Abstract
Endothelial blood-brain barrier (BBB) dysfunction is critical in the pathophysiology of brain injury. Rho-associated protein kinase (ROCK) activation disrupts BBB integrity in the injured brain. We aimed to test the efficacy of a novel ROCK2 inhibitor in preserving the BBB after acute brain injury. We characterized the molecular structure and pharmacodynamic and pharmacokinetic properties of a novel selective ROCK2 inhibitor, NRL-1049, and its first metabolite, 1-hydroxy-NRL-1049 (referred to as NRL-2017 hereon) and tested the efficacy of NRL-1049 on the BBB integrity in rodent models of acute brain injury. Our data show that NRL-1049 and NRL-2017 both inhibit ROCK activity and are 44-fold and 17-fold more selective towards ROCK2 than ROCK1, respectively. When tested in a mouse model of cortical cryoinjury, NRL-1049 significantly attenuated the increase in water content. Interestingly, 60% of the mice in the vehicle arm developed seizures within 2 hours after cryoinjury versus none in the NRL-1049 arm. In spontaneously hypertensive rats, NRL-1049 attenuated the dramatic surge in Evans Blue extravasation compared with the vehicle arm after transient middle cerebral artery occlusion. Hemorrhagic transformation was also reduced. We show that NRL-1049, a selective ROCK2 inhibitor, is a promising drug candidate to preserve the BBB after brain injury.
Introduction
Rho-associated protein kinase (ROCK) activity is a critical determinant of structure and function in virtually all cells, regulating vascular tone, leukocyte chemotaxis, platelet activation, and other processes. ROCK plays a role in the pathophysiology of diverse diseases, including cardiovascular, immune, and neurologic disorders.1 –5 Therefore, ROCK has been an attractive therapeutic target for almost three decades. 6 However, because of its widespread expression and multitude of functions, pharmacologically targeting ROCK in any one system or cell type without unintended side effects has been challenging. Despite intense efforts, systemic treatment with ROCK inhibitors found limited clinical utility around the world, entering the standard of care only in the Far East for short-term treatment after aneurysmal subarachnoid hemorrhage. 7
Most kinase inhibitors have limited selectivity for ROCK over other kinases such as the cyclic AMP- and GMP- dependent protein kinase (PKA and PKG, respectively) and protein kinase C (PKC). Those more selective towards ROCK often do not distinguish between the two ROCK isoforms, ROCK1 and ROCK2, with differential expression and roles in different cell types.8 –14 ROCK1 is predominantly expressed in the lung, liver, testes, blood, and immune system, whereas ROCK2 is more abundant in the brain, heart, smooth muscle cells, and vascular endothelium.8,15,16 Therefore, recent drug development programs have focused on the generation of isoform-selective ROCK inhibitors for more selective and targeted therapy to minimize potential side effects.6,17
ROCK inhibition has been uniformly efficacious in experimental brain injury.4,5,18 –20 Although most studies used isoform non-selective inhibitors such as fasudil, data suggest that ROCK2-selective inhibitors may be equally efficacious.21,22 ROCK is also an important regulator of the blood-brain barrier (BBB) in cerebral endothelial cells.23 –27 Upregulation of ROCK activity increases BBB permeability with potentially deleterious consequences ranging from brain edema to hemorrhage. Indeed, ROCK inhibition has been efficacious in preserving the BBB after various insults both in vitro and in vivo,22,28,29 and ROCK2 isoform appears to be the relevant target.22,30 –33
The novel small molecule ROCK2-selective inhibitor, BA-1049 (now listed as NRL-1049), 34 has recently been shown to reduce lesion volume and hemorrhagic transformation in a mouse model of cavernous angiomas, 35 and has a favorable profile for clinical development. Here, we report on the pharmacology of NRL-1049 and its efficacy in preserving the BBB in animal models of acute brain injury.
Materials and methods
Cell cultures and treatment
At this proof-of-concept stage, data were obtained from two independent vascular beds (brain microvascular and umbilical cord) since they have differential ROCK isoform expression (ROCK2 being more abundant in the cerebral vasculature) to corroborate findings in one cell line with the other for robust conclusions.
Human brain microvascular endothelial cells (hBMVECs, P7, Lot. 1796655; Cell Systems, Kirkland, WA, USA) were cultured in EGM-2 medium (Cat# CC-3202; Lonza, Cambridge, MA, USA) for 3 days. Thereafter cells were exposed to starvation medium (DMEM-F12 supplemented with 0.1% Human Serum Albumin (HSA)) for 2 hours. At 1 hour, NRL-1049 or it’s metabolite (NRL-2017) (0–1000 μM) was added to the starvation medium. At 2 hours, Lysophosphatidic acid (LPA, 40 µM) was added for 5 minutes where after cell cultures were harvested, lysated (using 4× Laemmli buffer with b-mercaptoethanol (9:1)), boiled for 5 minutes at 95 °C and stored at −80°C until further analyses. Three experiments, each carried out in triplicate were averaged to give a 50% effective concentration (EC50).
Human umbilical cord vein endothelial cells (HUVEC, Cat# C-003-5C; Gibco™ Cascade Biologics, Thermo Fisher Scientific, Waltham, MA, USA) were cultured for 3 days in medium 200 (Cat# M-200-500; Life technologies, Thermo Fisher Scientific, Waltham, MA, USA) with Large vessel endothelial supplement (LVES, Cat# A14608-01, Life technologies), whereafter cells were treated with NRL-1049, it’s metabolite (NRL-2017) (10 µM) or vehicle for 1 hour. Thereafter, each hour (for up to 4 hours), cells were activated by LPA (20 µM) for 5 minutes and harvested afterwards as described above. A n = 3 in triplicate for each drug and time point was performed.
Compounds were incubated in duplicate with cryopreserved hepatocytes (mouse, rat, dog, cynomolgus monkey and human, Cyprotex, Watertown, MA, USA) at 37 °C. Cells were thawed, viable cells counted, and equilibrated according to the supplier’s directions. After 30 min equilibration at 37 °C with gentle agitation, the test compound was added to the cells to give the desired final concentration of 1 μM. The cell suspension was incubated at 37 °C. At the indicated times, samples were removed and mixed with an equal volume of ice-cold stop solution (methanol containing the internal analytical standard, propranolol). Stopped reactions were incubated for at least ten minutes on ice, and an additional volume of water was added. The samples were centrifuged to remove precipitated protein, and the supernatants were analyzed by LC-MS/MS to quantitate the remaining parent.
Liquid chromatography mass spectrometry (LC-MS/MS)
Liquid Chromatography/Mass Spectrometry (LC-MS/MS) was developed and validated by MPI research (Kalamazoo, MI, USA; now Charles River) to analyze drug concentration in plasma. In order to determine the amounts of specific analytes in plasma or homogenized tissue, three volumes of methanol were added to individual samples containing an internal analytical standard (propranolol). Following centrifugation to remove precipitated protein, supernatants were analyzed by LC-MS/MS. 25 μL of each standard or study sample was mixed with 200 μL of working internal standard solution (10 ng/mL in acetonitrile/methanol (50/50, v/v)). The sample was vortexed and centrifuged. A 100 μL aliquot of the resulting supernatant was mixed with 300 μL of water/formic acid (100/0.1, v/v) and an aliquot was injected onto an LC-MS/MS system for analysis. Samples were run on an ACE 5 C18 column (5 μm particle size) with a gradient flow consisting of water/formic acid (100/0.1, v/v) and acetonitrile/formic acid (100/0.1, v/v) at a flow rate of 750 μL/minute. The analyte (NRL-1049), metabolite NRL-2017, and internal standard (propranolol) were detected using a SCIEX API 5000 triple quadrupole LC-MS/MS system equipped with an ESI (TurboIonSpray®) ionization source operated in the positive ion mode. Data were converted to % remaining by dividing by the time zero concentration value. All data were fitted to a first-order decay model to determine half-life.
