Abstract
In this study we explored if the secondary bioenergetic failure, which occurs a few hours after recirculation, following transient middle cerebral artery occlusion (MCAO) in rats, is caused by a compromised reflow. We induced 2 hours of MCAO and measured CBF at the end of the ischemia, as well as 15 minutes, 1, 2, and 4 hours after the start of recirculation, using autoradiographic or tissue sampling 14C-iodoantipyrine techniques. After 2 hours of MCAO, the autoradiographically measured CBF in the ischemic core areas was reduced to 3 to 5% of contralateral values. The reduction in CBF was less in neighboring, penumbral areas. After recirculation, flow already normalized in core tissues after 15 minutes, and remained close to normal for the 4 hours recirculation period studied. However, in penumbral tissues, recovery CBF values were usually below normal. The results show that tissues that are heavily compromised by the 2-hour period of ischemia and are destined to incur infarction, show a “relative hyperemia” during recirculation. In fact, some areas of the previously densely ischemic tissue showed overt hyperperfusion. This finding raises the question whether the relative or absolute hyperemia reflects events that are pathogenetically important. Because drugs that clearly ameliorate the final damage incurred fail to alter the relative hyperperfusion of previously ischemic tissues, it is concluded that vascular events in the reperfusion period do not play a major role in causing the final damage.
Focal ischemia caused by middle cerebral artery occlusion (MCAO) affects two types of tissues, “focal” and “penumbral” (Astrup et al., 1981; Hakim, 1987; Pulsinelli 1992; Siesjö 1992a, b ; Hossmann, 1994). In the focus, the tissues are so densely ischemic that they can only be salvaged by prompt reperfusion, whereas the penumbra, with its less-dense ischemia, is potentially salvageable.
In dense ischemia of the global/forebrain type, reflow usually leads to reactive hyperemia, followed by a period of secondary hypoperfusion, when blood flow decreases to values around 50% of control (Pulsinelli et al., 1982; Kågström et al., 1983a, b ; Steen et al., 1979). This hypoperfusion, which usually matches the corresponding decrease in metabolic rate, is probably not of pathogenetic importance, i.e., it does not reduce blood flow to values that jeopardize the survival of the ischemic tissues.
The situation may be different in ischemia of long duration, such as in MCAO. The results of del Zoppo et al. (1991) focused interest on the possibility that perfusion of microvessels becomes compromised in the recirculation period after 3 hours of MCAO, which is assumed to be caused by upregulation of adhesion molecules for polymorphonuclear (PMN) leucocytes, and obstructive adhesion of PMN and thrombocytes. This microvascular hypothesis of secondary tissue damage in transient ischemia of the MCAO type has received support from experiments in which the tissue damage was ameliorated by administration of antibodies to PMN leucocytes, or their adhesion molecules on endothelial cells (Chopp et al., 1994; Matsuo et al., 1994; Zhang et al., 1995). However, recent results on rats suggest that the secondary bioenergetic failure during recirculation (Folbergrová et al., 1995) is, at least in part, caused by secondary mitochondrial dysfunction (Kuroda et al., 1996a, b ), in this context, it is of interest that infarct size after transient middle cerebral artery (MCA) is ameliorated by the spin trap α-PHENYL-n-TERT-BUTYL NITRONE (PBN) (Zhao et al., 1994b) and by the immunosuppressant FK 506 (Sharkey and Butcher 1994; Kuroda et al., 1996). Results showing that the early bioenergetic failure and the mitochondrial dysfunction are ameliorated by the spin trap PBN support the notion of mitochondrial dysfunction playing an important pathogenetic role. However, it cannot be excluded that mitochondrial dysfunction is secondary to events occurring at the blood-endothelial cell interface.
The present study addresses the question of the adequacy of reflow after transient MCAO. Relatively little is known about this subject. Previous results hint that flow is not decreased in the reperfusion period. In one previous study, CBF was measured after 1 hour of MCAO followed by 2 hours of recirculation (Nagasawa and Kogure, 1989). This study, and another one from the same group in which tissue PO2 was measured (Matsuo et al., 1994), suggest that CBF is uncoupled from local metabolic rate and sufficient to increase tissue PO2. Another group studied 30 minutes of MCAO followed by 15 or 120 minutes of recirculation (Takagi et al., 1995). The results showed incomplete recovery of CBF after 15 minutes. After 120 minutes of reperfusion, CBF had normalized in the neocortex but not in the caudoputamen. The data thus suggest that reperfusion may be a limiting factor for recovery. In the present study, we induced 2 hours of MCAO and measured reflow at the end of ischemia, as well as after 15 minutes, 1, 2, and 4 hours of recovery. Thus, the reperfusion period studied covers the period in which secondary bioenergetic failure develops (Folbergrová et al., 1995; Kuroda et al., 1996 a, b ). The result suggest that in MCAO, ischemia-reflow does not follow the classical scheme of postischemic hyperperfusion and secondary hypoperfusion. Rather, densely ischemic tissues showed a relative increase in CBF during reperfusion, and hypoperfusion did not develop. In fact, tissues destined to incur infarction frequently showed areas of absolute hyperperfusion. The question arose as to whether the relative or absolute hyperperfusion reflects microvascular damage that could contribute to the final ischemic injury. To get some information on this issue, one group received PBN after 1 hour of recirculation, and CBF was measured with tissue sampling 3 hours later.
