Abstract
Wnt morphogens released by neural precursor cells were recently reported to control blood–brain barrier (BBB) formation during development. Indeed, in mouse brain endothelial cells, activation of the Wnt/
INTRODUCTION
The blood–brain barrier (BBB) is a physiologic interface between the central nervous system and the blood. It strictly limits the passive diffusion of polar substances from the blood to the brain, providing brain parenchyma with nutrients and efflux from the brain of toxic metabolites and xenobiotics by active mechanisms and controlling the migration of circulating immune cells. 1 The unique phenotype of BBB endothelial cells is characterized by the expression of tight junctions (TJ) and adherens junctions, two distinct, although functionally related, junctional complexes. Tight junctions are essential for BBB properties not only by restricting the passive diffusion of ions or macromolecules, 2 but also by preventing the lateral diffusion of lipids and integral membrane proteins, thus controlling the brain endothelial apicobasal polarity. 3
Tight junction complexes are composed of transmembrane proteins, notably claudin (cldn) −3, −5
2
and occludin,
4
which are crucial to maintain TJ integrity together with several immunoglobulin superfamily members, such as junctional adhesion molecules.
5
In addition, cytosolic proteins, such as the ZO family (ZO−1, −2, −3) directly bind the intracellular domains of these transmembrane proteins and act as molecular links to the cytoskeleton and signal transducers. We recently identified Gαi2 as a new cldn-5 partner that regulates TJ integrity in the hCMEC/D3 human brain endothelial cell line, an extensively used
Development and maintenance of the BBB needs a dynamic dialog between endothelial and perivascular cells of the so-called neurovascular unit: indeed, neural precursor cells were shown to secrete factors that have a central role in BBB formation and integrity.
9
Among these factors, Wnt morphogens appear as key regulators of the BBB phenotype. The Wnt family includes 19 secreted glycoproteins, which primarily signal through seven-pass transmembrane receptors, the Frizzled (Fzd) family in various cell types, including epithelial and endothelial cells.10, 11 Wnts activate several signaling pathways known as the
The present study aimed to assess whether the Wnt/Par/aPKC PCP pathway, in parallel to the Wnt canonical pathway, might directly control TJ integrity and apicobasal polarity in brain endothelial cells. Here we show that the Par/aPKC PCP complex controls hCMEC/D3 cell polarity; in addition, PAR-3 downregulation by specific small interfering RNAs (siRNAs) increases endothelial permeability and delays TJ recovery after hyperosmotic treatment. Finally, we demonstrate that Wnt5a, through a selective activation of the Wnt/Par/aPKC PCP pathway, significantly contributes to TJ integrity, strongly suggesting that the Wnt/Par/aPKC PCP pathway is a key regulator of the BBB.
MATERIALS AND METHODS
Cell Culture Conditions
The immortalized human brain microvessel endothelial cell line (hCMEC/D3) was cultured at density of 50,000 cells per cm2 on Transwell inserts (0.4
Small Interfering RNA Experiments
Small Interfering RNA transfections were performed using various Stealth RNAi duplexes against PAR-3 (#HSS183488, #HSS183489), aPKC-ζ (#HSS183348, #HSS183325), cldn-5 (#HSS186370), and nontargeting siRNA as control (Life Technologies SAS). Cells were plated onto culture inserts (Corning, Lowell, MA, USA) or culture E-plates wells for xCELLigence technology in culture medium without antibiotics. One hour after cells seeding (day 0), the transfection mix containing Lipofectamine RNAiMAX (Life Technologies SAS), 50 nmol/L of siRNAs and Opti-MEM Reduced Serum Medium (Life Technologies SAS) was added to the culture medium according to the manufacturer's instructions. Cells were incubated for 6 days at 37°C in a 5% CO2 incubator and culture medium was changed at day 3 with fresh siRNA-supplemented transfection medium. The efficacy of PAR-3, aPKC-ζ, and cldn-5 knockdown was assessed by immunoblotting (see below).
Permeability Assays
HCMEC/D3 cells were seeded onto culture inserts for 6 days and permeability assays with 50
Immunofluorescence Microscopy
HCMEC/D3 cells were cultured for 6 days on insert filters, washed with phosphate-buffered saline (PBS) and fixed with 2% paraformaldehyde for 10 minutes at room temperature; then cells were permeabilized with 0.1% Triton X-100 in PBS for 5 minutes at room temperature, blocked in 3% bovine serum albumin in PBS (blocking buffer) for 30 minutes, and stained with the indicated primary antibodies (diluted in the blocking buffer overnight at 4°C), directed against the following proteins: ZO-1 (monoclonal or polyclonal antibodies, Life Technologies SAS), VE-cadherin (Clone BV6; Merck Millipore SAS), Podocalyxin (Santa Cruz Biotechnology, Heidelberg, Germany), PAR-3 (Merck Millipore SAS), GLUT-1 (Epitomics, Burlingame, USA). After three washes with PBS, cells were incubated for 1 hour with Alexa488- or Alexa568-conjugated secondary antibodies (Life Technologies SAS). Cells were then washed three times with PBS and preparations were mounted in Glycergel medium (Dako, Carpinteria, CA, USA) supplemented with 0.1% 4′,6-diamidino-2-phenylindole. Cells were observed with a Zeiss Axio Observer Z1 microscope using a × 40 Oil Objective (Olympus, Rungis, France). For apicobasal polarity ratio assessment, cell membranes were stained before cell fixation with 5
Image Analysis
Analyses were performed using homemade ImageJ routine (http://www.imagej.nih.gov/ij/). For apicobasal polarity ratio assessment, the quantification of fluorescence intensity was estimated at the apical and basal plasma membranes, respectively above and below the nucleus, as illustrated in Figure 2B, taking advantage of the larger thickness of the cells at the level of the nucleus. Using nuclear signal
To establish the apicobasal ratio, CM-Dil labeling was used to define the apical and basal membrane boundaries. Then total fluorescence intensity of targeted protein was evaluated in those both domains and the apicobasal ratio was calculated.