In vitro kinase specificity
Kinase screening was carried out by DiscoverX (Fremont, CA, USA) as described in their protocol. Briefly, the assay utilizes a competition binding assay for any given compound of interest to compete with binding by a DNA-tagged kinase to an immobilized ligand on streptavidin-coated magnetic beads. Binding reactions were with the ligand beads, the DNA-tagged kinases, and test compounds (NRL-1049 or NRL-2017). After 1 hour reaction the beads were washed and bound kinase eluted and measured by qPCR. Binding reactions are expressed as percent of control.
Rho-associate protein kinase (ROCK) activity assay
The ability of NRL-1049 and both of its identified metabolites to inhibit ROCK was measured using the colorimetric Rho-associated Kinase Activity Assay according to the manufacturer’s instructions (Cat# CSA001; EMD-Millipore, Merck, Burlington, MA, USA). Comparisons were made over an 11-point semi-log dilution curve of test inhibitors (in triplicate wells) with Y-27632 serving as a positive control. The multi-well plate assay is based on the phosphorylation of an immobilized Rho kinase substrate (MYPT1) by purified, recombinant ROCK2 and using an antibody based method for detecting the change in the abundance of phospho-MYPT1 (Thr696) after incubation of the reaction at 30°C in the presence or absence of inhibitors, followed by colorimetric development.
Compound IC50 determination versus ROCK1 and 2
Inhibition dose response curves for determining the IC50 values of each compound were generated by radiometric filter binding assay using the Kinase Profiler service provided by Eurofins (Dundee, Scotland). Inhibition by specific compounds of purified, recombinant ROCK1 and ROCK2 was assayed in the presence of radioactive ATP at the Km for each enzyme. Ten-point IC50 curves were generated for each compound.
Western blot
Samples were centrifuged before loading. Samples were separated using SDS-PAGE and transferred to PVDF membrane for immune-blot analysis. Blots were blocked for 60 minutes at room temperature in TBS-Tween (0.1%) blocking buffer (Cat# IBB-592, Boston Bioproducts, MA, USA) with 4% Bovine serum albumin (BSA, Cat# BP9703-100, Thermo Fisher Scientific). Subsequently, blots were incubated overnight at 4 °C in blocking buffer with primary antibodies against: phosphorylated Myosin Light Chain 2 (pMLC2 S19/T18, 1:1000, Cat# 3674S, Cell Signaling Technology Inc., Danvers, MA, USA); ROCK1 (rabbit mAB, 1:1000, Cat# 4035, Cell Signaling Technology Inc.); ROCK2 (rabbit mAB, 1:10,000, Cat#ab125025; Abcam, Waltham, MA, USA) and Anti-GAPDH antibody (Cat# SC-365062, Santa Cruz Biotechnology Inc., Dallas, TX, USA), were used for sample normalization purposes. Primary antibodies were visualized by incubation with anti-mouse (1:10.000) and -rabbit (1:5000) IgG, HRP-linked antibody (Cell Signaling Technology Inc., Danvers, MA, USA) for 60 minutes at room temperature. Membranes were scanned using the FluoroChem SP Imaging System (Alpha Innotech Corp., Santa Clara, CA, USA). Bands were analyzed and quantified using ImageJ, and the results were normalized to the GAPDH fraction. Original Western blot images can be found in Supplemental Figure 1.
Experimental animals
All animal procedures were approved by the Institutional Animal Care and Use Committee and carried out in accordance with the Guide for Care and Use of Laboratory Animals (NIH Publication No. 85-23, 1996). Procedures were approved by the institutional review boards at the Massachusetts General Hospital (focal cerebral ischemia experiments) and the Tufts Division of Laboratory Animal Medicine (cryoinjury experiments). The cryoinjury studies were carried out in the pay-for-use space in the Department of Laboratory Animal Medicine at Tufts University School of Medicine, overseen by their veterinary medicine staff, reviewed and approved through the Tufts IACUC. Experiments are reported in compliance with the ARRIVE guidelines. All experiments were randomized and blinded to the identity of the treatment until after analysis was completed. All animals used for survival experiments received preoperative subcutaneous buprenorphine-SR for pain management. Test solutions were made up and test article identities were masked by a scientist not involved in the stroke experiments. Spontaneously hypertensive rats (SHR, male, 170–300 gram) or C57Bl/6 mice (male and female, 18–22 gram) were used.
Focal cerebral ischemia
Middle cerebral artery occlusion (MCAO) was performed to induce focal ischemia under isoflurane anesthesia (3–4% induction, 1.5–2% maintenance in 70% N2O and 30% O2). Via a midline incision in the neck of the rat (in supine position), the thoracic muscles were gently separated and retracted to the side to reveal the left common carotid artery (CCA) and its bifurcation into the internal and external carotid artery. All three arteries were carefully tied using a suture, whereafter a nylon monofilament (4041910Re, Doccol Corporation, Sharon, MA, USA) was inserted into the external carotid artery and advanced via the internal carotid artery into the Circle of Willis, to block the middle cerebral artery (MCA) at its origin. Reperfusion was allowed by removal of the filament after 90 minutes. During the occlusion period, animals woke up in their own cage, supplemented with a heating lamp to prevent hypothermia. The sham procedure included the same surgical steps and filament insertion halfway without blocking the MCA. To minimize stress during the awake period, we did not measure rectal temperatures while awake. However, no hypothermia was seen upon re-anesthetization for MCA reperfusion, and rectal temperature monitoring was resumed. MCA occlusion was confirmed by laser Doppler flowmetry (LDF) measurement of cerebral blood flow in the MCA territory and by neurological deficits (after reperfusion) indicating successful ischemia. The LDF fiber probe was placed onto the thinned skull after removal of the temporalis muscle. Rectal temperature was maintained around 37 °C using a thermostatic heating pad (FHC, Bowdoinham, ME, USA). NRL-1049 (0.1–20 mg/kg, free-base weight) or vehicle ip injection was given 15 minutes post reperfusion. Animals that died before the readout time point of 9 hours (n = 6; n = 3 in treatment group), with non-optimal perfused brains for Evans Blue (EB) quantification (n = 9; n = 5 in treatment group), or due to technical (measurement) problems (n = 5; n = 3 in treatment group) were excluded from the analysis.
Blood-brain barrier disruption
In rats, EB (2%, 3 ml/kg, 60 mg/kg) was administered via the tail vein at 6 hours post-MCAO. Three hours later, the animal was killed and circulating EB was washed via intra-cardiac saline perfusion. Ipsilesional and contralesional MCA territories were dissected, to reduce the effect of infarct size variation on the outcome. Cerebellum and liver tissue were used as negative and positive control, respectively. Tissue sections were stored in −80°C until EB analysis. The occurrence of intracerebral hemorrhages was examined at the single coronal sectioning line, posterior (−4 mm) from bregma. Hemorrhagic grading was performed using a previously described grading scale: 0 = no hemorrhage; 1 = single small hemorrhage and 2 = a single large or multiple small hemorrhages. 19
For the EB analyses a standard protocol was used. 22 In brief, tissue samples were thawed in 5 mL tubes, and homogenized in 7 tissue volumes of 50% trichloroacetic acid using sonication (2 minutes) on ice. Samples were centrifuged at 3000 G for 20 minutes where after supernatant was collected and diluted in ethanol 4 times. Fluorescence intensity was measured by a microplate fluorescence reader (620 nm excitation, 680 nm emission; Wallac, Perkin Elmer, Wellesley, MA, USA). Hemisphere samples were measured in quadruple, cerebellum in duplicate. A standard titration curve was added and measured for each measurement as a reference. EB concentrations (ng/mg) for each sample was calculated using the mean of all standard reference curves.