MATERIALS AND METHODS
Animal preparation and middle cerebral artery occlusion
Male Wistar rats (Møllegaard's Breeding Center, Copenhagen), weighing 310 to 340 g, were used for the experiments. The animals were fasted overnight but had free access to water. Anesthesia was induced by inhalation of 3.5% halothane. The animals were intubated and ventilated on 1 to 1.5% halothane in N2O/O2 (70:30) during the operation. The tail artery and tail vein were cannulated for blood sampling, blood pressure recording, and drug infusion. Blood pressure, P
During the operation, an electrical temperature probe was inserted 7 cm into the rectum to record core temperature, which was regularly maintained at 37°C. The animals were cooled by an air cooling system during ischemia and recirculation to keep core temperature close to normal levels (Zhao et al., 1994a).
Cerebral blood flow measurements
Cerebral blood flow was measured in awake animals placed in a small tube-shaped Plexiglass cage, restricting the movements of the animal, and giving access to the catheters for blood sampling and infusions (Dahlgren et al., 1981). A total of 44 rats were used for this study. Local CBF was measured in 8 animals by the autoradiographic technique described by Sakurada et al. (1978), as modified by Dahlgren et al. (1981). These were divided into 2 groups of 4 animals each. In one group, rats were subjected to 2 hours of MCAO without recirculation, while the other group was allowed a recovery period of 4 hours after 2 hours of MCAO. 14C-iodoantipyrine was used as the diffusible tracer, with 30 μCi of the isotope being infused at constant rate during 45 seconds. Ten arterial blood samples (29 μL each) were collected during the tracer infusion. The animal was decapitated simultaneously with the last blood sample. The brain was then quickly removed and frozen in 2-methylbutane chilled to −50°C. The 14C-activity in the blood samples were counted in a liquid scintillation counter. The brains were sectioned coronally at 20 μm in a cryostat at −20°C and exposed to an X-ray film for one week together with a set of calibrated 14C standards. After development of the exposed film the brain sections were analyzed on a Macintosh Quadra 950 using the public domain National Institutes of Health Image program (written by Wayne Rasband at the U.S. National Institutes of Health and available from the Internet by anonymous ftp from zippy.nimh.nih.gov.).
In the remaining 36 animals, regional CBF was measured with the tissue sampling technique described by Eklöf and Siesjö (1973). These animals were divided into 6 groups of 6 animals each. Middle cerebral artery occlusion was induced for 2 hours in all animals. In one group, CBF was measured at the end of the 2-hour MCAO period. The other groups were allowed recovery periods of either 15 minutes, 1, 2, or 4 hours. Two groups were studied with a recirculation time of 4 hours: nontreated and PBN-treated. In the PBN-treated group, PBN (Sigma, St. Louis, MO, U.S.A.) was dissolved in saline (10 mg·mL−1) and administered intraperitoneally in a dose of 100 mg·kg−1 after 1 hour of recirculation. Isotope administration (20 μCi), blood sampling, and decapitation were performed as in the autoradiographic CBF study. The brain was then rapidly removed and frozen as described above. Brain samples from lateral caudoputamen and somatosensory cortex were dissected from coronal sections of 3-mm thickness through the caudoputamen, and were cut into 2 pieces. The brain samples were dissolved in Soluene (Canberra-Packard, Groningen, The Netherlands) over 24 hours in room temperature and then prepared for scintillation counting together with the blood samples. Calculation of both local and regional CBF was made according to Sakurada et al. (1978).
Statistics
Statistical differences were calculated with analysis of variance followed by Scheffe's F test. A value of <0.05 was considered as significant.
