Abstract
The role of the phosphodiesterase type IV isozyme (PDE IV) in the regulation of cerebrovascular tone was investigated in the canine basilar artery in vitro and in vivo. The PDE isozymes extracted from the canine basilar artery were isolated by diethylaminoethanol (DEAE)-Sepharose affinity chromatography and identified based on sensitivity to tsozyme-selective PDE inhibitors. [3H]cAMP hydrolysis was observed in one major and one minor peak of activity. The predominant peak was inhibited by the addition of cGMP (25%), siguazodan (26%), rolipram (39%), and the combination of siguazodan and rolipram (95%). Selective PDE IV inhibitors BRL 61063, rolipram, and denbufylline were equieffective inhibitors of [3H]-ccAMP hydrolysis mediated by PDE IV isolated from the canine basilar artery [concentrations producing 50% inhibition (IC50s) = 0.21 ± 0.05 μM, 0.67 ± 0.23 μM, and 0.73 ±0.16 μM, respectively]. In precontracted isolated ring segments of the canine basilar artery, selective PDE IV inhibitors produced potent and complete relaxation (IC50s <150 nM). In contrast, zaprinast (a selective PDE V inhibitor) and siguazodan (a selective PDE III inhibitor) produced only weak relaxation of the basilar artery (IC50s = 4.5 μM and >10 μM, respectively). Vasorelaxation produced by PDE IV inhibitors was not altered by removing the endothelium, l-NAME, or adenosine receptor antagonism. In a canine model of acute cerebral vasospasm, all three selective PDE IV inhibitors reversed basilar artery spasm produced by autologous blood without altering mean arterial blood pressure. In contrast, prolonged treatment with BRL 61063 failed to alter the development of basilar spasm in the two hemorrhage canine models of chronic cerebral vasospasm. Denbufylline-induced relaxation in vitro was also significantly impaired in basilar arteries obtained from the model of chronic vasospasm. In conclusion, PDE IV appears to be the predominant isozyme regulating vascular tone mediated by cAMP hydrolysis in cerebral vessels. In addition, vasorelaxation modulated by PDE IV is compromised in chronic cerebral vasospasm associated with subarachnoid hemorrhage.
Keywords
The family of phosphodiesterase (PDE) isozymes is responsible for the intracellular hydrolysis of cAMP and cGMP. Some members of the seven PDE gene families differ in their structure, substrate specificity, sensitivity to selective inhibitors, and tissue distribution (for review, see Beavo et al., 1994). PDE
III (a cAMP-preferring enzyme) is deficient in the brain, and the majority of cAMP hydrolysis is via the PDE IV isozyme, where it is an important regulator of neuronal excitability and activity (Nemoz et al., 1989; Challiss and Nicholson, 1990). In this regard, selective PDE IV inhibitors (i.e., denbufylline and rolipram) possess a variety of CNS effects, including anti-ischemic, antidepressant, and nootropic activity (Angersbach, 1991; Nicholson et al., 1991).
In peripheral arteries (e.g., rat and bovine aorta), membrane-bound and cytosolic PDE III and PDE IV catalyze cAMP hydrolysis (Nicholson et al., 1991; Ivorra et al., 1992; Komas et al., 1993). A variety of PDE III inhibitors produce complete, endothelium-independent relaxation in precontracted rat aorta (Komas et al., 1991) and potentiate the vasorelaxant effects of isoprenaline (Lindgren et al., 1990). In contrast, selective PDE IV inhibitors alone had modest endothelium-dependent vasorelaxant properties (Komas et al., 1991) and did not potentiate isoprenaline vasorelaxation (Lindgren et al., 1990).
The regulation of cAMP hydrolysis is also important in modulating the vascular tone in the CNS (Suzuki et al., 1988; Parfenova et al., 1993); however, the PDE isozymes that regulate cAMP in cerebral vessels have not been well characterized. The superfusion of selective PDE III and IV inhibitors on mouse and cat pial arteries produced mild to moderate vasodilation, respectively (Parfenova et al., 1993; Rosenblum et al., 1993).
The purpose of the present study was to identify and characterize the cAMP PDE isozymes (PDE III and IV) in a major cerebral artery and to assess their functional role. Specifically, selective PDE inhibitors denbufylline, (±) rolipram, and BRL-61063 (Fig. 1) were evaluated for their effects on (1) cerebrovascular cAMP hydrolysis, (2) spasmogen-induced contractions in isolated canine cerebral vessels, and (3) acute and chronic cerebral vasospasm in a model of subarachnoid hemorrhage (SAH).
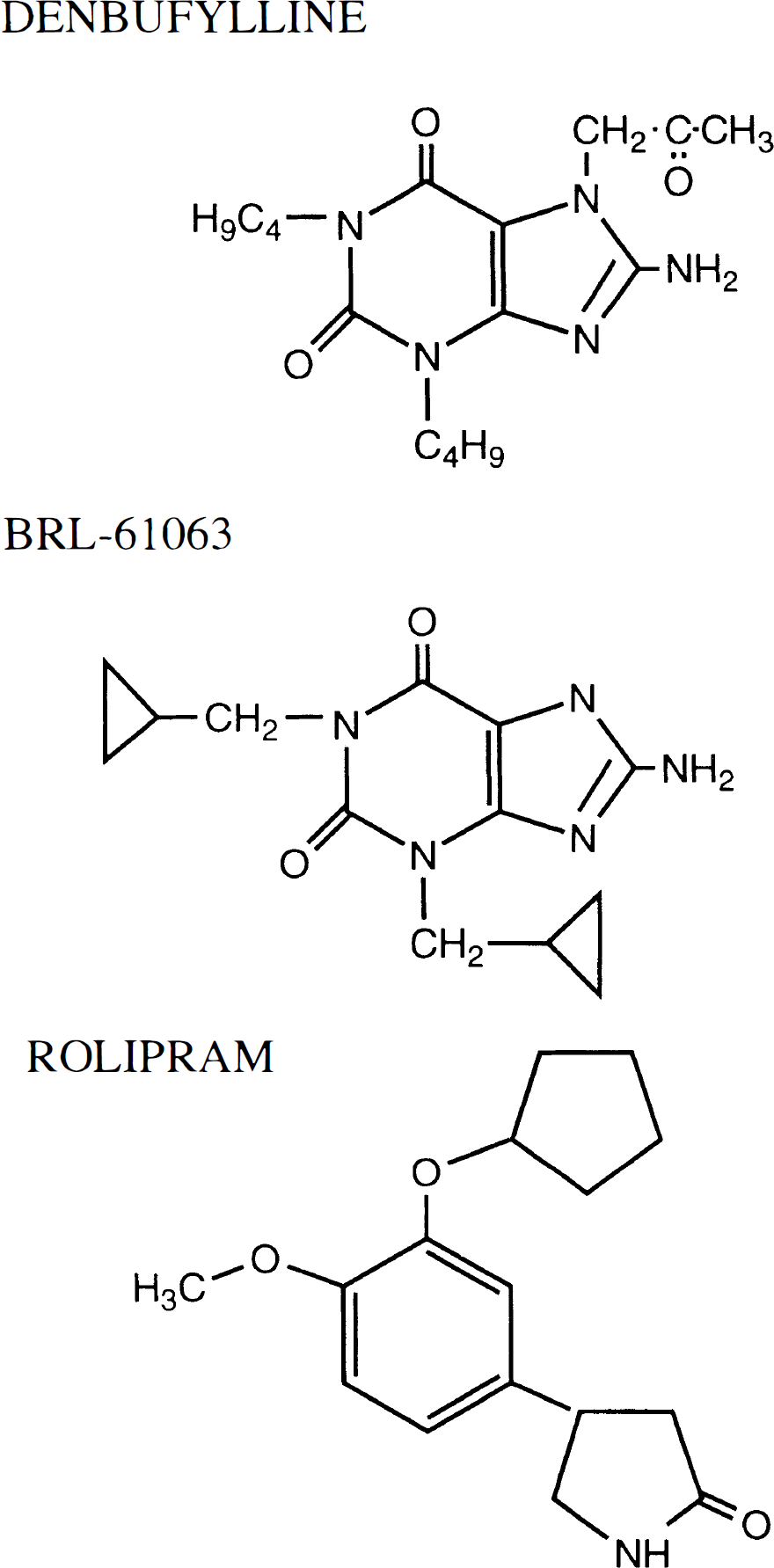
Chemical structures of the three selective phosphodiesterase type IV (PDE IV) inhibitors employed in the present study [denbufylline, BRL 61063 and (±) rolipram].
