Abstract
We tested whether cerebral arteriovenous malformations (AVM) alter brain tissue oxygen pressure, Po2, carbon dioxide pressure Pco2, and pH before, during, and after hypercapnia. A craniotomy was performed and a sensor inserted into normal brain tissue (control) (n = 7) or into tissue adjacent to an AVM (n = 9). Under baseline conditions, tissue Po2 was 80% lower in AVM compared to control patients, but Pco2 and pH were normal. During a 10 mm Hg increase in Paco2, tissue Po2 increased only in AVM patients, Pco2 increased in both groups, and pH decreased only in controls. When hypercapnia was reversed, tissue Pco2 decreased below baseline and pH increased in AVM patients. Results suggest that tissue CO2 washout and elevated pH result from increases in blood flow during hypercapnia. This response may be related to symptoms of hyperperfusion during AVM resection.
Arteriovenous malformations (AVMs) produce high flow shunts for cerebral blood flow (CBF), which decrease perfusion pressure in surrounding tissue and may lead to ischemia. However, the presence of ischemia has not been confirmed by direct measurement of brain tissue Po2, Pco2, and pH in AVM patients. It is hypothesized that chronic decreases in vascular resistance in tissue adjacent to an AVM produce vascular paralysis (Spetzler et al., 1978). This is suggested to produce hyperperfusion and tissue injury when perfusion pressure is normalized. However, vasodilatory reactivity is normal or increased in hypoperfused regions adjacent to AVMs, and autoregulation to increases in blood pressure is intact (Batjer et al., 1988; Young et al., 1990, Young et al., 1993). This suggests that mechanisms other than vasoparalysis are responsible for hyperperfusion complications after AVM resection.
In this study, we measured brain tissue Po2, Pco2, and pH before, during, and after hypercapnia. Results show that AVM patients are hypoxic under baseline conditions. Hypercapnia increases CBF, which results in tissue CO2 washout and elevated pH in AVM patients.
METHODS
Patient selection
These studies were approved by the institutional review board for clinical research, and informed consents were obtained. Group 1 (n = 7) comprised control patients undergoing craniotomy for cerebral aneurysm clipping. These patients had no evidence of cerebral hemorrhage before the surgery and had no signs of ischemia, according to neurologic testing or cerebral angiograms. Group 2 (n = 9) comprised patients with cerebral AVMs. Eight of these patients underwent one-two radiological procedures for AVM nidus embolization several days before the neurosurgical procedure to resect the AVM, which partially obstructed flow to the AVM.
For the study, patients were anesthetized with 10–15 μg/kg fentanyl and 3–5 mg/kg thiopental, paralyzed with 100 μg/kg vecuronium, intubated, and ventilated with 0.5% end-tidal isoflurane. FIo2 was 0.4 and Paco2 was maintained between 30 and 35 mm Hg. A radial artery catheter was inserted to measure arterial pressure (Marquette, Milwaukee, WI, U.S.A.). Following the craniotomy, the dura was opened and the tissue sensor inserted 4 cm into the cortex. After a 30 min equilibration period, baseline measures of end-tidal CO2 and anesthetic gases (Datex Instruments, Helsinki, Finland), mean arterial pressure (MAP), and brain tissue Po2, Pco2, pH, and temperature were recorded continuously by computer throughout the study.
The Paratrend 7 sensor (Biomedical Sensors, Malvern, PA, U.S.A.) is 0.5 mm in diameter and is supplied as a sterile, disposable device comprised of two modified optical fibers for Pco2 measurements and pH, a miniaturized Clark electrode for Po2 measurement, and a thermocouple for temperature determination. The sensor is packaged with a tonometer containing buffer solution, which serves as a calibrating medium, and is calibrated before insertion into the patient. For insertion of the sensor, the outer nonsterile introducer system is covered with a sterile sheath. The sterile sensor is then extended from the end of the system and visually inserted (by the neurosurgeon) 4 cm into cortex tissue 2–3 cm from the AVM.
A Vasamedics laser Doppler flow probe (Vasamedics, St Paul, MN, U.S.A.) was placed on the cortex surface in the vicinity of the Paratrend 7 sensor in one control subject and four AVM patients.
Protocol
After steady state baseline values were obtained for arterial blood gases, MAP and tissue Po2, Pco2, and pH. Paco2 was increased 10 mm Hg by decreasing the ventilation rate. The increase in Paco2 was made during a 10-min period and then returned to baseline levels. Arterial blood gases were measured at the end of the baseline period and during CO2 challenge. Recording was terminated 10 min after end-tidal gases were returned to baseline levels.
