Abstract
Periventricular white matter (PVWM) injury is the leading cause of neurologic disability in survivors of prematurity. To address the role of ischemia in PVWM and cerebral cortical injury, we hypothesized that immaturity of spatially distal vascular ‘end zones’ or ‘border zones’ predisposes PVWM to greater decreases in cerebral blood flow (CBF) than more proximal structures. We quantified regional CBF with fluorescently labeled microspheres in 0.65 gestation fetal sheep in histopathologically defined three-dimensional regions by post hoc digital dissection and coregistration algorithms. Basal flow in PVWM was significantly lower than in gyral white matter and cortex, but was equivalent in superficial, middle, and deep PVWM. Absolute and relative CBF (expressed as percentage of basal) did not differ significantly during ischemia or reperfusion between PVWM, gyral white matter, or cortex. Moreover, CBF during ischemia-reperfusion was equivalent in three adjacent PVWM levels and was not consistent with the magnitude of severity of PVWM injury, defined by TUNEL (terminal deoxynucleotidyltransferase-mediated dUPT nick end labeling) staining. However, the magnitude of ischemia was predicted by the severity of discrete cortical lesions. Hence, unlike cerebral cortex, unique CBF disturbances did not account for the distribution of PVWM injury. Previously defined cellular maturational factors, thus, appear to have a greater influence on PVWM vulnerability to ischemic injury than the presence of immature vascular boundary zones.
Introduction
Periventricular white matter injury (PWMI) is the major form of brain injury and the leading cause of chronic neurologic disability in survivors of premature birth (Volpe, 2000). It occurs in up to 25% of preterm survivors who acquire the permanent motor deficits of cerebral palsy (Miller et al, 2005) and in 25% to 50% who develop cognitive and learning disabilities by school age (Litt et al, 2005). Increasing evidence supports that PWMI is initiated in critically ill neonates by cerebral hypoxia-ischemia and accompanying oxidative stress that targets susceptible late oligodendrocyte progenitors (preOLs) (Back, 2006). The spatial distribution of PWMI is related to the relative density of susceptible preOLs and more resistant myelinating oligoden-droyctes (Riddle et al, 2006). Periventricular white matter injury may also occur in association with cerebral gray matter injury of unclear etiology that manifests as a reduction in the volume of cortical and subcortical structures (Back, 2006).
The extent to which spatial heterogeneity of cerebral vascular supply contributes to regional predilection for PWMI remains controversial. It has been proposed that immaturity of microvascular end zones and border zones predisposes periventricular white matter (PVWM) to hypoperfusion in the setting of systemic hypotension, a common clinical scenario for critically ill premature neonates (Volpe, 2001). Systemic hypotension may also pose particular risks for the premature infant, because of a lack of cerebral autoregulation (Greisen, 2005; Tsuji et al, 2000; Volpe, 2001). In the setting of a pressure-passive circulation, immature vascular zones may coincide with more severe ischemic injury. However, anatomic support for vascular zones remains inconclusive (Nelson et al, 1991), and, unlike term fetal sheep (Ashwal et al, 1980), virtually no physiologic studies of cerebral blood flow (CBF) have been performed in preterm sheep. Prior studies have not established a relationship between PWMI and perturbations in periventricular flow. Commonly employed measures of global CBF lack the spatial resolution to define cerebral hemodynamics in human PVWM. In addition, small fetal and neonatal animal models have been uninformative because of a paucity of PVWM, a propensity for mixed gray and white matter injury, and the technical limitations of invasive CBF measurements (Back et al, 2006b).
To circumvent these limitations, we developed an in utero preparation in the immature instrumented sheep fetus (0.65 gestation). We used fluorescent microspheres to quantify CBF in situ in anatomically defined three-dimensional (3D) regions by means of post hoc digital dissection. Global cerebral ischemia-reperfusion was initiated when normal CBF was interrupted by carotid occlusion. Ischemia, which was severe but not complete, reduced global CBF by ˜80% to 90% and generated graded injury to frontal and parietal PVWM, two regions of predilection for human PWMI (Riddle et al, 2006). We tested the hypothesis that spatially distinct vascular ‘end zones’ or ‘border zones’ exist that predispose more distal regions, such as the PVWM, to be more susceptible to decreases in blood flow than more proximal regions, such as cerebral cortex. We determined if physiologically distinct zones of CBF were present in PVWM during conditions of basal, ischemia or reperfusion flow. We sought evidence that during global cerebral hypoperfusion, CBF in the deep PVWM decreased proportionally more than that in more superficial regions of the PVWM, gyral white matter (GWM), or cerebral cortex. Lastly, we determined if the magnitude of ischemia to prospectively defined gray or white matter lesions was predicted by the severity of injury (quantified by TUNEL (terminal deoxynucleotidyltransferase-mediated dUPT nick end labeling). We identified distinct regional heterogeneity in basal CBF between cerebral gray and white matter. However, neither during ischemia nor reperfusion was the PVWM subject to distinct differences in CBF, nor were there more pronounced CBF disturbances identified in the regions of the PVWM that were more susceptible to injury. In contrast, small discrete cerebral cortical lesions were identified whose magnitude predicted the severity of ischemia. Hence, regional heterogeneity of CBF disturbances appears to contribute more directly to the severity of cerebral cortical than PWMI.
Materials and methods
Surgical Procedure
An instrumented fetal cerebral hypoperfusion preparation (Reddy et al, 1998) and the general surgical procedures (Chao et al, 1991) were modified as recently described (Riddle et al, 2006). Time-bred sheep of mixed western breed (91 to 92 days gestation; term, 145 days) were operated with sterile technique under general anesthesia (1% halothane in O2 and N2O). Vinyl catheters were placed in the amniotic cavity and into either a fetal axillary artery or nonocclusively into one carotid artery and a fetal hind-limb vein. Hydraulic occluders (silastic) were placed on each carotid artery. To confine the cerebral blood supply to the carotid arteries, the vertebro-occipital anastomoses were ligated bilaterally. These anastomoses connect the vertebral arteries with the external carotid arteries that are fed by the brachiocephalic artery (Baldwin and Bell, 1963). After closure of the uterus, the lines were externalized and 1 million units of penicillin G was infused into the amniotic cavity. Postoperative housing was on clean dry bedding in an individual cage with access to food and water ad libitum as well as with close visual contact with other sheep.
Electroencephalogram Recording
One or two pairs of electroencephalogram (EEG) electrodes (AS633-5SSF, Cooner Wire, Chatsworth, CA, USA) were secured on the dura over the parasagittal parietal cortex (5 and 10 mm anterior to bregma and 5 mm lateral) with a reference electrode attached over the occiput. The electrodes were connected to high-impedance amplifiers to provide a filtered input to a Stellate Systems (Montreal, QC, Canada) digital EEG recording and analysis system. We recorded two to five channels of continuous EEG from 10 animals for up to 16 h before and after global ischemia. Seizure detection was performed by visual analysis and confirmed with a newborn seizure detection algorithm (Stellate Systems).
Blood Analysis
One mL blood samples, taken anaerobically from the fetal axillary artery, were analyzed for arterial paH, PaO2, PaCO2 (corrected to 39°C; IL Synthesis 10 pH/Blood Gas Analyzer; Instrumentation Laboratory, Lexington, MA, USA), hemoglobin content (Thb), arterial oxygen content (CaO2), arterial oxygen saturation (SatO2; IL 682 Co-Oximeter; Instrumentation Laboratory), and hematocrit (Hct; capillary microfuge). Fetuses were studied only if they showed normal fetal oxygenation, defined as >6mL O2/100mL blood, at the time of the cerebral hypoperfusion studies.
Cerebral Hypoperfusion Studies
Seven animals were studied on the second or third postoperative day. Pressure transducers and a strip-chart recorder (TA 6000; Gould Instruments, Valley View, OH, USA) recorded mean arterial blood pressure in the fetal artery relative to amniotic fluid pressure. Fetal heart rate was calculated from triplicate measurements of the arterial pressure pulse intervals over a continuous recording of >20 sees. Under basal conditions, physiologic parameters and hemodynamic values (heart rate, mean arterial blood pressure, PaO2, SatO2 and CaO2) did not differ significantly between control and experimental groups and were similar to those previously reported (Riddle et al, 2006). Sustained cerebral hypoperfusion of 37 or 45mins duration was initiated by carotid artery occlusion after inflation of the carotid occluders. All occlusion CBF studies were performed at ˜30mins of ischemia. Cerebral reperfusion was established by deflation of the occluder and was studied at 15 and 60mins after blood flow restoration.
