Abstract
Endothelial cells and pericytes are closely associated in brain capillaries. Together with astrocytic foot processes, they form the blood–brain barrier. Capillaries were isolated from bovine brain cortex. Pure populations of endothelial cells and pericytes were isolated and cultured in vitro. Polarized monolayers of endothelial cells preferentially secreted immunoreactive endothelin-1 (Et-1) at their abluminal (brain-facing) membrane. They did not express receptors for Et-1. Pericytes expressed BQ-123-sensitive ETA receptors for endothelins as evidenced by 125I-Et-1 binding experiments. These receptors were coupled to phospholipase C as demonstrated by intracellular calcium measurements using indo-1-loaded cells. Addition of Et-1 to pericytes induced marked changes in the cell morphology that were associated with a reorganization of F-actin and intermediate filaments. It is concluded that Et-1 is a paracrine mediator at the bovine blood–brain barrier and that capillary pericytes are target cells for endothelium-derived Et-1.
Brain microvessels form the anatomical and functional basis of the blood–brain barrier. They are composed of vascular endothelial cells linked together by tight junctions, by pericytes, and by astrocytic foot processes. Pericytes are adventitial cells of mesodermal origin that are embedded in the abluminal basal lamina of endothelial cells. Their functions are still unclear, although there is evidence that they control proliferation of endothelial cells, the integrity of capillaries, transport, and phagocytic processes (Broadwell and Salcman, 1981; Sims, 1986; Farrell et al., 1987; Antonelli-Orlidge et al., 1989; Shepro and Morel, 1993). Expression of α smooth muscle actinand extensive studies performed on retinal pericytes and renal mesangial cells have also suggested that pericytes have contractile properties similar to those of smooth muscle cells in large vessels. It is, however, recognized that pericytes have organ-specific functions and that their properties are different in different tissues (Shepro and Morel, 1993). The properties and functions of pericytes from brain cortex capillaries are largely unknown.
The contractility of vascular smooth muscle cells is regulated by endothelium-derived relaxing factors such as nitric oxide (Moncada et al., 1991) and contracting factors such as endothelin-1 (Et-1) (Yanagisawa et al., 1988). Et-1 acts in both autocrine and paracrine manners. It recognizes ETB receptors on macrovascular endothelial cells and induces NO-mediated vasorelaxations. It also recognizes ETA receptors on smooth muscle cells and induces long-lasting and almost irreversible vasoconstrictions (Rubanyi and Parker Botelho, 1991). Increasing evidence suggests that vasoactive factors are present at the blood–brain barrier (Frelin and Vigne, 1993). Their functions in capillaries that lack smooth muscle cells remain, however, to be elucidated.
In this article, we analyze the properties of endothelial cells and pericytes isolated from bovine brain capillaries. The results indicate that Et-1 is a likely mediator of endothelial cell–pericyte interactions in brain capillaries.
MATERIALS AND METHODS
Materials
Peptides and biochemical reagents were from the Sigma Chemical Co. unless otherwise indicated.
Cell cultures
Capillaries were isolated from bovine brain cortex according to Méresse et al. (1989). Primary cultures consisting of mixtures of pericytes and endothelial cells were prepared. Brain capillary endothelial cells (BCEC) were cloned as previously described (Méresse et al., 1989) and grown in Dulbecco's Modified Eagle Medium (DMEM) supplemented with 10% (vol/vol) heat-inactivated calf serum and 10% (vol/vol) horse serum (Hyclone Laboratories, Logan, UT, U.S.A.), 2 mM glutamine, 50 μg/ml gentamycin, and basic fibroblast growth factor (1 ng/ml added every other day).
Primary cultures were rapidly overgrown by pericytes. Confluent cultures, which consisted almost exclusively of pericytes, were dissociated with a trypsin/ethylenediaminetetraacetate (EDTA) saline solution [0.05% (wt/vol) trypsin, 0.02% (wt/vol) EDTA in Ca2+- and Mg2+-free phosphate-buffered saline (TE)], and cells were frozen in liquid nitrogen (one 60-mm Petri dish per vial). For experiments, cells were rapidly thawed and grown in DMEM supplemented with 10% (vol/vol) calf serum and 10% (vol/vol) horse serum, 2 mM glutamine, and 50 μg/ml gentamycin. Pericytes were subcultured at a split ratio of 1/10 and used at passages of ≤3.
