Abstract
Brain pericytes not only maintain the anatomical, biochemical and immune blood–brain barrier, but display features of mesenchymal stem cells (MSCs) in vitro. MSCs have pro-regenerative properties attributed to their secretome. However, whether also brain pericytes possess such pro-regenerative capacities is largely unknown. Here we characterize the secretome and microvesicle (MV) release of human brain pericytes mediated by platelet-derived growth factor-BB (PDGF-BB)/PDGF receptor beta (PDGFRβ) signalling. Upon PDGF-BB, pericytes release not only a plethora of growth factors and a panel of cytokines, but also MVs containing BDNF, FGFb, βNGF, VEGF and PLGF, a response that is specific for PDGFRβ signalling and activation of the ERK 1/2 pathway. In contrast, lipopolysaccharide (LPS), an activator of the innate immune system, stimulates the secretion of much higher amounts of mainly inflammatory cytokines and activates the NFκB pathway. Pericytes change their morphology and undergo opposite changes in surface marker expression, respectively. Our findings provide evidence that the secretome of human brain pericytes varies greatly depending on the exogenous stimulus. The differential secretory functions of pericytes may play an important role in either regulating neuroinflammation or contributing to neurorestoration and identify a possible new target cell for neuroregeneration.
Keywords
Introduction
Brain pericytes are perivascular cells that are uniquely positioned at the blood–brain interface. Pericytes maintain the anatomical, biochemical and immune blood–brain barrier (BBB) and regulate capillary flow, angiogenesis, clearance of toxic byproducts and immune responses.1,2 This strategic position and their multiple functions indicate a crucial role in the pathogenesis of brain disorders. In other tissues, pericytes are known to promote repair and regeneration by contributing to wound healing. 3 Their possible reparative function in the brain, however, is largely unknown.
Pericytes highly express the receptor for platelet-derived growth factor-BB (PDGF-BB) (PDGFRβ). PDGF-BB signalling is important for pericyte recruitment to the blood vessels, proliferation and migration. 4 PDGFRβ-positive cells are, e.g. increased after stroke,5,6 suggesting an important role for PDGFRβ signalling. Interestingly, PDGF-BB has been shown to have a partial restorative effect on the nigrostriatal system in Parkinson’s disease (PD),7,8 findings that have led to clinical trials investigating PDGF-BB as a possible neurorestorative compound in PD. 9 How this restorative effect of PDGF-BB is mediated is currently not known. Most importantly, the function of PDGF-BB/PDGFRβ activation in pericytes with respect to secretion of growth factors has so far not been investigated.
Pericytes share several features of mesenchymal stem cells (MSC) when isolated and expanded in vitro, such as surface marker expression and mesodermal differentiation capacity.10,11 MSCs have considerable pro-regenerative, trophic and anti-inflammatory capacities when grafted, features that may also apply to pericytes. 12 Pericytes seem to play a role in the cerebral immune response. Evidence is currently accumulating that brain pericytes not only control immune cell recruitment 13 in response to certain pathological stimuli, but may also adopt a microglial phenotype in vitro and in vivo14,15 and secrete cytokines in vitro.16,17 These secretory capacities of pericytes in response to different exogenous stimuli may be a previously unrecognised feature of pericytes and particularly important for their contribution to regeneration.
MSCs are widely used for grafting in clinical trials due to their trophic and immunomodulating capacities. It has been shown that MSCs release microvesicles (MVs), small cell membrane particles that contain complex biological information (e.g. lipids, microRNAs, proteins). Microvesicles allow the emitting cell to modify gene and protein expression in the receiving cell and enable cell communication over long distances. 18 Mesenchymal stem cell-derived MVs are currently gaining attention as a new source of protective and regenerative molecules that modulate the microenvironment in neuroinflammatory and neurodegenerative disorders.19,20 It is, however, not known whether pericytes release MVs with similar properties as MSCs.
Here we show that human brain pericytes release neurotrophic growth factors (brain-derived neurotrophic factor (BDNF), nerve growth factor beta (βNGF)) and a panel of cytokines as well as MVs, in response to PDGF-BB. We characterize the secretome and the composition of MVs released by pericytes upon PDGF-BB stimulation and compare their cytokine release to stimulation with lipopolysaccharide (LPS), an activator of the innate immune system used to induce immunoreactivity of brain pericytes.16,21 Finally, we analyze cell-signalling mechanisms of brain pericytes after PDGF-BB and LPS treatment in vitro and demonstrate the specific PDGF-BB/PDGFRβ-mediated secretome.
Material and methods
Primary human brain pericytes
Brain pericyte lines were obtained from adult human brain tissue, as previously described.14,22 All procedures were performed with informed written consent by the patient for the donation of brain tissue and approved by the ethical committee of the Scania University Hospital, Lund, Sweden, according to the Declaration of Helsinki Principles.
Briefly, brain tissue was previously harvested from individuals undergoing surgery for ventriculostomy or shunt-placement (n = 2) or surgery for intractable temporal lobe epilepsy (n = 2). Fresh tissue samples were stored in Leibowitz-15 media (Invitrogen) at 4℃, cut and enzymatically digested in enzyme solution (Collagenase 1 mg/ml (Sigma); Dispase 1.6 mg/ml (Roche); Trypsin 0.25 mg/ml (Sigma); DNase I 80 U/ml (Sigma) in Dulbecco’s modified Eagle Media (DMEM) and 4.5 mg/ml glucose (Invitrogen) at 37℃/20 min. Cells were plated on 24-well culture dishes and incubated at 37℃/5% CO2 in DMEM/F-12/Glutamax/B27 (Invitrogen).
For this study, pericytes were expanded at passage 34 in Stemline medium (Sigma-Aldrich) supplemented with 2% fetal bovine serum (Invitrogen), 1% Penicillin/Streptomycin (Gibco), 20 ng/ml basic fibroblast growth factor (bFGF, Invitrogen) on gelatin-coated culture flasks (Nunc) and incubated at 37℃ in 5% CO2 conditions (Heraeus HERAcell 150 CO2 incubator, Thermo Scientific). Cells grew exponentially with a doubling time of approximately 48 h, reaching ca. 85% confluence after 48–72 h. The cells were seeded in six-well culture plates at 100,000 cells/well for the following experiments.
MTT assay
Human brain pericytes were plated in 96-well plates at a density of 3 × 103 cells per well, treated with PDGF-BB (20 ng/ml, 50 ng/ml, RD systems) or LPS (10 µg/ml, 50 µg/ml, Sigma) and incubated for 72 h at 37℃ in 5% CO2. The proliferation ability was assayed by using 3 -(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT). The absorbance was read at a wavelength of 570 nm in ASYS Expert 96 microplate reader.
Flow cytometry
Human brain pericytes were treated with PDGF-BB (20 ng/ml) or LPS (50 µg/ml) for 72 h, then harvested (20,000 cells/condition) and washed three times with phosphate-buffered saline. Cells were incubated with anti-human CD140b-PE, anti-human CD14-FITC, anti-human CD11b-PE, anti-human CD31-FITC, anti-human CD45-PerCP (all BD Biosciences, San Jose, CA) and anti-human CD146-FITC (AbD Serotec) fluorochrome-conjugated antibodies, in separated tubes for 45 min at 37℃ in the dark. IgG1-PE, -FITC or -PeCy5 mouse antibodies (all BD Biosciences) were used as isotype control to set threshold and gates in the cytometer. Finally, samples were analyzed by flow cytometry with an Accuri C6 (BD Biosciences). A total of 500,000–1000,000 events were recorded for each sample and analyzed with the FACS Diva software (Tree Star, Inc.).