MATERIALS AND METHODS
Separation and partial purification of PDE isozymes from canine basilar arteries
Tissue preparation.
Basilar arteries were pooled (n = 8), and PDE isozyme separations and partial purification were performed according to Torphy and Cieslinski (1990). Briefly, the arteries were quickly frozen in liquid N2 and stored at −70°C until being used. All further procedures were performed at 4°C. The arteries were chopped and homogenized with a Brinkman PT 10/35 polytron in 10 vol of buffer (pH 6.5) containing 20 mM bis-Tris, 2.5 mM dithiothretol, 10 mM ethylenediaminetetra-acetic acid (EDTA), 0.1% Triton x-100, plus a protease inhibitor mixture of 2 mM benzamidine, 20 μg/ml of soybean trypsin inhibitor, 100 μg/ml of bacitracin, 100 μM p-tosyl-L-lycine chloromethyl ketone, 100 nM leupeptin, and 50 μM phenyl-methylsulphonyl fluoride. The homogenate was centrifuged at 20,000 g for 30 min, and the supernatant was applied to a DEAE-Sepharose CL-6B (Pharmacia Fine Chemicals, Inc. Piscataway, NJ, U.S.A.) column for the separation of PDE activities.
DEAE-Sepharose chromatography.
PDE isozyme separation was performed on a DEAE-Sepharose column (1.6 cm i.d.) with a 10-ml bed volume equilibrated with homogenization buffer. A sample of the 20,000 g supernatant (1.8 ml) was applied to the column, followed by a wash with 25 ml of homogenization buffer. No detectable PDE activity was in this initial wash. The PDE isozymes then were eluted with 50 ml of a linear (0–1.0 M) sodium acetate gradient in the homogenization buffer at a flow rate of 10 ml/h. One-milliliter fractions were collected over the entire gradient. Bovine serum albumin was added to each fraction to a final concentration of 0.1%. The PDE activity was determined in every other fraction by using 1 μM [3H]cAMP or 1 μM [3H]cGMP as substrate (see below for assay method). At least 80% of the PDE activity was recovered from the anion-exchange column.
Phosphodiesterase assay.
Phosphodiesterase activity was determined as described previously (Torphy and Cieslinski, 1990). The reaction was conducted in a 0.1-ml buffer containing (final concentration) 50 mM Tris-HCl buffer (pH 7.5), 5 mM MgCl2, 50 μM 5′[14C]AMP, or 50 μM 5′-[14C]GMP (∼400 dpm/nmol) as carrier and for recovery of product, 1 μM [3H]cAMP or 1 μM [3H]cGMP (∼2,000 dpm/pmol), and enzyme. The reaction was initiated with either enzyme or substrate and conducted at 30°C. Incubation time varied depending on the amount of enzyme activity. The reaction was terminated by placing reaction vessels in a 100°C heating block for 1 min before they were placed in an ice bath. Cyclic nucleotide substrates were separated from 5′-nucleotide products by using polyacrylamide–boronate gel columns according to the method of Davis and Daly (1979). The 5′-monophosphate products were eluted with 8 ml of 0.25 M acetic acid into a scintillation vial containing 10 ml of Beckman Ready Solv-MP scintillation cocktail. Radioactivity was measured via scintillation spectroscopy. Recovery of 5′-[3H]AMP and 5′-[3H]GMP, as determined with the 5′-[14C]AMP or 5′-[14C]GMP, was 80 to 90%.
Isolated canine cerebral vessels
The methods used to investigate vasorelaxation in isolated segments of canine cerebral arteries have been described in detail elsewhere (Willette et al., 1994). Briefly, adult mongrel dogs were killed via i.v. pentobarbital overdose, and basilar, middle cerebral, and anterior spinal arteries were carefully dissected into a Krebs-Henseleit buffer (composition below). Ring segments (2–3 mm long) were prepared from each vessel with the endothelium intact and suspended for isometric tension recordings in (Grass FT.03, Quincy, MA, U.S.A.) in 50-ml organ baths containing aerated (95% O2/5% CO2) Krebs-Henseleit buffer (inmmol/L: NaCl, 119; KCl, 4.7; KH2PO4, 1.2; CaCl2, 2.5; MgSO4, 1.5; NaHCO3, 25 and glucose, 11) maintained at 38°C. An optimum resting tension of 750 mg was applied to each segment and equilibrated for 45 min before testing. The reactivity of each vascular segment was determined by increasing the concentration of KCl (by addition to the bath) in the bath buffer to 60 mmol/L to induce contraction and by adding arginine-vasopressin (100 nM) to produce an endothelium-dependent relaxation. After washing the tissue completely, submaximal (∼80%) concentrations of spasmogens (U46619, a thomboxane mimetic; oxyhemoglobin; and endothelin-1) were used to contract tissues for vasorelaxation experiments. In some experiments, spontaneous tone was allowed to develop. Pretreatments (e.g., L-NAME, a NOS inhibitor; and aminophylline, a nonselective adenosine antagonist) were added to the bath and equilibrated for 30 min before constructing a cumulative concentration-relaxation curves with selective PDE inhibitors [denbufylline, (±) rolipram, siguazodan, zaprinast]. In some basilar segments, the endothelium was removed by gently rolling the artery over a rod placed in its lumen. Vasorelaxation was expressed as the percent of the spasmogen-induced tension, and potency was estimated by comparison of the concentrations need to produce 50% relaxation (EC50s) or corresponding values.
Canine hemorrhage models of chronic and acute cerebral vasospasm
The 35 male mongrel dogs used for these experiments weighed 10 to 12 kg and were housed in an accredited laboratory animal facility. All procedures were performed in accordance with the Guide for Care and Use of Laboratory Animals (DHEW/DHHS Publication no. NIH 85-23) and were approved by the Institutional Animal Care and Use Committee at SmithKline Beecham Pharmaceuticals.
The procedure employed for producing delayed cerebral vasospasm was similar to the two-hemorrhage model described by Chyatte et al. (1983). On day 1, anesthesia was induced in all dogs by administering thiamylal sodium (8 mg/kg, i.v.). The dogs were intubated immediately and placed supine on a heated fluoroscopy table. Anesthesia then was maintained by mechanical ventilation with isoflurane (2.5% in oxygen), and the femoral artery was exposed using aseptic technique. A hemostasis device (Cordis, Miami, FL, U.S.A.) was placed in the left femoral artery, which allowed continuous monitoring of arterial blood pressure, sampling for arterial blood gas analysis, and passage of the vertebral catheter. The end-tidal CO2 also was monitored continuously. A transfemoral modified 5F Lehman catheter (Bard, Tewksbury, MA, U.S.A.) was guided fluoroscopically into the left vertebral artery and advanced to the vertebral column. The animals then were placed in a prone position. Automated arterial blood gases were determined periodically, and ventilation parameters were adjusted, if necessary, to maintain normocapnia within a limited range (PCO2 = 35–40 mm Hg) before obtaining the baseline (day 1) vertebrobasilar angiogram. All angiograms were recorded at identical magnification and injection parameters. Contrast (Omnipaque 300, Winthrop Pharmaceuticals, New York, NY, U.S.A.) was delivered rapidly (5 ml/s) with a pneumatic syringe (Cook Inc., Bloomington, IN, U.S.A.).
