Abstract
We examined the effects of FK506, a specific inhibitor of calcineurin, on the binding capacity of cyclic AMP-dependent protein kinase (cAMP-DPK) in gerbils subjected to 2-h cerebral hemispheric ischemia. FK506 (0.1 mg/kg) was infused intravenously at 15 min prior to the induction of ischemia by common carotid artery occlusion. The binding capacity of cAMP-DPK was evaluated by autoradiographic analysis of the cAMP binding, and cerebral blood flow (CBF) was measured by the [14C] iodoantipyrine method. In the sham-operated gerbils, FK506 significantly increased mean arterial blood pressure and tended to decrease CBF, suggesting that FK506 may constrict systemic blood vessels as well as cerebral blood vessels. On the other hand, cAMP binding was not altered by FK506 in the sham-operated gerbils. In the ischemia group of gerbils, FK506 prevented any significant reduction of cAMP binding in the hippocampus CA1 and cerebral cortices on the ischemic side, whereas it exerted no significant influence on the cAMP binding of the nonischemic side. The values of CBF were comparable between the vehicle-treated gerbils and FK506-treated gerbils in the ischemic regions. Preservation of cAMP binding indicates that intracellular signal transduction via cAMP-DPK can be maintained by FK506 despite ischemia, suggesting that this agent may be beneficial for reducing ischemic tissue damage.
Calcineurin is a Ca2+/calmodulin-dependent protein phosphatase and is highly abundant in the brain (Klee et al., 1983, 1988; Steiner et al., 1992; Dawson et al., 1994). Studies have demonstrated multiple roles for calcineurin in diverse signal transduction cascades of excitable cells (Klee et al., 1988). This enzyme is the only known protein phosphatase that is under the direct control of intracellular Ca2+ (Guerini et al., 1990) and can be activated by the elevation of the intracellular Ca2+ levels that occurs under ischemic conditions.
On the other hand, cyclic AMP (cAMP) plays an important role as one of the cardinal intracellular signaling systems in the central nervous system (Blitzer et al., 1995). cAMP binds to the regulatory subunit of cAMP-dependent protein kinase (cAMP-DPK), and the cAMP-DPK then promotes phosphorylation of various substrates. On the other hand, calcineurin dephosphorylates the substrates, including cAMP-DPK itself, so a close functional relationship between calcineurin and cAMP-DPK has been observed (Klee et at., 1983; Guerini et al., 1990; Baukal et al., Antoni et al., 1995; Blitzer et al., 1995; 1994; Coghlan et al., 1995).
We found previously that cAMP binding was significantly reduced in various regions after 2 h of cerebral ischemia (Tanaka et al., 1995), suggesting that the intracellular signal transduction via cAMP-DPK may be seriously compromised in ischemia. Our recent experiments demonstrated that this reduction closely reflected early ischemic neuronal damage and was especially prominent in vulnerable regions such as the hippocampus CA1 (Tanaka et al., 1996).
FK506 has been shown to be neuroprotective against glutamate toxicity (Dawson et al., 1993), focal cerebral ischemia (Sharkey and Butcher, 1994), and transient global ischemia (Ide et al., 1996; Tokime et al., 1996). Such neuroprotective effects of FK506 are considered to be due mainly to inhibition of calcineurin, since FK506 is a selective inhibitor of calcineurin, when FK506 binds to endogenous FK506-binding proteins (FKBPs) (Liu et al., 1991; Mukai et al., 1993; Kissinger et al., 1995). FKBPs are highly concentrated in discrete neuronal sites, including the hippocampus CA1 (Steiner et al., 1992; Dawson et al., 1994). Colocalization of FKBPs and calcineurin has also been demonstrated in the brain (Steiner et al., 1992; Dawson et al., 1994).
Based on these considerations, the present study was undertaken to examine whether neuroprotective effects of FK506 can be observed from the standpoint of cAMP-DPK during cerebral ischemia. We therefore assessed the effects of FK506 on the cAMP binding in sham-operated gerbils and in gerbils subjected to 2-h ischemia induced by occlusion of the common carotid artery (CCAO). The autoradiographic method employed in the present study enabled us to measure local cerebral blood flow and cAMP binding in the same brain (Tanaka et al., 1991).
MATERIALS AND METHODS
Animal preparation
The protocol described here had been approved as meeting the Animal Experimentation Guidelines of Keio University School of Medicine. A total of 36 Mongolian gerbils of either sex, weighing 60–100 g, were divided into four groups: a vehicle + sham group (n = 6), an FK506 + sham group (n = 5), a vehicle + CCAO group (n = 16), and an FK506 + CCAO group (n = 9). In all of these groups, polyethylene catheters (Clay–Adams PE 10; Becton–Dickinson, Parsippany, NJ, U.S.A.) were inserted into the femoral artery and vein under light anesthesia with pentobarbital sodium (20 mg/kg, i.p.). In addition, the right CCA was exposed, and a 5-0 silk suture was loosely looped around the artery for the future ligation. The arterial blood pressure and blood gases were monitored through the arterial catheters. After the gerbils had recovered from the anesthesia to a fully alert state at 2 h following catheterization, FK506 (0.1 mg/kg, freshly dissolved in saline) was injected intravenously in the FK506 + sham and FK506 + CCAO groups, whereas vehicle (saline, 0.5 ml) was injected in the vehicle + sham and vehicle + CCAO groups. At 15 min after the above injections, the right CCA was doubly tied with the silk suture to induce 2-h cerebral ischemia in the vehicle + CCAO and FK506 + CCAO groups under light anesthesia with 1.5% halothane. The halothane inhalation was terminated following the ligation of the artery. At 15 min after the ligation, the gerbils were evaluated for various neurologic signs to calculate the stroke index (McGraw, 1977; Nozaki et al., 1996). The gerbils that scored more than 5 for the stroke index were subjected to further study as the vehicle + CCAO group (n = 8) and the FK506 + CCAO group (n = 5). On the other hand, the CCA was merely exposed and left unoccluded in the vehicle + sham and FK506 + sham groups. The rectal temperature was monitored continuously by a digital thermometer (Shibaura Electronics) and maintained at 37 ± 0.5°C throughout the experiments by using a heating pad and lamp.