Growth factor screening analysis
Forty-one growth factors were analyzed in the cell supernatant of human pericytes under control conditions and after treatment with PDGF-BB (20 ng/ml) for 72 h. Growth factor production was evaluated by Ray Bio Human Growth Factor Antibody Array C1, following the manufacturer instructions (RayBiotech, Inc.). Images were acquired using a ChemiDoc™ MP System (BioRad) and densitometry was quantified using the Image J software (National Institutes of Health).
Microvesicle preparation and quantification
MVs were isolated from the cell supernatant of control, PDGF-BB-treated (20 ng/ml) or LPS-treated (50 µg/ml) pericyte cultures for 72 h by successive centrifugations. Briefly, cell medium was centrifuged at 1500g for 5 min and the supernatant free from cells debris was collected. In order to pellet MVs, the supernatant was centrifuged at 16,000 g for 45 min. MVs were washed three times in 1 ml of 0.9% NaCl at 16,000 g for 45 min. After that, the pellet of MVs was suspended in 100 µl of 0.9% NaCl and stored at 4℃ until subsequent analysis. For quantification, MVs were analyzed by flow cytometry (flow cytometer with an Accuri C6; BD Biosciences) using flow count beads (Beckman Coulter). The region corresponding to the total number of MVs was identified in FSC and SSC intensity dot plot representation, set at logarithmic gain, according to their diameter (0.1–1 µm), compared to the beads size. Samples were analyzed until 10,000 events occurred.
Cytokine screening
Forty-two cytokines were analysed in cell supernatant of control cultures, PDGF-BB-treated (20 ng/ml) or LPS-treated (50 µg/ml) pericyte cultures for 72 h. Cytokine production was evaluated by Ray Bio Human Cytokine Antibody Array C3 following the manufacturer instructions (RayBiotech, Inc.). Images were acquired using a ChemiDoc™ MP System (BioRad) and densitometry was quantified by Image J software.
Multiplex sandwich Elisa
Human growth factors and cytokines were quantified from medium or MVs of controls or PDGF-BB-treated (20 ng/ml, 72 h) pericytes with or without a 1 h Sunitinib (PDGFRβ kinase inhibitor, 2 µM, Selleck Chemicals) exposure prior treatment, or of LPS-treated (50 µg/ml, 72 h) pericytes, using human multiplex sandwich Elisa (Meso Scale Discovery, Gaithersburg, MD), according to the manufacturer instructions. Each sample (25 µl) was measured in duplicate, and the mean was used in the statistical analysis. The coefficient of variation was below 20% for all assays.
Western Blot analysis
Proteins were purified from pericytes treated with (i) PDGF-BB (20 ng/ml) for 15 min, 90 min or 72 h, (ii) Sunitinib (2 µM) for 1 h before treatment with PDGF-BB (20 ng/ml) for 15 min, 90 min or 72 h, or (iii) LPS (50 µg/ml) for 24 h. Briefly, cell pellets were resuspended and incubated in RIPA lysis buffer (Thermo Scientific) with phosphatase and protease inhibitors (Thermo Scientific) 15 min on ice with gentle shaking. Samples were then centrifuged at ∼15,000 g for 15 min at 4℃, and the supernatant protein concentration was quantified with BCA Protein Assay (Pierce). For Western blot analysis, 10 µg of the total proteins were resolved by 4–15% acrylamide SDS-PAGE and incubated with the following primary antibodies: rabbit anti-phospho-PDGFRβ antibody (pPDGFRβ; 1:1000, Cell Signaling), rabbit anti-PDGFRβ (1:1000, Cell Signaling), rabbit anti-Extracellular signal regulated kinase (ERK; 1:1000, Cell Signaling), rabbit anti-phospho-ERK (pERK; 1:1000, Cell Signaling), rabbit anti-Nuclear factor-kappa B (NFκB; 1:2000, Abcam), rabbit anti-phospho-NFκB (pNFκB; 1:2000, Abcam) and anti-βactin-HRP antibody (1:10,000, Sigma). Bound antibodies were detected with an anti-rabbit-HRP antibody (1:3000, Dako). Images were acquired using a ChemiDoc™ MP System (BioRad) and densitometry was quantified using Image J software.
Statistics
Data were analyzed using GraphPad Prism Software and are expressed as mean ± standard deviation (SD). For the growth factor and cytokine screening arrays generating semiquantitative data, a one-sample two-tailed t-test was performed to compare each treatment to the control set at 100%. A two-way ANOVA followed by a Sidak post hoc was used for comparison between treatments. For three-group comparison, a one-way ANOVA with a Tukey post hoc. For two-group comparison, an unpaired two-tailed Student t-test was used. The p-value was corrected for multiple comparisons, and comparisons were considered significant with a p-value < 0.05.
Results
Human brain pericytes express typical surface markers
First, we verified the surface marker expression of the previously established adult human brain pericytes under control conditions (Supplementary Figure 1).
22
Using flow cytometry, cell surface marker expression was then analysed for the presence of the pericyte markers PDGFRβ (CD140b) and CD146 and for the absence of the monocyte/macrophage markers CD14, the microglial marker CD11b and the endothelial marker CD31 (Figure 1(a) to (f)). Our results confirmed that CD140b and CD146 were highly expressed on human brain pericytes (96.3% ± 0.15 and 61.9% ±1.56, respectively). These results confirm that proliferating pericytes are highly purified (Figure 1(a) to (f)).
