Abstract
In the cerebral circulation, endothelin-A receptor activation mediates marked prolonged vasoconstriction whereas endothelin-B (ETB) receptor activation effects dilation. In contrast to some peripheral vascular beds, ETB receptor–induced vasoconstriction has not yet been demonstrated in brain vessels. In this study in chloralose-anesthetized cats, with perivascular microapplications of ETB selective agonist (BQ-3020) and antagonist (BQ-788), we investigated whether ETB receptor–mediated constriction could be uncovered in cortical arterioles in vivo. In addition, we examined whether normal dilator response to ETB receptor activation is preserved in postishemic cerebral arterioles. The first microapplication of the selective ETB receptor agonist BQ-3020 (1 μmol/L) onto a pial cortical arteriole elicited marked dilation (caliber increased by 26.3 ± 15.1% from preinjection baseline). A second application of BQ-3020 (10-minute interval) onto the same vessel failed to evoke any significant vasomotor response. Subsequent (third and fourth) adventitial microapplication of the ETB receptor agonist on the same arteriolar site effected a significant constriction of cerebral arterioles (−15.3 ± 12.7% and −9.7 ± 6.3% from preinjection baseline, respectively, at 20 and 30 minutes after the first application). The pial arterioles did not display tachyphylaxis to repeated applications of potassium (10 mmol/L). The perivascular application of the ETB receptor antagonist BQ-788 (0.001 to 1 μmol/L) had no effect on arteriolar caliber per se but blocked both BQ-3020–induced dilation (inhibitory concentration ∼ 5 nmol/L) and vasoconstriction elicited by repeated activation of ETB receptors. After middle cerebral artery occlusion, most of the arterioles examined displayed a sustained dilation. The microapplication of BQ-3020 into the perivascular space surrounding postischemic dilated arterioles elicited a constriction of a similar magnitude to that induced by application of CSF (−17 ± 7% and −17 ± 7% from preinjection baseline, respectively). The adventitial microapplication of the ETB receptor antagonist (BQ-788, 0.1 μmol/L) on postocclusion dilated pial arterioles effected no change in the arteriolar caliber when compared with preinjection baseline. This BQ-788–induced response was significantly different from that induced by perivascular microinjection of CSF (P < 0.001, analysis of variance). These investigations indicate that (1) repeated activation of ETB receptors displays tachyphylaxis of the vasodilator response but also uncovers significant constriction of cerebral arterioles in vivo; (2) the ability of BQ-3020 to elicit dilation is lost within 30 minutes of induced focal ischemia; and (3) ETB-mediated contractile tone contributes in a small but significant manner in limiting postischemia dilation of cortical pial arterioles.
Keywords
Endothelin-1 (ET-1) is a 21–amino acid peptide produced by vascular endothelial cells and has a high degree of structural homology with the other members of the endothelin family, ET-2 and ET-3 (Yanagisawa et al., 1988; Inoue et al., 1989). The combination of functional and molecular studies of endothelin receptors has demonstrated the existence of at least two distinct subtypes, namely endothelin-A (ETA) and endothelin-B (ETB). The ETA receptor is characterized by a higher affinity toward ET-1 than ET-2 and ET-3, whereas the ETB receptor displays a similar affinity toward the three endothelins (Sakurai et al., 1990; Haynes et al., 1993; Masaki et al., 1994). ETA receptors are mainly located in vascular smooth muscle cells, with ETB receptors being present on neuronal, glial, endothelial, and smooth muscle cells. The proportion of each class of endothelin receptor depends on species, vascular bed, and vessel type, and they may mediate contraction or relaxation (Rubanyi and Polokoff, 1994). In most vascular beds (including cerebrovascular), endothelin-induced vasoconstriction normally is attributed to activation of ETA receptors (based on the relative potency of endothelin and the ability of selective ETA receptor antagonists to attenuate the responses) (Robinson and McCulloch, 1990; Adner et al., 1993; Feger et al., 1994, Patel et al., 1996c). In contrast, in some peripheral vascular tissue, ET-1–mediated vasoconstriction is resistant to the actions of ETA receptor antagonists and therefore cannot be entirely attributed to ETA receptor activation (Summer et al., 1992; LaDouceur et al., 1993; Davenport and Maguire, 1994). The function of ETB receptors is more controversial and less well elucidated. It is widely accepted that stimulation of ETB receptors induces vasodilation through generation of the endothelium-derived relaxing factor (nitric oxide and/or prostayclin) (DeNucci et al., 1988; Karaki et al., 1993; Kitazono et al., 1995a). However, ETB receptor–mediated vasoconstriction has been described in some vascular beds but not yet in vessels of the brain (Clozel et al., 1992; Seo et al., 1994; Haynes et al., 1995). The qualitatively different effects of the endothelins at the ETB receptor have led to subdivision of ETB receptors into ETB1 receptors (on endothelial cells) that mediate vasodilation and ETB2 receptors (on smooth muscle cells) that mediate vasoconstriction (Warner et al., 1993; Masaki et al., 1994). The function of the different endothelin receptor subtypes in vessels is unclear. The ETA receptor has been proposed to mediate constriction on the high-pressure side of the systemic circulation, whereas contractile ETB receptors are more abundant in low-pressure systems such as the pulmonary and venous circulation (Moreland et al., 1994). In a defined vessel, the relative amount of ETA and contractile ETB receptors varies according to the physiologic or pathologic state of the vessel (Kitazono et al., 1995b; Adner et al., 1996; Dagassan et al., 1996).
The recent development of selective peptidic and non-peptidic antagonists and agonists for ETB receptors provides useful tools for the investigation of the physiologic and pathophysiologic role of ETB receptors (Ihara et al., 1992; Takai et al., 1992; Ishikawa et al., 1994; Breu et al., 1996). BQ-3020 (N-acetyl-Leu Met Asp Lys Glu Ala Val Tyr Phe Ala His Leu Asp Ile Ile Trp), is a linear ET-1 analogue that has demonstrated a ∼4700-fold selectivity for the ETB receptor subtype compared with the endothelin ETA receptor in vivo and in vitro (Ihara et al., 1992). BQ-788 (N-cis-2,6-dimethylpiperidinocarbonyl-
To establish a basis for selection of optimal pharmacologic profile (ETA selective or mixed ETA/ETB) of endothelin antagonists that could be used in cerebrovascular disease, the current investigation was designed to examine (1) whether ETB receptor–mediated constriction could be uncovered in cerebral circulation, and (2) whether the vasomotor effects of the ETB agonist and the ETB antagonist on resistance cerebral arterioles were modified by acute focal cerebral ischemia.
