Abstract
Interleukin (IL) 8 was measured in CSF of 14 patients with severe traumatic brain injury. IL-8 levels were significantly higher in CSF (up to 8,000 pg/ml) than serum (up to 2,400 pg/ml) (p < 0.05), suggesting intrathecal production. Maximal IL-8 values in CSF correlated with a severe dysfunction of the blood–brain barrier. Nerve growth factor (NGF) was detected in CSF of 7 of 14 patients (range of maximal NGF: 62–12,130 pg/ml). IL-8 concentrations were significantly higher in these patients than in those without NGF (p < 0.01). CSF containing high IL-8 (3,800–7,900 pg/ml) induced greater NGF production in cultured astrocytes (202–434 pg/ml) than samples with low IL-8 (600–1,000 pg/ml), which showed a smaller NGF increase (0–165 pg/ml). Anti-IL-8 antibodies strongly reduced (52–100%) the release of NGF in the group of high IL-8, whereas in the group with low IL-8, this effect was lower (0–52%). The inability of anti-IL-8 antibodies to inhibit the synthesis of NGF completely may depend on cytokines like tumor necrosis factor α and IL-6 found in these CSF samples, which may act in association with IL-8. Thus, IL-8 may represent a pivotal cytokine in the pathology of brain injury.
A traumatic event to the brain leads to an inflammatory response that is modulated by the release of cytokines through a complex cytokine cascade (for review see Morganti-Kossmann and Kossmann, 1995). Although chronic inflammation within the CNS can be considered deleterious for the brain tissue, cytokines have also the ability to promote the induction of nerve growth factor (NGF) (Nieto-Sampedro et al., 1982; Gadient et al., 1990; Spranger et al., 1990; Morganti-Kossmann et al., 1992a).
Induction of interleukin (IL) 1, IL-6, and tumor necrosis factor a (TNF-α) was found in rodent brain following different types of injuries (Giulian and Lachman, 1985; Giulian et al., 1988; Nieto-Sampedro and Berman, 1987; Woodroofe et al., 1991; Taupin et al. 1993). Furthermore, enhanced production of IL-1 and IL-6 was shown in serum and CSF of brain-injured patients (McClain et al., 1987, 1991; Kossmann et al., 1995) as well as the release of IL-8, the latter, however, only in serum (Ott et al., 1994). Recent studies from our laboratory demonstrated that increased levels of IL-6 persist for a long time period in the CSF and serum of patients with severe head injury (Kossmann et al., 1995, 1996). In addition, we showed that the concentrations of IL-6 and nerve growth factor (NGF) in human CSF strongly correlate with each other and that IL-6 produced in vivo induces NGF in cultured astrocytes (Kossmann et al., 1996). However, these results also indicate that further cytokines may be involved in promoting the synthesis of NGF following traumatic brain injury (TBI). Therefore, IL-8 was monitored in the CSF and serum of patients with severe brain injury, and an additional mechanism of NGF induction triggered by IL-8 was investigated.
IL-8 is a pro-inflammatory cytokine, with potent chemotactic and activating properties on neutrophils (for review see Baggiolini et al., 1992). A variety of cell types can synthesize IL-8, such as monocytes/macrophages, endothelial cells, and neutrophils following stimulation with IL-1 and TNF-α (Bazzoni et al., 1991; Dinarello, 1991). Astrocytes produce IL-8 in response to IL-1 and TNF-α challenge, whereas constitutive expression of IL-8 was demonstrated in activated and transformed astrocytes (Aloisi et al., 1992; Nitta et al., 1992). In addition to its properties shown toward neutrophils, IL-8 displays neurotrophic activity on hippocampal neurons (Araujo and Cotman, 1993). Moreover, the expression of the IL-8 receptor on glial cells suggests that this factor may regulate other activities in these cells (Lacy et al., 1995).
IL-8 is synthesized in a variety of diseases of the CNS such as bacterial meningitis, meningoencephalitis (Handa, 1992; van Meir et al., 1992; Halstensen et al., 1993; Seki et al., 1993; Mastroianni et al., 1994), and malignant brain neoplasms (Nitta et al., 1992; van Meir et al., 1992; Morita et al., 1993; Tada et al., 1993). However, the role of IL-8 in neuropathologies including TBI has not yet been fully elucidated.
The objective of this study was to monitor the production of IL-8 in CSF and serum of patients with severe TBI and to investigate whether IL-8 may play a role in the induction of NGF that may be relevant for the repair of the lesioned nervous system.
MATERIAL AND METHODS
Patients
Fourteen patients (three women, 11 men; age range 18–74 years, mean age 38 years) with the diagnosis of severe isolated TBI, admitted to the Division of Trauma Surgery at the University Hospital of Zürich, Switzerland, were included in the study. Of these patients, 13 suffered from a closed TBI, while only one patient had an open skull fracture (Patient 8). All patients had a Glasgow Coma Score (GCS) of <9 (Teasdale et al., 1974) at admission and alterations in the computed tomography (CT). After clinical and CT evaluation, the patients received an indwelling ventricular catheter for intracranial pressure (ICP) monitoring and therapeutic drainage of CSF within 2–4 h of admission. Four patients needed craniotomy for evacuation of hematomas. Three died as a result of untreatable elevated ICP. The remaining 11 patients recovered to various degrees as determined by the Glasgow Outcome Score (GOS) (Jennett and Bond, 1975). Table 1 shows a summary of the clinical data. After surgery all patients were transferred to the intensive care unit and treated following a standardized protocol (Stocker et al., 1995), which provides CSF drainage whenever ICP exceeds 15 mm Hg. Intraventricular catheters were removed after the ICP remained stable (< 15 mm Hg) for at least 24 h. Patients with significant systemic injuries requiring surgical intervention for thoracic, abdominal, or spinal cord injuries as well as pelvic or long bone fractures were excluded from the study. CSF from eight patients without traumatic head injuries was used as control following either lumbar diagnostic puncture or ventriculoperitoneal shunt. The study protocol was approved by the University Hospital Medical Ethics Board, Zürich, Switzerland.