Polarity of Et-1 secretion by BCEC
BCEC were cultured on collagen-coated Millicell CM inserts as described previously (Descamps et al., 1996). After 12 days of coculture with rat astrocytes, which corresponded to the establishment of tight properties (Dehouck et al., 1994), BCEC were cultured alone in serum-free DMEM. Culture media of the upper chamber (luminal side) and lower chamber (abluminal side) of the inserts were collected after 20 h, and Et-1 was assayed by radioimmunoassay according to the instructions of the manufacturer (Amersham, Les Ulis, France). Sucrose permeability and transendothelial resistance were determined as previously described (Dehouck et al., 1992).
Binding experiments
BCEC or pericytes (at passage 2) were scraped with a rubber policeman into a homogenizing buffer (250 mM sucrose, 1 mM EDTA, 10 mM Tris-Cl at pH 7.5) supplemented with a cocktail of protease inhibitors (0.1 mM bacitracin, 0.1 mM phenylmethylsulfonyl fluoride, 1μM leupeptine) and homogenized using a polytron (position 5, three times 5 s). After centrifugation at 100,000 g for 30 min, membranes were resuspended into an Earle salt solution [140 mM NaCl, 5 mM KCl, 1.8 mM CaCl2, 0.8 mM MgSO4, 5 mM glucose, and 25 mM N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid (HEPES) at pH 7.4] at a protein concentration of 0.9 mg/ml and stored at −20°C. For binding experiments, membranes (30 μg protein/ml) were incubated in an Earle salt solution supplemented with 0.05% (wt/vol) bovine serum albumin, 32 nM 125I-Et-1 (2,200 Ci/mmol; Amersham), and competitor drugs. After 2.5 h of incubation at 20°C, aliquots were filtered on 0.22-μm Sartorius filters under reduced pressure and the radioactivity remaining associated with the filters was determined. Nonspecific binding was determined in the presence of 100nM unlabeled Et-1. Binding data were analyzed using the Ligand software.
The presence of receptors occupied by endogenous Et-1 was assessed using the acid wash technique (Resink et al., 1990). Membranes prepared from BCEC were acidified to pH 2.5 with 25 mM glycine and left at room temperature for 90 min. Membranes were pelleted by centrifugation at 100,000 g and resuspended in an Earle salt solution. 125I-Et-1 binding sites were titrated as described already.
Intracellular Ca2+ measurements
BCEC or pericytes were loaded with 5 μM indo-1/AM for 2 h in complete culture medium at 37°C. After dissociation from the culture dishes using TE, cells were centrifuged at 1,000 g for 10 min and suspended in an Earle salt solution. Flow cytometric analysis of indo-1 fluorescence ratio was performed as previously described (Vigne et al., 1990) using a FacStar Plus (Becton-Dickinson). Indo-1 fluorescence ratio were measured in single cells and collected in real time at a rate of 500 cells/s. Means of 1,000 individual cell measurements were computed and collected at different times following the addition of agonists. To define dose–response curves for agonists, cells were treated with agonists and the mean indo-1 fluorescence ratio of 1,000 cells, sampled during 2 s, was collected 30 s after addition of the agonists. Solutions containing a low Ca2+ concentration (50 nM) were prepared using 1.8 mM CaCl2 and 4 mM ethyleneglycol bis-(β-aminoethylether)-N,N,N′,N′-tetraacetic acid. Dose–response curves were analyzed using the Sigma Plot software.
Fluorescence microscopy
Cells grown on coverslips were fixed at room temperature for 20 min with 4% paraformaldehyde in a fibrous component stabilizing buffer [60 mM piperazine-N,N′-bis(2-ethane)sulfonic acid, 25mM HEPES, 10 mM ethyleneglycol bis-(β-aminoethylether)-N,N,N′,N′-tetraacetic acid, 2 mM MgCl2, and 140 mM NaCl, pH 6.9]. Cells were then washed with this buffer and permeabilized with cold acetone (−20°C) for 1 min, followed by two more washes with the stabilizing buffer. Cells were stained with the F-actin probe BODIPY phallacidin (165 nM; Molecular Probes) for 30 min at room temperature. For the localization of vimentin and α smooth muscle actin, free aldehyde groups were quenched with 0.1 M l-lysine for 4 h at 4°C. Coverslips were then incubated for 30 min in Tris-Cl-buffered saline [20 mM Tris-Cl, 0.5 M NaCl, pH 7.0 (TBS)] containing 5% (wt/vol) ovalbumin (OVA) and 1% (vol/vol) heat-inactivated normal goat serum. This was followed by a 1-h incubation with mouse monoclonal antibodies to swine vimentin (Clone 9, diluted 1:10 in TBS/OVA; Dako) or to α smooth muscle actin (Clone 1A4, diluted 1:100 in TBS/OVA; Biogenex Laboratories). Fluorescein isothiocyanate–conjugated affinity-purified goat anti-mouse IgG (Cappel Laboratories, West Chester, PA, U.S.A.) was used as secondary antibody at a 1:100 dilution in TBS/OVA. After several washes, coverslips were mounted on a microscope slide in Mowiol.