Cryoinjury model
In mice, a 3 mm copper rod, equilibrated in liquid nitrogen, was placed on the exposed skull over the somatosensory cortex with 100 g of pressure for 30 seconds. Sham animals had the identical procedure, except the rod was at room temperature. Immediately following injury, there was a loss of cells on the dorsal surface of the brain, followed by an expanding area of neuronal death over 24 hours. 17 We confirmed the presence of a lesion area by Nissl staining 24 hours after injury (n = 6; not shown). Following cryoinjury, animals were treated with a single ip injection of either 10 mg/kg NRL-1049 or vehicle. In the 24 hour readout group, mice additionally received 10 µg/h NRL-1049 (or vehicle) via a subcutaneous osmotic pump (ALZET® Osmotic Pumps, Cupertino, CA, USA). The pump treatments were prepared aseptically. For pump loading, NRL-1049 or vehicle was dissolved in an appropriate volume of sterile PBS to reach the dose of 10 µg/hour. Following recovery from anesthesia, all animals were continuously observed by the scientific staff, and behavioral alterations were assessed once they had returned to sternal recumbence. Seizure activity was defined as loss of postural control, myoclonus, rapid ambulation, barrel rolling (≥3 turns), tonic limb extension or flexion, and tail erection, as previously described.37,38 To analyze edema formation, animals were sacrificed at 1 or 24 hours post-injury, and brain tissue water content (TWC) was analyzed by heat-drying the individual hemispheres.
In a second experiment, blood-brain barrier disruption was investigated using EB dye. Here we aimed to test an alternative route that may be translationally more appropriate for chronic use. Therefore, NRL-1049 (20 mg/kg) or vehicle was administered intraperitoneally immediately after cryoinjury, and then continued administration of NRL-1049 or vehicle in drinking water to deliver 100 mg/kg/day (assuming normal water intake, as recently shown in cavernous malformations 35 ), acknowledging that water intake may be reduced after this procedure. EB (2%, 100 µL) was administered via the tail vein at 6 hours after cryoinjury. The circulation time can be varied from one model to another based on the anticipated magnitude and timeline of BBB opening and the mechanism being targeted to block it to get the best signal-to-noise ratio. Since the timing of BBB opening is unknown in our cryoinjury model, we kept the circulation time longer (18 hours), compared with the MCAO model. But regardless of the circulation time, the control (i.e., vehicle) group has the same latency, and therefore, the leakage can be compared to the treatment arm the same way.
Brains were harvested at 24 hours after cardiac perfusion was performed. After dissecting the brain, a 3 mm biopsy punch of the cortex, centered directly over the lesion or the same area of the contralateral side, was analyzed for EB content using the same protocol as described above.
In both experiments, animals were euthanized before reaching the experimental endpoint when showing excessive seizure activity or other behavior leading to the humane endpoint upon the veterinarian’s directions.
Statistics
All statistics were carried out using Graphpad Prism (version 9.2.0). All individual datasets passed the test for normal distribution (D’Agostino & Pearson and/or Shapiro-Wilk tests) except for the NRL-1049 group in the MCAO EB experiment. Pilot data were present for in vitro experiments and used for sample size determination, and no exclusions were made. For the in vivo experiments, in the absence of prior experience with the models and species used, sample sizes were calculated empirically per model to achieve 80% power with α = 0.05, to detect a 10, 30 and 20% effect size based on an assumed standard deviation of 10, 20 and 20% for the cryoinjury TWC, cryoinjury EB, and the MCAO EB model, respectively. In the cryoinjury model in mice, 60% of animals developed severe convulsive seizures and thus had to be euthanized per institutional and national guidelines. Therefore, only 4 animals could be included in the tissue water content endpoint. Data are plotted as individual data points including mean and standard deviation. Graphs show animal numbers or mean ± SD. Biological half-life values were calculated using the freely available PK-solver software. 39
Results
Structure and activity of NRL-1049 and its metabolites in vitro
NRL-1049 is a piperidine derivative containing an isoquinoline group. It has an additional tail, including a chiral center with associated methyl and amino groups (Figure 1(a), left), which makes it a unique molecular structure compared to existing ROCK inhibitors.17,40
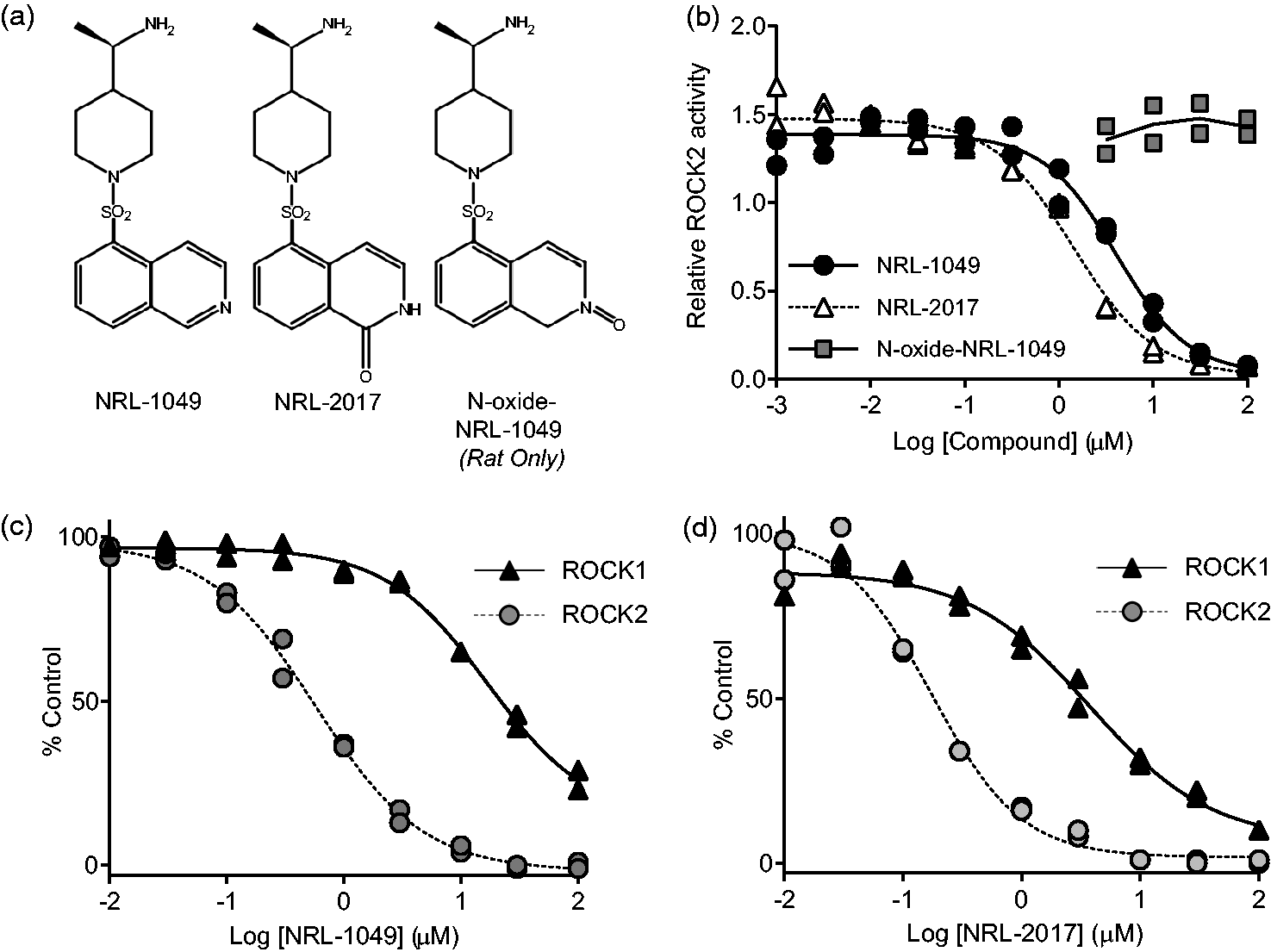
Structure and activity of NRL-1049 and metabolites in vitro. (a) Structures of NRL-1049, the 1-hydroxy-NRL-1049 (from here on referred to as NRL-2017) metabolite generated in rat, mouse, cynomolgus monkey and human hepatocytes in vitro, and the second metabolite generated solely in rat hepatocytes (N-oxide-NRL-1049). (b) Inhibition of ROCK2 activity by NRL-1049 and both metabolites in an in vitro activity assay. N-oxide-NRL-1049 did not inhibit ROCK2 (n = 2 in each group). (c and d) Filter-binding radiometric assay to calculate the IC50 (half-maximal inhibitory concentrations) of NRL-1049 and NRL-2017 against ROCK1 and ROCK2 at each kinases’ Km for ATP (n = 2 in each group).