Measurement of cerebral blood flow
At 2 h after the ligation or sham surgery of the CCA, the cerebral blood flow (CBF) was measured by the [14C]-iodoantipyrine (IAP) method as described previously (Tanaka et al., 1991). At the end of CBF measurement, the gerbils were decapitated, and within 30 s their brains were quickly removed and frozen in liquid Freon 22 (Asahi Glass). Consecutive 20-μm-thick coronal sections were prepared on a cryostat (Cryocut 1800; Leica, Nussloch, Germany). Double sets of sections were used for autoradiographic measurements of the CBF and for incubation studies with [3H]cAMP, respectively. The sections assigned for CBF measurement were exposed to x-ray films (SB-5; Eastman Kodak) together with calibrated 14C-embedded methylmethacrylate standards for 10 days.
Measurement of cAMP binding
The labeling procedure with [3H]cAMP was basically similar to that described by Gundlach and Urosevic (1989). The cut brain sections assigned for measurement of the cAMP binding were preincubated for 20 min at 22°C in Krebs–Hepes buffer to remove endogenous cAMP and to wash out [14C]IAP completely from the brain tissue, as confirmed previously (Tanaka et al., 1991). Subsequently, the sections were incubated for 90 min at 22°C in buffer containing 5 nM [3H]cAMP ([5′, 8-3H]cAMP ammonium salt; specific activity, 42 Ci/mmol; Amersham) and 1 mM 3-isobutyl-1-methylxanthine (IBMX; Sigma). The IBMX was included to prevent hydrolysis of [3H]cAMP by phosphodiesterase in the brain tissue (Gundlach and Urosevic, 1989). Nonspecific binding was defined as that in the presence of 10 μM unlabeled cAMP (Sigma). Autoradiograms were generated by apposing the labeled sections to tritium-sensitive Ultrofilm (Leica) with calibrated 3H-embedded polymer standards (Amersham) for 3 weeks.
Data analysis
Quantitative densitometric analysis of the autoradiograms was performed with the computerized digital image-processing system (Tanaka et al., 1991). The data obtained were tested by analysis of variance. When this procedure indicated a significant difference, the t test with the Bonferroni correction was performed for simultaneous multiple comparisons (Wallenstein et al., 1980). Values are presented as the means ± SD.
RESULTS
As shown in Table 1, the physiological parameters determined immediately before CBF measurement were within the normal ranges. However, the mean arterial blood pressure (MABP) was slightly higher in the FK506 + sham group as compared to that in the vehicle + sham group with statistical significance. Similarly, the MABP in the FK506 + CCAO group tended to be slightly higher than that in the vehicle + CCAO group. None of the other parameters differed significantly among the groups.
Physiological parameters
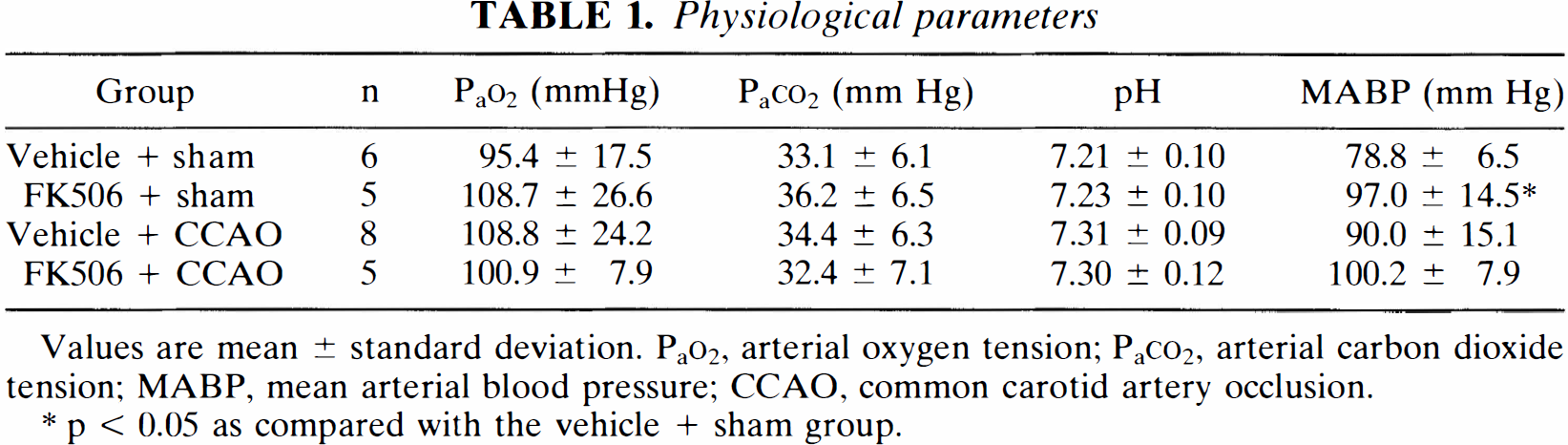
Values are mean ± standard deviation. PaO2, arterial oxygen tension; PaCO2, arterial carbon dioxide tension; MABP, mean arterial blood pressure; CCAO, common carotid artery occlusion.
