Abstract
Nitric oxide synthase (NOS) participates in the regulation of cerebral blood flow and neurotransmitter release and as a second messenger of glutamatergic and cholinergic systems. Developmental differences in NOS activity have been described in the rat, but not in a species with longer gestation and a larger, lobulated brain at birth. We assayed NOS activity by conversion of [14C]L-arginine to [14C]L-citrulline in 50-mg tissue samples from eight brain regions in sheep at 70, 92, 110, and 135 days gestation (term = 145 days); newborns (<7 days); and adults to test the hypothesis that NOS activity in the brain is developmentally regulated from midgestation through adulthood and matures along the neuroaxis in parallel with the known development of cerebral blood flow and neuronal activity. Three patterns of maturation of NOS activity were evident: increasing to or exceeding adult levels before 70 days gestation in the thalamus, cerebellum, and medulla; increasing to adult levels between 70 and 92 days in the hippocampus; and increasing to adult levels after 92 days in the cortex and caudate. Additionally, there were regional differences in cortical NOS activity: at 70 and 92 days of gestation, frontal cortex NOS activity was greater than parietal or occipital activity, and at 135 days gestation and in the newborn and adult, cortical and caudate activity exceeded that in most of the more caudal regions. The up to fourfold increase in regional cortical NOS activity between 92 and 135 days gestation was associated with twofold increases in cerebral blood flow and oxygen consumption during this period. Inhibition of NOS activity with administration of 60 mg/kg of NG-nitro-L-arginine methylester (L-NAME) resulted in 27% and 25% reductions in cerebral blood flow at 93 and 133 days gestation. While the associated increases in NOS activity with increases in CBF and CMRO2 do not appear causative, at various points in gestation the development of NOS activity may participate in the development of mature patterns of cerebral blood flow regulation in parallel with development of synaptic and electrical activity.
The role of nitric oxide (NO) in the regulation of basal CBF and activity-dependent cerebrovasodilation in adult models is supported by a significant body of literature (Faraci et al., 1993; Northington et al., 1992; Dirnagl et al., 1994; Faraci and Brian, 1994; Zhang et al., 1995). In vitro studies in the rat and sheep suggest developmental differences in nitric oxide synthase (NOS) expression, which could have functional significance during development (Ma et al., 1991; Bredt and Snyder, 1994; Northington et al., 1996). In sheep, fetuses are sufficiently large for making CBF measurements by ∼90 days gestation (term, 145 days). By that time, neuroblast differentiation and migration are complete (Astrom, 1967; Barlow, 1969), and DNA concentration has stabilized at adult levels (McIntosh et al., 1979). Protein concentration and histochemical staining of cytochrome oxidase in cortex increase abruptly between 80 and 100 days gestation (Barlow, 1969; McIntosh et al., 1979). Sensory evoked responses are converted from positive to negative voltages, indicative of maturation of synaptic circuits, and spontaneous electrocortical activity becomes continuous during this time period (Bernhard et al., 1967; Molliver, 1967). Between 90 and 130 days gestation, there is a corresponding twofold increase in CBF, oxygen consumption, and glucose consumption (Abrams et al., 1984; Gleason et al., 1989). Furthermore, neuronal maturation and increased cytochrome oxidase staining and glucose consumption occur earlier in more caudal regions of brain (Barlow, 1969; Abrams et al., 1984). At 90 days, CBF in caudal regions is greater than that in cortex, and cerebrovascular autoregulation is also more highly developed in these regions (Helou et al., 1994). Therefore, although the ovine brain is more highly developed by birth than in most species, the sheep is one of the few species in which fetal CBF and metabolism measurements can be made in the normal intrauterine environment at relatively immature stages of development.
If CBF, neuronal activity, and autoregulation develop in concert with mechanisms coupling neuronal activity to CBF, and if nitric oxide participates in coupling of neuronal activity to CBF during fetal life, then NOS activity should be developmentally regulated in a manner similar to CBF and neuronal activity. We sought to test this hypothesis by measuring NOS catalytic activity from eight brain regions at six stages of development in sheep. The developmental ages and regions were chosen because they allowed study of a full temporal range of brain development following the end of neuronal migration in brain regions with various levels of metabolic and synaptic activity and various rates of maturation. Furthermore, we measured regional CBF in chronically catheterized fetal sheep at 93 and 133 days gestation, which we found to bracket the period of the greatest increase in cortical NOS activity. We tested a second hypothesis that inhibition of NOS would result in a greater decrease in CBF in the more mature fetus with greater NOS activity.
