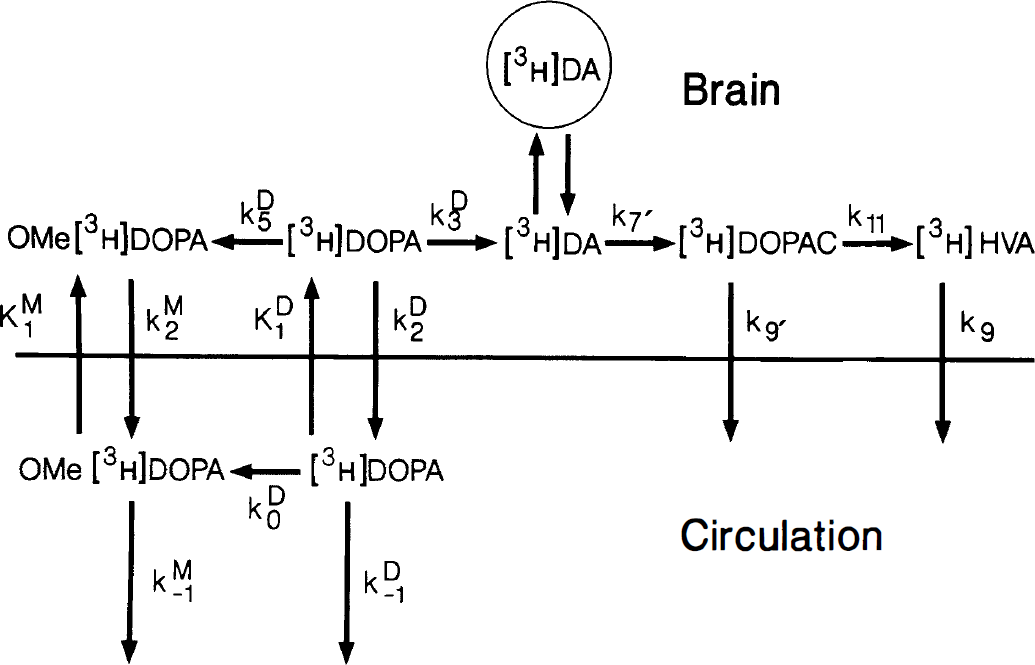
The kinetic model for the uptake and metabolism of [3H]DOPA in the carbidopa-treated rat. 0-Methyl-[3H]DOPA is generated from [3H]DOPA in circulation by catechol-O-methyltransferase activity in all tissues (k0DOPA)- Both tracers are cleared from circulation (k−1DOPA, k−1OMD), and transferred reversibly across the blood-brain barrier (K1DOPA, k2DOPA, K1OMD, k2OMD). [3H]DOPA within brain may be O-methylated (k5DOPA) or decarboxylated (k3DOPA). [3H]DA formed in a cytosolic compartment, if not stored in vesicles (circle), is deaminated by monoamine oxidase (k7′) to form [3H]DOPAC, which is cleared from brain (k9a), or O-methylated to form [3H]HVA (k11), which is eliminated from brain (k9b).
Male Wistar rats (225 to 275 g, Charles River, Québec, Canada) were anesthetized with halothane (2%, Ayerst, Canada) and received heparinized catheters in a femoral artery and vein as described previously (Cumming et al., 1994, 1995, 1997 a,b). Rats received carbidopa (5 mg/kg, intraperitoneally, Merck Sharp and Dohme Kirkland, Quebec, Canada) to block the peripheral decarboxylation of [3H]DOPA, and saline (intraperitoneally, n = 10), cis-flupenthixol (2 mg/kg, intraperitoneally, Research Biochemicals, Inc., [Natick, MA, U.S.A.] n = 12), or apomorphine (200 μg/kg, subcutaneously, Sigma [St. Louis, MO, U.S.A.], n = 14), before immobilization in a plaster body cast. After 45 minutes recovery from anesthesia, rats received 100 μCi [3H]DOPA (L-3,4-[ring-2,5,6-3H]dihydroxyphenylalanine, 80 Ci/mmol; New England Nuclear, Wilmington, DE, U.S.A.) as an intravenous bolus in 500 μL saline. Arterial blood samples were collected at intervals. The radiochemical composition of portions of plasma extracted with an equal volume of perchloric acid (0.5 mol/L) was determined by HPLC fractionation combined with scintillation spectroscopy as described elsewhere (Cumming et al., 1995 a,b). At circulation times ranging from 2.5 to 90 minutes, rats were killed by decapitation. Brains were rapidly dissected on ice into striatum, olfactory tubercle, cortex, hippocampus, and hypothalamus. Brain extracts, prepared by sonication in 10 volumes of 0.1 mol/L perchloric acid, were fractionated by HPLC.
