Abstract
Our previous study on the ischemia-induced expression of platelet-derived growth factor (PDGF)-B chain in the rat brain prompted us to examine expression of PDGF β-receptor in the ischemic brain. Focal ischemia was induced by permanent tandem occlusion of middle cerebral and common carotid arteries in spontaneously hypertensive rats. Northern analysis revealed that ischemia significantly increased expression of the receptor in the ischemic neocortex at 4 and 7 days (328 ± 109%; 323 ± 119%, respectively, over control: n = 4, p < 0.05 versus sham). Neurons in infarct transiently snowed increased immunostaining for the receptor at 1 day, whereas neurons in periinfarct area showed sustained and increased immunoreactivity from 1 to 14 days postischemia. Reactive glial cells in the external capsule and in molecular layer of the neocortex adjacent to infarct possessed enhanced immunoreactivity from 1 to 21 days. Furthermore, marked immunoreactivity was observed on brain macrophages in infarct and on the abluminal side of capillaries surrounding infarct from 4 to 7 days. These results demonstrated that ischemic insult increases expression of the PDGF β-receptor at both the mRNA and protein level in the brain, suggesting its important role in cellular cascade of the ischemic brain.
Platelet-derived growth factor (PDGF) (a homodimer of PDGF-A or PDGF-B chain or an AB heterodimer) is a growth-regulatory molecule and chemoattractant for mesenchymal and glial cells (Ross et al., 1986; Heldin and Westermark, 1990). The PDGF isoforms induce complex and diverse functions through two distinct cell-surface receptors designated α and β, respectively (Hart et al., 1988; Heldin et al., 1988). Although the PDGF α-receptor binds to the A or B chain with high affinity, the β-receptor binds only to the B chain. On binding of PDGF to receptors of either type, dimerization of receptor molecules and activation of the receptor tyrosine kinase ensue. These events lead to the autophosphorylation of the receptor and phosphorylation of various substrates including phospholipase C-γ (Kumjian et al., 1989; Meisenhelder et al., 1989), GTPase activating protein (Molloy et al., 1989; Kazlauskas et al., 1990), phosphatidylinositol-3′-kinase (Coughlin et al., 1989), and members of the src family (Kypta et al., 1990).
Our previous study on the ubiquitous neuronal expression of PDGF-B chain with the expression of PDGF receptors in the brain suggested a novel role of the B chain as a neuronal regulatory agent (Sasahara et al., 1991, Sasahara et al., 1992). Recently, we showed that ischemia increases expression of the B chain mRNA, but not that of the A chain mRNA, and enhances PDGF-B chain-like immunoreactivity in neurons and brain macrophages in the rat brain (Iihara et al., 1993; Iihara et al., 1994b). These observations indicate that the PDGF-B chain in the brain plays an important role in the healing process following ischemic damage. On the other hand, the PDGF β-receptor, which is specific for the B chain, was shown to be expressed on brain capillary endothelial cells (Smits et al., 1989) and on cultured neuronal cells (Smits et al., 1991). However, the functional significance of β-receptor expression in the brain remains unknown.
To address this issue, Northern blot analyses and immunohistochemical studies were performed to examine the expression of PDGF β-receptor in the rat brain after focal ischemia. The results presented here indicate that the enhanced expression of the β-receptor, which is induced coordinately with up-regulation of the B chain, should contribute to the survival of neurons at risk, glial reaction, neovascularization, and the accumulation of brain macrophages in and around the ischemic lesion. Preliminary results of this study were published previously in abstract form (Iihara et al., 1994a).
MATERIALS AND METHODS
Surgical procedure
Male spontaneously hypertensive rats (SHRs; 250–300 g) (Charles River) were used in the experiment because of their higher reproducibility of stroke compared with normotensive rats (Brint et al., 1988). Focal ischemia of the right neocortex was produced by permanent occlusion of the right common carotid artery and the right middle cerebral artery just superior to the rhinal fissure as described previously (MCA/CCAO) (Brint et al., 1988; Iihara et al., 1994b). Sham operations were performed in a similar manner without occluding the arteries.
Northern blot analysis
At 1, 4, 7, 14, and 21 days after MCA/CCAO and 4 days after sham operations (n = 12 each), the animals were anesthetized and decapitated. The right neocortex, which included the infarct and the adjacent neocortex, was dissected out from the coronal slices cut into 6-mm-thick sections (interaural line 9.2–3.2 mm), and quickly frozen in liquid nitrogen. Reproducibility of the size and location of infarct was confirmed by hematoxylin and eosin staining of the adjacent sections from the same animals used for the RNA preparation. Poly A+ RNA (1.4–3.7 μg, each), extracted directly as described previously (Wilkinson, 1991) from the neocortices of 3 animals, was separated on a 1% agarose gel (Sigma) containing 20 mmol/L 3-(N-morpholino)-propane sulfonic acid 5 mmol/L sodium acetate, 1 mmol/L EDTA, and 6% formaldehyde, transferred onto nylon filters (NYTRAN, Schleicher & Schuell) in 10 × SSC, cross-linked with ultraviolet light, stained with methylene blue, and photographed as reported previously (Iihara et al., 1994b). The filters were hybridized at 65°C overnight with a 32P (3,000 Ci/mmol; Amersham)-labeled cDNA probe of EcoRI/Hind III fragment of human PDGF β-receptor (supplied by Zymogenetics; Seattle, Washington, U.S.A.) in 0.25 mol/L Na2HPO4, 1% bovine serum albumin, 1 mmol/L EDTA, and 7% sodium dodecyl sulfate (SDS) at pH 7.2. After hybridization, the blots were washed twice at room temperature in 2 times; SSC, 0.1% SDS, and then with 0.4 × SSC, 0.1% SDS at 65°C. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) hybridization was used as a reference for relative mRNA per lane. Autoradiography was performed with Amersham hyperfilm (Amersham) and intensifying screens (Kodak) at −70°C.
The signal intensity of PDGF β-receptor was analyzed with a densitometer interfaced with an integrator (Shimadzu) at each time point in four separate experiments. The optical densities from the four experiments were combined to obtain the mean value at each time point, and the mean value from these experiments was expressed as fold increase over the normal controls.
Western blot analysis
To confirm crossreactivity of the rabbit anti-mouse antibody to the β-receptor (Oncogene Science) of the rat brain, immunoblot analysis was performed. The cerebral cortex of the SHR was removed under deep anesthesia, quickly frozen in liquid nitrogen, and disrupted in 50 mM Tris-HCl pH 7.4, 0.32 M sucrose, 2 mM EDTA, 0.5 mM EGTA, 2 mM phenylmethylsulfonyl fluoride, 10 μg/ml aprotinin, 5.0 μg/ml pepstatin, 0.5 μg/ml antipain, and 1.0 mM benzamidine with a Teflon homogenizer. The lysate was centrifuged at 10,000 g for 10 min at 4°C. The resulting supernatant was further centrifuged at 100,000 g for 30 min at 4°C. Subsequently the supernatant was discarded and the pellet was suspended in the same buffer. Ten micrograms of the extract was separated using 2% SDS-polyacrylamide gel under reducing condition and transferred onto a polyvinylidene-difluoride membrane (Biorad). The PDGF β-receptor protein was detected using the rabbit anti-mouse PDGF β-receptor polyclonal antibody (1:5,000) with the avidin-biotin-peroxidase method. To confirm the specificity of the detected band, the primary antibody was preincubated with a 10-fold excess of the immunizing peptide (Oncogene Science) in otherwise the same procedure before the immunoblot analysis.
