Abstract
The wider clinical acceptance of thrombolytic therapy for ischemic stroke has focused more attention on experimental models of reversible focal ischemia. Such models enable the study of the effect of ischemia of various durations and of reperfusion on the development of infarctions. We used high-resolution positron emission tomography (PET) to assess cerebral blood flow (CBF), cerebral metabolic rate of oxygen (CMRO2), oxygen extraction fraction (OEF), and cerebral metabolic rate of glucose (CMRglc) before, during, and up to 24 h after middle cerebral artery occlusion (MCAO) in cats. After determination of resting values, the MCA was occluded by a transorbital device. The MCA was reopened after 30 min in five, after 60 min in 11, and after 120 min in two cats. Whereas all cats survived 30-min MCAO, six died after 60-min and one after 120-min MCAO during 6–20 h of reperfusion. In those cats surviving the first day, infarct size was determined on serial histologic sections. The arterial occlusion immediately reduced CBF in the MCA territory to <40% of control, while CMRO2 was less affected, causing an increase in OEF. Whereas in the cats surviving 24 h of reperfusion after 60- and 120-min MCAO, OEF remained elevated throughout the ischemic episode, the initial OEF increase had already disappeared during the later period of ischemia in those cats that died during the reperfusion period. After 30-min MCAO, the reperfusion period was characterized by a transient reactive hyperemia and fast normalization of CBF, CMRO2, and CMRglc, and no or only small infarcts in the deep nuclei were found in histology. After 60- and 120-min MCAO, the extent of hyperperfusion was related to the severity of ischemia, decreased CMRO2 and CMRglc persisted, and cortical/subcortical infarcts of varying sizes developed. A clear difference was found in the flow/metabolic pattern between surviving and dying cats: In cats dying during the observation period, extended postischemic hyperperfusion accompanied large defects in CMRO2 and CMRglc, large infarcts developed, and intracranial pressure increased fatally. In those surviving the day after MCAO, increased OEF persisted over the ischemic episode, postischemic hyperperfusion was less severe and shorter, and the perfusional and metabolic defects as well as the final infarcts were smaller. These results stress the importance of the severity of ischemia for the further course after reperfusion and help to explain the diverging outcome after thrombolysis, where a relation between the residual flow and the effectiveness of reperfusion was also observed.
Keywords
Thrombolytic therapy can be effective in ischemic stroke as long as recanalization of an occluded vessel can induce recovery of reversibly ischemic tissue. This “therapeutic window” for improvement of flow is rather short and depends on the existence of viable, but functionally impaired (penumbra), tissue (Astrup et al., 1981) and the extent of residual perfusion within the ischemic territory (Heiss and Rosner, 1983). Since neither viability of tissue nor residual perfusion can be assessed reliably in the peracute stage of a stroke, the effectiveness of recanalization can not be predicted. This is the main reason why results of thrombolytic therapy have been unsatisfactory and controversial (ECASS, 1995; NINDS rt-PA Stroke Study Group, 1995): With recanalization rates of 35.9–83.0% after intraarterial (Zeumer et al., 1983) and 34–50% after intravenous (Del Zoppo et al., 1991; Hacke, 1994) application of recombinant-tissue plasminogen activator (rtPA), not all patients with effective reperfusion improved, but a varying percentage experienced poor outcome after development of space-occupying infarction (Koudstaal et al., 1988) and secondary hemorrhage (Wolpert et al., 1993; Hacke et al., 1995). The incidence of hemorrhagic events is higher with delayed treatment (Levy et al., 1994), and prognosis is poor in patients with a severe hemispheric stroke syndrome including impairment of consciousness and forced head and eye deviation early after onset. Both, large infarction and secondary hemorrhage, signal a deleterious outcome despite thrombolysis and are caused by the early development of ischemic damage related to poor collateral circulation insufficient to preserve the integrity of brain cells and vascular endothelium even for a rather short period.
Early perfusional disturbances eventually leading to fast destruction of tissue and their consequences for recovery after reperfusion can be studied in experimental models of transient focal ischemia. Recent developments in functional imaging technologies enable such changes to be followed noninvasively even in small animals. With high-resolution positron emission tomography (PET), regional cerebral blood flow (rCBF), regional metabolic rate for oxygen (rCMRO2), and glucose (rCMRglc) can be measured repeatedly before and after occlusion of the middle cerebral artery (MCA) in baboons (Tenjin et al., 1992; Pappata et al., 1993) and in cats (Heiss et al., 1994), and the development of the penumbra and the eventual progression to infarction can be followed. We have used repeat determinations of rCBF, rCMRO2, and rCMRglc by PET in transient MCA occlusion (MCAO) and related the observed temporal and spatial patterns of changes in flow and energy metabolism to those observed in a few cases during and after thrombolytic therapy.
MATERIALS AND METHODS
Eighteen adult cats of either sex, weighing 2.5–4.1 kg, were used. Anesthesia was induced with 25 mg/kg i. m. ketamine hydrochloride. After catheterization of the left femoral artery and vein, the cats were tracheostomized, immobilized with 0.2 mg/kg i.v. pancuronium bromide, and artifically ventilated. Anesthesia was continued with 0.8–1.5% halothane in a 70%/30% N2O/O2 gas mixture to circumvent protective effects of ketamine against excitotoxic and ischemic damage (Lees, 1989). An i.v. infusion of 2 ml/kg/h Ringer solution containing 5 mg/kg/h gallamine triethiodide for muscle relaxation was maintained throughout the experiment. Physiologic variables were kept in the normal range known for awake cats (Herbert and Mitchell, 1971). Deep-body temperature was kept at 37°C by means of a controlled heating blanket. The left MCA was exposed transorbitally, and an occluding device was implanted as described elsewhere (Graf et al., 1986). This device enabled occlusion and reopening of the MCA with a microdrive after the orbita was sealed. By this procedure, leakage of cerebrospinal fluid was avoided and intracranial pressure was maintained. In five cats the MCA was reopened after 30 min, in 11 cats after 60 min, and in two cats after 120 min of occlusion. Reopening of the vessel led in all cats to immediate reperfusion to the previously badly supplied territory of the MCA.
Mutiple consecutive PET studies were performed in each cat before and up to 24 h after permanent MCAO. Using a head holder and a cross-hair laser-beam system, the cats were positioned in the scanner gantry such that coronal brain sections corresponding to a stereotaxic cat-brain atlas (Reinoso-Suárez, 1961) were obtained. To guarantee positional stability, the cats were kept in the scanner throughout the entire experiment. Photon attenuation was corrected in each cat by using a transmission scan performed with rotating 68Ge rod sources. For the assessment of CMRO2 CBF, and CMRglc, bolus applications were used (Mintun et al., 1984; Herscovitch et al., 1983; Reivich et al., 1979). For CMRO2 determination, 10 mCi 15O2 was administered in a single breath by the respirator, followed by a 30-s breathhold; a blood volume of 6 ml/100 g was assumed (Heiss et al., 1994). CBF was determined after i.v. bolus injection of 20 mCi 15O-labeled water. CMRglc was measured after injection of 5 mCi [18F]fluoro-2-deoxy-
Serial PET scanning was performed with a 24-ring, high-resolution camera (Siemens/CTI ECAT EXACT HR; Siemans, Knoxville, TN, U.S.A.) with a 15-cm field of view, an in-plane spatial resolution of 3.6 mm full width at half-maximum (FWHM) and an axial resolution of 4.0 mm FWHM (Wienhard et al., 1994). For CMRO2 and CBF studies, a total of 6 × 106 and 107 counts, respectively, were collected for 2 min. For CMRglc studies, a total of 2 × 108 counts was collected for 40 min, starting at 20 min after injection, thus enabling the reconstruction of transaxial slices from 4 million counts per slice. To increase count rates and statistics, activity accumulation was started 20 min after injection, since metabolic rates calculated with individual rate constants stabilized after that period (Heiss et al., 1985). Rate constants normalized to local activity can be used instead of individually determined rate constants (Wienhard et al., 1985). During H215O and 15O2 scans, activity in arterial blood was measured continuously in an arteriovenous shunt using an automatic, calibrated blood-sampling system (Eriksson et al., 1988). Additionally, three arterial blood samples were taken during 15O2 scans for determination of blood gases and for whole-blood and plasma radioactivity measurements in a sample changer cross-calibrated to the camera; mean values were used for parametric image generation. During the FDG studies, eight blood samples were taken starting at tracer injection, and plasma radioactivity was used for CMRglc calculations according to the model equation (Reivich et al., 1979). Additionally, plasma glucose content was determined. At the end of the experiment, usually 24 h after MCAO, the cats were perfusion fixed with formalin (4%) and their brains were removed. Serial 7-μm sections (hematoxylin–eosin-stained or luxol fast blue stained) were obtained in parallel with the PET planes according to the stereotaxic cat-brain atlas (Reinoso-Suárez, 1961). Corresponding to the PET slice thickness, serial sections were analyzed at a section-to-section distance of ∼3 mm for histologic verification of infarcts and to match morphology with the various functional images. Correction for brain, edema was performed according to the method of Swanson et al. (1990).