METHODS
The experimental protocol was approved by the Animal Care and Use Committee of the Johns Hopkins Medical Institutions. To obtain fetal brain tissue, time-dated pregnant ewes (n ≥ 6 at 70, 92, 110, and 135 days gestation) were anesthetized with pentobarbital, and a midline hysterotomy was performed to deliver the fetus. After the umbilical cord was clamped and divided, the fetus was given an overdose of pentobarbital, and a craniotomy was performed to allow removal of the brain. Adult (n = 10) and newborn sheep brains (n = 6) were removed after each animal was given an overdose of pentobarbital. The ages examined bracket the time period during which mature levels of synaptic, neuronal, and metabolic activity develop, myelinization occurs, and mature patterns of CBF develop (Bernhard et al., 1967; Molliver, 1967; Barlow, 1969; Abrams et al., 1984; Gleason et al., 1989).
Nitric oxide synthase activity
In vitro NOS catalytic activity was measured by modification of the techniques described by Bredt and Snyder (1989). Using [14C]L-arginine as a substrate, NO synthase generates equimolar products of NO and [14C]L-citrulline. Brain tissue was harvested and immediately placed in 20 vol ice-cold buffer containing 50 mmol/L Tris and 2 mmol/L ethylenediamine tetraacetic acid (EDTA) (pH 7.4). Tissue samples were taken from frontal, parietal, and occipital cortex; caudate nucleus; hippocampus; thalamus; cerebellum; and medulla at the level of the obex. These areas represent brain regions known to vary in levels of CBF and metabolic activity (Abrams et al., 1984; Gleason et al., 1989; Helou et al., 1994). Samples were placed in Tris buffer, ultrasonicated for 10 s, and then centrifuged at 10,000 g for 15 min. The supernatant was kept on ice until used. In each assay, 25 μl of supernatant was added to 100 μl of reaction mixture containing 1 mmol/L NADPH, 1.25 mmol/L CaCl2, and 500 nmol/L [14C]L-arginine. Samples were run in duplicate. Incubation was carried out at 22°C for 30 min and terminated with 2 ml of buffer containing 30 mmol/L N-2-hydroxy methyl piperazine-2-ethyl ethane sulfonic acid (HEPES) and 3 mmol/L EDTA, pH 5.5. Eachtube was applied to a chromatography column of 0.5 ml Dowex AG50WX-8 (Na+ form), which was then eluted with 2 ml of water. [14C]L-citrulline in the column flow-through was quantified by liquid scintillation spectroscopy (Beckman LS 1800), with an efficiency of 90–94%. NG-nitro-L-arginine methylester (L-NAME; 100 μmol/L) was added to a third aliquot from each supernatant. L-NAME inhibited NOS activity by >98%. Protein was measured according to the method described by Bradford (1976). Results were expressed as the activity per milligram protein (picomoles citrulline · min–1 · mg protein–1). [14C]L-arginine (≈317 Ci/mmol) was obtained from NEN Life Sciences Products (Boston, MA, U.S.A.). All other chemicals were obtained from Sigma Chemical (St. Louis, MO, U.S.A.).
Cerebral blood flow measurements
For CBF measurements, time-dated pregnant ewes were chronically catheterized under halothane anesthesia as previously described (Harris et al., 1989). Through midline abdominal and uterine incisions, catheters were inserted into amniotic fluid and the fetal hindlimb pedal vein, axillary arteries, and superior sagittal sinus. Incisions were closed, and CBF was measured 2 days later in the unanesthetized fetus in utero by the radiolabeled microsphere technique (Harris et al., 1989). Fifteen-micron-diameter spheres labeled with 153Gd, 114In, 113Sn, 103Ru, 95Nb, or 45Sc (NEN Life Sciences Products) were injected at a dose of approximately one million spheres into the fetal inferior vena cava while a reference sample of arterial blood was withdrawn from the axillary artery. The arterial withdrawal lasted from 30 s before the sphere injection to 90 s after the injection catheter was flushed. The withdrawal rate was 1.0 ml/min in 93-day-old fetuses and 2.5 ml/min in 133-day-old fetuses.
