Abstract
The flow threshold for alterations of the in vitro [3H]cyclic AMP (cAMP) binding, an indicator of the total amount of particulate cAMP-dependent protein kinase, was evaluated in the gerbil brain after 30 min, 2 h, and 6 h of unilateral common carotid artery occlusion, respectively. The autoradiographic method developed in our laboratory enabled us to measure the [3H]cAMP binding and local CBF in each region of the same brain. The ischemic flow thresholds for reduction of the cAMP binding in the hippocampus CA1 were 18, 34, and 49 ml 100 g–1 min–1 after 30-min, 2-h, and 6-h ischemia, respectively. These values were higher than those in other regions such as the hippocampus CA3 and temporal cerebral cortex in each duration of ischemia. These findings indicate that (a) the ischemic flow threshold for perturbation of the cAMP system may be higher in the hippocampus CA1 than in other brain regions, suggesting that the hippocampus CA1 could be especially vulnerable to acute ischemic stress; and (b) the level of the aforementioned threshold may increase progressively during the time course of ischemia in particular regions such as the hippocampus CA1 and CA3, suggesting that the duration of ischemia exerts a definite influence on the viability of the ischemic neuronal cells in these regions.
In cerebral ischemia, the cell viability depends on the degree and duration of ischemia (Heiss, 1983). The ischemic thresholds for electrical activities, histological damage, and various neurochemical events have been extensively analyzed (Astrup et al., 1977; Naritomi et al., 1988; Shimada et al., 1989; Matsumoto et al., 1993; see review by Hossmann, 1994). Despite these extensive investigations, however, the ischemic threshold for perturbation of the cyclic AMP system still remains unknown.
Cyclic AMP (cAMP) binding protein is considered to be identical to the regulatory subunit of cAMP-dependent protein kinase (cAMP-DMK) (Walter et al., 1978; Lohmann and Walter, 1984; Gundlach and Urosevic, 1989). As has already been demonstrated by several investigators (Gundlach and Urosevic, 1989; Hara et al., 1992), the in vitro autoradiographic labeling with [3H]cAMP of the brain tissue can be assumed to reflect the regional distribution of membrane-bound, particulate cAMP-DPK regulatory subunits. Binding of cAMP to the particulate form of cAMP-DMK is an essential step for the control of synaptic transmission as well as the regulation of receptors and ion channels (Lohmann and Walter, 1984). On this basis, evaluation of alterations in the cAMP binding with concomitant CBF measurements can offer one means of elucidating the ischemic flow threshold for perturbation of the cAMP system. The present study was therefore undertaken to evaluate the [3H]cAMP binding and CBF in each brain region employing our autoradiographic technique (Tanaka et al., 1991) in experimental cerebral ischemia.
MATERIALS AND METHODS
The protocol described herein had been approved as meeting the Animal Experimentation Guidelines of Keio University School of Medicine. Fifty-two Mongolian gerbils of either sex weighing 60–100 g were employed and divided into six groups: a 30-min occlusion group (n = 10), a 30-min sham group (n = 5), a 2-h occlusion group (n = 15), a 2-h sham group (n = 5), a 6-h occlusion group (n = 9), and a 6-h sham group (n = 8). In all of these groups, polyethylene catheters (Clay-Adams PE 10, Becton-Dickinson) were inserted into the femoral artery and vein under light anesthesia with pentobarbital sodium (20 mg/kg, i.p.). Through the arterial catheters, the arterial blood pressure and blood gases were monitored. After the animals had recovered from the anesthesia to a fully alert state at 2 h after catheterization, the right common carotid artery was doubly tied with silk thread under anesthesia with 1.5% halothane to induce ischemia in each occlusion group. In the sham groups, the carotid artery was merely exposed. The skin was then sutured, and the halothane was discontinued. The body temperature was maintained at 37 ± 0.5°C throughout the experiments.
At 30 min, 2 h, or 6 h after the ligation or sham surgery of the carotid artery, the CBF was measured by the [14C]iodoantipyrine (IAP) method (Sakurada et al., 1978) in the 30-min occlusion and sham groups, the 2-h occlusion and sham groups, or the 6-h occlusion and sham groups, respectively, as described previously (Tanaka et al., 1991). Consecutive 20-μm-thick coronal sections were prepared on a cryostat (Cryocut 1800, Leica) at the level of the hippocampus. Double sets of sections were used for autoradiographic measurements of the CBF and for incubation studies with [3H]cAMP, respectively. The sections assigned for CBF measurement were exposed to x-ray films (SB-5, Eastman Kodak) together with calibrated 14C-embedded methylmethacrylate standards for 10 days.
