Abstract
Activation of CBF by hypercapnia or functional stimulation has been attributed to multiple mediators, most of which are thought to interfere with cerebrovascular reactivity in a closely time-related manner. Here we describe that brief hypercapnia produces marked up-regulation of somatosensory activation of blood flow that outlasts carbon dioxide exposure for at least 60 min. In chloralose-anesthetized, mechanically ventilated rats, somatosensory activation was carried out by electrical stimulation of the forepaw. Blood flow was measured in the contralateral primary somatosensory cortex by laser-Doppler flowmetry (LDF). Under control conditions, somatosensory stimulation increased LDF by 38.8 ± 11.0%. Ventilation with 6% CO2 for 3 min caused a rise of LDF by 28.0 ± 8.7%. Baseline CBF and Paco2 returned to control values within 20 min. Repetition of somatosensory stimulation after hypercapnia revealed a long-lasting up-regulation of the flow response: 25 min after hypercapnia, functional stimulation increased LDF by 86.0 ± 18.1%, and 60 min after hypercapnia even by 96.0 ± 26.0%. This is the first demonstration of CO2-induced up-regulation of functional activation of blood flow and an example of the importance of general physiological variables for the modulation of the coupling process.
The coupling between central nervous function, blood flow, and metabolism is a highly complex process that, despite considerable research over many decades, still remains to be elucidated. There is evidence that a multitude of chemical mediators is involved in this process, notably protons and potassium (Urbanics et al., 1978), adenosine (Ko et al., 1990), and nitric oxide (Dirnagl et al., 1993). The abundance of nerve terminals at the vascular wall suggests that functional activation is also under neurogenic control (Purves, 1978).
An important prerequisite for the biological relevance of any of these mediators is the spatial and temporal correlation with the functional activation pattern. In fact, blood flow begins to increase within seconds after the onset of functional activation, it declines promptly after cessation of stimulation (Leniger-Follert and Hossmann, 1979; Lindauer et al., 1993), and it is strictly confined to the region in which the activated neuronal network is located (Ueki et al., 1988). None of the mediators, however, fully complies with these observations. Potassium rises promptly after the onset of functional activation, but it declines before the end of functional hyperemia. Hydrogen ions and adenosine, in contrast, increase later than blood flow, but remain elevated throughout the functional activation. Nitric oxide is involved in the response of all these putative mediators, but its inhibition does not fully abolish functional activation of blood flow. It therefore has been suggested that the combined action of the mediators is responsible for the coupling process [for a recent review see Villringer and Dirnagl (1995)].
The complexity of the coupling process is further complicated by more diffuse modulating systems that are able to influence the set point and the sensitivity of the flow response. Up to now, these modulating effects have been little investigated, but there are obvious examples of pathophysiological and pharmacologically induced changes of the coupling process. Long-lasting reductions of the flow response during functional activation have been described following transient cerebral ischemia (Hossmann et al., 1973; Ueki et al., 1988). Flow activation is also reduced by most anesthetics like barbiturates (Lindauer et al., 1993) and a variety of other drugs including scopolamine (Dauphin et al., 1991) and theophylline (Ko et al., 1990). On the other hand, drugs have been described that lead to an enhancement of the flow coupling, such as physostigmine (Dauphin et al., 1991), S-emopamil (Szabo, 1989), and the N-methyl-
Recently, we noticed that the postischemic suppression of the flow response to somatosensory stimulation in rat is partly reversed by ventilating the animal for a few minutes with 6% carbon dioxide. When we looked into this phenomenon in more detail, it turned out that hypercapnia did not just accelerate the recovery of the disturbed functional activation, but led to a lasting up-regulation also in the nonischemic intact animal.