Clinical data from 14 patients with severe traumatic brain injury
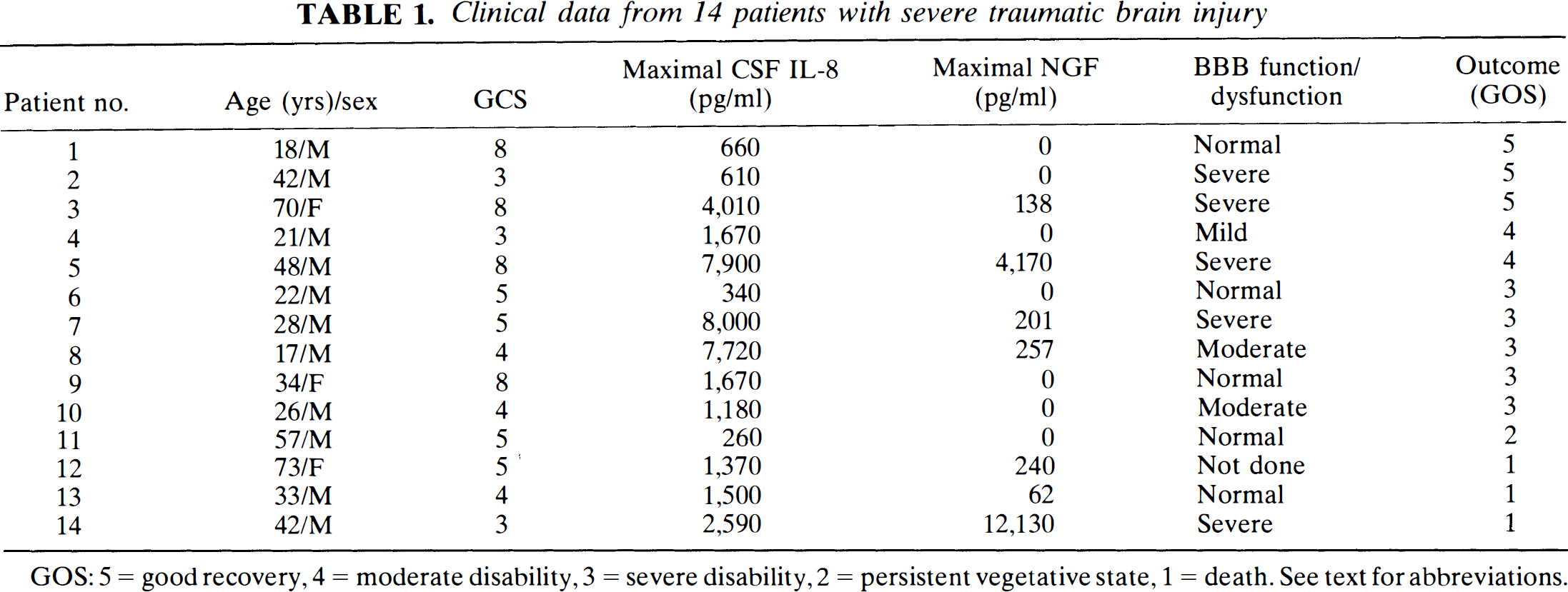
GOS: 5 = good recovery, 4 = moderate disability, 3 = severe disability, 2 = persistent vegetative state, 1 = death. See text for abbreviations.
Serum and CSF sampling
Drained CSF from patients was collected over 24 h at 4°C, centrifuged at 170 g (1,000 rpm) for 10 min at 4°C, aliquoted, and frozen at −70°C until analysis. In addition, parallel blood samples were collected once a day and immediately processed as described.
Blood–brain barrier
According to Reiber and Felgenhauer (1987), the CSF/serum albumin ratio (QA) is considered a sensitive parameter for the dysfunction of the blood–brain barrier (BBB). This was calculated daily to monitor the alteration of BBB permeability during the entire study period. The disturbance of the BBB was assessed as follows: QA values below 0.007 were regarded as normal, between 0.007 and 0.01 as a sign of a mild dysfunction, between 0.01 and 0.02 of a moderate dysfunction, and above 0.02 of a severe BBB dysfunction. Albumin levels were measured by automatized laser photometry (BNA Automat; Behring Werke, Marburg, Germany).
Analysis of cytokines and NGF
The levels of cytokines and NGF were analyzed by sandwich enzyme-linked immunosorbent assay (ELISA) as established by Halstensen et al. (1993) for IL-8 and by Kossmann et al. (1996) for NGF. IL-8 and NGF concentrations were calculated from a standard curve using purified human IL-8 (Sandoz, Basel, Switzerland) and mouse 2.5 S NGF purified from mouse submaxillary glands or human recombinant NGF-β (Boehringer Mannheim, Mannheim, Germany) depending on the samples (cell supernatants from mouse astrocytes or human CSF). The sensitivity of the assays was 20 and 5 pg/ml for IL-8 and NGF, respectively. A few CSF samples were also assayed for IL-1, IL-6, and TNF-α using two commercial kits (R & D Systems, Minneapolis, MN, U.S.A.). A microplate reader (Dynatech Laboratories, Alexandria, MD, U.S.A.) was used for measuring the absorbance.
Astrocyte cultures
Purified astrocytes were isolated from newborn mouse brains. Brain hemispheres were separated from the cerebellum, and after removal of the meninges, trypsinized for 15 min in a solution of 0.5% trypsin/1 mM ethylenediamine-tetraacetate/0.25 mg/ml DNase I (Boehringer Mannheim) in Hanks' Balanced Salt Solution without Ca2+ and Mg2+ (CMF-HBSS). The tissue was then resuspended in Dulbecco's Modified Eagle Medium (DMEM) and mechanically dissociated in CMF-HBSS containing 0.5 mg/ml DNase I using a fire-polished pasteur pipette. Cells were resuspended in DMEM supplemented with 10% low endotoxin (< 10 pg/ml) and heat-inactivated fetal calf serum (FCS) and plated in tissue culture flasks previously coated with 0.1 mg/ml poly-L-lysine (Sigma, Buchs, Switzerland). Cultures were maintained for 8–10 days in a humidified incubator at 37°C and 5% CO2 to obtain a mixed glial cell culture. Microglia and oligodendrocytes were separated from astrocytes by mechanical shaking as described elsewhere (Morganti-Kossmann et al., 1992b). Culture purity was determined by immunostaining using a polyclonal antibody to glial fibrillary acidic protein (Boehringer Mannheim), and >95% of the cells were found positive.
Stimulation of astrocytes
Astrocyte cultures were grown to confluency. Prior to stimulation, the cells were washed three times with CMF-HBSS and incubated overnight with DMEM + 1% FCS, then washed twice and incubated with CSF of three brain-injured patients for 3 days. Following stimulation, culture supernatants were collected, centrifuged at 170 g, and assayed for NGF as already described. For each patient, two samples were used: one with high and one with low IL-8. This was equivalent to, for Patient 1, 7,900 and 600 pg/ml for high and low IL-8, respectively, which were collected on days 17 and 21; for Patient 2, 3,800 and 700 pg/ml collected on days 1 and 7; and for Patient 3, 6,230 and 970 pg/ml collected on days 4 and 14.