Microsphere Injection Protocol
Fetal brain blood flow was measured spatially by the fluorescent microsphere distribution and reference sample method (Bernard et al, 2000). Fluorescent microspheres with four different colors (15 μm diameter; F-17047, F-17048, F-17048, and F-17050; Molecular Probes, Eugene, OR, USA) had the following peak excitation and emission: green (450/480), yellow (515/534), red (580/605), and scarlet (650/685). Approximately 3 × 106 microspheres suspended in 1mL of saline with 0.05% Tween were sonicated and then injected over 30 sees into the fetal hind limb vein followed by a 2mL flush with saline. Starting just before and continuing 2mins after each injection, a reference blood sample was drawn at 0.75 mL/min into a syringe mounted in a syringe pump (Harvard Apparatus Co., Dover, MA, USA).
Tissue Handling
At 24 h after the initiation of cerebral reperfusion, the ewe was given an intravenous injection of Euthasol (Virbac Inc., Ft Worth, TX, USA) to euthanize both ewe and fetus. Fetal brains were collected and immersion fixed at 4°C in 4% paraformaldehyde in 0.1mol/L phosphate buffer, pH 7.4, for 48 h and then stored in phosphate-buffered saline. Brains were subsequently immersed in 20% sucrose until they sank and then were rapidly frozen in OCT (a cryostat embedding compound).
Acquisition of Microsphere Distributions with the Imaging Cryomicrotome
Fluorescent microsphere detection and localization were performed with the imaging cryomicrotome (ICM) (Barlow Scientific Inc., Olympia, WA, USA) (Kelly et al, 2000), which combines fluorescence digital imaging of frozen tissue and serial cryostat sectioning to determine the 3D location of each microsphere. The accuracy of the ICM approach for measurement of regional CBF versus radioactive and standard fluorescence microsphere methods has been previously validated (Bernard et al, 2000). The entire frozen fetal brain containing microspheres was mounted in the ICM and serially sectioned to yield ˜1,000 38-μm-thick sections. Fluorescence images of the microspheres in the tissue block face were acquired at 25 μm resolution. At regular intervals, adjacent sections were retrieved from the ICM for histologic analysis. Fluorescent microspheres in reference blood samples were counted so that the distribution of blood flow could be related to absolute blood flow for each color. A hole drilled into a block of frozen OCT was filled with blood that had been sonicated to disperse microspheres and the sample was frozen rapidly. An aliquot of each reference sample was serially sectioned and imaged as for the tissue samples.
Image Analysis and Measurement of Regional Blood Flow
Imaging cryomicrotome-acquired fluorescence images were preprocessed to correct for variations in illumination intensity and detector response. The images were analyzed as previously described to distinguish microspheres from background tissue fluorescence and to determine the 3D location of each microsphere (Bernard et al, 2000). Unbiased in situ measurements of blood flow were made in frontal and parietal regions in a blinded manner by digital dissection of anatomically defined regions (Gluckman and Parsons, 1983). Imaging cryomicrotome tissue block face images were imported to Amira +3D image analysis software (TGS, San Diego, CA, USA). All the regions were sampled in the coronal plane and were contained within a region that extended 4,300 μm posterior to the genu of the corpus callosum (frontal blood flow) or posterior to the level of the ventral thalamic nucleus, tail of the caudate, and mammillary bodies (parietal blood flow). Analyzed within the frontal region were the cerebral cortex, GWM, body of the caudate nucleus, head of the caudate nucleus, PVWM, corpus callosum, and subventricular zone (SVZ), whereas the parietal region contained the cortex, GWM, PVWM, hippocampus, and thalamus.
Quantification of TUNEL-Labeled Nuclei
We recently showed that counts of TUNEL-labeled nuclei and 04 antibody-labeled degenerating cells were equivalent to quantify cell death in PVWM (Riddle et al, 2006). TUNEL staining was a sensitive and specific marker of total cell degeneration in fetal ovine PVWM. TUNEL-labeled nuclei were visualized in tissue sections obtained from six animals and counterstained with methyl green to delineate anatomical boundaries. As shown in Figure 1A, digitized images of medial (yellow) and lateral (violet) cortex, medial (blue) and lateral (brown) GWM, and PVWM (green) were acquired (Openlab 4.0.2; Improvision, Boston, MA, USA) using a motorized x-y stage mounted on an inverted microscope (Leica DMIRE2) equipped with a Hammamatsu Orca ER cooled CCD camera. Images were stitched together as a montage with ImageJ software, which resulted in large-scale, high-resolution, 2D images. Each montage comprised a range of 12 to 60 fields acquired with a × 10 objective. Within each montage, ROIs (regions of interest) (2.1 ± 0.2 mm2) TUNEL-labeled nuclei were selected. These ROIs were confirmed to localize to a minimum of two near-adjacent sections (30 μm) from each of two frontal levels separated by ˜1,500 μm. Regions of interest were imported into Openlab, and the mean density of TUNEL-labeled nuclei was determined using image-threshold analysis software (Improvision; Open Lab) as described (Back et al, 2006a). Within the cerebral white matter (PVWM or GWM), ROIs were defined as TUNELhigh (≤10 labeled nuclei/mm2) or TUNELlow (<10 labeled nuclei/mm2). Cortical ROIs were defined as TUNELhigh (≤ 20 labeled nuclei/mm2) or TUNELlow (<20 labeled nuclei/mm2). In general, the density of nuclei in TUNELlow lesions was very similar to normal control lesions in which degeneration arises from normal programmed cell death (Falkowski et al, 2002). The animals subjected to ischemia for 37 or 45mins were grouped in subsequent analyses because discrete lesions had a similar range of damage that showed no significant differences in mean TUNEL density (F(1,46) = 0.774, P = 0.4).
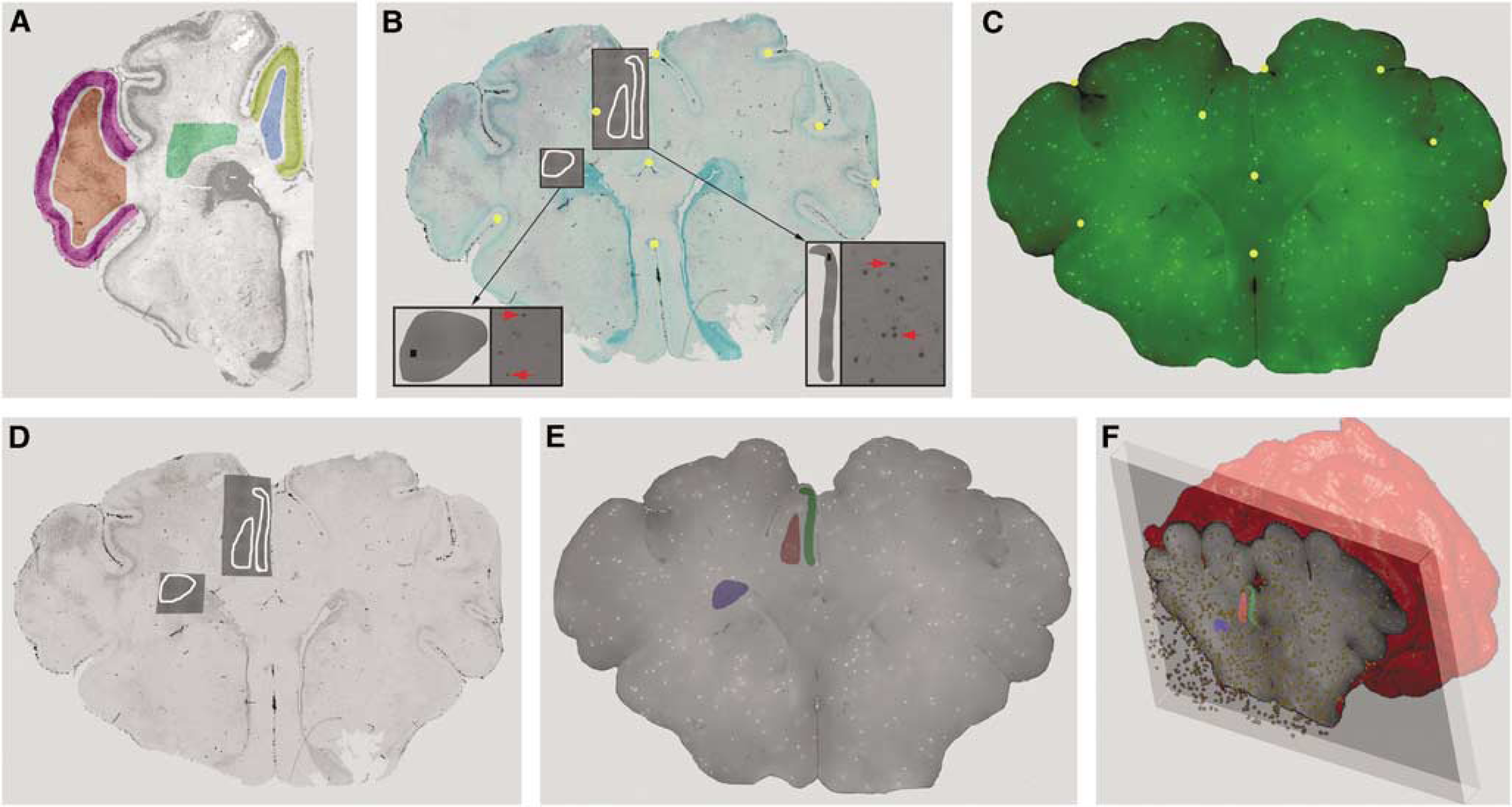
Our approach for 3D coregistration and analysis of CBF and cerebral injury data sets acquired from the same brain. (
Coregistration of Damage Assessment with Blood Flow
Individual ROIs of TUNEL-labeled regions were superimposed onto and linked with the low-magnification methyl green image (Figure 1B) and then imported into a commercial 3D image viewer (Amira). Next, 30 to 35 fiduciary landmarks (gold spheres in Figure 1B) were matched with the corresponding ICM images (Figure 1C) for which the unique 3D location of each green microsphere was known. Image ID was, thus, digitally ‘warped’ to minimize residual spatial error between the landmarks in the blood flow image (Figure 1C), thereby precisely coregistering both image data sets. The TUNEL-defined ROIs were then identically warped and ROIs of focal damage superimposed onto a 3D set of ICM images (Figure 1E). Amira scripts were next executed to count the number of microspheres in these damaged and undamaged ROIs in a volume defined by a depth of 1,500 μm (Figure 1F). Cerebral blood flow in the ROIs was quantified as mL per min per 100 g, thus allowing for comparison between CBF and TUNEL density in various regions under basal, occlusion, and reperfusion conditions (Figure 1).