The concentrations of [3H]DOPA and its peripheral metabolite O-methyl-[3H]DOPA in plasma, as functions of circulation time, were calculated from HPLC fractionation results. For experiments in which [3H]DOPA circulation lasted more than 10 minutes, the rate constants describing peripheral metabolism of [3H]DOPA were calculated as described previously (Cumming et al, 1993, 1994, 1995 a,b). For a population of rats, the normalized arterial integrals (θ, min−1, Gjedde, 1981, 1982, Patlak et al., 1983) for [3H]DOPA,
Using Matlab software for nonlinear regression (Mathworks, Natick, MA, U.S.A.) we fitted the model arterial curve and the measured brain [3H]DOPA concentrations (MtDOPA, DPM/mg) to a one-compartment model defined only by the unidirectional blood-brain clearance of [3H]DOPA (K1DOPA) and the rate constant for the net clearance of [3H]DOPA from brain (kc1DOPA, min−1), comprising the sum of the rate constants of clearance of [3H]DOPA from brain by transport from brain to blood (k2DOPA), decarboxylation in brain (k3DOPA) and O-methylation in situ (k5DOPA);
The kinetic constants so calculated were used to generate time-functions of the concentrations of [3H]DOPA in each brain region. The time-activity curves of the calculated concentrations of [3H]DOPA in brain, and the scaled concentrations of [3H]DA measured in brain (MtDA), were used together to calculate k3DOPA and k7′ by the linear regression described previously (Cumming et al., 1994),
RESULTS
In carbidopa-treated rats, the mean (± SEM) apparent rate constants for the O-methylation of [3H]DOPA in circulation was 0.014 ± 0.002 min−1, while the mean rate constant for the elimination of OMe-[3H]DOPA from circulation was 0.006 ± 0.003 min−1 (Table 1). The elimination rate constant increased to 0.022 ± 0.007 min−1 in flupenthixol-treated rats (P < 0.001). The relationship between tracer circulation time and the normalized arterial integrals in a population of carbidopa-treated rats is illustrated in Fig. 2A. The linear regression slopes of the ratios (α in Equation 1, ± 95% confidence