Measurements were made before and after fetal administration of the NOS inhibitor L-NAME (60 mg/kg i.v.). This dose was chosen to compensate for the increased blood volume in the fetal-placental unit and because preliminary experiments with 20 mg/kg produced less complete inhibition of in vitro NOS activity (data not shown). Samples of axillary artery and sagittal sinus blood were simultaneously drawn for measurement of oxygen content (OSM3; Radiometer Co., Copenhagen, Denmark). Global cerebral oxygen consumption (CMRO2) was calculated using blood flow to the entire cerebrum. At the end of the experiment, each ewe was euthanized with an anesthetic dose of pentobarbital (30 mg/kg i.v.) followed by potassium chloride injection to arrest the heart. The fetal brain was harvested and dissected into cerebrum, thalamus, cerebellum, and medulla. Arterial blood reference samples and tissue samples were counted for radioactivity, and blood flow was calculated (Harris et al., 1989). The hippocampus and caudate nucleus were too small in 93-day-old fetuses for obtaining accurate blood flow measurements to relate to NOS activity.
Statistical analysis
Activity levels of NOS between regions and age groups were compared with two-factor analysis of variance (ANOVA). Because highly significant differences (p < 0.0001) were found in addition to a highly significant region × age interaction, one-factor ANOVA with the Newman–Keuls multiple-range test was used to identify mean differences within regions across ages and between regions at each age. CBF values before and after L-NAME administration were compared by paired t test. A p value < 0.05 was considered significant. Data are presented as the mean ± SD.
RESULTS
NOS activity
NOS catalytic activity from the cortical regions and caudate nucleus showed overall similarities in the pattern of maturation. At 70 and 92 days of gestation, NOS activity was substantially less than at 135 days of gestation. After 135 days, NOS activity did not increase further in corresponding newborn and adult regions (Fig. 1). Region-specific differences in the rate of maturation to adult levels was evident. Parietal cortex, occipital cortex, and caudate nucleus were the latest