p < 0.05 as compared with the vehicle + sham group.
Figure 1 shows representative color-coded autoradiograms of the CBF at the hippocampus level. The FK506 + sham group (the top right image) showed slightly lower CBF in each region as compared to the vehicle + sham group (the top left image). On the other hand, both the vehicle + CCAO group (the bottom left image) and the FK506 + CCAO group (the bottom right image) demonstrated a severe reduction in CBF on the occluded side.
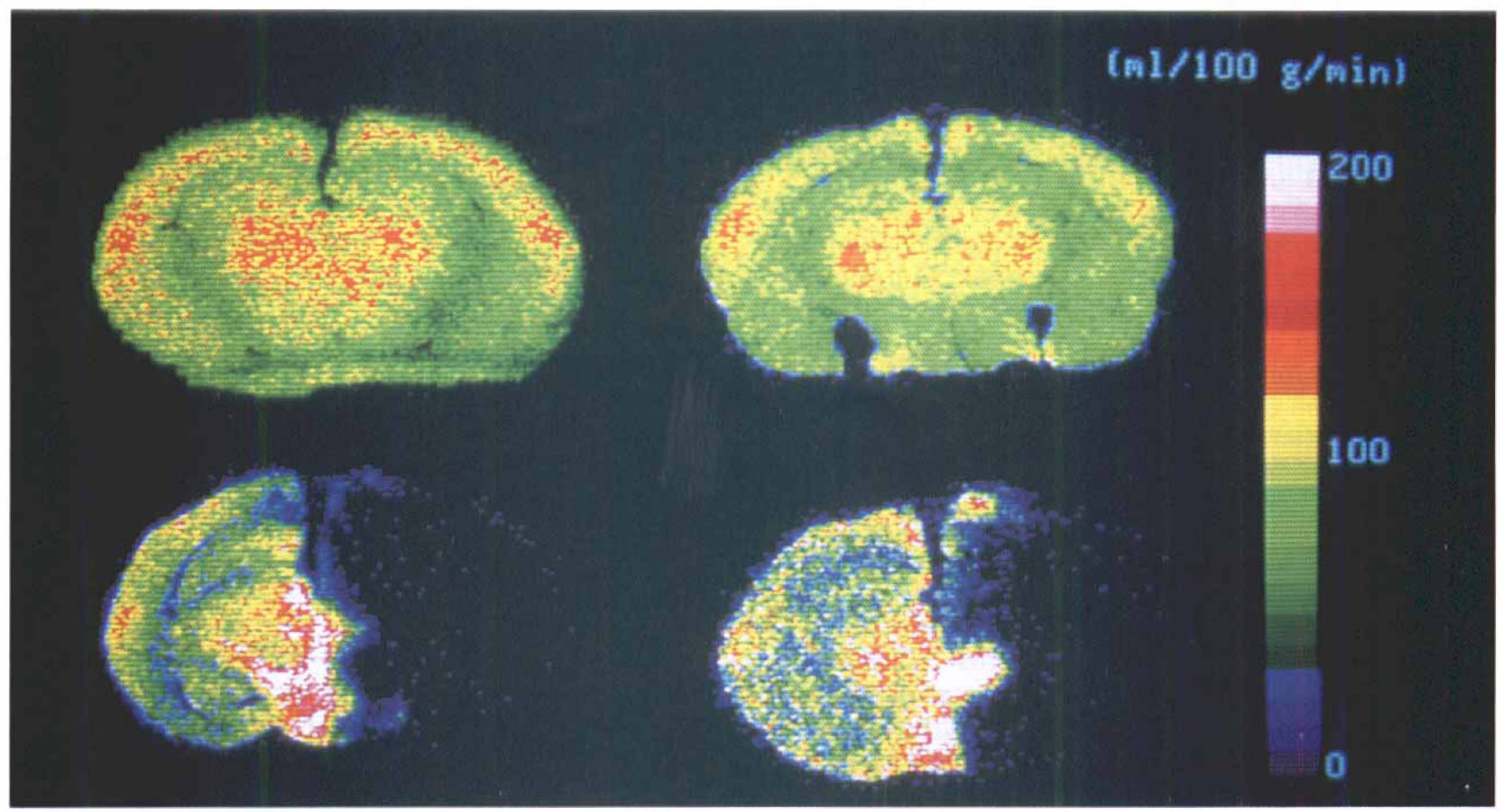
Representative color-coded autoradiograms of the cerebral blood flow at the hippocampus level. The color scales represent levels of cerebral blood flow (ml/100 g/min). The
Figure 2 shows representative color-coded autoradiograms of the cAMP binding at the hippocampus level, which were obtained from the same gerbils as in Fig. 1. Both the FK506 + sham group (the top right image) and the vehicle + sham group (the top left image) revealed a comparable distribution pattern of cAMP binding in each region. On the other hand, the vehicle + CCAO group (the bottom left image) showed a significant reduction in cAMP binding in the hippocampus and temporal cerebral cortex on the ischemic side, whereas the FK506 + CCAO group (the bottom right image) did not reveal such definite reduction.