The labeling procedure with [3H]cAMP was basically similar to that described by Gundlach and Urosevic (1989). The cut brain sections assigned for measurement of the cAMP binding were preincubated for 20 min at 22°C in Krebs-HEPES buffer to remove endogenous cAMP and to wash out [14c]IAP completely from the brain tissue, as confirmed previously (Tanaka et al., 1991). Subsequently, the sections were incubated for 90 min at 22°C in buffer containing 5 n
Each data point given in this report represents the specific binding by subtracting the value for the nonspecific binding from that for the total binding with the digital image analyzer (Tanaka et al., 1988). Because the grain density measured in the brain sections incubated for nonspecific binding did not show any significant difference in any region between the ischemic and nonischemic hemispheres, we assume that the calculation of radioactivity without correcting for quenching level provides no fatal errors in the present study.
The relationship between the cAMP binding and CBF was examined in detail in the hippocampus CA1, CA3, and the temporal cerebral cortex. Three individual measurements of the optical density (corresponding to a range of blood flows) at different points within each brain region were made from each brain. The values of the physiological parameters are presented as the means ± SD.
RESULTS
As shown in Table 1, the physiological parameters measured immediately before the CBF measurement were within the normal ranges, and none of them differed significantly among the groups.
Physiological parameters measured immediately before CBF measurement
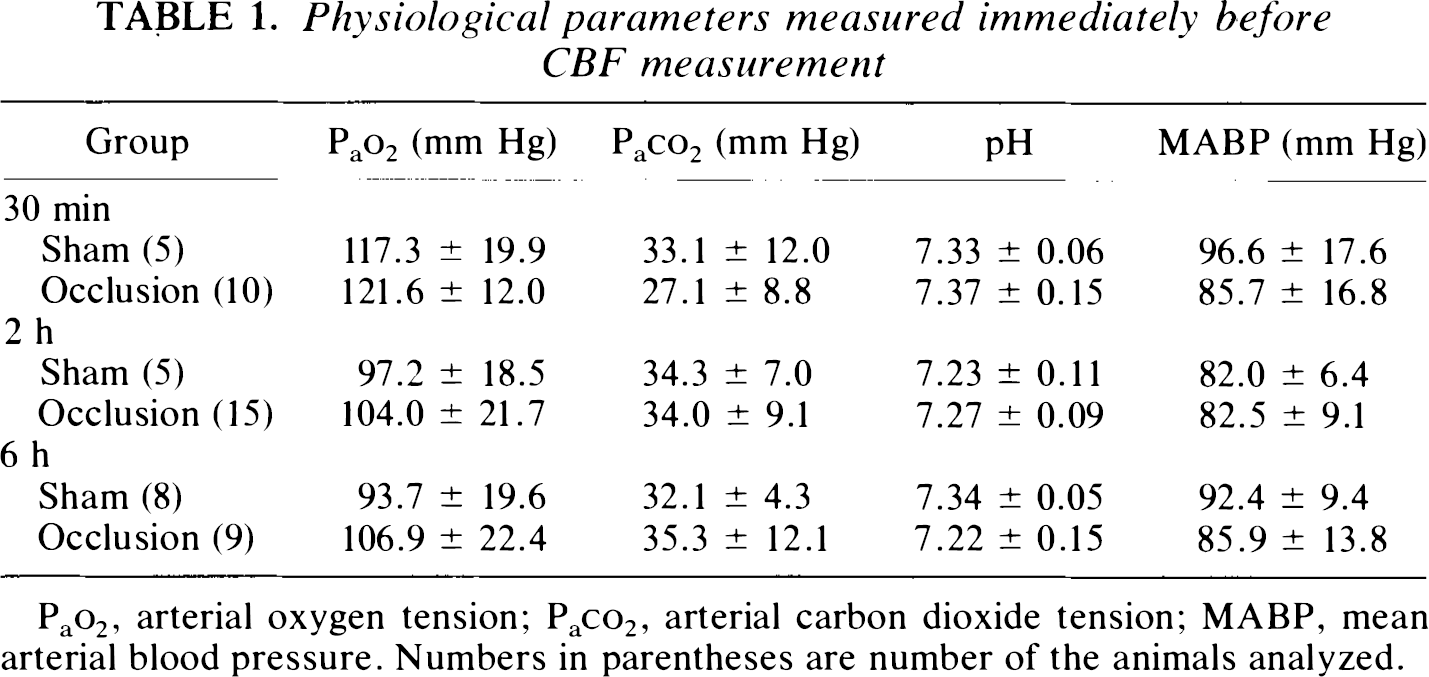
Pao2, arterial oxygen tension; Paco2, arterial carbon dioxide tension; MABP, mean arterial blood pressure. Numbers in parentheses are number of the animals analyzed.