maturing regions, with mature levels of NOS activity attained by 135 days of gestation. The rate of maturation to adult levels was intermediate in the frontal cortex, with NOS activity levels reaching adult levels between 92 and 110 days gestation (Fig. 1). Hippocampal activity was comparable to that in the parietal and occipital cortex at 70 days but reached mature levels during the next 22 days of gestation. Levels of NOS activity in the hippocampus at 92 days were nσt different from those in the newborn or adult (Fig. 1).
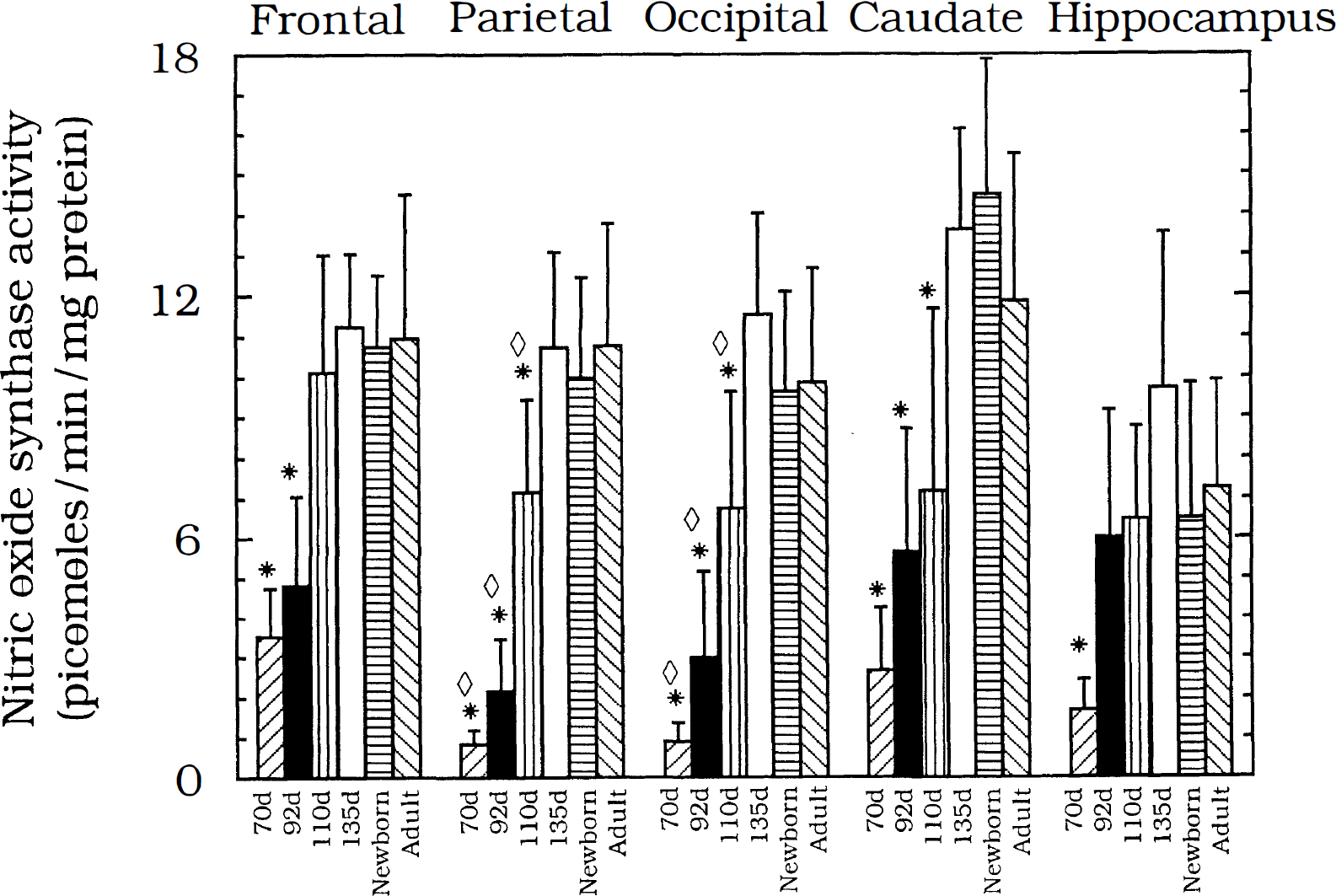
NOS activity (picomoles · min–1 · mg protein–1) in the cortical regions and caudate is shown for the six developmental ages tested: 70 days gestation, left-hatched (n = 6); 92 days gestation, solid bars (n = 8); 110 days gestation, vertical stripes (n = 7); 135 days gestation, open bars (n = 6); newborn, horizontal stripes (n = 6); and adult, right-hatched (n = 10). Within-region differences are seen in all five regions, with activity at 70 days uniformally less than in adulthood (*, p < 0.05 vs. adult) in all regions. At 92 days the frontal, parietal, and occipital cortex and caudate have immature levels of activity (*, p < 0.05 vs. adult). Differences from adult levels of activity persist through 110 days gestation in the parietal and occipital cortex and caudate nucleus (*, p < 0.05 vs. adult). At 70, 92, and 110 days, NOS activity in the parietal and occipital cortex is less than the corresponding activity in the frontal cortex (⋄, p < 0.05 vs. corresponding frontal value). Data are given as means ± SD.
Regional differences in activity within the frontal, parietal, and occipital cortex were present in the least mature animals. At 70, 92, and 110 days of gestation, levels of activity in the frontal cortex were consistently higher than those in the parietal or occipital cortex (Fig. 1). Even earlier maturation of NOS activity to adult levels was observed in the thalamus and medulla (Fig. 2). At no time in the last 0.52 of gestation were activities in these two regions different from those in the newborn or adult. In the cerebellum, in contrast, fetal levels of NOS activity generally exceeded those in newborns and adults (Fig. 2).
