Abstract
The role of GABA in regulating cerebral microvessels was examined in the parenchyma of the hippocampus and the surface of the neocortex. Microvessels were monitored in in vitro slices using computer-assisted videomicroscopy, and synaptically evoked field responses were simultaneously recorded. γ-Aminobutyric acid (GABA) and the GABAA receptor agonist, muscimol, elicited vasodilation in hippocampal microvessels, whereas the GABAB receptor agonist, baclofen, elicited constriction. The muscimol-induced dilation persisted in the presence of the nitric oxide synthase inhibitor, N-nitro-
Converging lines of evidence suggest that local neurons are major participants in the regulation of regional cerebral blood flow (CBF). At rest, regional CBF is directly correlated with the level of local neuronal activity (Lou et al., 1987). Furthermore, CBF increases in a spatially and temporally precise manner during enhanced neuronal activity (Fox and Raichle, 1986). This vascular response frequently is dissociated from changes in regional cerebral metabolism, suggesting that these blood flow changes are not necessarily a consequence of metabolic alterations (Lenniger-Follert and Lübbers, 1976; Nakai et al., 1983; Fox and Raichle, 1986; Lou et al., 1987; Fox et al., 1988). Finally, the destruction of local neurons has been shown to block the cerebrovascular response to stimulation of distant neural centers (Iadecola et al., 1987). Taken together, these findings strongly implicate a form of direct signaling of cerebral blood vessels from their local neuronal environment.
Local neurovascular signaling in the CNS necessarily involves the modulation of parenchymal microvessels, which are the ultimate supply vessels for neurons and the least understood of all cerebral vessels. These vessels are embedded within the brain neuropil and may therefore be exposed to a variety of signaling molecules released from neurons and glia in their immediate environment. Neurotransmitters and neuromodulators could conceivably not only serve to alter local neuronal activity but also could regulate local CBF. One possible source of vasoregulatory substances is the local interneuron. Interneurons are often associated with cerebral microvessels, forming close neurovascular appositions (Hendry et al., 1983; Schottler et al., 1996). The primary neurotransmitter released from interneurons is γ-aminobutyric acid (GABA), the major inhibitory neurotransmitter in the brain (Roberts, 1988; Goodchild, 1993). In the hippocampus, approximately 90% of interneurons are GABAergic (Woodson et al., 1989). Many of these interneurons exhibit increased activity during enhanced regional activation (Miles, 1990). In addition, GABAergic interneurons in the hippocampus exhibit high levels of spontaneous activity and GABA release (Lacaille and Williams; 1990), and spontaneous activation of GABA receptors occurs at a high frequency in the hippocampus (Soltesz and Mody, 1994). To evaluate the role of the interneuron in microvascular regulation, the vascular effects of GABA must be considered. GABA may be a messenger to local microvessels and induce changes in hippocampal blood flow. Previous studies show that exogenously applied GABA elicits dilation in isolated large cerebral blood vessels (Edvinsson and Krause, 1979). Although the role of GABAergic mechanisms in regulating parenchymal microvessels is unknown, it is possible that these vessels are also responsive to GABA and that endogenous GABA could modulate their resting and/or activity-dependent states.
There are several mechanisms through which GABA could influence the parenchymal microvasculature. GABA may have direct actions on nearby blood vessels or may operate through neuronal or glial intermediaries. GABA may exert its vascular effects through an inhibition of neuronal discharge activity (i.e., action potential generation). By suppressing the discharge activity of local neurons, GABA might indirectly alter microvascular tone. Also, GABA-induced dilation in the cerebral microvasculature could be mediated by chemical intermediaries such as the well-characterized vasodilator nitric oxide (NO). Recent evidence indicates that NO is a critical signaling molecule in the regulation of hippocampal microvessels (Fergus et al., 1995, 1996). In addition, NO-mediated GABAergic relaxation has been described in peripheral smooth muscle (Boeckxstaens et al., 1991).
GABA could participate in the vascular responses to more profound physiologic stimuli such as hypoxia. The level of GABA increases significantly in response to hypoxia (Young et al., 1992; Nilsson and Lutz, 1993; Romijn et al., 1994), and hypoxia elicits a vasodilatory response in the brain (e.g., Kontos et al., 1978). Moreover, GABA receptor agonists have proven to be neuroprotective in rat, rabbit, and gerbil models of ischemia (Sternau et al., 1989; Lyden and Hedges, 1992). The possibility therefore exists that GABA contributes to vasodilation during metabolic challenge and that the neuroprotective effects of GABAergic agents are mediated, partly by actions on the cerebrovasculature.
The in vitro slice preparation permits the examination of parenchymal microvessels within their “normal” cellular environment. Although the blood vessels in this preparation lack intraluminal flow and pressure, which have been shown to play a role in the regulation of tone (Johnson, 1980; Johansson, 1989; Mellander, 1989), the physiologic reactivity of vessels in the brain slice preparation has been demonstrated (Sagher et al., 1993; Fergus et al., 1995, 1996). The present study used in vitro brain slices to examine the roles of exogenous and endogenous activation of GABA receptors in the regulation of cerebral microvessels, to investigate the involvement of potential intermediaries in GABAergic signaling of cerebral microvessels, and to examine the role of GABA receptor activation in hypoxic vasodilation.
MATERIALS AND METHODS
All studies were performed using protocols approved by the Animal Review Committee at the University of Virginia (Charlottesville) and were in accordance with the Guide for the Care and Use of Laboratory Animals by the National Institutes of Health.
Slice preparation
In vitro brain slices were prepared from adult male Sprague Dawley rats (150 to 250 g), as described previously (Sagher et al., 1993; Fergus et al., 1995). Animals were anesthetized with sodium pentobarbital (60 mg/kg given intraperitoneally) and perfused intracardially with 60 mL of artificial cerebrospinal fluid (aCSF) containing 12.5 mg of indigo carmine (Sigma Chemical Co., St. Louis, U.S.A.). Indigo carmine is used as a nontoxic dye to improve visualization of the blood vessel walls within the brain slice. The animals then were killed by decapitation, and the brains were placed in ice cold oxygenated aCSF. Parenchymal and surface brain slices were cut transverse to the longitudinal axis of the hippocampus (350 μm in thickness) and parallel to the surface of the neocortex (approximately 400 μm), respectively, using a McIlwain tissue chopper. Cortical slices were taken from the frontal and parietal regions. The slices were placed in a holding chamber and maintained on a nylon net at interface between aCSF and a humidified, oxygenated atmosphere. The slices were kept in this holding chamber at 32°C for at least 1 hour after slicing. Individual slices then were transferred to a recording chamber mounted on the stage of a light microscope, where they were submerged and superfused continuously with aCSF that was oxygenated by constant bubbling with 95%/5% oxygen/carbon dioxide at 33°C.
Examination of microvessels
Parenchymal and surface microvessels were examined using a computerized videomicroscopy system described previously (Sagher et al., 1993; Fergus et al., 1995). In the hippocampal slices, vessels were chosen either in stratum radiatum of area CA1 or in the middle molecular layer of the dentate gyrus. The resting luminal diameter of these parenchymal microvessels ranged from 8 to 25 μm. These diameters reflect an underestimation of outer vessel diameter and are consistent with those of cerebral arterioles. Vessels with thin walls, indicative of a lack of smooth muscle, were not used; moreover, the smooth muscle surrounding the vessel lumen could be visualized above and/or below the plane of the vessel lumen in many instances. Luminal diameter of the microvessels was monitored continuously throughout each experiment. The drugs under investigation were added to the medium superfusing the slice. Because of the limited myogenic tone present in such in vitro preparations, microvessels were preconstricted with a V1 receptor agonist, vasopressin [Phe2, Ile3, Orn8] (V1 ag) to examine vasodilatory responses. Dilatory responses in preconstricted vessels are expressed as a percentage of the preconstriction for normalization.