General Statistical Analysis
Data analysis was performed using SPSS 12.0 statistical software. Repeated measures analyses of variance were used for the analysis of differences between independent variables with a time-dependent/repeated variable (i.e., blood flow measures taken in the same subjects/groups repeated over time). One-way analysis of variance and independent samples t-tests were performed to probe simple effects resulting from repeated measures analyses of variance showing significance, as well as to compare differences between two or more independent variables. One-sample t-tests were also performed to test differences between single variables expressed as a percentage from a basal score; thus, 100 represents the deviation test value.
Statistical Analysis for Studies of Coregistration of Damage Assessment with Blood Flow
Because CBF measurements during ischemia were associated with relatively low numbers of microspheres relative to basal (Polissar et al, 2000), multiple TUNELhigh or TUNELlow regions within each animal were combined and averaged before analysis across animals. The averaging provided a single, more accurate value to represent CBF in a given ROI of an animal. The paired t-test was used to compare values paired within animals.
Results
Basal CBF in the PVWM is Significantly Lower than in Cortical or Subcortical Gray Matter
Frontal PVWM and parietal PVWM are two regions of particular predilection for PWMI in human preterm infants. We, thus, first addressed the hypothesis that the susceptibility of the PVWM to ischemia-reperfusion injury is related to vascular boundary zones that manifest as lower basal CBF relative to less vulnerable cortical and subcortical gray matter structures. To test this hypothesis, we quantified basal CBF by in situ analysis of fluorescent microsphere distributions in seven anatomically defined regions at the level of frontal PVWM and four regions at the level of parietal PVWM (Figure 2). Basal CBF in frontal PVWM was very similar to the adjacent corpus callosum and SVZ (Figure 3). Basal CBF was also similar between cortical and subcortical gray matter structures (i.e., cerebral cortex, putamen, caudate head, and caudate body). As a whole, frontal PVWM and adjacent regions (corpus callosum and SVZ) had significantly lower basal CBF compared with cortical and subcortical gray matter structures (P<0.001). At the parietal level, comparison between cortical and subcortical regions showed that basal CBF in thalamus was significantly higher than in cortex and hippocampus (P = 0.001). The CBF in the cortex and hippocampus was similar and significantly higher than parietal PVWM (P<0.001). Thus, CBF was higher in all cortical and subcortical gray matter regions relative to PVWM and adjacent structures during basal conditions, with the thalamus showing the greatest flow.
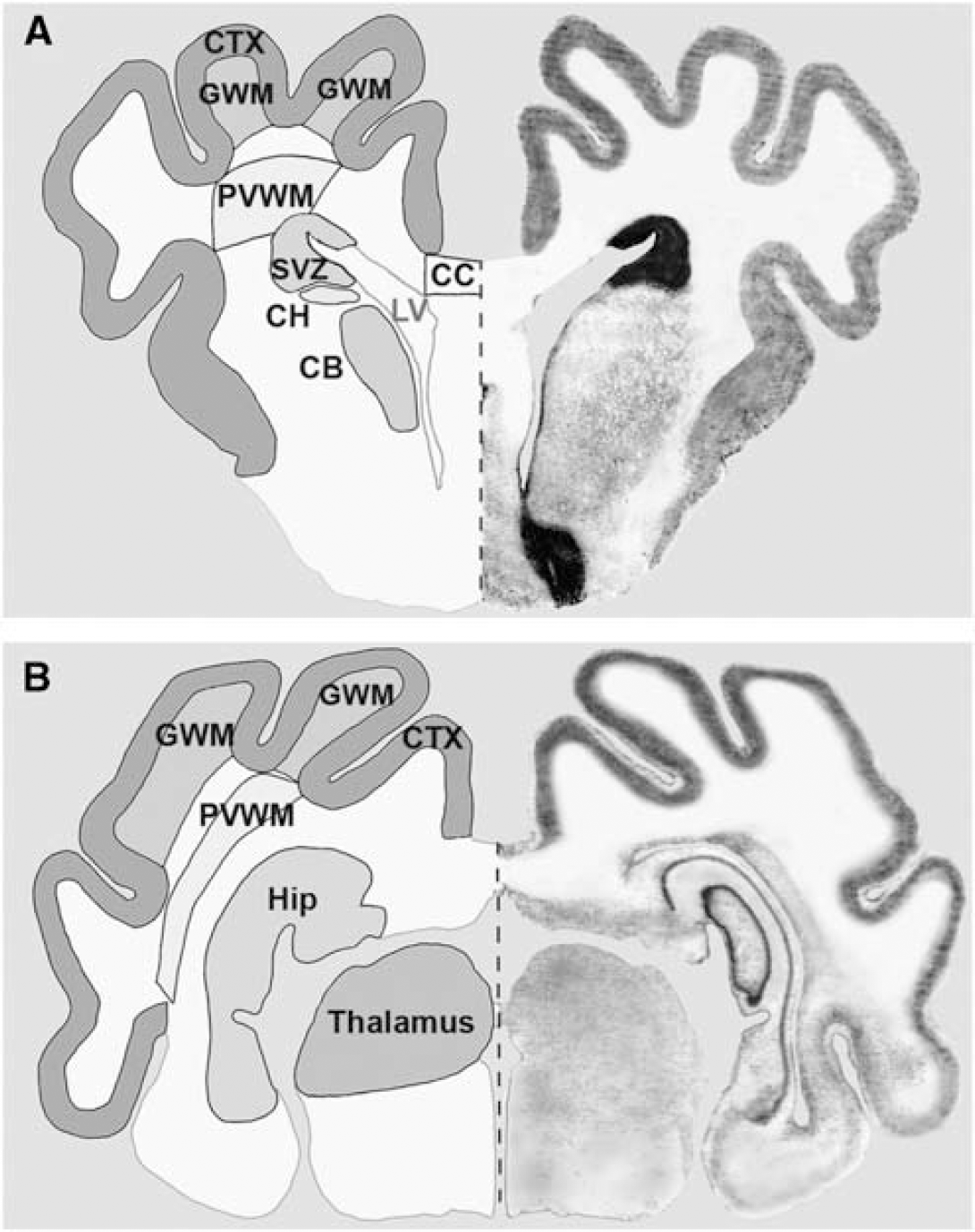
To determine distinct regional CBF values before, during, and at 15 and 60mins after ischemia (see Table 1), the brain was segmented into distinct frontal (
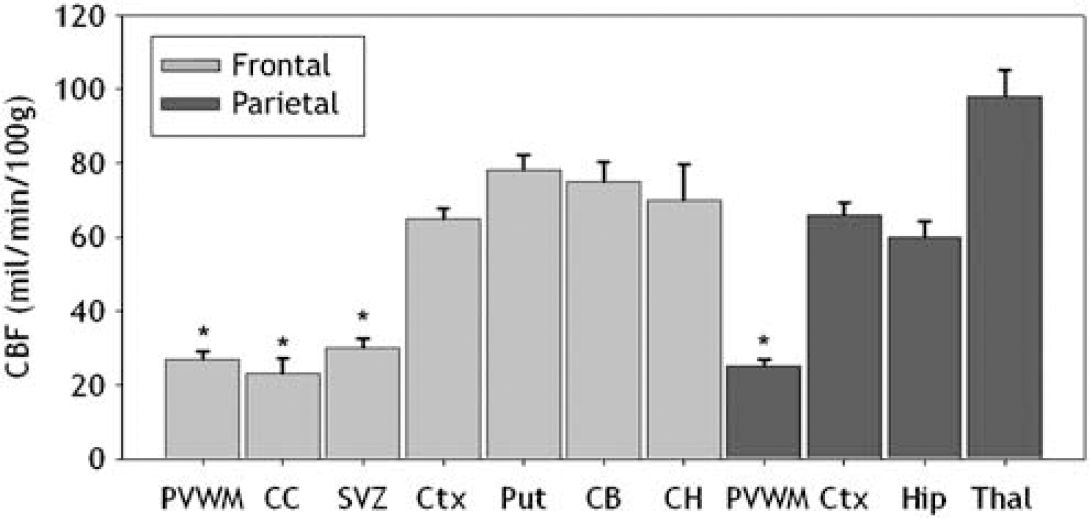
Basal CBF values (mean ± s.e.m.) are shown for seven frontal and four parietal regions. These areas included the periventricular white matter (PVWM), corpus callosum (CC), subventricular zone (SVZ), cortex (Ctx), putamen (Put), caudate body (CB), caudate head (CH), hippocampus (Hip), and thalamus (Thai). There were significant differences (*P<0.001) between deep cerebral structures (PVWM, SVZ, and CC) and all gray matter structures.