22
Characterisation of primary human brain pericytes by flow cytometry. Untreated human brain pericytes were characterized by flow cytometry. (a) Cells were identified according to forward scatter (FSC) and side scatter (SSC) intensity. (b–f) Grey histogram represents isotype control, and blue histogram shows the fluorescence intensity of pericytes after incubation with CD140b, CD146, CD14, CD11b and CD31 antibodies, respectively.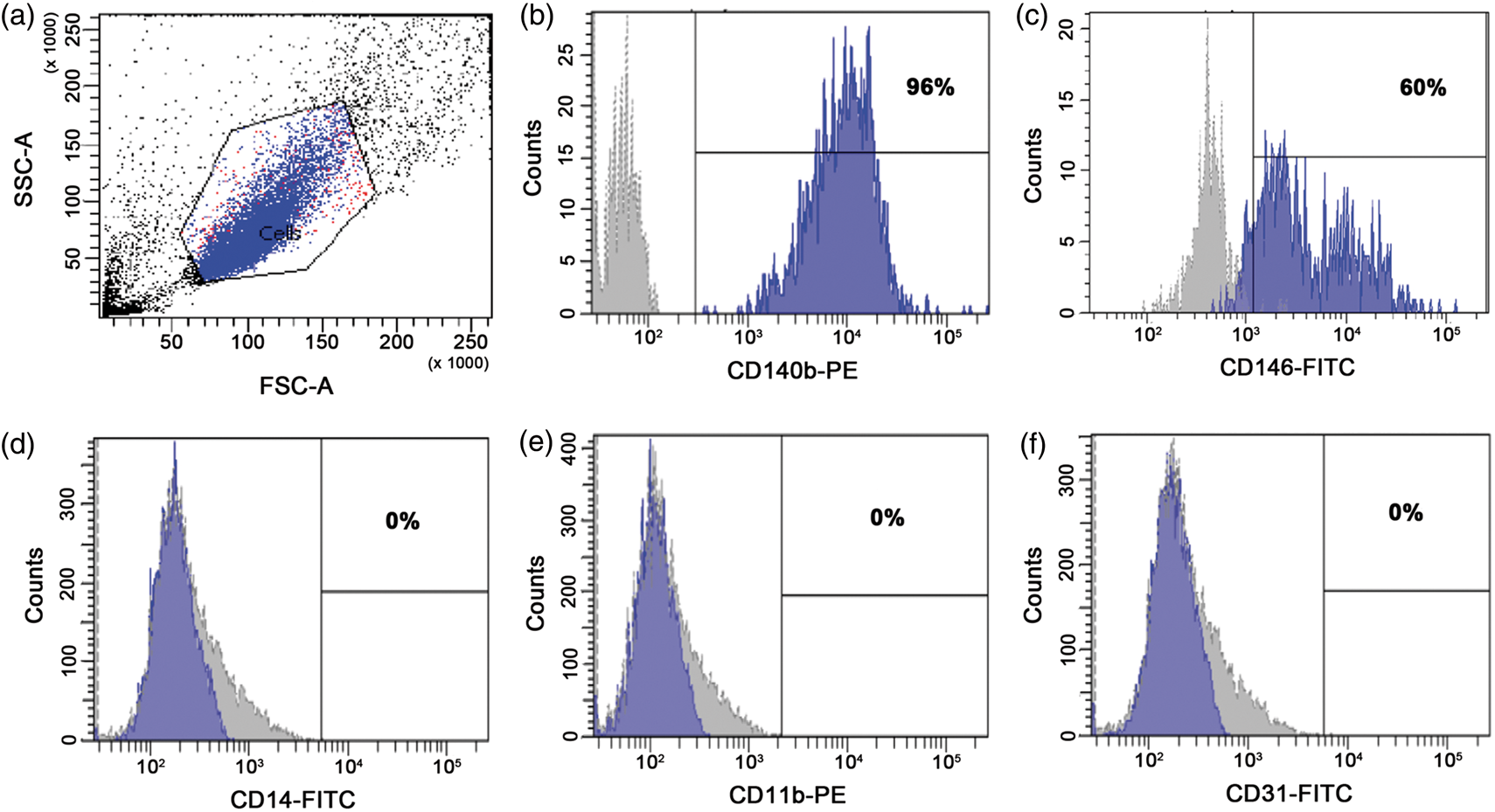
Pericytes secrete growth factors in response to PDGF-BB
In the central nervous system (CNS), PDGF-BB secreted by endothelial cells binds PDGFRβ on pericytes and stimulates proliferation and migration of pericytes and their recruitment to blood vessels and into the wound area.23,24 However, whether PDGF-BB in addition also modulates the secretome of pericytes is not known. Since PDGF-BB has restorative effects in Parkinson’s disease,7–9 we next analyzed if PDGF-BB signalling stimulates secretion of growth factors in brain pericytes. We examined the cell culture medium using a human growth factor array, a semi-quantitative analysis based on spot intensity quantification. When treated with PDGF-BB, human brain pericytes released significant amounts of several trophic growth factors such as insulin-like growth factor-binding protein (IGFBP), neurotrophin 3 (NT3) and heparin-binding EGF-like growth factor (HB-EGF), whereas the release of stem cell factor (SCF) was decreased after PDGF-BB treatment compared to controls (Figure 2(a)). Using a high sensitive quantification analysis (human multiplex sandwich Elisa), we analyzed a group of particularly interesting growth factors with neuroprotective, neuroregenerative or angiogenic capacities and observed a significant increase of BDNF (1.4-fold), bFGF (2-fold), βNGF (1.8-fold), and vascular endothelial growth factor (VEGF) (2-fold) released by brain pericytes treated with PDGF-BB compared to controls. Sunitinib, a PDGFRβ kinase inhibitor,
25
inhibited the PDGF-BB-induced release of the growth factors, confirming that the secretion of the different growth factors was specifically mediated via PDGFRβ (Figure 2(b)). The PDGF-BB concentration used (20 ng/ml) did not affect cell proliferation (Supplementary Figure 2).
PDGF-BB induces release of growth factors by human brain pericytes. (a) Human growth factor array analysis showing concentration of several growth factors. Medium was obtained from untreated human brain pericytes (Ctrl), or pericytes treated with PDGF-BB (20 ng/ml, 72 h). Increase in growth factor secretion is indicated by a green bar, while decrease of growth factors by a red bar. Data represent the mean ± SD (n = 4). One-sample two-tailed t-test: *p < 0.05 vs. each control set at 100%. (b) Quantification by multiplex sandwich Elisa of the concentration of BDNF, bFGF, βNGF and VEGF in the medium of untreated human brain pericytes (Ctrl), pericytes treated with PDGF-BB (20 ng/ml, 72 h), or treated with Sunitinib (2 µM) 1 h before treatment with PDGF-BB (20 ng/ml, 72 h). Data are expressed in pg/ml and represent the mean ± SD (n = 3–6). One-way ANOVA with Tukey post hoc: *p < 0.05 **p < 0.01 ***p < 0.001 vs. control group, #p < 0.05, ##p < 0.01, ###p < 0.001 vs. PDGF-BB-treated group.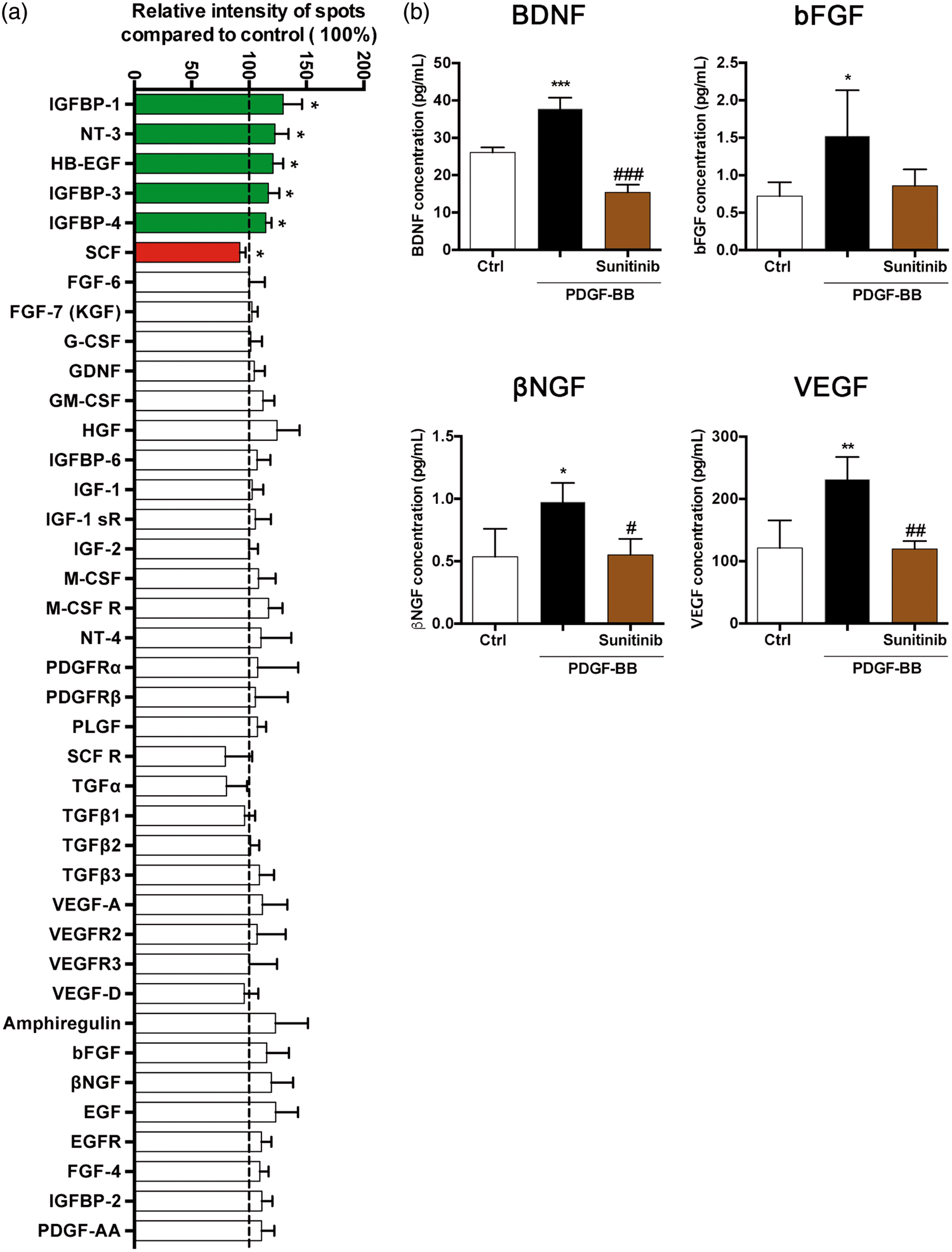
Human brain pericytes release microvesicles containing growth factors in response to PDGF-BB
MVs are small membrane vesicles that are shed from a variety of cell types and form a vehicle for cell–cell communication. They represent a reservoir of bioactive molecules that can be transported over longer distances. MVs are currently identified as important mediators of tissue regeneration.