Statistics
Data are reported as mean ± SD. Baseline arterial blood gases, MAP and tissue Po2, Pco2, and pH were compared between groups using t-tests. Peak changes during CO2 challenges were compared to baseline measures using paired t-tests. A Bonferroni correction was made for multiple comparisons.
RESULTS
Under baseline conditions, MAP was 84 ± 18 mm Hg in control and 81 ± 12 mm Hg in AVM patients. Paco2 was 33 ± 3 mm Hg in control and 32 ± 4 mm Hg in AVM patients and increased 10 ± 1 mm Hg during the CO2 challenge in both groups. Pao2 was maintained at >100 mm Hg in all patients and did not change during hypercapnia. Changes in tissue gases and pH are shown in Fig. 1. Under baseline conditions, tissue Po2 was lower in AVM compared to control patients. During hypercapnia, Po2 increased in AVM but not in control patients. Tissue Pco2 increased in both groups during the increase in Paco2 but decreased below baseline levels in AVM patients during the recovery period. Tissue pH decreased in control but not in AVM patients during the CO2 challenge and increased in AVM patients during the recovery period. Laser Doppler blood flow increased 48% in one control patient and 55 ± 22% in four AVM patients during the CO2 challenge. No bleeding or tissue injury was observed as result of insertion of the sensor.
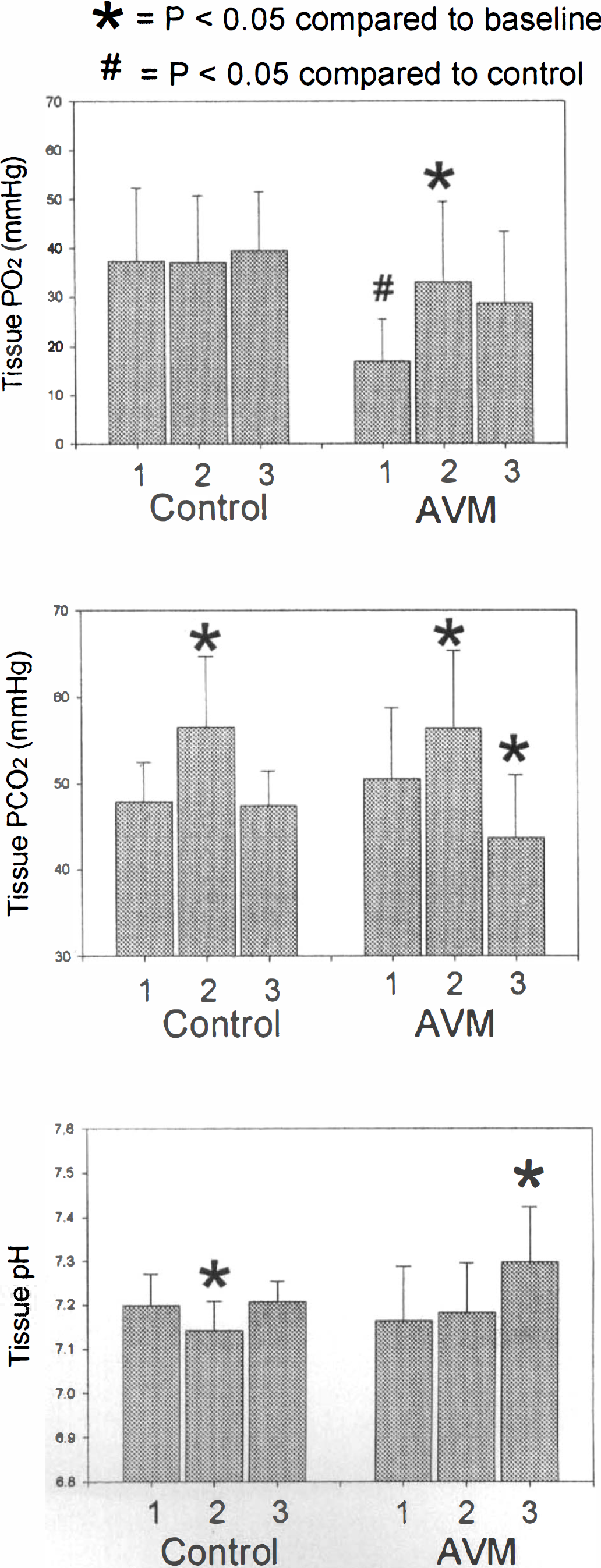
Brain tissue Po2, Pco2, and pH in control (n = 7) and AVM patients (n = 9) during hypercapnia. Under baseline conditions, AVM patients had low Po2 compared to controls. Hypercapnia increased Pco2 in both groups and decreased pH only in controls. During the recovery period, Pco2 decreased and pH increased compared to original baseline measures in AVM patients. 1, baseline; 2, hypercapnia; 3, recovery.