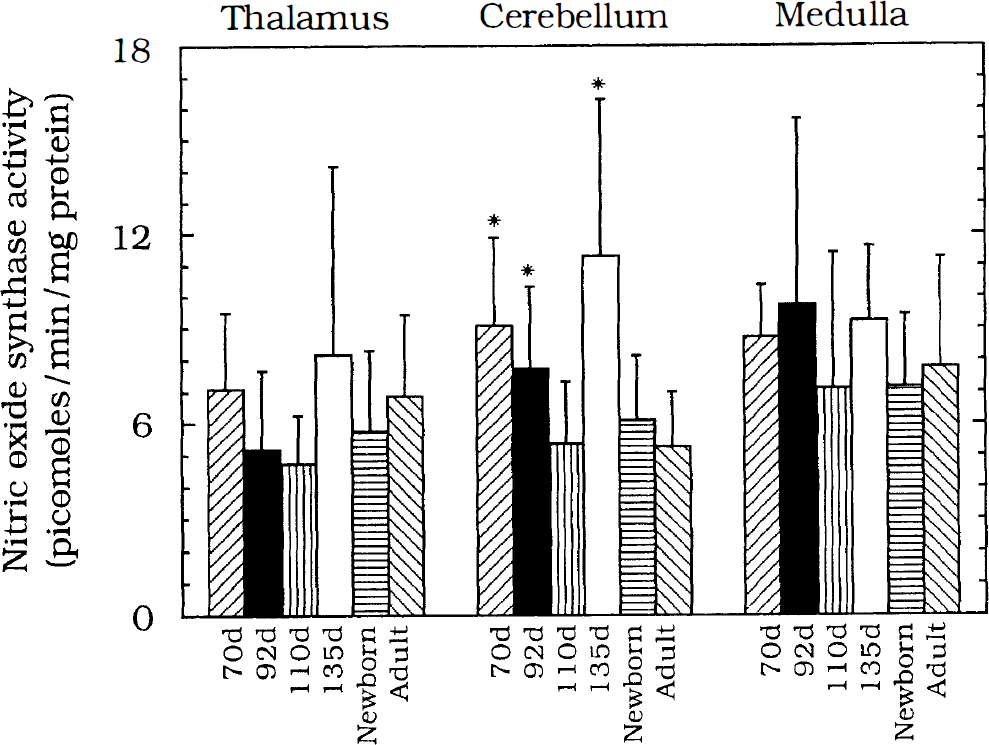
NOS activity (picomoles · min–1 · mg protein–1) in the thalamus, medulla, and cerebellum for the six developmental ages tested: 70 days gestation, left-hatched (n = 6); 92 days gestation, solid bars (n = 8); 110 days gestation, vertical stripes (n = 7); 135 days gestation, open bars (n = 6); newborn, horizontal stripes (n = 6); and adult, right-hatched (n = 10). No developmental differences were seen in the thalamus or medulla. NOS activity in the cerebellum is greatest prenatally. Activity at 70, 92, and 135 days gestation is greater than adult levels (*, p < 0.05 vs. adult).
Cerebral blood flow
Because the greatest increase in NOS activity in cortex occurred between 92 and 135 days, we measured the effect of NOS inhibition on CBF at these ages. A dose of 60 mg/kg of L-NAME administered intravenously to two 92-day-old fetal sheep resulted in NOS activity of 0.37 pmole/min/mg protein in the parietal cortex, representing 82% inhibition. This dose in four 135-day-old fetuses resulted in NOS activity of 1.27 pmole/min/mg protein, representing 89% inhibition.
Cerebral blood flow was measured in utero in six unanesthetized 93-day-old fetuses (range, 92–94 days) and seven unanesthetized 133-day-old fetuses (range, 132–135 days). Within each age group, blood flow increased in a caudal-to-rostral direction along the neuroaxis (Fig. 3). In 133-day-old fetuses, blood flow to the cerebrum, thalamus, cerebellum, and medulla was 298%, 276%, 233%, and 260%, respectively, of blood flow to the corresponding regions in 93-day-old fetuses. Likewise, CMRO2 in 133-day-old fetuses was 262% of that in 93-day-old fetuses. Administration of L-NAME did not alter CMRO2, but it decreased blood flow to the cerebrum by 27% in 93-day-old fetuses and by 25% in 133-day-old fetuses and to the cerebellum by 30% in 93-day-old fetuses and by 31% in 133-day-old fetuses (Fig. 3). Blood flow to the thalamus and medulla decreased by 31% and 39%, respectively, in 133-day-old fetuses, but it was not significantly changed in 93-day-old fetuses (Fig. 3). In response to administration of L-NAME, mean arterial pressure increased from 43 ± 4 to 53 ± 4 mm Hg in 93-day-old fetuses and from 52 ± 4 to 72 ± 4 mm Hg in 133-day-old fetuses. Because blood flow did not increase in any brain region in response to L-NAME, the substantial increase in fetal arterial pressure indicates that L-NAME increased vascular resistance throughout the cerebrovasculature.
