Abstract
The choroid plexuses (CPs) form a protective interface between the blood and the ventricular cerebrospinal fluid (CSF). To probe into the pathways by which CPs provide brain protection, we sought to evaluate the efficiency of glutathione conjugation in this barrier as a mechanism to prevent the entry of blood-borne electrophilic, potentially toxic compounds into the CSF, and we investigated the fate of the resulting metabolites. Rat CPs, as well as human CPs from both fetal and adult brains, displayed high glutathione-S-transferase activities. Using an in vitro model of the blood-CSF barrier consisting of choroidal epithelial cells cultured in a two-chambered device, we showed that glutathione conjugation can efficiently prevent the entry of 1-chloro-2,4-dinitrobenzene (CDNB) into the CSF, a model for electrophilic compounds. The duration of this enzymatic protection was set by the concentration of CDNB to which the epithelium was exposed, and this barrier effect was impaired only on severe epithelial intracellular glutathione and cysteine depletion. The conjugate was excreted from the choroidal cells in a polarized manner, mostly at the blood-facing membrane, via a high-capacity transport process, which is not a rate-limiting step in this detoxification pathway, and which may involve transporters of the ATP-binding cassette c(Abcc) and/or solute carrier 21 (Slc21) families. Supplying the choroidal epithelium at the blood-facing membrane with a therapeutically relevant concentration of N-acetylcysteine sustained this neuroprotective effect. Thus, glutathione conjugation at the CP epithelium coupled with the basolateral efflux of the resulting metabolites form an efficient blood-CSF enzymatic barrier, which can be enhanced by pharmacologically increasing glutathione synthesis within the epithelial cells.
Keywords
Introduction
The cerebrospinal fluid (CSF) circulatory system fulfills several functions, which range from mechanical and buffering protection of the brain to drainage of the cerebral parenchyma, neuroimmune regulation and paracrine transmission (Davson and Segal, 1996; Kivisakk et al, 2003; Knopf et al, 1995; Nicholson, 1999; Redzic and Segal, 2004). The choroid plexuses (CPs), localized in all four ventricles of the brain, are responsible for most CSF secretion. They are formed by an epithelium enclosing a highly vascularized stroma. This epithelium displays a large surface of exchange, at the interface between the blood and the ventricular CSF. In addition to CSF secretion, this epithelium also fulfills neuroprotective functions by preventing the entry of various blood-borne compounds into the newly formed CSF, and by actively clearing deleterious organic ions out of the CSF. This is achieved, with some similarities with the brain capillary endothelial cells that form the blood–brain barrier, by interepithelial tight junctions, and by different transporters of the ABC, SLC21 and SLC22 families (Strazielle and Ghersi-Egea, 2000; Strazielle et al, 2004). Xenobiotic metabolism at the choroidal tissue also participates in this neuroprotective function. In particular, the choroidal epithelium can form an efficient enzymatic barrier preventing the entry into the CSF of neurotoxic compounds by a process of conjugation to glucuronic acid, and subse quent active efflux into the blood of the formed glucurono-conjugate (Strazielle and Ghersi-Egea, 1999).
In detoxifying organs such as the liver, conjugation to glutathione (Throughout the text, glutathione refers to the reduced, SH function-bearing tri-amino-acid form of the compound.) represents a mechanism of protection towards different electrophilic drugs or endobiotics which are potentially toxic, provided the resulting conjugates are actively eliminated from the cells in which they are produced and further cleared from the body via the bile tract or the urine (Chasseaud, 1979; Hayes et al, 2005; Hayes and Pulford, 1995; Rinaldi et al, 2002; Whalen and Boyer, 1998). Immunohistochemical studies have located various classes of glutathione-S-transferases in the rodent CP (Cammer et al, 1989; Johnson et al, 1993; Philbert et al, 1995), and this tissue also comprises the complete set of enzymes involved in glutathione synthesis (reviewed in Ghersi-Egea and Strazielle, 2001; Monks et al, 1999). We thus hypothesized that a glutathione-linked detoxification process contributes to the barrier between the blood and the CSF and initiated a functional investigation of this mechanism.
In this paper, we report a high enzymatic glutathione conjugation activity in both human and rodent choroidal tissues. Then, using a highly differentiated in vitro cellular model of the choroidal epithelium that allows to study transport, metabolic and polarized secretion processes at the blood-CSF barrier (Strazielle and Ghersi-Egea, 1999; Strazielle et al, 2003a, b), we investigated the neuroprotective capability of this interface towards electrophilic, potentially toxic species, taking 1-chloro-2,4-dinitrobenzene (CDNB) as a model for such molecules. We show that the CSF can be protected from blood-borne reactive molecules by a glutathione-dependent detoxification mechanism involving both a metabolic and a transport step, and that the efficiency of this neuroprotective choroidal function can be pharmacologically sustained.
Materials and methods
Choroid Plexus Isolation and Primary Cultures of Choroidal Epithelial Cells
Autopsic human samples of choroidal tissue were obtained according to the ethical guidelines approved by INSERM, from the Service d'Anatomie et Cytologie Pathologiques, Hôpital E Herriot, and the Service de Neuropathologie, Hôpital Neurologique centralized via the Biological Resource Center NeuroBioTec Banques, Lyon. Post-mortem delays ranged from 8 to 36 h. Tissue samples 6 and 7 were surgery resections of choroidal tissue, also obtained from NeuroBioTec Banques.
Animal care and procedures have been conducted according to the guidelines approved by the French Ethical Committee (decree 87 to 848) and by the European Community directive 86 to 609-EEC. Oncins France Strain A (OFA) OFA males or OFA timed pregnant rats (200 to 240 g) were obtained from Harlan, Gannot, France. Choroid plexuses from both adult male and newborn rats were sampled intact under a stereomicroscope, rinsed in ice-cold 0.32 mol/L sucrose, 50mmol/L phosphate, 1 mmol/L ethylene diamine tetraacetate (EDTA), 0.1 mmol/L dithiothreitol, pH 7.4 buffer, and kept at −80°C until used for enzymatic measurement. Alternatively, 1- or 2-day-old rat CPs, sampled in sterile conditions and kept in fetal calf serum supplemented Dulbecco's modified Eagle's medium (DMEM)/F12 (1/1) medium, were used to prepare primary cultures of epithelial cells, which were seeded on Transwell-Clear filter inserts (1 cm2 surface, 0.4 μm pore size, Costar Plastics, Cambridge, MA, USA). Inserts were precoated on the upper side with laminin (Becton Dickinson, Bedford, MA, USA), as described previously in detail (Strazielle and Ghersi-Egea, 1999). Cultures were initiated from fourth ventricle choroidal tissue, unless otherwise stated. Laminin-coated inserts (without cells) were kept in the same conditions. Experiments were performed within 5 to 7 days after confluence.
Permeability and Transport Studies
Culture inserts with and without cells were rinsed once on both sides before initiating the transfer measurement. All incubations were performed on a rotating platform (250r.p.m.) at 37°C, in Ringer's solution-N-hydroxyethyl-piperazine-N-2-ethanesulfonic acid (HEPES) buffer (in mmol/L: 150 NaCl, 5.2 KCl, 2.2 CaCl2, 0.2 MgCl2, 6 NaHCO3, 2.8 glucose, 5 HEPES, pH 7.4). Paracellular permeability was measured using [14C]sucrose (Amersham, Little Chalfont, UK) as described previously in detail (Strazielle and Ghersi-Egea, 1999; Strazielle and Preston, 2003). Briefly, after addition of the sucrose-containing buffer to the upper compartment, the clearance volume of the tracer was measured during four consecutive time intervals, and a permeability × surface area coefficient was calculated for both laminin coated-filters (PSf) and laminin-coated filters with cells (PSt). The permeability × surface area of the epithelial cell monolayer (PSe) was then obtained according to the equation 1/PSe = 1PS 1/PSf, and the permeability coefficient Pe (in cm/min) was calculated by dividing the value of PSe by the surface area of the insert.