An example of the response in an AVM patient is shown in Fig. 2, which shows the increase in tissue Po2 during the CO2 challenge and the decrease in Pco2 and increase in pH when Paco2 was returned to baseline. Tissue bicarbonate significantly increased in both groups during hypercapnia: controls: baseline, 18.5 ± 3.4 meq/L; hypercapnia, 20.1 ± 4.4 meq/L; AVM: baseline, 17.2 ± 5.5 meq/L; hypercapnia, 20.2 ± 5.7 meq/L.
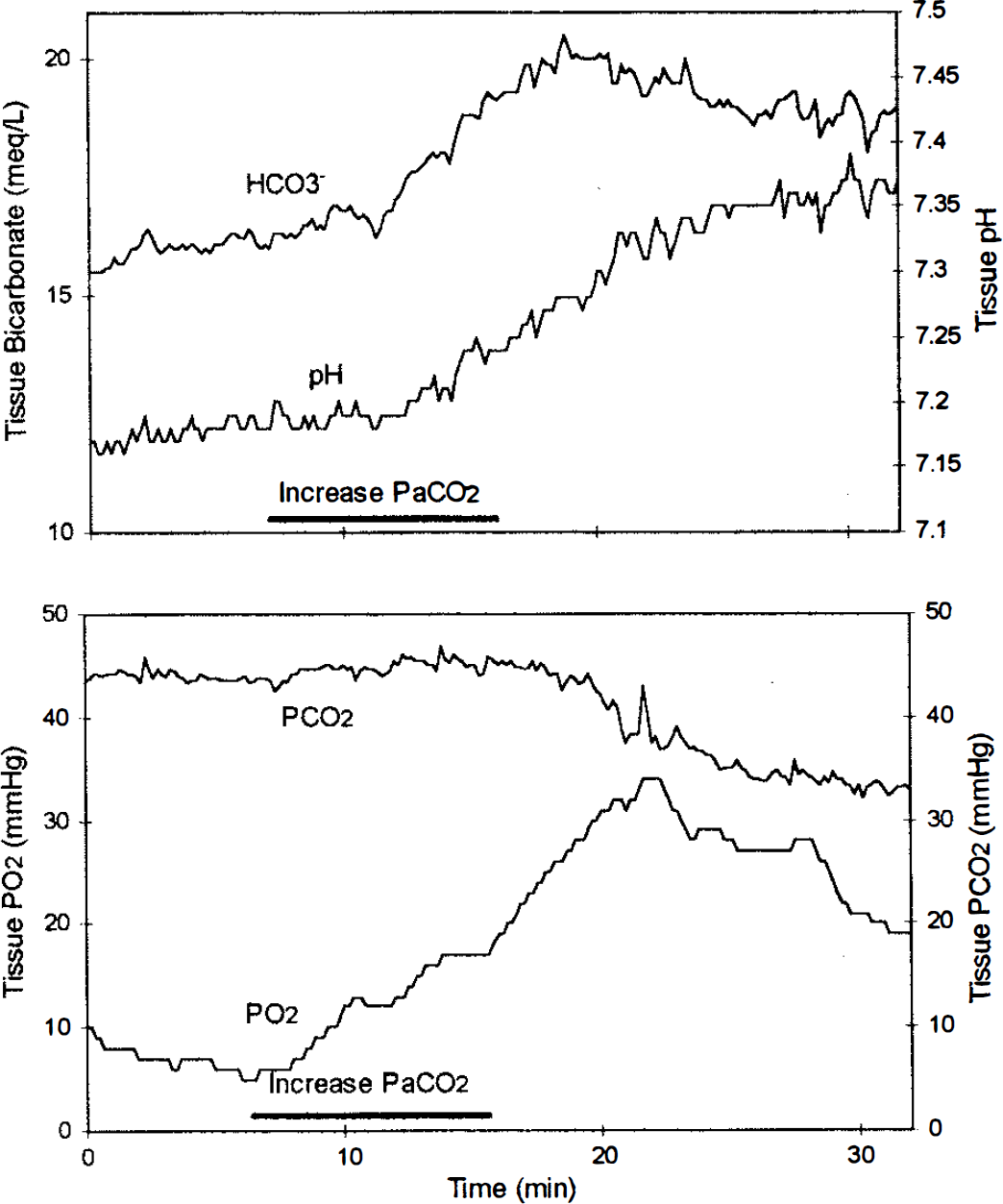
Changes in tissue Po2, Pco2, pH, and bicarbonate in an AVM patient during hypercapnia. During hypercapnia, Po2, Pco2, and bicarbonate increased. When the CO2 challenge was terminated, Pco2 decreased below baseline and pH increased above baseline levels.