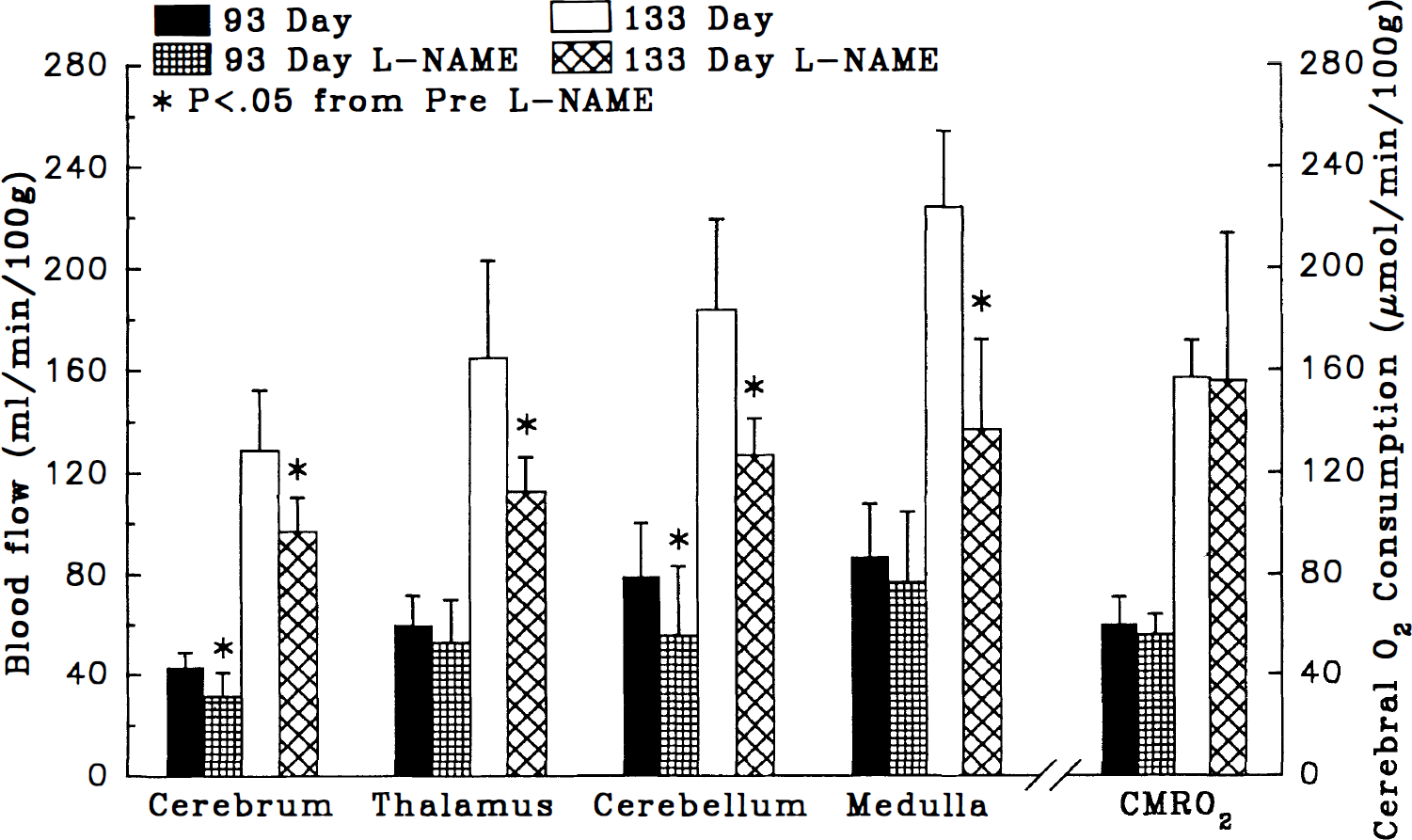
Cerebral blood flow (ml · min–1 · 100 g–1) and cerebral O2 consumption (μmol · min–1 · 100 g–1) for fetuses at 93 (n = 6) and 133 (n = 7) days gestation before and after administration of 60 mg/kg of L-NAME. L-NAME significantly depressed basal CBF in cerebrum, thalamus, cerebellum, and medulla in near-term fetuses and decreased CBF in cerebrum and cerebellum in the midgestation fetuses (*, p < 0.05 vs. pre-L-NAME baseline). CMRO2 triples from midgestation to near term but is unaffected by the administration of L-NAME.
DISCUSSION
These results demonstrate that NOS catalytic activity is developmentally and regionally regulated in the sheep brain. Levels of NOS activity equaled or exceeded adult values by 135 days gestation in all brain regions examined, but maturation to adult levels proceeded most rapidly in the more caudal structures. Maturation to levels equal to or greater than adult levels was present by 70 days, 0.48 of completed gestation, in the medulla, cerebellum, and thalamus. This finding was in contrast to findings in the cortex and caudate, where adult levels were generally reached by only 135 days. Maturation was intermediate in the hippocampus, where mature levels were attained by 92 days.