Extracellular recording in hippocampal slices
Synaptic responses were elicited using a bipolar stimulating electrode, consisting of a pair of twisted 0.05-mm Formvar-insulated Nichrome wires. Field excitatory postsynaptic potentials (fEPSP) were recorded with a glass micropipette filled with aCSF with a tip resistance of 1 to 3 megaohms. This extracellular recording electrode was placed approximately 20 μm from the vessel. The stimulating electrode was placed at least 1 mm away along the trajectory of the afferent fibers projecting to the region of the microvessel (i.e., Schaffer collateral/commissural pathway for vessels in area CA1 and perforant path fibers for vessels in the dentate gyrus). Input/output (i.e., stimulus intensity versus fEPSP slope) curves were generated for the synaptic responses in the region of the microvessel. Stimulus intensity then was adjusted to 70% of the maximal response, and single stimuli were delivered at a low rate (1 per 20 seconds) for the remainder of the experiment.
Pharmacologic manipulations
The pH was adjusted to within physiologic range (7.35 to 7.45) for all solutions used. In the first series of experiments, the effects of GABA receptor agonists on hippocampal microvascular tone were examined. Pilot experiments performed with GABA receptor agonists in both resting and preconstricted vessels indicated that GABAA receptor activation results in vasodilation and GABAB receptor activation results in vasoconstriction. Therefore, GABAA-mediated responses were examined in preconstricted vessels and GABAB-mediated responses were examined in resting vessels. To examine the effects of GABA and a selective GABAA receptor agonist, one of the following treatments was performed after a stable constriction was achieved with V1 ag (25 nmol/L): (1) GABA (10 μmol/L to 10 mmol/L); or (2) muscimol, a GABAA receptor agonist (1 to 100 μmol/L). A second series of experiments was performed in which baclofen, a GABAB receptor agonist (10 nmol/L to 100 μmol/L), was added directly to the superfusing medium without preconstriction. Previous studies using in vitro slice preparations suggest that these GABAergic agents are pharmacologically effective and selective within the concentration ranges examined (e.g., Okada and Ozawa, 1982; Newberry and Nicoll, 1985; Xie and Tietz, 1992). Based on the dose–response curves generated from these experiments (Fig. 1), doses eliciting asymptotic responses were chosen for the next series of experiments.
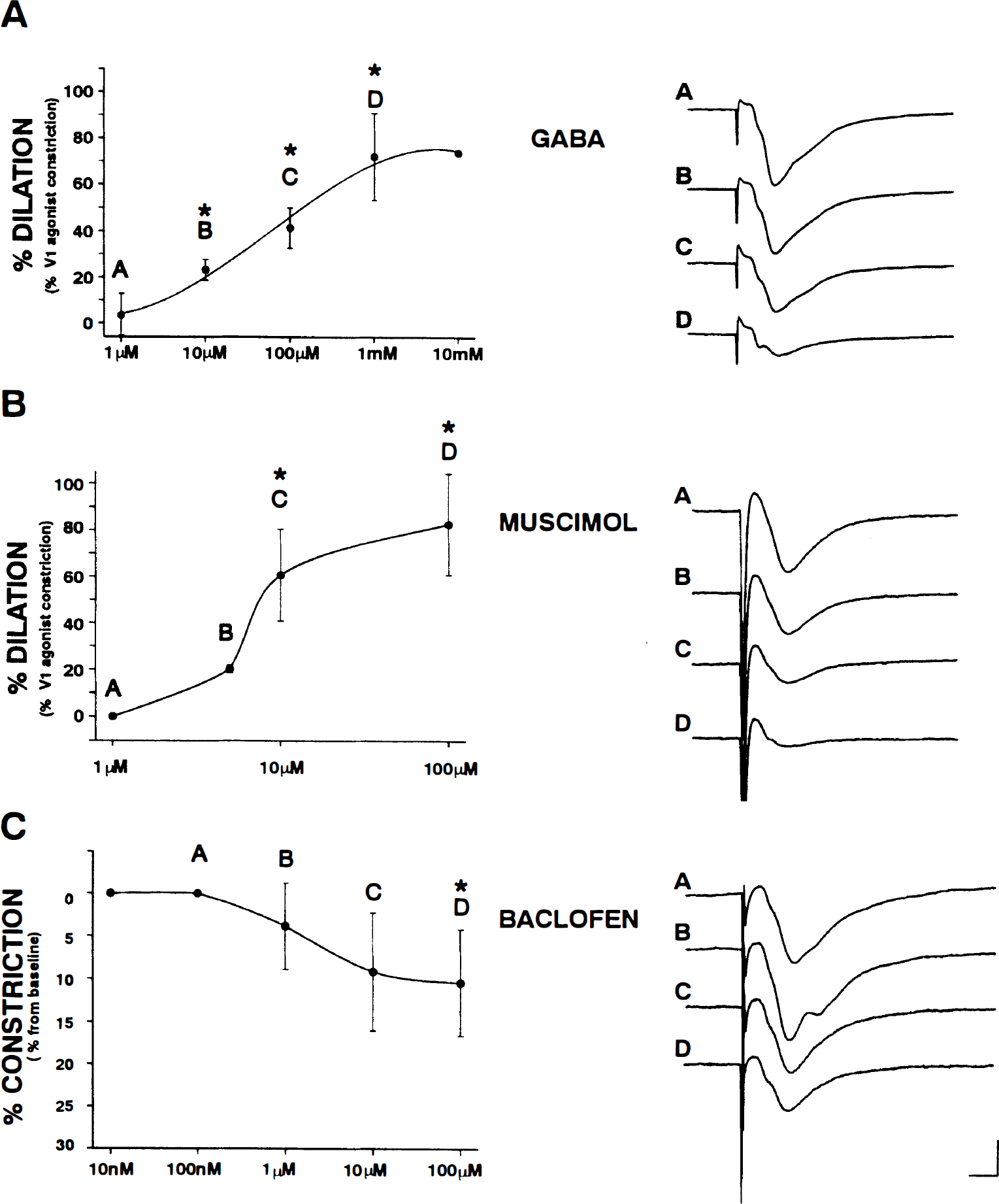
GABA-mediated microvascular responses in the hippocampus.