To determine the number and identities of metabolites produced in the mouse, rat, dog, cynomolgus monkey and human, we performed an in vitro assay in which existing cell lines of hepatocytes from each species were incubated with NRL-1049, and the resulting metabolites were analyzed by LC/MS. We found the presence of a single major hydroxylated metabolite (1-hydroxy-NRL-1049; NRL-2017) after incubation with mouse, cynomolgus monkey, and human hepatocytes (Figure 1(a), center). Only rat hepatocytes produced an additional metabolite (N-oxide-NRL-1049) (Figure 1(a), right), whereas dog hepatocytes did not form any detectable metabolite, in vitro.
To better understand the efficacy of the parent compound and its metabolites, we synthesized both metabolites (1-hydroxy-NRL-1049, referred to as NRL-2017 hereon, and N-oxide-NRL-1049) and compared their concentration-dependent inhibitory effects to those of the parent compound in an in vitro activity assay using phosphorylation of MYPT1 by purified ROCK2 as a readout (Figure 1(b)). We found NRL-2017 at least as potent as the parent compound, whereas the N-oxide-NRL-1049 metabolite detected only in rats was not active against ROCK2 in this assay. Moreover, N-oxide-NRL-1049 also lacked activity on the phosphorylation of downstream ROCK targets in cell cultures (phospho-myosin light chain or phospho-cofilin; data not shown). Therefore, N-oxide-NRL-1049 was not further investigated.
We next examined ROCK2 vs ROCK1 isoform selectivity of NRL-1049 and NRL-2017 using a filter-binding radiometric assay performed at each kinase’s respective Km for ATP. Both NRL-1049 (Figure 1(c)) and NRL-2017 (Figure 1(d)) achieved substantially more inhibition of ROCK2 than ROCK1. NRL-1049’s half-maximal inhibitory concentration (IC50) was 43-fold lower for ROCK2 vs ROCK1 (0.59 µM and 26 µM, respectively), while NRL-2017 IC50 was 17-fold lower for ROCK2 vs ROCK 1 (0.18 µM and 3 µM, respectively).
We also tested the selectivity of NRL-1049 and NRL-2017 (10 µM each) using a thermodynamic kinase screening assay against a panel of 468 human protein kinases (Kinome Scan, DiscoverX- Fremont, CA, USA). Thermodynamic binding characteristics against the kinase targets versus control molecules allowed the determination of inhibitory potential. NRL-1049 inhibited ten kinases at a predicted greater than 95% level, including ROCK1 and ROCK2 (Bike, CLK1, CLK4, HASPIN, PRKCE, RIOK1, ROIK3, ROCK1, ROCK2, RPS6KA5). NRL-2017 inhibited seven kinases at greater than 95%, including ROCK1 and ROCK2 (CLK1, DMK, GRK1, PRKCE, PRKD2, ROCK1, ROCK2).
NRL-1049 inhibits ROCK activation in endothelial cells
Lysophosphatidic acid (LPA) rapidly activates ROCK. 36 ROCK2 is the predominant isoform expressed in human brain microvascular endothelial cells (hBMVEC).32,41 We measured the induction of myosin light chain 2 (MLC2) phosphorylation as a biomarker for LPA-induced ROCK activity in hBMVEC grown to confluence (Figure 2(a)). 42 Incubation with either NRL-1049 or NRL-2017 at 0–1000 µM for one hour concentration-dependently inhibited MLC2 phosphorylation (pMLC2). The 50% effective concentrations (EC50) were 26.3 µM for NRL-1049 (n = 4) and 0.95 µM for NRL-2017 (n = 3).
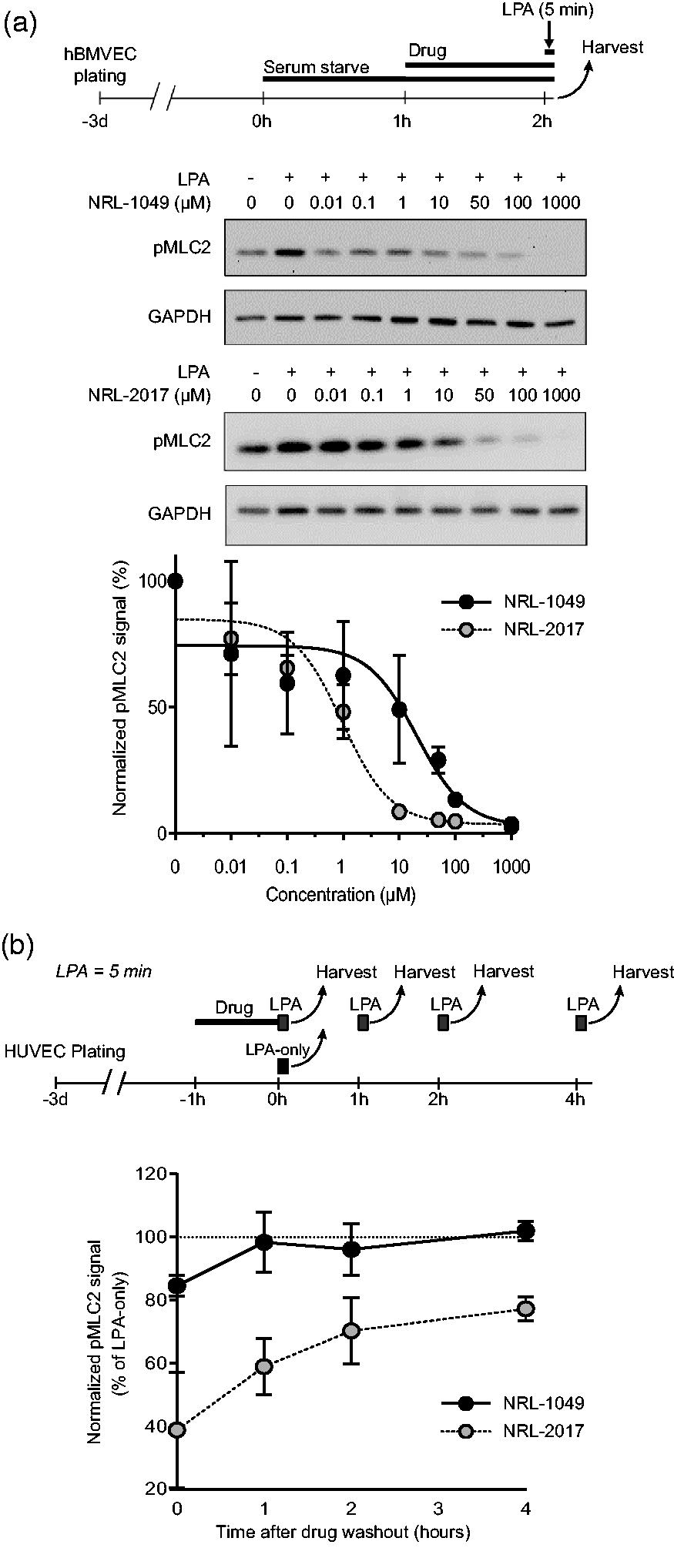
Dose-response and treatment duration to reverse ROCK activation in vitro. (a) Experimental timeline and representative Western blots with corresponding dose-response curves showing phosphorylated MLC2 (pMLC2) levels in hBMVECs after serum starvation for 2 hours and incubation with NRL-1049 or NRL-2017 for 1 hour at increasing concentrations (0–1000 µM) followed by activation with lysophosphatidic acid (LPA) for 5 minutes (n = 4 and 3, respectively). Data are mean ± SD (b) Experimental timeline and activity duration of both compounds, tested by pretreating HUVECs with NRL-1049 or NRL-2017 for one hour followed by 5 minutes of LPA stimulation and immediate cell extraction at 0, 1, 2, or 4 hours after drug washout. At time point 0, NRL-1049, and to a greater extent NRL-2017, reduced pMLC2 levels compared with LPA-only controls (n = 3 in all groups; 15% (p = 0.043) and 61% (p < 0.0001), respectively; one-way ANOVA). The effect of NRL-2017 persisted for at least 4 hours after the washout.