For CDNB permeability measurement, the cells were incubated with variable concentrations of CDNB (Sigma, St Louis, MO, USA) in the lower compartment. At various time intervals thereafter, the inserts were transferred into another well containing a similar concentration of CDNB, and 3/4 of the volume in the upper compartment was sampled and replaced with fresh solution kept at 37°C. The clearance volume was then calculated for each time interval, taking into account the residual amount remaining in the upper compartment from the previous time, as described previously (Strazielle and Ghersi-Egea, 1999; Strazielle and Preston, 2003). The media from the upper and lower compartments were also analyzed for their dinitrophenyl-glutathione (GS-DNP) content, with a similar correction for the data generated from upper compartment medium samples.
High-Performance Liquid Chromatography (HPLC) Analysis of 1-Chloro-2,4-Dinitrobenzene and Dinitrophenyl-Glutathione
Analysis was performed on a LC10 Shimadzu system (Duisburg, Germany) as follows: Samples (20 or 40 mL) were applied with an autoinjector device cooled at 4°C, onto an Ultraphere ODS RP-18 analytical column (5 mm, 4.6 × 250 mm; Beckman, Fullerton, CA, USA). The elution of GS-DNP was isocratic, using a mobile phase constituted by a 30/10/60 mixture of methanol/0.1 mol/L K-phosphate buffer, pH 6/water pumped at a constant rate of 1 mL/min. After 13 mins, the methanol concentration was increased from 30% to 50% in 2 mins, and kept at 50% for an additional 15 mins, to allow CDNB elution. Absorbance of the effluent was monitored, respectively, at 340 and 250 nm for GS-DNP and CDNB. The retention times of the two compounds were, respectively, 9 and 26 mins. Quantification was performed using CDNB and GS-DNP standards.
The latter was produced by enzymatic synthesis and quantified as follows: CDNB (200 μmol/L) was incubated in 1 mL of 0.1 mol/L K-phosphate buffer, pH 6.5, with an excess of glutathione (1 mmol/L, Sigma) and newborn rat liver homogenate (60 μg of proteins) as a source of enzyme for 20 mins at 37°C under continuous agitation. The enzymatic reaction was stopped by transferring the tubes on ice and adding 500 μL of acetonitrile. The incubation medium was centrifuged at 8000g for 10 mins, and the supernatant transferred in HPLC glass vials. Control incubations included incubation without enzymes, and/or glutathione, and/or CDNB. The exact GS-DNP concentration in the standard solution was calculated by coupling spectrophotometric measurement of the samples to their HPLC analysis at 340 nm. The optical density (OD)1 recorded for the supernatant coming from the incubation of CDNB with the liver homogenate in the absence of exogenous glutathione was the result of the absorbances of CDNB, the homogenate, and of GS-DNP formed by the endogenous glutathione present in the homogenate. The difference between the OD2 measured in the supernatant from the complete incubation and OD1 corresponded to the additional production of GS-DNP due to the exogenous glutathione added. The GS-DNP concentration resulting from this additional production is equal to (OD2-OD1)/ε, where ε is the extinction coefficient of GS-DNP (9600/ mol cm), and is directly related to the difference between the surface area of the GS-DNP peaks eluted when equivalent volumes of supernatant from incubation reactions with and without exogenous glutathione were injected into the HPLC. From this equivalence, the total GS-DNP concentration in the standard solution could be accurately determined. This determination was performed in triplicate on three separate enzymatic reaction tubes, which were then pooled before establishing the final GS-DNP concentration by HPLC (87.5 μmol/L), and kept at 80° C until use. Incubations without CDNB did not generate any peak with the retention time of GS-DNP.
Measurement of Glutathione and Cysteine Intracellular Content
Cell-covered filters were rinsed in ice-cold Ringer's solution HEPES buffer, the membranes were quickly cut from their plastic support, placed in a microcentrifuge tube and covered with 200 μL of 500 μmol/L 5,5′-dithio-bis(2-nitrobenzoic) acid (DTNB, Sigma) prepared in 10 mmol/L K-phosphate buffer, pH 7.4. After a freeze–thaw cycle, the biological material was further homogenized with a pellet mixer, heated at 37°C for 10 mins to allow for a complete reaction of DTNB with SH-containing residues, and centrifuged at 8000g for 10 mins. Standard solutions of mixed cysteine and glutathione were prepared extemporaneously and processed in a similar way. The supernatants were analyzed for their content in glutathione- and cysteine-derived disulfides by HPLC, as described previously (Strazielle et al, 2003b).
RNA Isolation and Reverse Transcriptase-Polymerase Chain Reaction (RT-PCR) Analysis
Total cellular RNA was isolated from CPs of newborn rat and from CP epithelial cells, either freshly isolated or after 7 days in culture, according to Chomczynski and Sacchi (1987), and used as a template for reverse transcription using oligo-dT as a primer. Specific primers corresponding to nucleotides 35 to 60 and 994 to 1017 of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) complementary deoxyribonucleic acid (cDNA) sequence (Genbank accession # NM_017008), nucleotides 1849 to 1866 and 2218 to 2235 of the rat organic anion transport polypeptide oatp2 (Slc21a5) cDNA sequence (Genbank accession # U88036), nucleotides 2456 to 2475 and 2830 to 2849 of the rat multidrug resistance protein mrp1 (Abcc1) cDNA sequence (Genbank accession # AJ277881) and nucleotides 686 to 705 and 722 to 742 of the rat multidrug resistance protein mrp4 (Abcc4) cDNA sequence (Genbank accession # NM_133411) were used to amplify fragments of 983, 387, 394 and 57 bp, respectively, in a Robocycler Gradient 96 (Stratagene, Amsterdam, NL, USA).
Enzymatic and Protein Content
Choroidal tissue was homogenized in 0.32 mol/L sucrose, 50 mmol/L K-phosphate, 1 mmol/L K-EDTA, 0.1 mmol/L dithiothreitol buffer, pH 7.4, using a glass–glass homogenizer. Glutathione-S-transferase activity was determined in a dual-beam Cary 100 spectrophotometer according to the method of Habig et al (1974). Assays were conducted in 0.1 mol/L K-phosphate, pH 6.5 at 25°C, using 1 mmol/L glutathione and 1 mmol/L CDNB as substrates. The complete assay mixture without the protein homogenate was used in the reference cuvette. The activity was strictly dependent on glutathione addition and totally inhibited on addition of 250 μmol/L of the glutathione-S-transferase inhibitor ethacrynic acid, in all samples analyzed. For kinetic parameter determination, concentrations of either CDNB or glutathione were varied from 10 μmol/L to 1 mmol/L. The total protein content of the samples was determined by the method of Peterson (1977), with bovine serum albumin as the standard.