MATERIALS AND METHODS
Anesthesia and general preparations
The experiments were carried out on 11 (5 ischemic and 6 nonischemic) adult female cats weighing between 3 and 4 kg. The anesthesia regimen and the surgical procedure are fully described elsewhere (Patel et al., 1996b). Briefly, anesthesia was induced with alphaxalone/alphadolone (Saffan, Glaxo Harefield, U.K.) administered into the radial vein. The animals were intubated, and positive-pressure ventilation with nitrous oxide/oxygen (70%/30%) initiated. The femoral artery and vein were cannulated for monitoring arterial blood pressure and arterial blood gas status and also for administering anesthetic agent and fluids. Anesthesia was maintained using α-chloralose (60 mg/kg) and supplemented as required to prevent the return of the corneal reflex during the course of the experiment. The inspired gas mixture was altered to oxygen supplemented air (25% oxygen and arterial blood samples were taken at regular intervals to monitor blood gas status. The animals were maintained normocapnic by adjusting the stroke volume. Metabolic acidosis was controlled by administration of sodium bicarbonate (8.4% solution) when necessary. Core temperature was measured by a rectal thermometer and maintained at 37°C by means of a thermostatically controlled heating blanket. At the outset of the study, mean arterial pressure was 100 ± 18 mm Hg, arterial carbon dioxide tension was 29.1 ± 4.0 mm Hg, arterial pH was 7.44 ± 0.05, and arterial oxygen tension was 149 ± 39 mm Hg. These physiologic parameters did not vary significantly during the course of the microapplication study.
The animals were placed in a stereotactic frame and after midline incision, the scalp retracted and sutured onto a metal ring to form a well over the calvaria. The temporalis muscle was retracted and a rectangular craniectomy (∼2.5 × 1.5 cm) was made over the parietal cortex using a saline-cooled dental drill. The exposed dura was bathed continuously with mineral oil at 38°C. With the aid of a stereomicroscope (Bausch and Lomb), the dura was excised and then reflected laterally. The preparation was allowed to equilibrate for approximately 30 minutes before any adventitial microapplications were performed.
Measurement of cerebral resistance arteriolar caliber
Cerebral resistance arteriolar caliber was measured by the method originally developed by Beaz (1966) using an image splitter linked through a closed-circuit video display system. The individual cerebral resistance arterioles were viewed through a stereomicroscope and arteriolar diameter measured from the degree of shear applied to the image splitter to tangentially oppose the two images. The system was calibrated at ×40 and ×70 against threads of known diameter, which allows the direct measurement of the vascular diameter in absolute units (micrometers). Vessel diameter was measured before and after injection of substances and monitored over a period of 1 to 2 minutes. In this study, the size of the arterioles examined ranged between 40 and 290 μm.
Administration of drugs
Artificial CSF was prepared with the following composition: Na+ 156 mmol/L, K+ 3 mmol/L, Ca2+ 2.5 mmol/L, HCO3− 12 mmol/L, and Cl− 152 mmol/L. The pH of the CSF was adjusted to 7.2 by aeration with 95% oxygen–5% carbon dioxide. The agents to be administered by adventitial microapplication were dissolved in CSF. All solutions were prepared freshly on the day of the study. Glass micropipettes (tip diameter 10 to 12 μm) were filled under vacuum with the solution to be studied and stored under mineral oil in CSF until required. Adventitial microapplication of substances was carried out using a micromanipulator to position the pipettes in the adventitial space around individual cerebral resistance arterioles. Small volumes (approximately 5 μL) were delivered by a pressure ejection system. Repeated application of the agents was performed in the same arteriolar site.
Focal cerebral ischemia studies
The middle cerebral artery (MCA) was exposed using a transorbital approach as previously described by Chen and colleagues (1991). Briefly, the contents of the left orbit were exenterated, and the optic foramen was enlarged to expose the dura and the MCA underlying it. The dura was excised, the MCA and its branches were occluded by means of bipolar diathermy, and the artery was sectioned. In each cat, perivascular microinjections of CSF (pH 7.2), BQ-3020 (1 μmol/L), and BQ-788 (0.1 μmol/L) were made at different sites after 30 to 210 minutes of cerebral ischemia. The arterioles studied were located in the ectosylvian and suprasylvian gyri (i.e., the regions supplied by the MCA and representing ischemic penumbra).
Statistical analysis
All results are expressed as means ± SD of the percentage change in arteriolar caliber from baseline immediately before microapplication of substances. The data were analyzed using a one-way analysis of variance followed by two-tailed Student's unpaired t test using a Bonferroni correction factor for multiple group comparisons.
RESULTS
Adventitial microapplication of BQ-3020
The application of CSF minimally altered pial arteriolar caliber (arteriolar caliber changed by 0.7 ± 4.8% from baseline). The first microapplication of BQ-3020 (1 μmol/L) mediated a marked and transient dilation of pial arterioles (caliber increased by 26.3 ± 15.1%). A second injection of the ETB receptor antagonist (10 minutes later) at the same site failed to induce any arteriolar dilation. Subsequent (third and fourth) adventitial microapplications of BQ-3020 effected a significant (P < 0.001, analysis of variance) constriction of cerebral arterioles (Fig. 1). Potassium-induced marked dilation, subsequent to repeated injection of BQ-3020, indicated that the pial arterioles were still responsive to vasodilator agents (Fig. 1). In separate experiments, repeated adventitial microapplication of potassium (10 mmol/L) at the same site did not effect any tachyphylaxis of the dilator response (Fig. 2). Both CSF- and potassium-induced vascular effects were independent of the preinjection diameter (caliber range, 45 to 239 μm). However, the dilation induced by the first microapplication of BQ-3020 was significantly dependent on the preinjection caliber. Pial arterioles with diameter less than 100 μm demonstrated a greater vasodilator response to ETB receptor agonist. The vasoconstriction, observed after repeated activation of ETB receptors, also displayed some size dependency, with arterioles less than 100 μm displaying more pronounced vasoconstriction.