RESULTS
Physiological variables
Table 1 shows arterial blood pressure, body temperature, blood gases, and glucose concentrations in all groups studied. There were no significant differences in these parameters during MCA occlusion and recirculation. However, as animals were unanesthetized during CBF measurements, their body temperature tended to be somewhat higher than when maintained during ischemia (Dahgren et al., 1981). All animals had a slight decrease in P
Physiological parameters
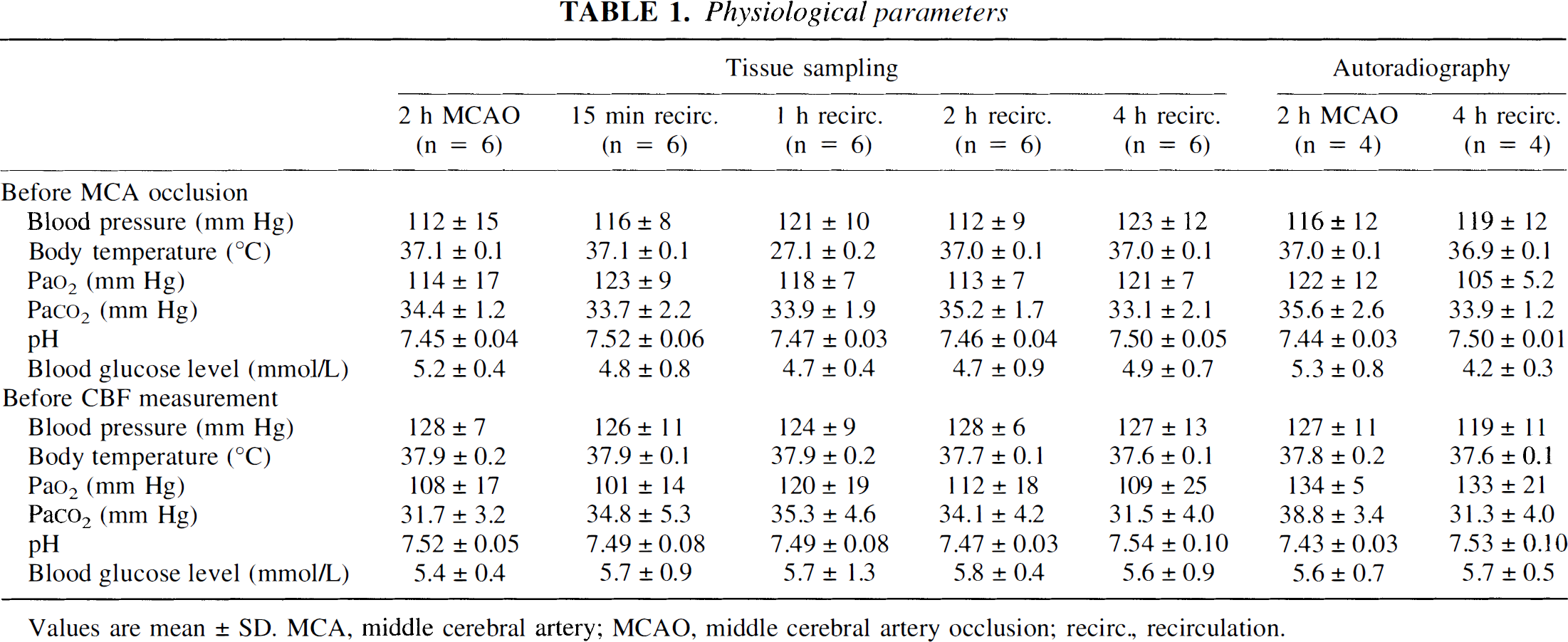
Values are mean ± SD. MCA, middle cerebral artery; MCAO, middle cerebral artery occlusion; recirc., recirculation.
Changes in CBF: autoradiography
Figure 1 shows a representative illustration of the autoradiographically measured alterations of local CBF after 2 hours of MCAO. The collected data are shown in Table 2, whereas Fig. 2 shows the percentage reduction of CBF, compared with the corresponding regions of the contralateral hemisphere. In this figure, the structures analyzed were arranged in the order of increasing CBF values during the ischemia. Thus, the areas with the lowest flow rates are at the top, while those better perfused are at the bottom of the graph. At the end of 2 hours of MCAO, CBF values in the ipsilateral hemisphere supplied by the occluded MCA were greatly decreased compared with the contralateral hemisphere. The core area, i.e., lateral caudoputamen and somatosensory cortex, had CBF values that were about 3 to 5 percent of contralateral side, respectively. The parietal, piriform, and motor cortexes also had very low values, as had the amygdala and anterior hypothalamus. The other neocortical areas and the medial caudoputamen had CBF values exceeding 20 mL 100g−1 min−1. Moderate hypoperfusion was observed in the medial caudoputamen, globus pallidus, anterior cingulate cortex, and hippocampus. Hyperperfusion was observed in the ipsilateral substantia nigra and thalamus, although the changes did not reach statistical significance. The mechanisms behind this increase in substantia nigra are not known. However, one can speculate that the reduction in CBF in the caudoputamen bereaves the substantia nigra of its GA-BAergic inhibition, causing an increase in metabolic rate, and blood flow.