Results
Choroid Plexuses from Both Human and Rodent Display a High Glutathione-S-Transferase Activity Toward Nitro-Compounds
We previously showed that a strong glutathione-S-transferase activity, as measured toward nitro-compounds, was associated to the CPs issued from both lateral and fourth ventricles of newborn rats (Strazielle and Ghersi-Egea, 1999). In the present study, 12 choroidal tissue samples obtained from either fetal or adult human brain were analyzed for this enzymatic activity. An activity was readily detected in all human tissues (Table 1). For three cases (3, 4 and 5), cortical parenchyma tissue was also available, and the recorded activities (respectively, 32.6, 50 and 35.5 nmol/min mg protein) were several-fold lower than those recorded in the corresponding choroidal tissue. In addition, the choroidal activities were comparable to those measured in homogenates from freshly isolated CP of newborn and adult rats, indicating that the strong glutathione-dependent metabolic capacity of the choroidal tissue reported previously in rodent (Strazielle and Ghersi-Egea, 1999) is also a characteristic of the human tissue.
Glutathione-S-transferase activity in individual human and in rat choroidal tissue samples
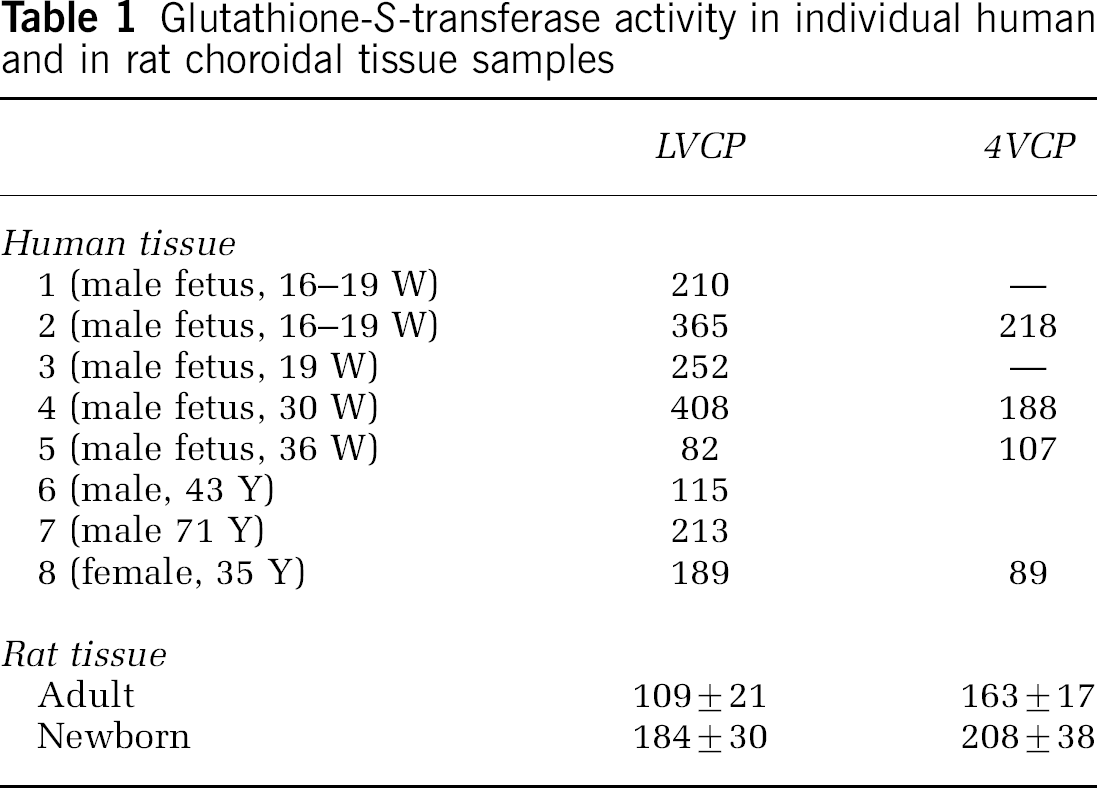
The specific activity is measured toward CDNB in whole-tissue homogenates, and is expressed as nmol/min mgprotein. Values from individual human samples represent the average of 3 measurements, which do not differ from the mean value by more than 13%. W: weeks of pregnancy, Y: year-old, —: tissue not available. Rat data are means7s.d. from four to six different pools of animals. LVCP: sample from lateral ventricle CP, 4VCP: sample from fourth ventricle CP.
Glutathione Conjugation forms an Effective Metabolic Barrier at the Choroidal Epithelium
We investigated whether glutathione conjugation at the CP influences the transfer of blood-borne electrophilic, potentially deleterious compounds across the blood-CSF barrier. The choroidal epithelium was reconstituted on the surface of a porous filter. 1-Chloro-2,4-dinitrobenzene was applied in the basolateral, that is, blood/stroma, compartment, and its rate of appearance in the apical, that is, CSF, compartment was followed by HPLC. For a 2 μmol/L concentration, the clearance rate of CDNB measured across the choroidal epithelium reconstituted from the fourth ventricle CP (Figure 1A) or from the lateral ventricle CP (not shown) was very low for the entire duration of the experiment (PSt = 0.06 ± 0.03 μL/min), and was below the clearance rate of sucrose (0.11 ± 0.03 μL/min), which reflects the paracellular permeability of the cell monolayer. This indicates that, when CDNB is used at the 2 μmol/L concentration, the choroidal epithelial barrier completely prevents its basolateral-to-apical transfer, despite the lipophilic nature of the compound, which readily crosses cell membranes (Boyer et al, 1983). A full barrier effect was also observed when higher concentrations of CDNB were used, but for a shorter duration of 15 and 7 mins for concentrations of 10 and 20 μmol/L, respectively. Thereafter, CDNB clearance increased, indicating that the epithelium ability to prevent CDNB entry into the CSF compartment became impaired. The increase in CDNB clearance rate was sharper when 20 μmol/L CDNB was used, as compared with 10 μmol/L (Figure 1A). Raising further the concentration of CDNB led to an even more rapid appearance of the compound in the apical compartment, but did not increase significantly the 15-to-30 min clearance rate measured at 20 μmol/L (data not shown), which therefore is considered maximal. Changes in CDNB transfer rate were independent of the epithelial paracellular permeability, which remained unchanged at all CDNB concentrations used (data not shown).