limits) for [3H]DOPA were 2.1 ± 0.1 in carbidopa-treated rats, 2.3 ± 0.1 in flupenthixol-treated rats, and 2.2 ± 0.1 in apomorphine-treated rats. An arterial curve common to all rats was calculated from Equation 1 with the mean coefficient of 2.2. This common arterial curve was plotted along with the [3H]DOPA concentrations measured in an individual rat after scaling to this common input function (Fig. 2B).
Calculated fractional rate constants for the O-methylation of [3H]DOPA in circulation (k0DOPA, min−1) and for the elimination of OMe-[3H]DOPA from circulation (k−1OMD, min−1)

Each estimate is the mean ± SEM of estimates obtained in (n) animals in which tracer circulation lasted longer than 15 minutes. Rats were treated with carbidopa (5 mg/kg, intraperitoneally) and saline (n = 6), flupenthixol (2 mg/kg, intraperitoneally, n = 8) or apomorphine (200 μg/kg, subcutaneously, n = 7). Significance of difference from saline group:
P < 0.001
not significant.
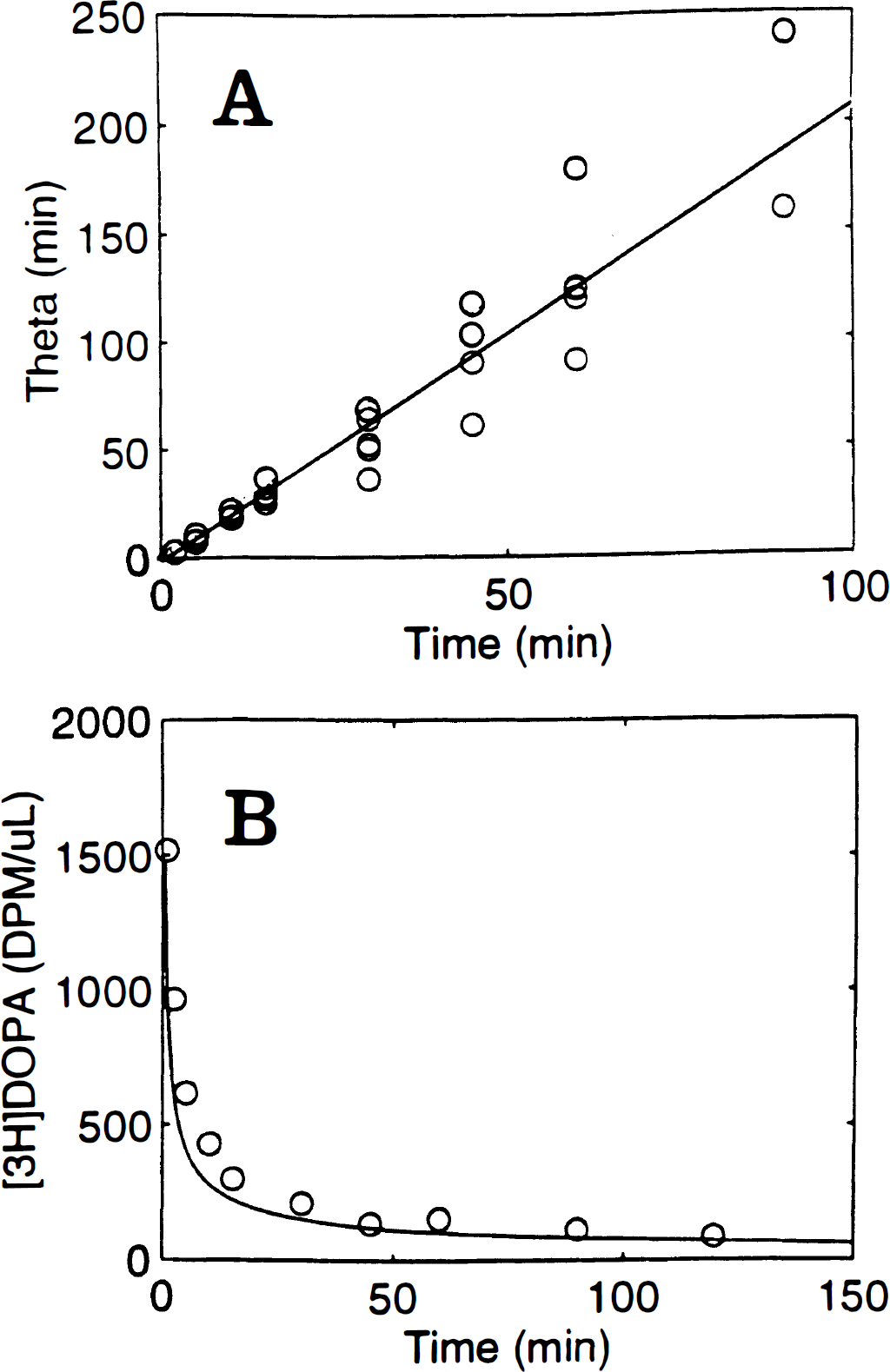
The correction of arterial input functions of [3H]DOPA measured in individual rats to a common input function for a population of rats.
The measured and calculated [3H]DOPA concentrations in cerebral cortex and in striatum of carbidopa-treated rats, and rats also receiving flupenthixol or apomorphine, are illustrated in Fig. 3. The mean (± SEM) unidirectional blood-brain clearances of [3H]DOPA (K1DOPA) were 0.030 ± 0.004 mL g−1 min−1 in five brain regions of carbidopa-treated control rats, 0.047 ± 0.009 mL g−1 min−1 in five regions of rats also receiving flupenthixol (P < 0.001 in each of five regions), and 0.027 mL g−1 min−1 in two regions of apomorphine-treated rats (Table 2).
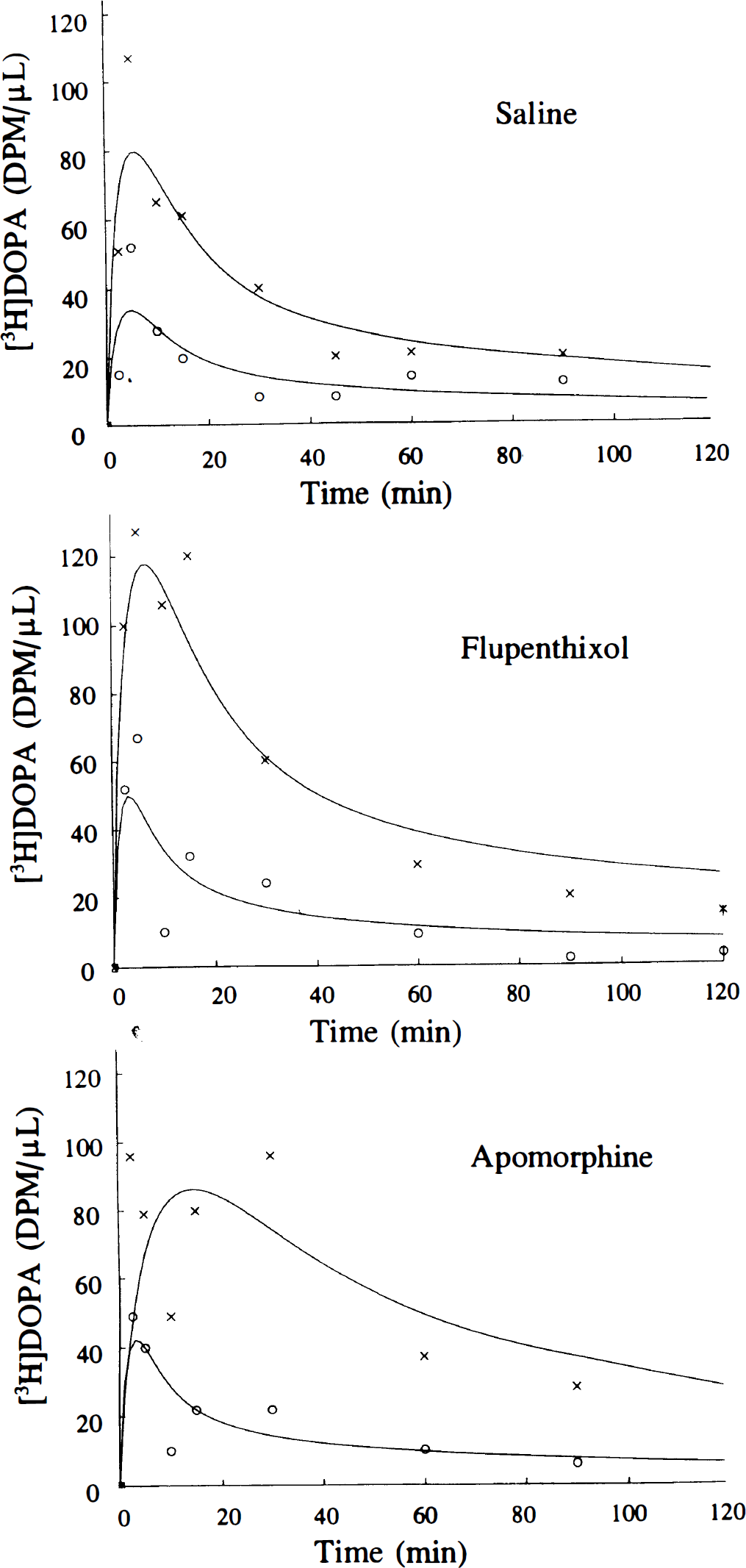
The concentrations of [3H]DOPA measured in cerebral cortex (×) and striatum (○) of carbidopa-treated rats (n = 10) and rats also treated with flupenthixol (2 mg/kg, intraperitoneally, n = 12) or apomorphine (200 μg/kg, subcutaneously, n = 14) as a function of time after [3H]DOPA injection (100 μCi, intravenously). Each point is