Immediately after the day 1 angiogram, the dog then was placed in a head-down position (∼30°) and the atlanto-occipital membrane was punctured aseptically with a 22-gauge spinal needle, 4 ml of cerebrospinal fluid were removed, and 4 ml of autologous venous blood were injected slowly (2 min). The dogs remained in the head-down position for 30 min. During this time, the dorsal surgical wounds were closed, and the dogs were prepared for recovery. The procedure for the intracisternal administration of autologous blood was repeated on day 3 of the study. On day 7, each animal was again prepared for a transfemoral vertebrobasilar angiogram to assess delayed vasospasm of the basilar and anterior spinal arteries.
Angiographic images of the basilar and anterior spinal arteries were captured every 100 msec and stored digitally for offline analysis and animation (NIH IMAGE 1.43, Rockville, MD, U.S.A.) The diameters of the spastic segments in placebo and treated animals were measured during systole using an edging routine, and the cross-sectional area was calculated. The cross-sectional areas of the spastic segments on day 7 were compared with the same segments in the baseline angiograms. The percentage of reduction in the cross-sectional area was determined, and the results were compared in vehicle and treated dogs.
Acute cerebral vasospasm of the basilar and anterior spinal artery was produced by preparing the animal as on day 1 in the delayed vasospasm model above; however, repeat angiograms were performed at 30 min after the intracisternal administration of the autologous blood. Dogs exhibiting an acute cerebral vasospasm (≥40% reduction in the control angiogram) were included in the study (∼80%). Immediately after the second angiogram, infusions of vehicle (5% polyethylene glycol 200) or denbufylline, BRL-61063, or (±) rolipram were begun, and angiograms were repeated and compared at 30 and 60 min after the infusion to pretreatment angiograms demonstrating acute vasospasm. Acute vasospasm was quantified as indicated above.
Drugs and dose regimens
Selective PDE IV inhibitors were synthesized at SmithKline Beecham Laboratories (King of Prussia, PA, U.S.A.). All other materials were obtained from common commercial sources. All PDE IV inhibitors solutions used for in vivo infusions were prepared fresh as a stock solution (100 μg/ml) in 5% polyethylene glycol 200 and filtered (Millex-GV, 0.2 μm, Bedford, MA, U.S.A.) before administration. Dosing regimens for i.v. administration in acute experiments were 5% polyethylene glycol 200 (1.2 ml i.v. bolus + 2 ml/h for 1 h), denbufylline (10 μg/kg, i.v. bolus + 3 μg/kg/min for 1 h), (±) rolipram (10 μg/kg, i.v. bolus + 1 μg/kg/min for 1 h), and BRL 61063 (10 μg/kg, i.v. bolus + 1 μg/kg/min for 1 h). In chronic experiments, i.v. infusions of vehicle and BRL-61063 were administered on days 1 and 7 (as above) and oral dosing of placebo (lactose) and BRL-61063 was administered in gel caps on days 2–6 (0.2 mg/kg, p.o., b.i.d. on days 2–6).
Statistical analysis
All values were expressed as the mean ± the standard deviation (SD). Multiple treatments were compared with an analysis of variance, followed by a Bonferroni t test. IC50 values were calculated by a four-parameter logistic-curve fitting routine which best described the data (GraFit, 3.01, Erithicus Software Ltd.). Vehicle and treatment results were compared with an unpaired and, in some cases, a paired Student's t test. Differences were considered statistically significant at p ≤ 0.05.
RESULTS
DEAE-Sepharose chromatography
The elution profile of cAMP PDEs obtained from pooled canine basilar arteries (n = 8) is shown in Fig. 2. DEAE chromatography of the soluble fraction produced one major peak of [3H]cAMP hydrolysis at 0.5 M sodium acetate and one minor peak at 0.1 M sodium acetate (Fig. 2A and B). The hydrolysis of cAMP was reduced in column fractions from the large peak by cGMP, siguazadon, (±) rolipram, and the combination of siquazadon and rolipram. The area analysis of the cAMP hydrolysis peaks indicates that the inhibitory activity of cGMP (25%) = siguazodan (26%) < (±) rolipram (39%) < siguazodan and rolipram (95%). These results suggest that cAMP hydrolysis was mediated by PDE IV predominantly and, to a lesser extent, by PDE III. The addition of Ca++/calmodulin stimulated several-fold the small peak observed at 0.1 M sodium acetate and produced a shoulder of activity at 0.25 M sodium acetate (Fig. 2A). These results suggest that this peak contains one or more PDE I subtypes. It is noteworthy that cGMP did not produce the expected increase in cAMP hydrolysis in column fractions at 0.3 M sodium acetate, suggesting a lack of PDE II activity in the basilar artery.
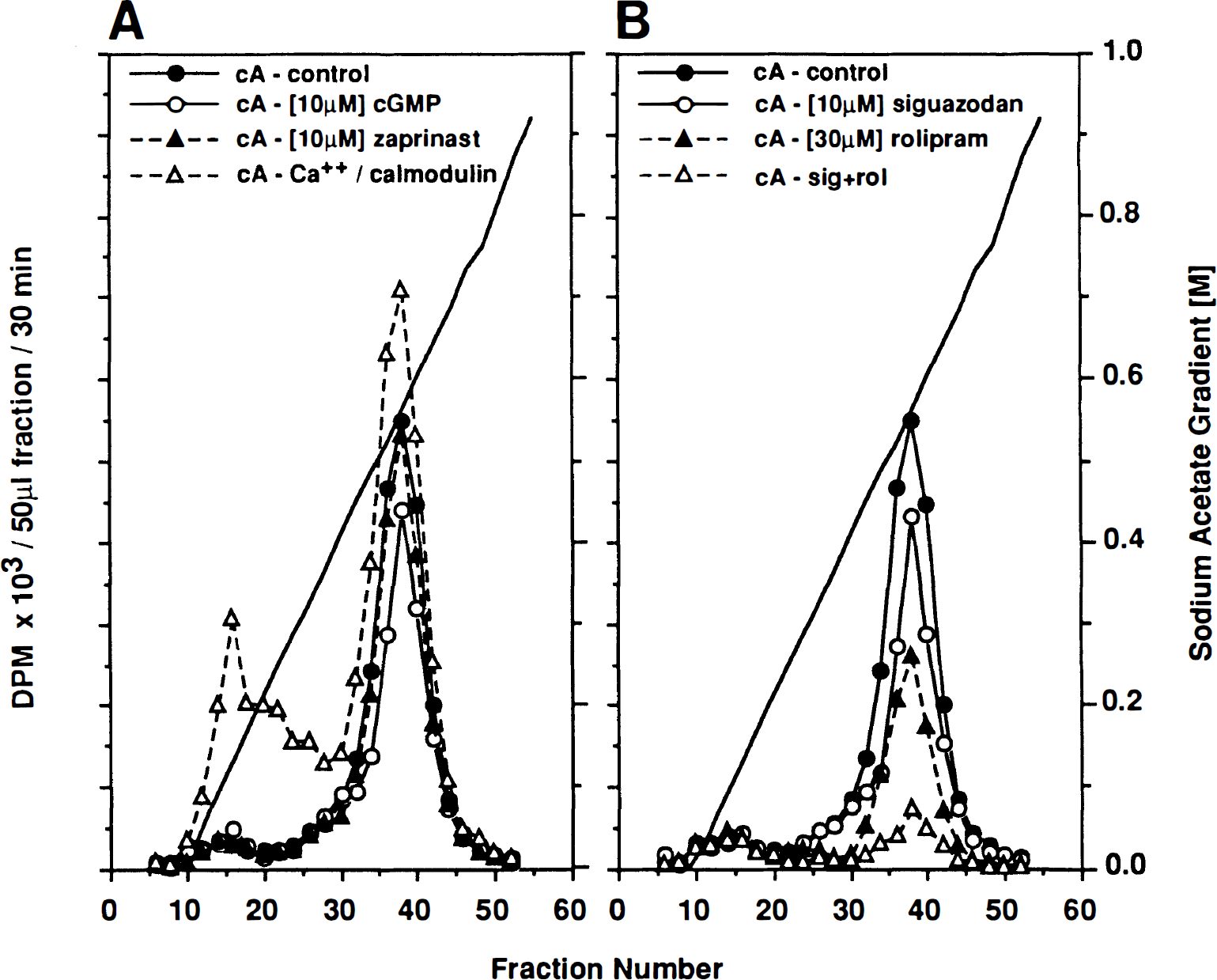
Elution profile of canine basilar artery (n = 8) cAMP-phosphodiesterase (PDE) activity (measured as[3H]cAMP, 1 μM, hydrolysis, [in disintegrations/min (dpm) × 103/50 μl fraction/30 min incubation] from DEAE-sepharose anion exchange columns. Crude enzyme preparations from tissue homogenates were applied to the column and eluted with a linear sodium acetate gradient. [3H]cAMP hydrolysis was determined in column fractions in the absence (control A & B) or in the presence of (
Further characterization of the effects of PDE IV inhibitors was obtained by assessing PDE activity in fractions (34–44) pooled from the large 0.5 M sodium acetate peak (Fig. 3). In the presence of siguazodan (10 μM), the selective PDE IV inhibitors, BRL-61063, (±) rolipram, and denbufylline produced complete concentration-related inhibition of PDE activity (IC50s = 0.21 ± 0.05 μM, 0.67 ± 0.23 μM, and 0.73 ± 0.16 μM, respectively). Thus, BRL-61063, (±) rolipram, and denbufylline were potent, and efficacious inhibitors of partially purified PDE IV were obtained from the canine basilar artery.