In general, there were similarities in the regional development of NOS activity with regional neuronal development and metabolic activity in fetal sheep. Barlow (1969) recognized neuronal differentiation in brain-stem nuclei by 60 days gestation, in cerebellum by 60–80 days, and in cerebral cortex by 100–110 days. The intensity of histochemical staining of cytochrome oxidase paralleled neuronal differentiation. Regional glucose metabolism begins to increase in auditory and vestibular brain-stem nuclei by ∼90 days (Abrams et al., 1984). In the thalamus and hippocampus, peak glucose consumption is attained at 114 days, whereas in cerebral cortex and caudate nucleus, peak glucose consumption is attained after birth (Abrams et al., 1984). Because glucose consumption parallels neuronal activity in the mature brain, the regional development of NOS activity may be regulated by the amount of neuronal activity.
It should be noted that we normalized NOS activity by protein concentration. However, the amount of protein per DNA increases abruptly between 80 and 100 days in both cerebrum and brain stem (McIntosh et al., 1979), and water content begins to decrease after 106 days gestation (Bradbury et al., 1972). Therefore, in regions where NOS activity per milligram of protein reaches a plateau in midgestation, activity per unit volume of tissue may still be increasing during development. Nitric oxide has been shown to participate in the maintenance of basal CBF in several species (Northington et al., 1992; Tanaka et al., 1993; Dirnagl et al., 1994; Faraci and Brian, 1994). A few studies have extended this observation to the newborn (Busija et al., 1990; Northington et al., 1995). These findings suggest that development of NOS activity could be important in the pattern of development of basal levels of CBF.
We examined the effect of NOS inhibition on regional CBF in the 93- and 133-day-old fetuses. Because these ages bracketed the up to fourfold increase in cortical NOS activity and the twofold increase in basal CBF, we anticipated that inhibition of NOS would reduce blood flow to a greater extent at 133 days gestation. However, we found that NOS inhibition caused significant reduction in blood flow to the cerebrum and cerebellum at 93 days gestation. These results indicate that NO is produced in sufficient quantities to affect cerebrovascular tone by ≤0.64 of full gestation in fetal sheep. Moreover, the percentage reduction in blood flow was similar at 93 and 133 days gestation. This observation might be anticipated in the cerebellum, where NOS activity was similar at these ages, but not in the cerebrum, where regional NOS activity increased as much as fourfold during this time period.
There are several possible explanations for the lack of an enhanced effect of L-NAME on basal CBF in cortex at term. First, the relative contributions of endothelial-versus neuronal-derived NO to basal cerebrovascular tone is unknown, and tissue NOS activity does not differentiate endothelial- and neuronal-based NOS. Immunocytochemistry indicates that expression of endothelial NOS protein varies very little during this period in any area of the sheep brain (Northington et al., 1996). The present cerebrovascular findings taken together with the consistent expression of endothelial NOS protein throughout this period of gestation suggest that endothelial-derived NO makes a significant contribution to basal cerebrovascular tone, but endothelial NOS accounts for only a small fraction of total NOS activity. Second, catalytic activity of NOS measured in homogenate with excess calcium and NADPH represents near maximum activity. Activity in vivo may not correlate with maximum activity measured in vitro.
Third, catalytic activity was measured in the supernatant, the supposed site of most of the neuronal-derived activity (Forstermann et al., 1991a, 1996). It is thought that ≥95% of the activity attributable to endothelial NOS activity is found in the particulate fraction (Forstermann et al., 1991a), and this fraction was not measured in these experiments. However, the present study did not use high-speed or differential centrifugation to separate the supernatant and particulate fractions; therefore, some of the reported activity is possibly due to endothelial NOS activity. Because some NOS activity has been found in the pellet resulting from differential centrifugation (Forstermann et al., 1991a), it is likely that the NOS activity reported here represents an underestimation of total brain NOS activity and an even more significant underestimation of endothelial NOS activity. Some cells may also express immunologic NOS during development (Galea et al., 1995; Bertini et al., 1996). Fourth, cerebrovascular architecture and smooth muscle tone is different at 93 days, giving rise to a greater vascular resistance. It is possible that basal levels of NO actually are less at 93 days, as suggested by these data, but that elimination of that smaller amount of NO exerts a relatively larger effect on blood flow because of inherent differences in architecture and smooth-muscle tone. Between 70 days gestation and term, cerebrovascular architecture changes from a primarily radial arrangement to a complex branching arrangement (Northington et al., 1996). In addition, differences in smooth-muscle tone at 93 and 133 days gestation are evident from the present data, as reflected in differences in basal cerebrovascular resistance. Fifth, the twofold increase in CBF between 93 and 133 days gestation was accompanied by a twofold increase in CMRO2. It is possible that the higher metabolic demand in the 133-day-old fetus results in a greater release of other vasodilatory mediators, which act to limit vasoconstriction when basal NO production is inhibited.