The role of neuronal discharge activity in the responses mediated by GABA receptors was examined by treating hippocampal slices with tetrodotoxin (TTX, 1 μmol/L) 15 minutes before the addition of a selective GABA receptor agonist (i.e., 100 μmol/L muscimol or 100 μmol/L baclofen) to the superfusing medium. At this concentration, TTX abolishes neuronal action potential generation completely in the slice preparation, whereas it is not sufficient to block Na+ channels in vascular smooth muscle cells (Sturek and Hermsmeyer, 1986). In addition, astrocytes do not appear to contain TTX-sensitive sodium channels in situ (Sontheimer and Waxman, 1993; Kressin et al., 1995). This concentration of TTX also is comparable with that used in previous in vitro slice studies (e.g., Soltesz and Mody, 1994; Newberry and Nicoll, 1995). To evaluate the role of NO in the vasodilatory response to muscimol, hippocampal slices were treated with the nitric oxide synthase inhibitor, N-nitro-
The role of endogenous GABA receptor activation in the regulation of resting tone was tested by treating hippocampal slices with cumulative ascending concentrations of the competitive GABAA receptor antagonist bicuculline methiodide (bicuculline; 1 to 200 μmol/L), the noncompetitive GABAA receptor antagonist picrotoxin (1 to 200 μmol/L), or the competitive GABAB receptor antagonist 2-hydroxysaclofen (1 to 200 μmol/L). Previous studies using in vitro slice preparations suggest that these GABAergic agents are pharmacologically effective and selective within the concentration ranges examined (e.g., Newberry and Nicoll, 1985; Davies et al., 1990). The concentration of bicuculline eliciting an asymptotic response (based on the dose–response curve generated, Fig. 2) was selected for the remaining experiments. In these experiments, hippocampal slices were treated with TTX (1 μmol/L) before the addition of bicuculline (100 μmol/L). A series of experiments then was performed in which hippocampal slices were first treated either with 100 μmol/L of
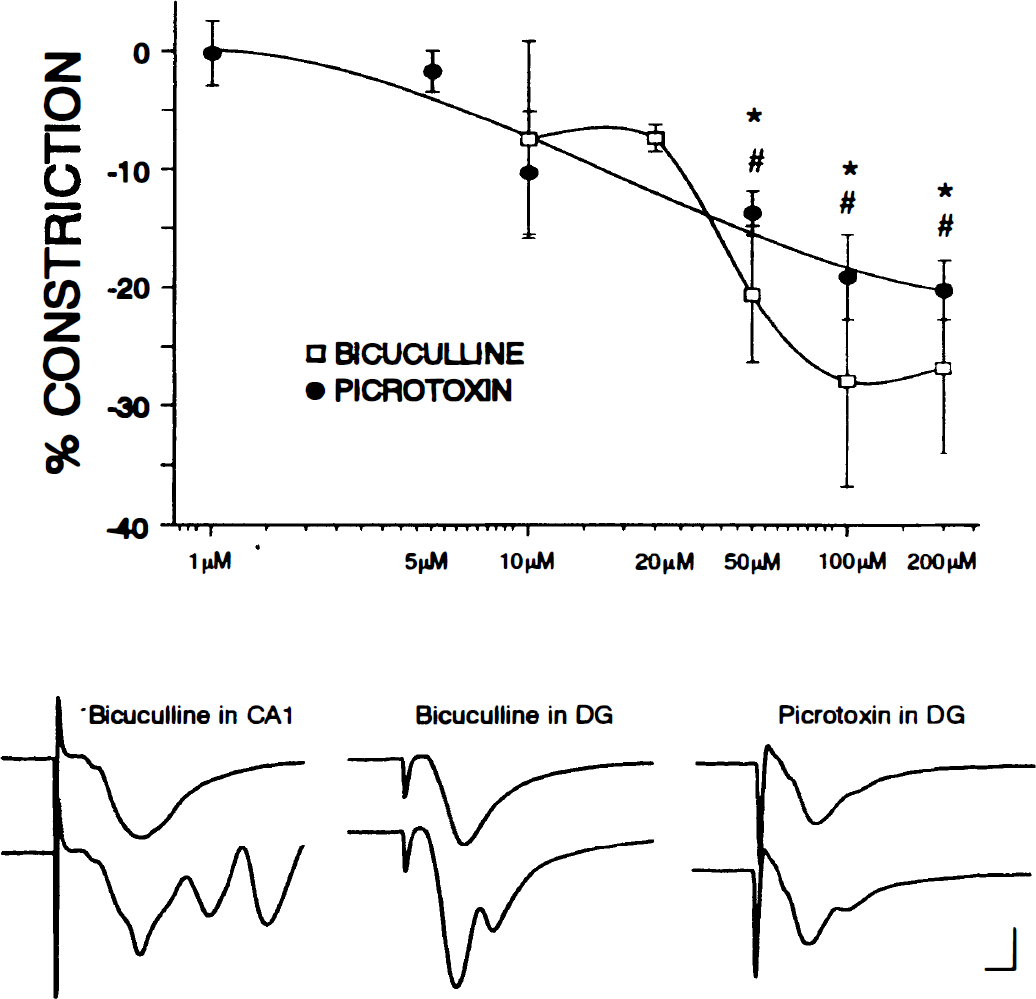
GABAA receptor antagonists elicit vasoconstriction.
The role of GABA in hypoxia-induced vasodilation also was examined. In these experiments, vessels in hippocampal slices were preconstricted with either V1 ag (25 nmol/L) or bicuculline (100 μmol/L). After a stable constriction was established, hypoxic conditions were achieved by replacing the 95%/5% oxygen/carbon dioxide, which bubbled through the superfusing medium with 95%/5% nitrogen/carbon dioxide for 60 minutes; this reduced the oxygen tension in the superfusing medium from approximately 500 to approximately 60 mm Hg.
A final series of experiments examined the role of GABAergic signaling in surface arterioles of the neocortex. The resting luminal diameter of the cortical vessels ranged from 30 to 50 μmol/L. In one set of experiments, vessels on the surface of the neocortex were preconstricted with V1 ag (25 nmol/L) and then treated with 100 μmol/L of muscimol. In other experiments, resting (i.e., not preconstricted) cortical vessels were treated cumulatively with ascending concentrations of bicuculline (100 to 200 μmol/L) or 2-hydroxysaclofen (100 to 200 μmol/L).
Chemicals
The aCSF consisted of 124.0 mmol/L NaCl, 25.7 mmol/L NaHCO3, 3.3 mmol/L KCl, 1.3 mmol/L KH2PO4, 2.4 mmol/L MgSO4, 2.0 mmol/L CaCl2, and 10.0 mmol/L glucose. Bicuculline methiodide, baclofen, muscimol, L-NNA, TTX, and GABA were obtained from Sigma Chemical Co (St. Louis, MO, U.S.A.). Vasopressin [Phe2, Ile3, Orn8] was obtained from American Peptide Co. (Sunnyvale, CA, U.S.A.) and 2-hydroxysaclofen was obtained from Research Biochemicals Int. (Natick, MA, U.S.A.).
Statistical analyses
One-way analysis of variance with Bonferroni post-hoc analyses, paired and unpaired Student's t-tests, and repeated measures one-way analysis of variance were used for statistical analyses. Statistical significance was taken at the P = 0.05 level. Values are depicted as mean ± standard deviation.
RESULTS
The effects of GABA receptor agonists on hippocampal microvessels
V1 ag (25 nmol/L) constricted hippocampal microvessels by 30.2 ± 8.8% (mean ± standard deviation) from baseline (n = 25). The onset of this response was rapid, the constriction stabilized after approximately 5 minutes, and it was maintained for at least 90 minutes in the continued presence of the agonist (Fig. 3). In addition, no obvious alterations in the neuronal field response were observed during treatment with V1 ag (Fig. 3).