Preparation of frozen sections
At 2 and 8 h and 1, 2, 4, 7, 10, 14, and 21 days after MCA/CCAO and 1 and 4 days after sham operations (n = 3 each), the animals were anesthetized, and decapitated after the brains were perfused with cold normal saline and then with a combination of 0.01 M NaIO4, 0.75 M L-lysine, and 2% paraformaldehyde in 0.075 M phosphate-buffered saline (PBS) at pH 7.4. The brains were removed, cut into 2-mm-thick coronal sections, and postfixed with the same fixative at 4°C overnight before being incubated sequentially in 10 and 15% sucrose in PBS for 24 h. Specimens were embedded in optimal cutting compound and frozen by immersion in liquid nitrogen. Serial 8-μm-thick frozen coronal sections were cut using a cryostat and mounted onto poly L-lysine-coated glass slides for hematoxylin-eosin staining and for immunohistochemical examination.
Immunohistochemistry
Immunostaining for PDGF β-receptor was performed on frozen sections using a rabbit antimouse PDGF β-receptor polyclonal antibody (Oncogene Science) by the streptavidin-immunoperoxidase method. This antibody is raised against the synthetic peptide, which corresponds to amino acid 425–446 of murine PDGF β-receptor. We used this antibody in the experiment because this amino acid sequence is identical to the corresponding region of the rat receptor (Herren et al., 1993). The slides were developed using 3,3′-diaminobenzidine as a chromogen. To determine the specificity of the immunostaining in the lesion, the primary antibody was substituted for nonimmune rabbit IgG at equivalent concentration or for the primary antibody preincubated with a ten-fold excess of synthetic peptide of PDGF β-receptor (Oncogene Science) in otherwise the same procedure. To identify macrophages, lectin histochemistry was performed using Ricinus communis agglutinin-120 (RCA-1) (Seikagaku, Tokyo, Japan) (Mannoji et al., 1996). After PBS washes, endogenous peroxidase activity was blocked by incubating the slides with 0.3% H2O2 for 20 min. After PBS washes, incubation with RCA-1 was performed at 4°C overnight. The slides were developed after additional washes in PBS, using 3,3′-diaminobenzidine as already mentioned.
Immunohistochemical staining for glial fibrillary acidic protein (GFAP) was carried out using rabbit polyclonal antibody against GFAP (Dako, Kyoto, Japan). Immuno-reaction was visualized by the streptavidin-immunoperoxidase method using Histofine SAB-PO kits following the manufacturer's instructions (Nicheri, Tokyo, Japan).
Statistical analysis
The results of Northern analysis are expressed as fold increase over normal control (means ± SD). The significance was tested with one-way analysis of variance (ANOVA) followed by Tukey's test. When the p value was <0.05, differences were considered statistically significant.
RESULTS
Northern blot analysis
Northern blot analysis revealed weak but distinct constitutive expression of PDGF β-receptor (5.6 kb in size) in normal neocortex of the rat brain. Ischemic insult dramatically increased mRNA expression of the β-receptor in the injured neocortex at 4 and 7 days (328 ± 109%, 323 ± 119%; n = 4, respectively, over normal control) post-MCA/CCAO. The increased expression of the receptor was significantly higher as compared with that of the sham operation (p < 0.05; one-way ANOVA followed by Tukey's test). This enhanced expression of the β-receptor rapidly declined after 7 days post MCA/CCAO (Figs. 1 and 2). Similar results were obtained in four independent experiments.
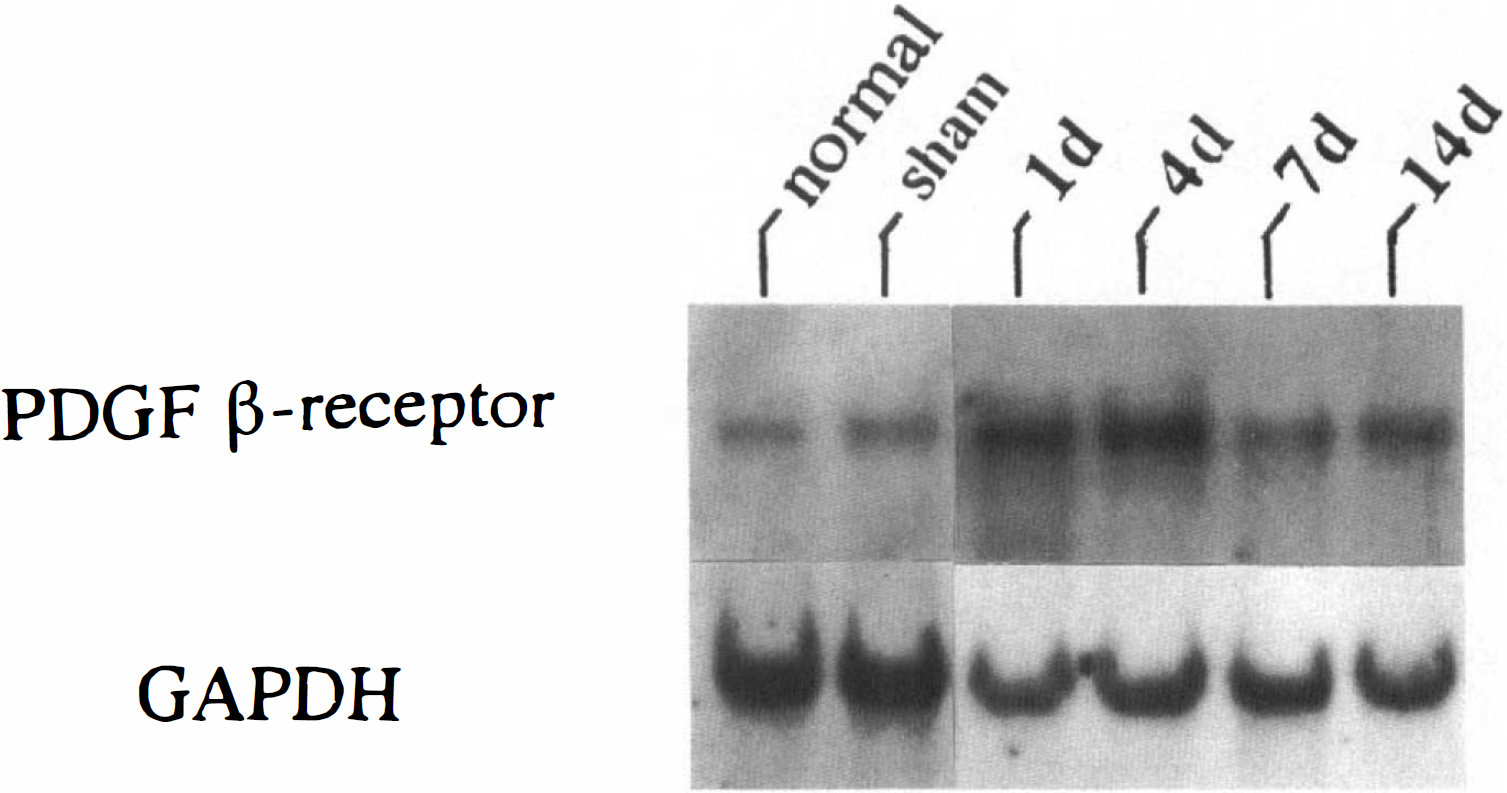
Northern blot analysis of platelet-derived growth factor (PDGF) β-receptor and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) in the rat neocortex ipsilateral to the focal ischemic lesion. Poly A+ RNA of 3.0 μg each, extracted from the right neocortices of 6-mm-thick coronal sections, were used for each lane. Ischemic insult markedly increased PDGF β-receptor mRNA expression at 4 and 7 days post-middle cerebral artery/common carotid artery occlusion.