To determine the duration of biological action, we incubated HUVEC, which expresses both ROCK1 and ROCK2, with NRL-1049 (50 µM) or NRL-2017 (50 µM) for one hour, followed by washout for 0, 1, 2, or 4 hours in separate cultures. We then exposed the cells to LPA for 5 minutes at the end of each washout duration and harvested the cells immediately thereafter to measure pMLC2 (n = 3 each; Figure 2(b)). Before washout (i.e., time 0), NRL-1049, and to a greater extent NRL-2017, reduced pMLC2 levels compared with LPA-only controls (15% (p = 0.043) and 61% (p < 0.0001; Chi-square (Mantel-Cox) test), respectively). The inhibitory effect of NRL-1049 on ROCK activity was reversed after only 1 hour of washout. In contrast, the effect of NRL-2017 persisted for at least 4 hours after the washout (p < 0.001 NRL-1049 vs. NRL-2017; two-way ANOVA). These results suggested that the metabolite NRL-2017 is responsible for most of the drug activity in vivo.
Pharmacokinetics of NRL-1049
Next, we studied the pharmacokinetics (PK) of NRL-1049 after intraperitoneal administration in the mouse and rat, since pharmacokinetics in the mouse after PO administration has already been described. 35 In mice, plasma levels of both the parent compound and the metabolite NRL-2017 slowly decreased over time after a single 10 mg/kg ip dose of NRL-1049. They were still detectable for up to 4 hours for NRL-1049 and up to 8 hours for the metabolite (Figure 3(a)). In rats, plasma levels of both the parent compound and the metabolite were still detectable hours after a single ip dose of NRL-1049 (10 mg/kg). Plasma levels of the metabolite were higher than the parent compound and were detectable for hours after administration (Figure 3(b)). Based on these, the T1/2 of NRL-1049 was 0.33 hours (λz = 2.11) in mice after a 10 mg/kg ip dose and 0.89 (λz = 0.78) in rats after a 10 mg/kg ip dose. In PK analyses, λz is the terminal phase associated elimination rate constant. These data also showed that rats metabolize and eliminate NRL-1049 and its metabolite more slowly than mice. 35
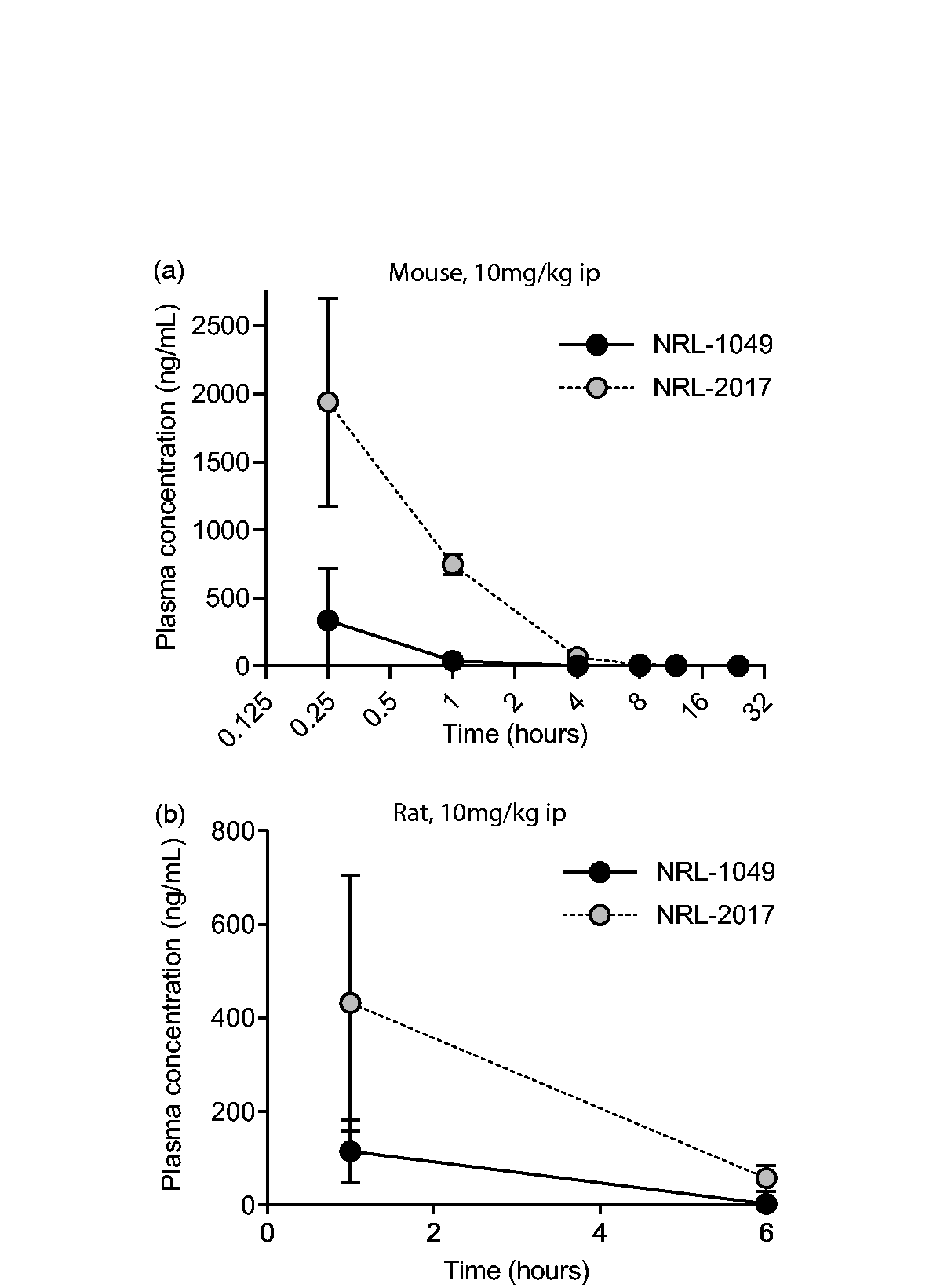
Pharmacokinetic analyses of NRL-1049 and metabolite NRL-2017. (a) Mouse plasma levels of a single 10 mg/kg dose of NRL-1049 via ip injection (n = 4, both groups) and (b) Rat plasma levels after a 10 mg/kg intraperitoneal dose of NRL-1049 (n = 4, both groups). All drug concentrations are measured in duplicates and expressed in free-base weight. Data are mean ± SD. Please note, due to small SDs some error bars are not visible in the graphs.