the mean of two or three determinations, all scaled to a single common arterial curve for [3H]DOPA. The smooth curves are the nonlinear regressions of the fitted to the data for a one-compartment model described by the unidirectional blood brain clearance (K1DOPA, mL g−1 min−1) and the fractional rate constant for the net clearance of [3H]DOPA from brain (kclDOPA, min−1) using the common arterial input.
The unidirectional blood-brain clearances for [3H]DOPA (K1DOPA, mL g−1 min−1)

Clearances estimated in five brain regions of (n) rats treated with carbidopa (5 mg/kg, intraperitoneally) and saline (n = 10), flupenthixol (2 mg/kg, n = 12), or apomorphine (200 μg/kg, subcutaneously, n = 14) 45 minutes before tracer administration (100 μCi, intravenously). Kinetic parameters were calculated from the concentrations of [3H]DOPA in the common arterial curve and the scaled [3H]DOPA concentrations measured in brain as a function of circulation time. A two-compartment kinetic model defined by the unidirectional blood-brain clearance (K1DOPA) and the rate constant for the elimination of [3H]DOPA from brain (kclDOPA) was fitted to these data by nonlinear regression. Each estimate is reported ± 95% coincidence intervals. NA, not analyzed. Significance of difference from estimate in saline-treated group:
P < 0.001.
The time-integrals of the concentrations of [3H]DA and [3H]DOPA were used to calculate the magnitude of the rate constants k3DOPA and k7′ in striatum of rats treated with carbidopa and saline, flupenthixol, or apomorphine (Fig. 4). In the saline-treated group, the magnitude of k3DOPA was 0.02 min−1 in cortex, 0.15 min−1 in hypothalamus, 0.11 min−1 in olfactory tubercle and 0.20 min−1 in striatum (Table 3). Flupenthixol-treatment produced twofold increases in the magnitude of k3DOPA in striatum, olfactory tubercle and hypothalamus (P < 0.001), but was without effect in cortex. In apomorphine-treated rats, the magnitude of the estimate of k3DOPA decreased to 0.16 min−1 in striatum (P < 0.05), but increased to 0.05 min−1 in cortex (P < 0.001).