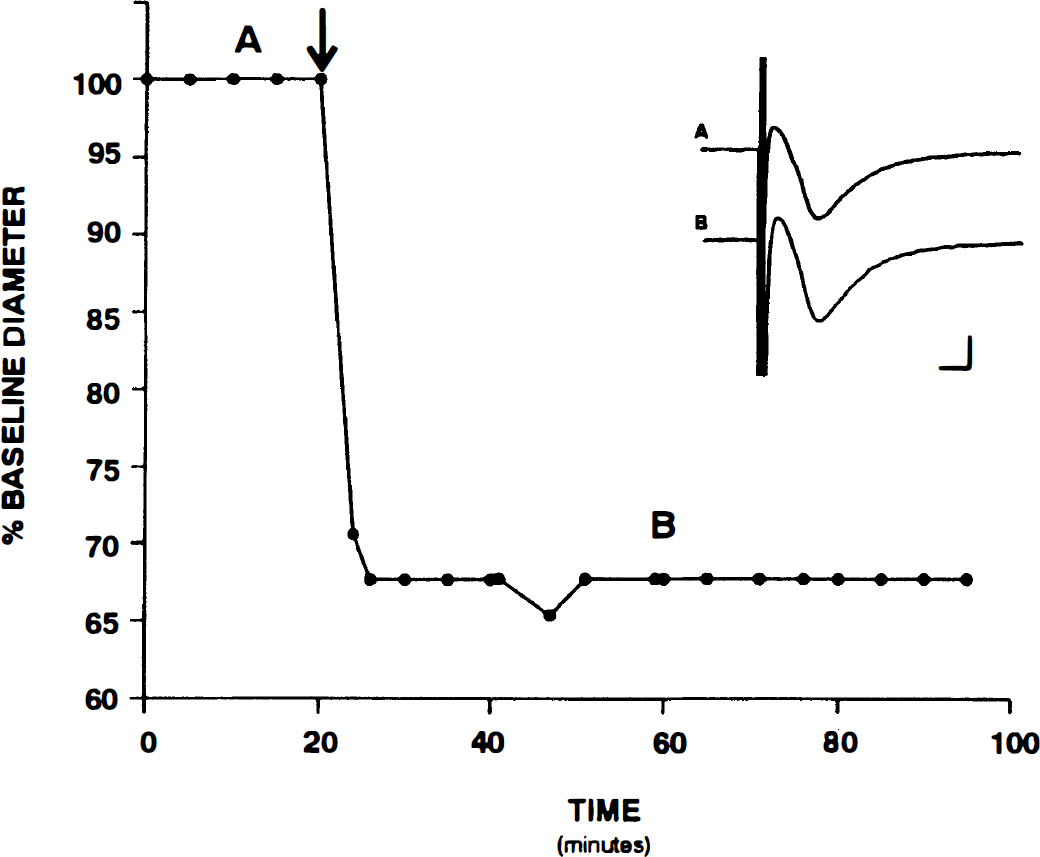
V1-mediated microvascular constriction in the hippocampus. A representative vascular response to a prolonged treatment with vasopressin [Phe2, Ile3, Orn8] (25 nmol/L) is shown.
GABA elicited a dose-dependent dilation of hippocampal microvessels preconstricted with V1 ag (n = 4; Fig. 1A). The average maximal dilatory response to the cumulative application of GABA was 62.0 ± 18.7% (n = 4) and was observed at a concentration of 1 mmol/L. Electrophysiologic recordings from the same slices demonstrated that GABA induces a dose-dependent suppression of the neuronal field response (Fig. 1A); this effect is in keeping with a substantial literature on the electrophysiologic effects of GABA, suggesting that pharmacologically effective and selective concentrations were used (Okada and Ozawa, 1982). Muscimol, a GABAA receptor agonist, also elicited a dose-dependent vasodilation in hippocampal microvessels (n = 4; Fig. 1B). Application of cumulative ascending concentrations of muscimol resulted in a maximal response around 100 μmol/L; the average maximal response was 82.6 ± 19.7% (n = 4). The slope of the dose-response curve for muscimol was steeper than that observed with GABA, but the difference between the average maximal dilatory responses to muscimol and GABA did not achieve statistical significance. Dose-dependent suppression of the neuronal field response also was observed with muscimol treatment in the hippocampus (Fig. 1B); this effect is consistent with previous electrophysiologic findings in the hippocampus (Zinebi et al., 1988). Taken together, these findings indicate that GABA elicits hippocampal vasodilation and that this effect is mediated by GABAA receptors.
Baclofen, a GABAB receptor agonist, elicited a dose-dependent constriction from baseline (Fig. 1C). The cumulative application of ascending concentrations of baclofen resulted in an average maximal constriction of 10.4 ± 6.2% (n = 4), and this was achieved at a concentration of 100 μmol/L. These observations indicate that, in addition to its vasodilatory actions at GABAA receptors, GABA is capable of eliciting vasoconstriction through an action at GABAB receptors. Baclofen also induced complex alterations in the neuronal field response. A small increase in neuronal excitability was occasionally elicited at lower doses, as evidenced by population spike contamination in the fEPSP. However, a substantial suppression of the field response was consistently elicited at higher doses. These electrophysiologic observations are consistent with previous reports of complex effects of GABAB receptor activation on neuronal responses (Davies et al., 1990; Steffensen and Henriksen, 1991; Otis and Mody, 1992; Morishita and Sastry, 1994).
The role of neuronal discharge activity in GABA receptor–mediated responses
The sodium channel blocker, TTX, was used to determine whether the microvascular responses to GABA receptor activation are dependent on the generation of action potentials (discharge activity) in the hippocampus. Muscimol (100 μmol/L) dilated preconstricted vessels by 74.1 ± 10.9% (n = 6) in the presence of TTX, and by 80.1 ± 16.1% (n = 5) in the absence of TTX (Fig. 4). The muscimol-induced dilation in the presence and absence of TTX did not differ significantly. These data indicate that the microvascular effects of GABA at GABAA receptors are independent of a GABAA-mediated suppression of neuronal discharge activity.
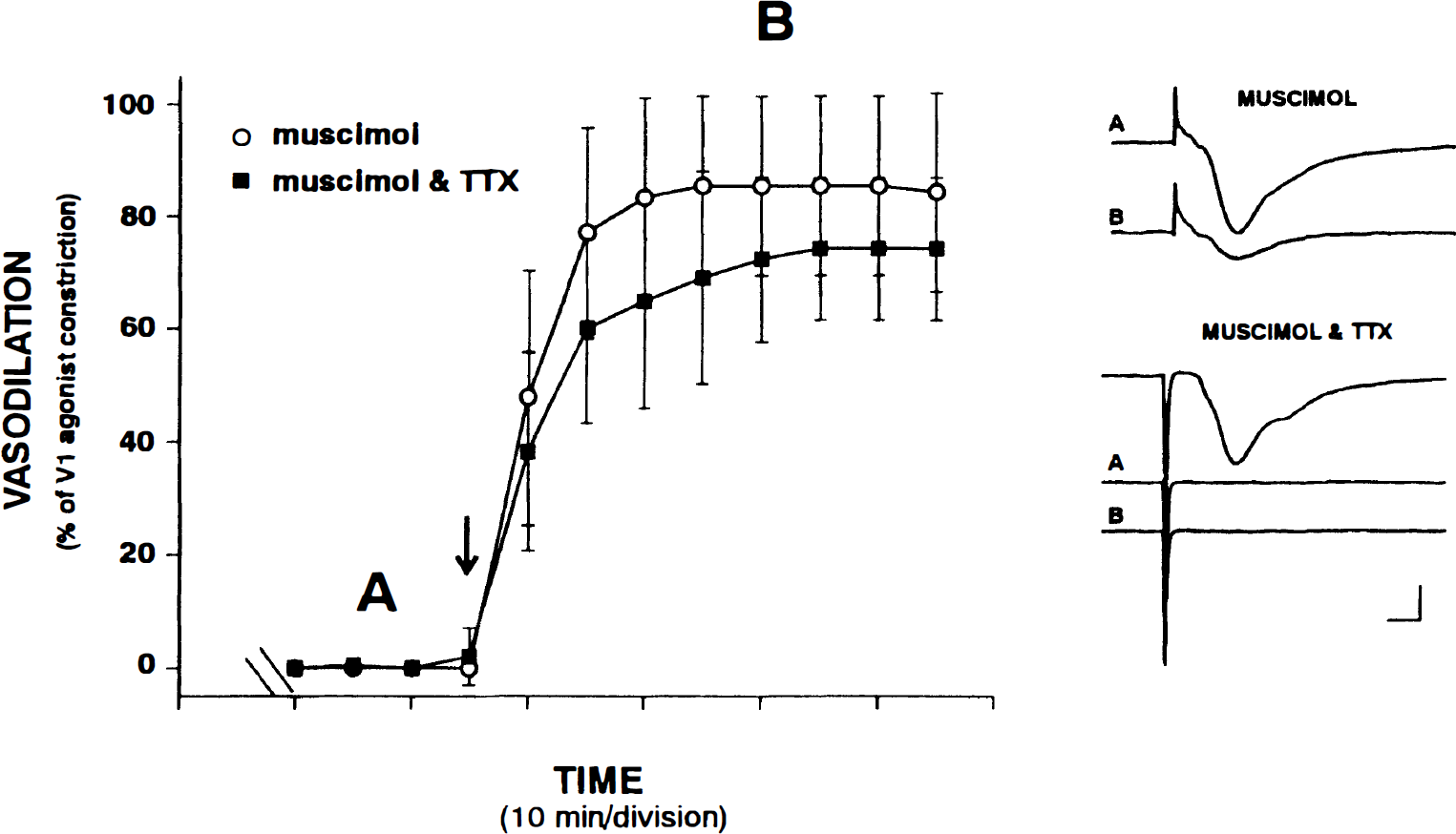
Muscimol-induced dilation is not activity-dependent.