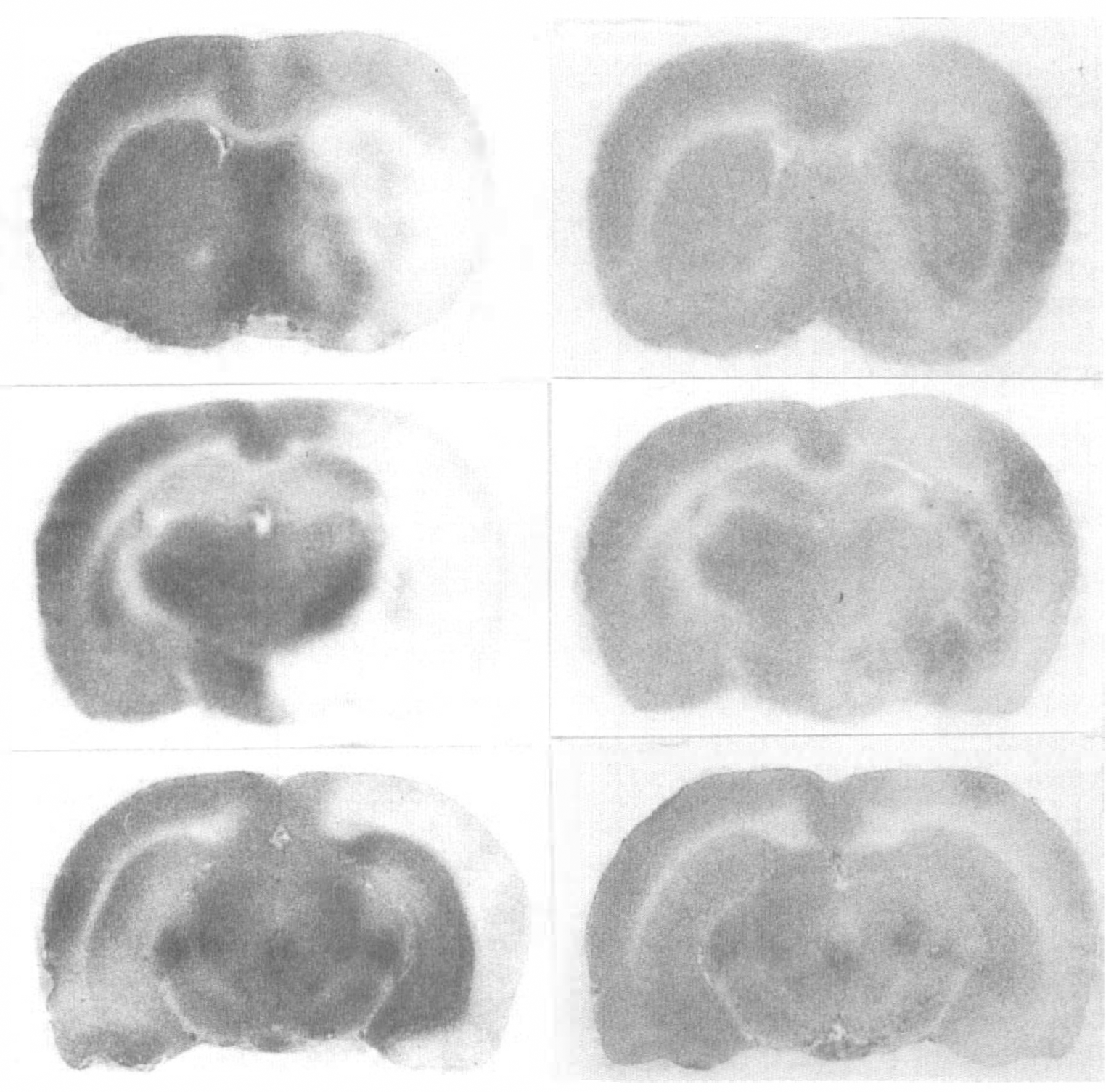
Representative local cerebral blood flow autoradiograms in cortical sections at level of caudoputamen (top), hippocampus (middle), and substantia nigra (bottom) from an animal subjected to 2 hours of middle cerebral artery occlusion (MCAO) (left). Note blood flow deficits in caudoputamen and overlying neocortex of the right hemisphere (ischemic focus). Corresponding brain sections from an animal with 4 hours reflow after 2 hours of MCAO (right). Note the darker areas (relative hyperemia) in the ischemic focus.