Immunoblotting Assays
HCMEC/D3 cells were washed with PBS, lysed for 10 minutes with ice-cold Laemmli lysis buffer and scraped. Proteins of the whole-cell lysate were heated at 95°C for 5 minutes and processed for immunoblotting as previously described. 6
xCELLigence Assays
The xCELLigence system (ACEA, San Diego, USA) is a cell-based label-free instrument that measure in real time electrical impedance across gold microelectrodes integrated on the bottom of culture E-plates. HCMEC/D3 cells were seeded in standard culture medium without antibiotics at a density of 50,000 cells per cm2 onto 96-well E-Plates coated with 150 μg/mL rat tail collagen type I. One hour after cell adhesion, the siRNA transfection mix was added (see protocol of siRNAs above) and cells were cultured for 7 days. As indicated, at 72 hours of culture, the confluent monolayer of hCMEC/D3 cells was treated with 1 mol/L mannitol (osmotic shock) in serum-free culture medium for 30 minutes. Then, mannitol solution was removed and standard culture medium was added for a recovery period up to 4 days. Impedance measurement was displayed in real time as a cell index (CI, an arbitrary unit) by the xCELLigence RTCA software (ACEA, San Diego, CA, USA). Results are presented as mean values of CI±s.d. against time in each condition.
Quantitative Real Time Polymerase Chain Reaction
Axin2 (a
The cDNA template (diluted 1:20) was then mixed with forward, reverse primers (0.5 μmol/L) and LightCycler 480 SYBR Green I Master (Roche, Basel, Switzerland) according to the manufacturer's instructions. Quantitative real time polymerase chain reaction was performed on a Roche Applied Science LightCycler 480 system (Roche,). Raw data were analyzed with LightCycler 480 software (Roche,) using the ΔΔ
Axin2: forward (5′-CGCCAACGACAGTGAGAT-3′); reverse (5′-TGCCCACACGATAAGGAG-3′)
RT2 Profiler Polymerase Chain Reaction Array
Total RNA was isolated using the RNeasy mini kit (Qiagen). RNA purity and concentration were determined with a NanoDrop 2000 spectrophotometer (Thermo Scientific). One microgram of total RNA was converted to cDNA using RT2 First Strand Kit (Qiagen). cDNA was mixed with RT2 SYBR Green Master mix and dispensed into the RT2 Profiler Array (Human WNT Signaling Pathway PAHS-043 F-2) (Qiagen). Quantitative real time polymerase chain reaction was performed on a Roche Applied Science LightCycler 480 system (Roche). Raw data were analyzed with LightCycler 480 software (Roche) using the ΔΔ
Statistical Analysis
Data are presented as means±s.d. Statistical analysis was performed using Student's
RESULTS
The Par/aPKC Planar Cell Polarity Complex is Expressed in hCMEC/D3 cells
Given that the Par/aPKC PCP complex is localized at TJs in epithelial cells, 19 we investigated its expression and localization in the human brain endothelial hCMEC/D3 cell line, a validated model of the human BBB. 8 As illustrated in Figure 1, PAR-3 expression could be detected at cell–cell junctions, in addition to nuclear and perinuclear localizations (Figure 1, left panel). PAR-3 expressed at cell–cell junctions colocalized with the TJ protein ZO-1 (Figure 1, center and right panels: arrows) suggesting that it might be located at TJs in brain endothelial cells, as previously reported in epithelial cells. In addition, we confirmed by western Blot analysis (data not shown) that hCMEC/D3 cells also express aPKC-ζ and PAR-6, the two other constituents of the Par/aPKC PCP complex, as recently reported. 22