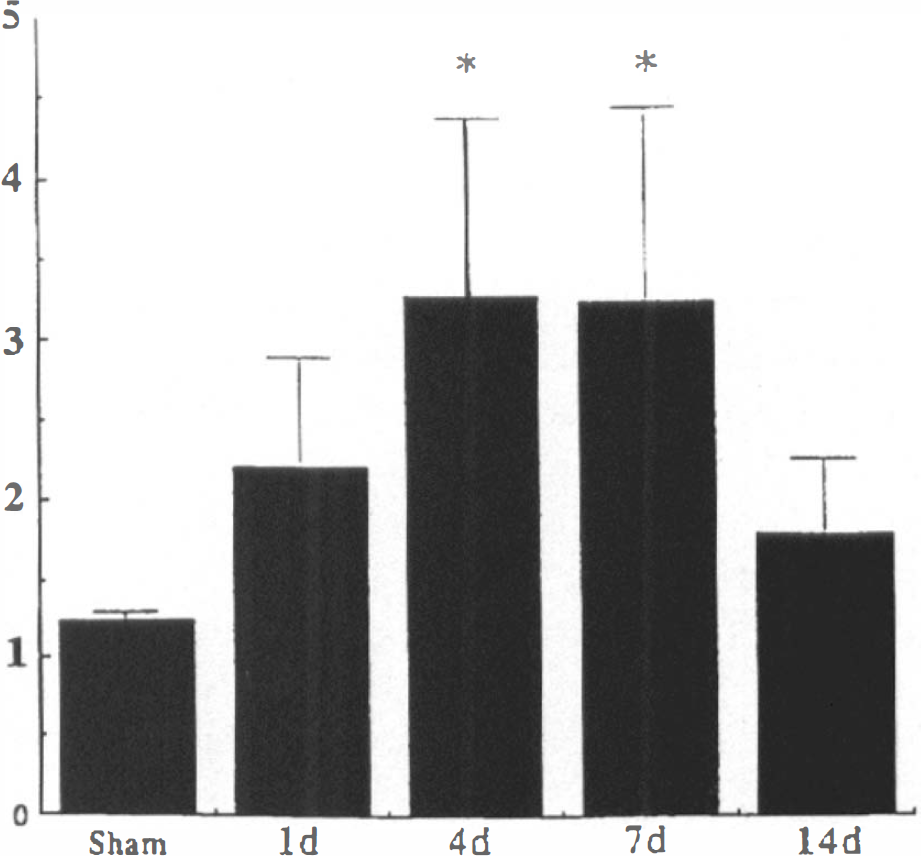
Densitometric analysis of mRNA level of platelet-derived growth factor (PDGF) β-receptor in the ischemic neocortex after middle cerebral artery/common carotid artery occlusion (MCA/CCAO). Symbols and bars represent means and SD, respectively, of four separate experiments (n = 4, each). The relative abundance of PDGF receptor mRNA was corrected by the expression of glyceraldehyde 3-phosphate dehydrogenase and measured in comparison with a normal control. The normal control values were taken as 1. *p < 0.05 between ischemic animals versus the animals that received sham operation (significance tested with one-way analysis of variance followed by Tukey's test).
Western blot analysis
Immunoblot analysis revealed that the antibody used recognizes two bands of 134 and 80 kDa, respectively. The detected bands were completely abolished by neutralizing the primary antibody with the immunizing peptide in otherwise the same procedure (Fig. 3).
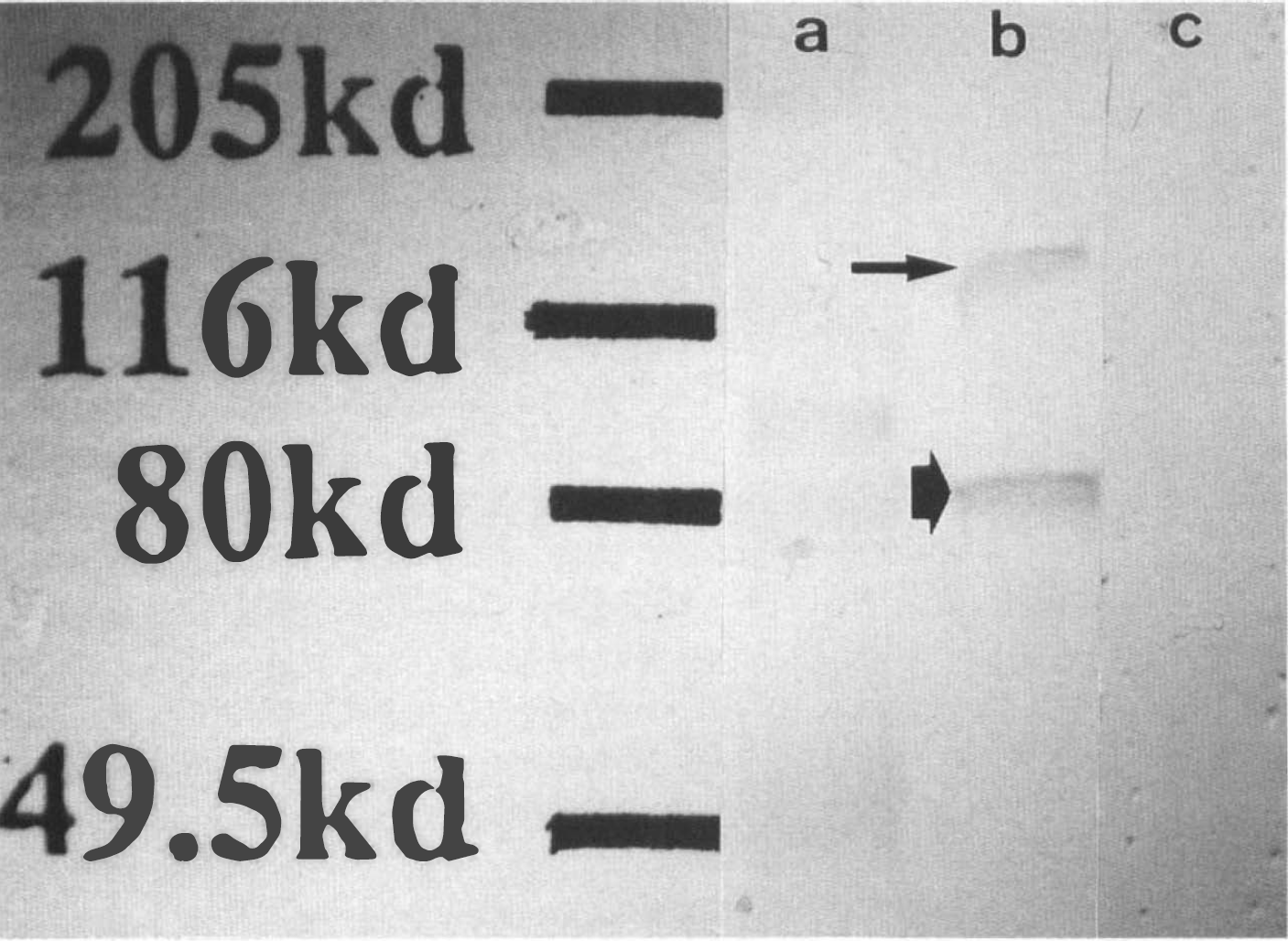
Immunoblot analysis on the homogenate of the normal cortex from spontaneously hypertensive rats showed a dense band of 80 kDa and a fainter band of 134 kDa (lane b) for platelet-derived growth factor (PDGF) β-receptor. These immunoreactions were completely abolished by neutralization of the primary antibody with the immunizing peptide of the mouse PDGF β-receptor (lane c). Lane a, size marker.