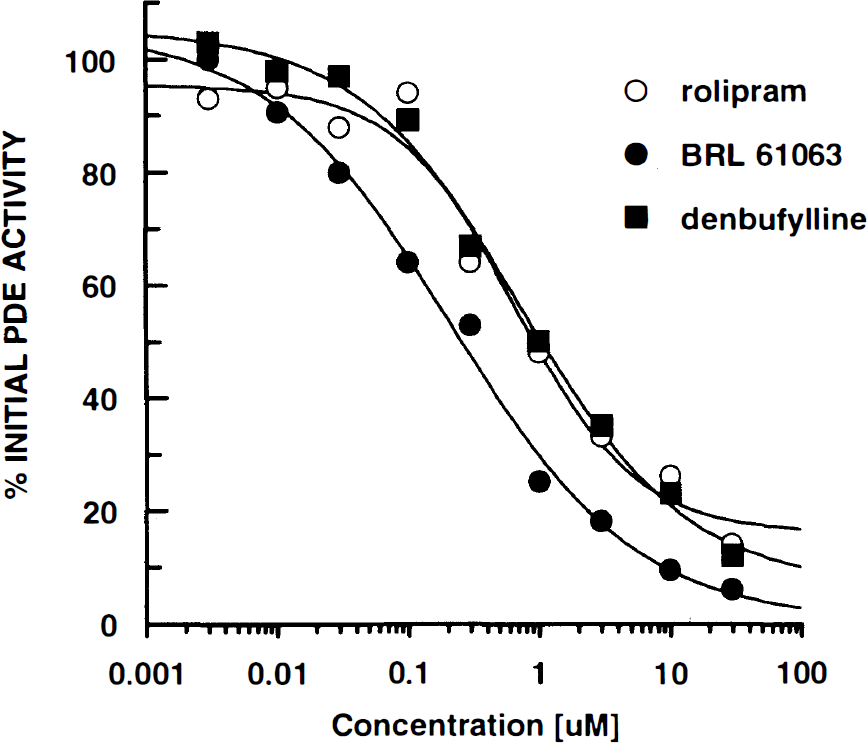
The effects of selective phosphodiesterase type IV (PDE IV) inhibitors ((±) rolipram, BRL 61063, and denbufylline) on canine basilar artery PDE activity (n = 8) were compared in pooled fractions (34–44) from DEAE-sepharose anion exchange columns containing siguazodan (10 μM).
Functional effects of PDE inhibitors in the canine basilar artery in vitro
In isolated ring segments of the canine basilar artery, denbufylline (a selective PDE IV inhibitor prototype) produced complete concentration-related relaxation when the tissues were precontracted with submaximal concentrations of several spasmogens as well as a spontaneously developed tone (Fig. 4A). The IC50s produced by denbufylline in vessels precontracted with U46619 (10 nM), oxyhemoglobin (1 μM), endothelin-1 (10 nM), and spontaneous tone were 144 ± 4 nM, 17 ± 15 nM, 21 ± 14 nM, and 5 ± 3 nM, respectively. Similar results were observed in segments obtained from the canine middle cerebral artery and anterior spinal artery (data not shown).
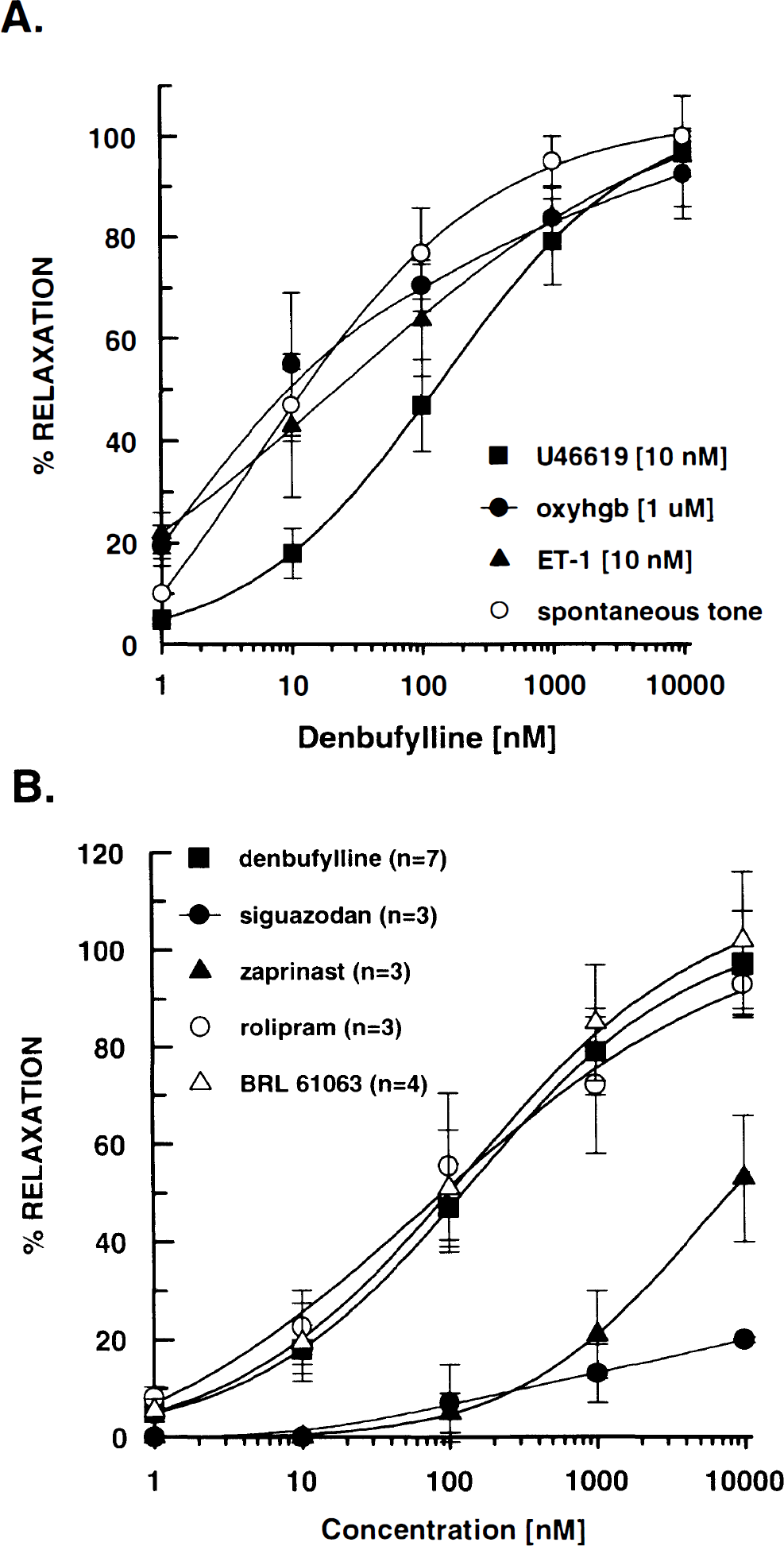
The vasorelaxant effects of denbufylline were compared in isolated segments of the canine basilar artery following precontraction with a variety of spasmogens. A: U46619 (a thromboxane mimetic), oxyhemoglobin (oxyhgb), and ET-1, as well as spontaneous tone. B: the vasorelaxant effects of selective phosphodiesterase (PDE) inhibitors (denbufylline, siguazodan, zaprinast, (±) rolipram and BRL 61063) were compared in isolated segments of canine basilar arteries pre-incubated with U46619 (10 nM). Each group contained basilar arteries from three to seven dogs and two to four observations per artery. All values are the mean ± SD.