Parametric PET images of CBF, CMRO2, and oxygen extraction fraction (OEF) were obtained before and up to 11 times within 24 h after MCAO, with each multitracer study taking ∼20 min. CMRglcs (additional 60 min) were determined at ∼6 and 24 h after MCAO. Data analyses were based on the parametric images of 16 transaxial brain slices. The images obtained enabled the identification of the main anatomic structures of the cat's brain and a distinction of gray and white matter with the best resolution obtained in the CMRglc images. Regionalization for quantitative data analysis was accomplished by stepwise definition of thresholds in control CMRglc and ischemic CBF image (see Fig. 1): In a first step, images were masked by including all intracerebral voxels with control CMRgls > 75% of the mean of the planes, and hemispheres were defined (Fig. 1A). In these images, cortical and caudate areas were marked by using a threshold of control CMRglc > 100% of the mean over both hemispheres, combined with rough anatomic definition (Fig. 1B). Thereafter, the earliest ischemic CBF image (Fig. 1C) was used to mark the ischemic territory by using a threshold of CBF < 50% of the mean over the contralateral hemisphere (Fig. 1D). In the surroundings of this empiric threshold, the spatial gradient of CBF was steepest and therefore enabled a good demarcation of the ischemic territory. In a last step, intersecting areas of cortical and caudate areas (Fig. 1B) with ischemic areas (Fig. 1D) were defined as ischemic areas of cortex and caudate nucleus, respectively (Fig. 1E). Means of these regions of interest in percent of individual preischemic controls were used for quantification in sequential multiparametric studies.
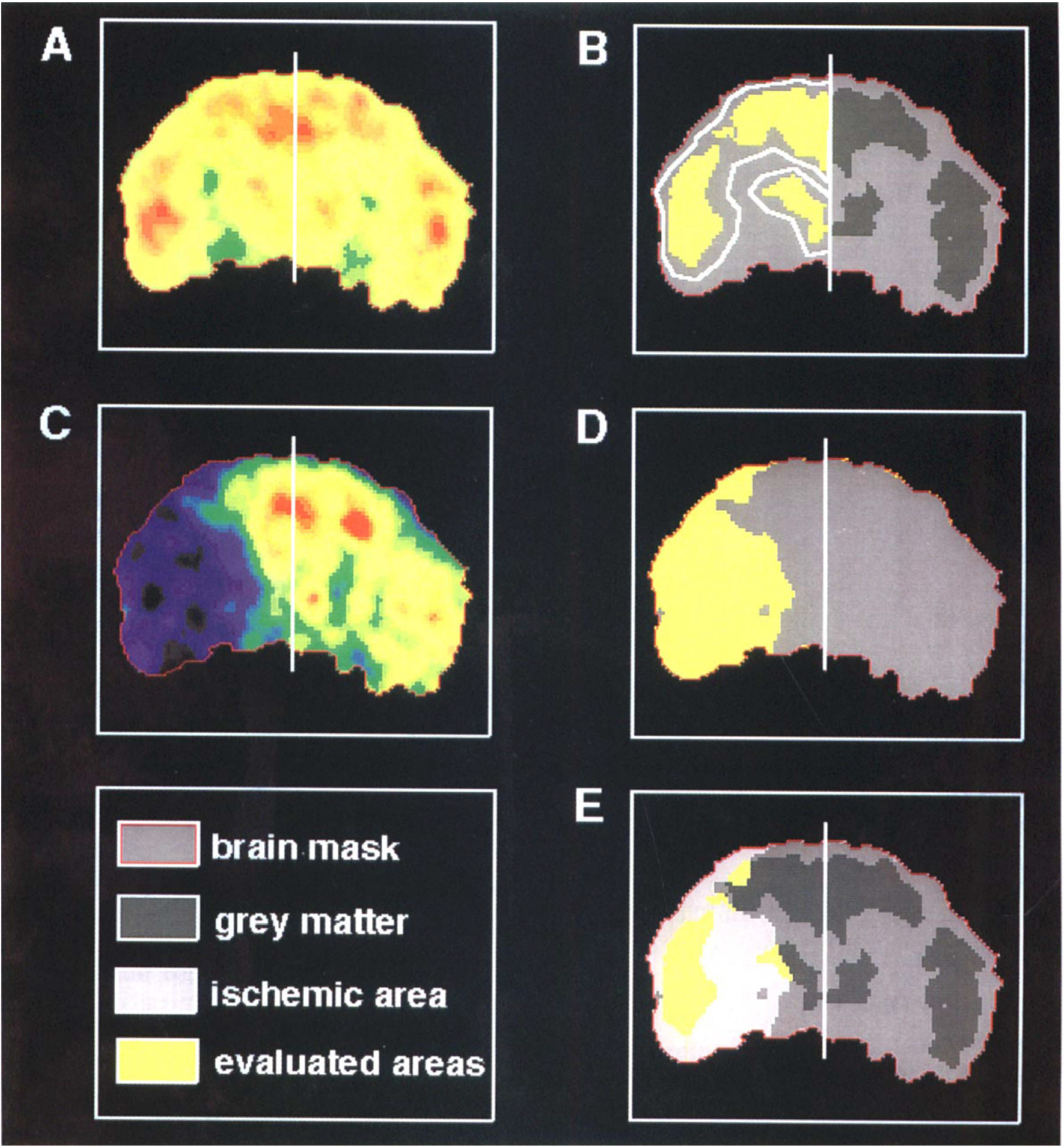
Definition of regions of interest (ROIs) for cerebral cortex and caudate nucleus in a control image of CMRglc (
The experimental protocol included control studies of CMRglc of ∼12 h and of CBF and CMRO2 of ∼0.5 h prior to arterial occlusion. During ischemia, the first measurements of CBF and CMRO2 were started 5 min after occlusion, followed by sequential measurements every 0.5 h (I1 … I4, depending on the duration of MCAO). During reperfusion, the first CBF and CMRO2 measurements were started after 5 min, followed by measurements at 0.5, 1, 2.5, and 4.5 h after reopening of the MCA (R1 … R5). Subsequently, CMRglc (R5) was measured. If possible, final CBF, CMRO2, and CMRglc measurements (R+) were performed ∼24 h after MCAO.