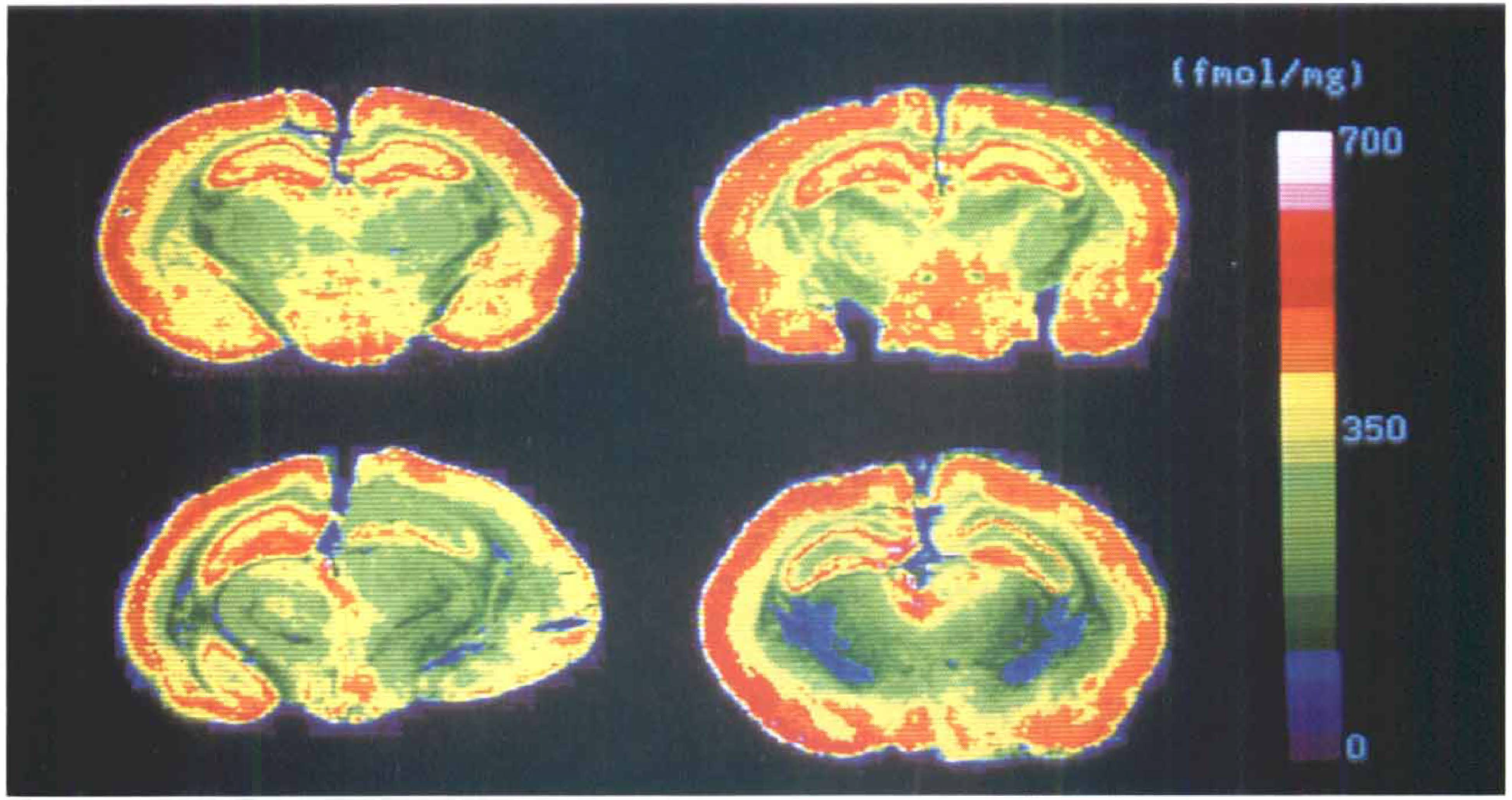
Representative color-coded autoradiograms of the cAMP binding at the hippocampus level. The color scales represent levels of cAMP binding (fmol/mg). The
Figure 3 summarizes the data for the CBF and cAMP binding in the hippocampus CA1. As shown in the top panel, the CBF in the FK506 + sham group was slightly lower than that in the vehicle + sham group on each side. On the other hand, both the vehicle + CCAO and FK506 + CCAO groups exhibited a significant reduction in CBF on the right side, while these groups also demonstrated a moderate decrease in CBF on the left side. The CBF in the FK506 + CCAO group was slightly higher than that in the vehicle + CCAO group on both sides.