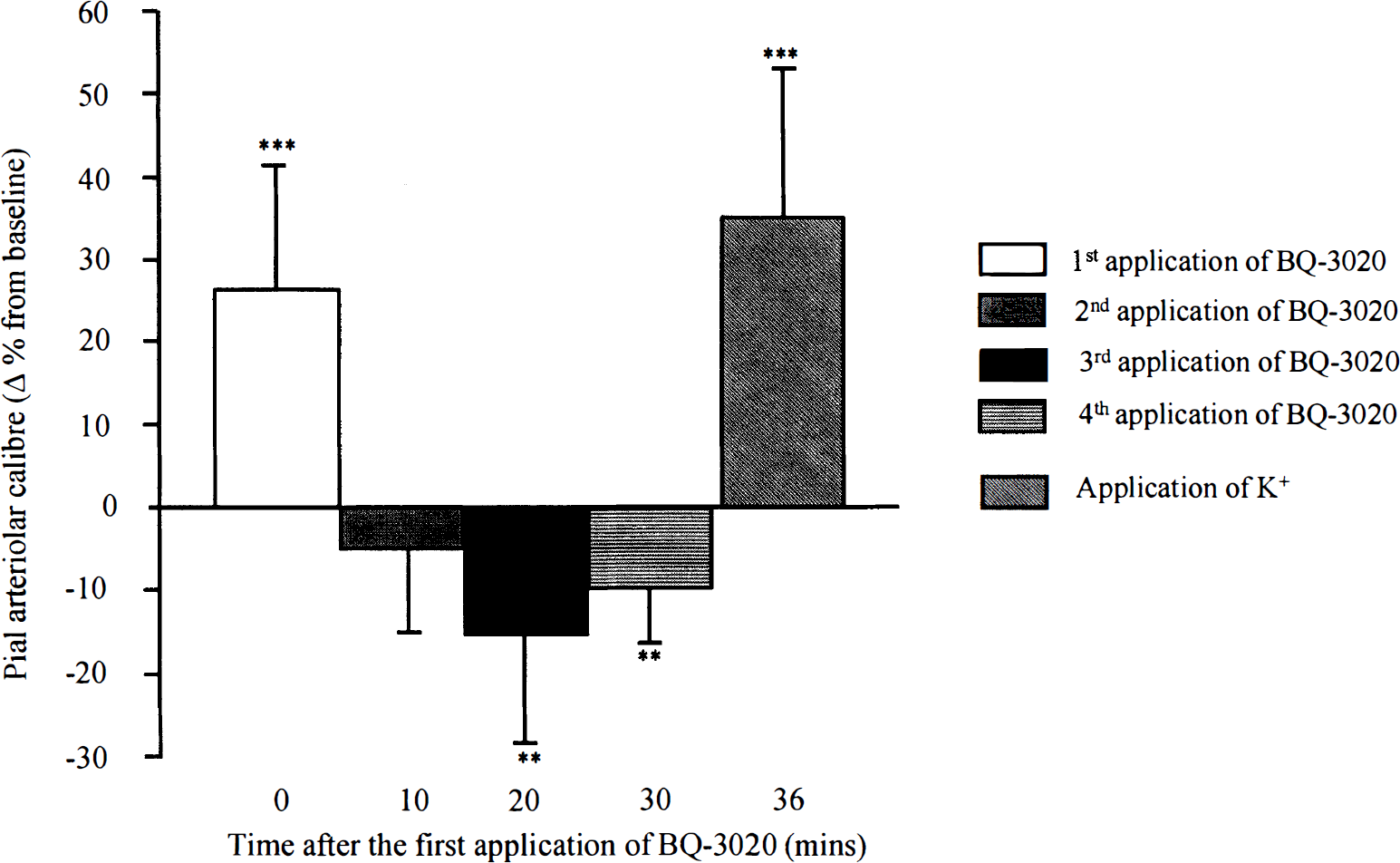
Vasomotor responses of pial arterioles to repeated perivascular microapplication of BQ-3020 (1 μmol/L) followed by adventitial application of potassium (10 mmol/L). Data are the percentage change from preinjection baseline in pial arteriolar caliber. **P < 0.001, ***P < 0.0001 for the comparison with CSF. Data are presented as mean ± SD. n, number of arterioles examined (n = 16).
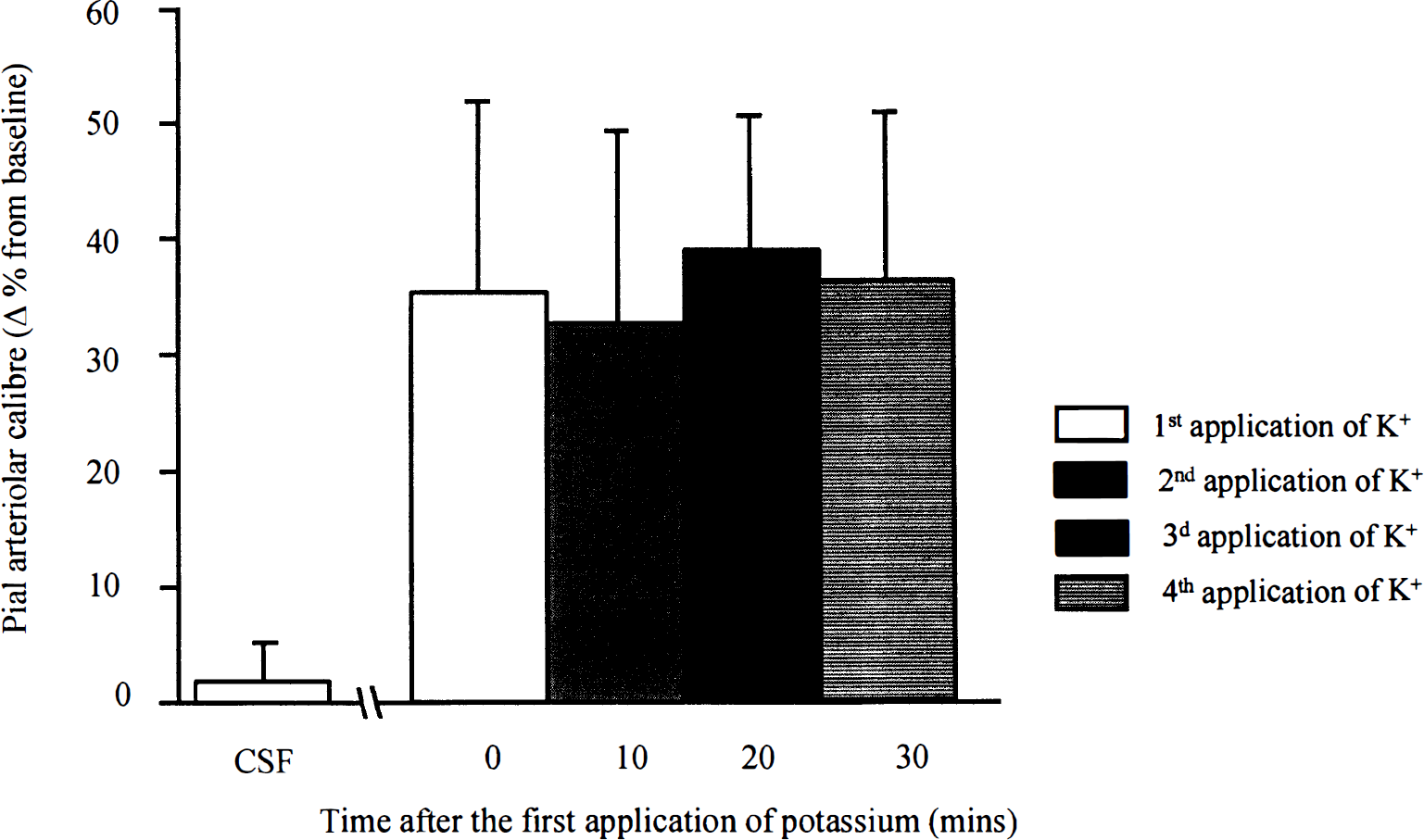
Vasomotor responses of pial arterioles to repeated perivascular microapplication of potassium (10 mmol/L). Repeated application of potassium elicited reproducible dilation of the arterioles. Data are the percentage change from preinjection baseline in pial arteriolar caliber. Data are presented as mean ± SD. n, number of arterioles examined (n = 7).
Adventitial microapplication of BQ-788
The subarachnoid perivascular application of the ETB receptor antagonist BQ-788 (0.001 to 1 μmol/L) had no significant effect on pial arteriolar caliber when compared with CSF injections (Fig. 3). This BQ-788–mediated response was similar, irrespective the size of the vessel examined (62 to 280 μm).
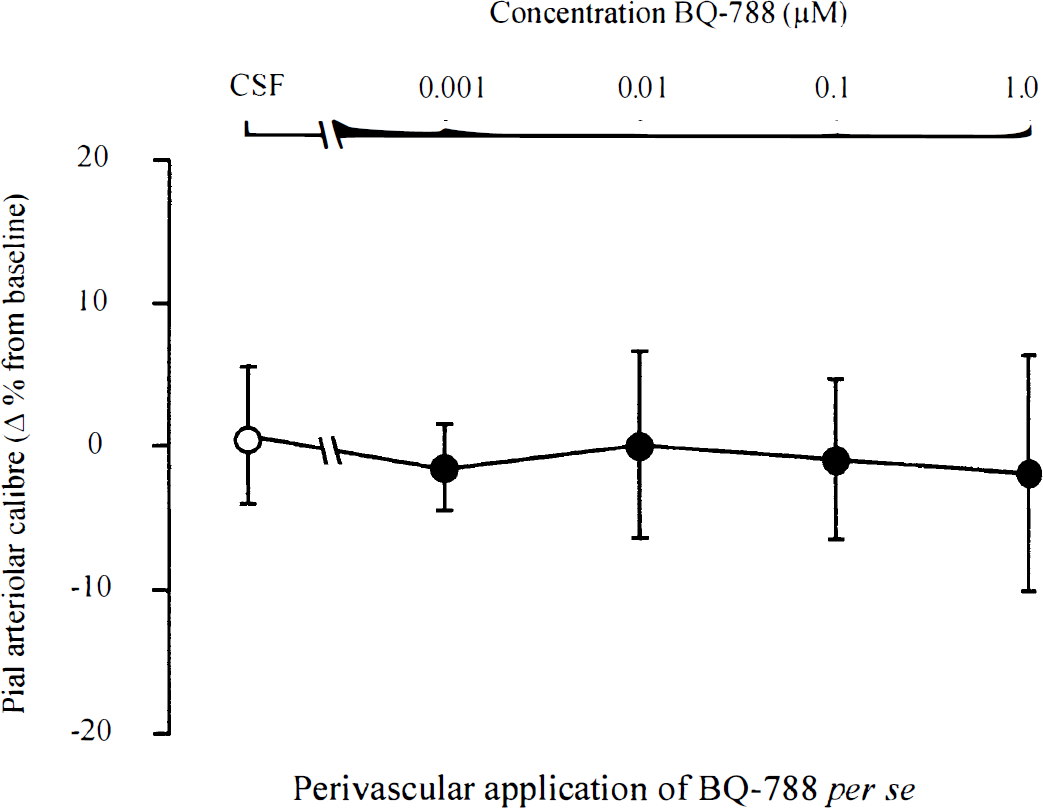
Vasomotor responses of pial arterioles to perivascular microapplication of BQ-788 per se. There are no significant alterations in pial arteriolar caliber at any concentration of BQ-788. Data are the percentage change from preinjection baseline in pial arteriolar caliber. Data are presented as mean ± SD. n, number of arterioles examined (n = 7 to 8 for each concentration).
Adventitial microapplication of BQ-788 and BQ-3020
Perivascular coapplication of BQ-788 with BQ-3020 (1 μmol/L) in vessels that have not been previously examined (preinjection caliber 114 ± 63 μm [44 to 286 μm]) produced a dose-dependent attenuation of BQ-3020–induced dilation (Fig. 4). The concentration of BQ-788 in the coinjectate, which produced half-maximal inhibition of dilatory response to the first application of BQ-3020, was estimated to be approximately 5 nmol/L. As shown earlier, the third microapplication of BQ-3020 effected a −15 ± 12% reduction in arteriolar caliber. In separate experiments, when BQ-788 (0.1 μmol/L) was coinjected with BQ-3020, after two previous applications of BQ-3020 alone, no change in arteriolar caliber was observed (0.2 ± 10.2% from baseline). This vasomotor response was significantly different from that elicited by the third application of BQ-3020 alone (P < 0.05).