The vasorelaxant effects of selective PDE inhibitors were also compared in canine basilar arteries precontracted with U46619 (10 nM). The selective PDE IV inhibitors, denbufylline (IC50 = 144 ± 4 nM), (±) rolipram (IC50 = 73 ± 9 nM), and BRL-61063 (IC50 = 36 ± 21 nM) produced complete concentration-related relaxation (Fig. 4B). In contrast, the selective PDE V inhibitor zaprinast and the selective PDE III inhibitor siguazodan produced only weak vasorelaxation of the canine basilar artery (IC50s = 4.5 ± 2.4 μM and >10 μM, respectively).
The mechanism of vasorelaxation produced by PDE IV inhibitors was explored in canine basilar arteries precontracted with U46619 (10 nM). Specifically, the role of the endothelium in mediating denbufylline- and (±) rolipram-induced vasorelaxation was evaluated in arteries in which the endothelium was removed mechanically. The lack of endothelium-dependent vasorelaxation mediated by arginine vasopressin (100 nM) was used to confirm removal of the endothelium (data not shown). The concentration-dependent relaxation produced by denbufylline was similar in intact (IC50 = 144 ± 4 nM) and denuded (IC50 = 322 ± 304 nM) basilar arteries (Fig. 5). Pretreatment with L-NAME (100 μM), a nitric oxide synthase inhibitor, was also effective in inhibiting the endothelium-dependent vasorelaxation induced by AVP (100 nM); however, L-NAME did not reduce the vasorelaxant potency of denbufylline (IC50 = 47 ± 2 nM) but reduced slightly maximal relaxation (Fig. 5). In addition, aminophylline (1 μM), a nonselective adenosine receptor antagonist, did not inhibit denbufylline-induced (IC50 = 36 ± 4 nM) vasorelaxation (Fig. 5). These results suggest that the endothelium and adenosine receptors do not play a significant role in the relaxation of the canine basilar artery produced by PDE IV inhibitors. Similar results were obtained in experiments performed with (±) rolipram (n = 2).
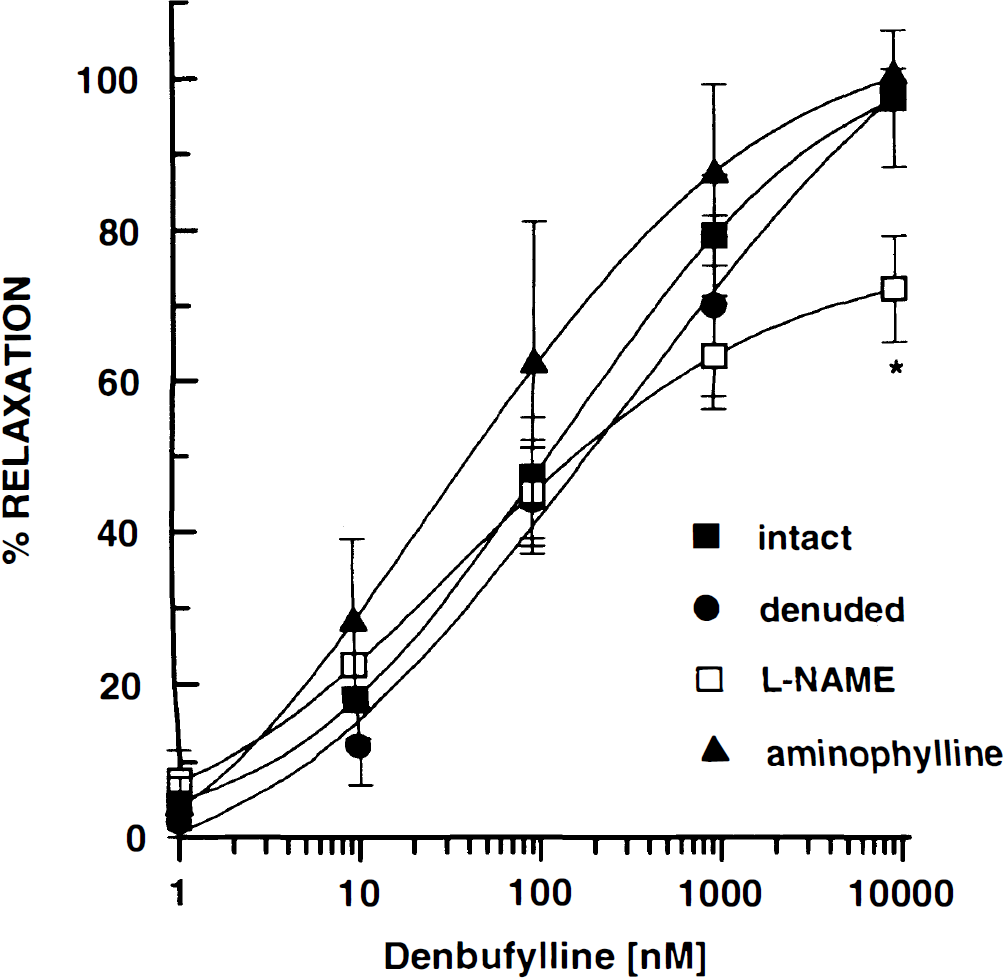
In canine basilar artery segments precontracted with U46619 (10 nM), vasorelaxation induced by denbufylline was compared in blood vessels pretreated with l-NAME (100 μM) or aminophylline (1 μM) and with intact or denuded endothelium. Each group contained basilar arteries segments from three to seven dogs and two to four observations per blood vessel. All values are the mean ± SD. *p < 0.05 compared to the corresponding response in the intact group.