The relative DOPA decarboxylase activities in vivo (k3DOPA, min−1)
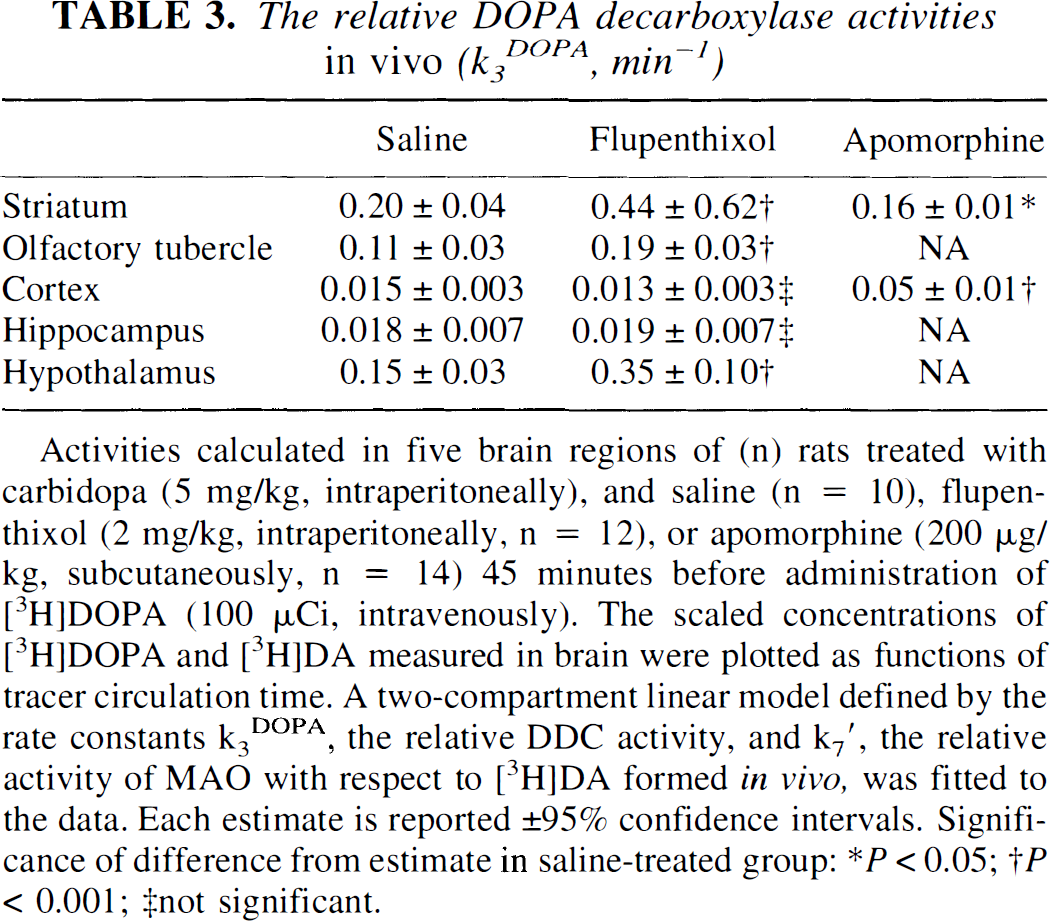
Activities calculated in five brain regions of (n) rats treated with carbidopa (5 mg/kg, intraperitoneally), and saline (n = 10), flupenthixol (2 mg/kg, intraperitoneally, n = 12), or apomorphine (200 μg/kg, subcutaneously, n = 14) 45 minutes before administration of [3H]DOPA (100 μCi, intravenously). The scaled concentrations of [3H]DOPA and [3H]DA measured in brain were plotted as functions of tracer circulation time. A two-compartment linear model defined by the rate constants k3DOPA, the relative DDC activity, and k7′, the relative activity of MAO with respect to [3H]DA formed in vivo, was fitted to the data. Each estimate is reported ± 95% confidence intervals. Significance of difference from estimate in saline-treated group:
P < 0.05
P < 0.001; ‡not significant.