Cell proteins were determined according to Bradford (1976).
Data are reported as means ± SD of n independent experiments.
RESULTS
Bovine BCEC produced Et-1 in a polarized manner and did not express ET receptors
Cultured bovine BCEC at early passage numbers secrete immunoreactive Et-1 into their culture medium. Because of the tight properties of the cerebral endothelium, it was important to determine whether this secretion occurred at the luminal (blood side) or at the abluminal (brain side) side of the blood–brain barrier. Polarity of Et-1 secretion was analyzed using a cell culture system consisting of a coculture of BCEC on one side of a porous filter and astrocytes plated on the plastic of a six-well dish. After 12 days of coculture, endothelial cell monolayers displayed tight properties as evidenced by a low permeability to sucrose (permeability coefficient = 0.4 ± 0.05 10–3 cm/min; n = 9) and a high electrical resistance (600–800 Ω · cm2). Conditioned media in the abluminal (astrocyte-facing) and luminal sides of Millicell CM inserts were harvested and assayed for the presence of immunoreactive Et-1. The results showed an abluminal rate secretion (665 ±30 fmol/cm2/20 h) 4.4 times higher than the rate of luminal secretion (150 ±8 fmol/cm2/20 h) (n = 3). This indicated that bovine BCEC preferentially secreted Et-1 toward the abluminal, brain-facing, side of the capillaries.
We next examined whether BCEC expressed receptor sites for Et-1, i.e., whether Et-1 could act in an autocrine manner at the bovine blood–brain barrier. No evidence for the presence of a specific 125I-Et-1 binding component was obtained in BCEC membranes. One possibility for this result could be that receptors were present but were masked by endogenous Et-1 as reported for mesangial cells (Clozel et al., 1993). To look for the presence of such cryptic receptors, titration experiments were also performed on acid-washed membranes but yielded negative results. These indicated that