RESULTS
Physiologic PET values obtained in the preischemic control period (Heiss et al., 1995) varied particularly among individual cats. Therefore, values of the consecutive multiparametric studies were normalized in relation to individual preischemic controls. While most cats survived permanent MCAO under prolonged experimental conditions for at least 24 h (Heiss et al., 1994), the survival rate is less with transient MCAO and complete reperfusion. Cats undergoing 30-min MCAO survived the observation period. After 60-min occlusion, however, six of 11 cats died between 6 and 20 h of reperfusion, and one of the two cats with 120-min occlusion died. The other had already developed pronounced spontaneous reperfusion during the early occlusion period and consequently had an even better outcome than most of the cats with 60-min MCAO. All cats not dying spontaneously were killed 24 h after transient ischemia, and their brains were processed for histologic evaluation. For the evaluation of changes in flow and energy metabolism, the cats were grouped according to the further course: five cats with 30-min MCAO; six cats dying within and five cats surviving the observation period after 60-min MCAO. Physiologic parameters did not vary significantly between the various groups and were therefore summarized (see Table 1). During the course of ischemia and reperfusion, neither of the parameters showed major variations. At the end of the experiment (R+), surviving cats showed a tendency for shifts in P
Physiologic parameters (mean ± SD)
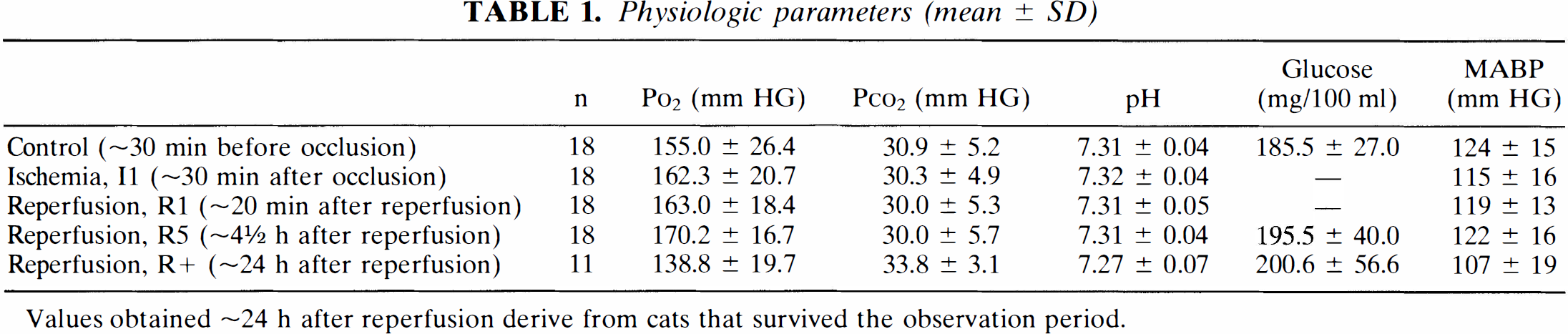
Values obtained ∼24 h after reperfusion derive from cats that survived the observation period.
Ischemic period
In all cats, the arterial occlusion immediately reduced CBF values in the respective supply territory to below ∼40% of its control level, with a distinct but graded transition to surrounding brain tissue (cf. Figs. 2 and 3, I1). At the first measurement during the ischemic period, that is, up to 30 min after MCAO, CMRO2 was less diminished, and consequently OEF was increased (cf. Figs. 2–3 and 4–5, I1) thus indicating that oxygen consumption was still preserved at a level indicating viability of tissue despite severe ischemia (misery perfusion) (Baron et al., 1981). This initial OEF response was small in the caudate nucleus of cats that died after 60-min MCAO (Fig. 5, 12). After the initial ischemic period, the two 60-min MCAO groups differed with respect to changes in blood flow and oxygen consumption: in cats that died, the initial OEF increase had nearly already disappeared during the later period of ischemia, whereas in cats that survived, OEF remained elevated throughout the ischemic episode (Fig. 5, I2). The difference between the two 60-min MCAO groups regarding the second measurement during ischemia (Fig. 5, I2) was significant for cortex and caudate nucleus (p < 0.05, analysis of variance, Newman–Keuls post hoc comparison) despite large variability and a small number of experiments. In several cases, the area with increased OEF spread also to the neighboring vascular territory, especially in the posterior portion off the hemisphere (Fig. 6, surface recordings).
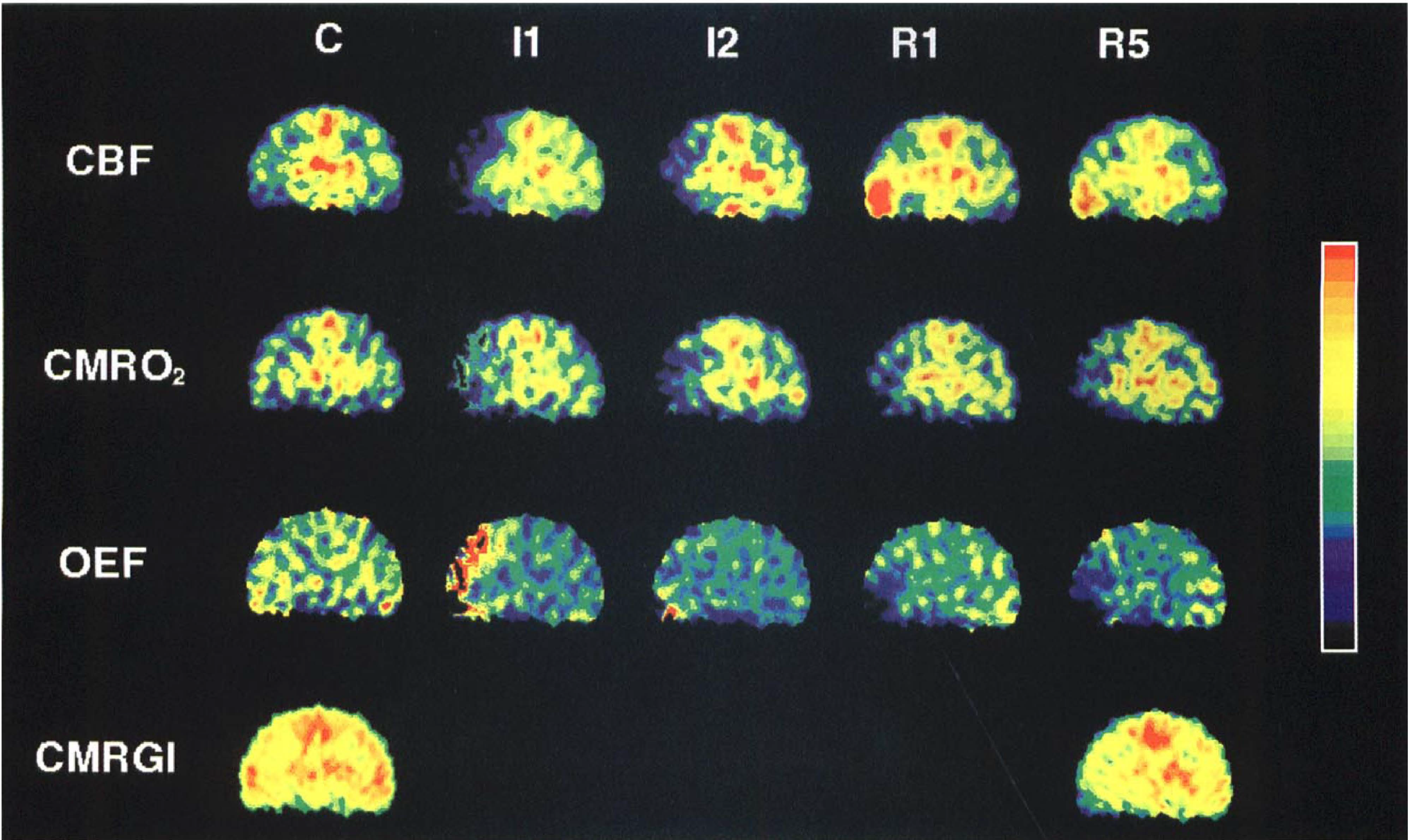
Sequential PET images of plane 7 of an individual cat that died during the experiment. Images represent CBF, CMRO2, OEF, and CMRglc before (C), immediately (I1) and 30 min after the start of 60-min MCAO (I2), and immediately (R1) as well as 4.5 h after reperfusion (R5). During the ischemic episode, CMRO2 deteriorates and OEF does not stay at an elevated level. Sustained hyperperfusion is observed during reperfusion.