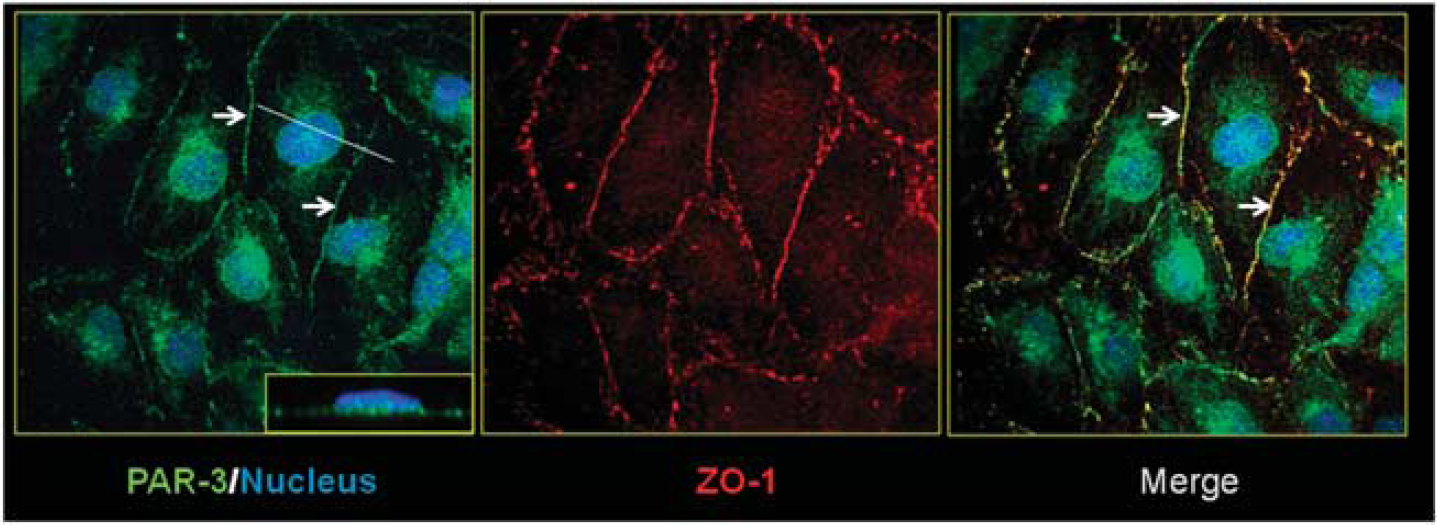
Par/aPKC planar cell polarity complex is expressed in hCMEC/D3 cells and colocalized with ZO-1. HCMEC/D3 cells grown at confluence for 6 days were fixed and permeabilized. Double immunofluorescence labeling was performed with a ZO-1 polyclonal antibody and PAR-3 monoclonal antibody. Nuclei were labeled with 4′,6-diamidino-2-phenylindole. Inset in right panel
The Par/aPKC Planar Cell Polarity Complex Controls Polarization of hCMEC/D3 Cells
It has been well established that the Par/aPKC PCP complex is a regulator of cell polarization in the epithelia. Because brain endothelial cells are also polarized cells, the putative role of Par/aPKC PCP complex in controlling hCMEC/D3 polarization was investigated. Together with podocalyxin (Podxl), an apical marker of endothelial cells, the ABC-transporter P-gp, selectively expressed by brain endothelial cells, is highly enriched at their apical membrane; by contrast, the glucose transporter GLUT-1 is known to be expressed at apical and basal membranes of brain endothelial cells. 23 By confocal immunofluorescence analysis of these proteins, we then compared their apical versus basal expression in hCMEC/D3 cells after siRNA-mediated PAR-3 knockdown or after treatment with scrambled siRNA as control.
Quantification of the apical versus basal expression of these three proteins was described in the M&M section and illustrated in Figure 2B. As expected, Podxl and P-gp were expressed in control cells at a significantly higher level in the apical membrane (apical/basal ratio: 2.53±0.43 and 2.12±0.45, respectively) (Figure 2C), whereas GLUT-1 expression was not significantly different in the two compartments (apical/basal ratio: 1.01±0.41) (Figure 2C). After confirming that two individual siRNAs substantially decreased PAR-3 expression in hCMEC/D3 cells (Figure 2A), we quantified the apical versus basal expression of Podxl, P-gp, and GLUT-1 in cells treated by PAR-3 siRNA (#A), the most efficient siRNA used. PAR-3 knockdown significantly decreased the polarization of Podxl and P-gp expression (apical/basal ratio: 1.60±0.36 and 1.11±0.32, respectively) (Figure 2C); as control, GLUT-1 localization was not affected (apical/basal ratio: 0.80±0.29) (Figure 2C). In Figure 2D, the same results are presented as the proportion of analyzed cells with increasing mean apical/basal ratios, from 0 to 0.5 (1) to 2.0 to 2.5 (5): the shift to the left of Podxl and P-gp expression in PAR-3 knockdown cells (Figure 2D: open bars) clearly illustrates that PAR-3 knockdown drastically reduced the spontaneous polarization of hCMEC/3 cells.
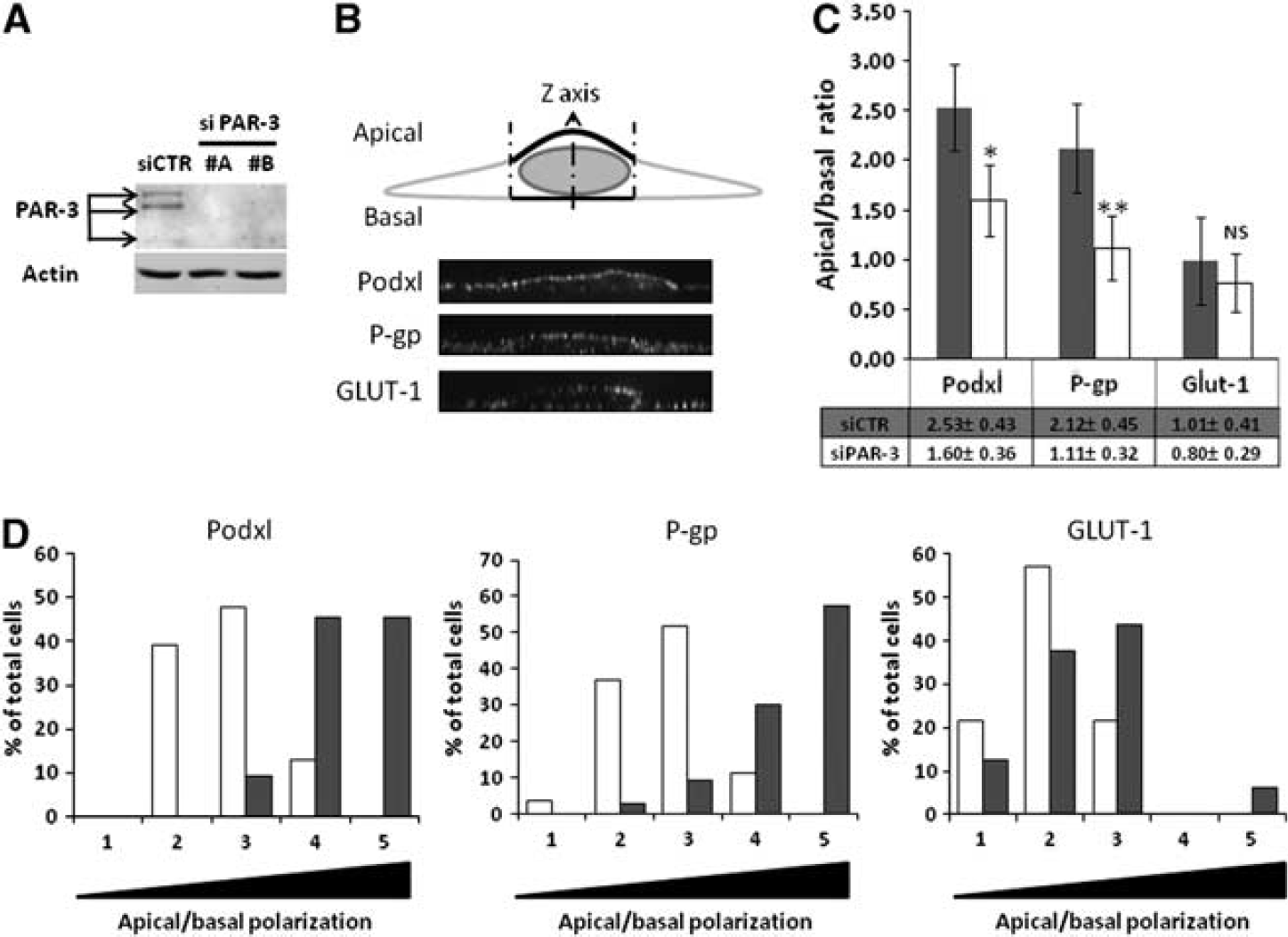
The Par/aPKC planar cell polarity complex controls hCMEC/D3 cells polarization. (
From this set of experiments, we concluded that the Par/aPKC PCP complex efficiently controls the polarization of brain endothelial cells.