Absolute regional CBF values (mean ± s.e.m.) and CBF values expressed as a percentage of basal CBF during ischemia and reperfusion at 15 and 60 mins, as obtained for seven frontal and four parietal structures
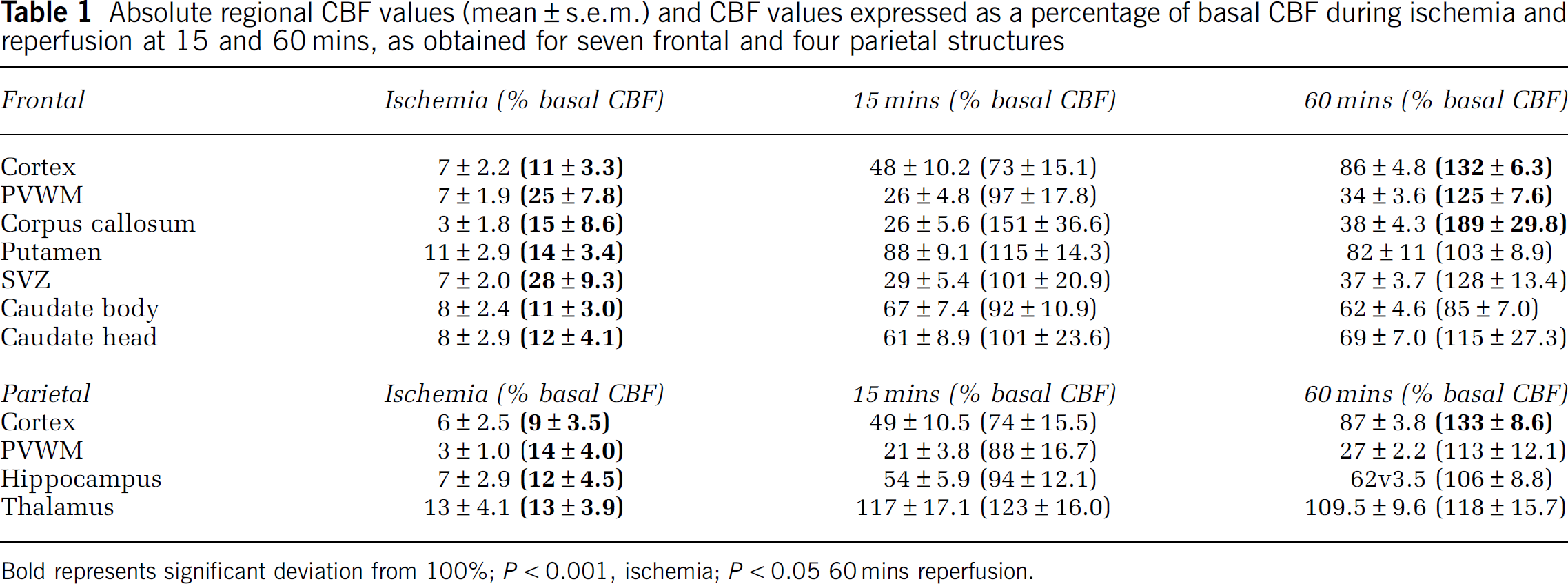
Bold represents significant deviation from 100%; P < 0.001, ischemia; P < 0.05 60mins reperfusion.
CBF during Ischemia and Reperfusion was Similar in PVWM and Cerebral Gray Matter Structures
We next hypothesized that during global ischemia, the PVWM sustains the most severe reduction in CBF and has the greatest hyperemia during early reperfusion. We first determined that in all regions, CBF during occlusion and reperfusion was independent of the duration of ischemia (37mins, n = 3; or 45 mins, n = 4). A 2 (occlusion duration: 37 versus 45mins) × 3 (CBF event: occlusion, 15 mins reperfusion or 60 mins reperfusion) × 7 (region) repeated measures analysis of variance showed no effect for the duration of ischemia (F(1,5) = 0.973) on frontal regional CBFs. A similar analysis of CBF in four parietal regions also showed no effect for the duration of ischemia (F(1,5) = 0.391). Hence, in subsequent analyses of both frontal and parietal regions, data were combined for animals subjected to 37 or 45 mins of ischemia. There were also no significant differences in CBF between medial and lateral cortex or between medial and lateral GWM (Figure 1F), and these respective regions were combined for all subsequent analyses.
We next determined mean absolute CBF and mean relative (percentage of basal) CBF values during occlusion and at 15 or 60 mins reperfusion for the same seven anatomically defined frontal and four parietal regions analyzed for basal CBF (Table 1). During ischemia, absolute CBF in all regions was reduced to a similar level. Similarly, there was no significant difference in relative CBF between frontal PVWM, which retained 25% of basal flow during the occlusion, and cortical CBF, which was reduced to 11% of basal flow. This was the case even though we recently found frontal PVWM to be more susceptible to acute damage than the cerebral cortex (Riddle et al, 2006). Hence, the heterogeneity in CBF under basal conditions was not observed during ischemia.
During early reperfusion (15 mins), frontal and parietal cerebral cortex showed a trend toward a modest hypoperfusion state that was not significantly different relative to the corresponding basal CBF. At 60 mins reperfusion, there was significant hyperemia in frontal cortex, PVWM, corpus callosum, and parietal cortex (one-sample t-tests showing deviation from 100%; P <0.05).
Electrographic Seizures are Delayed Beyond the Early Ischemia-Reperfusion Period
We hypothesized that electrographic seizures might provide an explanation for the hyperemia that was observed at 1 h of reperfusion. Figure 4 (EEC recordings) shows typical electrographic seizures that were seen in all of 10 animals that were recorded for 16 h after reperfusion. Seizures typically arose from a suppressed background and were brief (<10 sees). Rhythmic slow wave activity (upper panel; 1 to 1.5 Hz) or higher frequency activity (lower panel; 2 to 3.5 Hz) was the predominant pattern seen. Electrographic seizures were rarely seen within the first hour after ischemia (Figure 4, graph). The majority of electrographic seizures was observed between 2 and 6 h after ischemia and did not coincide with the first hour of reperfusion when moderate cortical hyperemia was observed.