cultured bovine BCEC did not express receptors for Et-1. In agreement with this conclusion, we observed that addition of 100 nM Et-1 to indo-1-loaded bovine BCEC did not increase cytosolic Ca2+ concentrations. Under the same conditions, BCEC responded to 1 μM bradykinin and to 100 μM ATP by large intracellular Ca2+ transients (data not shown). This result indicated that bovine BCEC, at least under culture conditions, did not express functional receptor sites for Et-1.
Bovine brain pericytes expressed functional ETA receptors
Because of their close proximity to BCEC in brain capillaries, pericytes are putative target cells for Et-1 produced by BCEC. Pure cultures of pericytes from bovine brain capillaries were prepared. Pericytes were characterized by their large size and branched morphology (Fig. 1A). They showed positive immunofluorescence for α smooth muscle actin antibodies (Fig. 1B). They were negative for Factor VIII-related antigen, a marker of vascular endothelial cells, and for glial fibrillary acidic protein, a marker of astrocytes (data not shown).
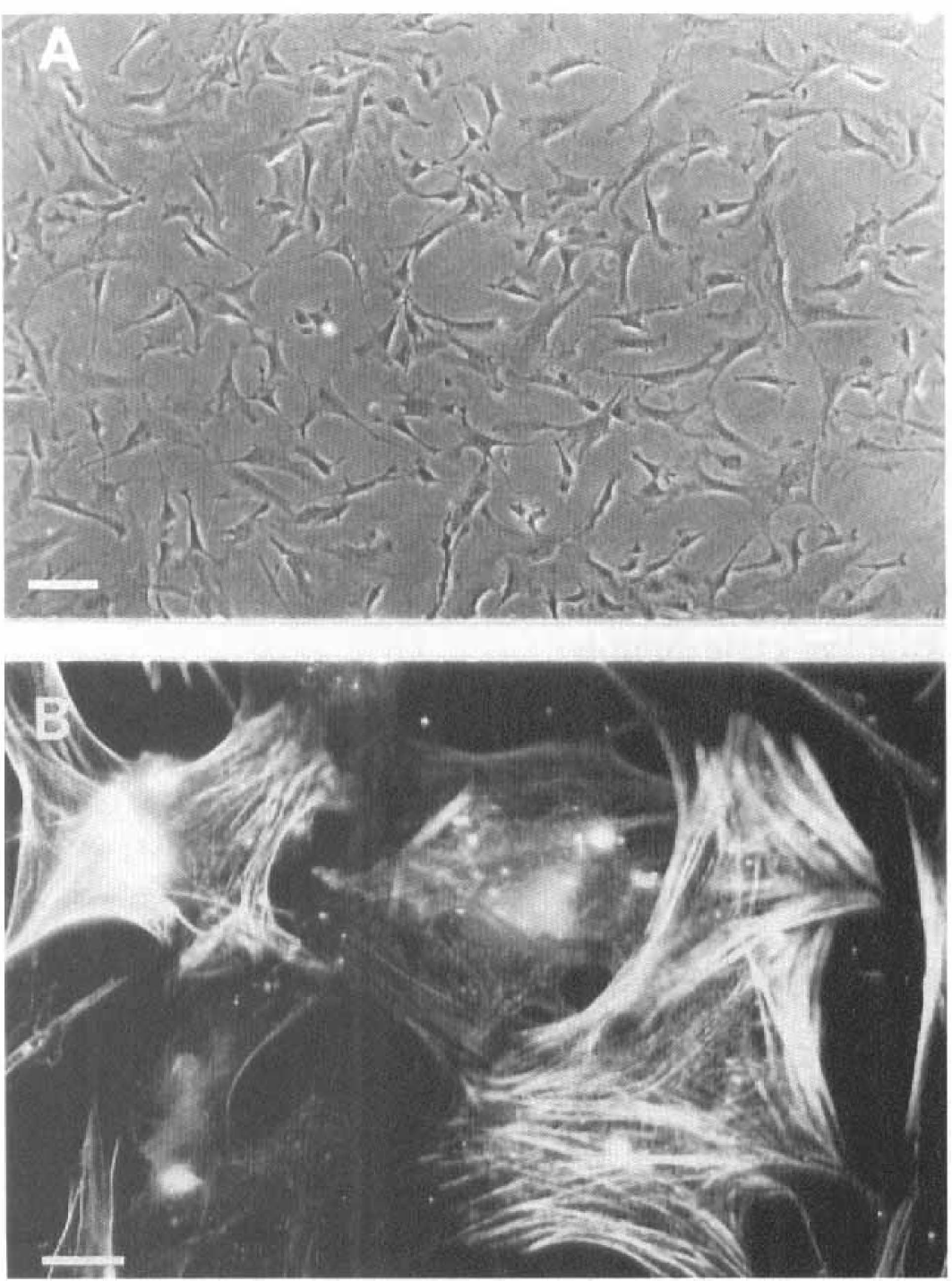
Properties of cultured bovine brain pericytes. (
Figure 2 shows that 125I-Et-1 bound specifically to pericyte membranes. The specific 125I-Et-1 binding was prevented by unlabeled Et-1, Et-3, and BQ-123. Apparent equilibrium dissociation constants were 100 ± 20 pM for Et-1 and 5 ± 1 nM for BQ-123. Et-3 was at least 1,000 times less potent than Et-1. The pharmacological profile of agonists (Et-1 >> Et-3) and the action of low concentrations of BQ-123, a selective antagonist of ETA receptors (Ihara et al., 1995), identified the presence of an ETA receptor subtype.
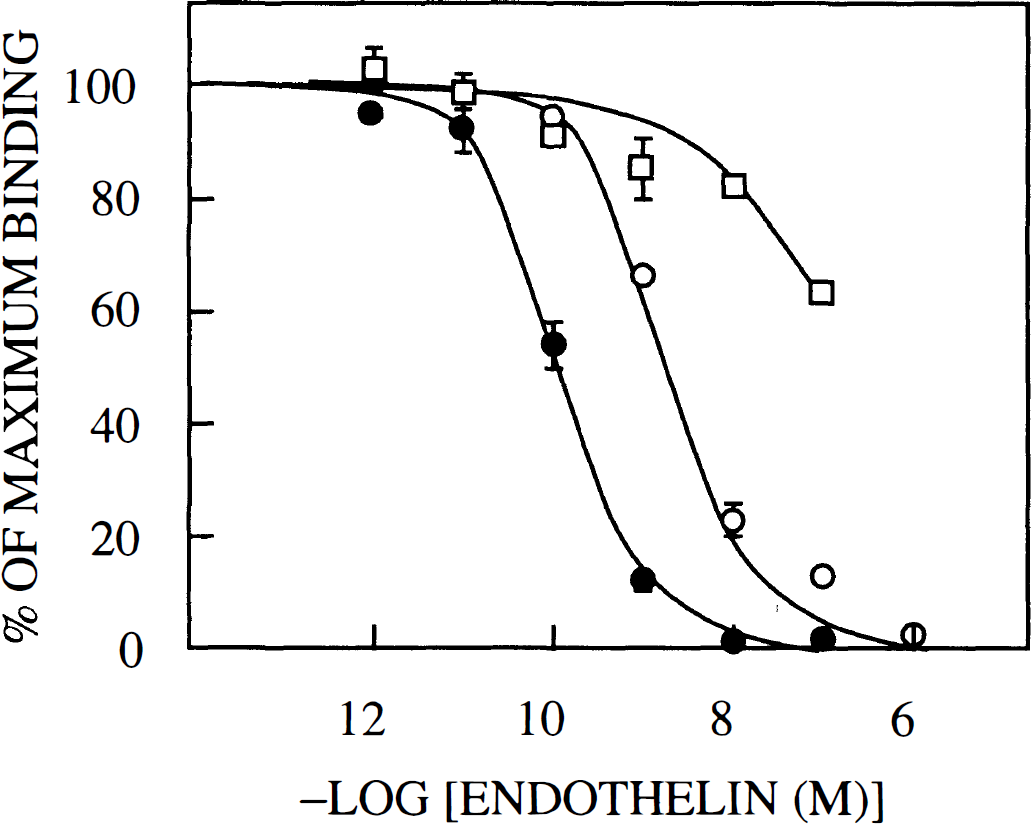
Pericytes express an ETA receptor subtype. Dose–response curves for the competitive actions of unlabeled endothelin-1 (Et-1) (filled circles), BQ-123 (open circles), and Et-3 (squares) on 125I-Et-1 binding to pericyte membranes. Means ± SD (n = 3) are shown.