MATERIAL AND METHODS
General preparations
All animal experiments were approved by the ethical committee of the local authorities and conducted following the principles of laboratory animal care. Five adult male Sprague–Dawley rats (330–400 g) were initially anesthetized with 1.5% halothane in a 7:3 N2O/O2 mixture delivered to the animal by a face mask. The left femoral artery and vein were cannulated with polyethylene catheters (PE-50) for blood pressure recording (Kombidyn Monitoring Set; Braun AG, Melsungen, Germany), blood sampling, and drug administration. To prevent dehydration and obstruction of the catheters, saline was continuously infused (2 ml/kg/h) throughout the experiment. Following tracheotomy, the animals were paralyzed with pancuronium bromide (0.2 mg/kg/h) and artificially ventilated (Harvard Rodent Ventilator; Harvard, South Natick, MA, U.S.A.). Respiration frequency and tidal volume were adjusted to keep arterial Pco2 within the physiological range.
The head of the animals was fixed in a stereotaxic frame, the apex of the skull was exposed, and the bone over the parietal cortex was thinned using a saline-cooled drill to allow transcranial laser-Doppler flowmetry (LDF) (Lindauer et al., 1993). The laser probe (PeriMaster 4001; PeriMed, Sweden) was placed ˜3.5 mm lateral and 1 mm rostral to the bregma according to the coordinates of the somatosensory cortex (Paxinos and Watson, 1982) and then fine-positioned for maximum signal changes during stimulation of the contralateral forepaw. Monopolar EEG recordings were carried out with an active calomel electrode at 0.5 mm behind the laser probe and an indifferent calomel electrode placed on the nasal bone. Rectal temperature was kept constant at 37°C using a feedback-controlled heating pad. LDF, systemic arterial pressure, and EEG were recorded continuously on a polygraph and processed on a PC running the DasyLab data acquisition software (DataLog, Mönchengladbach, Germany). For electrical stimulation, small needle electrodes were inserted under the skin of both forepaws. Arterial blood samples were taken at regular intervals and analyzed using a blood gas analyzer (model 288; Ciba-Corning, Fernwald, Germany).
Forty-five minutes before the stimulation experiments, halothane/N2O anesthesia was discontinued and replaced by intravenous anesthesia with α-chloralose (80 mg/kg). A supplemental dose of α-chloralose (25 mg/kg) was given 5 min prior to forepaw stimulation. During chloralose anesthesia, the animals were ventilated with 7:3 nitrogen/oxygen.
Activation protocol
Somatosensory activation was carried out by electrical stimulation of the contralateral forepaw with rectangular pulses (0.3 ms, 0.5 mA) at a frequency of 3 Hz for a duration of 1 min. Specificity of the response was ascertained by stimulation of the ipsilateral forepaw. To exclude a time-dependent modulation of the blood flow response, the control measurement was repeated after 45 min in one of the animals. Subsequently, 6% CO2 was added to the ventilation gas for 3 min, and stimulation was repeated 25 and 60 min later. Before, during, and after CO2 ventilation and during each stimulation sequence, the arterial carbon dioxide tension was measured. Primary somatosensory evoked potentials (SEPs) were averaged using the DasyLab system.
Data analysis
Changes in regional CBF during hypercapnia and functional stimulation were calculated from the average of the data points sampled over 60 s before and during stimulation and expressed as percentage of baseline. Induced flow changes were tested for significance using Student's paired t test and physiological variables using repeated-measures analysis of variance (STATISTICA for Windows 4.5; StatSoft, Tulsa, OK U.S.A.). Values are given as means ± SD. Statistical significance was assumed for p < 0.05.
RESULTS
Physiological variables were within the normal range throughout the experiment, except during ventilation with 6% CO2 (Table 1). The amplitudes of the SEPs and the mean amplitude and median frequency of EEG also remained constant (Table 2), indicating that the brief hypercapnic episode did not lead to lasting general physiological or electrophysiological changes.