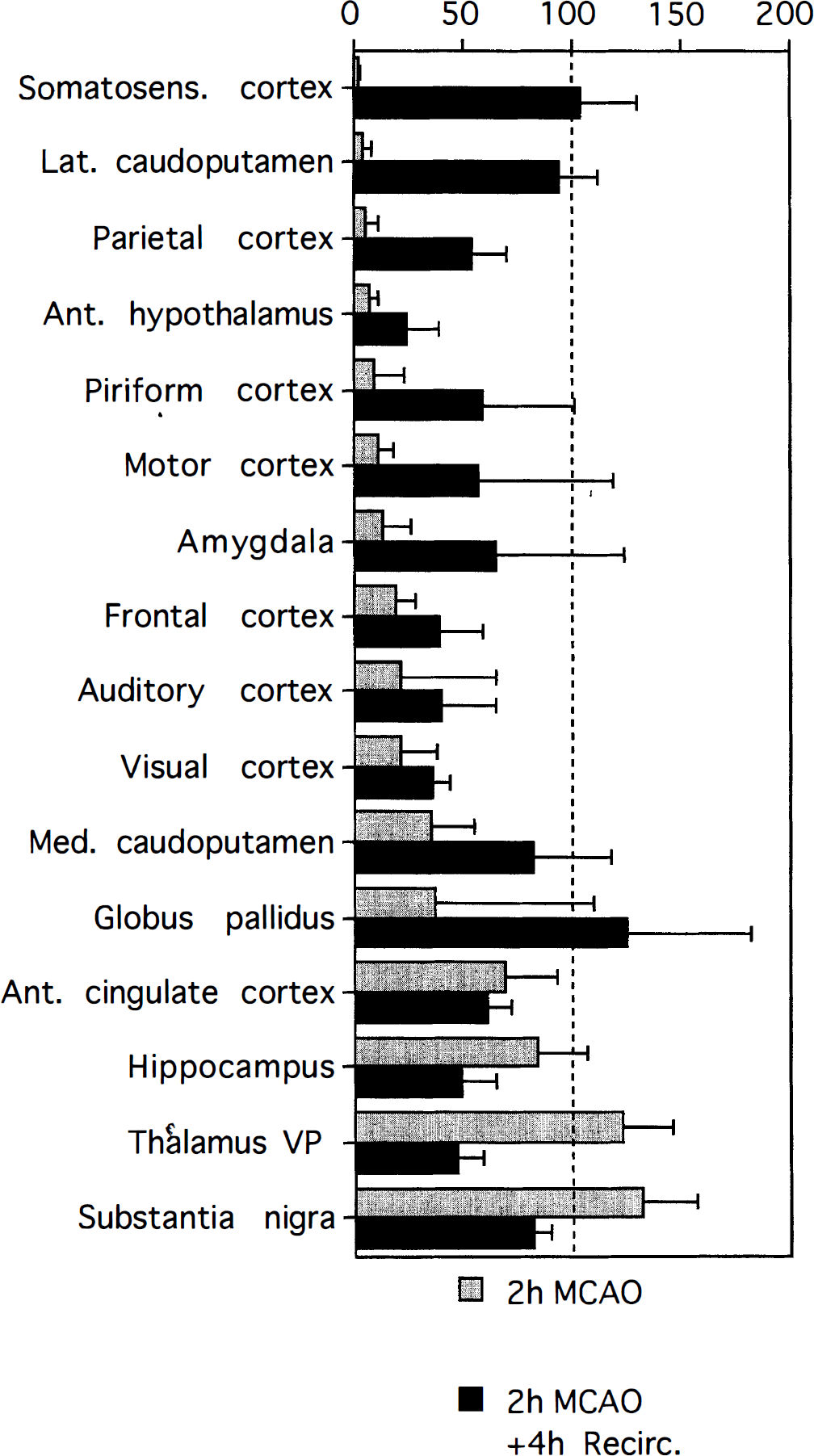
Local cerebral blood flow in the middle-cerebral- artery-occluded hemisphere, as percent of contralateral hemisphere. Values are means ± SD, n = 4. Ant., anterior; lat., lateral; med., medial; recirc., recirculation; somatosens., somatosensory; VP, ventroposterior nucleus.
Local cerebral blood flow during and after MCAO (autoradiography)
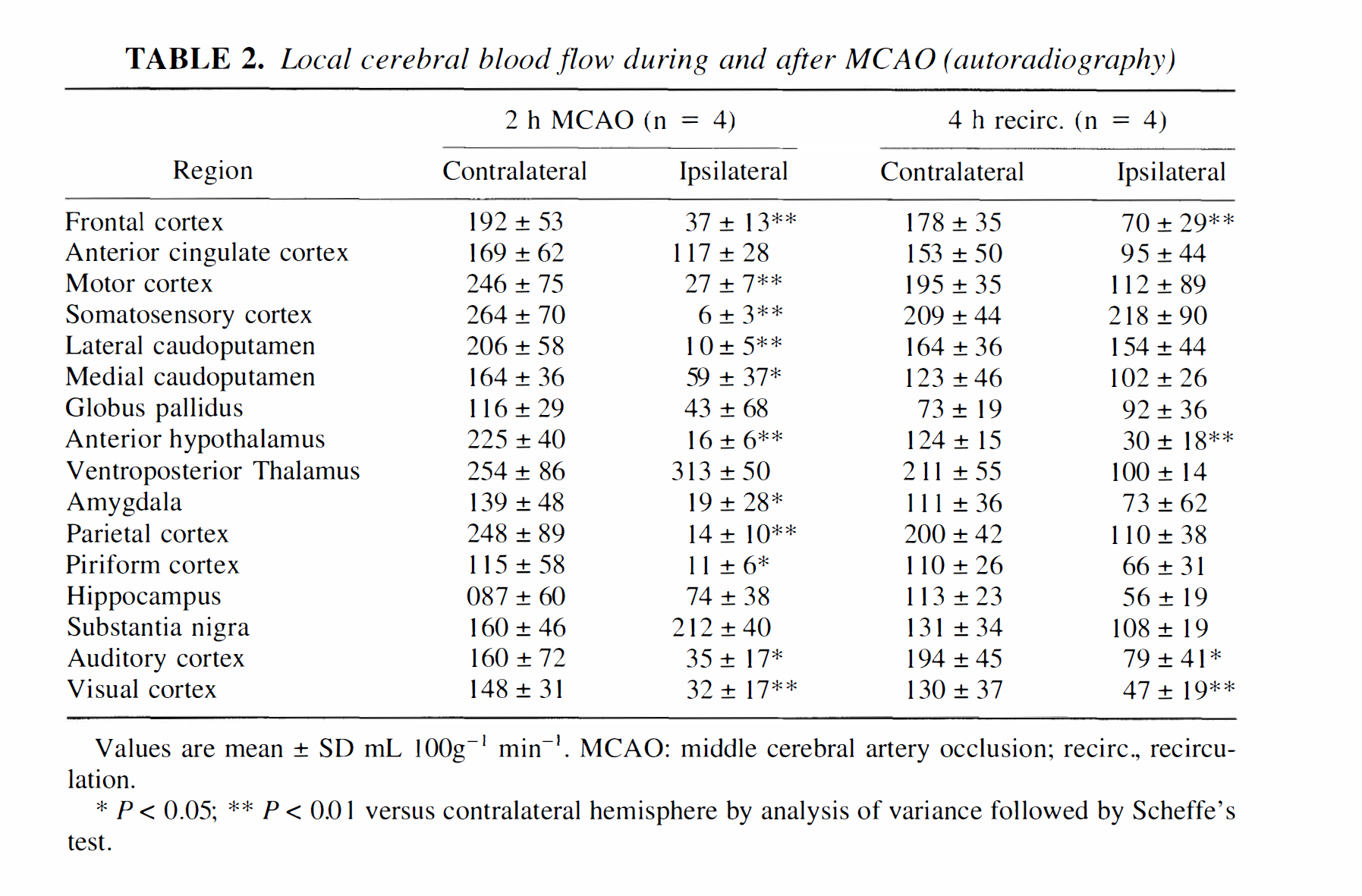
Values are mean ± SD mL 100g−1 min−1. MCAO: middle cerebral artery occlusion; recirc., recirculation.