The Par/aPKC Planar Cell Polarity Complex Controls Tight Junction Integrity
Because the Par/aPKC PCP complex has been described during epithelial morphogenesis to be involved in TJ complex formation and stability, we intended to investigate its function in brain endothelial cells, using the hCMEC/D3 model.
First, we assessed the consequences of PAR-3 or aPKC-ζ knockdown on hCMEC/D3 permeability to Lucifer Yellow (LY), a 457-Da fluorescent marker of paracellular permeability. As presented in Figure 3, PAR-3 knockdown significantly increased hCMEC/D3 permeability to LY. Similarly, two individual aPKC-ζ siRNAs increased LY permeability, although to various extents (likely due to incomplete protein knockdown, as shown in Figure 3A), suggesting that the Par/aPKC PCP complex is involved in the control of TJ integrity.
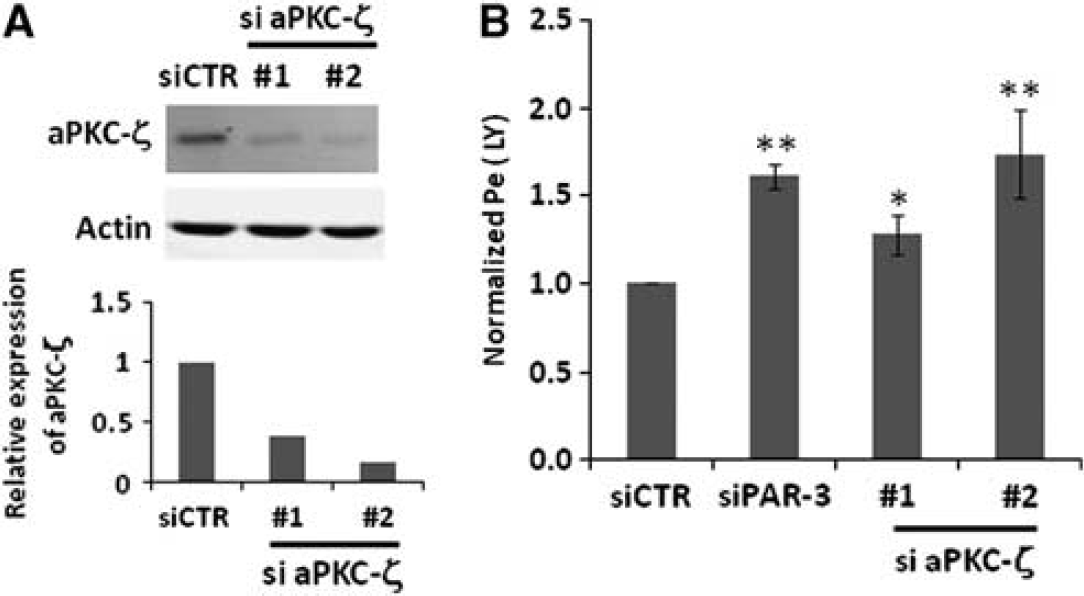
Par/aPKC planar cell polarity complex knockdown increases hCMEC/D3 permeabilty to Lucifer Yellow (LY). (
Second, immunofluorescence analysis of junction proteins showed that PAR-3 or aPKC-ζ knockdown decreased ZO-1 localization at cell–cell junctions, compared with the continuous staining observed in control cells (Figure 4, upper panel). By contrast, VE-cadherin expression/localization was not affected.