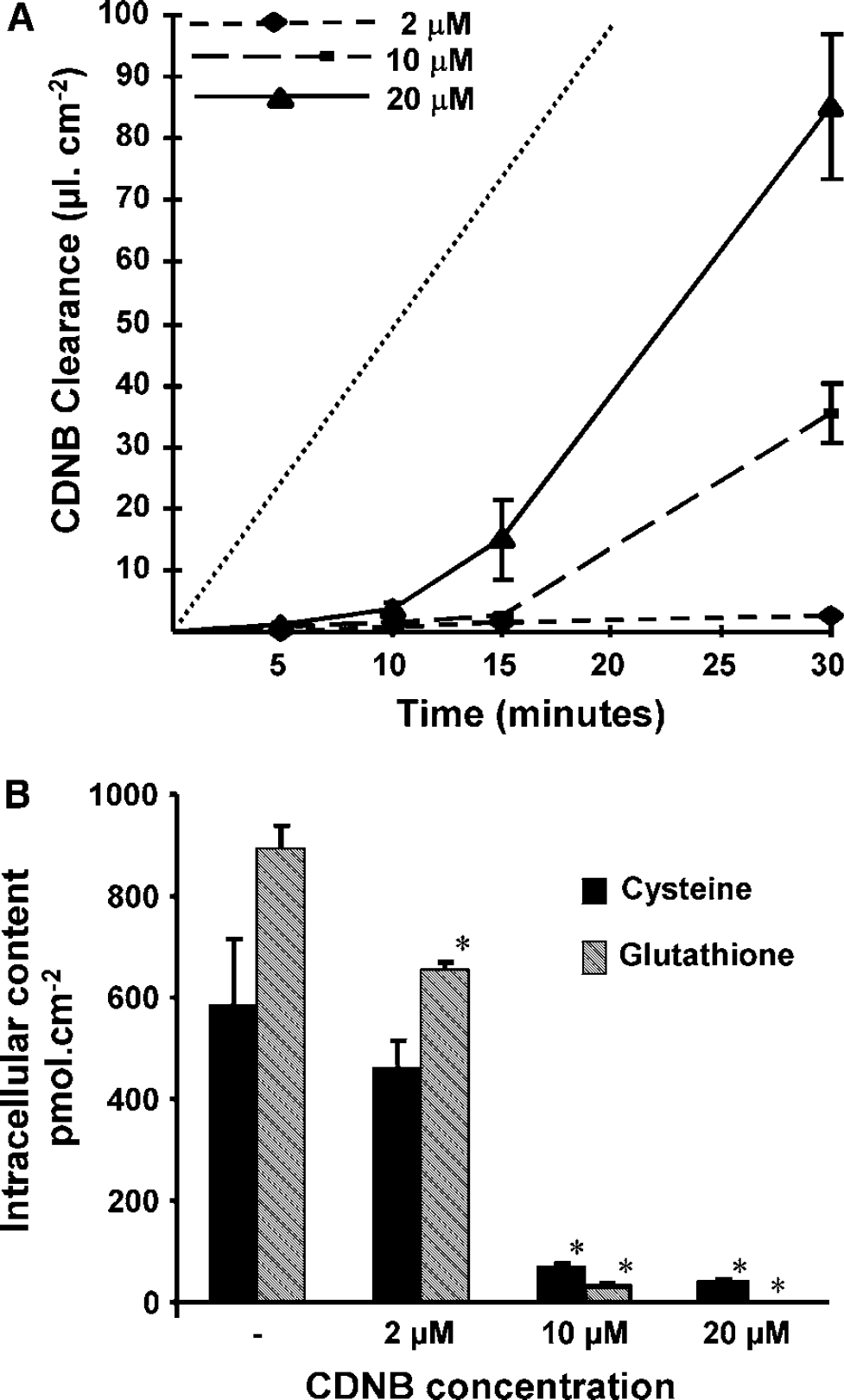
Glutathione-dependent metabolic barrier toward the transcellular passage of CDNB across the choroidal epithelial cells.
At the end of the CDNB transfer experiment, the intracellular concentrations of glutathione and its precursor cysteine were measured (Figure 1B). In cells exposed basolaterally to 2 μmol/L CDNB, the intracellular concentration of glutathione was reduced by only 25% by comparison to the control value, while it was drastically reduced and undetectable, respectively, in cells exposed to 10 and 20 μmol/L CDNB. Similarly, intracellular cysteine concentration decreased strongly in cells exposed to both highest doses of CDNB. These data show that the dose- and time-dependent increase in the transcellular clearance of CDNB (Figure 1A) is paralleled by a shortage of cellular glutathione and of its precursor, suggesting that glutathione is essential for the epithelial cells to function as an efficient barrier.
The subsequent HPLC analysis of both apical and basolateral medium revealed that the glutathione conjugate of CDNB (GS-DNP) was indeed produced and excreted by the cells, thereby providing a link between glutathione-dependent metabolic conjugation and barrier efficacy. The preparation of a GS-DNP standard solution (see Materials and methods) allowed us to quantify the released metabolite. When the concentration of 2 μmol/L CDNB was used, the metabolite excretion was continuous and linear over time in both compartments (Figure 2), thus allowing the calculation of an average export rate across each membrane (Figure 2, insert). The efflux was strongly polarized with 79% of the conjugate excreted at the basolateral, that is, blood-facing membrane (Figure 2, insert). The total amount of GS-DNP excreted over the 30-min period, (252 ± 5 pmol/cm2) accounts for the lack of CDNB clearance across the epithelial cells (see Discussion). When CDNB concentration was raised to 10 and 20 μmol/L, a proportional increase in GS-DNP production rate was observed with a similar polarity of secretion for as long as the barrier effect was complete (not shown).
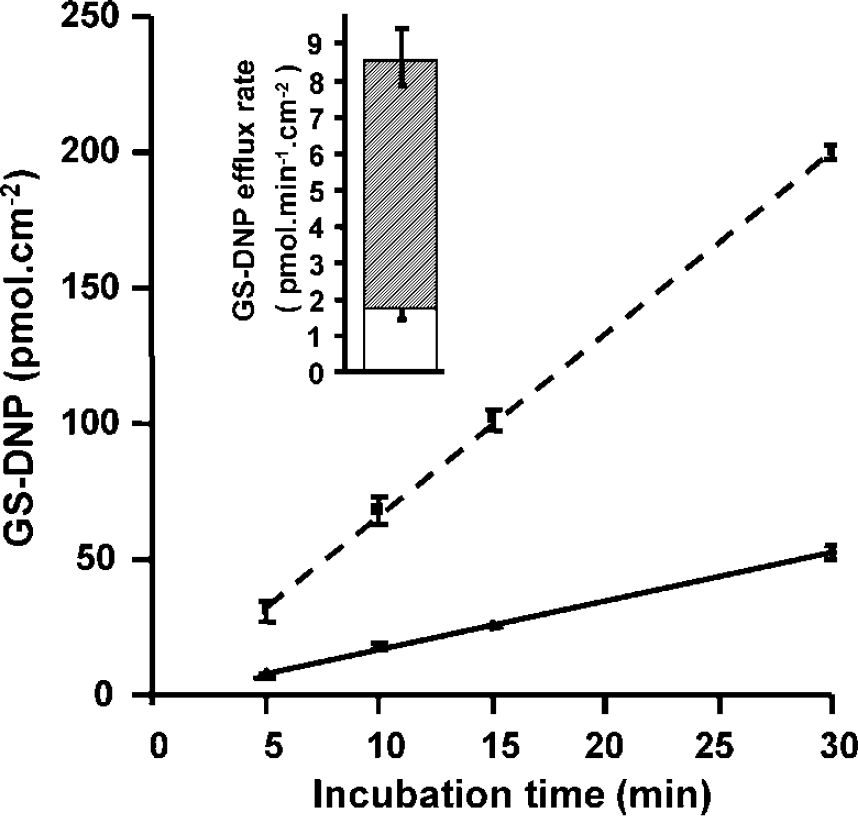
Polarized efflux of GS-DNP from CP epithelial cell monolayers. Cells from the fourth ventricle CPs were incubated with 2μmol/L CDNB (as in Figure 1) and samples from both apical and basolateral chambers were analyzed for GS-DNP by HPLC. The cumulated amount of GS-DNP is plotted versus time. Each time point is a mean value ± s.d. of three filters. The efflux process was linear in both the basolateral and apical compartments. Dashed line: basolateral compartment, continuous line: apical compartment. Inset: The export rates calculated from the clearance curves (mean + s.d.) indicate a strongly polarized efflux with 79% of the metabolite released at the basolateral membrane of the cells. Only half s.d. bars are presented. Downright-directed bars refer to single compartment data; light gray area: apical release; dashed area: basolateral release.