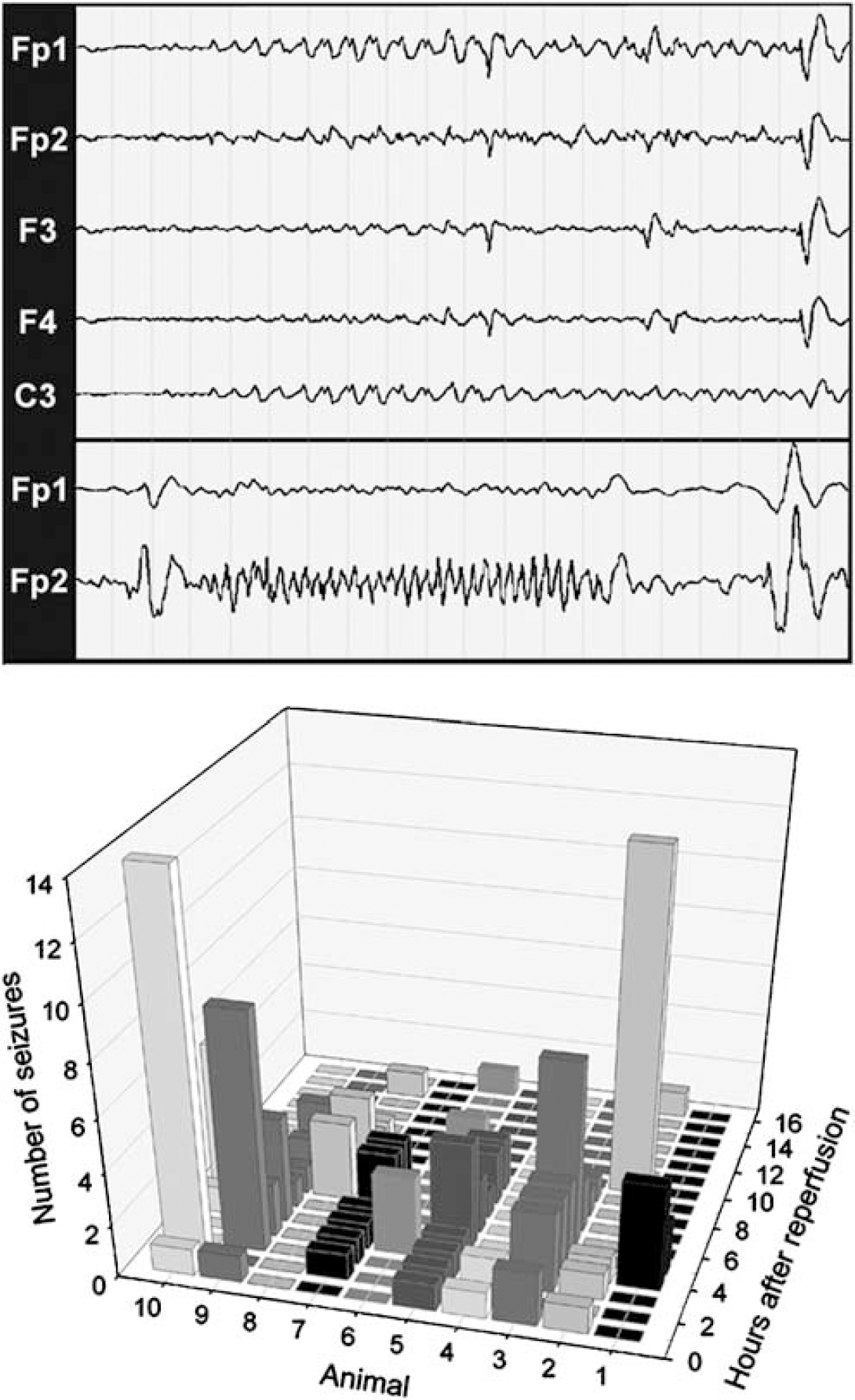
Electrographic seizures are delayed beyond the early ischemia-reperfusion period. Electroencephalogram tracings are from recordings performed during reperfusion. Representative electrographic seizures from different animals are shown in the upper and lower panels. The upper panel shows the initiation of a rhythmic slow wave (1 to 1.5 Hz) seizure of left hemispheric origin that arises from a suppressed background. These slow wave patterns are typical for seizures seen in human neonates (25 to 44 weeks gestation). Interestingly, they evolve in frequency and amplitude but remain focal, which may relate to the lack of mature myelination at 0.65 gestation. In the lower panel, a faster (2 to 3.5 Hz) seizure is shown that lasts for 12 sees. Higher frequency patterns coexist with slow wave seizures in preterm neonates. As shown graphically, electrographic seizures, recorded for a 16 h duration in 10 animals, were rare in the first hour after reperfusion. Between 2 and 6 h, there was a marked increase in seizure frequency in the majority of animals and then a diminishing number of events were seen in the last 10h. Scale for EEG recordings: 1 page = 20secs.
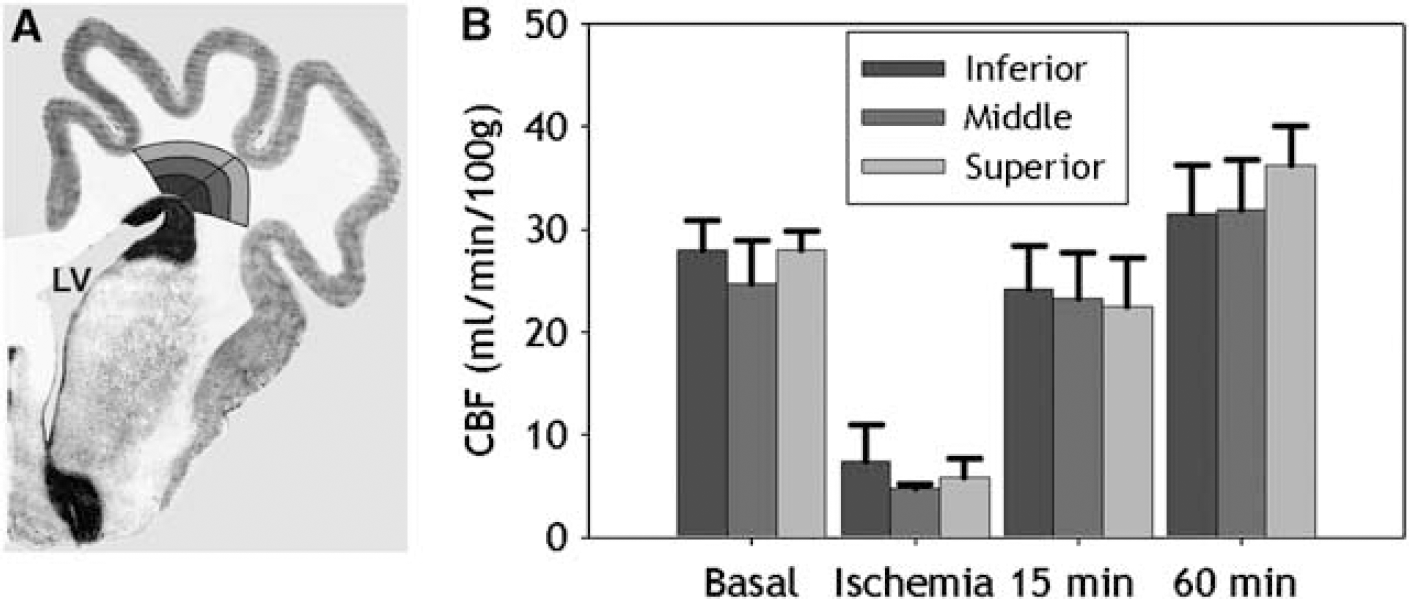
Gradients of CBF were not Present in PVWM
To further test the hypothesis that vascular boundary zones are present in the PVWM, we next determined if gradients of CBF were present between the deep and superficial PVWM. We quantified CBF during basal, ischemia, and reperfusion (15 and 60mins) conditions in frontal PVWM that was segmented into six regions (Figure 5A). The PVWM was divided into medial and lateral regions, which were further segmented into inferior, middle, and superficial zones. No differences in CBF were found between the medial and lateral frontal PVWM. Accordingly, data for the medial and lateral parts of each zone were grouped in subsequent analyses. Figure 5B shows that no significant differences in CBF were identified among the inferior, middle, and superior zones for basal, ischemia, and 15 and 60mins reperfusion conditions. There were, thus, no apparent gradients of CBF present in the frontal PVWM.