26
However, whether also pericytes release MVs is not known. We therefore next quantified and characterized the MVs released by pericytes upon different exogenous stimuli. After PDGF-BB treatment, human brain pericytes released a higher number of MVs compared to untreated cells or LPS-treated pericytes (Figure 3(a)), even though cell numbers were not affected by the treatments (Supplementary Figure 2). Then, we analyzed if released MVs contain the same trophic and angiogenic factors observed in the medium of PDGF-BB-treated pericytes. Multiplex sandwich Elisa showed that the MVs contained significantly more BDNF, bFGF, βNGF, VEGF and placental growth factor (PLGF) after treatment with PDGF-BB compared to MVs-derived from control cells (Figure 3(b)).
Human brain pericytes release microvesicles containing growth factors in response to PDGF-BB. (a) Number of MVs in medium collected from untreated human brain pericytes (Ctrl), pericytes treated with PDGF-BB (20 or 50 ng/ml, 72 h) or with LPS (10 or 50 µg/ml, 72 h). Data are expressed as number of MVs/µl of medium and represent the mean ± SD (n = 3–11). One-way ANOVA with Tukey post hoc: *p < 0.05 ***p < 0.001 vs. control group. (b) Quantification by multiplex sandwich Elisa of BDNF, bFGF, βNGF, VEGF and PLGF concentrations in MVs collected from medium of untreated human brain pericytes (Ctrl) and pericytes treated with PDGF-BB (20 ng/ml, 72 h). Data are expressed in pg/ml and represent the mean ± SD (n = 3–4). Two-tailed unpaired t-test: **p < 0.01 ***p < 0.001 vs. control group.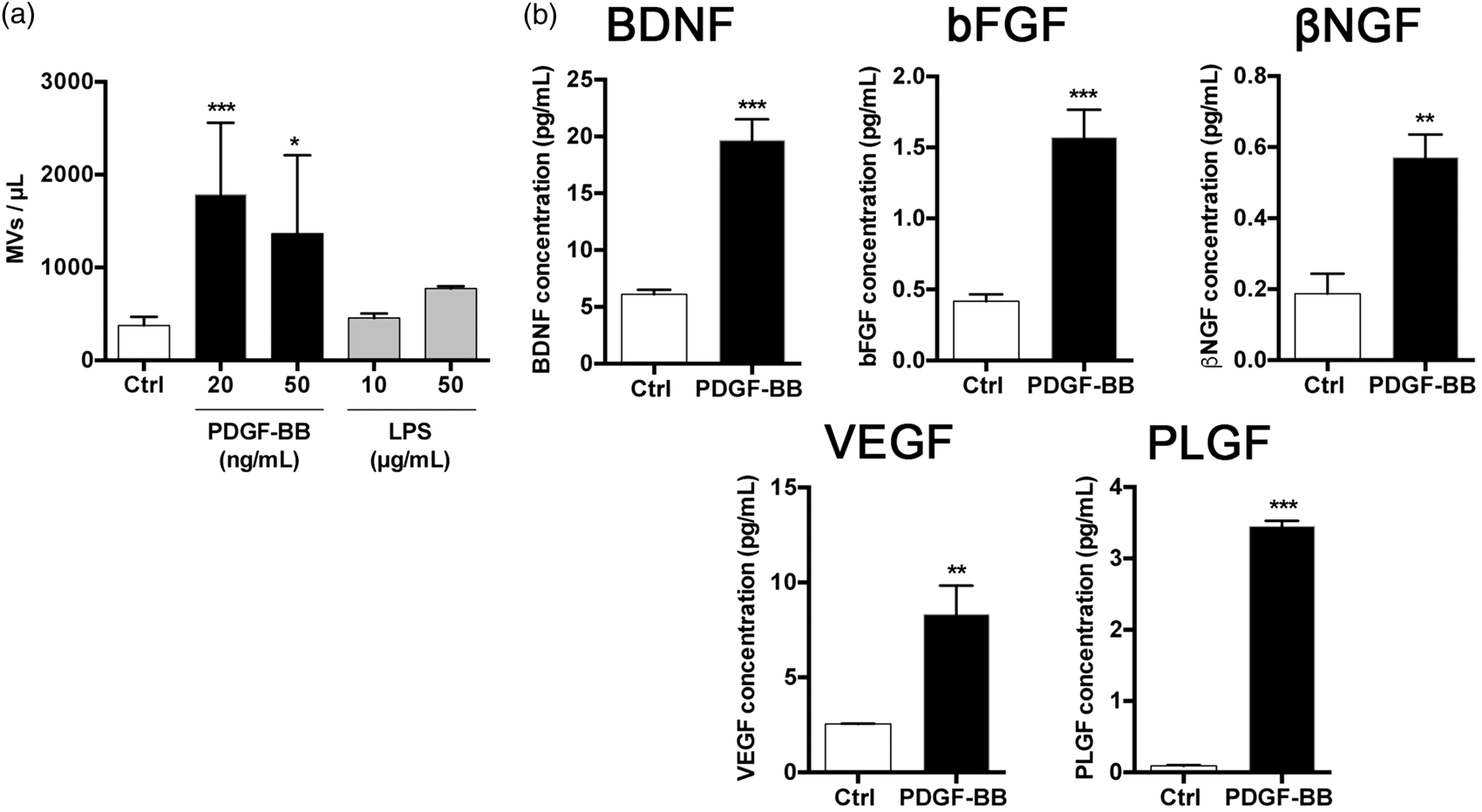
Pericytes release multiple cytokines in response to PDGF-BB and LPS
Pericytes have recently been shown to secrete pro-inflammatory cytokines in response to inflammatory stimuli,
21
but the cytokine response to PDGF-BB is not known. Next, we therefore asked whether human brain pericytes release cytokines in addition to growth factors when treated with PDGF-BB and, especially, how this cytokine release compares to the response to LPS, a strong inflammatory stimulus. Using a human cytokine array, we showed that LPS treatment induced a highly significant release of cytokines and chemokines with mainly pro-inflammatory properties such as growth-regulated protein alpha/beta/gamma (GROα/β/γ), interleukin (IL) IL-6, IL-8, granulocyte macrophage colony-stimulating factor (GM-CSF), IL-7, RANTES, chemokine CCL-17 (TARC), IL-1α, M-CSF, stromal cell-derived factor 1 alpha (SDF-1α) and IL-2, some of which have been previously described to be secreted by mouse and human brain pericytes after an inflammatory stimulus (Figure 4(a) and Supplementary Figure 3).16,27 PDGF-BB also led to the release of cytokines and chemokines but to a significantly lower amount (Figure 4(a)). Interestingly, PDGF-BB significantly decreased the secretion of tumor necrosis factor alpha (TNFα) and increased IL-13, IL12p40/70, IL-15 by brain pericytes compared to controls (Figure 4(a)) and Supplementary Figure 3).