NRL-1049 preserves the BBB and suppresses seizures after brain injury
We next examined the efficacy of NRL-1049 on acute cortical cryoinjury in mice (Figure 4), a widely used model to study BBB disruption, vasogenic edema, and seizures. 43 We first showed elevated perilesional phospho-cofilin levels in the cortex as a marker of ROCK activation 44 compared with the contralesional homotopic tissue as early as 1 hour after cryoinjury (Western blot, n = 2 mice; Figure 4(a)). We then tested the effect of NRL-1049 (20 mg/kg ip bolus followed by 10 µg/h via osmotic pump) when administered starting immediately after cryoinjury. Strikingly, 60% of vehicle-treated animals in the 24-hour group developed generalized seizures requiring euthanasia, compared with none in the NRL-1049 arm (p = 0.004; Mantel-Cox survival analysis, Figure 4(b)). Ipsilesional brain water content progressively increased at 1 and 24 hours after injury compared with the contralesional hemisphere (Figure 4(b)). NRL-1049 arm showed significantly lower brain water content compared with vehicle controls at 24 hours (p = 0.003) and a strong trend as early as 1 hour (p = 0.08) after cryoinjury (two-way ANOVA followed by Šídák’s multiple comparisons). We also examined the effect of NRL-1049 on large molecular leakage across the BBB using EB extravasation assay (Figure 4(c)). At 24 hours, there was strong EB extravasation compared with sham controls in the vehicle arm (p < 0.001; two-way ANOVA followed by Šídák’s multiple comparisons). NRL-1049 diminished EB leakage, which did not statistically differ from the sham group (p = 0.188; two-way ANOVA followed by Šídák’s multiple comparisons). Altogether, these data demonstrated the efficacy of NRL-1049 on BBB protection after acute brain injury.
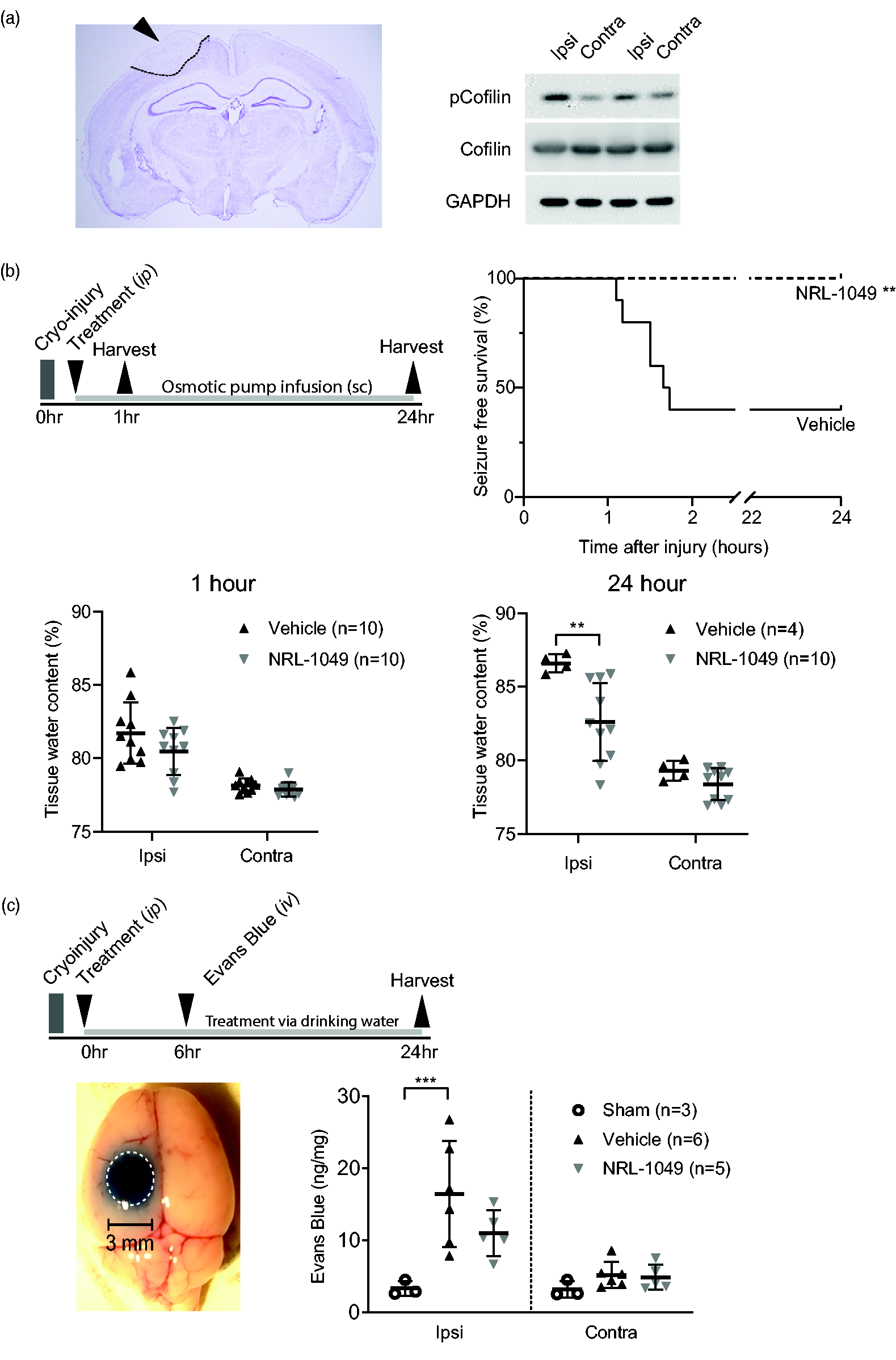
ROCK activity and reduced BBB permeability after NRL-1049 after cerebral cryoinjury in mice. (a) Example picture of H&E stained coronal brain section showing the cortical lesion due to cryoinjury (arrow and delineated area). Western blots showing increased pCofilin on the cryoinjured (ipsi-lateral) side of the brain at 24 hours after injury. (b) Experimental timeline of the edema measured as wet/dry weight at 1 and 24 hour after cryoinjury in the ipsi-lateral hemisphere (cryoinjured) and contra-lateral Continued.hemisphere. Kaplan-Meier curve demonstrating the percent of mice seizure-free in the first 24 after cryoinjury (**p = 0.004; Survival (Mantel-Cox) test, n = 10 for each group). There is a non-significant trend to less water in the NRL-1049 group compared with vehicle treated animals (n = 10 per group). At 24 hours after cryoinjury, NRL-1049 significantly reduced percentage of water (**p = 0.003; two-way ANOVA (Šídák’s multiple comparisons test), n = 10 NRL-1049 treated animals, n = 4 vehicle controls) and (c) Experimental timeline and example picture of EB leakage in and around the lesion (white dotted line) and quantification of EB leakage in the ipsi- and contralateral hemisphere in sham (n = 3), vehicle (n = 6) and NRL-1049 (n = 5) treated mice (***p < 0.001; two-way ANOVA (Šídák’s multiple comparisons test)). In the 1 and 24-hour readout groups, NRL-1049 or vehicle was administered via a single loading dose (20 mg/kg, ip), where only in the 24-hour group this was followed by subcutaneous delivery via osmotic pump (10 µg/hour).
NRL-1049 preserves the BBB and suppresses hemorrhagic transformation after ischemic stroke
To enhance the clinical relevance and seek efficacy in a particularly severe model, we induced focal cerebral ischemia in spontaneously hypertensive rats. 45 NRL-1049 (10 mg/kg ip) administered at reperfusion following 90 minutes MCAO significantly reduced EB leakage in the ischemic hemisphere when measured 9 hours after stroke onset (Figure 5 and Suppl. Figure 2, n = 18 vehicle and 16 NRL-1049, p = 0.047; Welch’s t-test). Moreover, when we qualitatively examined the brains before EB measurements, we observed a significantly lower proportion of SHR with hemorrhagic transformation in the NRL-1049 arm (78% in vehicle vs 25% in the NRL-1049 arm, p = 0.004; Chi-square test). The hemorrhage grade 21 was also worse in the vehicle group (mean score 1.22 and 0.43, vehicle and NRL-1049, respectively, p = 0.007; Mann-Whitney test). These data showed that NRL-1049 preserves the BBB and reduces the incidence of intra-infarct hemorrhage after ischemia-reperfusion injury in a clinically relevant animal model of essential hypertension.