(
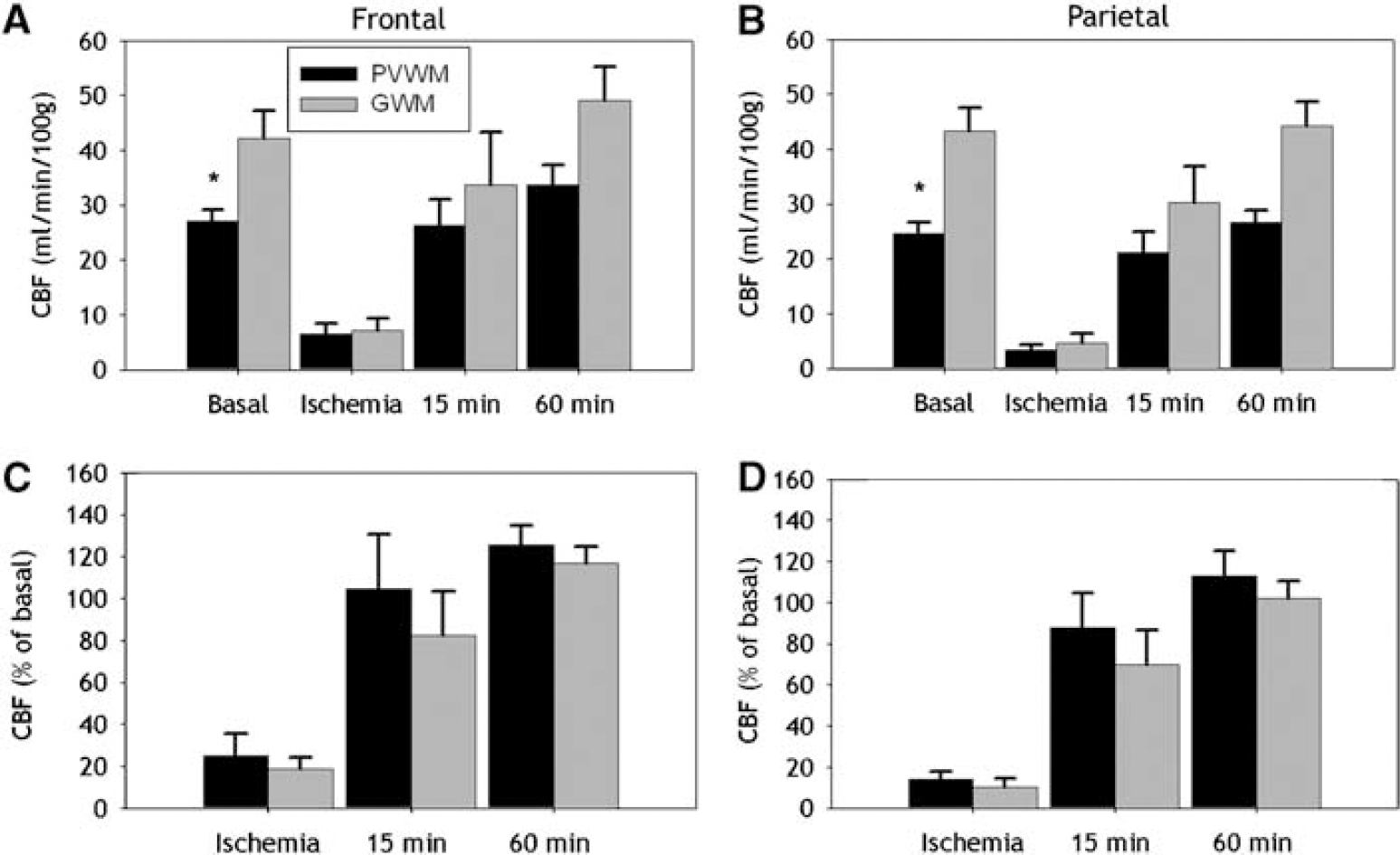
PVWM and GWM do not Differ in Absolute or Relative CBF
The cerebral white matter of the human premature infant is supplied by short penetrating arteries to the GWM and long penetrating arteries to the PVWM that contribute to apparent vascular end zones (Volpe, 2000). Thus, we next tested the hypothesis that during ischemia the PVWM will sustain more severe ischemia than the GWM. There were significant differences in absolute basal CBF between the PVWM and the GWM for both the frontal (Figure 6A; P <0.05) and parietal (Figure 6B; P < 0.001) levels. Absolute CBF in frontal (Figure 6A) and parietal (Figure 6B) PVWM and GWM showed no significant differences during ischemia or reperfusion (15 and 60mins). Similarly, there were no significant differences in relative CBF during ischemia or reperfusion between the PVWM and GWM for either the frontal (Figure 6C) or parietal (Figure 6D) levels. Hence, there were no differences in absolute or relative CBF between the PVWM and GWM during occlusion or at 15 or 60mins of reperfusion to support the presence of pathophysiologically significant gradients of flow between superficial and deep cerebral white matter.
(
The Magnitude of Histopathologically Defined White Matter Injury does not Predict CBF during Ischemia or Reperfusion
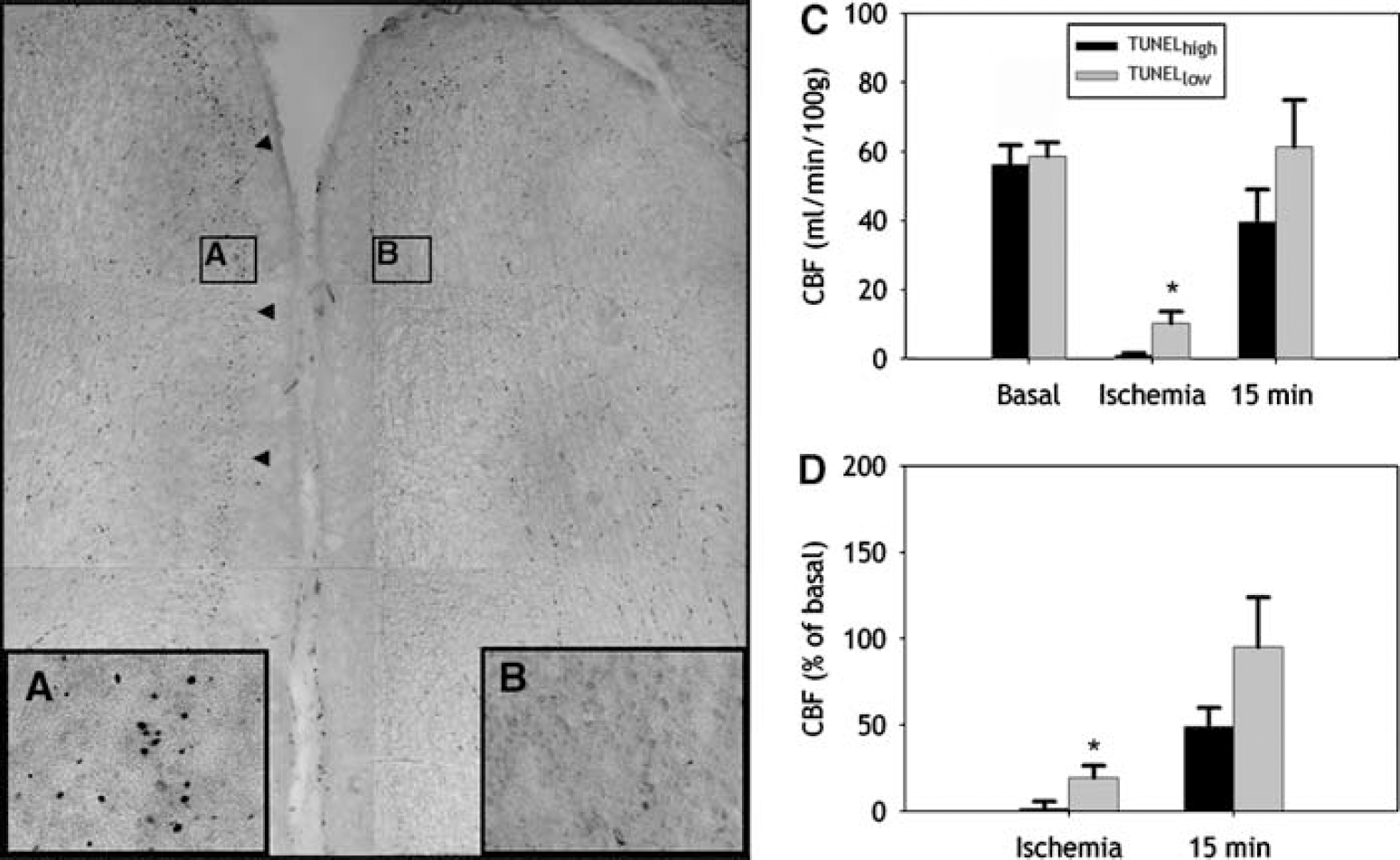
We next determined if the magnitude and distribution of cerebral white matter injury, defined by TUNEL staining, coincided with a greater reduction in flow in those regions that sustained greater injury. We hypothesized that regions of more pronounced white matter injury will show more severe ischemia than regions of minimal injury. To test this hypothesis, we segmented regions of histopathologically defined injury and coregistered these regions with the corresponding CBF data sets (see Materials and methods and Figure 1). TUNEL staining was quantified in 22 separate TUNELhigh (≤ 10 labeled nuclei/mm2) or 18 TUNELlow (< 10 labeled nuclei/mm2) lesions in frontal PVWM or GWM. Figures 7A and 7B show representative high-power images of TUNELhigh (A) and TUNELlow (B) ROIs from a portion of a digitized montage in the PVWM. There were no significant differences in absolute CBF during ischemia between TUNELhigh and TUNELlow regions in the PVWM (Figure 7C; P≤0.5; mean difference: 1.0 mL per min per 100 g; 95% confidence limits: +4.9 to −2.9). Similarly, there were no significant differences in absolute CBF for GWM (Figure 7D; P≤0.5). Also, there were no significant differences in CBF seen in PVWM and GWM at 15mins of reperfusion. Relative CBF in both PVWM (Figure 7E; P=0.5) and GWM (Figure 7F; P=0.5) was also not significantly different between TUNELhigh and TUNELlow regions. These data support that the distribution of cerebral white matter injury does not appear to be related to the magnitude of absolute or relative CBF during either ischemia or early reperfusion.