In contrast, alterations in neuronal discharge activity are necessary for the microvascular effects of baclofen. In a matched set of experiments in which a single concentration of baclofen was tested, baclofen (100 μmol/L) elicited a 9.0 ± 1.5% constriction of hippocampal microvessels (n = 5; P = 0.002). The microvascular response to baclofen was blocked completely by treatment with TTX; in the presence of TTX, 100 μmol/L baclofen constricted microvessels by 0.1 ± 1.2% (n = 4). The blockade of baclofen-induced constriction by TTX was highly significant (P < 0.001).
The role of nitric oxide in GABAA-mediated dilation
To investigate whether NO acts as an intermediary in the GABAA receptor-mediated dilation of hippocampal microvessels, nitric oxide synthase activity was inhibited with
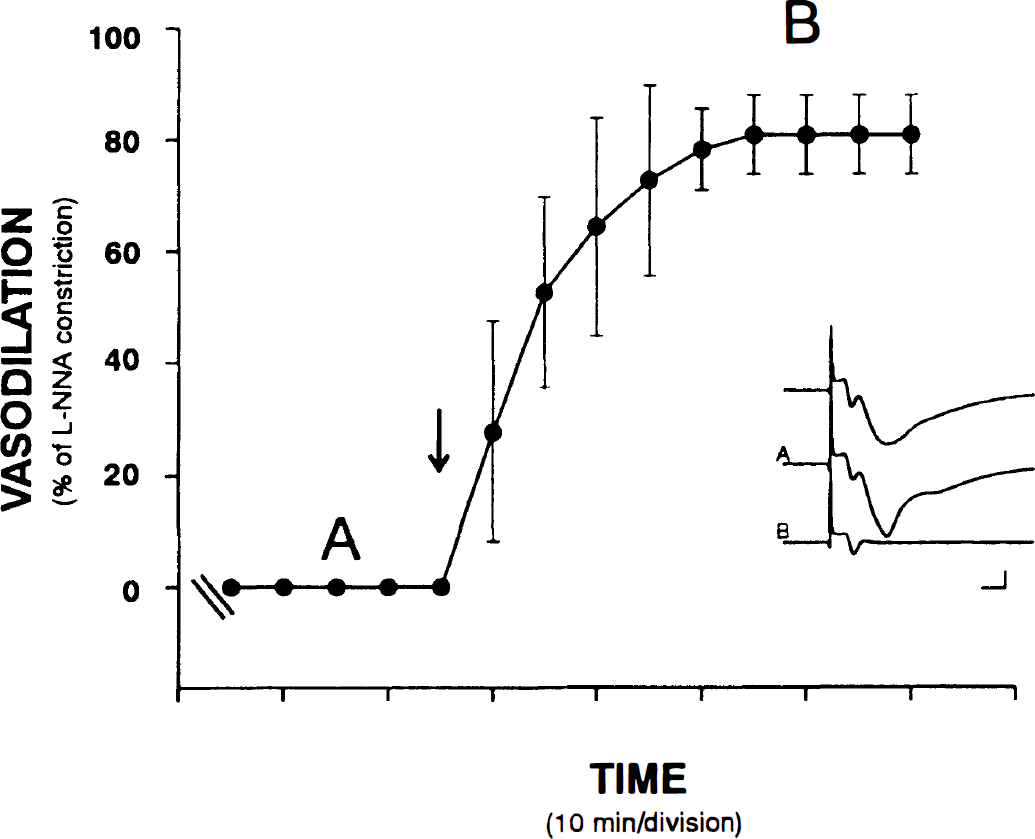
Nitric oxide is not an intermediary in muscimol-induced dilation. A dose of 100 μmol/L of muscimol elicits a significant dilation in the presence of the nitric oxide synthase inhibitor, 100 μmol/L of N-nitro-
The role of endogenous activation of GABA receptors in the resting state of parenchymal microvessels
The possible contribution of endogenous GABA to the resting tone of parenchymal microvessels was examined. The GABAA receptor antagonists bicuculline and picrotoxin induced dose-dependent constrictions in hippocampal microvessels. The dose–response curve to bicuculline, a competitive GABAA antagonist, was narrow compared with the dose–response curve for the noncompetitive GABAA receptor antagonist, picrotoxin (Fig. 2). The response to bicuculline reached a plateau around 100 μmol/L (n = 4). At 100 μmol/L, bicuculline induced a 27.9 ± 9.8% constriction from baseline. The average maximal response to picrotoxin was 21.7 ± 2.5% and was obtained at a concentration of approximately 200 μmol/L. Both bicuculline and picrotoxin elicited dose-dependent increases in the excitability of the neurons within the slice; population spikes were reflected in the field responses in both CA1 and the dentate gyrus (Fig. 2). However, epileptiform activity (as noted by spontaneous bursting activity and transient loss of the fEPSP) was not observed at doses up to and including 100 μmol/L. Epileptiform activity was present in one of the slices treated with 200 μmol/L of bicuculline. Dilation was observed with the onset of epileptiform activity, and this experiment was excluded from the quantitative analyses performed in this study.
In contrast to the effects of blocking GABAA receptors, the antagonism of GABAB receptors with 2-hydroxysaclofen did not elicit a significant vascular response at any of the concentrations examined (1 to 200 μmol/L; n = 5). 2-Hydroxysaclofen, within this concentration range, has been shown to effectively block GABAB-mediated responses (Davies et al., 1990; Diesz et al., 1993). In the current study, 2-hydroxysaclofen increased the excitability of the neurons within the slice, as evidenced by population spike contamination in the dendritic response (data not shown).
The role of neuronal discharge activity in endogenous GABAA-mediated dilatory tone
Application of 1 μmol/L TTX abolished the fEPSP completely but did not elicit a significant vascular response (n = 5). Furthermore, TTX did not significantly attenuate the bicuculline-induced microvascular constriction. Bicuculline (100 μmol/L) elicited a 26.7 ± 9.4% constriction (n = 15) in the absence of TTX and 23.5 ± 3.8% constriction (n = 5) in the presence of TTX. The time courses of the constrictions observed in the presence and absence of TTX did not differ significantly. Constriction was initiated within 1 minute of the time bicuculline was added to the superfusing medium and it reached a plateau after approximately 20 minutes. The time courses of the bicuculline-induced constriction in the presence and absence of TTX are shown in Fig. 6.