The kinetic parameters for CDNB conjugation were determined in newborn CP homogenates toward either CDNB or glutathione, the corresponding cosubstrate being added in saturating concentration (1 mmol/L). Affinity constants (Km) deduced from either the Lineweaver–Burk, Eadie–Hofstee or Hanes–Woolf representations did not vary by more than 11% of the average values, which were 51 μmol/L for CDNB and 74 μmol/L for glutathione. The maximal velocity measured (Vmax) were, respectively, 204 and 188 nmol/min mg protein. The low Km over Vmax ratios deduced from these data are in agreement with the finding of an efficient enzymatic blood-CSF barrier toward CDNB (Figure 1A). In particular, the affinity constant measured for glutathione is much lower than the intracellular concentration of glutathione estimated in the millimolar range, supporting the efficacy of the metabolic process for as long as the intracellular glutathione is not strongly depleted.
The polarized export of the glutathione conjugate suggests the involvement of (a) transport protein(s) asymmetrically distributed between both membrane domains. To probe into the kinetic parameters of the efflux system, we modulated the intracellular GS-DNP production rate or glutathione concentration, and followed the efflux of the metabolite in both compartments. Incubating the cells with a high (200 μmol/L) CDNB concentration was immediately followed by an important and polarized export of the glutathione conjugate (Figure 3). Over time, the reduced production of GS-DNP arising out of the shortage of intracellular glutathione resulted in a progressive decrease in its export rate that was much sharper at the basolateral membrane, and consequently led to a loss of polarity. This suggests that the apical and basolateral transport systems, not sharing the same kinetic properties, are different. Of interest, the polarity was lost for an overall export rate similar to the one measured when the 2 μmol/L CDNB concentration leading to a highly polarized efflux was used (compare the polarity in Figure 3, time interval 17.5 to 22.5 mins, and that in Figure 2, insert, for which total export rates were 8.7 ± 1.2 and 8.4 ± 0.2 pmol/min cm, respectively). This indicates that intracellular GS-DNP concentration is not the only factor setting the export rate at the basolateral membrane. 1-Chloro-2,4-dinitrobenzene concentration (200 μmol/L), like lower concentrations, did not alter the integrity of the paracellular barrier, as evaluated by the measurement of sucrose permeability, which remained low and constant for at least 1 h (Pe: 0.117 ± 0.03 × 10−3 cm/min in CDNB exposed cells versus 0.110 ± 0.029 × 10−3 cm/min for control cells). This indicates that the loss of polarity is not due to a general toxicity exerted by the electrophilic compound on the cellular tight junction integrity, which is necessary for effective epithelial polarity.
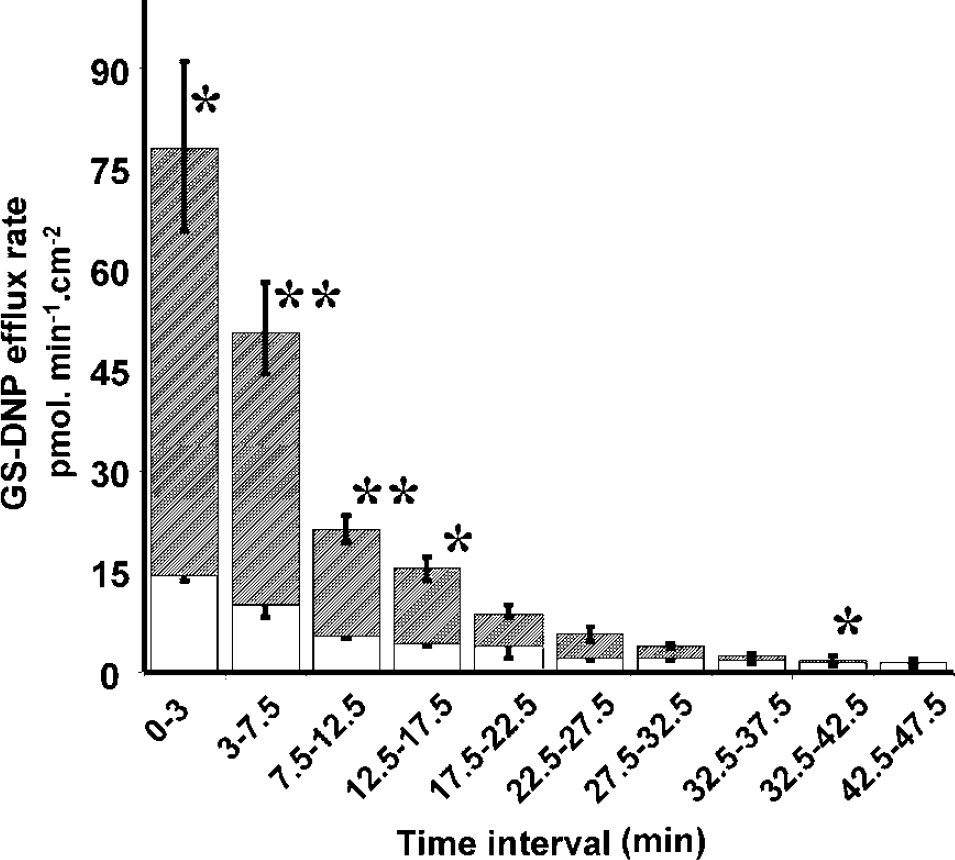
Time-dependent changes in the apical and basolateral efflux rate of GS-DNP after exposure to high CDNB concentration. Cells were exposed to 200 μmol/L CDNB and apical and basolateral compartments were sampled at various time intervals and assayed for GS-DNP by HPLC. For each time interval, average apical (light gray area) and basolateral (dashed area) efflux rates were calculated. Results are expressed as mean ± s.d., n = 3. Only half s.d. bars are presented. Downright-directed bars refer to single compartment data, upward-directed bars refer to the total GS-DNP excretion.*.** Basolateral export rate statistically different from apical export rate, P < 0.05 and 0.01, respectively, one-tailed Student's t-test for unequal variance. Basolateral excretion was undetectable in the last time interval.
We then attempted to modulate the de novo synthesis of intracellular glutathione by incubating the cells exposed to 20 μmol/L CDNB in the basolateral compartment, with a cocktail of amino acids involved in glutathione synthesis and with N-acetylcysteine (NAC), as a donor of cysteine. When CDNB was used alone, the export of GS-DNP was polarized and linear for 10 mins. In accordance with the previously described increase in CDNB clearance (Figure 1), the metabolite export rate thereafter decreased with a concomitant loss of polarity (5 pmol/min excreted in each compartment, compare the 15- to 30-min export rates at the two membranes, square symbols in Figure 4A). Adding the amino acids alone had limited effects on GS-DNP formation and excretion, suggesting that the rate of glutathione synthesis was not efficiently increased. By contrast, adding NAC at the therapeutic concentration of 15 μmol/L to the amino-acid cocktail yielded a strong increase in the export of GS-DNP, indicating an augmented glutathione availability within the cell. Only the basolateral export was increased, which led to the restoration of the polarity to a 1-to-4 apical-to-basolateral ratio. These data show that the apical efflux has a low saturation level in comparison to the basolateral efflux. HPLC analysis of CDNB in the apical compartment showed that CDNB clearance rate was little affected by amino-acid supply in the basolateral compartment, but was strongly reduced when NAC was also provided (Figure 4B). This is in accordance with the increased GS-DNP production on NAC treatment (Figure 4A), and indicates that NAC is efficient to restore a metabolic barrier when glutathione is extensively used as a conjugation cosubstrate.