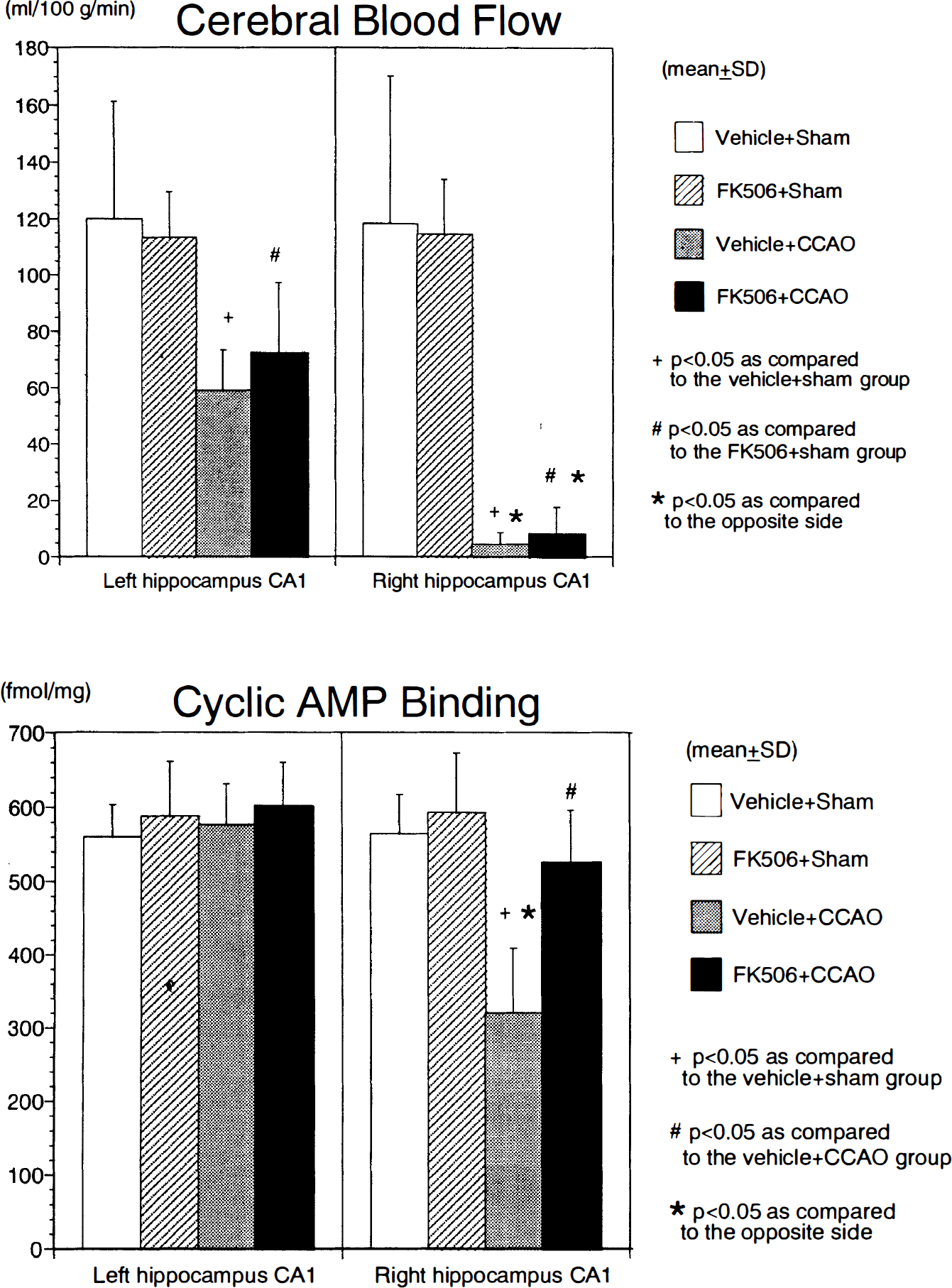
Summary of cerebral blood flow (
As shown in the bottom panel of Fig. 3, no significant differences in cAMP binding were noted between the vehicle + sham and FK506 + sham groups on each side of the hippocampus CA1. As reported previously (Tanaka et al., 1995), the cAMP binding on the right side of the vehicle + CCAO group exhibited a significant reduction as compared to the opposite side. In contrast, the cAMP binding on the right side of the FK506 + CCAO group demonstrated only a mild decrease with no statistical significance as compared to the opposite side, and its value was significantly higher than that on the right side of the vehicle + CCAO group.
Figure 4 summarizes the data for the CBF and cAMP binding in the temporal cerebral cortex. As shown in the top panel, the CBF in the FK506 + sham group was slightly lower that that in the vehicle + sham group on both sides. In the groups subjected to CCAO, the CBF was significantly reduced on the right side in both the vehicle + CCAO and FK506 + CCAO groups. It was also moderately decreased on the left side in these groups.