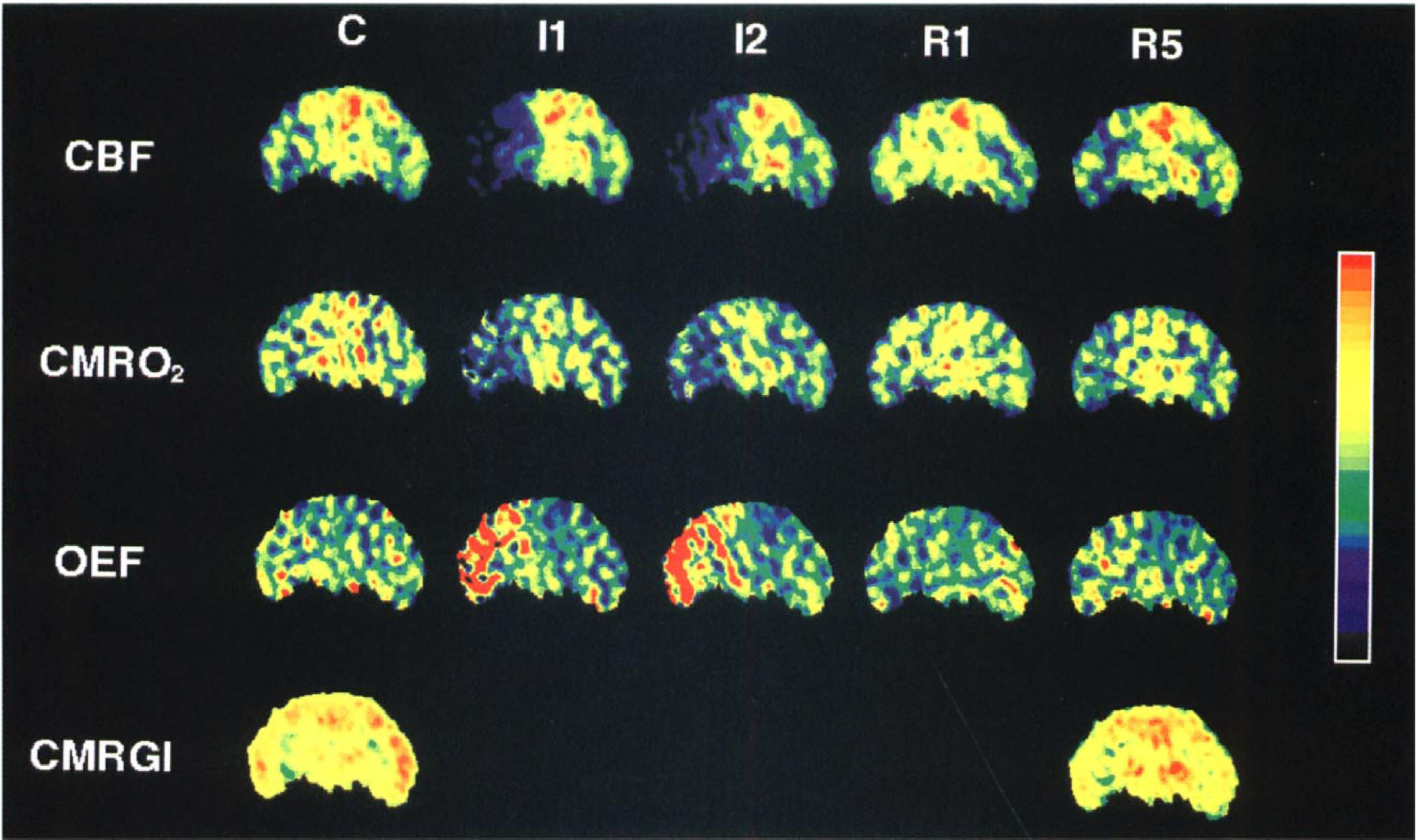
Sequential PET images of plane 7 of an individual cat that survived the 24-h observation period after MCAO/reperfusion. Images represent CBF, CMRO2, OEF, and CMRglc before (C), immediately (I1) and 30 min after the start of 60-min MCAO (I2), and immediately (R1) as well as 4.5 h after reperfusion (R5). During the ischemic episode, CMRO2 did not deteriorate further and OEF increase persisted. Hyperperfusion was not pronounced in this particular cat.
Reperfusion period
Reopening of the MCA induced immediate reperfusion of the previously ischemic territory, with flow values far above control in most instances. Hyperperfusion reached higher values, affected a larger territory, and lasted longer (up to 6 h in exceptional cases; cf. Figs. 2–5) in cats that were going to die than in those surviving the experimental period. A thresholdtype correlation existed between severity of ischemia during MCAO and extent of hyperperfusion after reopening of the vessel (Fig. 7). Since CMRO2 remained below control during reperfusion, OEF was decreased, with a tendency to return slowly to control levels in the later phase of the experiments. A second period of slight misery perfusion followed the early hyperperfusion in two cats after 30-min MCAO and in five after 60-min MCAO (see means of OEF in Figs. 4 and 5). The alternating pattern of OEF—markedly increased during MCAO, reduced during early hyperperfusion, and again moderately increased later—can be best seen in reconstructed OEF surface views of the cat's left hemisphere (Fig. 6). In some of the cases, flow and oxygen metabolism progressively deteriorated also in the hemisphere contralateral to the MCAO, and all of these cats died during the first day.
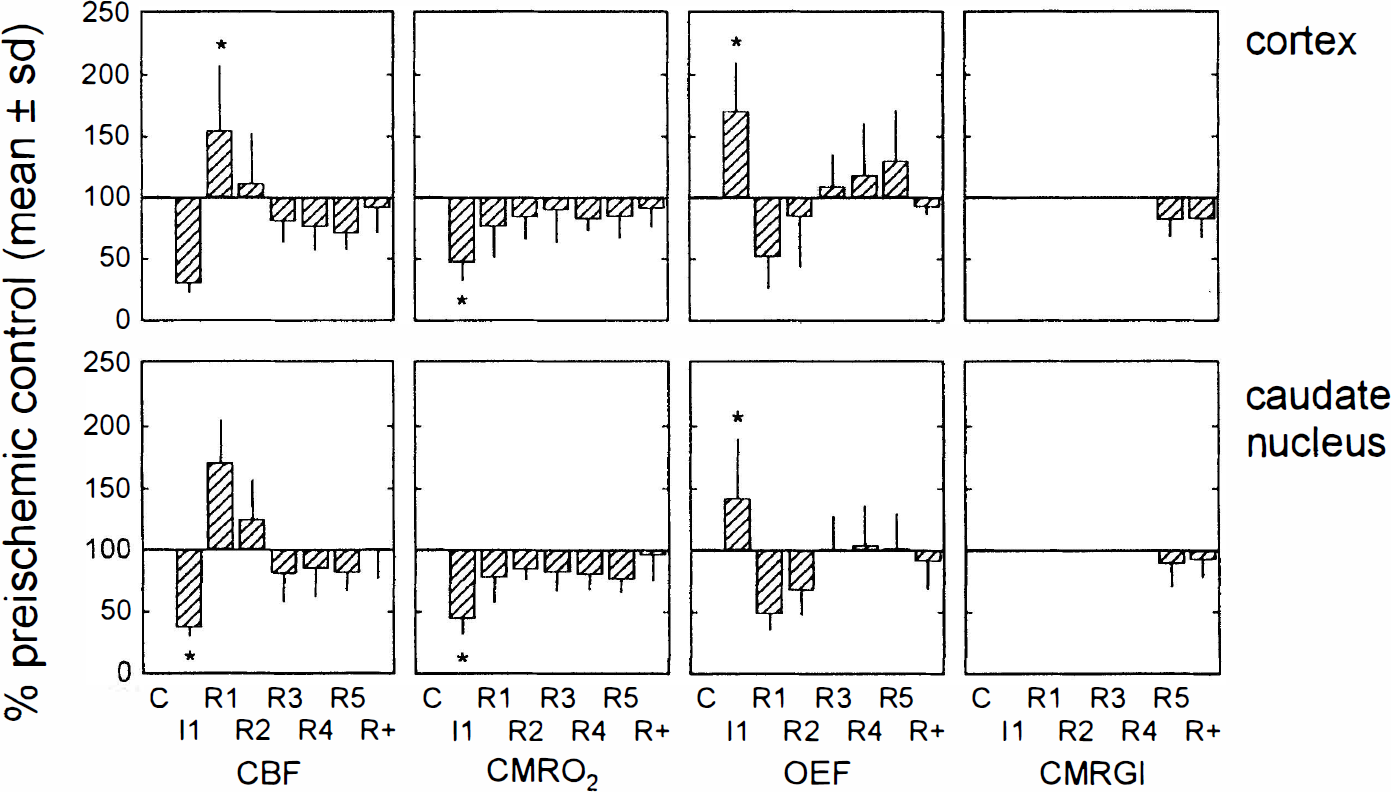
Time course of CBF, CMRO2, OEF, and CMRglc (% control) in ischemic regions of interest of cortex and caudate nucleus. Means ± SD are shown for all 16 planes. Significant differences of ischemic values from preischemic control (>0.05, analysis of variance, repeated-measures design). Measurements were taken immediately after the start of 30-min MCAO (I1), and at six time points during reperfusion (at 5 min and 0.5, 1, 2.5, 4.5, and 20–24 h after reperfusion, R1–R5 and R+).