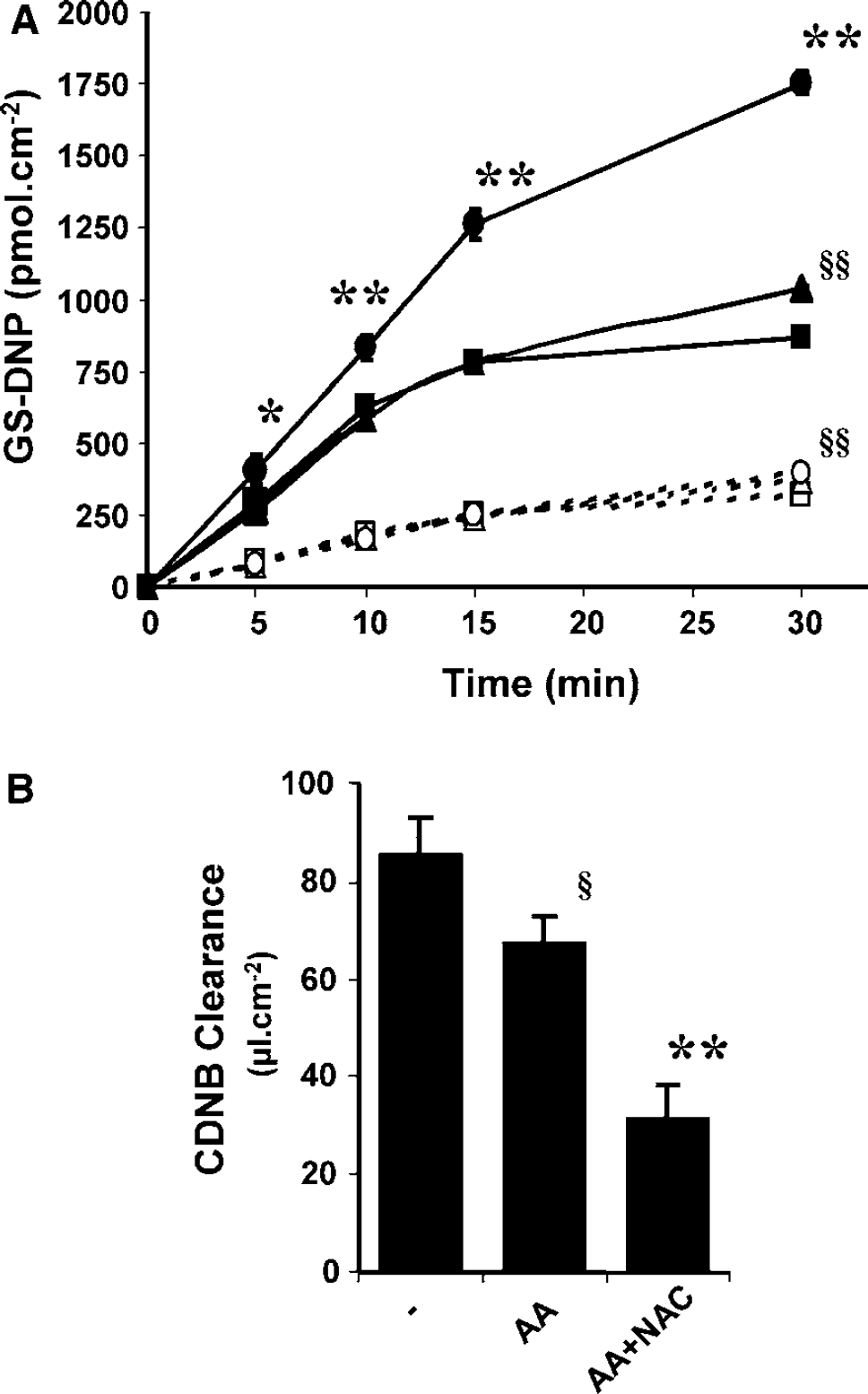
Effect of glutathione amino-acid precursors and NAC on GS-DNP efflux and CDNB transcellular passage across the CP epithelial cell monolayer. Cells were exposed at their basolateral side to 20 μmol/L CDNB-containing buffer with or without a cocktail of amino acids (AA) involved in glutathione synthesis and NAC. The amino-acid cocktail composition was 250 μmol/L glycine, 50 μmol/L L-methionine, 200 μmol/L serine, 100 μmol/L glutamic acid and 60 μmol/L L-cysteine (all concentrations similar to plasma and CSF concentrations; Davson and Segal, 1996). N-acetylcysteine was used at the pharmacological concentration of 15 μmol/L.
With respect to the molecular identity of the transport proteins responsible for GS-DNP efflux at the CP, two members of the Abcc subfamily of multidrug-resistant proteins, Abcc1 and 4, as well as Slc21A5, an organic anion transport protein, all the three susceptible to accept amphiphilic molecules such as glutathione conjugates as substrates, have been shown to be localized at the basolateral membrane of the epithelial cells from lateral ventricle CP (reviewed in Strazielle and Ghersi-Egea, 2005a). We investigated the expression of these trasnporters in cultured CP epithelial cells by RT-PCR, in comparison to freshly isolated CPs from both lateral and fourth ventricles. All three genes are expressed in CPs from both types as well as in corresponding CP epithelial cells after 7 days of culture (Figure 5).
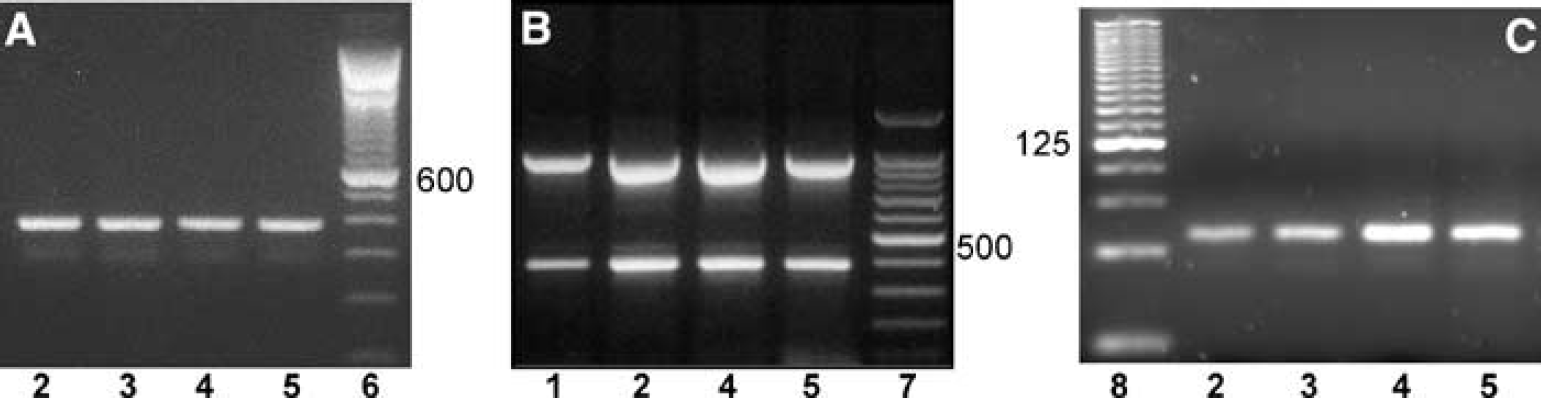
Expression of Abcc1, 4 and Slc21a5 in CPs and choroidal epithelial cells. Total mRNA preparations isolated from newborn rat fourth ventricle (lane 3) and lateral ventricle (lane 2) CP, 7-day-old lateral ventricle (lane 4) and fourth ventricle (lane 5) CP epithelial cells, were reverse transcribed with oligo-dT. The 387, 394 and 57-bp-long fragments amplified using oligonucleotides specific for Slc21a5
Discussion
This work shows that the glutathione S-transferase activities present at high levels in CP in humans as in rodents, and coupled to a polarized basolateral excretion of glutathione conjugates from the choroidal epithelial cells, represent an efficient enzymatic barrier mechanism at the blood-CSF barrier.