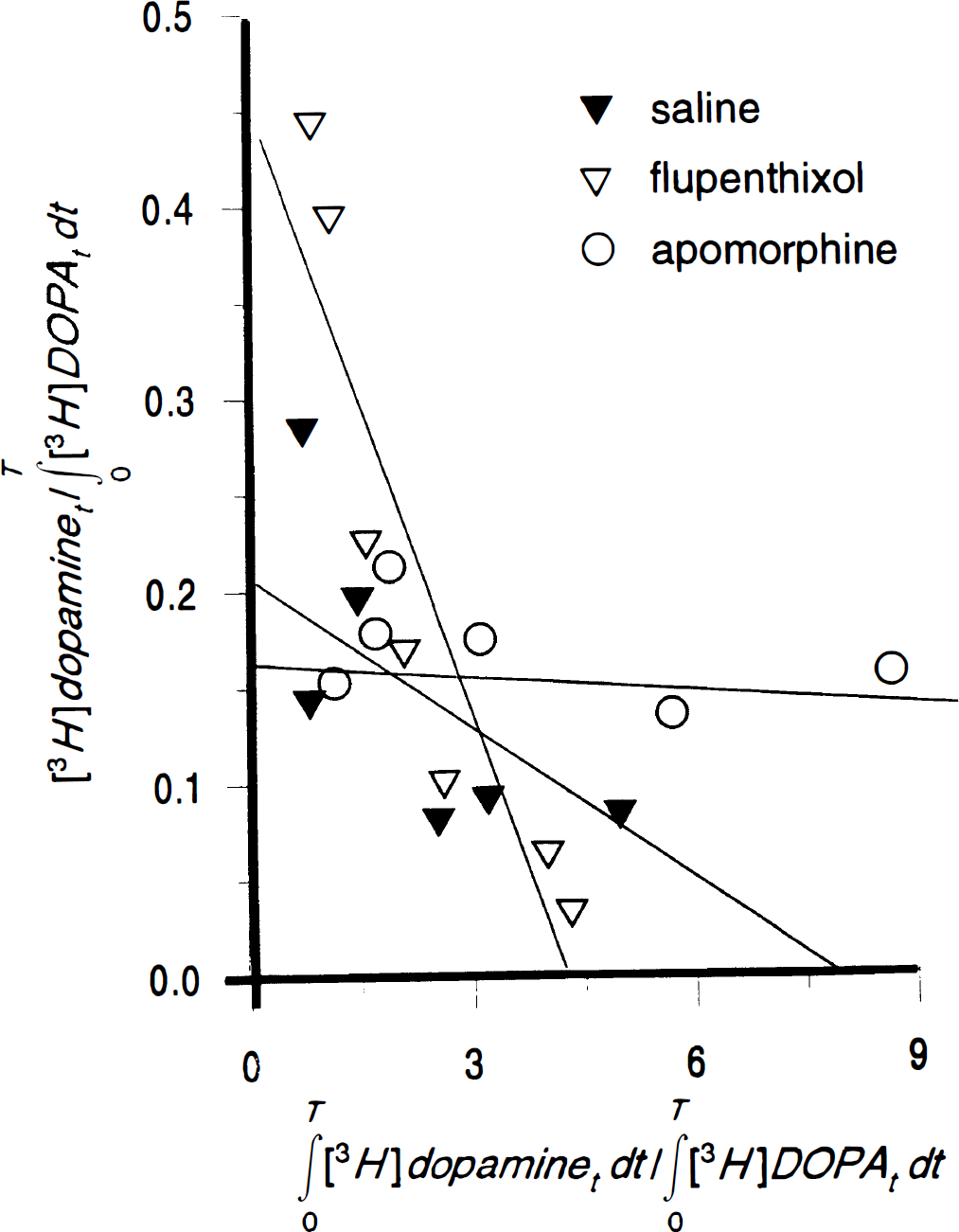
The integrated activities of [3H]DOPA and [3H]DA measured in rat striatum several times after bolus injection of [3H]DOPA (100 μCi, intravenously) yield a linear relationship of which the ordinate intercept corresponds to the relative activity of DOPA-decarboxylase (DDC) (k3DOPA), and the slope corresponds to the rate constant for the decomposition of [3H]DA in (k7′). Each point corresponds to the integration of the scaled tracer concentrations measured in 2 to 3 rats pretreated with carbidopa and saline (n = 10), flupenthixol (n = 12) or apomorphine (n = 14) 45 minutes before injection of [3H]DOPA.
The estimates of the rate constants of the catabolism of [3H]DA formed within brain from [3H]DOPA in carbidopa-treated rats (k7′) ranged from 0.025 min−1 in striatum to 0.08 min−1 in hypothalamus (Table 4). Flupenthixol-treatment increased the magnitude of the estimate of k7′ to 0.1 min−1 in striatum and olfactory tubercle, 0.2 min−1 in cortex, and 0.3 min−1 in hypothalamus (P < 0.001), but was without effect in hippocampus. Apomorphine decreased the estimate of k7′ to 0.003 min−1 in striatum (P < 0.001), but was without significant effect in cortex.
The rate constants for the decomposition of [3H]DA (k7′, min−1)

Rate constants estimated in five brain regions of (n) rats after administration of [3H]DOPA (100 μCi, intravenously). Rats were treated with carbidopa (5 mg/kg, intraperitoneally) and saline (n = 10), flupenthixol (2 mg/kg, intraperitoneally, n = 12), or apomorphine (200 μg/kg, subcutaneously, n = 14) 45 minutes before [3H]DOPA. The concentrations of [3H]DOPA and [3H]DA in brain were plotted as a function of tracer circulation time. A two-compartment linear model defined by the rate constants k3D, the relative DDC activity, and k7′, the relative activity of MAO with respect to [3H]DA formed in vivo, was fitted to the data. Each estimate is reported ± 95% confidence intervals. Significance of difference from estimates in saline-treated group:
P < 0.05
P < 0.001
not significant.