DISCUSSION
In this study, AVM patients had low baseline tissue Po2 but normal Pco2 and pH compared to control patients. This suggests that chronic decreases in cerebral vascular resistance and/or capillary recruitment can partially compensate for decreases in perfusion pressure produced by the AVM (Norbash et al., 1994; Halsey and McFarland, 1974). During hypercapnia, Po2 increased in AVM patients, suggesting that oxygenation was flow limited. However, when Paco2 was returned to normal, tissue Pco2 decreased below baseline levels and pH increased in AVM patients. This suggests that hypercapnia increased CBF and produced a washout of CO2 in AVM patients. Enhanced CO2 clearance, coupled with buffering mechanisms that increase tissue bicarbonate (Hoffman et al., 1995), produced an increase in pH in AVM patients during the recovery period. The CO2 washout produced in AVM patients during hypercapnia is probably due to a transient increase in CBF and may represent an adaptation of tissue vasculature to chronically low perfusion pressure.
Reduction in tissue perfusion pressure produced by an AVM has been characterized as producing a steal of blood flow from surrounding tissue (Norbash et al., 1994). Spetzler et al. (1978) performed studies in animals in order to mimic the changes produced by an AVM. After placement of shunts in the cerebral circulation, they showed that severe hypoperfusion and cerebrovascular paralysis can occur as a result of decreased perfusion pressure with loss of reactivity to hypercapnia. This vasoparalysis may to lead to hyperperfusion complications when the AVM is resected surgically and normal perfusion pressure is restored to the tissue. However, in patients, Batjer et al. (1988) have shown that AVMs that are complicated later by symptoms of hyperperfusion have augmented vasodilatory reactivity in tissue adjacent to the AVM. Other studies agree that CO2 reactivity is intact before AVM resection and show that tissue autoregulation is intact after AVM resection, (Young et al., 1990, Young et al., 1993). This does not support a theory of vasoparalysis which occurs as a result of steal and suggests that hyperemia may occur by other mechanisms.
When brain tissue becomes ischemic, Po2 and pH decrease and Pco2 increases (Ljunggren et al., 1974; Hoffman et al., 1996). However, AVM patients in this study were only hypoxic. Reduced metabolic function may compensate for tissue hypoxia to maintain Pco2 normal and to avert metabolic acidosis (Fink, 1992). Capillary recruitment could also compensate for hypoperfusion by decreasing oxygen diffusion distance and enhancing CO2 exchange. However, capillary recruitment could be detrimental if CBF is increased, even modestly, during AVM resection (Young et al., 1993). Elevated perfusion pressure with enhanced capillary surface area could promote tissue edema. After AVM resection, neurological symptoms and deficits are present in patients with evidence of tissue hyperperfusion, and complications such as hemorrhage and hemispheric midline shift can occur (Katayama et al., 1994). Enhanced tissue CO2 washout may also be detrimental by reducing blood flow and producing hypoxia (Sugioka and Davis, 1960).
Some factors may have affected the results of this study. The AVM was partially obstructed in eight of the 10 patients studied by endovascular procedures before the surgery. This reduced AVM flow and may have improved perfusion pressure in the surrounding tissue. However, this was only a partial obstruction, and the high flow status of the AVM remained. This suggests that although the hypoxic changes may have been worse in patients without partial obstruction, they would not be substantially altered. We inserted the sensor 4 cm into brain tissue in a region 2–3 cm from the AVM. This tissue appeared normal in spite of the fact that significant hypoxia was present. Since the Po2, Pco2, and pH sensors were separated by up to 3 cm along the length of the probe, it is possible that different degrees of hypoxia were present in each tissue region measured. Therefore, each variable, i.e., Po2, Pco2, and pH, should be considered separately in each patient. We assume that we are measuring Po2, Pco2, and pH in a region adjacent to an AVM. It is also possible that opening the skull has produced a change in tissue dynamics and altered tissue gas measurements compared to the closed skull. Our baseline measurements were made under steady state conditions and should be compared with the experimental controls of this study.
In conclusion, our results show that hypercapnia in normal patients increases tissue Pco2 without a change in oxygenation. Tissue buffering mechanisms increase bicarbonate concentration during hypercapnia and attenuate the decrease in pH. In AVM patients, hypercapnia produces an increase in tissue oxygenation, enhanced CO2 clearance, and elevated pH compared to baseline measures. The CO2 response in AVM patients appears to uncover cerebrovascular mechanisms that compensate for chronically decreased perfusion pressure and may produce hyperperfusion complications during AVM resection.