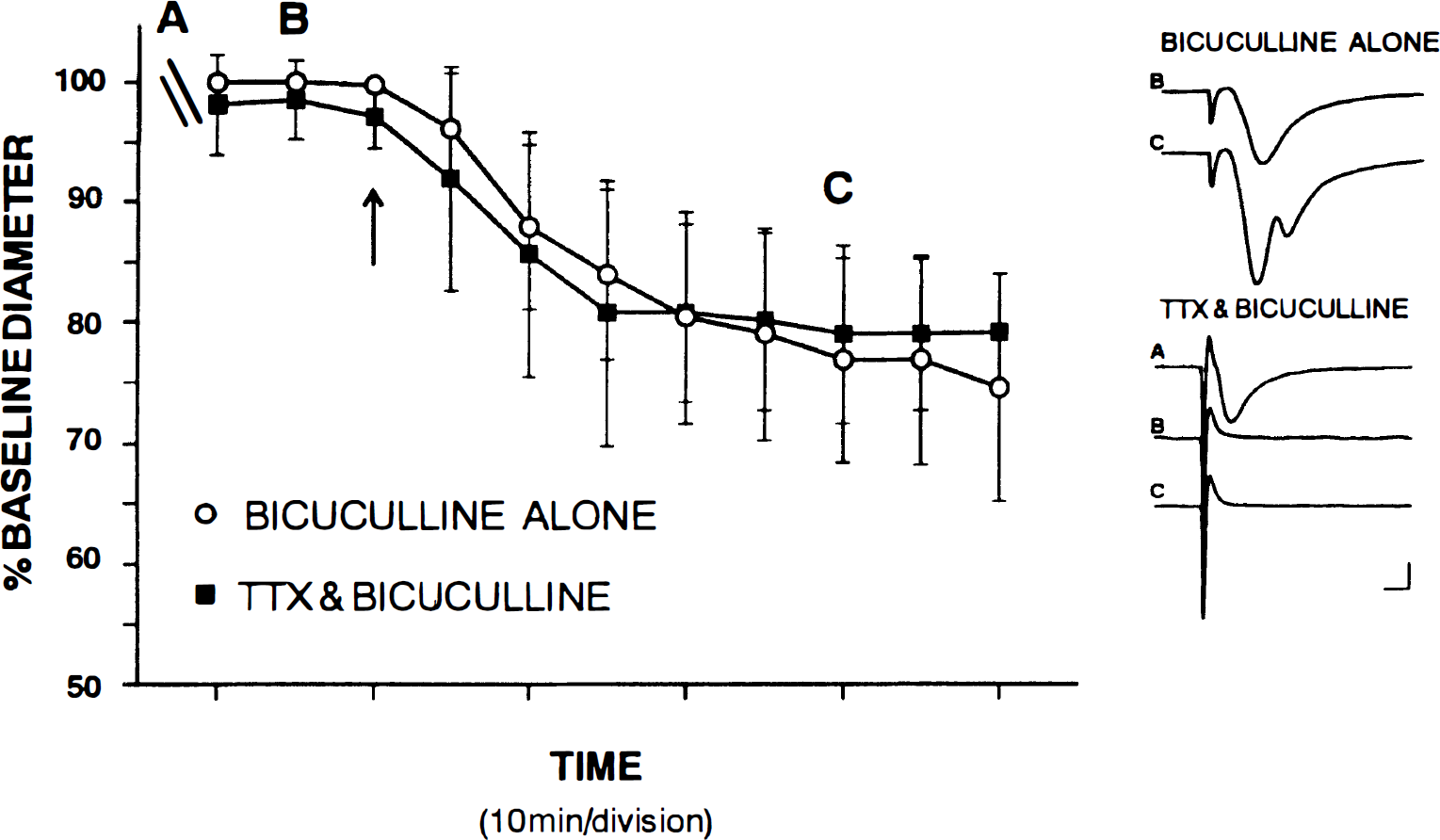
GABAA-mediated effects on resting tone are not activity dependent.
The combined effects of endogenous nitric oxide and GABA on resting tone
As described earlier, treatment with 100 μmol/L of
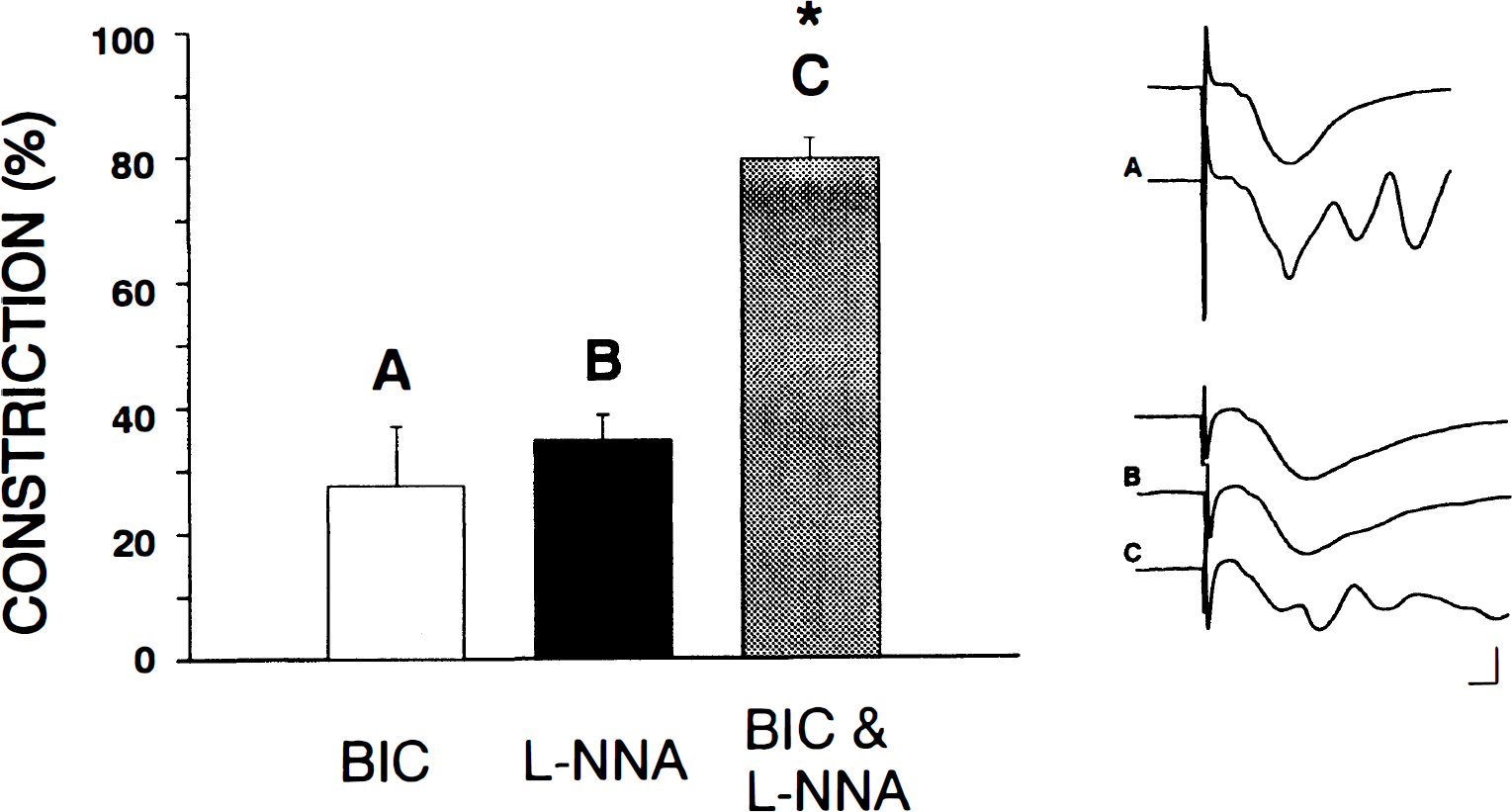
Constrictions induced by N-Nitro-
The role of GABA in hypoxia-induced microvascular dilation
The possible role of endogenous GABAergic mechanisms in hypoxia-induced vasodilation was examined. In slices treated with V1 ag, hypoxia induced a maximal dilation of 64.4 ± 16.9% (n = 6; P = 0.002). In the presence of bicuculline, the maximal dilatory response was 58.4 ± 25.4% (n = 7; P = 0.02). The difference between the hypoxic dilation in the presence of V1 ag or bicuculline did not differ significantly. Synaptic responses were lost during hypoxia in the presence of either V1 ag or bicuculline (Fig. 8). During reoxygenation, vascular tone recovered to a level that was usually stronger than the prehypoxic “constricted baseline.” Synaptic responses also recovered partially during reoxygenation.