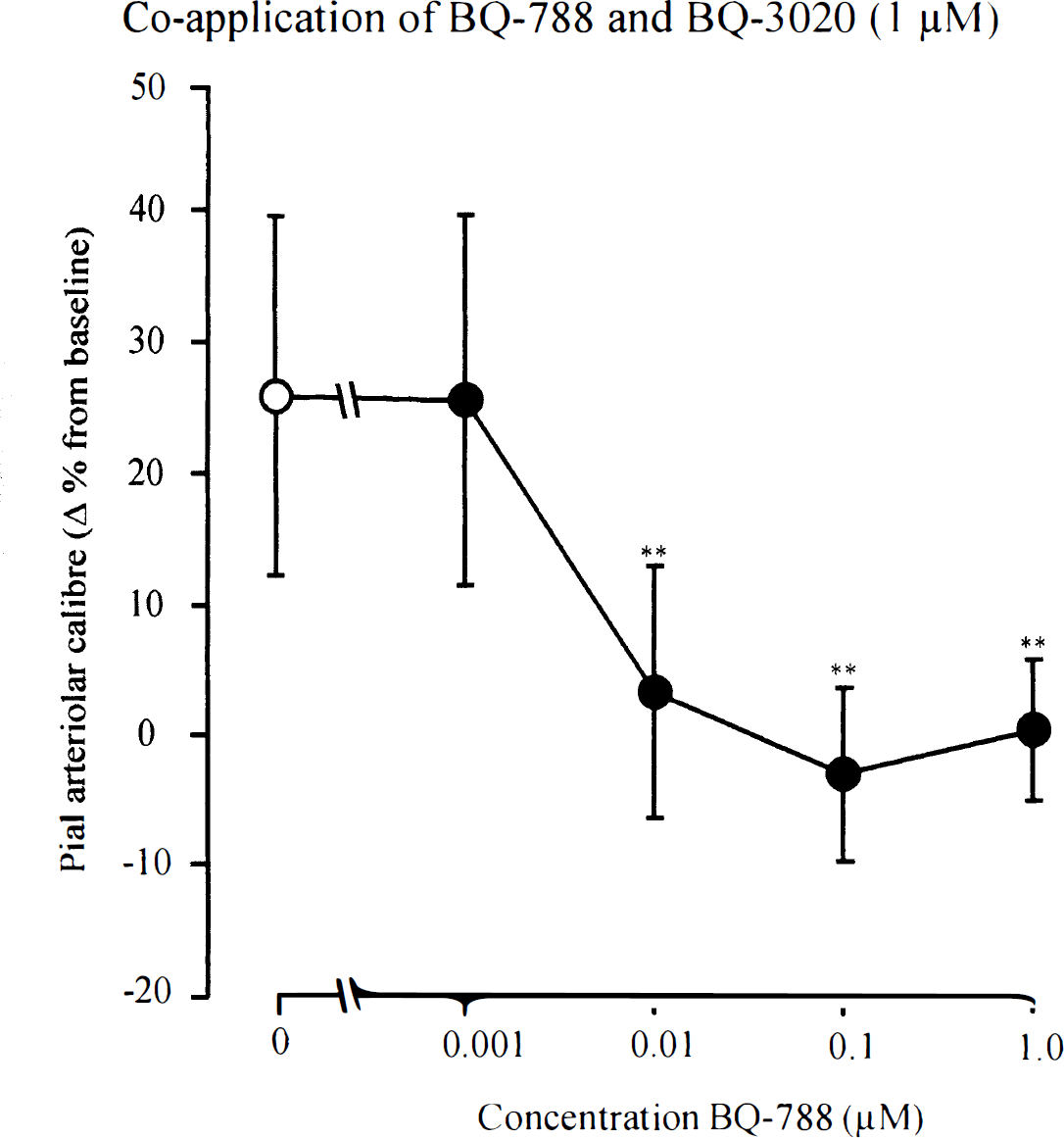
Vasomotor responses of pial arterioles to perivascular microapplication of BQ-3020 (1 μmol/L) and BQ-788. BQ-788 significantly reduces the dilation induced by BQ-3020. **P < 0.001 for the comparison with BQ-3020 alone (open circle). Data are the percentage change from preinjection baseline in pial arteriolar caliber. Data are presented as mean ± SD. n, number of arterioles examined (n = 11−13 for each concentration).
Middle cerebral artery occlusion–induced alterations in arteriolar diameter
The occlusion of the MCA effected an immediate dilation of all pial arterioles overlying the parasagittal, suprasylvian, and the ectosylvian gyri. This vasodilation was sustained in the parasagittal gyrus (zone irrigated by the anterior cerebral artery) over the entire experimental period. However, in the ectosylvian and the suprasylvian, few arterioles displayed a transient vasoconstriction. In two cats, a circumscribed zone of marked and sustained vasoconstriction was developed in the ectosylvian gyrus during the 30 minutes after MCA occlusion. Because of the limited availability of vasoconstricted arterioles in our preparation, we elected to investigate the effect of the ETB receptor agonist and antagonist only on dilated pial arterioles located within the suprasylvian and the ectosylvian gyri (i.e., the region supplied by the MCA and representing ischemic penumbra).
Adventitial microapplication of BQ-3020 after middle cerebral artery occlusion
The microapplication of CSF (pH = 7.2) on postischemic dilated arterioles elicited a small vasoconstriction (−17 ± 7%) compared with preinjection caliber (Fig. 5). The magnitude of this response did not alter significantly during the period of 30 to 210 minutes subsequent to induction of ischemia (Fig. 6). The perivascular microapplication of BQ-3020 (1 μmol/L) induced a vasoconstriction of a similar magnitude to that observed after injection of CSF (Fig. 5). Furthermore, the magnitude of the response to BQ-3020 did not vary with time after MCA occlusion (Fig. 6). No relation between the preinjection diameter, which ranged between 49 and 288 μm, and the BQ-3020–induced response was found.