* P < 0.05; ** P < 0.01 versus contralateral hemisphere by analysis of variance followed by Scheffe's test.
After 4 hours of recirculation, many areas, including most neocortical ones, showed moderate hypoperfusion, while some areas with moderate or no ischemia during the insult had relatively high CBF values (medial caudoputamen, globus pallidus, and substantia nigra). The most striking results, though, were the persisting hypoperfusion in the anterior hypothalamus, and the absence of hypoperfusion in the core areas, i.e., somatosensory cortex and lateral caudoputamen. In the latter, some areas had flow rates that increased above control. For example, when flow was averaged over such hyperemic areas in the caudoputamen, a value of 444 ± 98 mL 100 g−1 min−1 was obtained. This was significantly different from the value obtained for corresponding regions on the contralateral side (240 ± 52 mL 100 g−1 min−1, P < 0.02).
Changes in CBF: tissue sampling
Figure 3 shows the areas sampled to represent core areas in caudoputamen and neocortex, whereas Fig. 4 gives regional CBF values (mean ± SD) during ischemia, as well as after 15 minutes, 1, 2, and 4 hours of recirculation. At the end of 2 hours of MCAO, CBF in the ipsilateral somatosensory cortex and caudoputamen was greatly decreased compared with the contralateral hemisphere. After 2 hours of MCA occlusion, followed by 15 minutes of recirculation, CBF in the ipsilateral hemisphere was restored to the contralateral values, and remained at this level throughout the reperfusion period. There was no significant difference in CBF between groups during reperfusion. Delayed postischemic hypoperfusion was thus not observed in the ipsilateral core regions. After 2 hours of MCA occlusion followed by 4 hours of recirculation, CBF in the ipsilateral caudoputamen was similar in vehicle-injected and PBN-injected animals (136 ± 36 and 118 ± 31 mL 100g−1 min−1), i.e., PBN administered 1 hour after the start of recirculation did not change the overall CBF values, as measured after 4 hours.
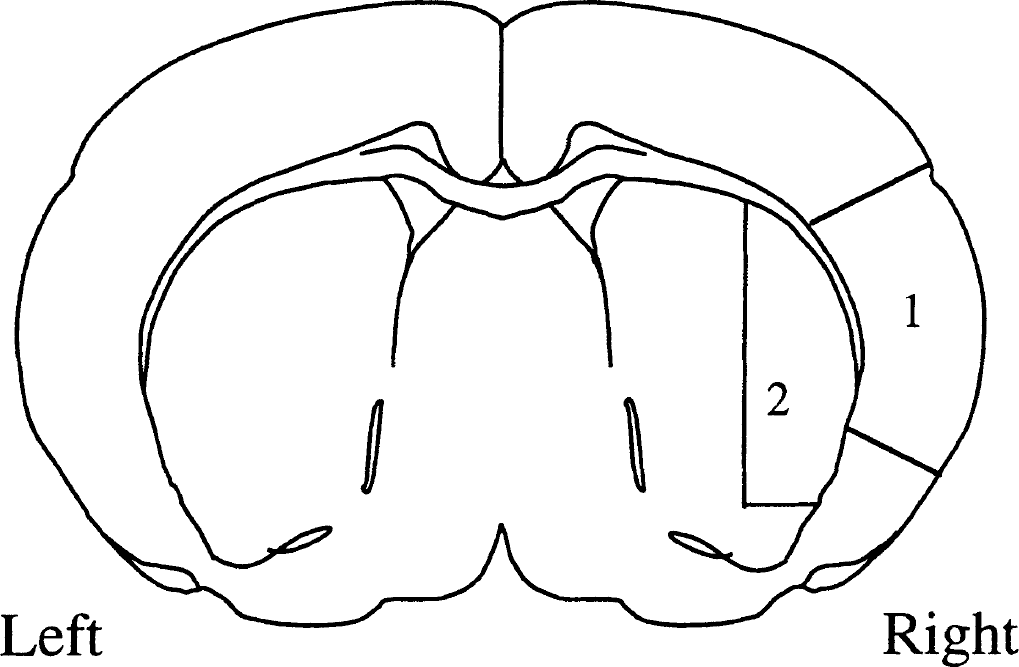
Anatomic regions of somatosensory cortex and caudoputamen used for measurement of regional CBF by a tissue sampling technique. Two pieces of 2.0-mm thickness were cut out from each hemisphere as illustrated. (1) somatosensory cortex; (2) caudoputamen.