DISCUSSION
The estimates of the magnitude of the fractional rate constants of formation and elimination of OMe-[3H]DOPA in the circulation in carbidopa-treated rats (Table 1) were nearly identical to earlier estimates (Cumming et al., 1995 a,b). Flupenthixol treatment substantially increased the magnitude of k−1OMD, an effect possibly indicative of increased renal clearance of OMe-[3H]DOPA in circulation. Age-related changes in this rate constant have been observed in humans (Cumming et al., 1993), possibly also related to renal clearance. It is the activation of dopamine D1-like sites that increases urine flow (Eisner et al., 1994); the mechanism by which flupenthixol may increase renal clearance of a large neutral amino acid is unclear. The present drug treatments were without effect on the estimates of k0DOPA, the rate constant describing systemic catechol-O-methyltransferase activity.
The relationship between the tracer circulation time and the normalized arterial integral for [3H]DOPA is highly linear within a population of rats (Fig. 2A). The slope of this relationship (α) for [3H]DOPA is identical to earlier estimate for [3H]tyrosine (Cumming et al., submitted), suggesting that distribution of the two labelled amino acids in circulation is very similar. Linear scaling of [3H]DOPA concentrations measured in an individual rat during 120 minutes of tracer circulation results in very close agreement with the normalized arterial curve (Fig. 2B). Thus, the modelling of the arterial [3H]DOPA curves from a population of rats with a common power function (Equation 1) provides a satisfactory means of pooling data for compartmental analysis of dynamic time-activity curves.
The magnitudes of the unidirectional blood-brain clearances of [3H]DOPA (K1DOPA) in carbidopa-treated rats were in the range of earlier estimates (Cumming et al., 1995 a,b). Apomorphine was without consistent effect on unidirectional blood-brain clearance, but flupenthixol globally increased the estimate of K1DOPA by 50% (Table 2). Unidirectional clearance of [3H]DOPA is potentially sensitive to plasma concentrations of amino acids and cerebral blood flow, both of which could, in principle, be drug sensitive. However, the magnitudes of K1DOPA in the flupenthixol group were still within the range of control values obtained previously (Cumming et al., 1995 a,b).
Acutely, neuroleptics stimulate catecholamine synthesis in rat striatum by activation of tyrosine hydroxylase (Zivkovic et al., 1974, Lazar et al., 1982, Cumming et al., submitted). Chronically, neuroleptics increase the expression of mRNA for DDC in nigrostriatal neurons (Buckland et al., 1992). In the present study, acute treatment with flupenthixol substantially activated DDC in striatum, olfactory tubercle, and hypothalamus (Table 4), a finding consistent with the results of DDC assays ex vivo after acute treatment with flupenthixol and other antagonists of DA (D1 and D2) receptors (Zhu et al., 1992; Hadjiconstantinou et al., 1993, Zhu et al., 1994). However, the binding sites in rat striatum that regulate DA synthesis are now understood to be, in part, D3 receptors (Meller et al., 1993). In the present study, the receptors and pathways mediating DDC activation in vivo cannot be identified with certainty because flupenthixol is a nearly equipotent antagonist of D1 (Gingrich et al., 1992), D2, and D3 receptors (Freedman et al., 1994) in vitro.
The dose of apomorphine administered in the present study is intermediate between the autoreceptor dose, and the higher psychomotor-stimulant dose activating postsynaptic D1 and D2 receptors (Widzowski and Cory-Slechta, 1993). Partial inactivation of DDC by apomorphine in synaptosomes from rat striatum could not be overcome by flupenthixol (Zhu et al., 1994), indicating a complex pharmacology for the regulation of DDC in vivo. In the present study, the relative activity of DDC in intact striatum was reduced by 20% by acute apomorphine. Nonetheless, continuous infusion of apomorphine has been reported to reduce substantially the required dose of DOPA in a group of patients with Parkinson's disease (Gancher et al., 1995). The content of the DA metabolites in cerebrospinal fluid of non-parkinsonian subjects was substantially reduced for several hours after acute apomorphine administration (Przedborski et al., 1995). In the present study, apomorphine nearly arrested the catabolism of [3H]DA formed in rat striatum, consistent with the substantial decreases in DOPAC and HVA content of rat striatum observed 60 minutes after apomorphine (Westerink and Spaan, 1982). Presumably the clinical antiparkinsonian postsynaptic effects of apomorphine can predominate over effects on DDC activity and DA metabolism mediated by presynaptic autoreceptors.