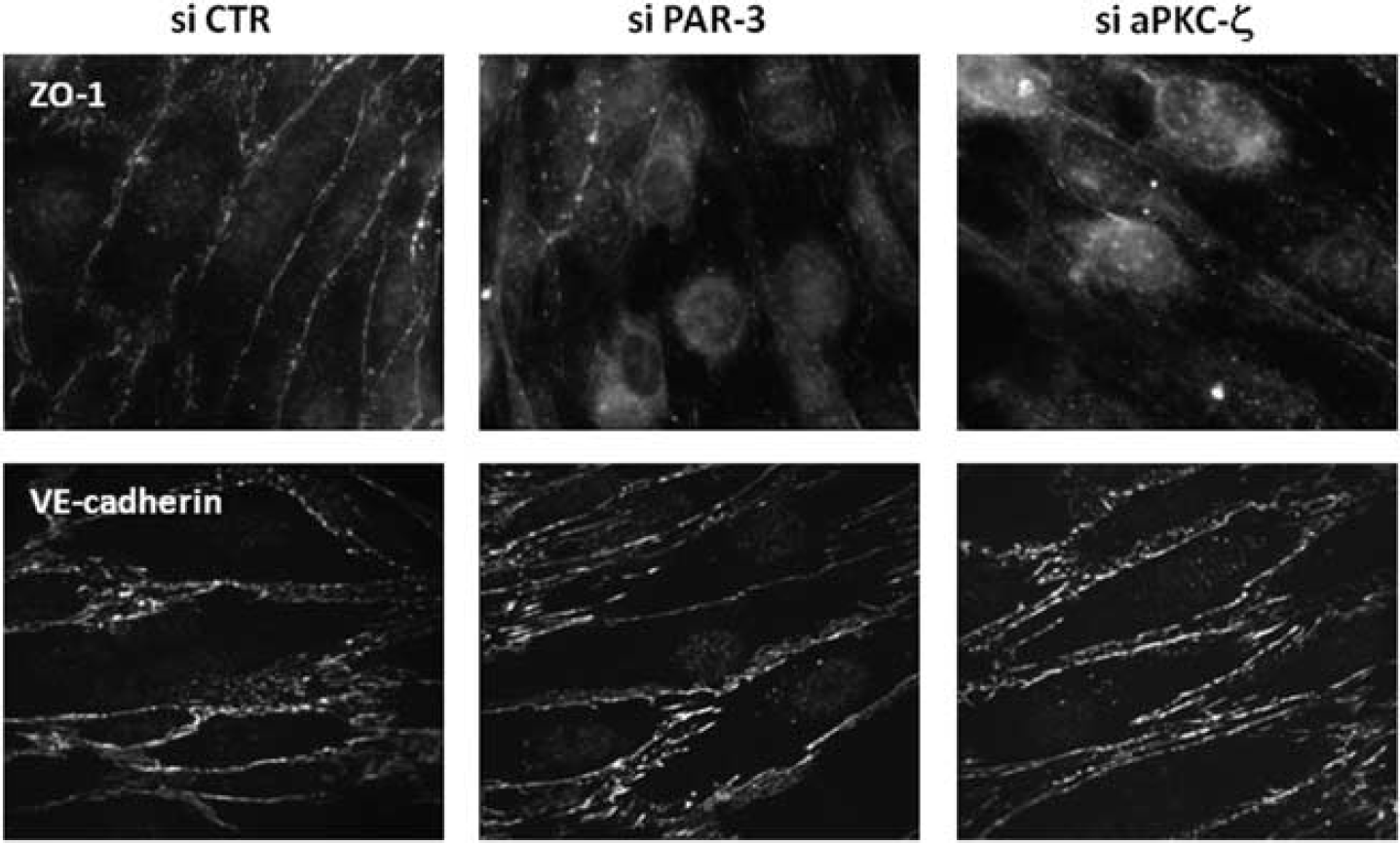
Par/aPKC planar cell polarity complex knockdown destabilizes tight junction complexes. HCMEC/D3 cells were treated with control small interfering RNAs (si CTR), PAR-3 siRNA (A) or atypical protein kinase C-ζ (aPKC-ζ) siRNA (#2), and grown at confluence for 6 days on culture inserts. Cells were fixed and permeabilized. Immunofluorescence labeling was performed with a ZO-1 monoclonal antibody and VE-cadherin (VE-cad) polyclonal antibody.
Altogether, these observations indicate that, although adherens junction complex assembly appears independent of the Par/aPKC PCP-complex, TJ complexes do not properly form in brain endothelial cells in absence of PAR-3 or aPKC-ζ, thereby affecting TJ integrity.
The Par/aPKC Planar Cell Polarity Complex Controls Tight Junction Reassembling after Osmotic Shock
We previously established that knockdown of cldn-5, a key TJ protein for BBB integrity
After a 72-hour period of proliferation, confluent control siRNA-treated hCMEC/D3 cells or PAR-3 or cldn-5 knockeddown cells were submitted to osmotic shock by 30-minute treatment with 1 mol/L mannitol; then monolayer impedance (assessed as CI) was monitored using the ACEA xCELLigence system over a time period of 96 hours. After cell proliferation, when cell confluence was reached, the CI of cldn-5 or PAR-3 knockeddown cells plateaued at similar levels (CI/cldn-5 knock-down=6.95±0.13; CI/PAR-3 knock-down=6.7±0.65), significantly lower than that of control siRNA-treated cells (CI=10.16±0.25) (Figure 5), which is in agreement with our observations that cldn-5 or PAR-3 deficiency did not prevent cells to reach confluence, but somehow destabilized TJs and increased LY permeability (Figure 4). 6