Effect of PDE IV inhibitors on cerebral vasospasm in vivo
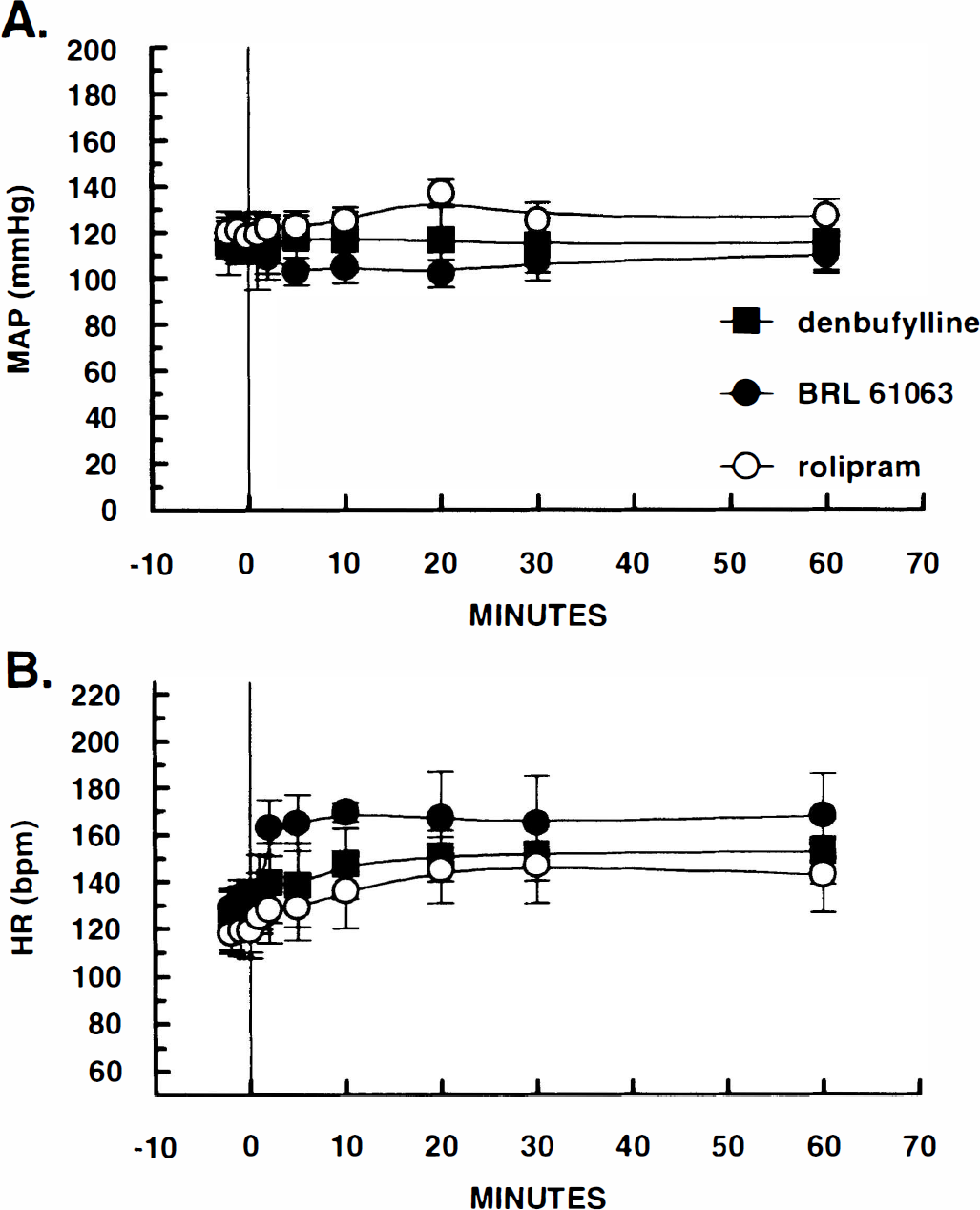
In the acute cerebral vasospasm model, the intracisternal administration of autologous blood produced a significant reduction in the cross-sectional area of the basilar and anterior spinal arteries (Fig. 6A and B) within 30 to 60 min. The acute vasospasm persisted throughout the experiment (≥3 h) and was uneffected by vehicle (5% polyethylene glycol 200) infusion (data not shown). In contrast, the administration of selective PDE IV inhibitors, denbufylline (10 μg/kg, i.v. bolus plus 3 μg/kg/min for 1 h), (±) rolipram, (10 μg/kg, i.v. bolus plus 1 μg/kg/min for 1 h), and BRL-61063 (10 μg/kg, i.v. bolus + 1 μg/kg/min for 1 h) produced a significant reversal of the acute vasospasm when angiograms were repeated at the conclusion of the infusion (Fig. 6A and B). None of the PDE IV inhibitors employed had a significant effect on mean arterial blood pressure (Fig. 7A); however, all three compounds increased heart rate moderately throughout the infusion period (Fig. 7B).
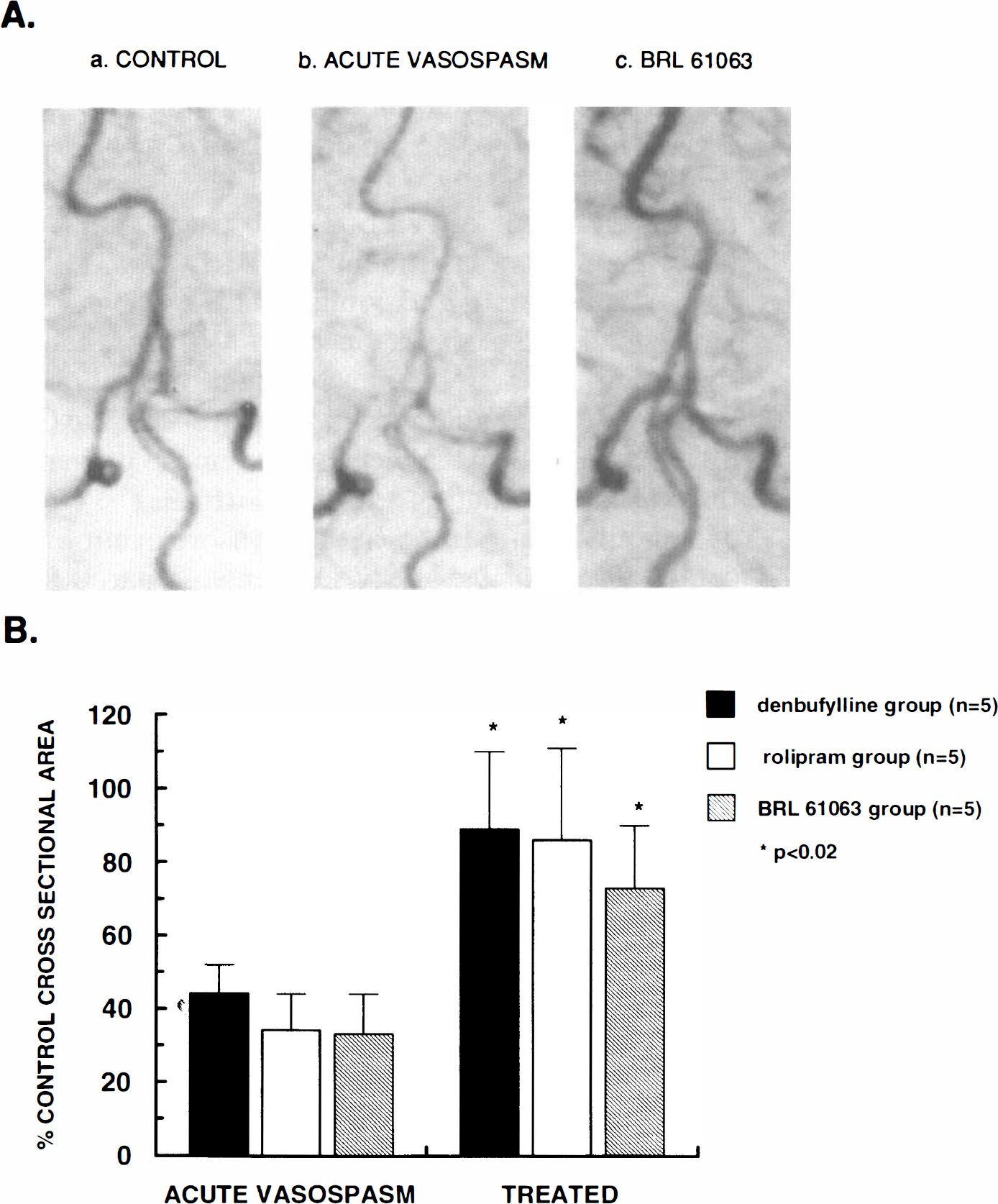
In the two-hemorrhage model of chronic cerebral vasospasm, a severe spasm was observed consistently in the basilar and anterior spinal arteries on day 7 (Fig. 8A). Placebo treatment (5% polyethylene glycol 200 i.v. infusion for 1 h on days 1 and 7, plus lactose capsules, p.o., b.i.d. on days 2–6) had no effect on the development of cerebral vasospasm (similar to historic controls). Treatment with BRL-61063 (10 μg/kg i.v. bolus plus 1 μg/kg/min for 1 h on days 1 and 7; 0.2 mg/kg, p.o., b.i.d. on days 2–6) also had no significant effect on the development of cerebral vasospasm when compared with the placebo group (Fig. 8B). An increase in the incidence of emesis prevented further dose escalation with BRL-61063.