Human brain pericytes release multiple cytokines in response to PDGF-BB and LPS. (a) Human cytokines array analysis of 42 cytokines expressed by relative intensity of spot obtained by analyzing medium of pericytes treated with PDGF-BB (20 ng/ml, 72 h) or LPS (50 µg/ml, 72 h). Data represent the mean ± SD (n = 4). Two-way ANOVA with Sidak post hoc: ##p < 0.01 ###p < 0.001 between LPS-treated and PDGF-BB-treated groups. (b) Quantification by multiplex sandwich Elisa of IL-6, IL-8, MCP-1 and IP-10 concentrations in the medium of untreated human brain pericytes (Ctrl), treated with PDGF-BB (20 ng/ml, 72 h), treated with Sunitinib (2 µM) 1 h before treatment with PDGF-BB or treated with LPS (50 µg/ml, 72 h). Data are expressed in pg/ml and represent the mean ± SD (n = 3–4). One-way ANOVA with Tukey post hoc for three-group comparison and two-tailed unpaired t-test for two-group comparison: *p < 0.05 **p < 0.01 ***p < 0.001 vs. control group; ###p < 0.001 vs. PDGF-BB-treated group.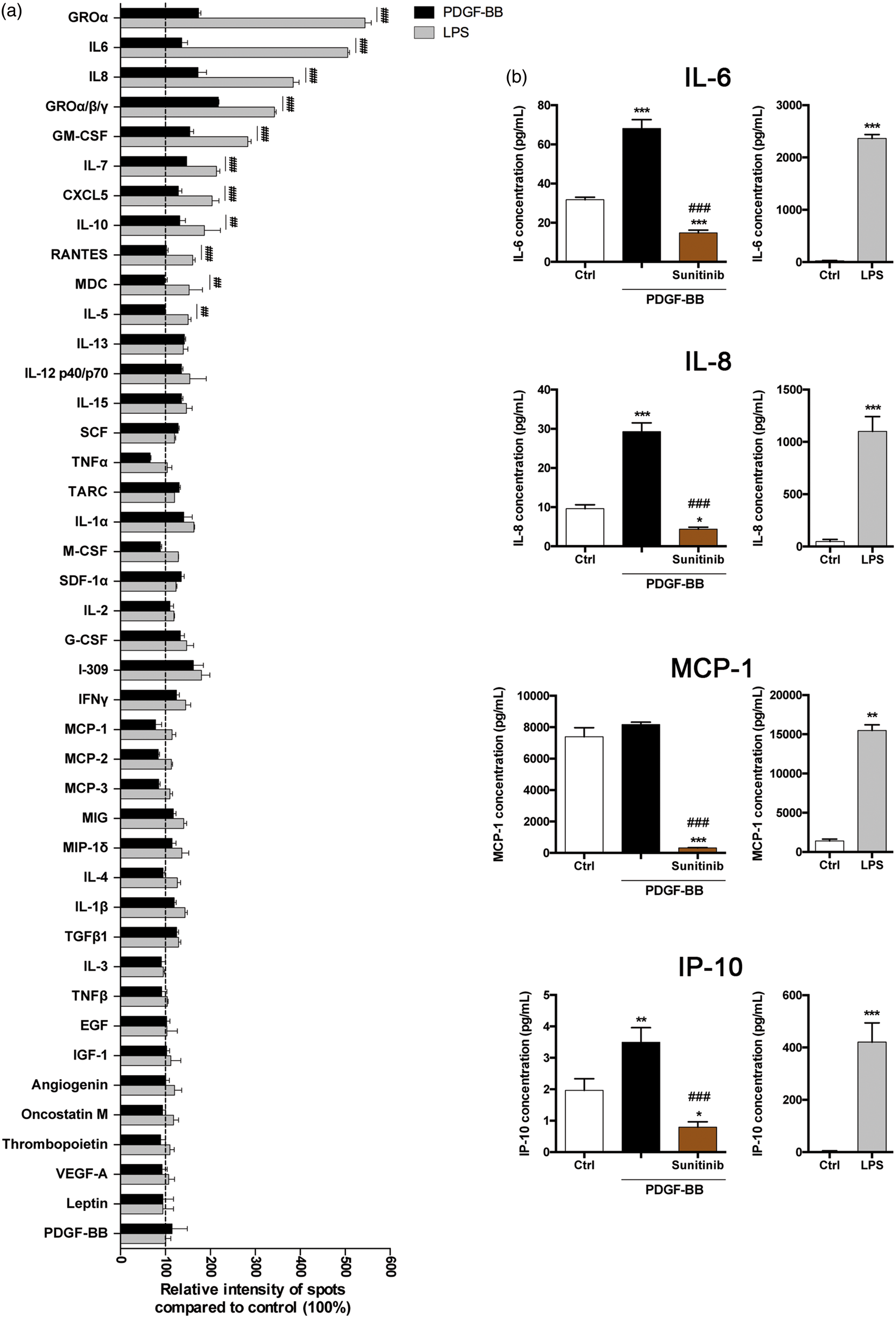
In order to study the implication of PDGFRβ signalling on PDGF-BB-mediated cytokine release and to quantify the release of cytokines, we used a multiplex sandwich Elisa analysis and focused on IL-6, IL-8, monocyte chemoattractant protein 1 (MCP-1) and chemokine CXCL10 or interferon gamma-induced protein 10 (IP-10). Our results showed that Sunitinib inhibited the PDGF-BB-induced secretion of IL-6, IL-8, MCP-1 and IP-10. This confirmed that the secretion of cytokines in human brain pericytes is PDGFRβ-mediated (Figure 4(b)). In contrast, LPS induced a much higher release of cytokines and chemokines compared to PDGF-BB, for IL-6 (90-fold increase), IL-8 (23-fold increase), MCP-1 (11-fold increase), IP-10 (131-fold increase) (Figure 4(b)), suggesting that pericytes have the capacity to modulate their secretome in response to environmental stimuli, both qualitatively and quantitatively.
Human brain pericytes release microvesicles containing cytokines in response to PDGF-BB and LPS
The secretome analysis showed that PDGF-BB and LPS induce a release of different panels of cytokines in human brain pericytes. We therefore next analyzed if released MVs contain the same cytokines observed in the medium of treated pericytes. Multiplex sandwich Elisa showed that MVs contained significantly more IL-6, IL-8, MCP-1 and IP-10 after treatment with LPS compared to MVs derived from untreated cells or from cells treated with PDGF-BB, which contained only low amount of cytokines (Figure 5).