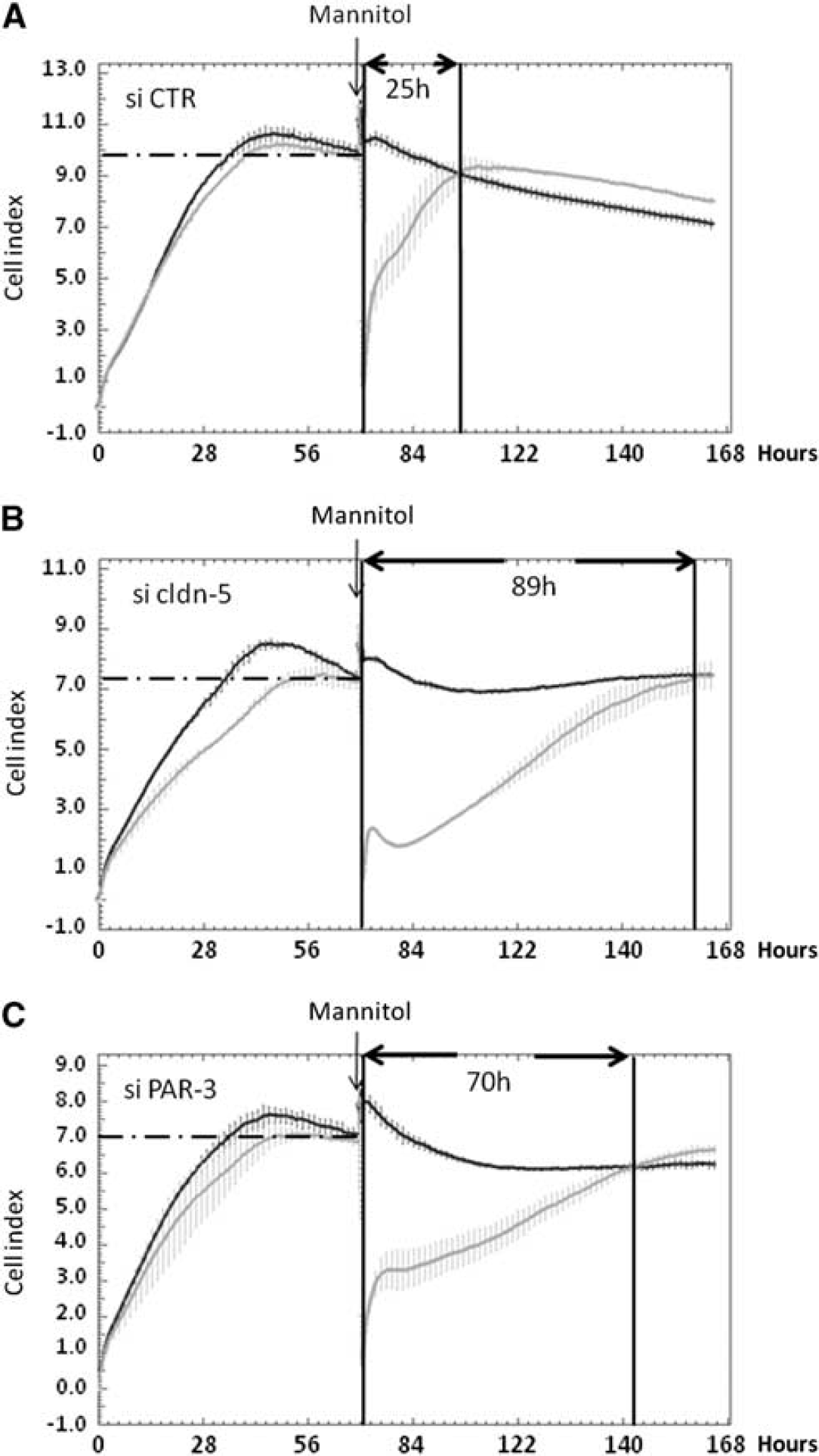
PAR-3 knockdown delays tight junction complex reassembling after 1 mol/L mannitol treatment. HCMEC/D3 cells treated with control (
Mannitol treatment, known to induce transient TJ destabilization at the BBB
Altogether, these data illustrate that the absence of PAR-3, like the absence of cldn-5, strongly affects the integrity of cell–cell junctions in hCMEC/D3 cells by disturbing the process of junction reassembly during recovery after an osmotic shock.
Wnt5a Regulation of hCMEC/D3 Permeability is Mediated by the Par/aPKC Planar Cell Polarity Complex
It is well established that the Par/aPKC PCP complex can be activated in epithelial cells by some Wnt factors, including Wnt5a. This functional link is being known as the Wnt/PCP pathway. Accordingly, we investigated a putative role of Wnt5a in controlling TJ integrity in hCMEC/D3 cells through the Par/aPKC PCP complex.
In a first step, we confirmed by qRT-PCR analysis that hCMEC/D3 cells express the major components of Wnt signaling pathways (Table 1), like Fzd −1 to −8 (except Fzd-3), Dishevelled proteins-1 and −2, together with Dishevelled-associated activator of morphogenesis 1, which is known to be triggered by Wnt5a.
25
As a next step, hCMEC/D3 cells were treated during 6 days with Wnt5a (40% conditioned medium of Wnt5a producing LM (TK) cells (ATCC #CRL-2814), as previously described.