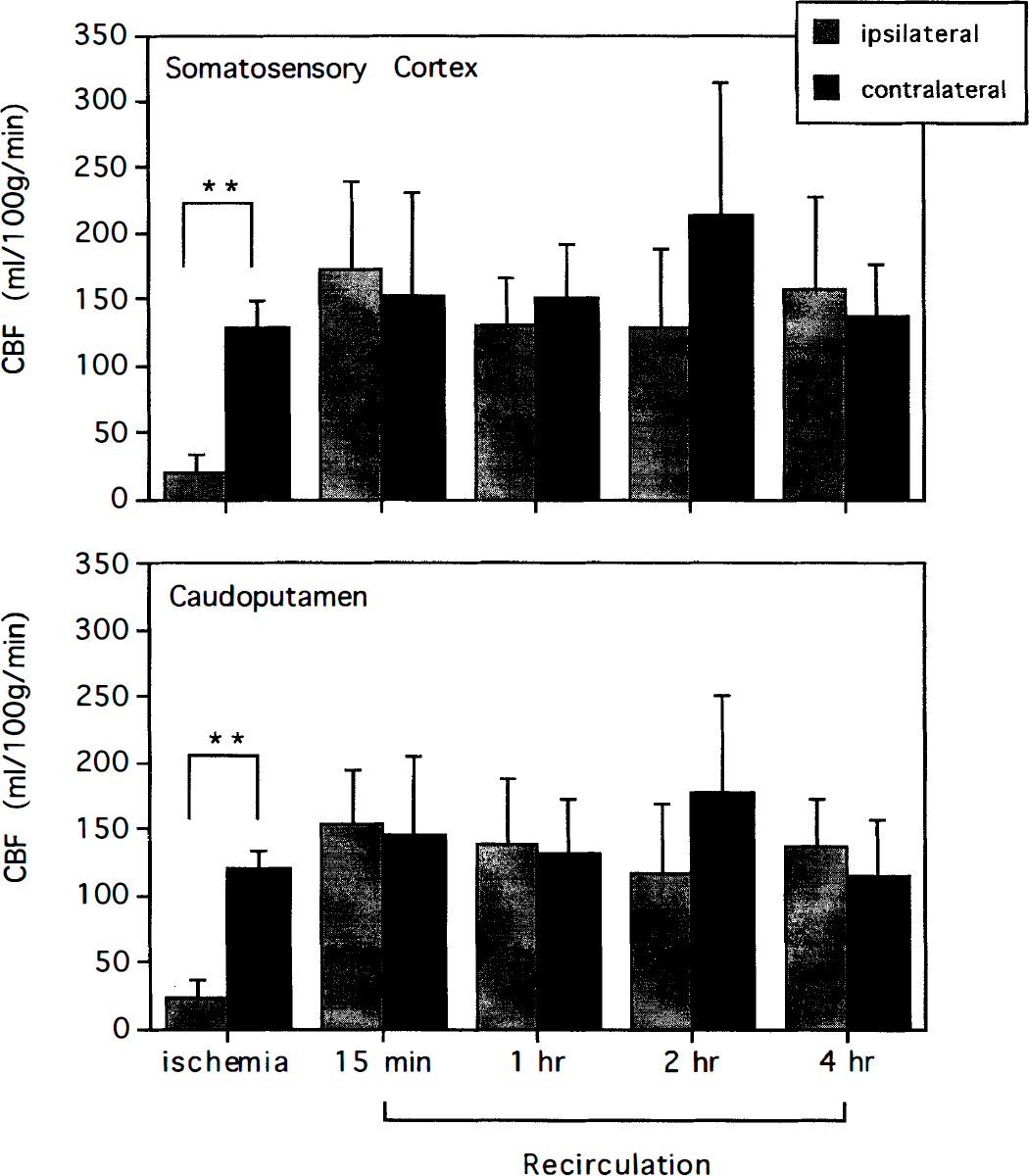
Regional cerebral blood flow during and after middle cerebral artery occlusion in the somatosensory cortex (A) and caudoputamen (B). Values are means ± SD, n = 6 in each group. ** denotes a significant (P < 0.01) difference between hemispheres (two-factor analysis of variance followed by Scheffé's F-test).
DISCUSSION
As remarked in the introduction, in focal ischemia of the type caused by occlusion of the MCA, there is a densely ischemic focus, supplied by the occluded artery, and perifocal penumbra areas in which CBF is higher but nonetheless in the ischemic range. In primates, irreversible ischemic changes occur if CBF decreases below 12 to 15 mL 100g−1 min−1 for 2 to 3 hours, or below 20 mL 100g−1 min−1 during permanent occlusion (De Girolami et al., 1984; Marcoux et al., 1982; Buchan et al., 1991; Dirnagl 1993). The thresholds are probably somewhat higher in the rat (Tyson et al., 1984; Kaplan et al., 1991; Mies et al., 1991; Heiss et al., 1992; Jacewicz et al., 1992; Paschen et al., 1992). The present data confirm the previous findings in revealing CBF values of <10% of control in the densely ischemic focal areas. In contrast, cells in the penumbra are at risk. They can survive for hours before they are recruited in the infarction process, and they can be salvaged by pharmacological treatment (Pulsinelli 1993; Siesjö 1992a, b ; Hossman 1994; Siesjö et al., 1995; Siesjö and Siesjö 1996). These characteristics correspond to intraischemic flow rates of 10 to 25% of control.