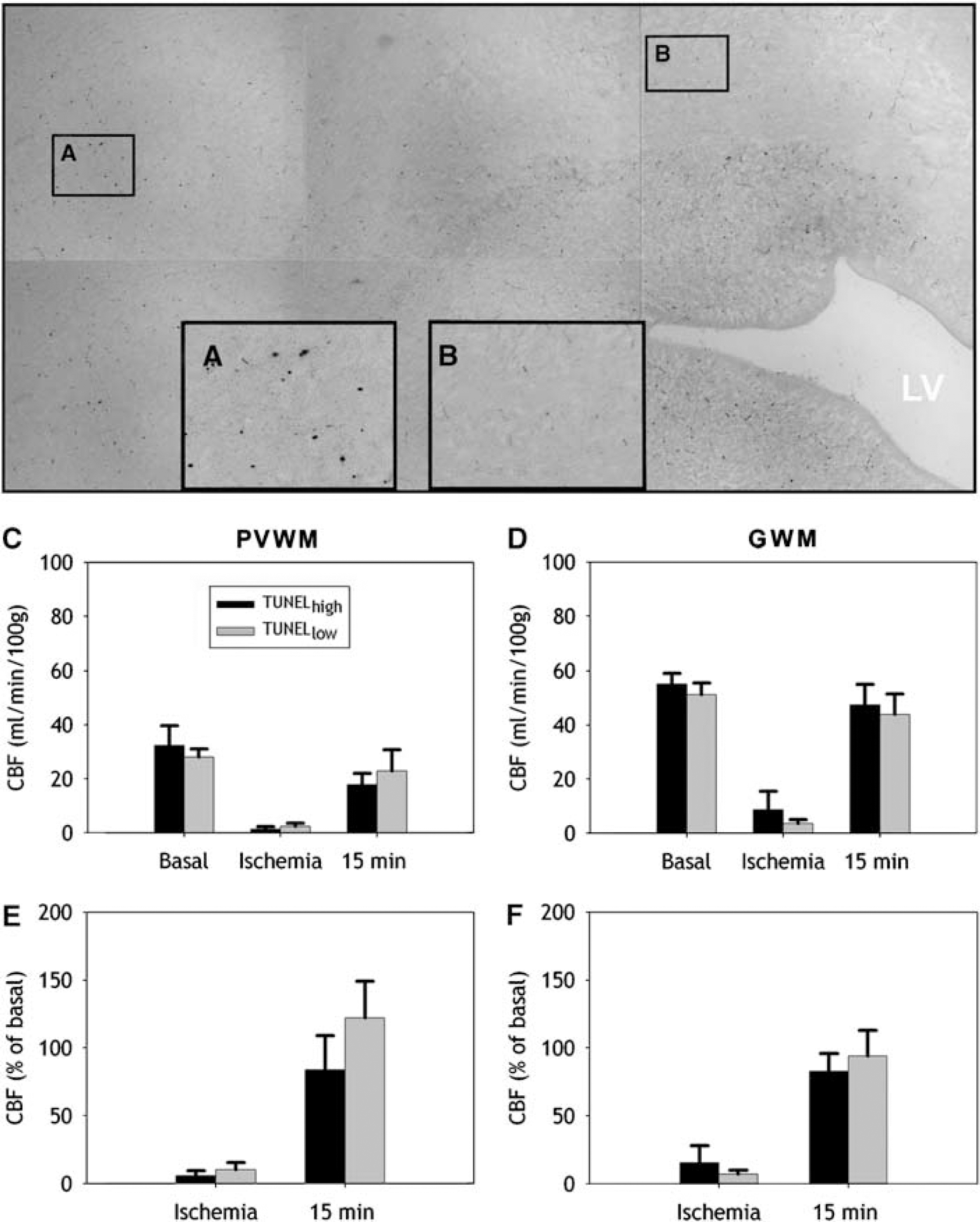
Absolute CBF and relative CBF in the PVWM and GWM are similar in regions of high and low injury defined by TUNEL staining. (
The Magnitude of Cortical Injury Predicts CBF during Ischemia
We next determined if the magnitude and distribution of cortical injury, defined by TUNEL staining, were related to the degree of CBF reduction during ischemia. We first segmented medial and lateral regions of frontal cerebral cortex (Figure 1F). At both levels, medial and lateral cortical regions did not differ in CBF for either basal, ischemia, or early reperfusion (15mins) CBF. In subsequent studies, medial and lateral regions of cerebral cortex were jointly analyzed. TUNEL staining was quantified in 14 separate TUNELhigh (≤20 labeled nuclei/mm2) or 9 TUNELlow (<20 labeled nuclei/mm2) lesions in frontal cortex (see Materials and methods). Figures 8A and 8B show representative high-power images of TUNELhigh (A) and TUNELlow (B) ROIs from a portion of a digitized montage of two adjacent frontal cortical gyri. Cerebral cortical lesions that were TUNELhigh showed a significantly greater reduction in absolute CBF during ischemia than regions that were TUNELlow (Figure 8C; P <0.05; mean difference: 9.3 mL per min per 100 g; 95% confidence limits: +18.4 to +0.2). Analysis of relative CBF showed a similar significant difference between TUNELhigh and TUNELlow regions (Figure 8D; P= <0.05). Hence, the predilection for cortical injury was associated with the severity of ischemia.
The magnitude of cortical injury is predicted by the severity of ischemia. (
Discussion
To address the role of physiologically defined vascular boundary zones in immature cerebral white matter injury, we developed a novel method to spatially quantify fetal CBF in histopathologically defined lesions in two regions of predilection for PWMI in the preterm infant. This study yielded several findings that did not support the presence of pathologically significant gradients of fetal CBF under conditions of ischemia-reperfusion: (1) Cerebral blood flow during both ischemia and reperfusion was equivalent throughout the PVWM. (2) Absolute and relative CBF did not differ significantly between the PVWM and GWM during ischemia or reperfusion. (3) Relative CBF in cerebral cortex during ischemia was lower than in PVWM. (4) Relative CBF during ischemia and reperfusion was not predictive of the severity of white matter injury. (5) The magnitude of ischemia to the cerebral cortex but not to the PVWM directly correlated with lesion severity defined by TUNEL staining. Hence, relative to more superficial cerebral structures, the PVWM did not display unique CBF disturbances. Periventricular white matter lesions did not localize to regions susceptible to greater hypoperfusion, nor did less vulnerable regions of the PVWM have greater CBF during ischemia.
To test the hypothesis that PWMI is related to regional heterogeneities of CBF, we took several departures from prior studies that quantified regional fetal CBF with radioactively labeled microspheres. Radioactive microspheres have provided limited information about regional differences in preterm CBF, because precise definition of regional boundaries in fresh unfixed tissue is not feasible. There is the potential for systematic error in the selection of tissue samples at necropsy, and histologic studies are also not possible. These limitations were circumvented with fluorescently labeled microspheres (Riddle et al, 2006) that allowed CBF to be quantified in an unbiased fashion by coregistration of microsphere data sets with histologically defined ROIs. Hence, a particular advantage for studies with large animals is that a single microsphere data set can be analyzed in conjunction with multiple histopathologic data sets.
We believe that microsphere flow measures are a valid method for analysis of fetal cerebral ischemia and subsequent cellular damage. An earlier study in adult cat raised a potential concern for overestimation of CBF in PVWM by microsphere-based measurements because of a discordance between flow as measured with radiolabeled iodoantipyrine relative to microsphere-derived measurements (Schuier et al, 1987). However, studies with 14C-iodoantipyrine in anesthetized, exteriorized 60-day fetal sheep showed distinct gradients of regional CBF between cortical plate and cerebral white matter (Habgood et al, 1991). These authors found a ratio of cortical to deep white matter CBF of 1.8, which is consistent with a similar trend that we found in 90-day fetal sheep with CBF measurements made by microspheres (ratio of 2.4). Taken together, these findings support that the vascular anatomy of the immature fetal sheep brain does not direct microspheres preferentially to the deep white matter.
Despite inherent differences in approach, our fetal CBF measurements with fluorescently labeled microspheres were in general agreement with previous studies of global CBF with radioactive microspheres (Gleason et al, 1989; Helou et al, 1994; Iwamoto et al, 1989; Szymonowicz et al, 1988). We found that basal CBF in frontal and parietal PVWM was ˜60% to 70% lower than in cerebral cortex. Similarly, a prior study found ˜50% lower rates of basal perfusion in undefined white matter relative to cortex in preterm and near-term sheep (Szymonowicz et al, 1988). Hence, the lower basal flow in PVWM does not, alone, support the notion of vascular immaturity or insufficiency. Regional differences in basal CBF are more likely related to differences in metabolic demand and a tight linkage between metabolism and blood flow. Basal glucose metabolism in ˜0.65 gestation ovine PVWM is about 70% lower than in cerebral cortex (Abrams et al, 1984; Iwamoto et al, 1989). Regional differences in basal flow and metabolism may also be related to the different cell types present in white matter/SVZ versus gray matter or to higher levels of neuronal activity.