Histological examination
In normal brain, immunostaining for the PDGF β-receptor revealed ubiquitous neuronal expression throughout the brain. The immunoreactive products were located mainly on the cell membranes of neuronal cell bodies and processes (Fig. 4A). The interfascicular glial cells in the subcortical white matter including the external and internal capsules, corpus callosum, olfactory tract, and optic tract, also showed weak immunoreactivity as well (Fig. 5). In contrast, glial cells in the gray matter showed no immunoreactivity.
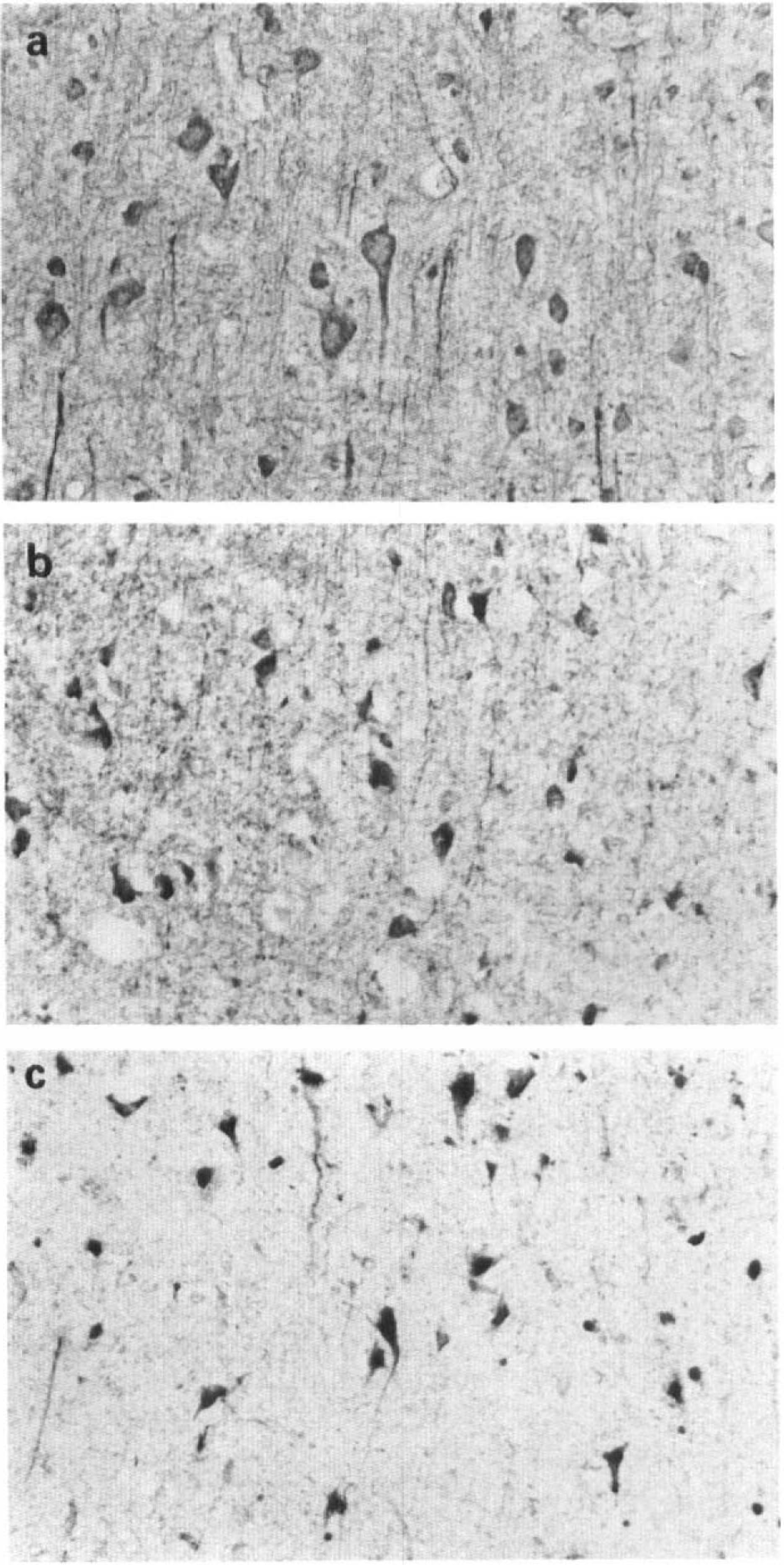
Platelet-derived growth factor β-receptor immunoreactivity in neurons in the normal brain (A), in the periinfarct area (B), and in the infarct (C) at 24-h post-middle cerebral artery/common carotid artery occlusion. Neurons both in the periinfarct area and in the infarct showed an enhanced immunostaining as compared with those in the normal brain.