Human brain pericytes release microvesicles containing cytokines in response to PDGF-BB and LPS. Quantification by multiplex sandwich Elisa of IL-6, IL-8, MCP-1 and IP-10 concentrations in MVs collected from medium of untreated human brain pericytes (Ctrl), pericytes treated with PDGF-BB (20 ng/ml, 72 h) and pericytes treated with LPS (50 µg/ml, 72). Data are expressed in pg/ml and represent the mean ± SD (n = 3–4). One-way ANOVA with Tukey post hoc: *p < 0.05 **p < 0.01 ***p < 0.001 vs. control group; #p < 0.05, ##p < 0.01, ###p < 0.001 vs. PDGF-BB-treated group.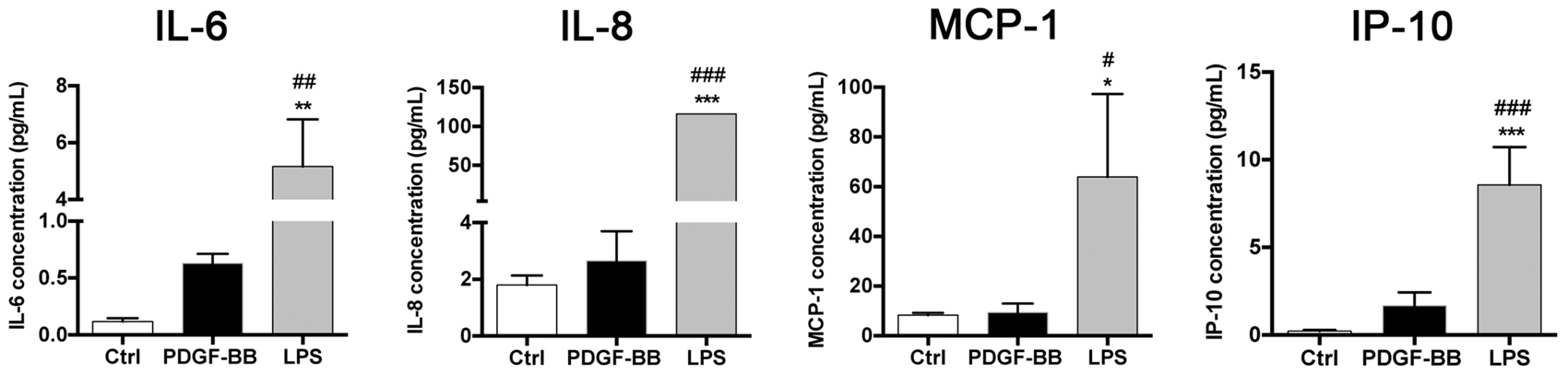
PDGF-BB and LPS activate different signal transduction pathways
In order to further investigate PDGFRβ-mediated cell signalling, we studied the chronology of PDGFRβ and signal transduction pathway activation. We treated human brain pericytes with PDGF-BB for 15 min, 90 min and 72 hours, and pre-treated with Sunitinib. As expected, after 15 min exposure, PDGF-BB activated PDGFRβ as seen by an increase in protein expression of phosphorylated PDGFRβ (Figure 6(a) and (b)). Pre-treatment with Sunitinib completely inhibited PDGF-BB-induced PDGFRβ phosphorylation. After 90 min of PDGF-BB treatment, a significant increase of ERK 1/2 phosphorylation, the main intracellular pathway activated by tyrosine kinase receptors, was observed. The ERK 1/2 activation was prevented by Sunitinib (Figure 6(a) and (b)), corroborating the PDGFRβ-mediated activation by PDGF-BB of this downstream pathway.
28
PDGF-BB treatment for 90 min and 72 h led to a downregulation of the expression of PDGFRβ, consistent with the previously described internalisation of the receptor.
29
At 72 h, both PDGF-BB and Sunitinib significantly decreased PDGFRβ and ERK 1/2 phosphorylation. In contrast, in pericytes treated with LPS (24 h), NFκB was activated as seen by its phosphorylation,
27
whereas there was a trend towards a decrease in ERK 1/2 phosphorylation after exposure to LPS that did not reach significance. Our findings demonstrate that PDGF-BB and LPS activate different signal transduction pathways in human brain pericytes (Figure 6(a) and (b)).
PDGF-BB and LPS activate different signal transduction pathways in pericytes. (a) Western blot analysis and (b) corresponding quantifications showing the expression of pPDGFRβ, PDGFRβ, pERK, ERK, pNFκB, NFκB and βactin in untreated human brain pericytes, pericytes treated with PDGF-BB (20 ng/ml) for 15 min, 90 min or 72 h, treated with Sunitinib (2 µM) 1 h before treatment with PDGF-BB or treated with LPS (50 µg/ml, 24 h). PDGF-BB increased PDGFRβ phosphorylation and ERK 1/2 pathway activation. LPS increased NFκB phosphorylation. Data represent the mean ± SD (n = 3–8). One-way ANOVA with Tukey post hoc for three-group comparison and two-tailed unpaired t-test for two-group comparison: *p < 0.05 **p < 0.01 ***p < 0.001 vs. control group; ##p < 0.01 ###p < 0.001 vs. PDGF-BB-treated group.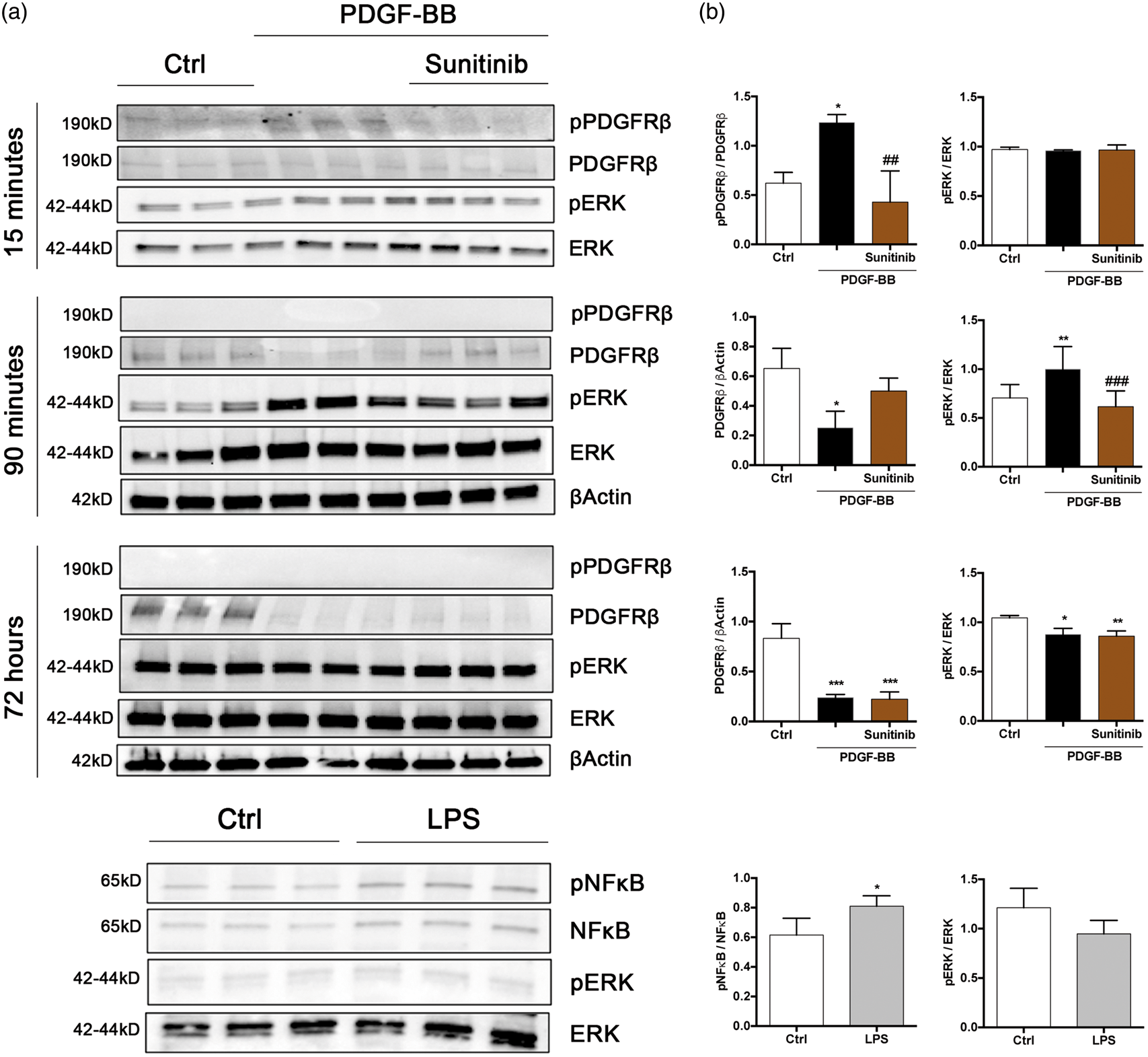
PDGF-BB and LPS treatments differently modulate pericyte morphology and surface marker expression
Finally, we analysed if pericytes show distinct morphological and phenotypic changes in response to two different stimuli. PDGF-BB treatment induced morphological changes in human brain pericytes whereby cells adapted an elongated shape (Figure 7(a)). In contrast, LPS treatment induced an amoeboid and ramified shape of the cells, reminiscent of a microglial/macrophage-like morphology. Using flow cytometry, we therefore analysed whether PDGF-BB and LPS also affect the surface maker expression on pericytes (Figure 7(b) and (c)). PDGF-BB significantly decreased the percentage of cells expressing PDGFRβ (CD140b) from 96.3% ± 0.15 in control cultures to 62.46% ±6.41 in PDGF-BB-treated cultures, suggesting internalization of the receptor. Both PDGF-BB and LPS increased the expression of CD146. The observed increase in CD146 was significantly higher when pericytes were treated with LPS compared to PDGF-BB (93.13% ± 0.504 with LPS vs. 71.5% ± 0.450 with PDGF-BB). CD146 is a membrane glycoprotein which functions as a calcium-independent cell adhesion molecule involved in cell–cell interactions
30
and has been implicated in leukocyte adhesion.