As reported recently (Zhao et al., 1994b), PBN markedly reduces infarct volume resulting from 2 hours of MCAO, even when administered 1 or 3 hours after the initiation of recirculation. Furthermore, recirculation was found to lead to partial recovery of the bioenergetic state at 1 hours, with no further recovery after 2 hours, and signs of a secondary deterioration at 4 hours; this secondary deterioration of the bioenergetic state was prevented by PBN administered 1 hour after the start of recirculation (Folbergrová et al., 1995). These results can be explained by the fact that ischemia elicits reactions producing mediators of rapidly developing secondary damage, affecting either microcirculation or mitochondria. Some of these mediators are probably free radicals, or nitric oxide, or other reactive metabolites, emanating from lipid hydrolysis and arachidonic acid metabolism (Siesjö 1992a, b , 1995; Feuerstein et al., 1994; Hallenbeck 1994, 1996; Siesjö and Siesjö 1996).
The original objective of this study was to explore CBF changes in the ipsilateral hemisphere after 2 hours of MCAO followed by recirculation. One could envisage a situation in which a secondary microvascular compromise is reflected in a change in CBF. However, our results showed the somewhat unexpected fact that, after 2 hours of MCAO followed by 15 minutes to 4 hours of recirculation, CBF in the ipsilateral somatosensory cortex and lateral caudoputamen was similar to that in the corresponding contralateral hemisphere. However, our results are in partial agreement with those of Nagasawa and Kogure (1989). These authors showed that, after 3 hours of MCAO followed by 3 hours of recirculation, CBF in the ipsilateral cortex was similar to that in the contralateral cortex, with no significant difference between CBF in the recirculated area and the corresponding area of the control rats. Other studies in which CBF was measured after temporary MCAO have also shown rapid restoration of CBF to near normal or increased levels after recirculation (Shigeno et al., 1985; Kuroiwa et al., 1988; Buchan et al., 1991). The combined data show high CBF values in the core area after reperfusion (Shigeno et al., 1985; Nagasawa and Kogure, 1989). Furthermore, the lowest CBF values during recirculation in the ipsilateral area were encountered in areas that did not suffer severe ischemia, and that will not usually develop an infarct (Nagasawa and Kogure, 1989; Memezawa et al., 1992b).
The present results extend these previous observations by providing flow values for a variety of structures and for recirculation periods varying between 15 minutes and 4 hours, i.e. the critical period in which secondary bioenergetic failure and mitochondrial dysfunction develop (Folbergrová et al., 1995; Kuroda et al., 1996a, b ). Furthermore, they show that PBN does not ameliorate this secondary “relative hyperperfusion”. The results thus lend no support to the notion that the relative hyperperfusion represents microvascular damage caused by free radicals of a type and degree which contributes to the secondary deterioration of the energy state. In discussing alternative mechanisms other than vascular obstruction, the following facts should be taken into consideration.
As discussed, ischemia triggers the expression or upregulation of adhesion molecules on endothelial cells and on PMN leucocytes, raising the possibility that obstruction of microvessels by activated PMN leucocytes gives rise to a “no-reflow” phenomenon (del Zoppo et al., 1991; del Zoppo 1994; Garcia et al., 1994). In support, focal ischemic damage can be ameliorated by blocking adhesion molecules on endothelial cells and PMN leucocytes by administration of antibodies to ICAM-1 and CD11b/18 (Chopp et al., 1994; Matsuo et al., 1994; Zhang et al., 1995). In the rat, though, we have been unsuccessful in showing a reduced capillary patency, when using the same experimental paradigm as in the present study (Vogel, Li, Smith, Kuschinsky and Siesjö, unpublished results).
Our previously published results show that animals subjected to 2 hours of focal ischemia, followed by recirculation, show an initial, partial recovery of mitochondrial O2 consumption, to be followed by a gradual decrease of respiratory control ratio (Kuroda et al., 1996a, b ). The results lead to the tentative conclusion that a substantial part of the secondary damage is caused by free radical-mediated injury to mitochondrial respiratory components, either pyruvate dehydrogenase (Zaidan and Sims, 1994; Bogaert et al., 1994) or cytochrome c oxidase (Almeida et al., 1995).
A tentative explanation for these different results is as follows. Oxidative damage to vessels during the immediate reperfusion period yields vascular dilatation, possibly also unresponsiveness to vasodilatory stimuli of the type described in trauma by Kontos et al. (Kontos, 1985, 1989). This would explain the relative hyperperfusion, and the lack of effect of PBN, given after 1 h of recirculation. The subsequent upregulation of adhesion molecules, and the adherence of PMN leucocytes, have the potential of obstructing capillaries. However, if the adhesion occurs in larger vessels, such as postcapillary venules, flow would not be obstructed. The adverse effects of such adhesion could then be related to the oxidative burst triggered by activated leucocytes. It is perhaps on such events that PBN acts. Clearly, this is a speculative deduction but it seems justified that interest is focused on PMN-endothelial cell interactions.