26
As control, lithium chloride 10 mmol/L was used as an activator of the Wnt/
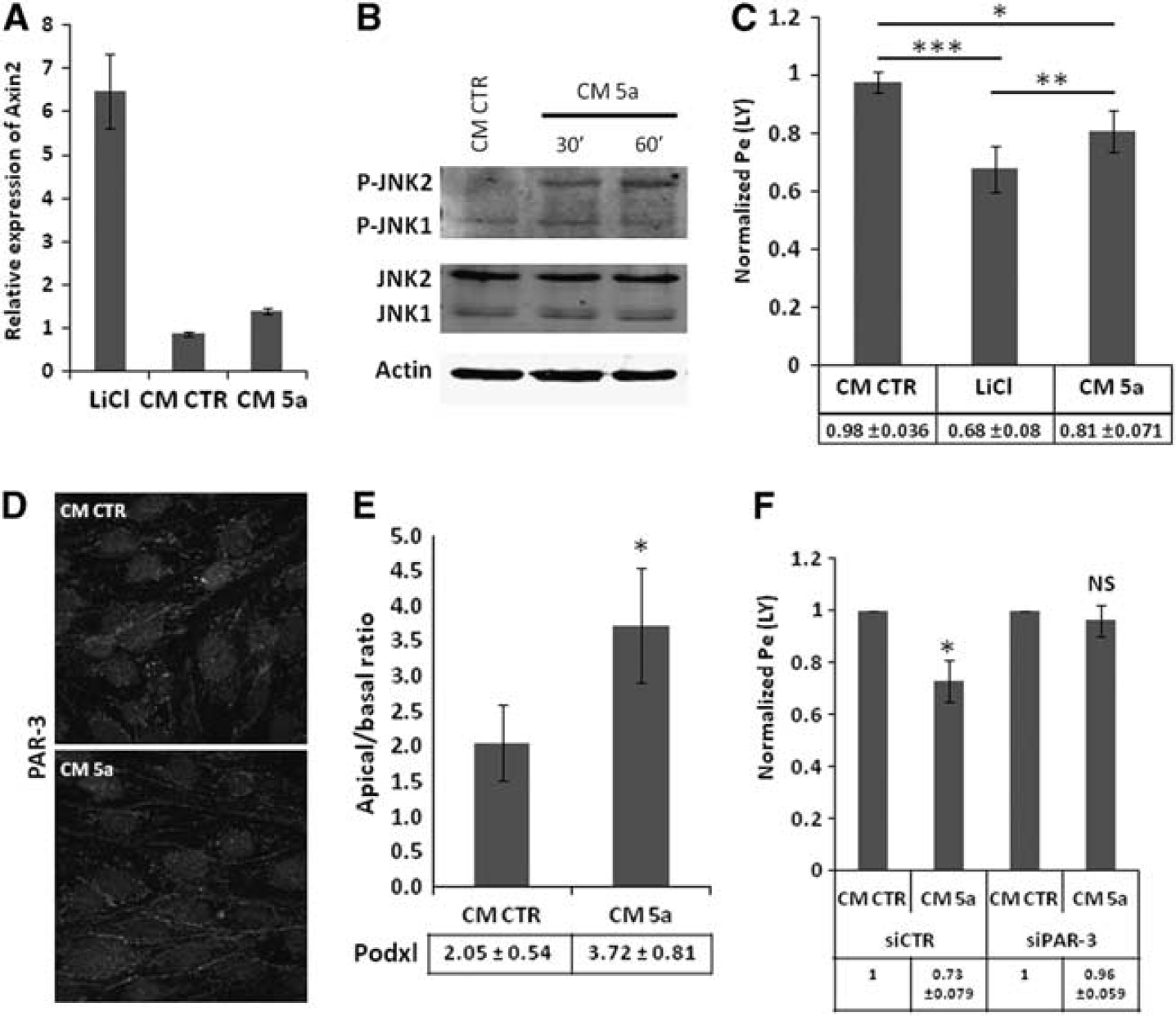
Wnt5a-triggered control of hCMEC/D3 permeability is mediated by Par/aPKC planar cell polarity complex. (
Expression of genes involved in Wnt signaling pathways in hCMEC/D3 cells
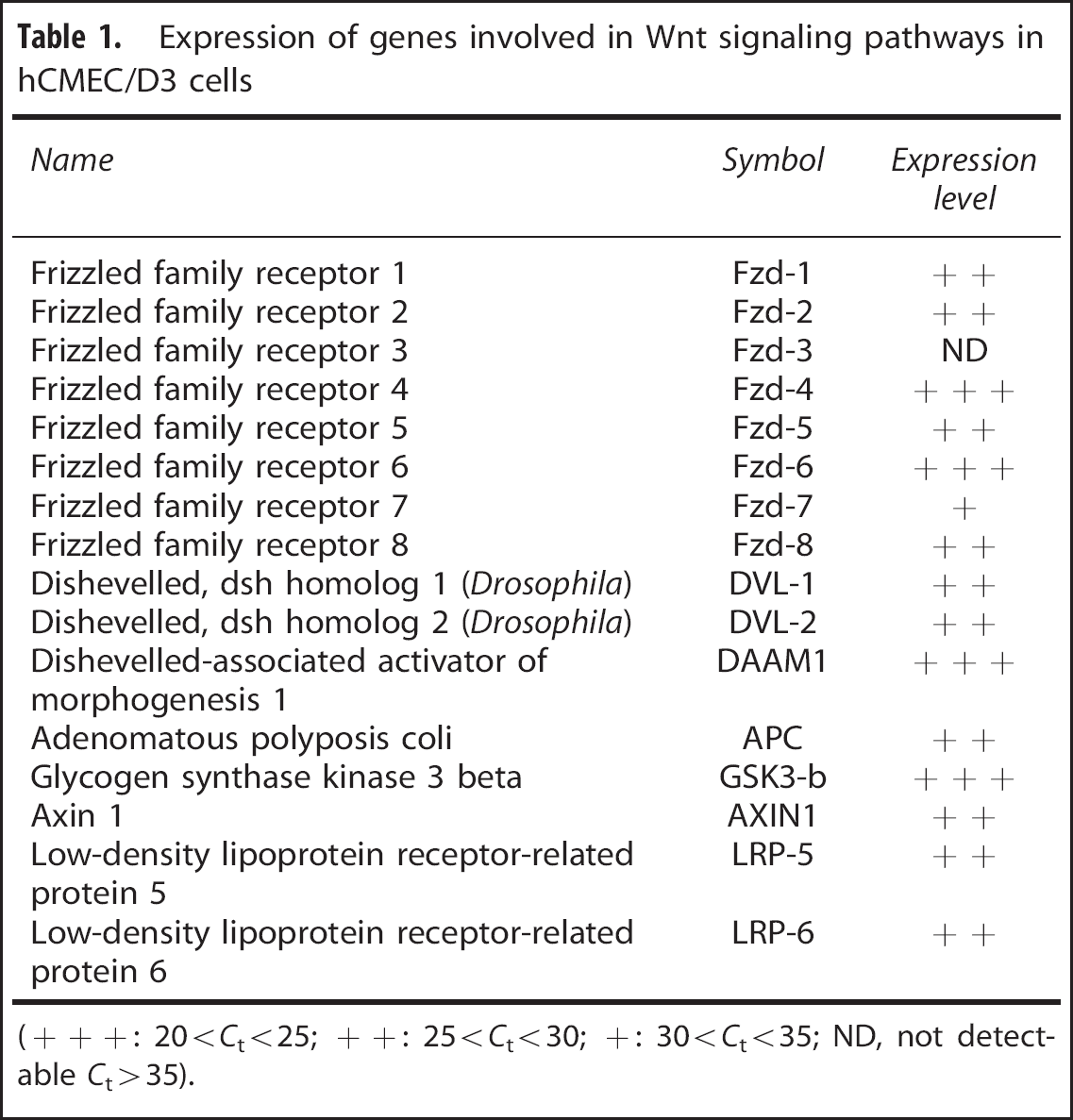
(+++: 20<
Because C-JUN-N-terminal kinase (JNK) has been reported as a downstream effector of the Wnt/PCP pathway, the putative phosphorylation of JNK, reflecting its activation, was assessed in hCMEC/D3 cells in response to Wnt5a. As illustrated in Figure 6B, Wnt5a-conditioned medium increased JNK-2 phosphorylation in 30 to 60 minutes, strongly suggesting that the Par/aPKC PCP complex is activated by Wnt5a. After 6 days of incubation with Wnt5a-conditioned medium, LY permeability of hCMEC/D3 cells was moderately but significantly decreased as compared with cells treated with control-conditioned medium (0.81±0.07 as compared with 1 for normalized permeability of control cells) (Figure 6C); as control, lithium chloride also decreased LY permeability (0.68±0.08), as previously reported. 15
In order to establish whether this effect of Wnt5a might be mediated by the Par/aPKC PCP complex, we first monitored PAR-3 expression and localization in hCMEC/D3 cells after Wnt5a stimulation. As illustrated in Figure 6D, PAR-3 localization to cell–cell contacts was stronger after Wnt5a treatment, in line with its putative activation-induced translocation to the plasma membrane. Furthermore, we observed by confocal fluorescence microscopy that the apical/basal ratio of Podxl expression was significantly increased after Wnt5a treatment: apical/basal ratio: 3.72±0.81 versus 2.05±0.54 in control condition (Figure 6E).
Finally, PAR-3-deficient or control hCMEC/D3 cells were treated with Wnt5a-conditioned medium, or control-conditioned medium. Whereas Wnt5a-conditioned medium significantly decreased LY permeability of control hCMEC/D3 cells (as reported above), no effect was observed in PAR-3-deficient cells (Figure 6F); as control, Wnt5a increased JNK phosphorylation in PAR-3-deficient as well as control cells, confirming that the proximal step of the Wnt/PCP pathway was activated by Wnt5a in both cell types (data not shown).
Altogether, these results indicate that Wnt5a stabilizes brain endothelial TJs through activation of the Par/aPKC PCP complex, independently of the Wnt canonical pathway.
DISCUSSION
It is now well established that the Wnt/
Although the role of the Par/aPKC PCP complex in epithelial polarity has been well established both in