31
PDGF-BB and LPS treatments differently affect the morphology and surface marker expression on human brain pericytes. (a) Light microscopy images of untreated human brain pericytes, treated for 72 h with PDGF-BB 50 ng/ml, showing an elongated morphology and for 72 h with LPS (50 µg/ml) illustrating an amoeboid shape. Scale bar = 100 µm. Arrows indicate cell morphological changes. (b) Flow cytometry analysis of the surface marker expressions of PDGFRβ (CD140b), CD146, CD11b, CD14 and CD45 on untreated human brain pericytes, treated with PDGF-BB (20 ng/ml, 72 h) or with LPS (50 µg/ml, 72 h). (c) Quantification of surface markers expression in controls (Ctrl), after PDGF-BB or LPS treatments. PDGF-BB-treated pericytes down regulated CD140b and slightly upregulated CD146 and CD11b expressions, whereas LPS-treated pericytes highly upregulated CD146 CD11b and slightly CD14. Data represent the mean ± SD (n = 3). One-way ANOVA with Tukey post hoc: *p < 0.05 **p < 0.01 ***p < 0.001 vs. control group; #p < 0.05 ##p < 0.01 ###p < 0.001 vs. PDGF-BB-treated group.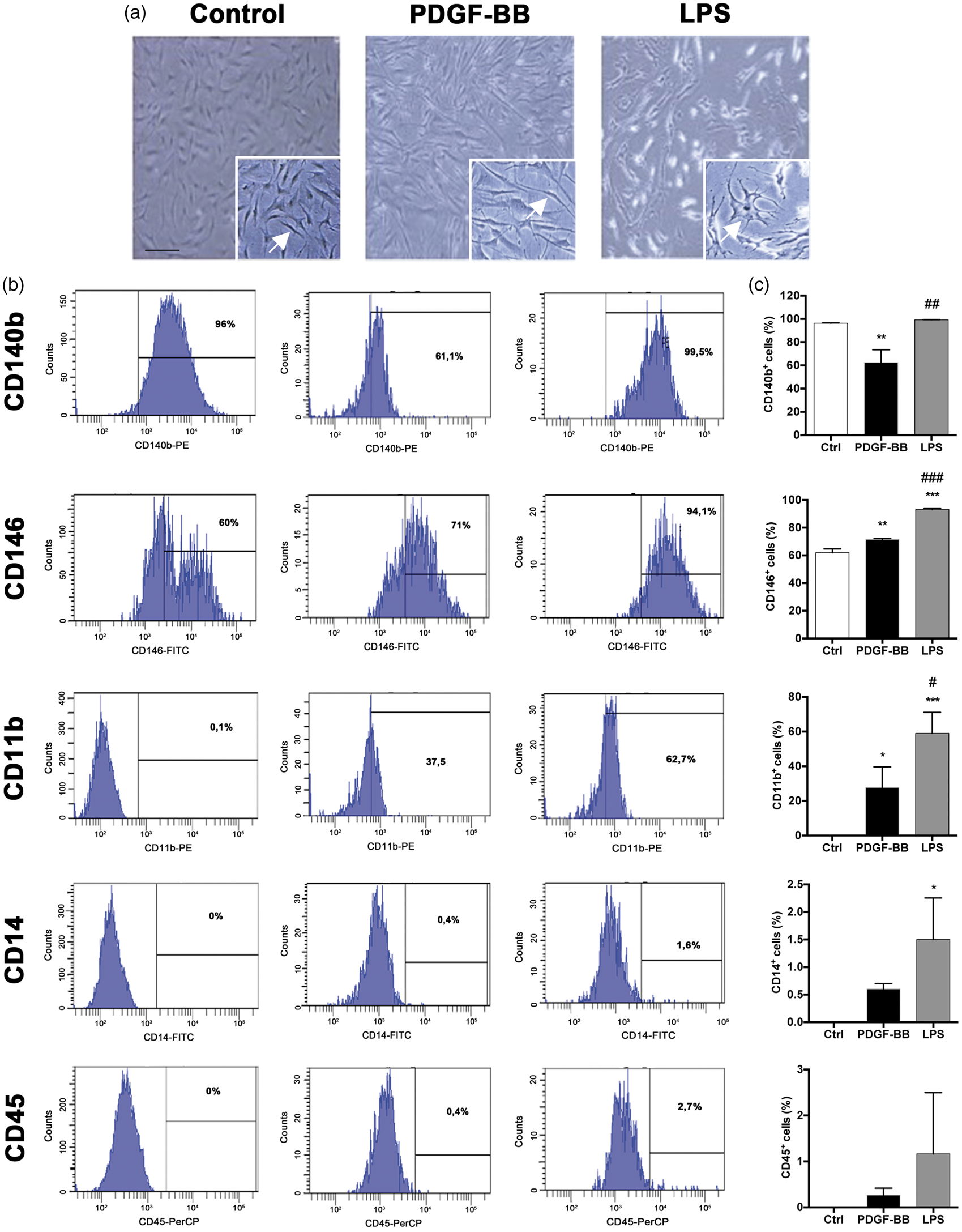
LPS exposure also resulted in a 60% increase in CD11b expression (60% ± 6.97 with LPS vs. 27.67% ± 6.92 with PDGF-BB), a microglial marker, consistent with the morphological changes observed after LPS stimulation in vitro. Only 1–2% of cells expressed the monocyte/leukocyte markers CD14 and CD45 after both treatments (Figure 7(c)).
Discussion
Here, for the first time, we demonstrate in human brain pericytes that the specific PDGF-BB-mediated signalling via PDGFRβ leads to secretion of different growth factors, cytokines and the release of MVs carrying growth factors that have been implicated in neuroprotection and neurorestoration.