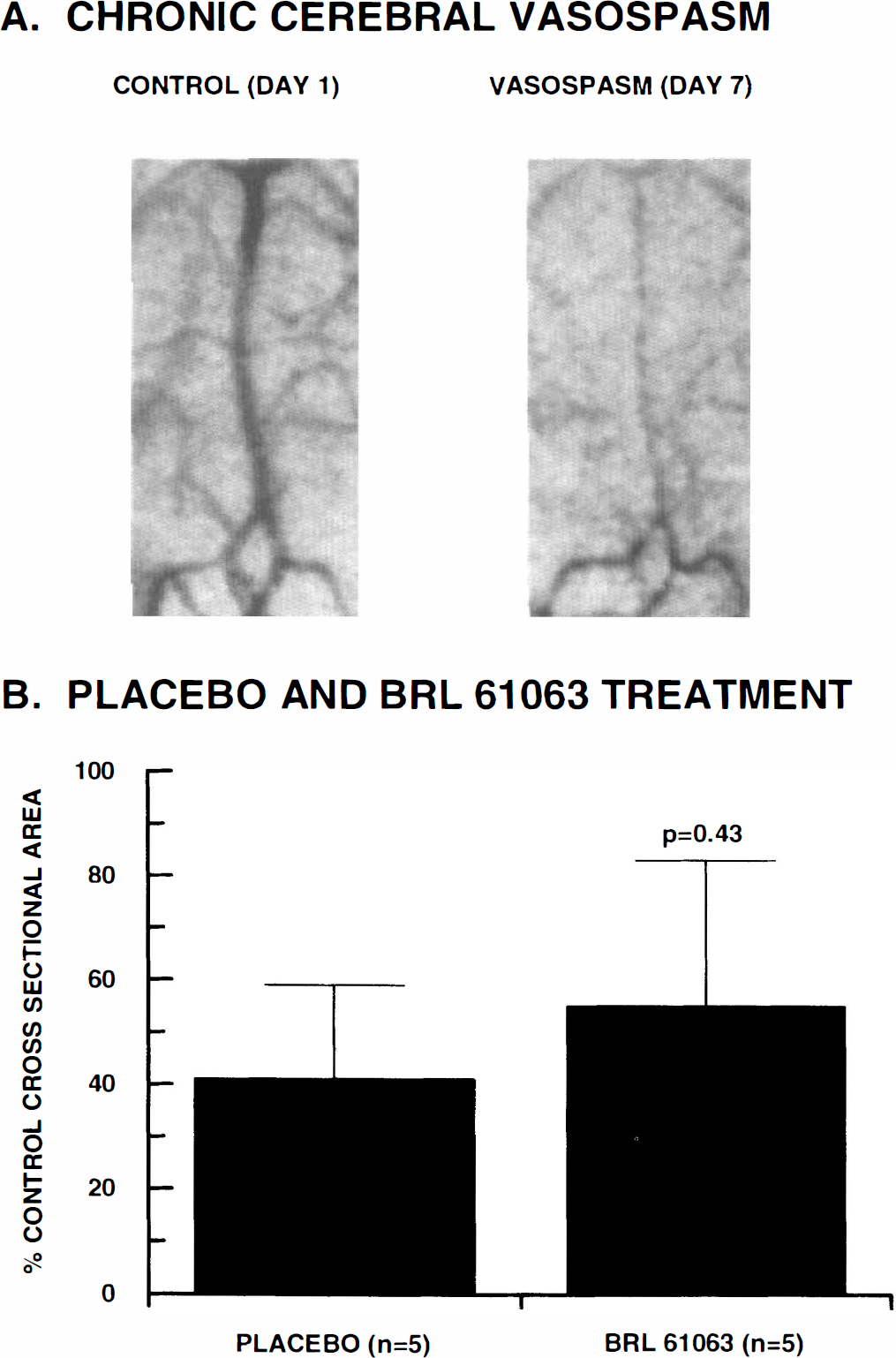
Denbufylline-induced in vitro vasorelaxation was compared in basilar artery segments prepared from some of the 7 day SAH placebo group in Fig. 8 and basilar artery segments obtained from a naive group. Vasorelaxation produced by denbufylline was impaired significantly in the basilar artery segments obtained from the subarachnoid hemorrhage (Fig. 9). There was approximately a 10-fold reduction in denbufylline's potency (IC50 = 144 ± 4 nM; naive group versus IC50 = 1,345 ± 205 nM; SAH group). A similar impairment of (+) rolipram-induced vasorelaxation (n = 1) also was observed in basilar segments obtained from the 7-day SAH placebo group (data not shown).
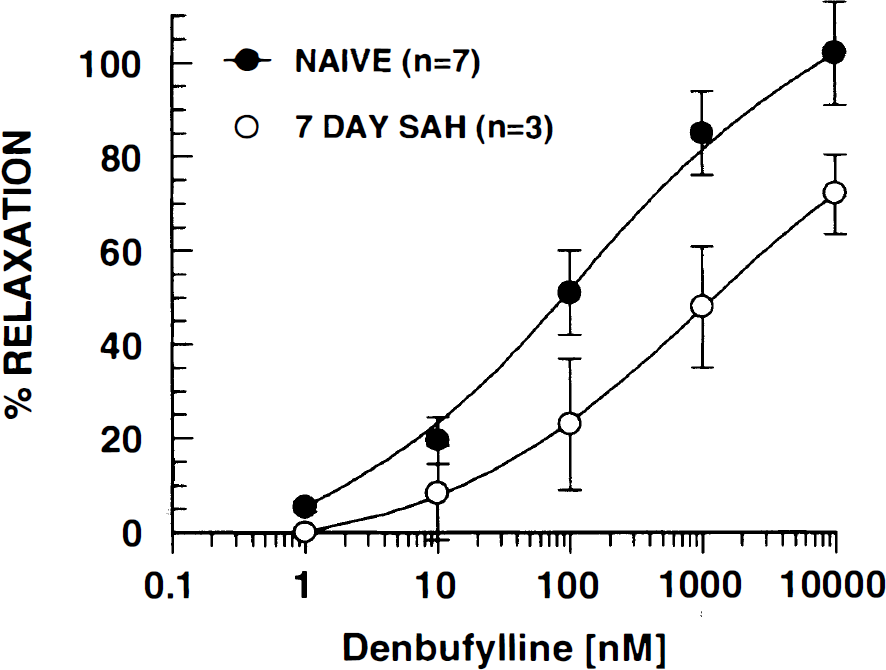
In canine basilar artery segments precontracted with U46619 (10 nM), vasorelaxation induced by denbufylline was compared in vessels obtained from naive animals and untreated animals at days 7 in the subarachnoid hemorrhage (SAH) group. Two to four segments were prepared from each artery. *Significant difference (p < 0.05, unpaired t test) when compared with the corresponding SAH value.