The in vitro cellular model used in this work retains the morphological characteristics of the blood-CSF barrier and presents functional tight junctions, as illustrated by a close match between the permeability coefficients determined in vivo and in vitro for various polar and slightly lipophilic compounds (Strazielle and Ghersi-Egea, 1999; Strazielle et al, 2003a). The model also displays the polarity, transport and secretion properties of the in vivo choroidal epithelium and, importantly, its metabolic capacity toward exogenous molecules, including glutathione-S-transferase activity, is preserved (Strazielle and Ghersi-Egea, 1999, 2005b; Strazielle et al, 2003a, b). We showed that the basolateral-to-apical flux of the cytotoxic compound CDNB across the choroidal epithelium was strongly impeded despite its lipophilic nature. This cellular barrier effect was observed for basolateral exposure of the choroidal epithelial cells to CDNB at concentrations up to at least 20 μmol/L; yet it was limited in duration for the higher concentration used. Exposure of the choroidal epithelial cells to CDNB resulted in its conjugation into GS-DNP. The cellular export of this metabolite occurred predominantly at the basolateral, that is, blood-facing, membrane, a mechanism favoring its further elimination from the body. For this metabolic process to fully explain the barrier action of the epithelium, the rate of metabolism Rm needs to be equal to the theoretical rate of CDNB diffusion (Rd) across the cell monolayer. The former could be accurately quantified after producing a metabolite standard. The latter can be estimated according to the equation: Rd = PeH × C, where C is the concentration of CDNB in the basolateral compartment, and PeH is the permeability coefficient of CDNB across the cell monolayer measured for a high CDNB concentration during the 15- to 30-min interval of transfer (Figure 1A), that is, in conditions in which metabolism is overwhelmed and becomes insignificant, leading to maximal diffusional transfer. Accordingly, when CDNB was used at 2 μmol/L, and after deduction of the minute amount of compound that reached the apical compartment, Rd could be estimated to be 267 ± 29 pmol/cm2 per 30 mins (n =5 from different PeH measurements). This is indeed similar to the rate of glutathione conjugate production (252 ± 5 pmol/cm2 per 30 mins, calculated from Figure 2). This conjugation/efflux mechanism at the CPs, translating into a metabolic barrier, represents therefore a new facet of the neuroprotective roles of glutathione, besides its known inactivating effect toward reactive oxygen species in glial and neuronal cells (reviewed in Dringen, 2000; Dringen and Hirrlinger, 2003), and toward peroxides involved in traumatic cerebrovascular injury (Gidday et al, 1999).
For CDNB concentrations up to 20 μmol/L, neither conjugation to glutathione nor the overall conjugate export was saturated, as the barrier efficacy was independent of the concentration applied to the epithelium during the first time interval evaluated (Figure 1A). This further emphasizes the potentiality of the protective mechanism, and highlights a difference between the glutathione-dependent barrier and the barrier mechanism linked to glucuronidation previously described in this interface (Strazielle and Ghersi-Egea, 1999). As for the latter, the factor limiting the efficiency of the metabolic barrier was the rate of the enzymatic reaction, while, in the former, the efficiency of the barrier is linked mainly to the availability of the cofactor glutathione, explaining the biphasic aspect of CDNB clearance curves across the epithelium when higher concentrations are used. Strategies to increase the duration and efficiency of this biochemical barrier should therefore aim at increasing the overall availability of glutathione. Cysteine is the amino-acid precursor that limits the rate of glutathione synthesis in several brain cell types (Dringen, 2000; Dringen et al, 1999). Being quickly oxidized, cysteine is not found in significant amounts in biological fluids such as plasma and CSF. Our data show that supplying the cells with therapeutically relevant concentrations of the mucolytic drug NAC, acting as a stable source of cysteine, increases the rate of glutathione synthesis (a requisite for the augmented production of glutathione conjugate that we observed). It thus enhances the barrier efficacy of the epithelium toward high concentrations of CDNB, and also favors the basolateral excretion of the detoxification product. Reinforcing brain protection at the CP by strengthening its enzymatic barrier capacity, especially in case of exposure to pharmacological compounds with potential central side effects or to toxicants, should therefore be an additional benefit of this drug in addition to its previously pointed out protective effects on other detoxifying organs such as the liver and the kidney, and its potential therapeutic indication in inflammatory diseases or carcinogenesis (Chyka et al, 2000; Tepel and Zidek, 2004; Zafarullah et al, 2003).
As expected from the anionic nature of the glutathione conjugate, and further indicated by its polarity, GS-DNP efflux from the CP epithelial cells is transporter-mediated and occurs for the main part via a high-capacity process localized at the basolateral membrane. Interestingly, while the decrease in CDNB conjugation arising out of the shortage of cellular glutathione led to a strong reduction in the metabolite export rate at the basolateral membrane, it affected less the low-efficiency export process at the apical membrane (Figure 3), indicating that mechanisms with different kinetics govern the efflux at each membrane. Sustaining this view, we observed that, for similar total glutathione conjugate production and secretion, the basolateral-to-apical polarity was maintained when intracellular glutathione concentration remained high (Figure 2), but was lost when glutathione depletion occurred, as during exposure to high concentrations of CDNB (Figures 3 and 4). This indicates that the intracellular GS-DNP concentration is not the only factor driving its rate of efflux across the basolateral membrane, and suggests that unconjugated glutathione is also involved in the clearance of organic anions from choroidal epithelial cells into the blood.
Among the various organic anion transport proteins identified to date, one member of the Slc21 family, Slc21a5, and two members of the Abcc family, Abcc1 and 4, have been shown to localize at the basolateral membrane of the rat choroidal epithelium (Gao et al, 1999; Leggas et al, 2004; Rao et al, 1999; Wijnholds et al, 2000). These three genes are expressed in both lateral and fourth ventricle CPs from newborn rats, as well as in cultured choroidal epithelial cells (this paper). Slc21a5 is a sodium-independent organic anion transporter, and experiments using Slc21a5-expressing Xenopus laevis oocytes have shown that glutathione conjugate cellular efflux can be mediated by this protein (Li et al, 2000). Abcc proteins are adenosine triphosphate (ATP)-dependent cellular export pumps, and there are direct and indirect evidence that Abcc1 (Jedlitschky et al, 1996; Muller et al, 1994) and, with a lower affinity, Abcc4 (Bai et al, 2004; Klokouzas et al, 2003), accept organic anions such as glutathione conjugates as substrates. In addition, other yet uncharacterized organic anion transporters may exist at the blood-CSF barrier. Thus, the choroidal basolateral efflux of glutathione conjugates can be mediated by several transport proteins. Delineating the relative contribution of these apparently redundant proteins will require a combination of strategies based on the specific modulation of expression as well as inhibition/competition approaches. A hint might be given, however, by the observation that cotransport and/or activation with unconjugated glutathione has been shown to increase the efflux of unconjugated drugs as well as glucurono-conjugates by Abcc proteins such as Abcc4 and 1, the Km value for glutathione activation being in the millimolar range (Leslie et al, 2001; Loe et al, 1998; Qian et al, 2001; Rius et al, 2003). Providing that glutathione-dependent activation of Abcc proteins applies to glutathione conjugate transport also, these proteins may therefore play a major role in conjugate efflux at the blood-CSF barrier, at least in conditions when intracellular glutathione content is not significantly lowered by oxidative stress.