ETA receptors usually couple to phospholipase C. This action leads to the production of inositol 1,4,5-trisphosphate from phosphatidylinositol bisphosphate and to the mobilization of intracellular Ca2+ stores. Activation of phospholipase C in pericytes was assessed by intracellular Ca2+ measurements using the fluorescent Ca2+ indicator indo-1. Figure 3 shows that the addition of 100 nM Et-1 to pericytes induced a large increase in the indo-1 fluorescence ratio, which was nearly identical at 1.8 mM or 50 nM external Ca2+. This indicated a likely contribution of intracellular Ca2+ stores to the measured indo-1 fluorescence signal. Figure 3 further shows that the action of Et-1 was almost completely suppressed by 10 μM BQ-123. Figure 4 presents the dose–response curves for Et-1 and Et-3 actions on intracellular Ca2+. The concentration of Et-1 that produced half-maximal increase in the indo-1 fluorescence signal was 1.3 ± 0.3 nM. The corresponding value for Et-3 was 134 ± 36 nM. The 100-fold difference in potency between Et-1 and Et-3 and the sensitivity to BQ-123 suggested the involvement of ETA receptors that were identified in 125I-Et-1 binding experiments (Fig. 2). Brain pericytes did not respond to bombesin, substance P, neurokinin A, neurokinin B, bradykinin, vasopressin, angiotensin II (all at 1 μM), or 0.1 mM glutamate by changes in the intracellular Ca2+ concentration.
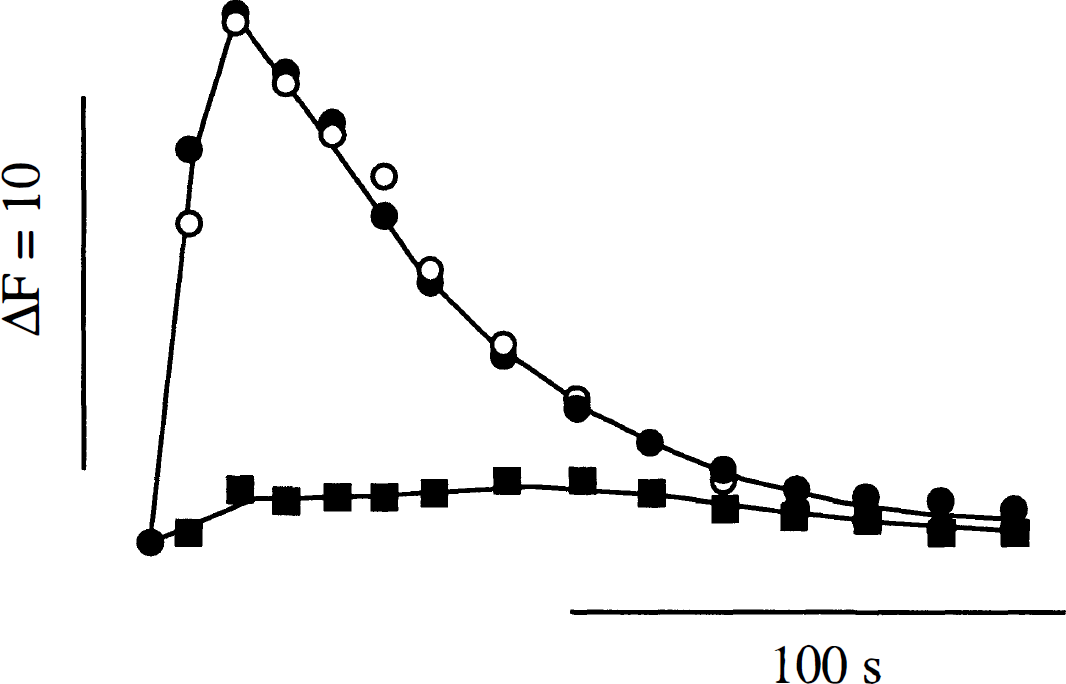
Intracellular Ca2+ transients induced by endothelin-1 (Et-1) in brain pericytes, Indo-1-loaded cells were treated with 100 nM Et-1 and the mean indo-1 fluorescence ratio from 1,000 cells was measured at different times after Et-1 addition. Experiments were performed in the presence of 1.8 mM external Ca2+ (filled circles and squares), of 50 nM external Ca2+ (open circles), and of 10 μM BQ-123 (filled squares). SD was smaller than the size of the points.