Furthermore, we demonstrate a differential response of pericytes by showing the stimulus-dependent secretion of different molecules. In addition, we provide novel evidence that brain pericytes release MVs in a stimulus-specific manner that highly reflects the characteristics of the secretome. Our data support the hypothesis that brain pericytes may present a new actor not only in the regulation of neuroinflammation, but also for CNS regeneration that may partially be mediated via PDGF-BB/PDGFRβ signalling and their secretome.
Brain pericytes highly express PDGFRβ, a tyrosine kinase receptor and PDGF-BB/PDGFRβ signalling is important for pericyte survival.32,33 Here, we show that PDGF-BB signalling via the PDGFRβ in brain pericytes promotes secretion of trophic and angiogenic growth factors that can be blocked by the tyrosine kinase inhibitor Sunitinib. We and others have recently demonstrated that PDGF-BB is neurorestorative in animal models of PD7,8 and PDGF-BB has, based on pre-clinical findings, entered clinical trials in PD. 9 The neurorestorative mechanism of PDGF-BB is not fully understood, why the secretion of neuroprotective molecules by pericytes in response to PDGF-BB is of high relevance. Among the factors released by brain pericytes via PDGF-BB/PDGFRβ stimulation, we focused particularly on growth factors with potential neuroprotective or neuroregenerative capacities. First, we show that pericytes release BDNF and βNGF, which may act on neurons to support their survival, growth and differentiation.34,35 Besides, human brain pericytes treated with PDGF-BB produced bFGF, which has been shown to enhance neuroprotection and recovery in rodent stroke and PD models.36,37 It is conceivable that pericytes contribute to neuroprotection under certain pathological conditions by secretion of some of the regenerative growth factors identified here. However, the exact role of the secretome of pericytes under these pathological conditions needs to be further investigated in in vivo models in the future.
Pericytes have been widely perceived as the in vivo presentation of MSCs in all tissues, 42 even though this view has recently been challenged. 11 We have previously shown that also adult human brain pericytes possess distinct features of MSC when isolated. 22 Recently, the therapeutic potential of MSCs upon grafting has been largely attributed to paracrine factors contained in MVs. 39 Here, we showed for the first time that human brain pericytes released MVs when stimulated in vitro with PDGF-BB or LPS, respectively. The number of MVs released after PDGF-BB stimulation was significantly higher compared to LPS-stimulation or control conditions, which did not differ from each other. However, the MVs released by LPS contained high amounts of the inflammatory cytokines IL-6, IL-8, MCP-1 and IP-10, all cytokines that are also released in the medium by pericytes after LPS stimulation. MVs released by pericytes upon PDGF-BB stimulation contained a 3–4 fold increase of the growth factors BDNF, bFGF, βNGF, VEGF and PLGF compared to controls. PLGF is an angiogenic factor that belongs to the VEGF family. PLGF has been shown to promote the survival of cultured primary cortical neurons under oxygen and glucose deprivation, 40 as it is upregulated in blood vessels, neurons and astrocytes after stroke 41 and contributes to neuroprotection in cerebral ischemia. 42 These growth factors transported in MVs may provide an effective mechanism for long-range communication of neuroprotective and angiogenic information between pericytes and other cells. Further studies are needed to explore the proteins and miRNA composition of pericyte-derived MVs and their potential function in brain disorders.
When we compared the specific cytokine responses of human brain pericytes to two different exogenous stimuli, PDGF-BB, a growth factor and LPS, an inflammatory stimulant, we found a differential response. PDGF-BB induced the release of well-described cytokines and chemokines, some of which have mainly anti-inflammatory properties, and decreased the release of TNFα, a cytokine with pro-inflammatory properties. Of interest is IL-13, shown for the first time to be upregulated upon PDGF-BB treatment by brain pericytes, as it promotes neuronal survival and induces death of activated microglia after LPS injection in vivo. 43 PDGF-BB induced only a low rate of inflammatory cytokines compared to a stimulus with LPS.
PDGF-BB first induced PDGFRβ activation at an early time point, as demonstrated by receptor phosphorylation, whereas longer exposure led to PDGFRβ downregulation as seen by flow cytometry and Western blot analysis. Internalization of PDGFRβ as part of a negative feedback mechanism of binding PDGF-BB is well known.29,44 Consistently, we show that PDGF-BB results in ERK 1/2 phosphorylation. 28 Both, PDGFRβ phosphorylation/downregulation and ERK 1/2 pathway activation are prevented by sunitinib, a receptor tyrosine kinase inhibitor, 45 confirming that PDGF-BB-mediated changes are signalled via PDGFRβ. In contrast, LPS led to NFκB phosphorylation. These findings are consistent with published data showing that LPS activates toll-like receptor 4-mediated NFκB signalling pathway in human pericytes. 27 Thus, pericytes respond differently to different exogenous stimuli and activate segregated intracellular pathways suggesting that pericytes may have clearly distinct functions depending on the different environmental circumstances.
Morphological and phenotypical changes in pericytes occur when exposed to pathological conditions such as hypoxia14,15 or inflammation. 17
We demonstrated that LPS exposure highly increased the percentage of pericytes expressing CD11b, whereas there was only a slight increase in CD11b-positive cells after PDGF-BB treatment. The prominent increase in CD11b was well consistent with the microglial morphology and the increase in CD146 expression, an adhesion molecule implicated in leukocyte adhesion31,46 observed in LPS-treated pericytes and suggested that brain pericytes change in response to LPS towards a microglial phenotype. Similar to our observations, LPS led to upregulation of CD11b in mouse pericytes 16 consistent with findings by others showing that cultured brain pericytes might present immune cell properties and exhibit phago- or pinocytic activity.14,21,47
The possible conversion of pericytes to an inflammatory phenotype is further supported by our findings that human brain pericytes secreted pro-inflammatory cytokines especially when stimulated with LPS. Cultured mouse brain pericytes have been shown to secrete 18 inflammatory cytokines and chemokines in response to LPS. 16 In our study, using human brain pericytes, we extended the cytokines screening by analyzing 42 cytokines. We found 12 cytokines and chemokines significantly released by human brain pericytes in response to LPS. The cytokines/chemokines newly described in our study were especially implicated in the proliferation, maturation and activation of immune cells such as T cells, B cells, Natural killer, and neutrophils (GRO α/β/γ, IL-7, IL-2, IL-5). This suggests that the pericyte secretome may contribute to the infiltration of circulating immune cells when exposed to an inflammatory stimulus such as LPS, a finding that will be of relevance for the design of new treatments addressing neuroinflammatory diseases by targeting pericyte-immune cell crosstalk. Our findings further support the notion that pericytes have the capacity to undergo phenotypic changes and acquire functional properties of inflammatory cells when stimulated by LPS.
Conclusion
Brain pericytes may constitute a novel target not only for BBB protection and immunomodulation in CNS disorders, but also for neuroprotection due their secretory capacities. The elucidation of receptors and down-stream pathways mediating such regenerative effects is essential to guide the development of novel drug targets for regeneration.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Swedish Medical Research Council, the Parkinsonfonden, the Brain Foundation, the Aners Foundation, Crafoord Foundation, Scania University Hospital donations and Multipark.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ contributions
AG designed the research project, conducted the analysis, interpreted the data and wrote the manuscript. TP contributed to cell culture work, manuscript writing and figure layout. MB contributed to analysis of multiplex sandwich Elisa. GP and IÖ designed and supervised the research project, interpreted the data and contributed to writing of the manuscript. All authors critically reviewed and approved the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