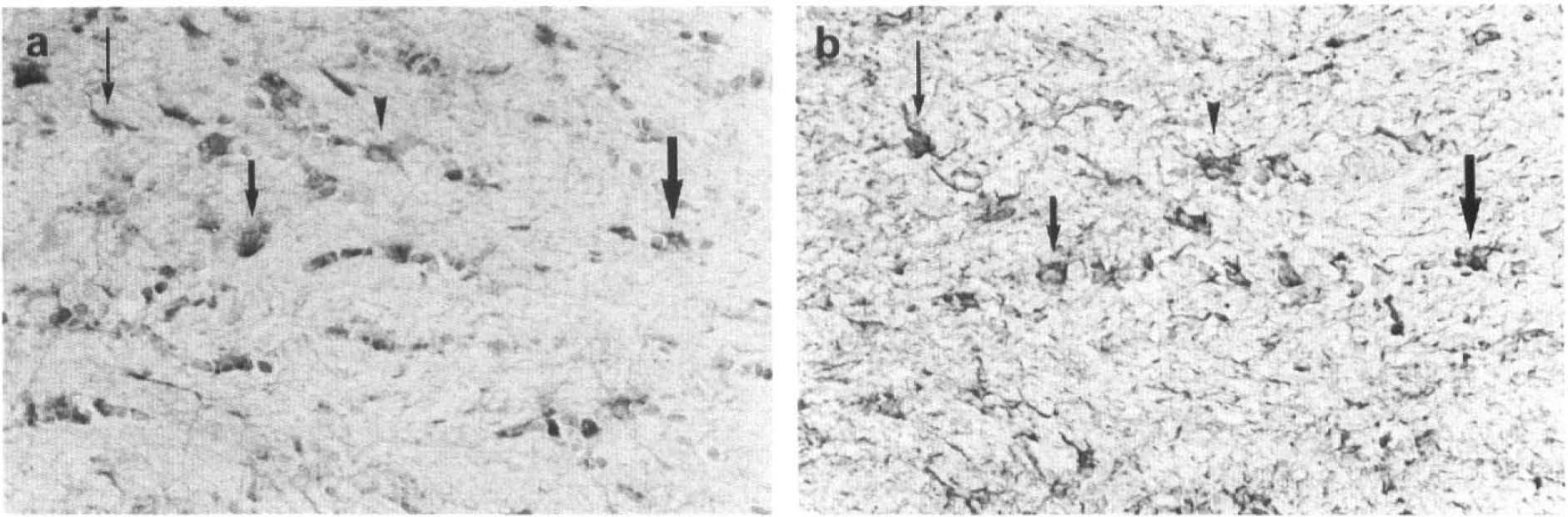
Platelet-derived growth factor β-receptor (a) and glial fibrillary acidic protein (GFAP) immunoreactivity (b) in the glial cells in the external capsule at 4 days post-middle cerebral artery/common carotid artery occlusion. Markedly enhanced PDGF β-receptor-like immunoreactivity was shown on GFAP-positive and hypertrophied glial cells in the external capsule.
In the MCA/CCAO groups, large neocortical infarcts, extending into the dorsolateral caudoputamen in some animals, were produced in all animals as reported previously (Brint et al., 1988; Iihara et al., 1994a, 1994b). At 24 h post MCA/CCAO, the shrunken angular neurons in the periinfarct (Fig. 4B) and the neurons in the infarct area (Fig. 4C) showed moderately enhanced immunoreactivity for the β-receptor. Although the immunoreactivity in the shrunken neurons in the infarct gradually decreased thereafter, the neurons in the periinfarct area sustained an increase in immunostaining from 1 day to 14 days post MCA/CCAO (Fig. 6B).
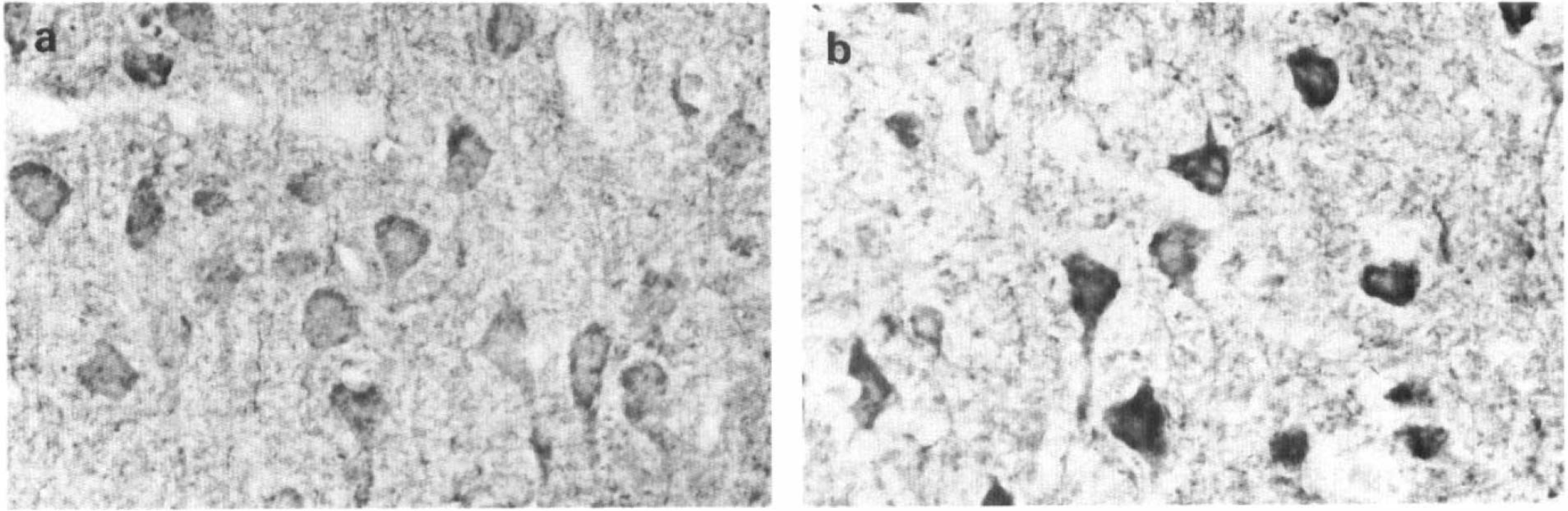
Platelet-derived growth factor-β-receptor immunoreactivity in neurons in the normal (a) and in the periinfarct area (b) at 4 days post-middle cerebral artery/common artery occlusion. Neurons in the periinfarct area sustained markedly enhanced immunoreactivity as compared with those in the normal brain.
GFAP-positive, reactive glial cells accumulated in the neocortex and the subcortical white matter surrounding the infarct from 24 h to 21 days post-MCA/CCAO. The hypertrophy and hyperplasia of the glial cells were more pronounced in the subcortical white matter just beneath the infarct than in the neocortex throughout the experiment and spread into the contralateral external capsule from 4 days to 21 days post-MCA/CCAO. Hypertrophic change in the glial cells in the external capsule was most prominent at 7 days post-MCA/CCAO. Immunohistochemistry for the β-receptor on serial sections showed that GFAP-positive, reactive glial cells in the external capsule showed intense immunoreactivity at 4 and 7 days (Fig. 5). The immunoreactivity in these cells gradually decreased thereafter but was still enhanced at 14 and 21 days post MCA/CCAO (Fig. 7). On the other hand, GFAP-positive reactive astrocytes in the neocortex, except in the molecular layer, showed no immunoreactivity for the β-receptor throughout the experiments.