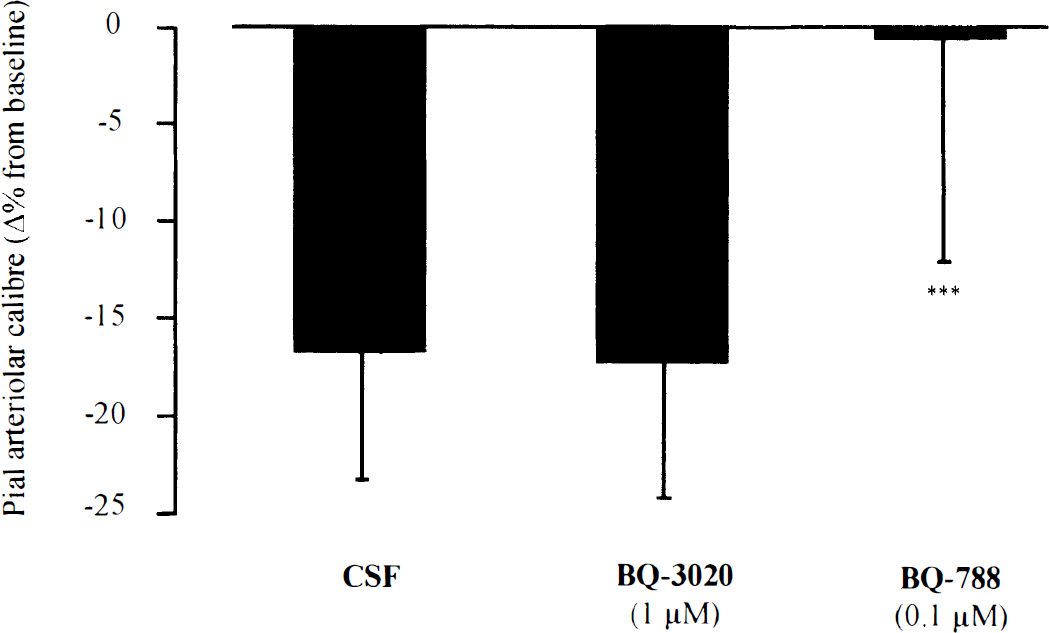
Vasomotor responses of postocclusion dilated pial arterioles to perivascular microapplication of CSF, BQ-3020 (1 μmol/L), and BQ-788 (0.1 μmol/L). Microinjections were performed 30 to 210 minutes after MCA occlusion. ***P < 0.001 for the comparison with CSF and BQ-3020. Data are the percentage change from preinjection baseline in pial arteriolar caliber. Data are presented as mean ± SD. n, number of arterioles examined (n = 34–43).
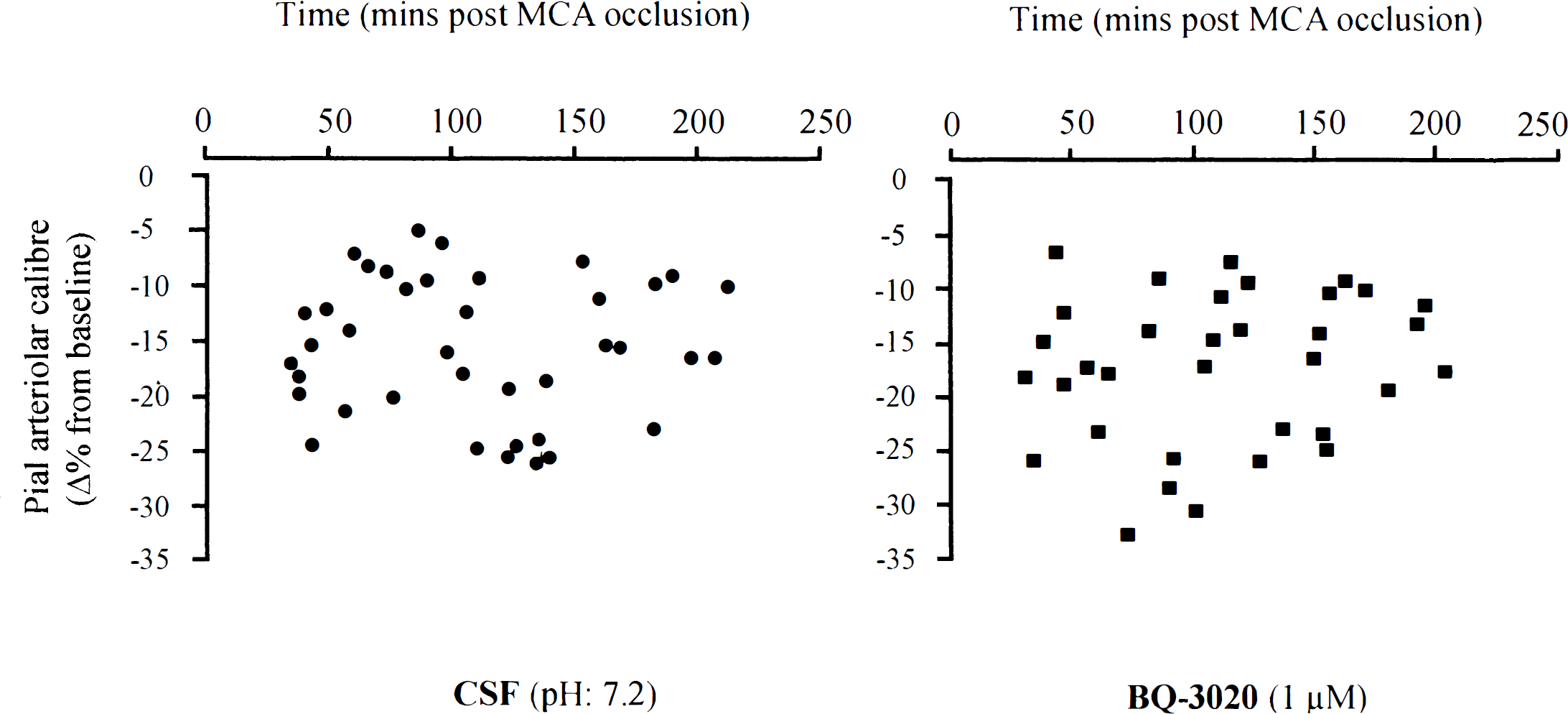
Vasomotor responses of postocclusion dilated pial arterioles to perivascular microapplication of CSF and BQ-3020 (1 μmol/L) at varying times after MCA occlusion. Each point represents the response of a single pial arteriole. The MCA was occluded at time zero. Data are the percentage change from preinjection baseline in pial arteriolar caliber.