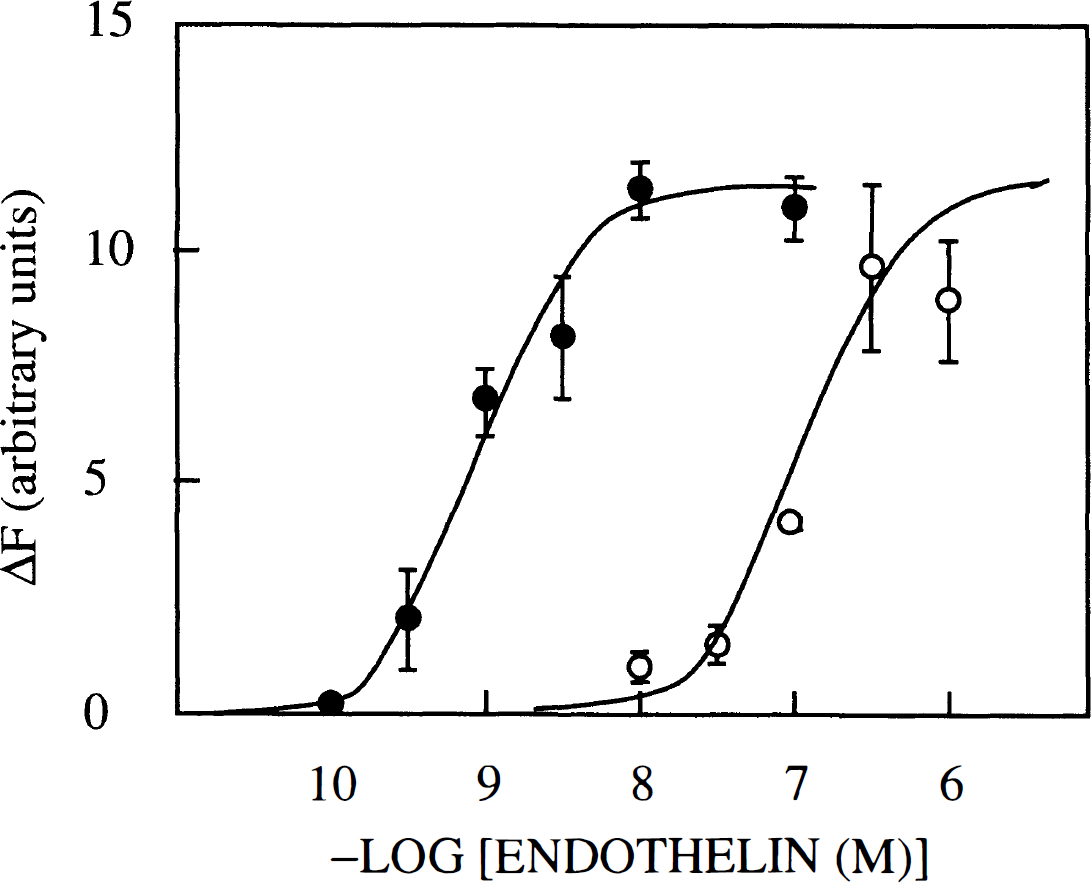
Dose-response curves for the actions of endothelin-1 (Et-1) and Et-3 on [Ca2+]. Indo-1-loaded cells were treated with the indicated concentrations of Et-1 (filled circles) and Et-3 (open circles) and the mean indo-1 fluorescence ratio from 1,000 individual cells was measured after 30 s. Means ± SD (n = 3) are indicated.