To demonstrate the specificity of IL-8 in the CSF for inducing NGF production, CSF samples were preincubated with anti-human IL-8 antibodies (1–10 μg/ml). The antibody concentrations needed for neutralization were calculated based on data provided by the manufacturer (R & D Systems). CSF samples were incubated with antibodies at 4°C for 12 h prior to the stimulation experiments. Since preformed NGF was present in four CSF samples before being added to the cultures, the synthesis of NGF of astrocyte origin was computed as
astrocyte NGF = (NGF in culture supernatants) − (NGF present in human CSF)
The extent of inhibition of NGF production was calculated as follows:
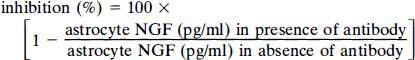
For control, astrocytes were incubated with DMEM + 1% FCS and with medium containing anti-IL-8 antibodies (10 mg/ml) as well as with human CSF without IL-8 or other cytokines. Similar experiments were performed by adding human recombinant IL-8 (R & D Systems) in increasing concentrations (10–1,000 ng/ml) to astrocyte cultures followed by incubation for 3 days.
Analysis of data
Statistical analysis was performed on commercially available software (Stat View 4.0; Abacus Concepts, Berkeley, CA, U.S.A.) using Wilcoxon signed rank test with a modified Bonferroni correction for repeated measurements, Spearman rank correlation test, linear regression analysis, Mann–Whitney U test, Student t test, and Kruskal–Wallis test as appropriate. A p value of <0.05 was considered significant. To approximate a normal distribution, IL-8 values were log-transformed and means as well as standard deviations calculated on these data. For these calculations, 0 pg/ml IL-8 concentrations were replaced by 19 pg/ml (20 pg/ml, detection limit of the assay) to allow logarithmic transformation. The same was done for calculation of the IL-8 ratio (QIL-8 = IL-8CSF/IL-8serum). A QIL-8 of > 1 indicates that CSF exceeded serum concentrations.
RESULTS
Release of IL-8 in CSF and serum after brain trauma
Elevated concentrations of IL-8 were measured in CSF samples of the 14 patients with head injury (range of maximal values: 260–8,000 pg/ml) for up to 21 days. Mean values were significantly higher compared with the CSF of control patients (range: 0–170 pg/ml) at all time points. Seven patients had always 2- to 136-fold higher IL-8 concentrations in CSF than serum. In one patient only, IL-8 levels were always higher in serum than CSF. In the remaining six patients, CSF concentrations of IL-8 were 2–256. times higher for the majority of the time points (42–95% of the measurements for each patient), excluding some days in which serum levels exceeded CSF levels up to 15-fold. IL-8 concentrations in serum showed individually different patterns (range of maximal values: 0–2,400 pg/ml, normal range 0–10 pg/ml) and were significantly lower than in CSF (Fig. 1A) (Wilcoxon signed rank test with Bonferroni correction; p < 0.05). Whenever IL-8 in CSF was below the detection limit of the assay (20 pg/ml), there was no IL-8 detectable in serum either. On the other hand, many patients showed at some time points high IL-8 concentrations in CSF without any measurable serum concentrations. Due to the high between- and within-patient variability of cytokine concentrations, calculating a mean ± SD only roughly reflects the differences of IL-8 concentrations in CSF and serum. Therefore, the IL-8 quotient (QIL-8) was calculated for each patient on each day. In this analysis, all values of >1 indicate that IL-8 levels are higher in CSF than serum as shown at most time points for all 14 patients (Fig. 1B).
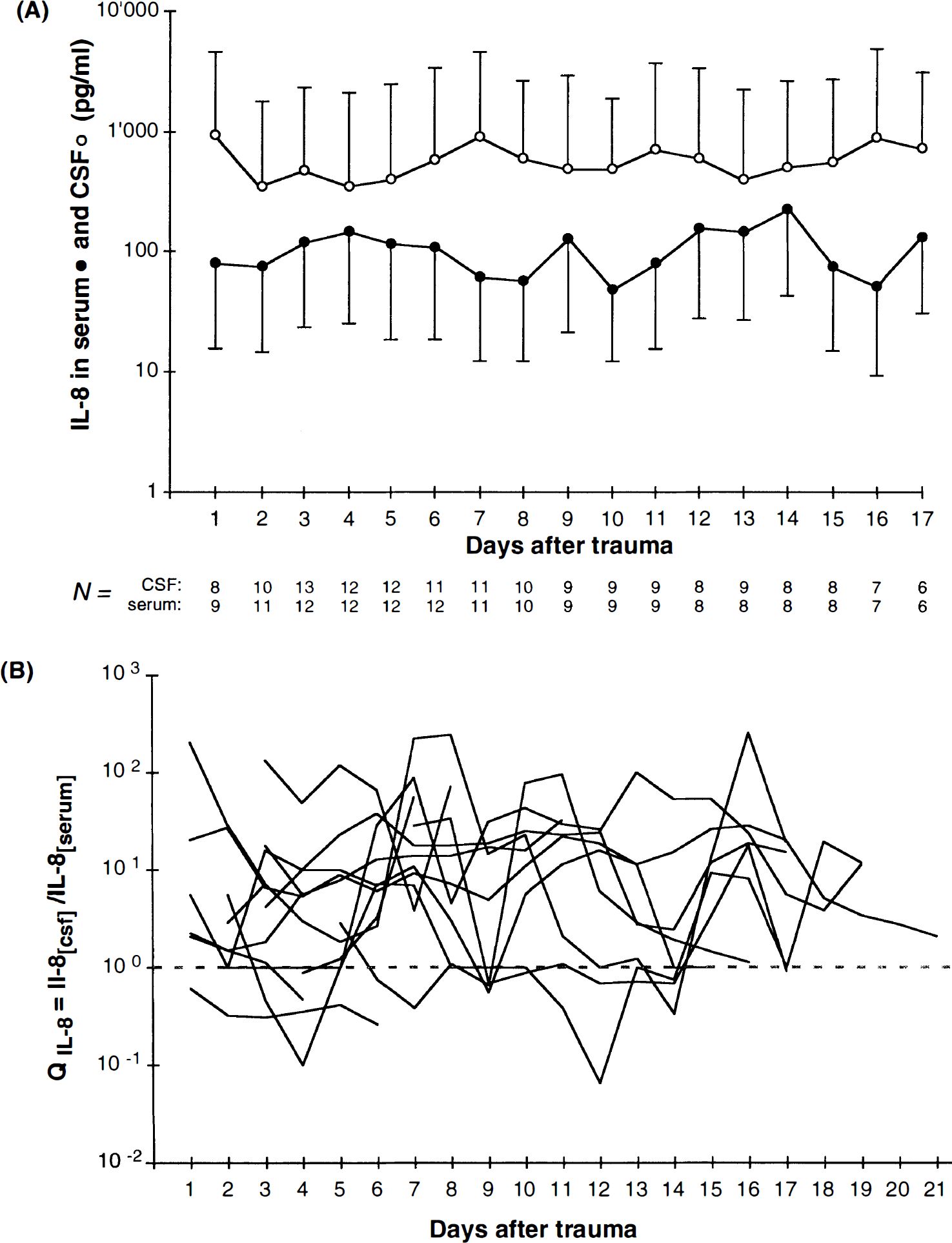
Release of interleukin (IL) 8 into CSF and serum of 14 patients with severe traumatic brain injury (TBI).