The finding of a postnatal decline in NOS activity in the cerebellum is not without precedent. Total cGMP release in rat cerebellar slices in response to both NMDA and non-NMDA receptor agonists is greatest between postnatal day 8 and day 14 and the relative efficacy of the various glutamate receptor agonists to elicit NO production changes markedly from day 8 to adulthood (Southam et al., 1991). Transient expression of NOS activity is also seen in a subpopulation of ventral horn cells in the spinal column and is thought to contribute to motor neuron differentiation and be a mechanism contributing to activity-dependent plasticity in the central nervous system during development (Kalb and Agostini, 1993; Wu et al., 1994). Furthermore, transient expression of neuronal NOS has been identified immunohistochemically in the cortical plate, olfactory epithelium, and sensory ganglia of the rat, while other regions display stable and persistent labeling after the period of cell division (Bredt and Snyder, 1994). Similar observations, of transient neuronal NOS immunoreactivity, have been made in the cortical subplate and subthalamic nucleus in developing sheep (Northington et al., 1996).
Techniques similar to those in the present study have been used to describe regional and developmental differences in neuronal NOS activity in the rat with somewhat different results than we report here. In the adult rat, cerebellar levels of NOS activity exceed those in the cortex (Ma et al., 1991). In the present study, this was true for all cortical regions and the caudate at the earliest gestational age examined, 70 days. At 92 days of gestation, cerebellar activity still exceeded that in the parietal and occipital lobes. In contrast, after birth cortical and caudate NOS activity exceeded that found in the cerebellum. Regional differences similar to what we report have been seen in bovine brain (Ohshima et al., 1992). Despite the different regional hierarchy of NOS activity in the adult rat, a developmental increase in NOS activity is seen in the rat. Levels of NOS activity in the adult rat cerebrum are two times that seen at 19 days of gestation (Ma et al., 1991). Immunohistochemical studies have also been used to examine regional differences in NOS protein expression in the rat (Bredt et al., 1990; Forstermann et al., 1990; Vincent and Kimura, 1992; Bredt and Snyder, 1994). Intense staining of the cerebellum, colliculi, and olfactory bulb, with less intense staining of the cortex and hippocampus, was reported with the initial use of a purified antibody to neuronal NOS (Bredt et al., 1990). An extensive survey of NADPH-diaphorase staining in rat brain documents the presence of NADPH-diaphorase-positive neurons in each of the areas we examined in the sheep (Vincent and Kimura, 1992). Therefore, although relative regional levels of activity appear to be species specific, widespread distribution of NOS protein is common among species.
In summary, maturation of the portion of NOS activity measured in this study proceeds in a caudal to rostral direction along the neuroaxis in parallel with the pattern of neuronal maturation and metabolism (Barlow, 1969; Bernhard et al., 1967; Abrams et al., 1984; Gleason et al., 1989). Furthermore, in sheep, a species with longer and more complex brain development, cortical levels of NOS activity exceed those in the more caudal regions at maturity, a finding not observed in the rat. These overall large increases in cortical NOS activity between midgestation and late gestation parallel large increases in CBF. However, the amount of NO production is already adequate at 0.64 of full gestation to exert an effect of CBF in utero that is unchanged in magnitude throughout the remainder of gestation.
Footnotes
Acknowledgment:
The authors thank Megan Williams, Ophelia Garalde, Ann Biser-Rohrbaugh, and Debra Flock for expert technical assistance with this project and Karen Lingerman for assistance with manuscript preparation. This study was supported in part by USPHS NIH grant NS20020 and an AHA Grant in Aid with funds provided by the Maryland Affiliate of the American Heart Association. Dr. Northington is supported by a Merck Clinician Scientist Award from Johns Hopkins University School of Medicine and by USPHS NIH grant NS1742-01.