Adventitial microapplication of BQ-788 after middle cerebral artery occlusion
The adventitial microapplication of the ETB receptor antagonist (BQ-788, 0.1 μmol/L) on postocclusion dilated pial arterioles elicited no change in the arteriolar caliber when compared with preinjection baseline (Fig. 5). However, this BQ-788–induced response was significantly different from that induced by perivascular microinjection of CSF (P < 0.001, analysis of variance). The vasomotor response to perivascular application of BQ-788 did not exhibit any marked dependency with respect to the time that had elapsed after occlusion (Fig. 7) or with respect to the size of the arterioles analyzed (diameter ranged between 41 and 277 μm).
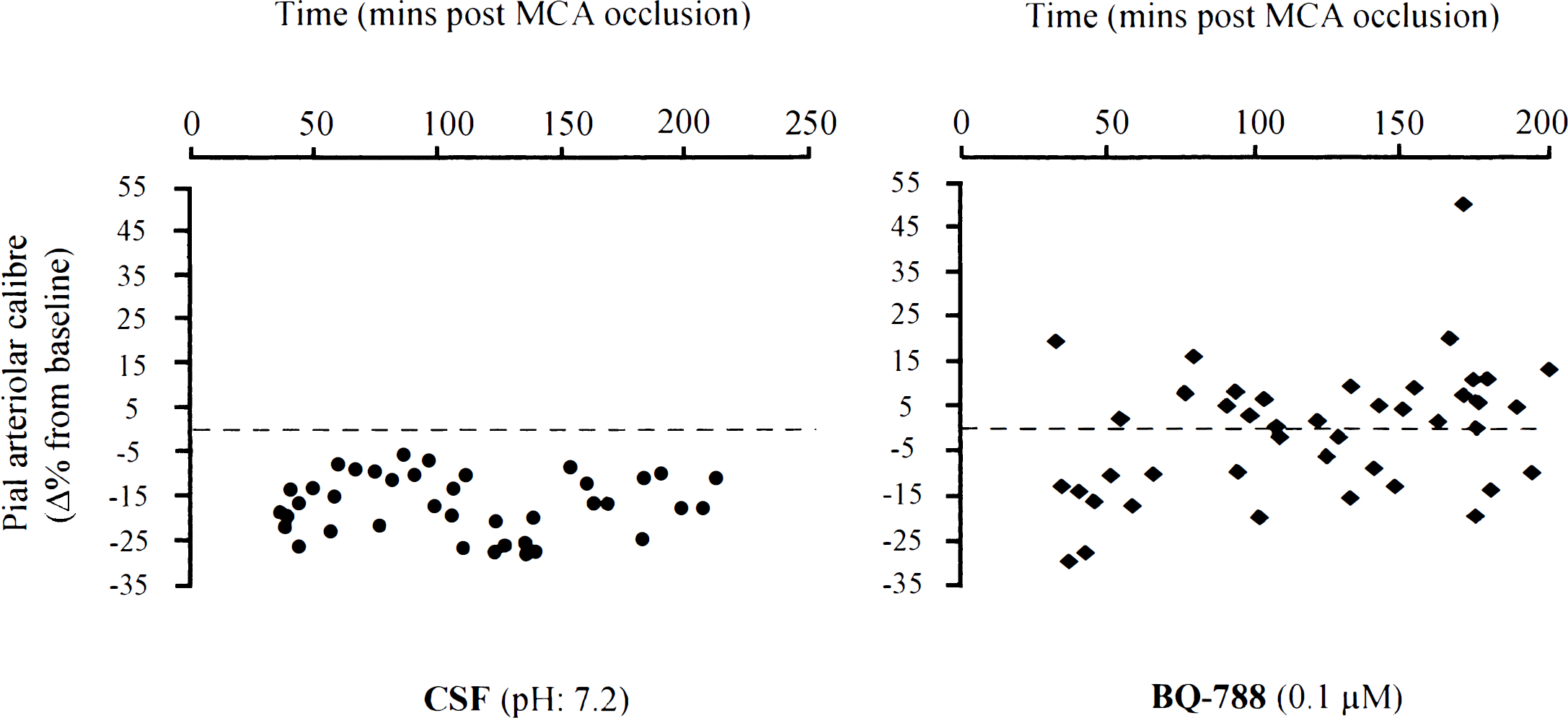
Vasomotor responses of postocclusion dilated pial arterioles to perivascular microapplication of CSF and BQ-788 (0.1 μmol/L) at varying times after MCA occlusion. The responses to CSF are identical to those in Fig. 6. Each point represents the response of a single pial arteriole. The MCA was occluded at time zero. Data are the percentage change from preinjection baseline in pial arteriolar caliber.
DISCUSSION
Four novel findings were demonstrated in the current investigation. First, repeated activation of ETB receptors induced not only tachyphylaxis of the vasodilator response, but also significant constriction of cerebral resistance arterioles in vivo. Second, perivascular microapplication of the selective ETB receptor antagonist (BQ-788) had no effect on pial arteriolar caliber per se but blocked both BQ-3020–induced dilation and BQ-3020–mediated vasoconstriction. Third, the ability of BQ-3020 to elicit dilation was lost within 30 minutes of induced focal ischemia. Fourth, ETB-mediated contractile tone contributes in a small but significant manner to limiting postischemia dilation of cortical pial arterioles.