No correlation could be found for IL-8 levels in the CSF of the brain-injured patients and clinical parameters such as the severity of the initial injury using the GCS and recovery as assessed by the GOS (GCS: p = 0.945, GOS: p = 0.548; Kruskal–Wallis test, data not shown).
IL-8 associated with disturbance of BBB
Each patient displayed an individual pattern of QA values over the study period. The most severe dysfunction was observed in five patients (QA > 0.02), whereas two had a moderate (QA = 0.01–0.02) and one a mild dysfunction (QA = 0.007–0.01) of the BBB. Five patients had normal QA values (< 0.007), and in one patient (Patient No. 12), no albumin could be measured due to the small amount of CSF sample (Table 1). Interestingly, the individuals with a severe to moderate disturbance of the BBB showed the highest maximal IL-8 levels in CSF, which were between 610 and 8,000 pg/ml (geometric mean = 3,227 pg/ml), whereas the patients with mild or normal BBB had maximal IL-8 values between 260 and 1,670 pg/ml (geometric mean = 790 pg/ml). The overall linear regression analysis of the logarithmically transformed maximal QA and maximal IL-8 showed a significant correlation (r = 0.639, p = 0.019) (Fig. 2). The association of high concentrations of intrathecal IL-8 and augmented permeability of the BBB suggests a potential role for this cytokine in the modulation of the BBB physiology.
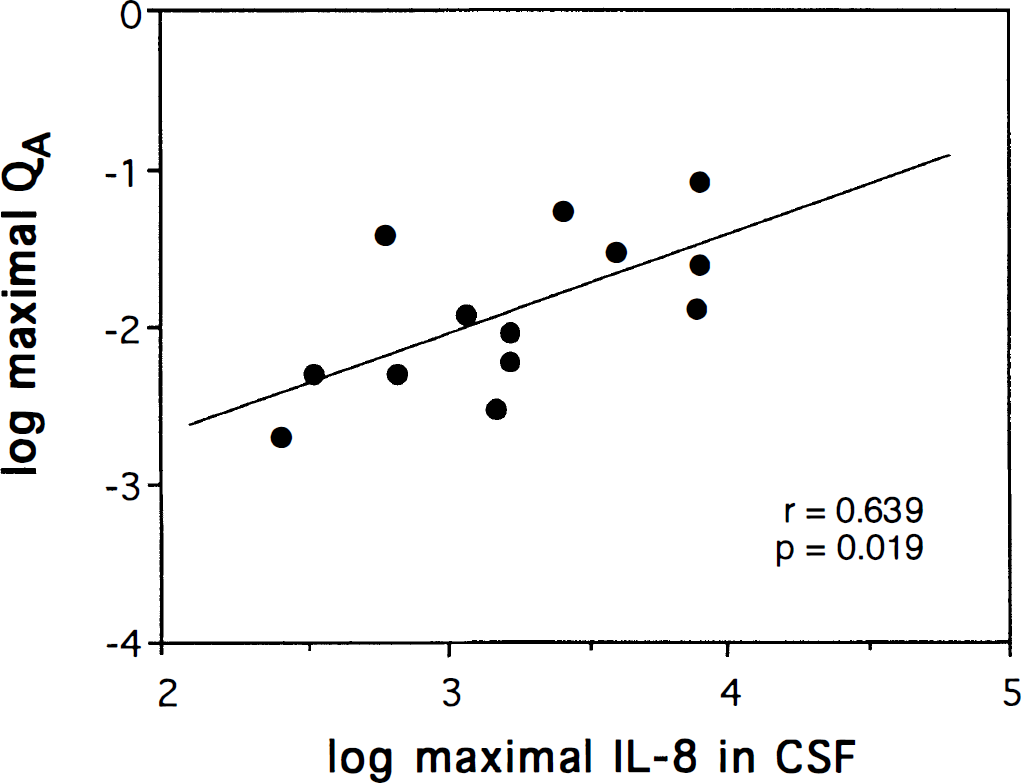
Maximal interleukin (IL) 8 correlates with the dysfunction of the blood–brain barrier. Linear regression analysis on logarithmically transformed maximal IL-8 concentrations in CSF and maximal QA revealed a significant correlation between these two factors as indicated.
IL-8 correlated with NGF secretion into CSF
NGF was present in the CSF of seven of 14 patients (range of maximal values 62–12,130 pg/ml). The NGF peaks were found in most patients within the first 5 days following trauma (n = 5), and in some individuals a second rise of NGF was also observed at later time points. Interestingly, the patients with measurable NGF in CSF had also higher CSF IL-8 levels (3,749 pg/ml geometric mean) over the days compared with the IL-8 levels in the CSF of patients with no detectable NGF (736 pg/ml geometric mean). Figure 3 shows a significant difference (p < 0.01, Mann–Whitney U test) between the maximal values of IL-8 in the group without NGF (NGF–) and the group with detectable levels of NGF (NGF+) in the CSF. Moreover, the highest NGF concentrations were detected in the CSF of those patients who had highest maximal IL-8 levels some time during the study period (not in the same CSF sample). Overall analysis of maximal IL-8 and maximal NGF concentrations in the CSF of all patients revealed a significant correlation between these two factors (Spearman rank correlation; p = 0.698, p = 0.01) (Fig. 3).