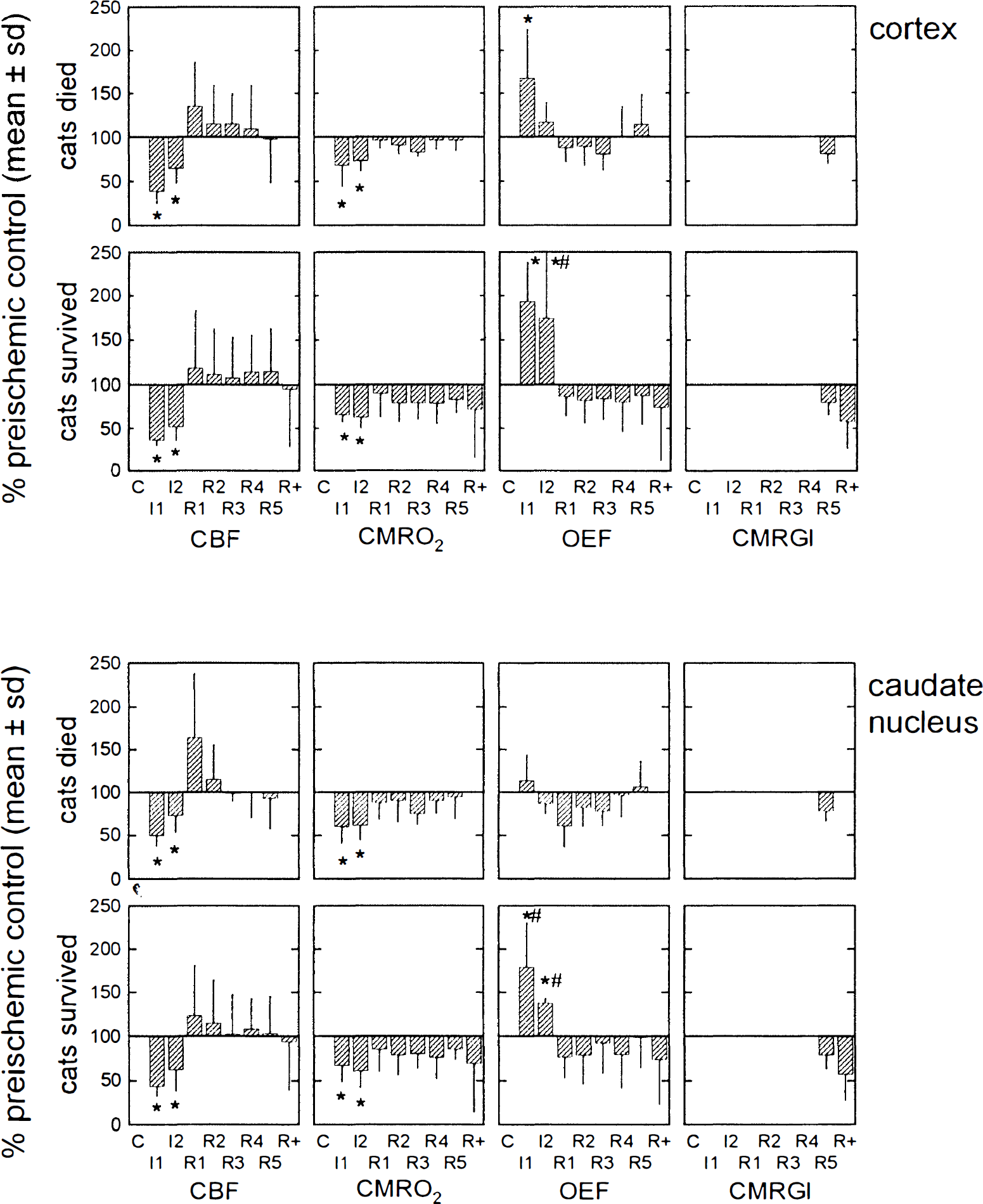
Time course of CBF, CMRO2, OEF, and CMRglc (% control) in ischemic regions of interest (ROIs) of cortex and caudate nucleus in cats that died during reperfusion (n = 6,
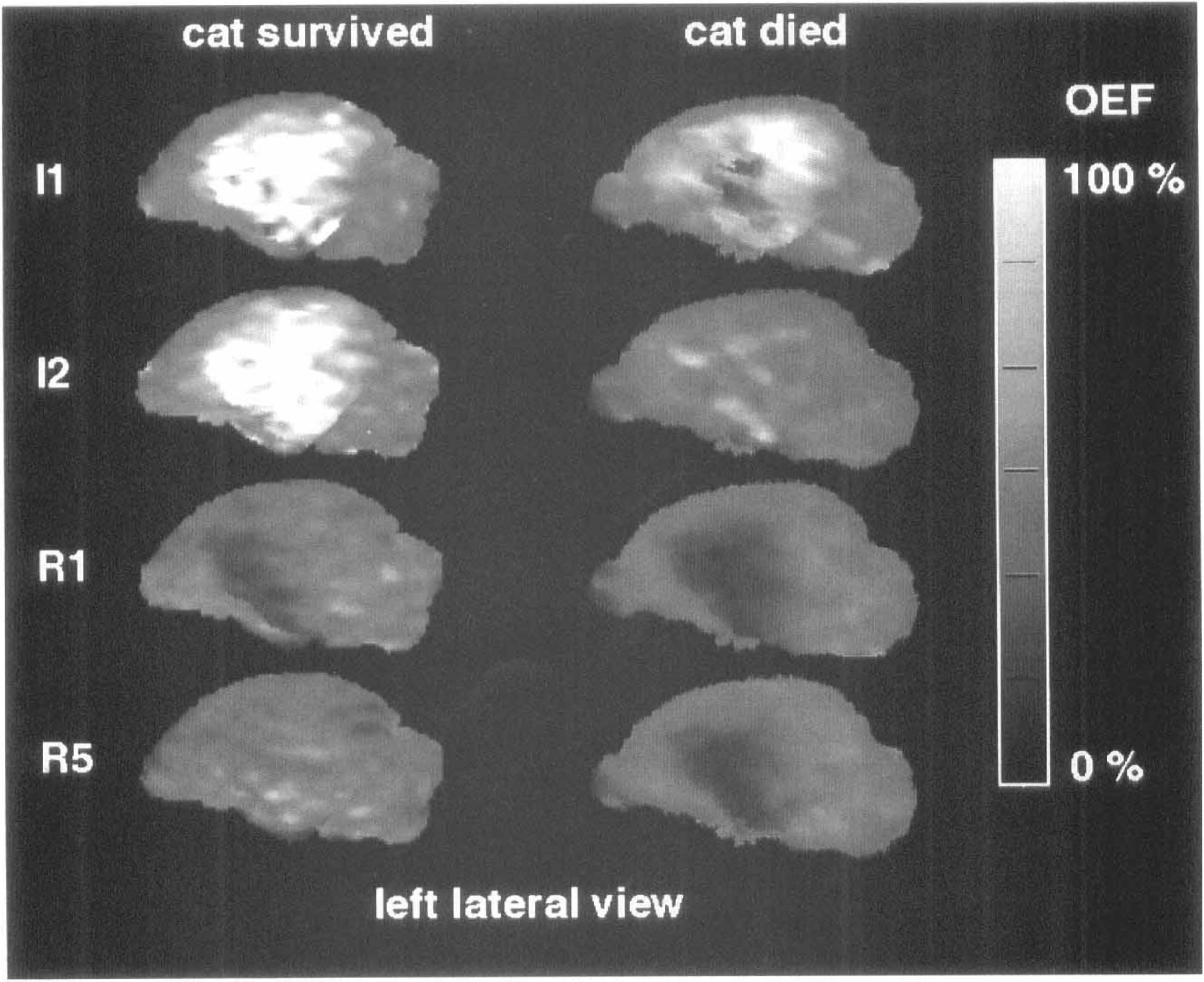
Two examples of variable development of OEF during ischemia (I1, I2) and reperfusion (R1, R5). Reconstructed surface views of OEF masked by control CMRglc in the cats demonstrated in Figs. 2 and 3. Note dynamic deterioration of OEF in the cat that finally died as compared to an almost complete recovery in the surviving cat.