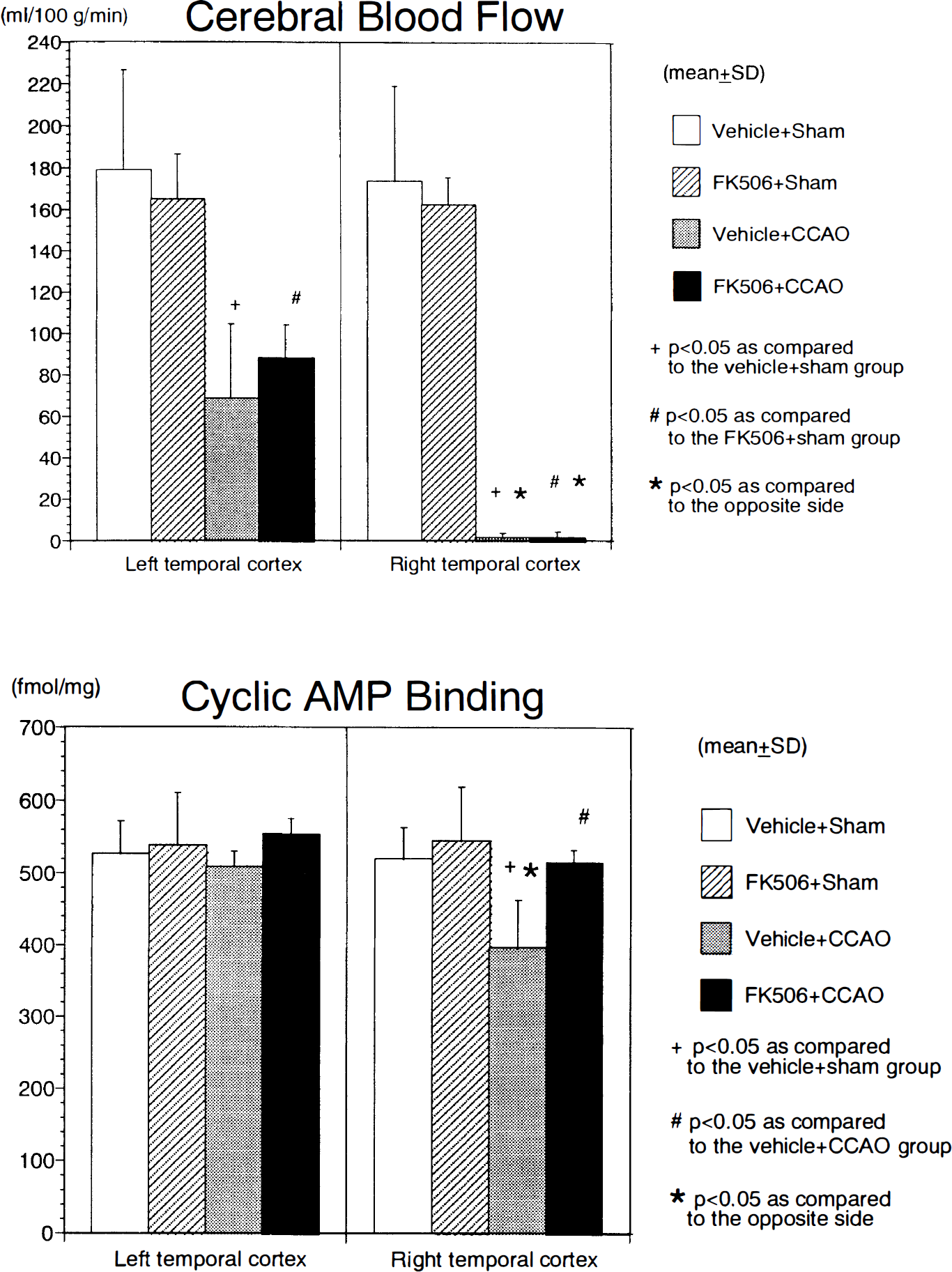
Summary of cerebral blood flow (
As shown in the bottom panel of Fig. 4, the cAMP binding in the FK506 + sham group displayed a similar value to that in the vehicle + sham group on each side of the temporal cerebral cortex. On the other hand, the vehicle + CCAO group on the right side revealed a moderate but statistically significant decrease as compared to the opposite side and the right side of the vehicle + sham group. In contrast, the right side of the FK506 + CCAO group demonstrated only a slight reduction in cAMP binding as compared to the opposite side, and its value was significantly higher than that on the right side of the vehicle + CCAO group. Similar results were obtained in other cerebral cortical regions such as the frontoparietal and occipital cortices.
Figure 5 summarizes the data for the CBF and cAMP binding in the cerebellar cortex. As observed in the temporal cerebral cortex, the CBF in the FK506 + sham group was slightly lower than that in the vehicle + sham group on both sides. The CBF in the vehicle + CCAO group exhibited a moderate reduction as compared to the vehicle + sham group, whereas the CBF in the FK506 + CCAO group did not undergo a definite reduction as compared to the FK506 + sham group (top panel).