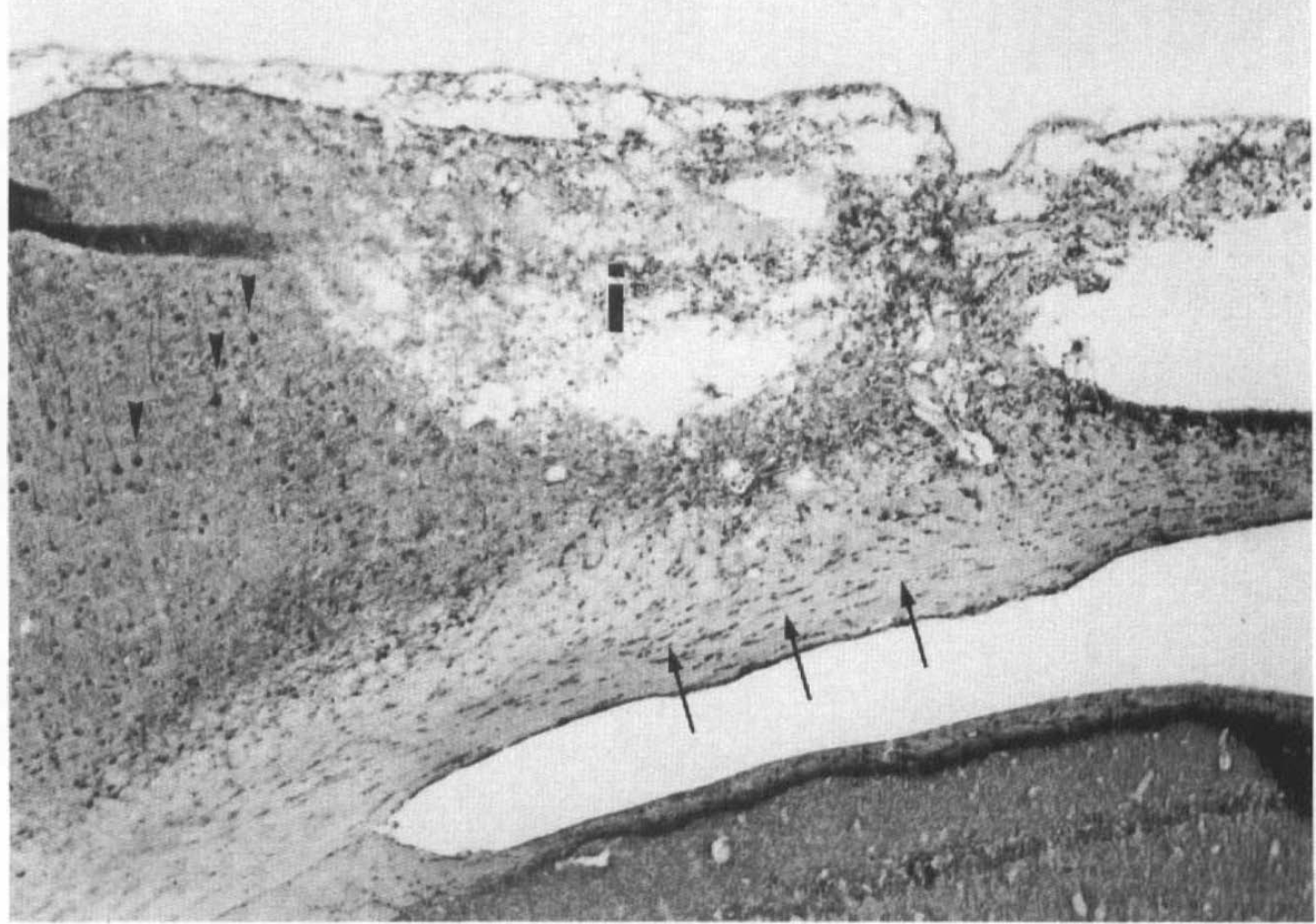
Platelet-derived growth factor-β-receptor immunoreactivity at 14 days post-middle cerebral artery/common carotid artery occlusion showed sustained and enhanced immunostaining in neurons in the periinfarct area (arrowheads) and in reactive glial cells in the external capsule (arrows) adjacent to the infarct (i).
RCA-1 histochemistry revealed the accumulation of brain macrophages in the infarct from 2 to 14 days with a peak at 7 days post-MCA/CCAO as described previously (Iihara et al., 1994a, 1994b). Immunostaining for the β-receptor on serial sections showed intense immunoreactivity on the cell membranes of these cells at 4 and 7 days post-MCA/CCAO (Fig. 8). No immunoreactivity was observed on the brain macrophages at 10 or 14 days post-MCA/CCAO.