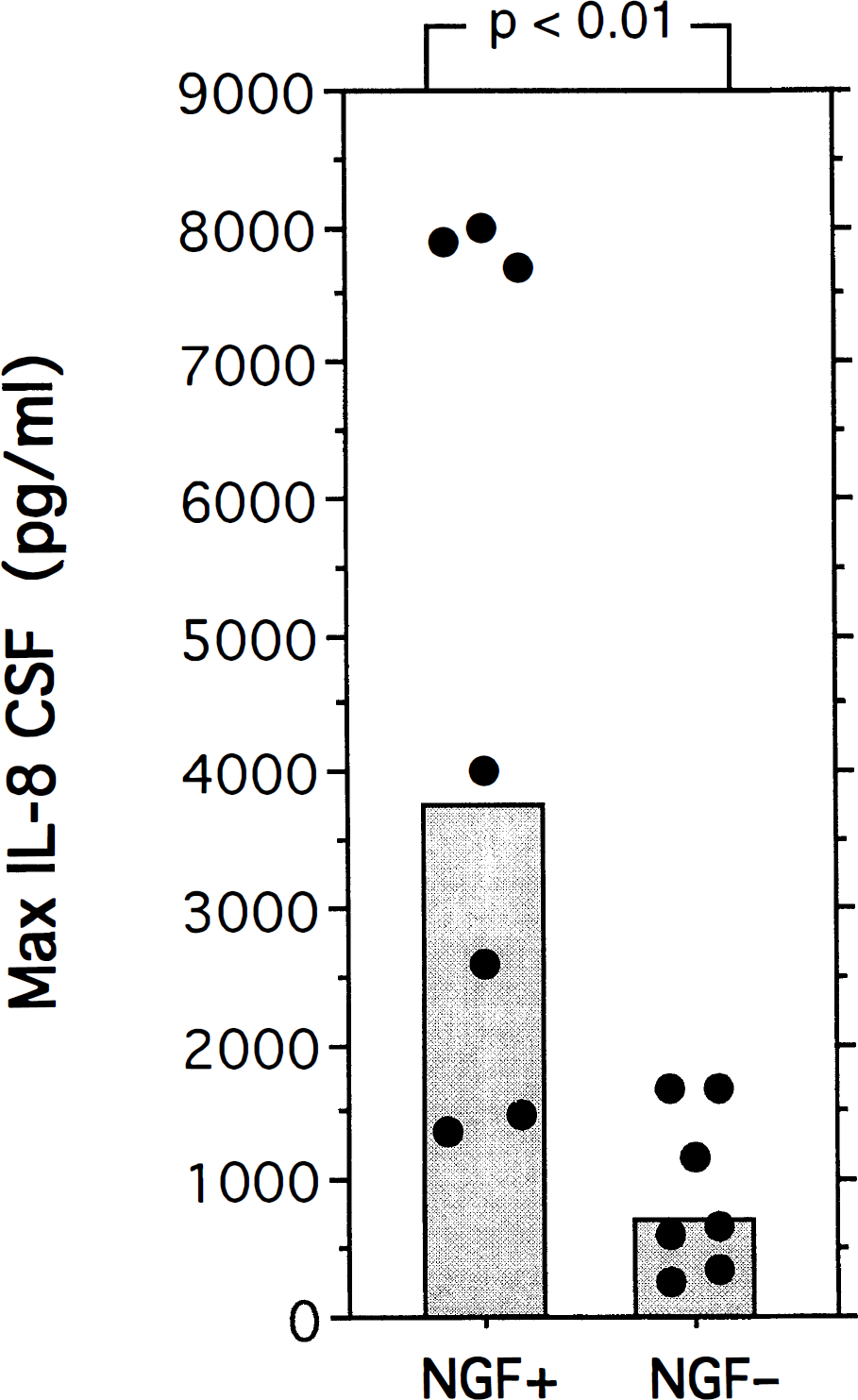
Nerve growth factor (NGF) is released into the CSF of patients with high interleukin (IL) 8. Maximal IL-8 concentrations of the patient group with detectable NGF (NGF+) were significantly higher (p < 0.01, Mann–Whitney U test) than the values in the patient group without NGF (NGF –). Boxes represent the geometric means of maximal IL-8 (NGF+: 3,749 pg/ml; NGF– : 736 pg/ml). Overall analysis of maximal IL-8 and maximal NGF concentrations in the CSF of all patients revealed a significant correlation between these two factors (Spearman rank correlation; r = 0.698, p = 0.01).
Production of NGF induced by Il-8 in astrocytes
To determine whether the production of NGF detected in the CSF of head-injured patients may be induced by IL-8, primary astrocyte cultures were stimulated with CSF samples containing IL-8 for 72 h, and NGF was measured by ELISA in the culture supernatants. Figure 4 shows NGF levels released by astrocytes after incubation with CSF from three patients with severe brain injury containing IL-8 collected at determined time points as described in Material and Methods and in Table 2. For each patient, two different CSF samples were used for stimulation: one with higher and one with lower IL-8 concentration (Table 2). With the exception of one sample (Patient 2, low IL-8), all samples obtained from head-injured patients induced an increased NGF production from 71 to 434 pg/ml in astrocytes compared with control cultures treated with medium alone (37 pg/ml). Stimulation with three control CSF samples collected from a ventriculoperitoneal shunt or diagnostic lumbar puncture, without detectable IL-8 or other cytokines, did not induce a significant NGF production in astrocytes (0–40 pg/ml). Measurement of NGF in the CSF samples prior to the incubation with astrocytes revealed the presence of NGF in four samples (range 9–448 pg/ml) as indicated in Table 2. This preexisting NGF was subtracted from the NGF measured in the culture supernatants as already described in Material and Methods. Since other cytokines that are produced after brain injury have the ability to induce NGF production by astrocytes (Gadient et al., 1990; Awatsuji et al., 1993; Kossmann et al., 1996), the astrocyte cultures were incubated with the same CSF samples after pretreatment with anti-IL-8 antibodies in neutralizing concentrations (1–10 mg/ml). Figure 4 shows NGF levels after incubation of astrocytes with CSF in the presence and absence of anti-IL-8 antibodies. The addition of anti-IL-8 antibodies resulted in a partial inhibition of the NGF production that was more evident in those samples containing more elevated concentrations of IL-8 (range 52–100%), whereas the inhibition obtained with lower IL-8 levels was less pronounced (range 0–52%). Although the release of NGF was increased following stimulation with CSF, the inability of anti-IL-8 antibodies to completely reverse the production of NGF indicates that other factors/cytokines contained in these samples may be involved.