Vasomotor effects of endothelin-B receptor agonist in normal vessels
In the cerebral circulation, the in vitro or in vivo activation of ETB receptors in rat basilar artery or in feline pial arteriole produces a marked vasodilation, which is dependent on the production of nitric oxide (Feger et al., 1994; Kitazono et al., 1995a; Schilling et al., 1995; Patel et al., 1996c). In contrast, in some peripheral vascular beds, an ETB receptor also has been linked to the contractile properties of endothelins. Indeed, it was observed that ET-1–mediated vasoconstriction was resistant to the actions of ETA receptor antagonists, and therefore could not be solely attributed to ETA receptor activation (Sumner et al., 1992; McMurdo et al., 1993; Shetty et al., 1993; Seo et al., 1994; Gellai et al., 1996; Awane-Igata et al., 1997). The current study provides the first definitive evidence of ETB–mediated constriction. Our data demonstrate that a single perivascular microapplication of selective ETB receptor agonist (BQ-3020, 1 μmol/L) induces vasodilation in cerebral resistance arterioles in the same magnitude to that reported by Patel and colleagues (1996c) in anesthetized cats. This ETB dilator response displays tachyphylaxis, as described by Kitazono and colleagues (1995a). In addition to this, we demonstrate, for the first time, that subsequent (third and fourth) activations of the ETB receptors (10-minute interval) induce significant constriction of pial arterioles (Fig. 1). The fact that repeated perivascular application of potassium revealed a reproducible degree of dilation of pial cortical arterioles and that vasoconstriction was inhibited by selective ETB receptor antagonist (BQ-788, 0.1 μmol/L) further implies that both responses (i.e., tachyphylaxis and vasoconstriction) were induced by specific ETB receptor activation. Evidence from in vivo and in vitro studies has indicated the susceptibility of endothelin receptors to desensitization. In the rat, it has been shown that in the basilar artery, vasodilation was markedly attenuated in response to a second application of IRL 1620 (ETB receptor agonist), suggesting desensitization to vasodilator effect of the ETB receptor agonist (Kitazono et al., 1995a). Nevertheless, no ETB receptor–mediated vasoconstriction was reported in this study, perhaps because of the lack of repeated activation of the ETB receptors. In awake rats, ET-1 induced depressor responses, which are probably mediated by ETB receptors and also display pronounced tachyphylaxis (Le Monnier de Gouville et al., 1996). Furthermore, an in vitro study by Clozel and Gray (1995) reports that in epithelium-denuded rat tracheal rings, the ETB receptor exhibited tachyphylaxis, since incubation of the rings for 45 minutes with sarafotoxin S6c (selective ETB agonist) induced desensitization to sarafotoxin S6c. In contrast, tachyphylaxis of the vasomotor response has not been shown to occur after application of some neuropeptides (i.e., calcitonin gene-related peptide, adrenomedullin) known to induce vasodilation in cerebral vessels (Wahl et al., 1994; Lang et al., 1997). An alternate explanation for the apparent tachyphylaxis to the vasodilation mediated by ETB receptor agonism is a disruption in vascular endothelial cell function. Repeated challenges with the ETB receptor agonist could have depleted the supply of the endothelial vasorelaxant substances that mediate the response (nitric oxide or prostayclin) or damage the endothelial cells. This mechanism also may be consistent with the lack of vasodilation with BQ-3020 after ischemia, since endothelial cell integrity could have been compromised with this insult. Nevertheless, Kitazono and coworkers (1995a) report that pretreatment of the basilar artery with an ETB receptor agonist did not affect acetylcholine-induced vasodilation (which is nitric oxide mediated). This may indicate that tachyphylaxis of the vasodilator response, because of repeated activation of ETB receptors, could not be ascribed to the depletion of nitric oxide.
The varied effect of the endothelins at the ETB receptor have led to the subdivision of the ETB receptor into ETB1 receptors (on endothelial cells), which mediate vasodilation, and ETB2 receptors (on smooth muscle cells), which mediate vasoconstriction (Warner et al., 1993; Masaki et al., 1994). The exact mechanisms underlying the contraction of pial arterioles, found in the current study, after repeated activation of ETB receptors are unknown. One hypothesis is that repeated perivascular microapplication of BQ-3020 induces desensitization of endothelial ETB (ETB1) receptors that are vasodilator and thus unmask vasoconstriction mediated by ETB2 receptors located on smooth muscle. These data argue for a potential role of ETB receptors in pathophysiologic situations where release of endogenous agonist is increased. The identification and characterization of ETB receptor subtypes, if any, would be of great importance to elucidate the function of ETB receptors in the cerebral vasculature, especially in resistance arterioles, which play an important role in the regulation of cerebral blood flow.
Endothelin-B receptor–mediated vasomotor effects in postischemic vessels
Several lines of evidence indicate the implication of endothelins in the pathogenesis of focal cerebral ischemia, although the precise definition of their pathophysiologic role has not been fully elucidated (Barone et al., 1995b; Patel, 1996). Endothelin receptor antagonists have been shown in some studies to reduce brain damage after induced focal cerebral ischemia (Barone et al., 1995a; Patel and Wilson, 1995; McAuley et al., 1996; Patel et al., 1996a,b). Critical evaluation of these studies is difficult because of differences in experimental design, species, and receptor antagonists used. Most of the studies that have intervened with endothelin receptors antagonists after induced focal ischemia have focused on the role of ETA receptors as mediator of brain damage. The role of ETB receptors in ischemic disease, however, is still to be elucidated. Endothelin ETB receptors do in part mediate vasospasm in large vessels after subarachnoid hemorrhage, since therapeutic intervention with a mixed endothelin ETA/ETB receptor antagonist was shown to be more effective than with an endothelin ETA receptor antagonist alone (Zuccarello et al., 1994). Moreover, it has been shown that there is an increase in ETB receptor agonist binding sites in basilar arteries after experimental subarachnoid hemorrhage, which may indicate a potential role of ETB receptors in these pathologic conditions (Roux et al., 1995; Hino et al., 1996). In our investigation, we addressed the implication of ETB receptor in vasomotor disturbances associated with acute focal cerebral ischemia. Perivascular microinjection of CSF (pH 7.2) onto dilated pial arterioles within the ischemic territory elicited small reduction in arteriolar caliber. This is most likely caused by the acidotic milieu within the MCA territory rendering the exogenously applied CSF alkalotic by comparison. The direct adventitial application of BQ-3020 onto dilated arterioles after MCA occlusion showed that ETB receptor–induced dilation, in normal pial arterioles, is lost in postischemic vessels. This indicates that the ETB receptor–induced vascular effects may vary in pathologic situations such as acute stroke. More importantly, the BQ-788–mediated effects in dilated arterioles, compared with the responses induced by CSF (vehicle), suggest that ETB receptors participate significantly in restricting postischemic dilation of cortical arterioles in the ischemic penumbra (Fig. 6). Nonetheless, this contribution of ETB receptors in limiting vasodilation is less pronounced compared with that of ETA receptors described by Patel et al. (1996b). These findings suggest that the use of broad-spectrum endothelin receptor antagonist (i.e., mixed endothelin ETA/ETB receptor antagonist) would be of greater benefit in reversing the increased cerebrovascular tone resulting from the release of endogenous endothelins in acute stroke.