Addition of 100 nM Et-1 to pericytes induced marked changes in cell morphology that were suggestive of contractions. This conclusion was ascertained by the fact that Et-1 induced the aggregation and realignment of F-actin into bundles that were oriented circularly around the nucleus (Fig. 5A). A representative control is shown in Fig. 5C. Interestingly, changes in cell morphology were also associated with a redistribution of vimentin, a major component of intermediate filaments (Fig. 5B and D).
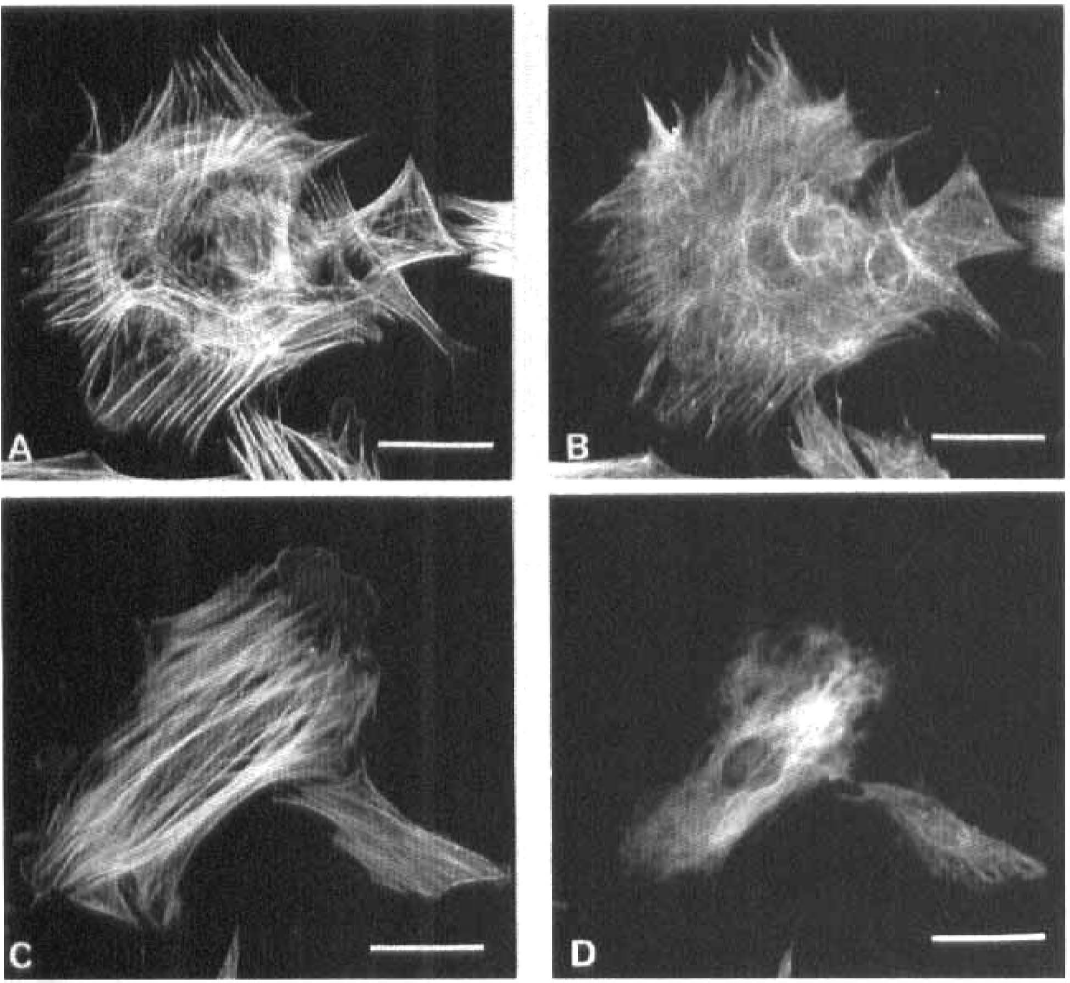
Endothelin-1 (Et-1) induced pericyte contractions. Pericytes incubated in HEPES/Ringer supplemented with 0.01% bovine serum albumin were exposed for 5 min to 100 nM Et-1, fixed, and stained with BODIPY phallacidin (
DISCUSSION
Et-1 was originally purified from culture media conditioned by porcine aortic endothelial cells (Yanagisawa et al., 1988). It was later found to be produced by a variety of macrovascular endothelial cells. Et-1 is a major regulator of the vascular tone. It controls smooth muscle cell contractility in two independent manners. An autocrine action induces NO-mediated relaxations. A direct action of Et-1 on vascular myocytes induces long-lasting contractions (Rubanyi and Parker Botelho, 1991).