Analysis of cytokines and NGF in human CSF used for astrocyte stimulation
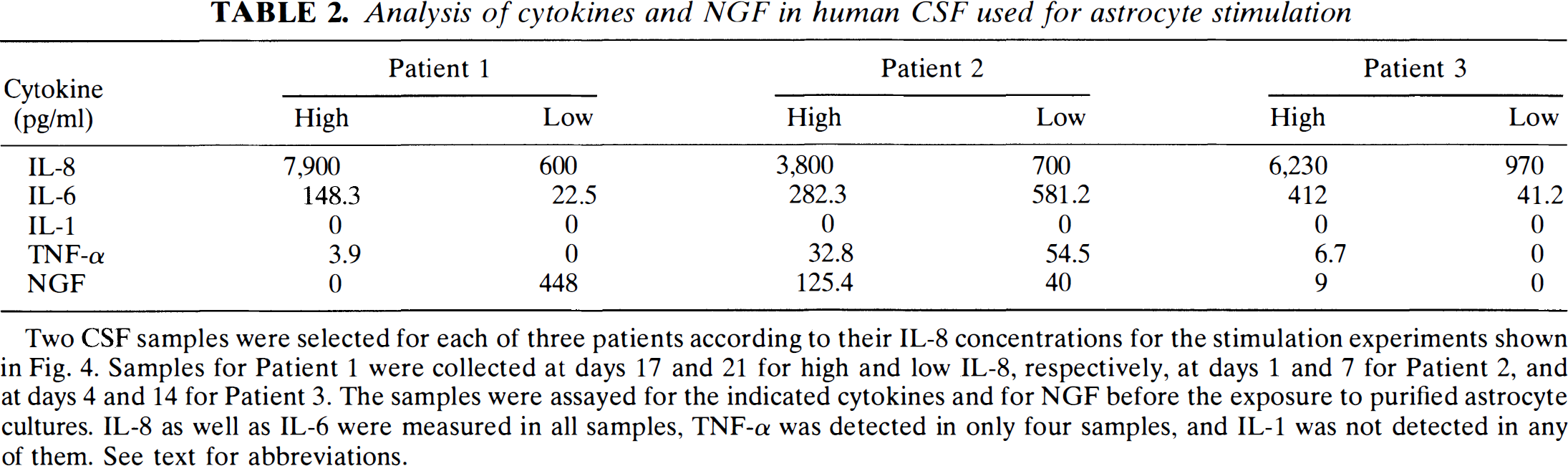
Two CSF samples were selected for each of three patients according to their IL-8 concentrations for the stimulation experiments shown in Fig. 4. Samples for Patient 1 were collected at days 17 and 21 for high and low IL-8, respectively, at days 1 and 7 for Patient 2, and at days 4 and 14 for Patient 3. The samples were assayed for the indicated cytokines and for NGF before the exposure to purified astrocyte cultures. IL-8 as well as IL-6 were measured in all samples, TNF-α was detected in only four samples, and IL-1 was not detected in any of them. See text for abbreviations.
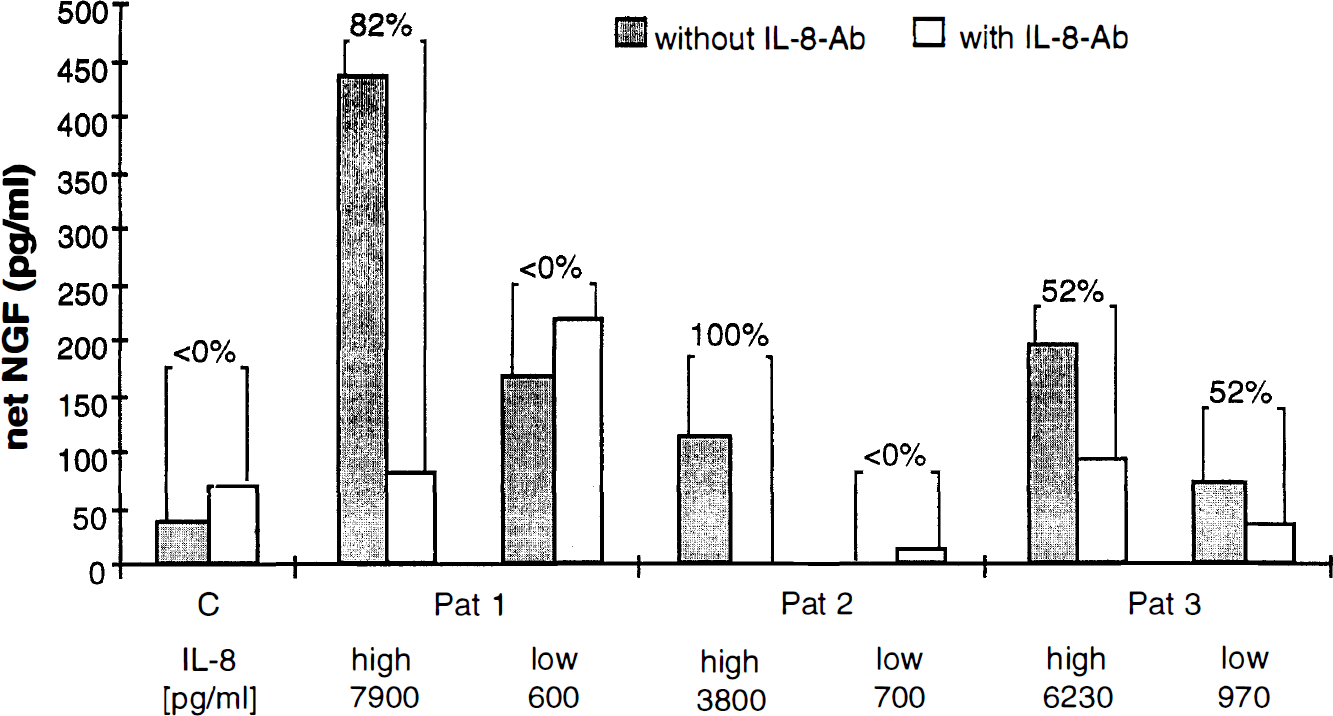
CSF containing interleukin (IL) 8 induces astrocytic nerve growth factor (NGF) production. NGF concentrations in mouse astrocyte supernatants following incubation with CSF samples of three brain-injured patients in the presence and absence of neutralizing anti-IL-8 antibodies. Confluent cell cultures were incubated with two CSF samples for each patient, the first containing a high and the second a low concentration of IL-8 as indicated. To demonstrate the specificity of IL-8 for inducing NGF production, the same CSF samples were incubated with anti-IL-8 antibodies (IL-8-Ab) prior to the exposure to astrocyte cultures. Following incubation for 72 h, the supernatants were collected and analyzed for NGF by enzyme-linked immunosorbent assay as described in Material and Methods. NGF produced by astrocytes was calculated as follows: (astrocyte NGF) = (NGF in culture supernatant) − (NGF present in human CSF).