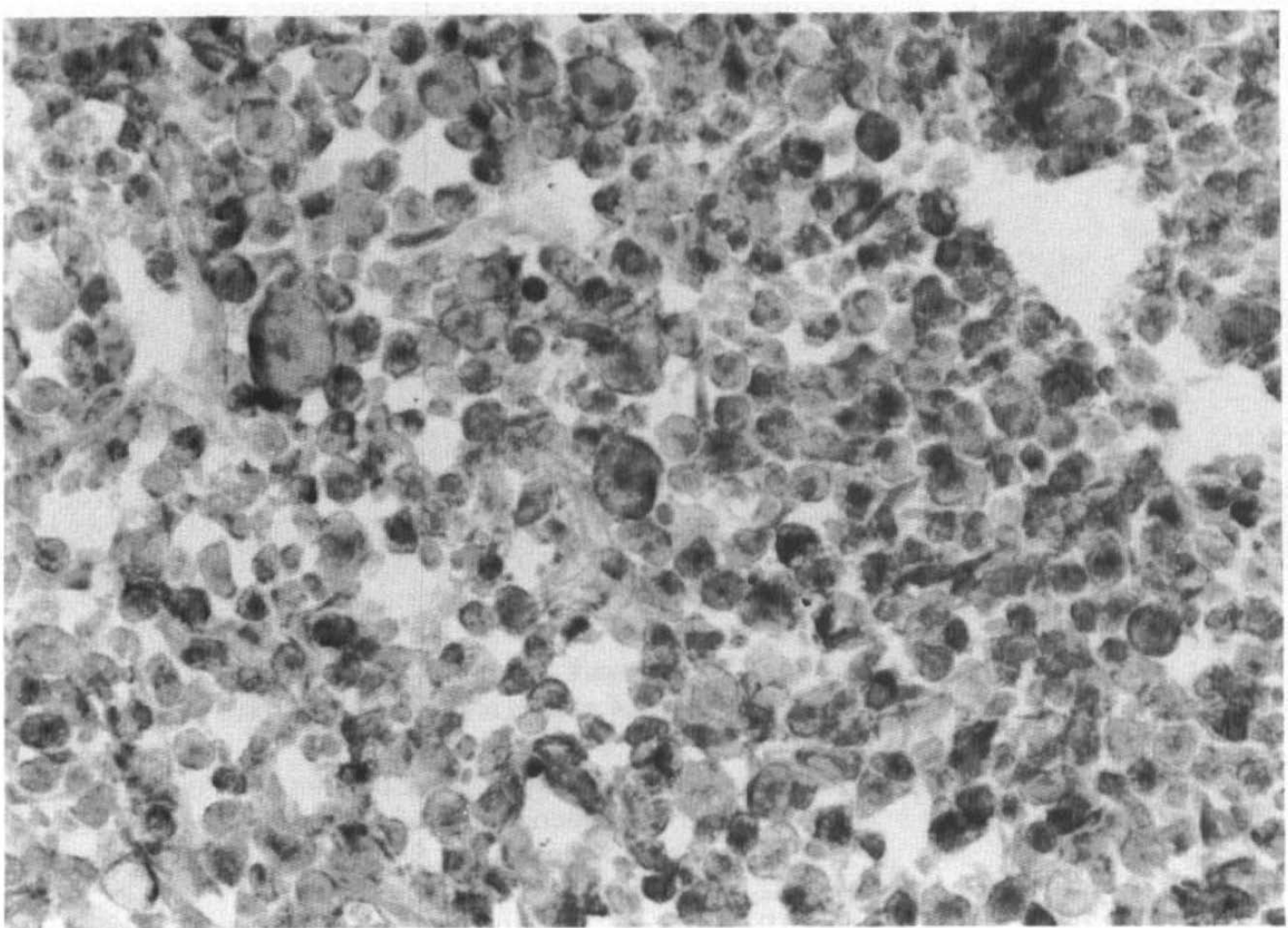
Platelet-derived growth factor-β-receptor immunoreactivity was observed in the brain macrophages migrating to the infarct at 7 days post-middle cerebral artery/common carotid artery occlusion.
The immunoreactivity for the β-receptor was also shown to be associated with the abluminal side of the capillary vessels at 4 and 7 days post-MCA/CCAO (Fig. 9).
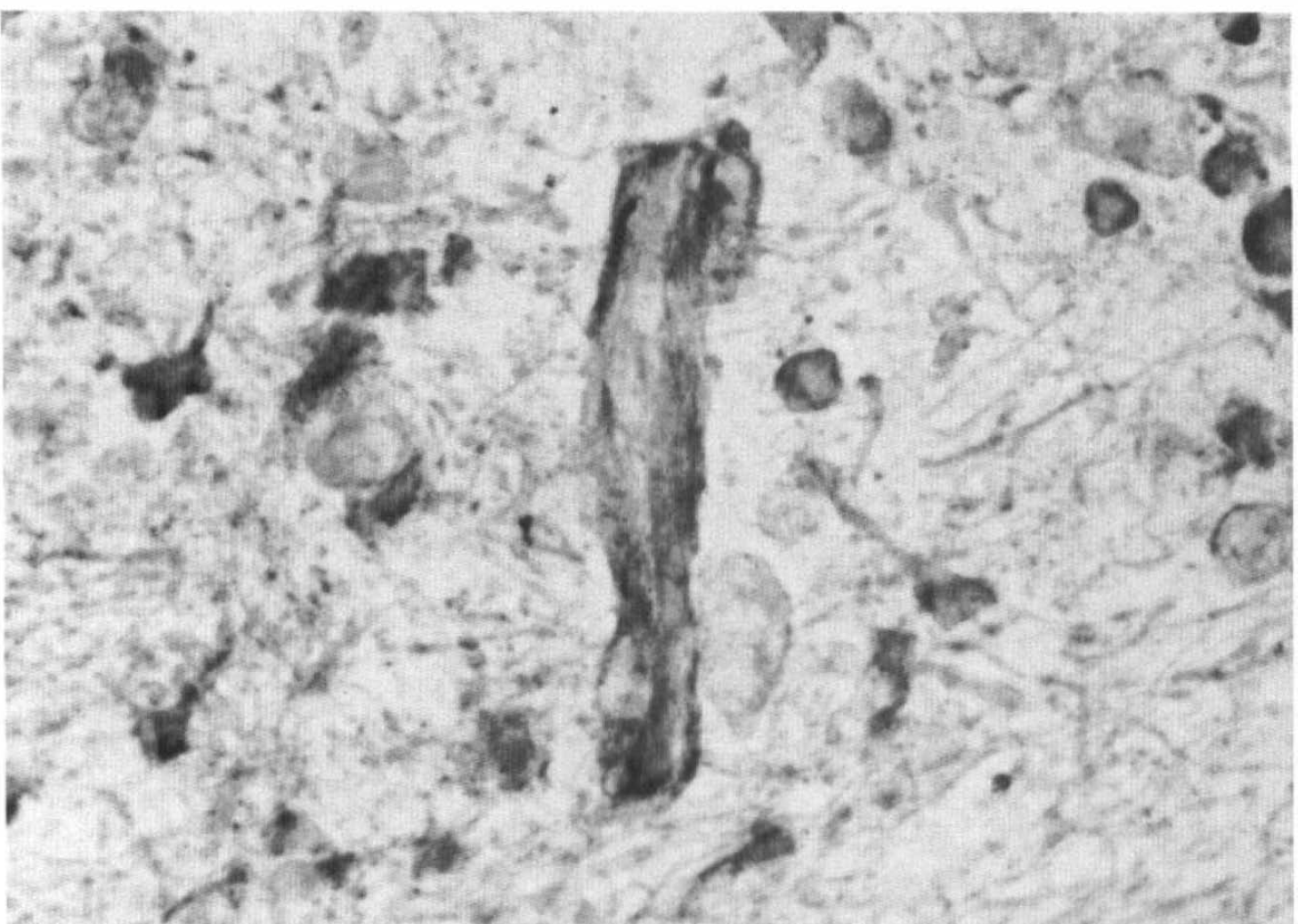
Platelet-derived growth factor-β-receptor immunoreactivity associated with capillary microvessies in the external capsule at 4 days post-middle cerebral artery/common carotid artery occlusion. Marked immunoreactivity was seen mainly on cells on the abluminal side of the capillaries.
Neutralizing the primary antibody with synthetic peptide of PDGF β-receptor and replacement of the primary antibody with nonimmune rabbit IgG both completely abolished the immunostaining already described.