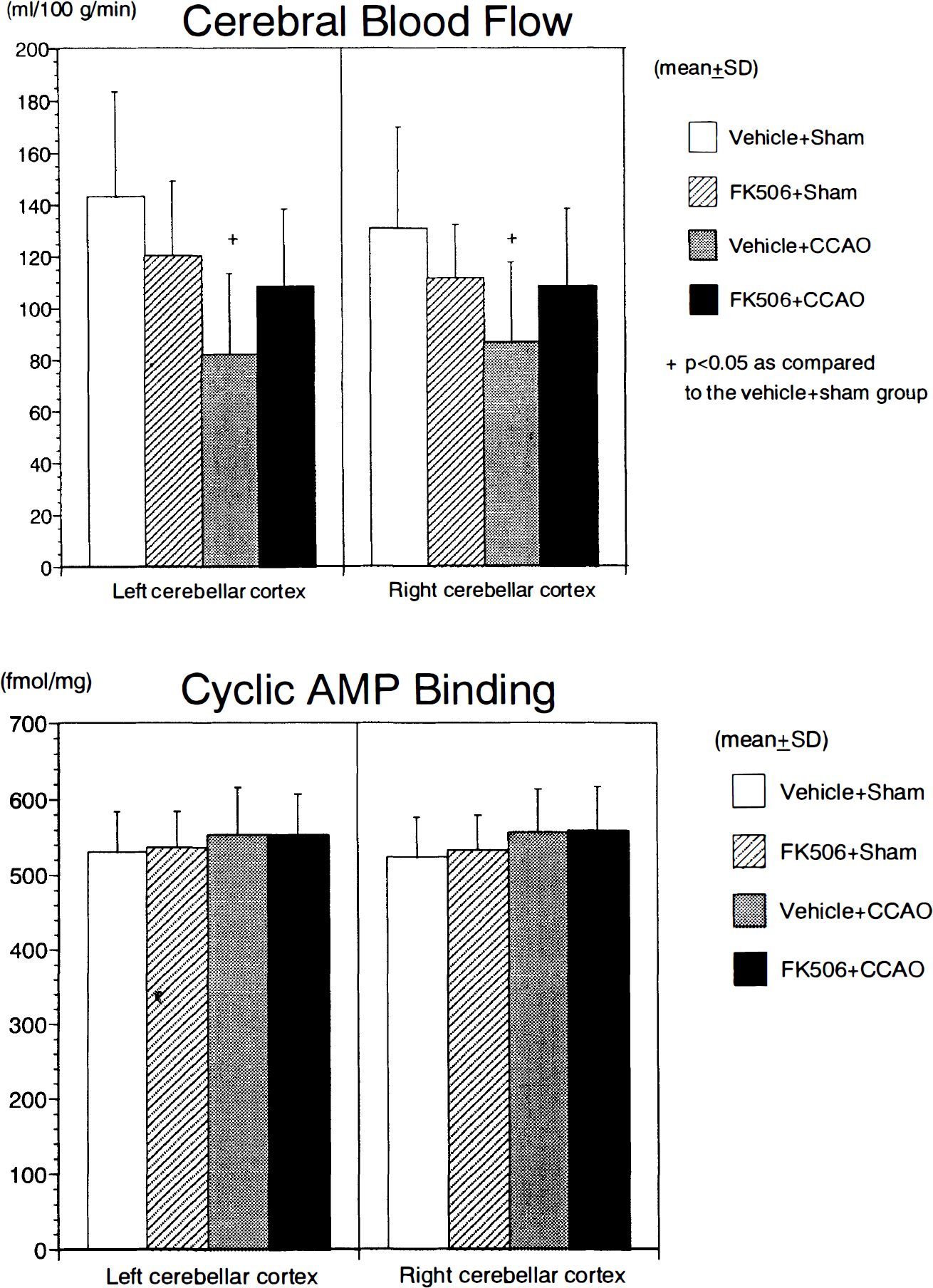
Summary of cerebral blood flow (
As shown in the bottom panel of Fig. 5, the cAMP binding in the cerebellar cortex of each group displayed a similar value on both sides, and no significant differences were noted among the groups.
DISCUSSION
The present experiments revealed that (a) FK506 slightly decreased the CBF while it increased the systemic arterial blood pressure in the sham-operated gerbils, (b) the CBF in the FK506 + CCAO group was slightly higher in the nonischemic regions such as the left hippocampus CA1 and bilateral cerebellar cortices as compared to the vehicle + CCAO group, (c) FK506 did not exert any definite influence on the cAMP binding in the sham-operated gerbils, and (d) FK506 significantly prevented a reduction in cAMP binding in the hippocampus CA1 and temporal cerebral cortex in the gerbils subjected to 2-h hemispheric ischemia.
FK506 has been reported to increase arterial blood pressure and sympathetic activity, and such actions are apparently related to inhibition of calcineurin (Lyson et al., 1993). FK506 also decreases endothelial release of prostacyclin, while it increases the release of endothelin (Yatscoff et al., 1993). These pharmacological properties may also contribute to the increase in arterial blood pressure induced by FK506. In fact, clinical studies have suggested that hypertension is an important side effect of FK506 (Armitage et al., 1991). In line with these reports, we found that intravenous injection of FK506 slightly increased the MABP as compared to that of the vehicle-treated groups as shown in Table 1. These observations imply that FK506 exerts vasoconstricting actions on the systemic vascular system. In the present study, the CBF in the FK506 + sham group was slightly decreased as compared to the vehicle + sham group. We infer that FK506 may constrict not only systemic blood vessels but also cerebral blood vessels to some degree.