In an earlier study, we observed that acute inhibition of MAO with pargyline resulted in 50% reductions of the magnitude of k3DOPA in striatum and olfactory tubercle, but no change in hypothalamus or cerebral cortex (Cumming et al., 1995a). To explain this phenomenon, we proposed that pargyline treatment, by blocking DA catabolism, increased the occupation of autoreceptors on nigrostriatal terminals by DA, causing in turn a partial inactivation of DDC. Conceivably the balance of factors regulating DDC activity in living hypothalamus favors the inactivated state of DDC such that acute pargyline was without effect on k3DOPA. However, decarboxylation of [3H]DOPA in vivo may substantially occur within serotonin terminals in some brain regions (Cumming et al., 1995a, Cumming et al., 1997). Furthermore, haloperidol-treatment failed to activate tyrosine hydroxylase activity in hypothalamus (Cumming et al., 1997a). Therefore, it is unclear why flupenthixol should activate DDC in hypothalamus in the present study. Flupenthixol was without effect on k3DOPA in cortex, whereas apomorphine decreased the magnitude of k3DOPA in that tissue. However, estimates of kinetic rate constants in cortex and hippocampus are based on measured [3H]DA concentrations that are generally so low as to be unreliable.
The relative activities of MAO with respect to [3H]DA formed in situ (k7′ Table 4) are close to the magnitudes of the rate constants reported in an earlier study (Cumming et al., 1995a). As before, the highest estimate of this rate constant was in hypothalamus, although the present result is 50% lower than the earlier estimate, possibly reflecting more rapid processing of postmortem specimens in the present study. Rate constants of intermediate magnitude for MAO activity, relative to [3H]DA, were again found in cortex, although with large relative errors caused by the low magnitude of k3D in that tissue. In the present study, we again find evidence for a dorsal-ventral gradient in the magnitude of k7′ within the basal ganglia consistent with the different DOPAC turnover rates observed in rat dorsal striatum and olfactory tubercle (Westerink and Kikkert, 1986). In the present study, acute treatment with flupenthixol greatly accelerated the rate constant of catabolism of [3H]DA formed from [3H]DOPA within brain, consistent with the effect of acute neuroleptics on DOPAC and HVA measured in extracts of rat striatum (Westerink and Spaan, 1982).
The schema in Fig. 1 suggests that DA catabolism in vivo proceeds in proportion to the distribution of DA between cytosolic and vesicular compartments, because only cytosolic DA is accessible to catabolic enzymes. Were DA synthesis acutely stimulated, the vesicular storage sites may plausibly become saturated, in which event newly synthesized DA would remain in the cytosol where it would be rapidly decomposed by MAO. Consistent with this model, acute treatment with haloperidol does not alter the total DA content of intact rat striatum, although DA metabolite concentrations are greatly increased (Westerink and Spaan, 1982). Thus, the present model suggests that stimulation of the pathway for DA synthesis is futile with respect to steady-state DA levels if the vesicular DA compartment is nearly saturated. The present finding that flupenthixol stimulates the formation of DA from exogenous L-DOPA in rat brain suggests that a selective autoreceptor antagonists might potentiate the antiparkinsonian action of DOPA. However, it is difficult to predict the clinical consequences of stimulated DA synthesis in brain of patients with Parkinson's disease, in whom the vesicular storage of DA may also be compromised.
In conclusion, the present in vivo tracer kinetic study provides further evidence for a regulation of DDC activity in brain of living rat. Chronic DOPA administration decreases the synthesis of DA and the activity of DDC in rat striatum measured ex vivo (Melamed et al., 1983; Takashima et al., 1987; Murata et al., 1993). In a recent PET study in anesthetized nonhuman primates, acute DOPA treatment increased the magnitude of k3DOPA in striatum by about 20% (Tedroff et al., 1997). We have elsewhere argued that the presence of competing pathways suggests that DOPA entering brain (Gjedde et al., 1993) or formed within brain (Cumming et al., in press) is not wholly committed to catecholamine synthesis. The activity of DDC in vivo determines the fraction of DOPA within brain that is transformed into DA. From earlier estimates of the rate constant for elimination of [3H]DOPA from rat brain (Cumming et al., 1995a), we can calculate that acute flupenthixol increased this fraction from 60% to 80%, thus contributing to the regulation of DA synthesis. Together with earlier findings, the present results suggest that DDC activity and expression may be regulated in proportion to the occupancy of DA autoreceptors, and that DDC activity in striatum of living rat is poised at an intermediate state of activation. The precise pharmacology of this regulation is at present uncertain. Studies with centrally acting drugs more selective for DA autoreceptors than flupenthixol or apomorphine may be informative about the presynaptic mechanisms regulating DDC activity in vivo. A clear understanding of these mechanisms could result in improved pharmacotherapy for Parkinson's disease, especially if DDC activity were stimulated with DA antagonists acting only presynaptically.