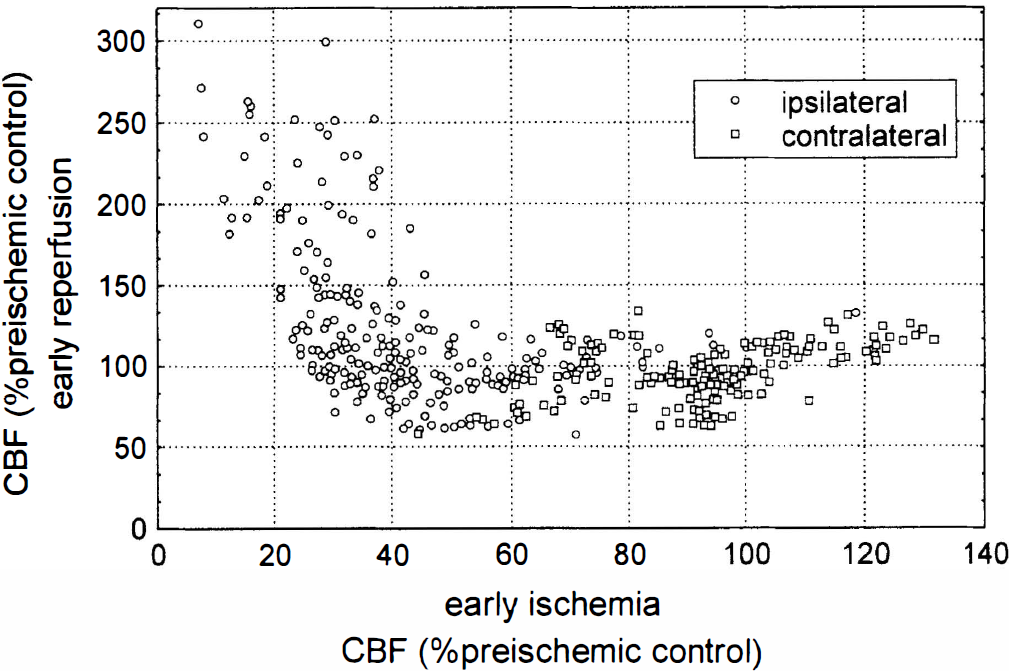
Relationship between early ischemic (I1) and early postischemic CBF (R1) in individual cats. Values are means of consecutive planes of hemispheres ipsilateral and contralateral to the occlusion side. Means obtained at I1 were plotted against those obtained at R1. Note the thresholdtype relationship between the severity of ischemic CBF reduction and the magnitude of postischemic hyperperfusion.
Final state
All cats with 30-min MCAO, five with 60-min MCAO, and one with 120-min MCAO completed the experimental protocol. CBF, CMRO2, and CMRglc were measured 20–24 h after reopening of the MCAO, and the brains were histologically examined after perfusion fixation. After 30-min MCAO, defects of flow and metabolism were small and restricted mainly to the basal ganglia, and the values had returned close to normal in most regions. Accordingly, histologic examination revealed no infarcts or only small lesions confined mainly to the caudate/putamen (Fig. 8). In most cats surviving 60-min and 120-min MCAO, flow and metabolism were depressed in the territory of the MCA, with values in the center diminished to a level indicating irreversible damage (Fig. 6). The size of the ultimate infarct as it was demonstrated in the histologic sections was reflected best by CMRglc images (Fig. 8). Decreases in CMRglc, however, were also observed in the ipsilateral hemisphere outside the infarction and also in the contralateral hemisphere in seven cats with large space-occupying lesions. In the cats that died during the first day, a satisfying histologic workup was not possible because their brains were severely damaged and could not be removed completely for serial sectioning. In all of these cats, gross morphologic examination revealed large infarctions comprising most of the hemisphere, and additional damage to the contralateral hemisphere and the brainstem, probably as a result of fatally increased intracranial pressure.
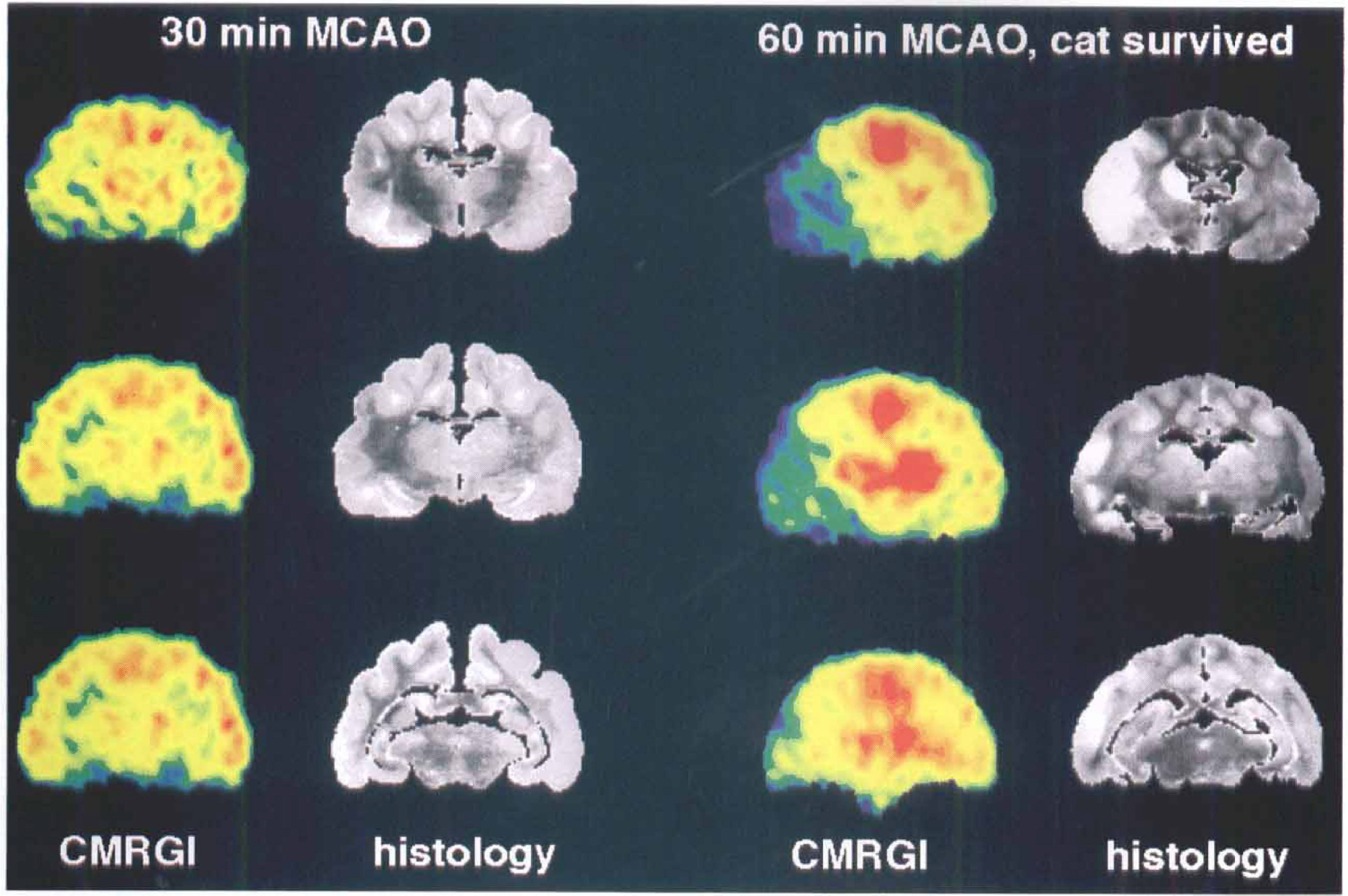
Comparison between final metabolic deficits and histologic damage in two individual cats with 30-mm and 60-mm MCAO, respectively, followed by 20–24 h of reperfusion. CMRglc images at three representative planes and corresponding histologic cross sections (chresyl violet/luxol fast blue stain) are shown. Infarcts are well demarcated in the cat after 60-min MCAO and correspond to areas with metabolic depression in the final stage of the experiment.