The CBF on the nonischemic side in the FK506 + CCAO group showed a slight increase as compared to the vehicle + CCAO group in each examined region. A slight increase in MABP in the FK506 + CCAO group as compared to the vehicle + CCAO group may be responsible for the above increase in CBF, since autoregulation of CBF is known to be extensively impaired in the acute phase of severe ischemia.
cAMP-DPK is a tetramer containing two regulatory subunits and two catalytic subunits. Phosphorylation of the regulatory subunit causes cAMP-DPK to dissociate readily into the regulatory subunits and active catalytic subunits. On the other hand, dephosphorylation of the regulatory subunit leads cAMP-DPK to assume the inactive state with association of each subunit. Calcineurin dephosphorylates the regulatory subunit of cAMP-DPK (Klee et al., 1983), where cAMP-binding sites are located (Taylor et al., 1990). According to earlier basic studies on purified cAMP-DPK (Rangel-Aldao and Rosen, 1976), however, the amounts of cAMP that can bind to the regulatory unit of cAMP-DPK were found to be identical for both the phosphorylated and dephosphorylated forms of cAMP-DPK, with approximately the same affinity. These pharmacological properties appear to be compatible with the present data, which revealed no definite difference in cAMP binding between the vehicle + sham and FK506 + sham groups.
We reported previously that cAMP binding is progressively reduced in the hippocampus CA1 and cerebral cortices at 30 min to 6 h of cerebral ischemia (Tanaka et al., 1996). In contrast, at 15 min of ischemia, we noted no significant reduction in cAMP binding in the brain (Tanaka et al., 1995). Such alteration of cAMP binding is consistent with a progressive decrease in cAMP-DPK activity during ischemia (Schwartz et al., 1976). On the other hand, according to Kobayashi et al. (1977), cAMP concentration in the brain increased to 13-fold greater than control at 1 min of ischemia and then declined to the level of fivefold greater than control during the late ischemic period in gerbils. Earlier investigation (Steiner et al., 1972) also demonstrated a transient, but significant, increase in the concentration of cAMP in the brain following decapitation. Therefore, the effects of endogenous cAMP on [3H]cAMP binding should be considered. However, as already discussed (Gundlach and Urosevic, 1989; Tanaka et al., 1996), the present binding protocol for [3H]cAMP can be expected to reduce the endogenous cAMP concentrations to negligible levels within the tissue. Consequently, we consider that the decrease in cAMP binding observed in our experiment is not parallel with cAMP production in the ischemic brain and not due to excessive production of cAMP. We speculate that the reduction in cAMP binding observed in the present study may have been attributed to ischemic structural damage of cAMP-DPK protein.
In the present study, administration of FK506 significantly prevented the reduction of cAMP binding in the ischemic regions. Several mechanisms should be considered to explain this effect of FK506. One of them may be a maintained phosphorylation of cAMP-DPK due to inhibition of calcineurin by FK506. The intracellular Ca2+ level increases significantly in cerebral ischemia, so calcineurin is expected to be markedly activated. As described above, dephosphorylation of cAMP-DPK may lead each subunit of cAMP-DPK to assume the associated state forming a tetramer, and cAMP-binding sites may thus be less accessible under ischemic conditions (Taylor et al., 1990). As mentioned previously, however, earlier basic studies have suggested that the binding capacity of cAMP-DPK may be identical for the phosphorylated and dephosphorylated forms (Rangel-Aldao and Rosen, 1976), so a direct influence of FK506 on the phosphorylation state of cAMP-DPK might not be involved in the present results.
The effect of FK506 on CBF needs some consideration. In the hippocampus CA1 on the ischemic side, the CBF was 4.3 ± 4.2 ml 100 g–1 min–1 in the vehicle + CCAO group, whereas it was 8.2 ± 9.3 ml 100 g–1 min–1 in the FK506 + CCAO group. According to our recent research (Tanaka et al., 1996), the cAMP binding was similarly reduced at 2 h of ischemia in the hippocampus CA1 irrespective of the value of CBF if the CBF was <20 ml–1100 g min–1. The slightly higher level of CBF in the FK506 + CCAO group may therefore not contribute to the prevention of a reduction in cAMP binding in this group. Further, the CBF in the temporal cerebral cortex in both the vehicle + CCAO and the FK506 + CCAO groups was at similar levels, so the effects of FK506 on the cAMP binding in this region cannot be explained from the standpoint of CBF.
Finally, we should evaluate the possible neuroprotective effects of FK506 against ischemic damage. Recent reports have indicated that FK506 prevents histological damage in focal cerebral ischemia (Sharkey and Butcher, 1994) as well as in transient global ischemia (Ide et al., 1996; Tokime et al., 1996). FK506 also attenuates glutamate neurotoxicity (Dawson et al., 1993). Such protective effects of FK506 may have reduced the ischemic neuronal damage in the present study so that a reduction in cAMP binding in the ischemic regions was, therefore, secondarily prevented. Deactivation of nitric oxide synthase by inhibition of calcineurin has been speculated to play an important role in the cytoprotective effects of FK506 (Dawson et al., 1993). Since the preservation of cAMP binding in the present study indicates that signal transduction via cAMP-DPK may be maintained despite ischemia, FK506 could be useful for supporting the tissue viability.
In conclusion, FK506, a specific inhibitor of calcineurin, clearly prevented a reduction in cAMP binding in ischemic brain regions such as the hippocampus CA1 and temporal cerebral cortex. The data obtained suggest that intracellular signal transduction via cAMP-DPK may be maintained by FK506 during ischemia, implying that this agent could be beneficial for reducing ischemic damage from the standpoint of cAMP-DPK. The precise mechanisms underlying this effect of FK506 need to be elucidated in the future.
Footnotes
Acknowledgment:
FK506 was a kind gift from the Fujisawa Pharmaceutical Company, Osaka, Japan.