In previous studies, we have shown that IL-6 contained in the CSF of brain-injured patients, as well as in recombinant form, stimulates NGF in cultured astrocytes (Kossmann et al., 1996). Therefore, the presence of IL-6 as well as of other cytokines such as IL-1 and TNF-α, which also promote NGF production in these cells (Gadient et al., 1990), was measured in the same CSF samples used for the stimulation experiments described (Table 2). IL-1β was not detected in any samples; IL-6 was found in all six samples, and the concentrations ranged from 22.5 to 581.2 pg/ml in the patients' CSF. TNF-α was measured in four samples ranging from 3.9 to 54.5 pg/ml (Table 2). The presence of these cytokines in the CSF provides a possible explanation for the inability of anti-IL-8 antibodies to completely reverse the induction of NGF (Fig. 4).
To confirm that IL-8 promotes the synthesis of NGF, astrocyte cultures were incubated with increasing concentrations of recombinant IL-8 (10–1,000 ng/ml) for 72 h. The supernatants were then collected and assayed for NGF. The concentrations of NGF measured in supernatants of astrocyte cultures stimulated with low IL-8 concentrations (10, 50, and 100 ng/ml) were not significantly higher than control. However, a statistically significant increase in NGF levels was achieved using 500 and 1,000 ng/ml of IL-8 (p < 0.005 and p < 0.002, respectively; unpaired t test) (Fig. 5). These results showed that a much higher concentration of recombinant IL-8 alone is required to induce NGF as compared with the concentrations present in the CSF samples. Astrocytes treated with medium alone produced lower levels of NGF (13 pg/ml mean value). Six experiments were performed.
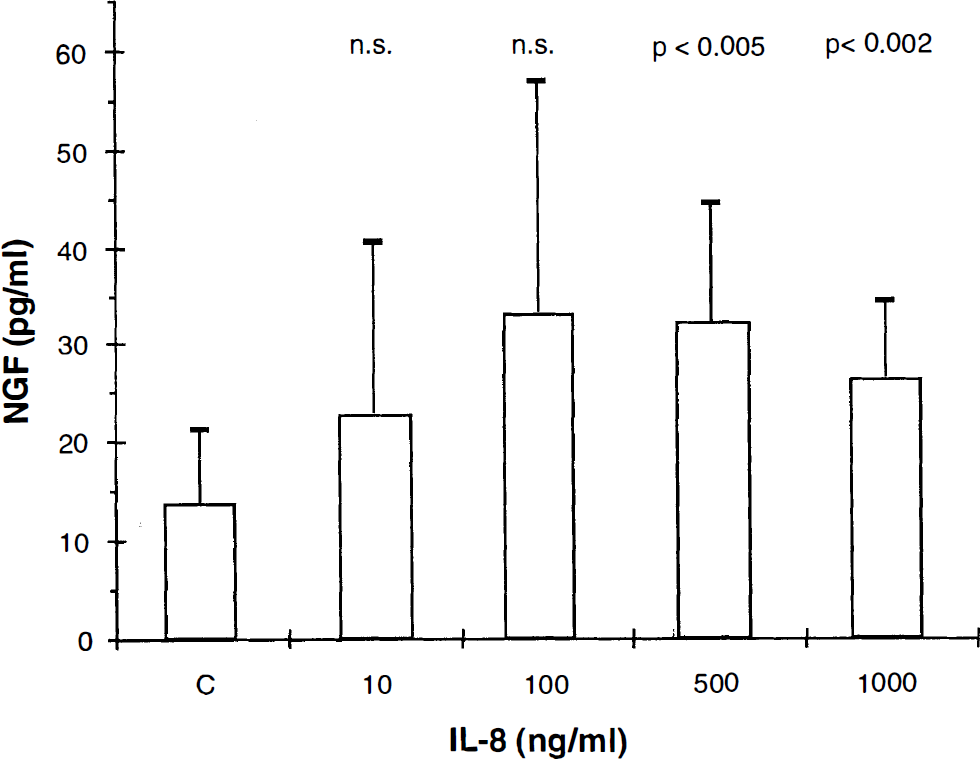
Recombinant interleukin (IL) 8 induces nerve growth factor (NGF) in astrocytes. NGF production after incubation of primary mouse astrocyte cultures with recombinant IL-8 (10–1,000 ng/ml). Prior to stimulation, the astrocyte cultures were washed three times and incubated for 12 h with Dulbecco's Modified Eagle Medium (DMEM) + 1% fetal calf serum (FCS). The cells were then washed again twice and incubated with increasing amounts of recombinant IL-8 for 3 days. As control (C), DMEM + 1% FCS was used. The supernatants were collected and analyzed for NGF by enzyme-linked immunosorbent assay. The figure shows the mean + SD of six separate experiments. Astrocyte cultures produced significantly higher amounts of NGF when stimulated with high doses of IL-8 (500 and 1,000 ng/ml) than control (p < 0.005 and 0.002, respectively, Student t test).
DISCUSSION
IL-8 was measured in CSF and serum of 14 patients with severe head trauma for a time period of up to 21 days. In serum samples, IL-8 was either not detected or found in concentrations up to 256 times lower than in CSF. This suggests that this cytokine may be produced in the cerebral compartment following the traumatic impact as supported by the findings that astrocytes and microglia are capable of producing IL-8 after challenge (Aloisi et al., 1992; Mukaida and Matsushima, 1992; Nitta et al., 1992).
Elevated levels of the pro-inflammatory cytokine IL-8 have been reported in numerous pathological conditions, including diseases of the CNS (Handa, 1992; Nitta et al., 1992; van Meir et al., 1992; Halstensen et al., 1993; Seki et al., 1993; Tada et al., 1993; Mastroianni et al., 1994). IL-8 is produced by numerous cells upon stimulation including glial cells and neutrophils (Bazzoni et al., 1991; Aloisi et al., 1992; Nitta et al., 1992). Cytokines such as TNF-α and IL-1 have the ability to induce the expression and secretion of IL-8 in astrocytes, and the production of TNF-α, IL-1, and IL-6 has been demonstrated in experimental head injury models and in humans very early after the traumatic impact (McClain et al., 1987, 1991; Nieto-Sampedro and Berman, 1987; Woodroofe et al., 1991; Taupin et al., 1993; Kossmann et al., 1995). The lack of detectable IL-1 in our samples may be due to the fact that the first CSF samples were collected only 24 h after the head injury, when IL-1 activity may already have dropped below the detection level of the assay used. In an animal model of shock induced by intravenous administration of endotoxin, the peak of IL-8 in serum was found succeeding the IL-1 and TNF-α peaks and occurring simultaneously with the IL-6 peak (Martich et al., 1991). Therefore, these cytokines immediately released after head trauma may promote the synthesis of later mediators such as IL-8 and IL-6. Both of these cytokines are present in higher concentrations in CSF than serum, suggesting that they may be produced intrathecally under similar regulatory mechanisms (Kossmann et al., 1995).