While we found no evidence of blood flow heterogeneity to explain the spatial distribution of cerebral white matter injury in our model of severe but not complete global ischemia, the microsphere technique we applied, like most other blood flow methods, was inherently limited. Specifically, the relative precision of blood flow measurements during severe global ischemia at low flows in small ROIs was limited by relatively low numbers of microspheres and the unavoidable error because of Poisson variation. Nevertheless, measurements of CBF during ischemia were unbiased and, therefore, valid. The percentage decrease in CBF in comparably large regions was similar in cortex, GWM, and PVWM. If anything, the flow to the cortex decreased proportionally more than the flow to the PVWM. Thus, the PVWM does not appear to be a vascular end or boundary zone. The precision of the CBF measurements would be most reduced when the ROIs were small, as was commonly dictated by the size of the TUNELhigh and TUNELlow regions of PVWM and cortex. We cannot conclusively rule out, but found no evidence for, pathophysiologically significant differences in CBF in relatively small discrete TUNELhigh or TUNELlow lesions in the ischemic PVWM. Evidence for such CBF differences was, however, found in the cortex. To further improve the accuracy of the CBF measurements in regions with microheterogeneity of damage, we need studies that use markedly more microspheres during ischemia in a larger cohort of animals.
The significance of the ˜30% hyperemia we found in most brain regions at 1 h of reperfusion remains to be determined. Hyperemia showed no apparent relation to seizure activity that was maximal several hours later. To our knowledge, only one other study measured reperfusion flow in immature fetal sheep. Reinfusion of blood after mild hemorrhagic hypotension resulted in a similar modest sustained immediate hypoperfusion in gray and white matter (Szymonowicz et al, 1990). Late gestation fetuses had substantial hyperemia to many brain regions that was not significant before 12 h after ischemia (Raad et al, 1999). Postischemia hyperemia has been linked to cerebral edema (Heiss et al, 1976).
Another advantage of our approach is that serial measurements of basal, ischemia, and early reperfusion flow were feasible in the same animal. We chose to measure CBF at the 15- and 60-min reperfusion times, because we expected that white matter injury may be related to a rise in free radical generation during the immediate reperfusion period. In adult rodents, major disturbances in reperfusion flow, either high or low, have been associated with increased tissue oxidative damage (Chan, 2001). However, in agreement with a recent study in preterm sheep (Welin et al, 2005), we did not detect a significant increase in F2-isoprostanes in tissue collected from the PVWM in three animals at 30 mins of reperfusion after 45 mins of ischemia (SA Back, AR Hohimer, and TJ Montine, unpublished findings).
It has been suggested that the predilection for PWMI during ischemia is related to a loss of cerebral autoregulation (pressure passive flow), whereas other regions such as the cortex are relatively protected, because they can autoregulate (Greisen, 2005). Immature fetal sheep do not appear to autoregulate flow in the cortex, white matter, thalamus, or brainstem (Muller et al, 2002; Szymonowicz et al, 1990), and data are also controversial in human (Menke et al, 1997; Soul et al, 2007; Tsuji et al, 2000; Tyszczuk et al, 1998). Although our experiments did not directly address this hypothesis, our current data do not support that explanation. Absolute CBF and relative CBF were similar between cerebral cortex and PVWM during ischemia. If anything, the PVWM seemed to retain a greater percentage of basal flow than cortex. Thus, both relatively protected and vulnerable regions with reference to damage have indistinguishable flow characteristics in our model of global cephalic hypotension.
Our physiologic findings are not consistent with the notion that two or more distinct microvascular-anatomic boundary zones explain the predilection for ischemic PWMI. One vascular supply is thought to arise from ventriculopetal arteries that arise from leptomeningeal arteries, penetrate the cerebral cortex, and terminate as capillary end zones adjacent to the ventricles (reviewed in Nelson et al, 1991). Second are ‘ventriculofugal’ arteries that arise from choroidal and striate arteries and project toward the lateral ventricles before deviating away from the ventricle to a final termination in PVWM capillary beds. Ventriculopetal end zones, and ventriculopetal and ventriculofugal arteries were also proposed to collectively form a vascular border zone in the PVWM. The existence of this border zone and the associated ventriculofugal arteries has been the subject of considerable controversy (Mayer and Kier, 1991; Nelson et al, 1991; Volpe, 2001). We previously found that relatively large adjacent regions of medial or lateral PVWM did not differ in CBF under conditions of basal or ischemia-reperfusion flows (Riddle et al, 2006). Here, we also found that microvascular heterogeneities in flow did not correlate in magnitude with the severity of PVWM injury.
An alternative explanation for the topography of PVWM lesions is the distribution of susceptible cell types. We recently studied the relative susceptibility of axons and glia (i.e., microglia-macrophages, astroctytes, and oligodendrocyte-lineage cells) to global ischemia in the 0.65 gestation sheep (Riddle et al, 2006). The distribution of PWMI coincided with the magnitude and distribution of preOL degeneration. PreOLs are particularly susceptible to oxidative stress and hypoxia-ischemia in human and ovine preterm white matter (Back, 2006). By contrast, other cell types are markedly more resistant to ischemic cell death. In fact, more differentiated OLs were more resistant to degeneration than preOLs (Back et al, 2002; Riddle et al, 2006). This resistance of OLs to ischemia coincides with less damage to OL-rich regions of PVWM. Injury to the PVWM and the GWM was, thus, variable and greatest in preOL-enriched regions. Our findings, thus, support that the topography of cerebral white matter injury is related to heterogeneity in the distribution of susceptible OL lineage stages.
The cerebral gray matter susceptibility to ischemia appears to be defined by vascular mechanisms distinct from those that injure white matter. Unlike the PVWM, the severity of cortical injury was predicted by the reduction in CBF during ischemia. We previously reported that cortical injury was related to the duration of ischemia between 30 and 45mins, and beyond 37mins it increased markedly both in magnitude and distribution in a nonlinear fashion (Riddle et al, 2006). With prolonged ischemia (45mins), pronounced cortical injury predominated in the medial cortex in a watershed pattern. Our findings suggest the possibility that the cortical vasculature has physiologically distinct microvascular zones that may reflect heterogeneity in vascular-anatomic maturation. Heterogeneity in the expression of substances that regulate cerebral vasodilation may also be maturation dependent. Regional differences may also exist in the closing pressure at which CBF approaches a no-flow state (Greisen, 2005).
Clinico-Pathologic Significance
Although increasing evidence supports a role for cerebral ischemia in the pathogenesis of PWMI in critically ill premature infants, routine clinical monitoring of CBF to identify high-risk infants is not currently feasible. One potential approach for bedside monitoring of global CBF is near-infrared spectroscopy, which may provide an indirect measure of PVWM flow. A limitation of near-infrared spectroscopy is that CBF measurements are restricted to superficial cerebral cortex (Wolfberg and du Plessis, 2006). Studies are needed to determine if cortical CBF measurements accurately reflect CBF changes deeper in the PVWM. The heterogeneity in cortical CBF that we identified during ischemia also indicates that clinical measurements may require greater sensitivity to discriminate maturation-dependent differences in microregional cortical CBF.
Although it appears that cortical-neuronal susceptibility occurs in association with PWMI (Volpe, 2005), the etiology of the reductions in cerebral cortical volume seen in many preterm survivors remains unclear. Also, there is no explanation for the significant cognitive and learning disabilities diagnosed in up to 50% of preterm survivors at school age (Litt et al, 2005). Our findings suggest that microvascular heterogeneity may contribute to primary cerebral cortical injury from ischemia, which may cause enduring changes in cerebral cortical connectivity at a critical window in development.
In summary, studies to date support that ischemia-reperfusion injury is clinically important for the initiation of PVWM injury in human preterm neonates. Our studies in preterm fetal sheep found no evidence for physiologically significant vascular boundary zones as the basis for the topography of white matter lesions. Rather, this topography appears to be better explained by the distribution of susceptible preOLs in regions subject to relatively uniform disturbances in CBF during ischemia-reperfusion. Future clinical studies are needed in preterm neonates that apply emerging real-time perfusion imaging approaches to define the degree of CBF heterogeneity under basal conditions and conditions of metabolic demand.
Discloure/Conflict of Interest
None.
Footnotes
Acknowledgements
We are grateful to Dr Lowell Davis, Dr John Bissonnette, Dr Kent Thornburg, Dr Samantha Louey, and the Oregon Heart Center for their support and to Loni Socha and Robert Webber for their technical support.