Representative pairs of color-coded autoradiograms of the cAMP binding and CBF are illustrated in Fig. 1 for 30-min (top panel), 2-h (middle panel), and 6-h ischemia studies (bottom panel), respectively.
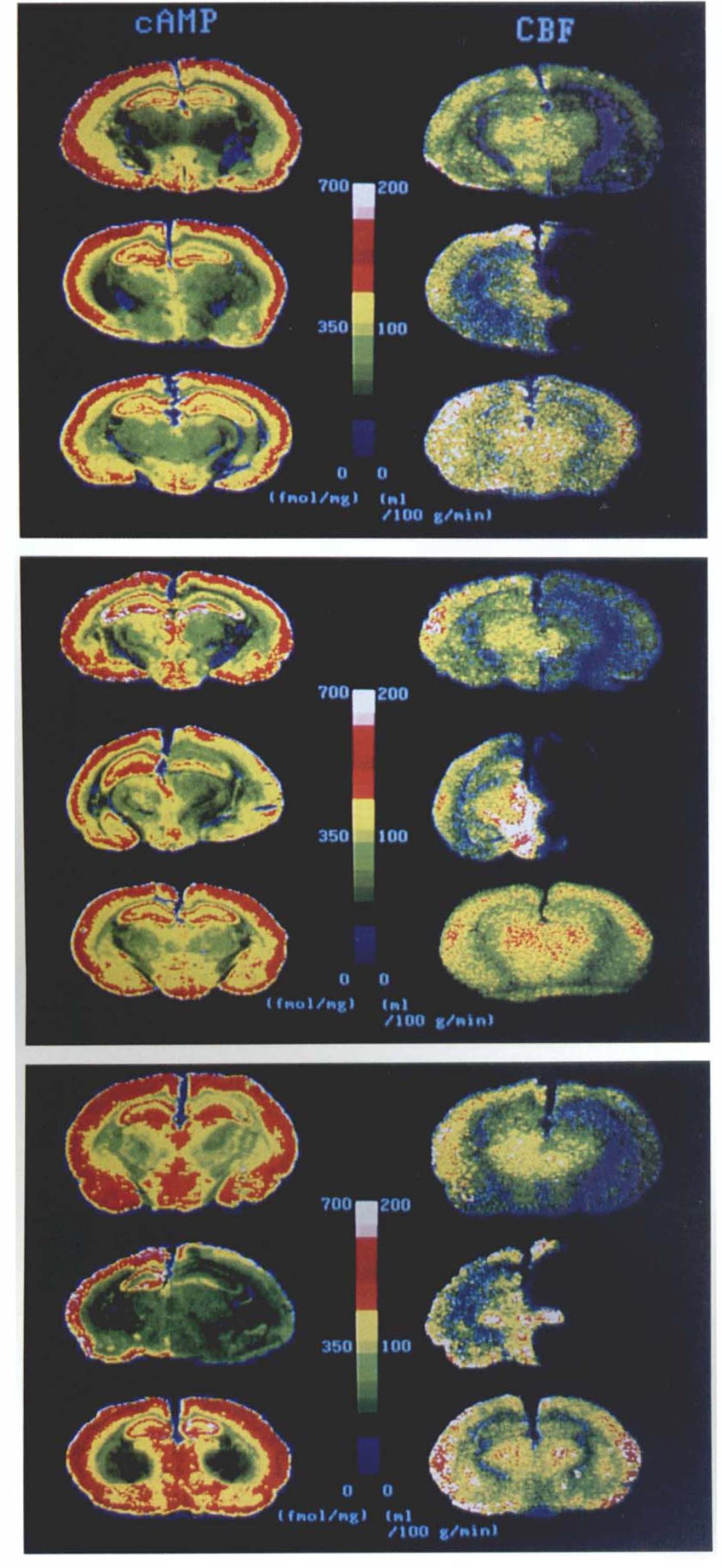
Representative pairs of color-coded autoradiograms of the [3H]cyclic AMP (cAMP) binding (left column) and CBF (right column) obtained from the 30-min ischemia study
In the 30-min ischemia study, the cAMP binding in the animal with mild ischemia revealed no definite alteration. On the other hand, the animal with severe ischemia revealed a definite reduction in cAMP binding in the hippocampus CA1 on the ischemic side.