DISCUSSION
The present study indicates that the PDE IV isozyme plays an important role in mediating vasorelaxation in large cerebral arteries and suggests that this mechanism is compromised in chronic cerebral vasospasm associated with subarachnoid hemorrhage. The cAMP/PDE isozyme profile in the canine basilar artery demonstrates that cAMP hydrolysis was catalyzed by PDE IV activity and, to a lesser extent, PDE III activity. Selective inhibition of both PDE III and IV abolished cAMP hydrolysis in the basilar artery. This cAMP PDE isozyme profile appeared similar to that reported in bovine aortic smooth muscle (Ivorra et al., 1992). It is noteworthy that little or no PDE II was demonstrable in the canine basilar artery. The significance of this observation remains to be determined.
In canine basilar artery segments, selective PDE IV inhibitors (denbufylline, BRL 61063, and rolipram) produced complete and potent relaxation in arteries contracted by spontaneous tone as well as oxyhemoglobin, U46619 (thromboxane mimic), and endothelin-1, all proposed mediators of cerebral vasospasm (Steele et al., 1991; Satoh et al., 1991; Willette et al., 1994). Thus, the relaxant effects of PDE IV inhibitors are not limited to inhibition of a specific spasmogen and are consistent with their known mechanism of action. In fact, the vasorelaxant effects were observed over approximately the same range of concentrations needed to inhibit the PDE IV isozyme in the canine basilar artery. In addition, the vasorelaxant potency and efficacy of all three selective PDE IV inhibitors were significantly greater than the selective PDE III inhibitor siguazodan and the selective PDE V inhibitor zaprinast. These results were consistent with the relative preponderance of the PDE IV isozyme in the canine basilar artery. In contrast, the relaxant effects of selective PDE IV inhibitors alone have been shown to be modest, at best, in a variety of peripheral vascular segments in vitro (Lindgren et al, 1990; Lindgren and Andersson, 1991; Komas et al., 1991). For example, (±) rolipram produced only slight relaxation in rat and human coronary and pulmonary arteries but showed more (35%) relaxation in rat and human renal arteries (Lindgren and Andersson, 1991); however, the modest relaxation produced by selective PDE IV inhibitors in the rat aorta is potentiated considerably by agents that increase cAMP and cGMP (Komas et al., 1991). Under these conditions, both endothelium-dependent and independent relaxation can be demonstrated.
Denbufylline and (±) rolipram were used as prototype PDE IV inhibitors to address the potential mechanisms of vasorelaxation in the canine basilar artery. Pretreatment with aminophylline, a nonselective adenosine receptor antagonist, had no significant effect on the vasorelaxant potency and efficacy of PDE inhibitors. It was important to perform this experiment given the known, albeit weak, interactions of some adenosine-like PDE IV inhibitors with adenosine receptors and reuptake sites (Nicholson et al., 1989). These results suggest that the vasorelaxant effects of PDE IV inhibitors are not mediated indirectly through an accumulation of adenosine or via direct interactions with adenosine receptors. In addition, the vasorelaxation produced by PDE IV inhibitors was not dependent on a functional intact endothelium. Endothelium-independent relaxation has also been reported for IBMX, a nonselective PDE inhibitor, in the rat and canine basilar artery (Nishimura et al., 1992; Cosentino et al., 1992). Surprisingly, pretreatment with L-NAME, a nitric oxide synthetase inhibitor, produced a small but significant reduction in the denbufylline maximum response. Similar results were reported for the suffusion of a PDE IV inhibitor (RO-201724) on mouses pial arteries following endothelial damage and L-NMMA (Rosenblum et al., 1993).
Cerebral vasospasm associated with subarachnoid hemorrhage is believed to involve both vasoconstriction and vascular remodelling of large cerebral arteries surrounded by the thick abluminal blood clot (Kim et al., 1989). The ability of PDE IV inhibitors to produce (a) potent vasorelaxation of spasmogens thought to be of importance in cerebral vasospasm (herein), (b) inhibition of vascular smooth-muscle proliferation (Takahashi et al., 1992; Souness et al., 1992), and (c) inhibition of TFN-α production (Kaplan et al., 1995) led to the proposal that selective PDE IV inhibitors may limit cerebral vasospasm by inhibiting vasoconstriction and vascular remodelling in vessels affected by subarachnoid hemorrhage. In a canine model of acute cerebral vasospasm, all three selective PDE IV inhibitors (denbufylline, (±) rolipram, and BRL 61063) reversed the acute basilar spasm observed on days 1 and 2 (data not shown) after the intracisternal administration of autologous blood; however, chronic treatment with BRL-61063, an orally active, metabolically stable analog of denbufylline (Kaplan et al., 1995), failed to prevent the development of basilar artery spasm in a 7-day two-hemorrhage canine model of chronic cerebral vasospasm. These results suggest that the development of acute cerebral vasospasm is not a prerequisite for chronic vasospasm, as PDE IV inhibitors clearly reverse acute cerebral vasospasm, at least during the first 2 days after SAH. In addition, the vasorelaxant potency of denbufylline was significantly reduced (∼10-fold) in isolated segments of vasospastic basilar arteries obtained on day 7 from the SAH group. The results also suggest that the pathomechanisms underlying chronic cerebral vasospasm differ from acute cerebral vasospasm and may limit, either directly or indirectly, cAMP-dependent vasorelaxation. It was shown previously that vasorelaxation and cAMP generation produced by vasoactive intestinal polypeptide and calcitonin gene-related peptide are comprised in isolated basilar arteries from a rabbit model of subarachnoid hemorrhage (Tsukahara et al., 1989; Sutter et al., 1995). In addition, Kim et al. (1992) also demonstrated impaired nitric oxide-mediated vasorelaxation in basilar arteries obtained from the canine chronic SAH model. These investigators determined that the altered cGMP-dependent relaxation was related to an impaired activation of soluble guanylate cyclase and not to cGMP insensitivity. Perhaps a similar impairment of adenylate cyclase regulation may account for the reduced potency and efficacy of PDE IV inhibitors in chronic cerebral vasospasm.
In conclusion, cAMP appears to be an important modulator of cerebrovascular tone, and the PDE IV isozyme plays a prominent role in the hydrolysis of cAMP in large cerebral arteries. In addition, selective PDE IV inhibitors produce potent and efficacious endothelium-independent vasorelaxation in these vessels both in vitro and in vivo. Finally, the vasomodulatory effects of PDE IV are significantly impaired in chronic cerebral vasospasm associated with SAH. This conclusion does not preclude the possibility that PDE IV inhibitors with fewer dose-limiting side effects may yet prove useful for the treatment of chronic cerebral vasospasm in SAH.
Footnotes
Acknowledgment:
We thank Dr. G. Z. Feuerstein for insightful discussions and Ms. Wendy Crowell for help in preparing the figures.