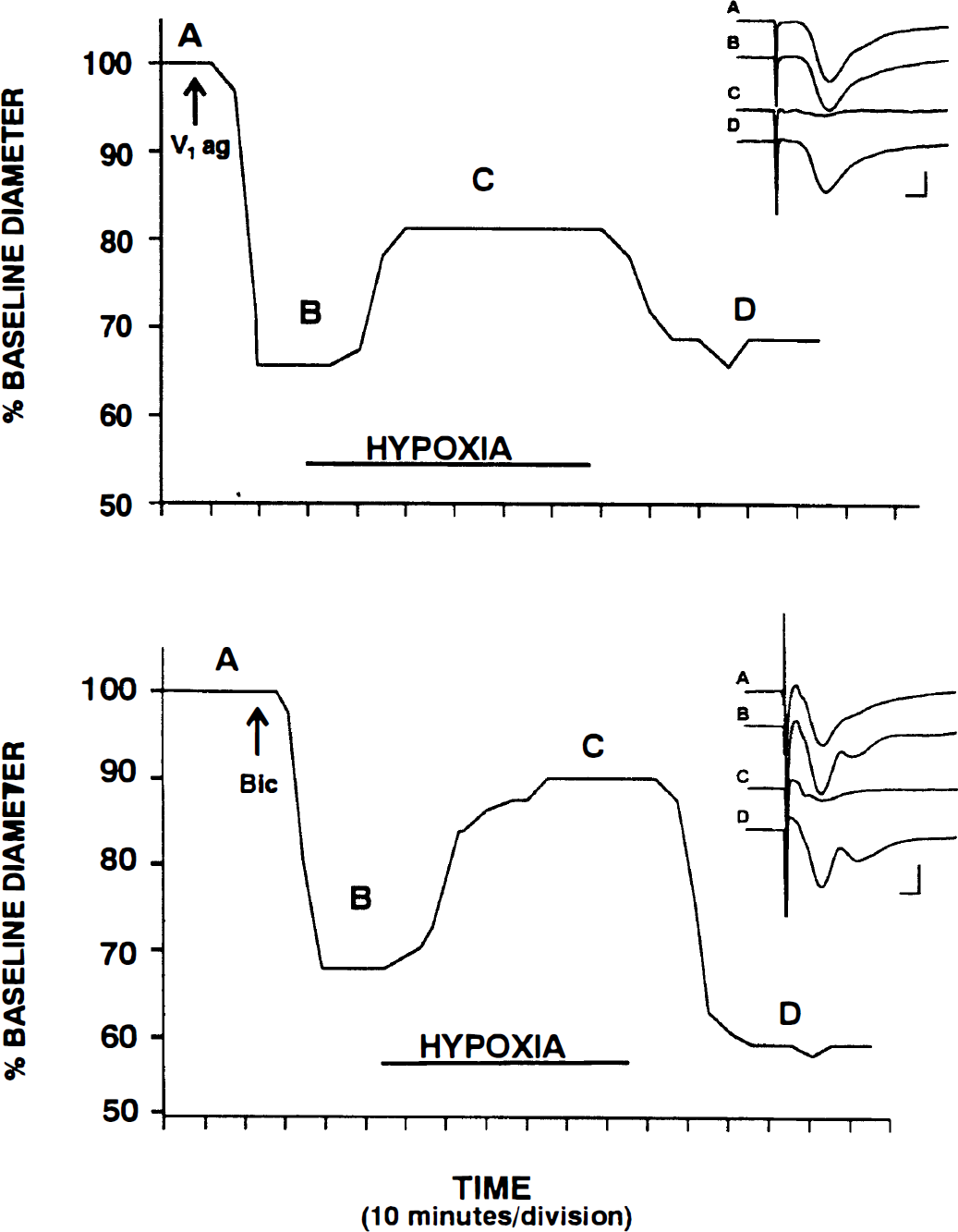
GABAA receptor activation is not involved in hypoxic vasodilation. Representative vascular responses to moderate hypoxia are shown in the presence of 25 nmol/L of V1 ag (
GABAergic signaling in cortical surface vessels
Selective activation of GABAA receptors resulted in vasodilation in cortical surface vessels preconstricted with V1 ag. V1 ag (25 nmol/L) elicited a 52.7 ± 17.4% (n = 4) constriction in cortical surface vessels with a time course similar to that in hippocampal vessels. Muscimol (100 μmol/L) elicited a significant dilatory response in the preconstricted cortical vessels (n = 4; 51.5 ± 8.5%; P = 0.02).
GABA receptor antagonists did not induce changes in the luminal diameter of resting cortical surface vessels. Specifically, bicuculline (100 or 200 μmol/L) did not elicit significant changes in vessel diameter (n = 5, −4.0 ± 9.6% for 100 μmol/L; n = 5, −3.3 ± 10.0% for 200 μmol/L). In addition, 2-hydroxysaclofen did not elicit a significant response in the surface vessels of the neocortex (n = 3, −0.1 ± 0.8% for 100 μmol/L; n = 4, −1.0 ± 1.5% for 200 μmol/L).
DISCUSSION
The signaling mechanisms responsible for regulating parenchymal microvessels in the brain are just beginning to be understood. The current study indicates a role for the inhibitory neurotransmitter, GABA, in this process. The findings demonstrate that both exogenous and endogenous activation of GABA receptors affect cerebral microvascular tone. In addition, the GABAergic influence on cerebral microvessels is heterogeneous. In the parenchymal microvessels of the hippocampus, application of exogenous GABA elicits a net vasodilation. Selective activation of GABAA receptors also results in dilation, suggesting that the GABAergic dilation is mediated by GABAA receptors. In contrast, activation of GABAB receptors in the hippocampus results in microvascular constriction. Although GABAB-mediated contraction has been reported in peripheral smooth muscle (Pencheva et al., 1991), this is the first evidence for cerebrovascular constriction resulting from GABAB receptor activation. The dilatory response in vessels on the surface of the neocortex to selective GABAA receptor activation is consistent with previous studies examining pial vessels and larger cerebral blood vessels (Fujiwara et al., 1975; Edvinsson and Krause, 1979). The slightly higher concentrations used in the current study may have been necessary because of the presence of neurons and glia in the slice, which also are influenced by GABAergic agents. In contrast to the current findings and observations in larger cerebral vessels (Fujiwara et al., 1975; Edvinsson and Krause, 1979), GABA does not dilate cerebral microvessels in isolation (Takayasu and Dacey, 1989). This may reflect a role for glial cells that remain intact in the slice preparation and not in the isolated vessel preparation. Alternatively, regional heterogeneity of vascular reactivity may account for the differences seen. Takayasu and Dacey (1989) examined segments from the first (M1) portion of the middle cerebral artery. Regional heterogeneity has been described for cerebrovascular responsiveness to other neurotransmitters and blood-borne substances (Hamel et al., 1988).