Physiological variables
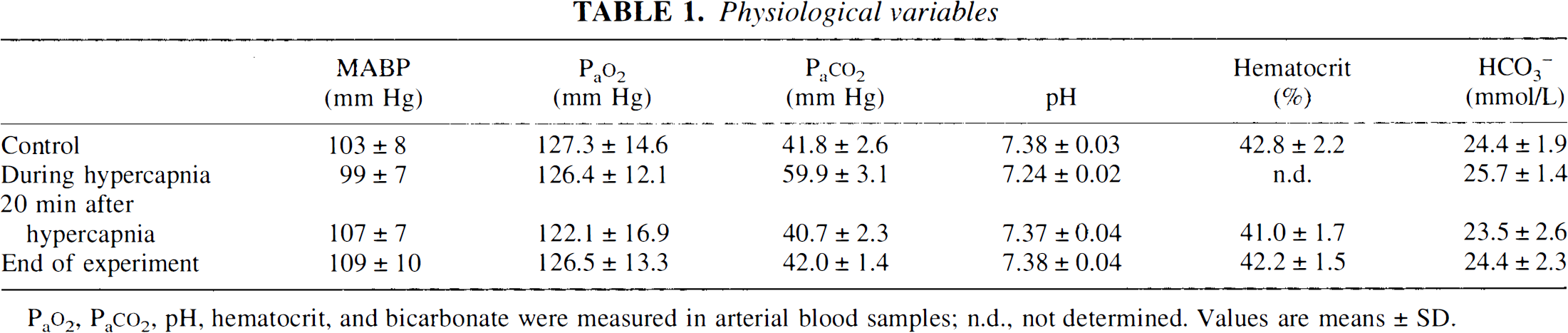
Pao2, Paco2, pH, hematocrit, and bicarbonate were measured in arterial blood samples; n.d., not determined. Values are means ± SD.
Ventilation for 3 min with 6% CO2 led to an increase of Paco2 from 41.8 ± 2.6 to 59.9 ± 3.1 mm Hg (p < 0.05) and to a decline of arterial pH from 7.38 ± 0.03 to 7.24 ± 0.02 (p < 0.05). LDF increased by 28.0 ± 8.7% (p < 0.05), corresponding to a rise by 1.58 ± 0.6%/mmHg. After termination of CO2 ventilation, blood variables and LDF promptly returned to normal.
Electrophysiological recordings
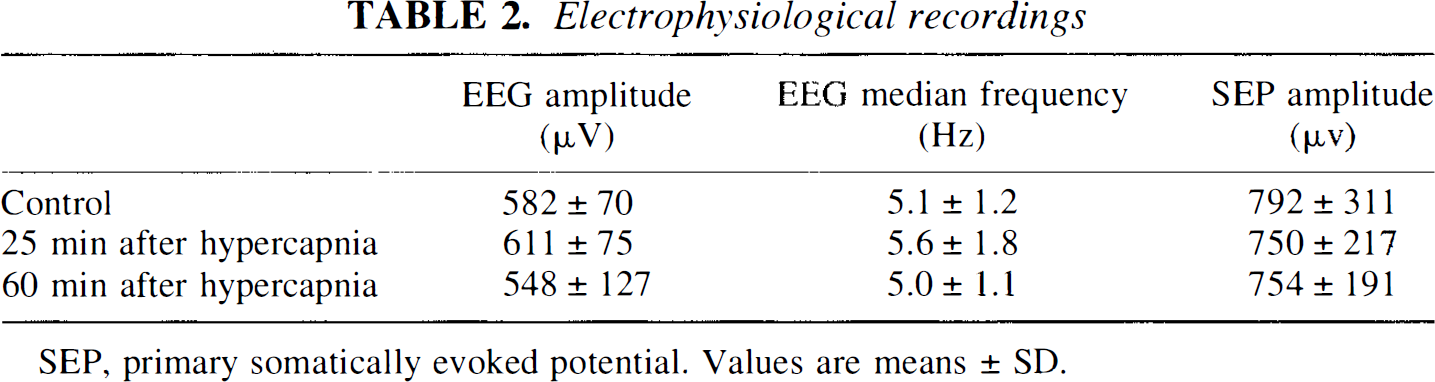
SEP, primary somatically evoked potential. Values are means ± SD.