Endothelial cells from brain capillaries, although they are not associated with smooth muscle cells, also secrete Et-1. Et-1 secretion was observed with porcine BCEC (Yoshimoto et al., 1991), bovine BCEC at low passage numbers (this study), immortalized bovine BCEC (Durieu-Trautmann et al., 1993), and human BCEC (Spatz et al., 1994). Rat BCEC express Et-1 prepro-mRNAs (Frelin and Vigne, 1993), but they do not secrete Et-1 into their culture medium (Vigne et al., 1990). One reason for the lack of Et-1 secretion by rat BCEC could be that secreted Et-1 is trapped onto endogenous receptors (Frelin and Vigne, 1993). Taken together, these results indicated that secretion of Et-1 is probably a general property of brain microvascular endothelial cells.
We have previously developed methods to obtain pure populations of bovine BCEC that acquire blood–brain barrier properties when cocultured with astrocytes. These properties are expression of tight junction–associated protein ZO-1 (Descamps et al., 1996), the development of a high electrical resistance and of a low permeability to sucrose and inulin (Dehouck et al., 1990), and the expression of enzyme activities specific to the blood–brain barrier such as γ-glutamyl transpeptidase (Dehouck et al., 1990), monoamine oxidase (Méresse et al., 1989), and carrier-mediated transport processes (Dehouck et al., 1994; Descamps et al., 1996). Results presented in this article illustrate another property of tight BCEC monolayers: a polarized secretion of Et-1. Secretion preferentially occurs at their abluminal face, i.e., toward brain tissues. This suggests that Et-1 secreted by BCEC serves local functions in the capillary walls rather than systemic functions.
Possible target cells for Et-1 secreted by BCEC are BCEC themselves, pericytes, and astrocytes. Responses of astrocytes to Et-1 have been well documented. They involve ETB receptors (MacCumber et al., 1990; Marsault et al., 1990). Responses of BCEC to endothelins are species specific. Rat and human BCEC express ETA receptors for Et-1. In these cells, Et-1 induces prominent cellular responses such as the activation of phospholipase C (Vigne et al., 1990; Stanimirovic et al., 1994), of the Na+/H+ antiporter (Vigne et al., 1991), of the Na+,K+,Cl– cotransporter (Vigne et al., 1994), and of Ca2+-activated K+ channels (Van Renterghem et al., 1995). These have suggested that one function of Et-1 in brain capillaries could be to control the permeability of the blood–brain barrier to K+ (Vigne et al., 1994; Van Renterghem et al., 1995). This study shows that bovine BCEC differ from human and rat BCEC in that they do not respond to Et-1. The lack of response was not due to a masking of receptors by endogenous Et-1 as reported for mesangial cells (Clozel et al., 1993). Provided that cultured cells have the same properties as in vivo, these would suggest that Et-1 has different functions in bovine and human (or rat) capillaries. It may have both autocrine and paracrine actions in human and rat capillaries. It may have only paracrine actions in bovine capillaries.
Two subtypes of Et receptors have been cloned. ETA receptors display a high affinity for Et-1 and a low affinity for Et-3. They are selectively antagonized by BQ-123 (Arai et al., 1990; Ihara et al., 1995). ETB receptors do not discriminate between Et-1 and Et-3 (Sakurai et al., 1990). 125I-Et-1 binding experiments (Fig. 2) and intracellular Ca2+ measurements (Figs. 3 and 4) indicated that bovine brain pericytes express functional BQ-123-sensitive ETA receptors. This property is shared by vascular smooth muscle cells (Rubanyi and Parker Botelho, 1991). It is also shared by retinal pericytes (Chakravarthy et al., 1992; Ramachandran et al., 1993) and renal mesangial cells (Simonson and Rooney, 1994). These suggest that ETA receptor-mediated reactivity to Et-1 is probably a tissue-independent property of pericytes.
Pericytes isolated from bovine brain capillaries have many features of smooth muscle cells. They express a α smooth muscle actin (Fig. 1) and ETA receptors for Et-1 (Fig. 2). The contractions observed in response to Et-1 (Fig. 5) suggest that one function of pericytes could be to regulate microvascular caliber and tone similarly to vascular smooth muscle cells in larger vessels. Et-1 has been proposed as a key mediator of cerebral vasospasm following subarachnoid hemorrhage (Asano et al., 1989). Extensive evidence for a protective effect of antagonists of ETA receptors has also been presented (Clozel and Watanabe, 1992; Itoh et al., 1993; Nirei et al., 1993). The fact that brain capillary pericytes are target cells for Et-1 suggests that pericytes could be involved in the pathogenesis of cerebral vasospasms in addition to macrovascular smooth muscle cells.
Footnotes
Acknowledgment:
The authors thank N. Boyer and J. Kervella for expert technical assistance.