DISCUSSION
Experimental model
The MCAO model in the cat has been used extensively to study electrophysiologic, hemodynamic, and biochemical changes induced by focal ischemia. By applying devices for reversible blockade of the vessel, the time windows for induction of neuronal necrosis or gross infarction and the relationship between duration and severity of ischemia could also be established [reviewed by Heiss (1992)]. Recently, state-of-the-art PET technology enabled perfusional and metabolic changes leading to infarction in permanent MCAO to be followed (Heiss et al., 1994). In these studies, the cat MCAO model had more similarities to the observations from single-shot anecdotal examinations of acute ischemic stroke in humans than MCAO in baboons, in which a more delayed and more moderate decrease in flow and metabolism was observed and infarcts were induced variably and confined often only to the basal ganglia (Tenjin et al., 1992; Pappata et al., 1993; Touzani et al., 1995; Young et al., 1995).
Multitracer PET studies yield quantitative assessment of flow and energy metabolism, but they require complex and expensive equipment and complicated logistics and therefore must be restricted to small samples of experimental animals or to a few time points in the course after stroke in selected patients. These technical limitations can be overcome by functional magnetic resonance technologies (fMRI) in which early changes of water diffusion and tissue perfusion (diffusion and perfusion weighted MRI) indicate ischemic compromise (reviewed by Hossmann and Hoehn-Berlage (1995)]. However, the clinical value of these new techniques in the evaluation of early ischemic damage must still be established in larger series (Warach et al., 1996). At present, flow and metabolic data collected by PET in early stroke in humans and in experimental focal ischemia provide an insight into pathophysiology still not surpassed by other noninvasive imaging modalities. Therefore, PET studies can hardly be replaced at present by these new techniques.
Clinical correlates of the various durations of ischemia
There are important differences between experimental animal models of focal ischemia and ischemic stroke in humans; the cat is especially sensitive to increases in intracranial pressure resulting from reperfusion to already damaged tissue. On the other hand, if surviving the critical acute period, cats are able to recover to a high level of function after large infarcts (Heiss et al., 1976). Therefore, a comparison of our results to clinical findings may be justified: Permanent MCAO resembles the natural course after vascular occlusion leading to large infarcts in most cases, with a chance of collateral reperfusion resolving misery perfusion and improving outcome. It was demonstrated in this model that areas of increased OEF spread out with time from the core of ischemia to the border zones of the vascular territory, and that misery perfusion is followed in most cases by necrotic transformation, which again shows a dynamic propagation from the center to the surroundings. Some animals experience collateral perfusion and favorable outcome with minimal permanent defects and small infarcts (Heiss et al., 1994). Even with permanent MCAO, excessive spontaneous reperfusion may occur in regions of initial low residual flow, and this may lead to large infarcts and early death due to severely increased intracranial pressure (Heiss et al., 1976; Hayakawa and Waltz, 1975).
Reopening of the MCA resembles the (spontaneous) dissolution of vascular occlusions in transient ischemic attacks, spontaneous lysis of emboli after a period beyond the tolerable time period, and therapeutic thrombolysis. Reperfusion after 30-min MCAO led to a brief hyperperfusion period and to a fast normalization of flow and metabolism (with or without a phase of delayed misery perfusion). These experimental ischemic episodes may be comparable to transient ischemic attacks and do not cause gross infarctions. During MCAO of longer duration—60 and 120 min—two patterns can be distinguished: in approximately half of the animals, decrease in the OEF during MCAO reflects fast, irreversible tissue damage, whereas the persistence in raised OEF indicates preserved viability of tissue over the ischemic period. During ischemia, OEF seems to be the better indicator of damage than are CBF and CMRO2, which are more affected by high variability. Forced reperfusion by reopening of the MCA cannot salvage already irreversibly damaged tissue, but may cause additional damage by inducing edema via leaking vascular endothelium, and this effect is aggravated by severe and prolonged hyperperfusion in paralyzed vessels. In such cases, the infarcts are large and animals die early due to increased intracranial pressure; these courses resemble the deleterious outcome of thrombolytic therapy that was initiated to late, could not prevent the development of large infarcts, and induced additional edema and secondary hemorrhagic transformation.
Some PET findings in six patients (Heiss et al., 1997) treated acutely with thrombolysis (rt-PA, 0.9 mg/kg) according to the protocol of the NINDS controlled trial (1995) underline the comparability with our experimental data. All cases showed a marked perfusional deficit in the area of the MCA, but the level of residual perfusion showed some differences: in four cases, uptake of 15O-labeled water into border zones of the ischemic territory indicated some spontaneous collateral circulation whereas, in two other cases, the markedly decreased isotope uptake suggests low residual flow (Fig. 9). A complete reperfusion of the ischemic territory with marked hyperperfusion was observed in four cases 1–3 days after the vascular attack, and these patients recovered completely from their neurologic deficit (Fig. 9, top row). In the further course, gross infarction could not be detected on computed tomography or MRI, and metabolic and flow images nearly normalized, leaving some regional defects as indicators of discrete cellular loss or ischemic white-matter lesions without clinical correlates. In the two patients with large flow defects with very low residual perfusion to the ischemic territory (Fig. 9, bottom row), reperfusion could only be achieved by the border zones of ischemia, and large defects persisted on the flow images. Large infarcts were seen on computed tomography on the following day, often with space-occupying effects. These patients suffered from a complete neurologic syndrome, the clinical course after stroke was severe, and clinical stabilization was delayed.