In the 2-h ischemia study, the animal with moderate ischemia revealed an apparent decrease in cAMP binding in the hippocampus CA1 on the ischemic side. On the other hand, the cAMP binding in the animal with severe ischemia demonstrated a profound reduction in the hippocampus CA1, while a moderate reduction in cAMP binding was also noted in the hippocampus CA3, the temporal cerebral cortex, and thalamus on the ischemic side.
In the 6-h ischemia study, the animal with mild ischemia revealed a definite decrease in cAMP binding in the hippocampus CA1 on the ischemic side. On the other hand, the cAMP binding in the animal with severe ischemia was significantly reduced in each region of the cerebral hemisphere on the ligated side.
Figure 2 illustrates the relationship between the CBF and [3H]cAMP binding in the hippocampus CA1 (left column) and CA3 (right column) on the operated side in each group of animals, respectively. In the hippocampus CA1, the CBF thresholds for reduction of the cAMP binding were 18, 34, and 49 ml 100−1 min−1 after 30 min (top graph), 2 h (middle graph), and 6 h (bottom graph) of ischemia, respectively.
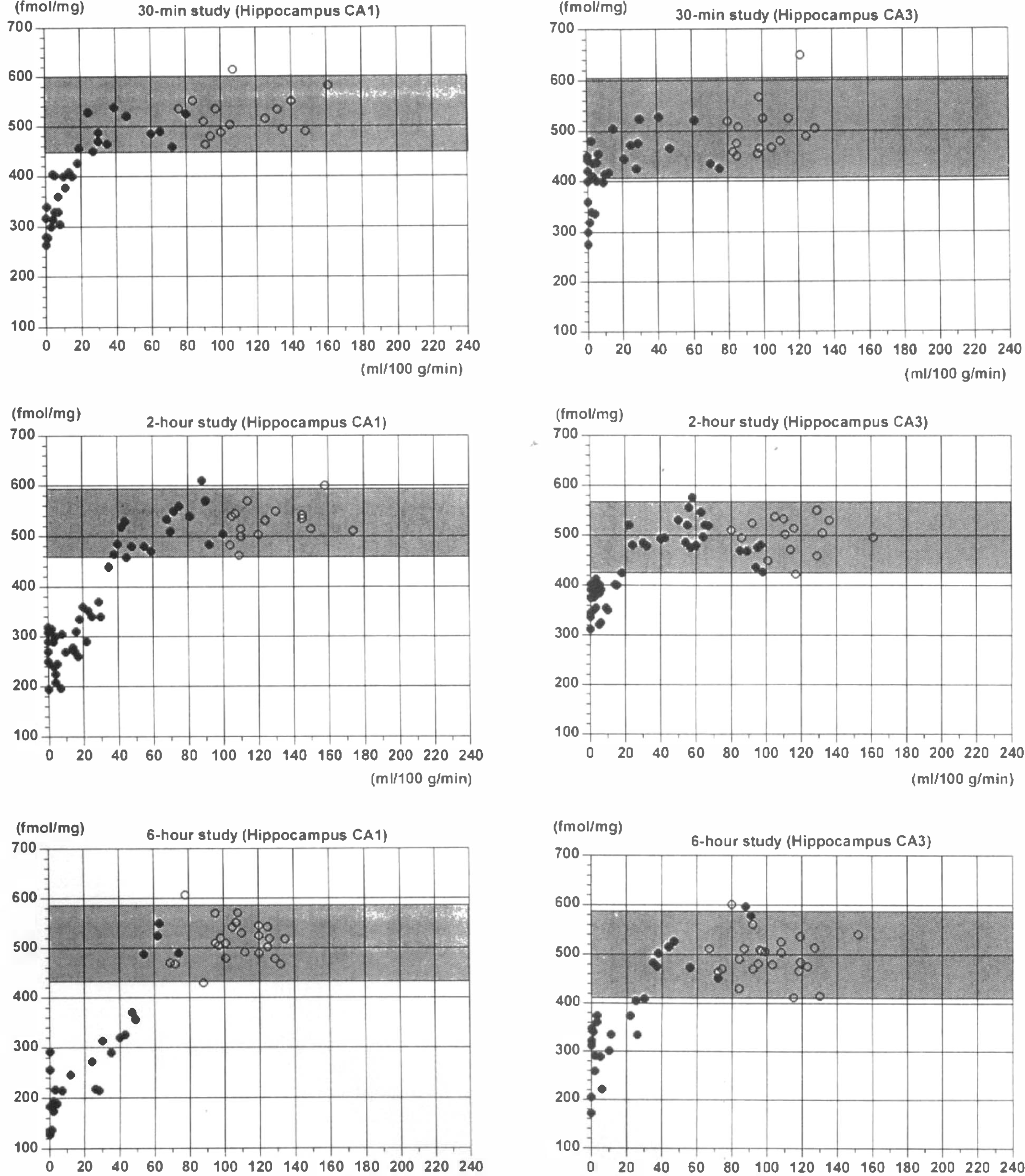
Scatterplots of the relations between CBF (
In the hippocampus CA3, at 30 min of ischemia (top graph), the [3H]cAMP binding showed a large variation from a significant reduction to no definite alteration, when the CBF was reduced to <10 ml 100 g−1 min−1. At 2 h (middle graph) and 6 h (bottom graph) of ischemia, the CBF thresholds for reduction of the cAMP binding were 18 and 30 ml 100 g−1 min−1, respectively.