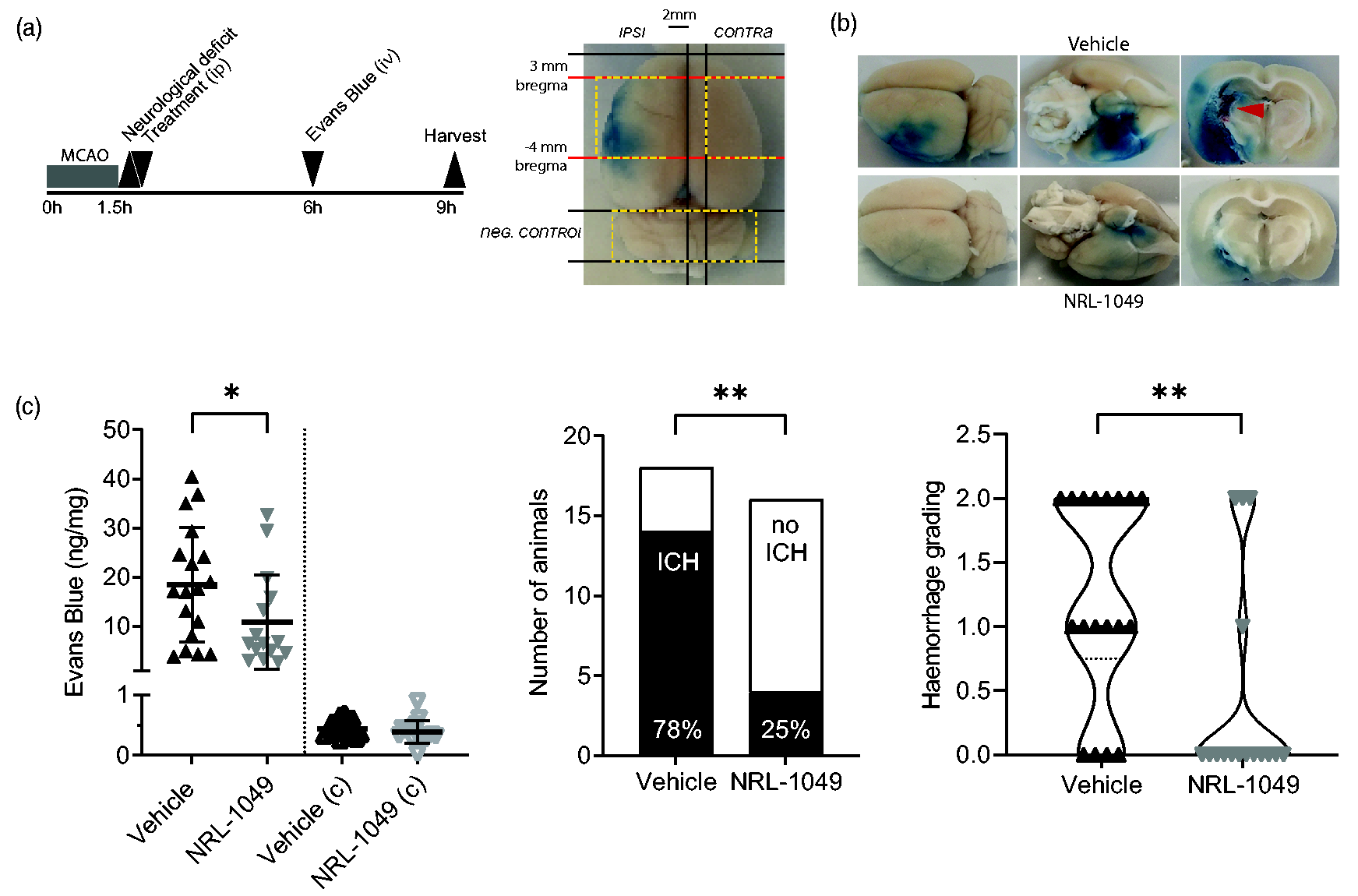
NRL-1049 reduced BBB disruption after experimental ischemic stroke. NRL-1049 reduces EB dye 13 extravasation and intracerebral hemorrhage after transient MCAO in rats. (a) Experimental timeline of 90-minute MCAO in spontaneously hypertensive rats. Treatment with NRL-1049 or vehicle was administered via a single loading dose (10 mg/kg ip) 15 minutes after reperfusion. EB was injected 4.5 hours after reperfusion and 3 hours before sacrifice. On the right side the brain sectioning scheme. Yellow dashed brain parts were analyzed (Ipsi-, contralateral and cerebellum). (b) Representative rat brains and their coronal sections with EB leakage and intracerebral hemorrhages (red arrow) and (c) EB leakage after transient MCAO and treatment with NRL-1049 or vehicle. Intraperitoneal administration of 10 mg/kg NRL-1049 at 15 minutes after reperfusion significantly reduces the concentration of EB leakage in the ischemic side of the brain 3 hours after EB injection and 7.5 hours after reperfusion (*p = 0.047, n = 18 vehicle, n = 16 NRL-1049-treated rats; Welch’s t-test). NRL-1049 administration significantly reduced the proportion of rats with detectable intracerebral hemorrhage 7.5 hours after reperfusion (**p = 0.004; Chi-square test). Grading intracerebral hemorrhages also showed a worse outcome in the vehicle group compared with the treatment group (**p = 0.007; Mann-Whitney test).
Discussion
We describe the pharmacology of a novel, orally bioavailable ROCK2 inhibitor NRL-1049 34 and show that it preserves the BBB after acute brain injury. NRL-1049 has ∼40-fold lower IC50 for ROCK2 over ROCK1 and does not inhibit PKA or PKC at physiological ATP concentrations. 17 It has an active metabolite, 1-hydroxy-NRL-1049, that we have synthesized and named NRL-2017. Pharmacokinetic studies show that in vivo the metabolite has biological activity to inhibit ROCK2.
ROCK1 and ROCK2 are highly homologous kinases with overlapping functions but different tissue distributions. 46 NRL-1049 is a selective ROCK2 inhibitor but not completely specific to ROCK2 because higher concentrations of NRL-1049 will also inhibit ROCK1 (Figure 1). NRL-1049 differs from other ROCK inhibitors in its ROCK2/ROCK1 inhibition profile, its clean-off target profile,10,12 and its promising safety profile. 35 Our data show that NRL-1049 ameliorates BBB disruption, protein extravasation, edema formation, and hemorrhagic transformation after acute ischemic stroke or cryoinjury. These results are in line with previous results of NRL-1049 given PO to transgenic mice bred to test efficacy for the treatment of cavernous malformations. In these studies, low doses of 1 mg/kg/d NRL-1049 significantly reduced lesion hemorrhage, and higher doses of 100 mg/kg/d significantly reduced lesion volume. 35 The effect of NRL-1049 on the reduction of edema formation and hemorrhagic transformation is in line with other, non-specific ROCK1/ROCK2 kinase inhibitors. For example, Y-27632 protects BBB integrity and function after acute ischemic stroke.11,47 Since ROCK1 inhibition can be associated with cardiovascular side effects,6,48 ROCK2 selectivity of NRL-1049 may retain efficacy while maximizing safety in indications where both ROCK1 and ROCK2 play a role.