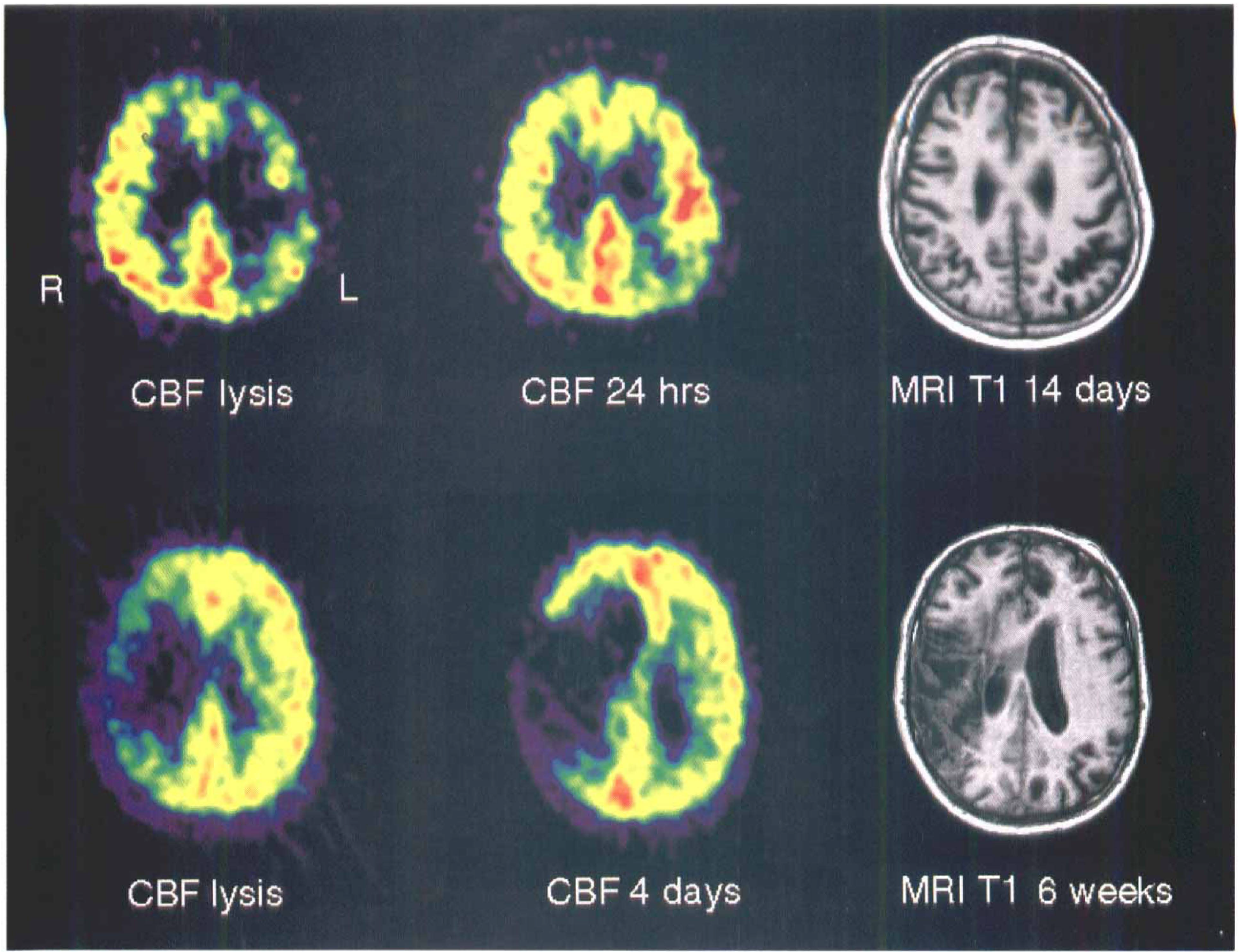
Examples of PET CBF and MRI studies in two patients with thrombolytic therapy.
Ischemic damage versus reperfusion injury
Reperfusion injury is seen as an important factor of brain damage initiated by ischemia [reviewed by Dietrich (1994)] and may represent a paradoxical consequence of spontaneous or pharmacologically induced reperfusion (Hallenbeck and Dutka, 1990; Hacke et al., 1996). Under certain experimental conditions, reperfusion may exacerbate brain injury and may be responsible for progression of neuronal loss over prolonged postischemic periods (Jenkins et al., 1981; Kirino, 1982; Pulsinelli et al., 1982; Nakano et al., 1990). In stroke patients, fetal edema formation during reperfusion after thrombolysis with rtPA was attributed to reperfusion injury (Koudstaal et al., 1988) even though experimental studies with transient vessel occlusion failed to demonstrate that reperfusion increased infarct size (Kaplan et al., 1991; Memezawa et al., 1992). There is strong evidence, however, that reperfusion aggravates edema formation (Ito et al., 1979; Nagasawa and Kogure, 1989; Nishigaya et al., 1991), which was shown to depend on severity and duration of ischemia and the extent of reperfusion (Symon et al., 1979; Bell et al., 1985; Kataoka et al., 1987; Slivka et al., 1995).
Our experimental results with reversible MCAO in cats and the observations in a few stroke patients with thrombolysis suggest that the severity of ischemia measurable as residual perfusion is the main factor determining the further course and the extent of tissue damage. If reperfusion is achieved in preserved tissue perfused at a level above the threshold for morphologic integrity (Heiss and Rosner, 1983; Garcia; Garcia et al., 1983), subsequent damage is small, metabolism and flow recover to values close to normal, and the clinical course is favorable, without delayed neurologic impairment indicating reperfusion damage. Reactive hyperemia as a functional overshoot reaction (De Crespigny et al., 1992) was observed after the 30-min ischemic episode. After 60-min MCAO, the grade of postischemic hyperperfusion was related to the severity of ischemia (Fig. 7). This result suggests that reperfusion forced through vessels maximally dilated by lactacidosis affected already damaged tissue, the additionally induced severe edema further increased intracranial pressure, and all factors together caused early death. The same results were obtained when thrombolysis induced reperfusion to some already irreversibly damaged areas bordering large infarcts, where the increased vascular permeability during the revascularization forces edema formation and parenchymal hemorrhage and enhances ischemic tissue damage by various mechanisms, including massive increase in excitatory amino acids (Benveniste et al., 1984), production of free oxygen radicals (Chan, 1994; Tasdemiroglu et al., 1994) and excessive Ca2+ influx into cells (Uematsu et al., 1988). This concept is supported by the experimental results demonstrating that early reperfusion preserves penumbra tissue whereas late reperfusion increases tissue injury (Yang and Betz, 1994) and the clinical observation that the quality of collateral flow is essential for the spontaneous recovery (Ringelstein et al., 1992) as well as for the beneficial effects of thrombolytic therapy (Frey et al., 1995; Baird et al., 1994). In permanent vessel occlusion, however, where misery-perfused potentially viable tissue can be observed for many hours, but is later included in growing infarctions (Heiss et al., 1992; Marchal et al., 1993; Higano et al., 1993), the penumbra zone propagates with time from the center to the border zone of ischemia and is eventually followed by necrotic transformation or recovery (Heiss et al., 1994); under these circumstances, molecular and biochemical mechanisms, eventually triggered by waves of spreading depression like depolarization (Hossmann, 1994) inducing progressive metabolic impairment, contribute significantly to a complex vicious cycle enhancing ischemic damage.
In conclusion, PET measurements in cat MCAO can serve as a model to explain varius courses and diverging outcomes after thrombolysis in acute ischemic stroke. While permanent MCAO mimics the spontaneous course of ischemic hemispheric stroke leading to large infarcts with the chance for collateral reperfusion improving outcome, 30-min occlusion is comparable to transient ischemic attacks and does not result in gross infarction. Reperfusion after 60 min of severe focal ischemia leads to large infarct and early death; this might be a correlate for the deleterious effects of thrombolytic therapy initiated at a state when tissue and endothelial damage have already developed and forced reperfusion causes fatal perifocal edema. Reperfusion after 60-min MCAO to moderately ischemic tissue (due to some spontaneous reperfusion) leads to small infarcts; this corresponds to the beneficial effects of thrombolytic therapy initiated within the therapeutic window when residual flow still preserves morphologic integrity of the tissue. Our findings also stress the importance of the residual flow or the severity of ischemia for the further course after reperfusion: Forced reperfusion to already irreversibly damaged tissue induces edema formation, enlarges developing infarcts, and thereby may cause a deleterious increase in intracranial pressure, whereas reperfusion to viable penumbra tissue prevents morphologic damage and reverses neurologic deficits.