Stimulation of the ipsilateral forepaw did not result in measurable changes of blood pressure or LDF. In contrast, stimulation of the contralateral forepaw caused an increase of LDF by 38.8 ± 11.0% (Figs. 1 and 2). Blood flow typically increased within 2–3 s after beginning of forepaw stimulation and reached its maximum a few seconds later (Fig. 1). After termination of stimulation, regional CBF abruptly fell slightly below normal before returning to baseline after ˜30–60 s. Repetition of the control experiment after 45-min delay in one animal revealed no difference in the magnitude of the LDF response (39 and 42%, respectively).
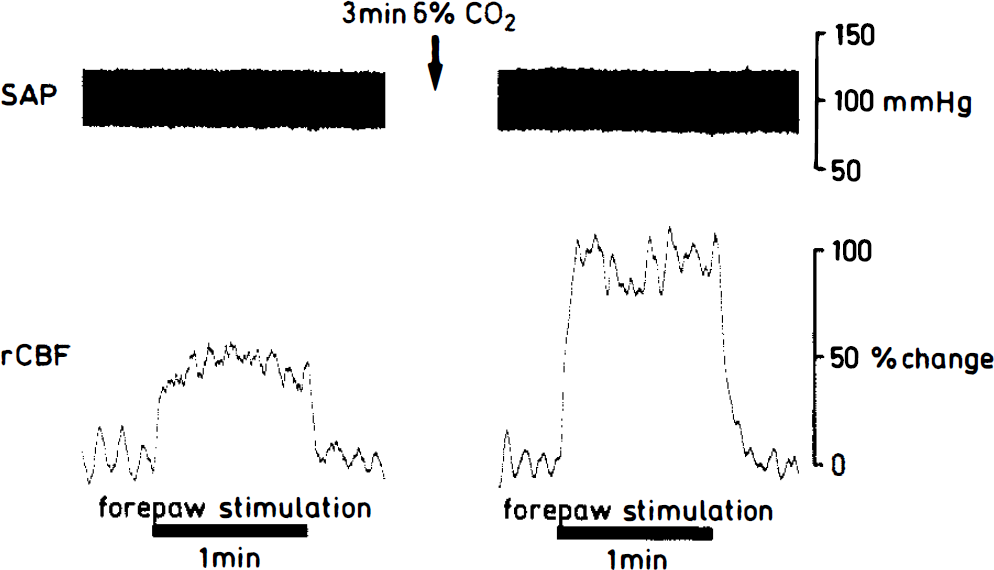
Regional CBF (rCBF) response to electrical forepaw stimulation before and after a brief episode of 6% hypercapnia. Blood flow was measured by laser-Doppler flowmetry (LDF) in the primary somatosensory cortex of the opposite hemisphere. Note marked up-regulation of the flow response after hypercapnia. SAP, systemic arterial pressure.
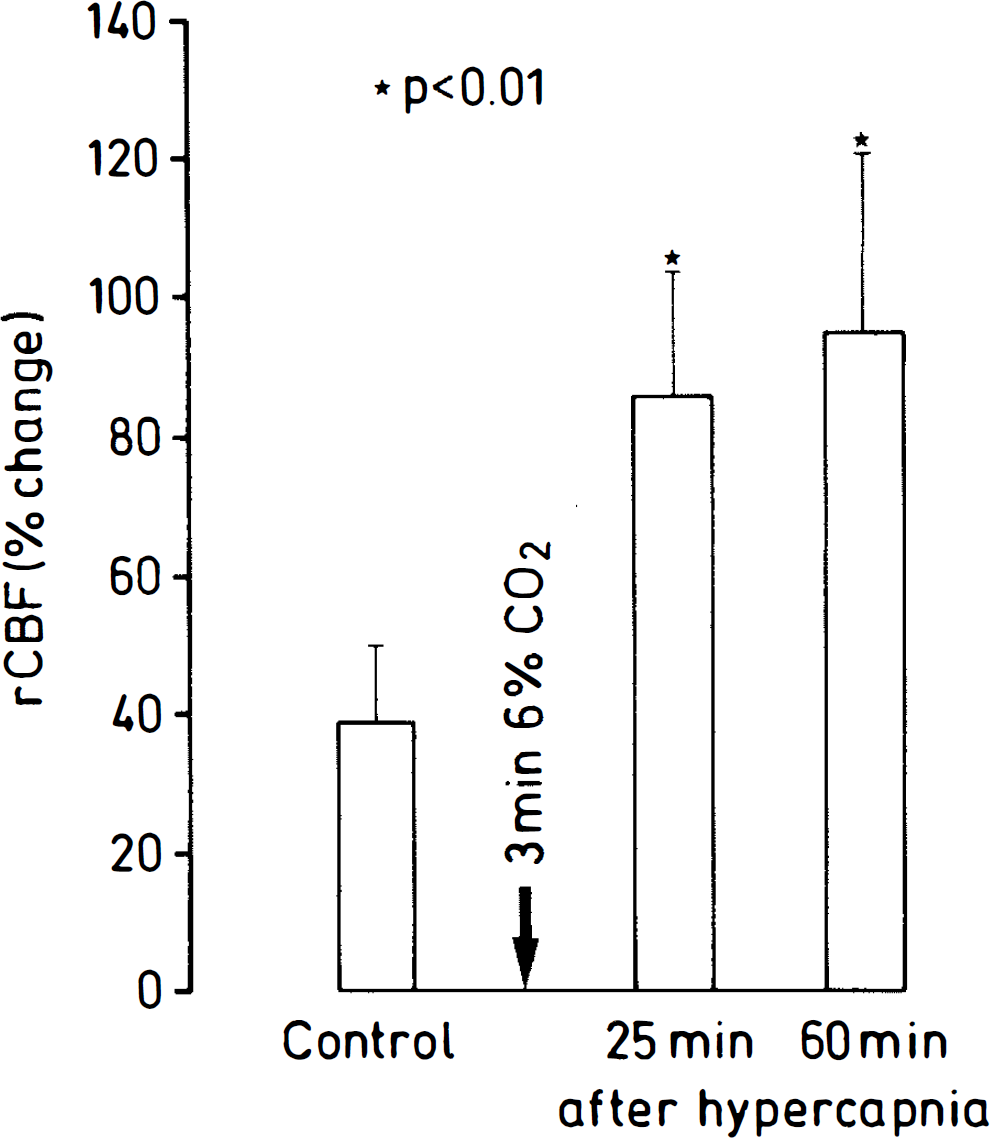
Up-regulation of the functional activation of blood flow after 3-min ventilation with 6% CO2. Blood flow was measured by laser-Doppler flowmetry (LDF) in the somatosensory cortex during stimulation of the contralateral forepaw. Note marked increase of the flow response at both 25 and 60 min after hypercapnia. rCBF, regional CBF.
After hypercapnia, the response of LDF during functional activation markedly changed. Twenty-five minutes after CO2 ventilation, somatosensory stimulation increased LDF by 86.0 ± 18.1%, and after 60 min even by 96.0 ± 26.0% (Fig. 2). Compared with the prehypercapnic response, these values represent an up-regulation of the flow response by 122 and 147%, respectively.
DISCUSSION
To the best of our knowledge, the here-described enhancement of the somatosensory activation of CBF by a brief episode of hypercapnia is the first report of a long-lasting up-regulation of the function/flow couple following transient increase of Paco2. This enhancement persisted throughout the 60-min observation period, although all blood variables promptly returned to normal after termination of hypercapnia. The changes of plasma bicarbonate also quickly normalized, excluding lasting changes of the buffer capacity of the blood. When stimulations were repeated, we did not see changes in the flow response, excluding a modulation of the flow couple by the stimulation itself. We therefore conclude that brief hypercapnia does, in fact, produce long-lasting changes in functional activation.
To appreciate the magnitude of the hypercapnia-induced up-regulation, our recordings should be compared with previous measurements in chloralose-anesthetized rats. Dirnagl et al. (1994) reported an increase of blood flow by 18% during 1-min whisker stimulation, and Ueki et al. (1988) an increase by 33% during electrical forepaw stimulation. These values are smaller than our control response of ˜40% and far below the flow increase by ˜90% after hypercapnic conditioning. It is therefore rather unlikely that the here-observed up-regulation is due to particularities of the stimulation paradigm.