Our results suggest that IL-8 may display functions additional to the regulation of the immune response, such as the up-regulation of NGF. Increased NGF expression has been described following neurotrauma in animals as well as in humans (Nieto-Sampedro et al., 1982; Heumann et al., 1987; Patterson et al., 1993; Kossmann et al., 1996). NGF has the ability to favor the development and survival of cholinergic neurons (for review see Levi-Montalcini and Angeletti, 1968). Much evidence supports a role for cytokines in the regulation of NGF synthesis (Lindholm et al., 1987, 1990; Gadient et al., 1990; Spranger et al., 1990; Carman-Krzan et al., 1991; Yoshida and Gage, 1991). Enhanced NGF production has been shown following intrathecal injection of IL-1, TNF-α, and transforming growth factor-β (Spranger et al., 1990). This production seems to be predominantly attributed to astrocytes and fibroblasts as a result of experiments performed in vitro (Furukawa et al., 1989; Gadient et al., 1990). We demonstrate for the first time that IL-8 may promote the synthesis of NGF as well, which may have beneficial consequences for the lesioned brain, since the administration of NGF was shown to result in increased neuronal survival (Kromer, 1987; Apfel et al., 1991). However, the observation that high levels of NGF have previously been found to be associated with the death of the patients indicates that NGF production may depend on the degree of the brain damage (Kossmann et al., 1996). In this study, the neurotrophic factor NGF was found to be associated with elevated levels of IL-8 in the CSF. Furthermore, maximal IL-8 levels measured in the patient group with NGF were significantly higher than those detected in the patient group without NGF. Thus, IL-8 may induce NGF production in vivo after brain injury. This hypothesis is also supported by the findings that astrocytes and microglia express the IL-8 receptor, and therefore these cells are likely candidates for the production of NGF following the challenge with IL-8 (Lacy et al., 1995). In addition, IL-8 itself has also been shown to promote the survival of hippocampal neurons (Araujo and Cotman, 1993) possibly through the release of factors by astrocytes that were present in these cultures (Morganti-Kossmann and Kossmann, 1995). It cannot be excluded, however, that the release of these factors is the consequence of the same cause, namely, the injury to the brain, and not the result of a functional relationship. Therefore, to elucidate whether IL-8 induces NGF production in astrocytes, CSF samples of brain-injured patients containing IL-8 were incubated with purified astrocyte cultures derived from newborn mouse brain. NGF production was induced by all samples, except for one containing low levels of IL-8, and the NGF levels appeared to depend on the concentration of IL-8. When CSF samples were pretreated with anti-IL-8 antibodies, a distinct but not complete inhibition of the astrocytic NGF production occurred, indicating that IL-8 may act in association with other factors/cytokines. For this purpose, the CSF samples utilized for the culture experiments were tested for other cytokines. IL-6 and TNF-α were found in different amounts, whereas IL-1 was not detected in any of the three samples. In this regard, we have previously shown that IL-6 secreted in the CSF in elevated concentrations (up to 7,324 pg/ml) has the ability to induce NGF in astrocytes. Therefore, the low amounts of IL-6 present in the CSF samples presently used for astrocyte stimulation may not be sufficient to promote NGF alone, but may be sufficient for a multifactorially regulated mechanism (Gadient et al., 1990; Kossmann et al., 1996). This hypothesis is confirmed by the fact that higher concentrations of recombinant IL-8 alone are necessary for stimulating astrocyte cultures to produce NGF as compared with the IL-8 concentrations present in the CSF used for the challenge. Furthermore, a greater amount of NGF was released by astrocytes stimulated with CSF than with recombinant IL-8 alone, supporting the role played by other factors present in the CSF. The in vivo produced NGF may also derive from other cell types such as fibroblasts, which like astrocytes have the ability to release NGF (Furukawa et al., 1989).
The functional significance of IL-8 production in the lesioned CNS remains to be further elucidated. Although other work showed a correlation between IL-8 levels and clinical status in meningococcal infections of the CNS (Halstensen et al., 1993), in the present study, IL-8 levels did not correlate with clinical status at admission or with the outcome of the patients. This may be due to the fact that the pathophysiology of head trauma differs considerably from the pathology of infectious diseases in the nervous system. We show that IL-8 was found to be associated with a severe dysfunction of the BBB. However, further experimental effort is needed to clarify whether or not the permeability of the BBB is indeed increased by this cytokine. In fact, controversial opinions exist on the ability of cytokines to increase BBB permeability (Banks et al., 1989, 1991; Kim et al., 1992). The passage of cytokines across the BBB has been demonstrated in both directions, blood to brain and brain to blood, through a saturable active mechanism (Banks et al., 1991). Although no data are available on the disruption of the BBB mediated by IL-8, the recruitment of neutrophils, which may be promoted by IL-8, followed by the release of free oxygen radicals and other factors by these cells, may cause vascular damage and increase brain edema (Chan et al., 1984; Baggiolini et al., 1989; Shiga et al., 1991). This is supported by the fact that antibodies to the leukocyte adhesion molecule ICAM-1 reduce the neurological damage in a model of stroke by inhibiting the adhesion of neutrophils to the endothelium (Bowes et al., 1993).
In conclusion, this study showed that IL-8 is released in the CSF and serum of brain-injured patients and that, apart from its pro-inflammatory properties, IL-8 may influence the function of the BBB and regulate the induction of NGF synthesis by astrocytes.
Footnotes
Acknowledgement
This study was supported by the Swiss National Foundation (no. 31-36375.92 and no. 31-42490.94). The authors thank Dr. V. Hans (Department of Surgery, University of Zurich) and Dr. Scott R. Barnum (Department of Microbiology, University of Alabama at Birmingham, AL, U.S.A.) for critically reviewing the manuscript. We are grateful to Mrs. Emerita Ammann and Mrs. Eva Paul for excellent technical assistance and to Miroslav Ceska (Sandoz Research Center Vienna, Austria) for providing the IL-8 reagents.