We carried out in vitro studies of NRL-1049 and its metabolite NRL-2017 on brain endothelial cells. We observed a prolonged duration of the inhibitory effect on the activation of the ROCK pathway. Treatment with NRL-1049 reduced MLC2 phosphorylation in 1-4 hours after drug or metabolite washout. Although non-ROCK substances may also act to trigger MLC2 phosphorylation, 49 the sustained effect in our culture experiments is consistent with in vivo results from our previous study 35 where we showed an increased level of NRL-2017 in blood vessels compared with plasma. The possibility that vascular cells export the drug only slowly, leading to prolonged inhibition of ROCK2, warrants further investigation.
We studied NRL-2017, the active 1-hydroxy-NRL-1049 metabolite, and found that it is highly selective for ROCK2 compared with ROCK1. NRL-2017 is more potent than NRL-1049 to block ROCK2 activity in vitro (Figures 1 and 2). The metabolite might lengthen the duration of action and contribute to the potency of the protective properties of NRL-1049 during acute brain injury. The higher activity of the metabolite compared to the main compound is also a feature of fasudil, a non-selective Rho kinase inhibitor, where hydroxy-fasudil (the metabolite) more selectively blocks Rho Kinase activity.11,50,51 Hydroxy-fasudil is also eliminated in vivo more slowly than fasudil,52,53 as we observe for NRL-1049 and its metabolite. Preliminary investigations of NRL-2017 indicate that it is not orally available (not shown), unlike NRL-1049. Further investigation of the in vivo activity of NRL-2017 should be done via intravenous delivery. Since NRL-1049 has good bioavailability when given po or ip and is largely metabolized into NRL-2017 after systemic administration (i.e., first metabolite), we chose to test NRL-1049 only, in vivo, in this study. We also tested different combinations of administration routes in this study (i.e., intraperitoneal followed by oral (cryoinjury for Evans blue leakage in mice), intraperitoneal followed by subcutaneous (cryoinjury for tissue water content in mice), and intraperitoneal alone (ischemic stroke for Evans blue leakage in spontaneously hypertensive rats). Although the oral route is clinically preferred for chronic use, in acute brain injury, it is often desirable to get the drug into the bloodstream as quickly and predictably as possible, and the oral route may not always be safe due to dysphagia and aspiration risk, which also applies to rodents immediately after anesthesia and brain injury. For these reasons, we chose the initial dosing to be intraperitoneal.
Our overarching focus was the BBB integrity after brain injury. The cryoinjury model yields robust BBB breakdown leading to vasogenic edema within 2 hours and has been used to examine the efficacy of interventions targeting the BBB.54 –59 We first tested the efficacy of NRL-1049 in the cryoinjury model in mice. After finding efficacy, we extended the testing to a more clinically relevant injury model, i.e., focal ischemic stroke. For the latter, we decided to use a different species to strengthen the translational value of the study. While our primary readout was the BBB integrity by EB, we also wanted to assess the incidence of hemorrhagic changes in the infarct. However, the assessment was done at a single coronal section because the EB protocol required rapid tissue processing to avoid signal loss due to light exposure; therefore, we might have missed hemorrhage at other rostrocaudal levels. While this is a limitation, we believe the hemorrhage rate at the imaged coronal plane likely reflects the overall propensity for hemorrhagic changes elsewhere.
There are 12.2 million strokes every year, and over 100 million people live with the consequences of a previous stroke. 60 Despite the magnitude of the problem and enormous research efforts to address it, the vast majority of stroke trials that evaluated neuroprotective compounds have failed. 61 However, there has not been a large randomized controlled trial published on ischemic stroke patients targeting the BBB. Clinical 62 and preclinical20,63 –65 studies with fasudil have demonstrated that targeting the BBB following stroke may be a promising strategy, but potential systemic (e.g., cardiovascular) side effects have hindered its clinical development. Fasudil inhibits not only ROCK1 and ROCK2 but also protein kinase A and other kinases. 12 It is approved in Japan to prevent vasospasm following subarachnoid hemorrhage, but its safety profile for long-term use is unknown. Accumulating data show that drugs that selectively target ROCK2 may have a better safety profile.6,48,66 NRL-1049 is currently in preclinical development to treat cerebral cavernous malformations. In conclusion, our study aligns well with STAIR recommendations, 67 and suggests that exploration of both NRL-1049 and NRL-2017 for preserving the blood-brain barrier integrity following ischemic stroke is further warranted. While NRL-2017 might be useful in an emergency setting, the ability to use NRL-1049 as an oral drug opens up the possibility of long-term treatment as well.
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X241238845 - Supplemental material for The novel ROCK2 selective inhibitor NRL-1049 preserves the blood-brain barrier after acute injury
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X241238845 for The novel ROCK2 selective inhibitor NRL-1049 preserves the blood-brain barrier after acute injury by Inge A Mulder, Matt Abbinanti, Sarah A Woller, Joerg Ruschel, Jonathan M Coutinho, Helga E de Vries, Ed van Bavel, Kenneth Rosen, Lisa McKerracher and Cenk Ayata in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was in part funded by The International Headache Society 2017 Fellowship Award (to I.A.M.); Royal Netherlands Academy of Arts and Sciences Van Leersum Grant 2017 (to I.A.M.); The Dutch Heart Foundation 2021 E. Dekker Grant (03-006-2021-T019 to I.A.M.); Amsterdam Neuroscience (to J.M.C. and E.vB.); Neurelis, Inc (San Diego, CA); BioAxone BioSciences, Inc (Boston, MA).
Acknowledgements
The authors would like to thank Ms. Radhika Barve for technical support with the in vitro experiments.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. McKerracher is CEO of BioAxone BioSciences, Inc. and hold an ownership interest in the company and has a significant competing interest. The data for this article were collected while Drs. Abbinanti, Woller, Ruschel and Rosen were employed at BioAxone BioSciences, and they have a modest conflict of interest. The opinions expressed in this article are the author’s own and do not reflect the view of the National Institutes of Health, the Department of Health and Human Services, or the United States government. Neurelis, Inc. acquired the rights to BA-1049 (now referred to as NRL-1049) from BioAxone in June 2021. Ayata is on the scientific advisory board of Neurelis, Inc. and declares sponsored research agreements with Praxis and Takeda Pharmaceutical Company. Mulder declares a past sponsored research agreement with BioAxone BioSciences, Inc. (concerning work not included in this manuscript). The other authors declare no conflict of interest. No writing assistance was utilized in the production of this manuscript.
Authors’ contributions
According to Brand et al. (2015), Learned Publishing 28(2).
Mulder; Conceptualization, Methodology, Validation, Formal analysis, Investigation, Writing – Original Draft Preparation, Writing – Review & Editing, Visualization, Project administration, Funding acquisition.
Abbinanti; Conceptualization, Methodology, Validation, Formal analysis, Investigation, Writing – Review & Editing, Project administration.
Woller; Methodology, Validation, Formal analysis, Investigation, Writing – Review & Editing, Project administration.
Ruschel; Methodology, Validation, Investigation, Writing – Review & Editing, Project administration.
Coutinho; Validation, Writing – Review & Editing, Funding acquisition.
de Vries; Validation, Resources, Writing – Review & Editing, Supervision, Funding acquisition.
van Bavel; Validation, Resources, Writing – Review & Editing, Supervision, Funding acquisition.
Rosen; Conceptualization, Methodology, Validation, Formal analysis, Resources, Writing – Original Draft Preparation, Writing – Review & Editing, Supervision, Funding acquisition.
McKerracher; Conceptualization, Validation, Resources, Writing – Original Draft Preparation, Writing – Review & Editing, Supervision, Funding acquisition.
Ayata; Conceptualization, Methodology, Validation, Formal analysis, Resources, Writing – Original Draft Preparation, Writing – Review & Editing, Visualization, Supervision, Funding acquisition.
Supplementary material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