An important methodological concern is the possibility that this phenomenon is related to the anesthesia with α-chloralose. In contrast to barbiturates, halothane, and most other anesthetics, chloralose does not suppress functional activation and therefore is widely used for studying the coupling process in experimental animals (Leniger-Follert and Hossmann, 1979; Ueki et al., 1988; Dirnagl et al., 1994; Hyder et al., 1994). Chloralose in the dose used in our study produces stable anesthesia for ˜4 h (Bonvento et al., 1994), which is in line with our observation of stable EEG and SEP data. It is therefore unlikely that the up-regulation observed here during the 1-h observation period is due to alterations in the depth of anesthesia. On the other hand, Bonvento et al. (1994) described a reduction of CO2 reactivity following chloralose anesthesia that lasted for ˜3 h. Since changes in functional activity are associated with changes in metabolic activity and hence CO2 production, this might also alter the extent of flow activation. However, the reduction in CO2 reactivity was stable for 3 h and therefore would not explain the dramatic change in vascular reactivity right after the hypercapnic episode.
A more likely mechanism for the up-regulation of the flow couple is interference of hypercapnia with other mediators of functional activation. CBF is sensitive to, among others, H+ and potassium (Kuschinsky et al., 1972), K+ (Urbanics et al., 1978), Ca2+ (Betz and Csornai, 1978), and adenosine (Berne et al., 1974), all of which change during functional activation. There is also increasing evidence that NO may play a role in functional activation (Dirnagl et al., 1994). In fact, inhibition of NO synthetase strongly attenuates the CBF response to such different mediators as H+, K+, and adenosine (Dirnagl et al., 1994; Iadecola et al., 1994; Dreier et al., 1995). It therefore would be of interest to find out if hypercapnia is able to up-regulate NO synthetase activity. In vitro experiments have, in fact, revealed that mild cytoplasmic acidification to ˜7.0 enhances NO synthetase activity (Heinzel et al., 1992). However, although CO2 is a freely diffusible molecule, moderate hypercapnia causes only minor intracellular pH changes (Jensen et al., 1988) because of potent intracellular buffering systems (Chesler, 1990). Moreover, the rapid normalization of Paco2 after hypercapnia makes it rather unlikely that the lasting up-regulation is mediated by pH-dependent changes of NO synthetase activity.
Another possible mechanism could be the Ca2+/calmodulin dependence of NO synthetase (Bredt and Snyder, 1990). In human fibroblasts, extracellular acidosis triggers inositol triphosphate-mediated mobilization of intracellular calcium (Smith et al., 1989). In primary neuronal cultures from rodents, intracellular acidification also results in direct release of Ca2+ from intracellular stores (OuYang et al., 1994). It therefore is conceivable that the brief episode of hypercapnia applied is able to produce long-lasting changes in intracellular Ca2+ activity. Finally, the possibility of an intracellular calcium influx via voltage-dependent calcium channels should be considered. It has been hypothesized that this mechanism is responsible for the activation of NO synthetase during functional activation (Iadecola, 1993), but it would be difficult to understand how a brief episode of hypercapnia is able to increase the conductance of these channels for such a long time. In conclusion, the present data clearly demonstrate that a short episode of moderate hypercapnia leads to sustained up-regulation of the flow activation to somatosensory stimulation. Although the underlying mechanism is unclear at the present, the functional significance of this observation may be substantial. Further experiments should therefore be carried out to clarify the underlying mechanism and the importance of this phenomenon for the coupling between function and metabolism under physiological and pathophysiological conditions.
Footnotes
Abbreviations used
Acknowledgment:
The authors gratefully acknowledge the artwork of Mrs. I. Mühlhöfer and Mr. B. Huth and the excellent secretarial help of Mrs. H. Hahmann and Mrs. D. Schewetzky.