The findings from this study also indicate that GABAA receptor-mediated dilation in hippocampal microvessels does not depend on NO signaling or action potential discharge in local neurons. Although action potential–independent neuronal effects cannot be excluded, these features are consistent with a direct action of GABA on microvascular or glial elements. The presence of GABAergic relaxation in isolated vessel preparations (Fujiwara et al., 1985; Edvinsson and Krause, 1979) also supports a nonneuronal mechanism for the GABAA-mediated responses observed in the current study. The existence of GABA receptors on hippocampal microvessels has not been examined; however, GABAA receptors have been identified on the walls of larger cerebral blood vessels, suggesting that the responses observed could be caused by a direct action on the microvascular wall (Krause et al., 1980a, b ; Napoleone et al., 1987). GABAA receptors also have been identified electrophysiologically and immunohistochemically on hippocampal astrocytes (Fraser et al., 1994; Fraser et al., 1995), and it is possible that astrocytes act as cellular intermediaries in the GABAA-mediated microvascular responses. In contrast to GABAA receptor-mediated dilation, the GABAB receptor-mediated (i.e., baclofen-induced) constriction is dependent on the conduction of neuronal action potentials. These data indicate that baclofen elicits its vascular effects through a neuronal intermediary. This is consistent with the apparent absence of GABAB receptors on cerebral blood vessels (Napoleone et al., 1990).
The overall effects of GABA on CBF are unclear from the existing literature. The findings from this study are consistent with reports of increased total CBF after application of GABA receptor agonists (Edvinsson et al., 1980; Alborch et al., 1984). However, other studies observed decreases in cerebral metabolism and local CBF after transient systemic administration of muscimol (Kelly and McCulloch, 1983; Kelly et al., 1989). In the current study, continuous treatment with GABA or muscimol resulted in sustained microvascular dilation with simultaneous suppression of the neuronal field response. These findings, along with the observation that the dilatory response to muscimol is TTX-insensitive, suggest that the vasodilatory effects of GABA occur independently of short-term effects on neuronal action potential generation. In contrast, the TTX-sensitivity of the baclofen-induced constriction may reflect an indirect vascular response to the suppression of neuronal activity through neuronal GABAB receptors. The GABA-mediated decrease in blood flow reported in previous studies may reflect such an indirect action. The current findings support the hypothesis that sustained activation of vascular GABAA receptors would override the metabolic effects of GABA on microvascular tone.
The current study also provides the first evidence that the activation of GABA receptors by endogenous GABA plays a role in the regulation of microvascular tone. GABAA receptor antagonists constrict hippocampal microvessels, suggesting that spontaneous activation of GABAA receptors by endogenous GABA modulates the resting state of parenchymal vessels. This finding is consistent with the high level of spontaneous GABA release from interneurons even in the presence of TTX (Otis and Mody, 1992). In contrast, resting tone in vessels located on the surface of the brain does not appear to be regulated by the endogenous activation of GABA receptors. Neither GABAA nor GABAB receptor antagonists elicit significant vascular responses in surface arterioles. These data corroborate previous observations of the responses of these vessels in situ (McCulloch et al., 1981) and basilar and Circle of Willis arteries in vitro (Edvinsson and Krause, 1979). Taken together, these results indicate that a wide range of cerebral vessels respond to pharmacologic application of GABA, but that tonic vascular regulation by endogenous GABA under basal physiologic conditions may be unique to parenchymal microvessels. The small caliber of the microvessels is one variable that may contribute to this difference in tonic GABAergic regulation. On the other hand, the existence of GABAergic tone in parenchymal microvessels may be a reflection of their location within the brain. Microvessels are embedded in neuropil, and interneurons frequently are associated with these microvessels (Schottler et al., 1996). Furthermore, the concentration of GABA varies regionally in the brain with the hippocampus exhibiting very high levels (Goodchild, 1993). Parenchymal microvessels of the hippocampus are therefore in a position to appraise the GABA released into the neuropil from GABAergic interneurons. In contrast, neocortical surface vessels are not embedded in GABA-generating neuropil to the same extent as the parenchymal microvessels and may not be subjected to high GABA levels under physiologic conditions. The overall densities of GABA receptors do not differ dramatically between hippocampal and cortical regions (Chu et al., 1990). Nevertheless, the presence of GABA receptors specifically on hippocampal microvessels has yet to be examined, and it is conceivable that the density of GABAA receptors on these vessels is greater than that observed on the larger surface vessels (Napoleone et al., 1987). In addition, the possibility exists that GABA produced within the walls of the cerebral microvessels themselves acts as an endogenous source of GABA. Glutamate decarboxylase activity, which is necessary for GABA production, has been demonstrated within the walls of cerebral blood vessels (Hamel et al., 1981; Gragera et al., 1993). Glutamate decarboxylase–positive nerve fibers have been identified within the walls of cerebral blood vessels (Okuno et al., 1994). These nerve fibers are in an ideal position to exert GABAergic influence on microvascular tone. Regardless of the local sources of GABA, the current data provide direct evidence of the existence of regional variability of GABAergic signaling in cerebral blood vessels.
Both NO and GABA participate in the regulation of basal tone in parenchymal microvessels. The additive effects of bicuculline and
This study also provides direct evidence that microvessels in the hippocampus dilate in response to hypoxia. This response is consistent with physiologic responses of other cerebral blood vessels in vitro and in vivo (e.g., Kontos et al., 1978; Gebremedhin et al., 1994). The failure of GABAA receptor antagonism to block hypoxic vasodilation indicates that endogenous activation of GABAA receptors does not mediate this response. Nevertheless, the findings from the current studies indicate that the use of GABAergic agents as a therapeutic strategy would not only have effects on neurons but also on nearby blood vessels, which must be considered. The current findings may therefore have far reaching implications for therapies targeting GABAergic systems. In addition, it is conceivable that endogenous GABA participates in vascular responses to stimuli such as increased neuronal activation. Although GABA does not participate in the CBF response to parallel fiber stimulation in the cerebellum (Li and Iadecola, 1994; Akgören et al., 1996), regional differences may exist in the role of GABAergic signaling of cerebral blood vessels. Furthermore, the role of GABAergic signaling may depend on the type and extent of neuronal stimulation.
The current study also underscores the utility of the brain slice preparation in the examination of cerebral microvessels from deep brain structures. Although a limitation of the brain slice preparation (and most in vitro vascular preparations) is the lack of intraluminal flow and pressure resulting in a decrease in myogenic tone, the responses of blood vessels in this preparation parallel those observed in vivo (see also Sagher et al., 1993; Fergus et al., 1995 and 1996). The physiologic response to hypoxia of microvessels described here provides further support for the use of brain slices and for their responsiveness to physiologic stimuli. Furthermore, the slice preparation allows direct visualization of parenchymal microvessels within their normal microenvironment, which is not otherwise possible. Potential signaling mechanisms from neuronal and glial processes remain intact in this preparation and regional neuronal responses can be monitored. These features support the utility of the slice preparation for evaluating cerebral microvascular function and may ultimately allow the identification and characterization of local neurovascular communication in the parenchyma of the brain.
In summary, these studies provide the first evidence for GABAergic signaling of parenchymal microvessels in the hippocampus. These studies demonstrate the participation of GABAA receptor activation in the modulation of resting tone in cerebral blood vessels. This may represent a means through which the resting brain couples the level of neuronal activity to an appropriate level of local CBF. Tonic inhibitory pathways are prevalent throughout the brain and the characteristics of inhibitory neurons put them in an ideal position to not only modulate the level of neuronal excitation in a particular region, but also to modulate local CBF by signaling nearby supply vessels. The roles of GABA in the regulation of microvascular tone and in neuronal signaling suggest that GABA is in a suitable position to serve as a messenger in the coupling of neuronal activity to local CBF under resting conditions and conditions of enhanced neuronal activity.