Both fetal and adult human CPs, like the rodent tissues, display a high enzymatic capacity of conjugation to glutathione (Table 1). In rodents, immunohistochemical studies have shown the presence of mu, pi and alpha classes of cytosolic glutathione S-transferases and a microsomal glutathione S-transferase in the choroidal epithelium (Cammer et al, 1989; Johnson et al, 1993; Otieno et al, 1997; Philbert et al, 1995). Among the three cytosolic classes, pi forms are present early in development, both in rats (Beiswanger et al, 1995) and humans (Carder et al, 1990). The choroidal localization and developmental profile of the more recently identified classes of cytosolic, mitochondrial and microsomal glutathione-S-transferases are unknown. As all the isoenzymes so far detected in the CPs accept CDNB as a substrate, their relative contribution to glutathione conjugation activity measured in human tissue cannot be defined. Nonetheless, these high enzymatic activities, in conjunction with the immunohistochemical evidence for ABCC1 and ABCC4 in the human choroidal epithelium (Leggas et al, 2004; Rao et al, 1999), indicate that the blood-CSF barrier mechanism, characterized in the rodent cellular model of this interface, is likely to be as efficient in human.
The blood-CSF barrier develops very early during development, and the CP-CSF system plays a major role in brain ontogenesis as a source of growth factors (Chodobski et al, 2005; Ek et al, 2005; Strazielle and Ghersi-Egea, 2000). The volumic ratio of choroidal stroma versus epithelium changes during lifetime, and this precludes a direct comparison between fetal-, newborn- and adult-specific activities measured in CP homogenates. The apparently higher activities measured at earlier developmental stages may only reflect the larger epithelial content of the choroidal tissue. Yet, the important glutathione-S-transferase activity measured in the developing CPs in both humans and rats and the expression of transporters in neonate rodent choroidal tissue strongly suggest that the glutathione-dependent detoxification pathway shown in this paper is already functional at early stages to protect both the fetal choroidal epithelium and the developing brain.
While the biochemical blood–CSF barrier shown in this work will limit the concentration of toxins in the CSF, and hence their effect on tissues surrounding the ventricular, subarachnoidal and cisternal compartments, the fate of the toxins potentially reaching the brain by diffusion through the blood-brain barrier remains to be ascertained. To some extent, glutathione-S-transferases and organic anion transporters have been found associated with cerebral capillaries. However, the pattern of expression, the membrane distribution and the functional relevance of the various protein isoforms differ between the blood-brain barrier and the blood-CSF barrier (Strazielle and Ghersi-Egea, 2005a). The presence and efficiency of a glutathione conjugation-dependent metabolic barrier at the former interface remains therefore to be evaluated. The CSF-CP system, via the conjugation-efflux process shown in this paper, may also act as a pump that maintains a steep extracellular fluid-to-CSF downward gradient for toxins and toxin conjugates formed within the brain, thereby decreasing the overall exposure of neural cells to such deleterious compounds. Finally, the efficacy of pharmacological agents which are directly infused into the CSF might as well be influenced by the choroidal biochemical barrier mechanism, provided the metabolism pathway of these drugs includes one of the conjugation steps active within the choroidal epithelium.
In conclusion, this work describes a novel glutathione-dependent neuroprotective mechanism (summarized in Figure 6) that takes place at the blood-CSF interface. Blood-borne electrophilic, potentially reactive compounds can undergo an enzymatic conjugation to glutathione (2), while passively diffusing through the blood-CSF barrier (1), which impedes their penetration into the CSF. The conjugate is excreted out of the cell, mainly at the basolateral, that is, blood-facing membrane, by transporters such as Abcc or Slc21 proteins (3). This polarized efflux, seemingly dependent on unconjugated glutathione, reinforces the barrier mechanism. Cerebrospinal fluid-borne organic anion conjugates may also use this pathway to leave the CSF, provided they are substrates for one of the inwardly directed transporters present at the apical membrane (Strazielle et al, 2004) (4). The capacity of this biochemical barrier can be increased or maintained by pharmacologically increasing glutathione synthesis within the choroidal epithelium (5).
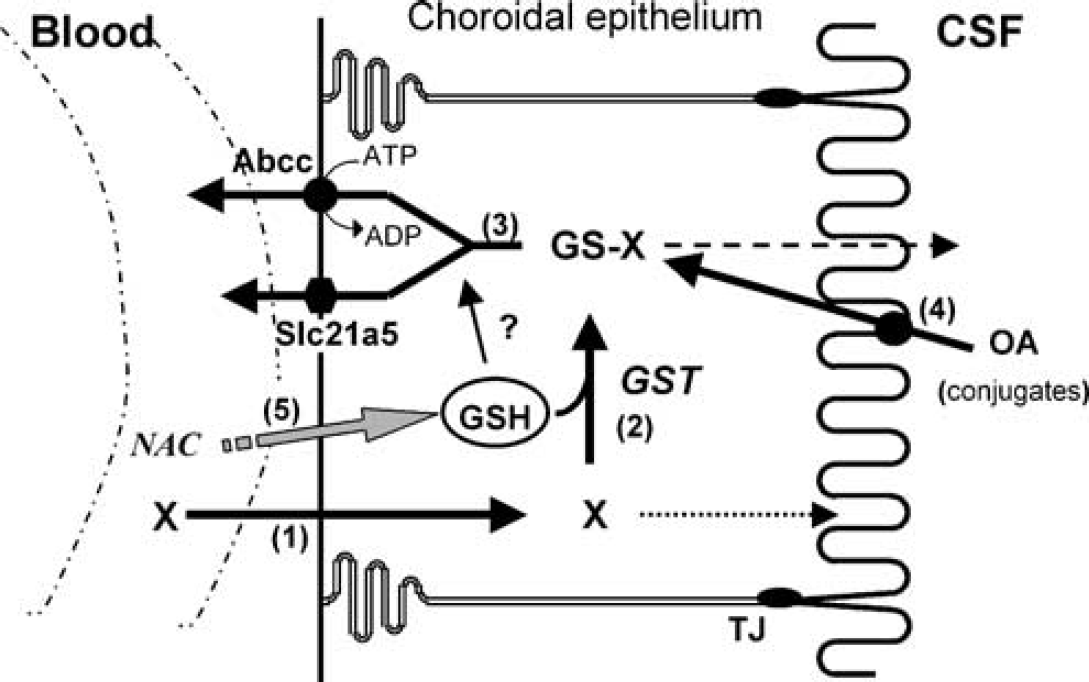
Schematic model of the glutathione-dependent metabolic barrier function of the blood-CSF interface. Numbers are referred to in the Discussion. TJ: apical tight junction, OA: organic anion, GSH: glutathione, GST: glutathione-S-transferases.
Footnotes
Acknowledgements
The authors thank Drs Raymonde Bouvier, Koué Folligan, Carmine Mottolese, Marc Sindou, and Jacqueline Trouillas for their involvement in human sample collection.