In the temporal cerebral cortex, at 30 min of ischemia, the [3H]cAMP binding remained within the normal range, even when the CBF was reduced to <10 ml 100 g−1 min−1. At 2 h of ischemia, a moderate reduction in cAMP binding was observed when the CBF fell below 18 ml 100 g− min−1. At 6 h of ischemia, a definite reduction in cAMP binding was associated with a CBF of <23 ml 100 g−1 min−1.
DISCUSSION
The autoradiographic techniques employed in the present study permitted us to measure the CBF only once at the end of the ischemic period. However, it seems rational to use the resultant CBF value for estimating the extent of CBF reduction during carotid occlusion, because Ishihara (1984) in our laboratory observed a stable reduction in CBF on the occluded side with the same ischemia model as that used in the present study. The autoradiographic [14C]IAP method is best suited for correlating the CBF value with the [3H]cAMP binding.
The major findings of the present study can be summarized as follows: (a) the hippocampus CA1 demonstrated higher values of CBF threshold for reduction in the cAMP binding than did the hippocampus CA3 and the temporal cerebral cortex at each duration of ischemia (30 min, 2 h, and 6 h), suggesting that the cAMP system in the hippocampus CA1 may be more vulnerable to acute ischemic stress than the other regions; and (b) a progressive increase in CBF threshold for reduction in the cAMP binding was noted during the time course of ischemia in the hippocampus CA1 and CA3, suggesting that the duration of ischemia exerts a definite influence on the viability of ischemic neuronal cells.
Although cAMP has little selectivity for the two major types of regulatory subunits (RI and RII), the brain contains significantly more RII than RI (Ludvig et al., 1990). We can assume, therefore, that the present data reflect mainly the changes in RII of cAMP-DMK. In the brain of mammalian species, particulate cAMP-DMK and soluble cAMP-DMK exist in an ∼1:1 ratio. Because our incubation protocol is expected to wash out soluble proteins from the unfixed brain tissue into the buffer solution, the present study is considered to demonstrate mainly the membrane-bound particulate cAMP-DMK (Gundlach and Urosevic, 1989). Indeed, the localization of the [3H]cAMP binding observed in the present study was consistent with that of particulate cAMP-DPK to neuronal somata and dendrites as determined by immunohistochemical techniques (Lohmann and Walter, 1984; Urosevic and Gundlach, 1988; Gundlach and Urosevic, 1989; Ludvig et al., 1990).
As stated by Gundlach and Urosevic (1989), the present binding protocol for [3H]cAMP including the 20-min preincubation treatment plus 90-min incubation of unfixed 20-μm thick slide-mounted brain sections, can be expected to reduce the endogenous cAMP concentrations to negligible levels within the tissue. It can be assumed, therefore, that the quantitative results presented here may not have been adversely affected by the presence of endogenous cAMP. Indeed, consistent with the present findings, cAMP-DMK activity has been reported to undergo a significant and progressive decrease at 1 h to 6 h of ischemia in the cerebral cortex of the gerbil (Schwartz et al., 1976).
A progressive increase in CBF threshold during the time course of ischemia as noted in the present study has also been reported for decline of the ATP content in the cerebral cortex of rats (Mies et al., 1991), for amino acid elevation in the feline cerebral cortex (Matsumoto et al., 1993), and for development of infarction in monkeys (Jones et al., 1981). However, a progressive increase in CBF threshold has not been reported for the hippocampus CA1 before the present study. Similarly, a higher CBF threshold for neuronal damage such as reduction of the cAMP binding in the hippocampus CA1 as compared with other brain regions has not been reported previously. The basic mechanisms underlying such specific vulnerability of the hippocampus CA1 to acute ischemic stress need to be investigated in detail in the future.