DISCUSSION
Recently, we showed that ischemia induces mRNA and protein expression of the PDGF-B chain, but not of the PDGF-A chain, in the rat brain and suggested an important role of the B-chain in a variety of cellular responses after cerebral injury (Iihara et al., 1993; Iihara et al., 1994b). This view was further supported by our following work on the stab wound model in the rat brain (Takayama et al., 1994). The results of the present study demonstrated, for the first time, that ischemic insult to the brain markedly increases PDGF β-receptor mRNA expression at 4 and 7 days postischemia. This reversible, controlled expression of the β-receptor reinforces our hypothesis that the PDGF-B chain plays an important role in the normal repair process of the injured brain through binding to PDGF receptors on responsive cells (Sasahara et al., 1991; Iihara et al., 1993; Iihara et al., 1994b; Takayama et al., 1994), because PDGF β-receptor binds only to the B chain with high affinity (Heldin et al., 1990). This in vivo coordinate expression of PDGF/PDGF receptors initiated by ischemic injury should amplify the cellular responses in the damaged brain (Iihara et al., 1994a), as suggested previously with the cutaneous injury model (Antoniades et al., 1991).
Immunoblot analysis demonstrated that the antibody used detects a dense band of 80 kDa and a fainter band of 134 kDa of the β-receptor protein. The specificity of the immunoreaction was confirmed by the neutralization of the primary antibody with the immunizing peptide. These results are similar to those of the previous study on the developmental distribution of the PDGF and the β-receptor in the mouse CNS (Hutchins and Jefferson, 1992). This study disclosed that the same antibody recognizes two bands of the β-receptor of 129 and 65 kDa, respectively.
Immunohistochemistry for the β-receptor in the normal rat brain showed moderate staining in neurons throughout the brain and weak staining in interfascicular glial cells in the subcortical white matter but not in the capillary endothelial cells. These results confirmed neuronal expression of the β-receptor reported previously both in vivo and in vitro (Smits et al., 1991; Giacobini et al., 1992; Hutchins and Jefferson, 1992; Yamada et al., 1994). However, the present findings were inconsistent with those of previous immunohistochemical studies, which have shown a positive staining in capillary endothelial cells (Smits et al., 1989) but no staining in glial cells in the brain (Smits et al., 1991). This difference in immunoreactivity in these cells cannot be fully explained, but may be due to the difference in fixatives or in the antibodies used in these studies.
In the present study, neurons in the infarct showed a moderate increase in the immunoreactivity at 1 day, but this immunostaining decreased rapidly thereafter as the neuronal degeneration progressed. This transient increase in immunoreactivity is in striking contrast to the sustained and marked increase in the immunoreactivity in neurons in the periinfarct area to 14 days post-MCA/CCAO. Our previous study revealed that PDGF-B chain-like immunoreactivity is increased in neurons both in infarct and periinfarct areas from 16 h to 5 days and 7 days postinsult, respectively. Taken together, these findings raise the possibility that sustained and enhanced expression of both the B-chain and the β-receptor is necessary for the survival of neurons at risk after ischemic insult to the brain, because the PDGF-B chain was reported to possess neurotrophic activities through binding to the β-receptor (Smits et al., 1991, Giacobini et al., 1992) In addition, the sustained expression of the β-receptor in neurons at risk in the periinfarct areas indicates that the brain macrophage-derived PDGF-B chain may also exert a neurotrophic effect on the neurons at risk in a paracrine fashion in the ischemic brain. Further study to examine the effects of antagonists or agonists of the receptor on neuronal death in the ischemic brain is required to make conclusions on this view.
Another important finding of this study is PDGF β-receptor expression in the brain macrophages that accumulated in the infarct at 4 and 7 days after ischemic insult. The presence of β-receptors on macrophages has only recently been reported (Inaba et al., 1993). In this in vitro study, PDGF β-receptor expression was associated with maturation and differentiation of cultured human monocyte-macrophages through the activation of protein kinase C. In this regard, it is interesting to note our previous finding that the expression of PDGF-B chain in the brain macrophages is also induced in a similar time course, with a peak at 5–7 days post-MCA/CCAO, with some delay from their migration into the infarct at 2 days post-MCA/CCAO (Iihara et al., 1994b). Taken together, these results indicate that both PDGF-B chain and β-receptor are transiently coinduced in brain macrophages during the process of differentiation from monocytes migrating into the infarct. In addition, these findings support our view that the PDGF-B chain induced in the neurons at risk or the brain macrophages themselves contributes to the proliferation and migration of brain macrophages in the infarct (Iihara et al., 1994b) through its mitogenic and chemotatic actions (Ross et al., 1986; Deuel et al., 1982) by binding to the receptors upregulated on the macrophages.
The present study demonstrated a clear difference in immunostaining for the β-receptor in GFAP-positive reactive glial cells in the subcortical white matter adjacent to the infarct and in the neocortex except in the molecular layer. The mechanism responsible for the expression of the β-receptor in this specific glial population remains unknown. Several lines of evidence suggested that cerebral injury provokes a heterogeneous response in glial cells in the brain. Reactive astrocytosis that develops in the undamaged brain is reversible with prolonged survival, whereas reactive astrocytosis that accompanies structural brain damage persists for a prolonged period and is associated with hyperplasia, as well as hypertrophy (Petito et al., 1990). In addition, it was reported that in the stab-injury model of the rat brain, reactive glial cells doubly labeled with GFAP and [3H]thymidine are localized in the area adjacent to the lesion, in the molecular layer of the neocortex, and in the white matter. In contrast, none of the cells was doubly labeled in the second to sixth layers of the neocortex (Takamiya et al., 1988). In view of these observations, the present study raises a possibility that enhanced expression of the β-receptor in specific reactive glial cells contributes to the proliferation of these cells adjacent to the infarct in the ischemic brain. Double labeling experiments using bromodeoxyuridine (BrdU) and β-receptor antibodies should help elucidate its role. This enhanced expression of the β-receptor in the glial cells surrounding the lesion indicates that PDGF-B chain induced in the brain macrophages and neurons at risk may influence the proliferation of glial cells in the injured brain as suggested previously (Iihara et al., 1994a and 1994b; Takayama et al., 1994). Although the nature of the β-receptor-positive cells in the abluminal side of capillaries remains to be determined, the expression of the β-receptor in these cells may be involved in the neovascularization in the injured brain.
In conclusion, the present study demonstrated enhanced expression of the PDGF β-receptor at both mRNA and protein levels in the ischemic brain. The close temporospatial synchronization between the expression of the receptor and the PDGF-B chain reported previously (Iihara et al., 1994b) suggests an important role in a variety of cellular responses such as glial proliferation, migration and proliferation of brain macrophages, and neovascularization as well as the survival of neurons at risk in the ischemic brain.
Footnotes
Acknowledgment:
We thank Zynogenetics for the gift of cDNA probes for the PDGF-β receptor. We also thank Drs. Eiji Yamada and Yoneko Hayase, Mr. Hideo Kataoka (Shiga University of Medical Science), Dr. Yasunobu Goto (National Cardiovascular Center), and Mr. Masahiro Sakata (Kyoto Pharmaceutical University) for important scientific discussions. This work was supported in part by a Grant-in-Aid for Scientific Research from the Ministry for Education, Science and Culture of Japan and by Special Coordination Funds for Promoting Science and Technology (Encouragement System of COE) from the Science and Technology Agency of Japan.