In addition, by knocking down the expression of PAR-3 or aPKC-ζ, we observed a significant increase in hCMEC/D3 permeability to the paracellular diffusion marker LY (Figure 3), together with a partial loss of ZO-1 expression at cell–cell contacts (Figure 4) and a delayed TJ reassembling after hyperosmotic shock (Figure 5). We recently reported similar results after siRNA-mediated deletion of cldn-5 expression in hCMEC/D3 cells,
6
strongly suggesting that the Par/aPKC PCP complex, like the TJ protein cldn-5, is actively involved in maintaining TJ integrity in brain endothelial cells. Compared with the very transient opening of the BBB
Our data are in agreement with the previous report that, although PAR-3 does not interact directly with cldns, it is recruited to TJ complexes through binding to JAM proteins. 32 In epithelial cells, two alternative polarization complexes, the Crumbs and Scribble complexes, are known to be involved in TJ formation and apicobasal polarization and to functionally interact with the Par/aPKC PCP complex. 27 Inasmuch as a recent report indicated that endothelial cells also express Scribble, 33 it is tempting to speculate that this complex might also contribute to the apicobasal polarity of brain endothelial cells and BBB integrity.
Activation of the noncanonical Par/aPKC PCP complex has been extensively described as a signaling pathway downstream of the interaction between Wnt morphogens and their membrane receptors, the Fzd family and associated coreceptors.
34
Because Wnts are now considered as key regulators of BBB formation and integrity, we intended here to explore the putative role of the Wnt/Par/aPKC PCP pathway in TJ integrity in hCMEC/D3 cells. The Wnt factor Wnt5a was previously described to selectively activate this pathway in various cell types,25, 35 to control multiple cellular functions (cell migration, proliferation, differentiation) and to promote proliferation or survival, particularly in endothelial cells.
36
These pleiotropic effects are mediated through Wnt5a binding to several Fzds, including Fzd-2, Fzd-4, Fzd-5, and Fzd-7.
37
In line with these observations, here we report in hCMEC/D3 cells, which express all these Fzds (Table 1), that Wnt5a induced JNK phosphorylation, a hallmark of activation of the Wnt/Par/aPKC PCP pathway,
38
without activating the canonical Wnt/
In conclusion, we propose that Wnt factors control BBB integrity through activation of multiple signaling pathways, including the canonical Wnt/
Footnotes
The authors declare no conflict of interest.
