Abstract
When used to measure blood flow in the brain, water leaves a residue in the vascular bed that influences the estimation of blood flow by current methods. To assess the magnitude of this influence, we developed a two-compartment model of blood flow with separate parameters for transport and vascular distribution of brain water. Maps of the water clearance, K1 into brain tissue, separated from the circulation by a measurably resistant blood–brain barrier (BBB), were generated by time-weighted integration. Depending on the validity of the assumptions underlying the two-compartment model presented here, the maps revealed a significant overestimation of the clearance of water when the vascular residue was ignored. Maps of Vo the estimate of the apparent vascular distribution volume of tracer H215O, clearly revealed major cerebral arteries. Thus, we claim that the accumulation of radioactive water in brain tissue also reflects the volume of the arterial vascular bed of the brain.
Keywords
The present study was designed to reveal the magnitude of the residue of water left in the vascular bed of the brain during the water's circulation after a bolus injection. Identification of this residue was intended to provide the basis for a method permitting the correct measurement of the clearance of water from the circulation into brain tissue as a more faithful index of perfusion than those currently used.
The permeability of the blood–brain barrier (BBB) to water is of such magnitude that radioactive water is often used as a tracer to measure cerebral blood flow (CBF), e.g., by positron emission tomography (PET). The use is based on the assumption that the tracer is instantly distributed to brain tissue as a whole, allowing blood flow alone, rather than the combination of blood flow, BBB filtrational and diffusional permeability, and capillary surface areas, to determine the water uptake.
It is, nonetheless, doubtful that the permeability of the BBB to water is sufficient to exclude a measurable resistance to instantaneous distribution. Limited diffusibility of water in the BBB has been reported frequently (Eichung et al., 1974; Gjedde et al., 1975; Herscovitch et al., 1983b; Raichle et al., 1983). Koeppe et al. (1987) demonstrated a significant overestimation of water clearance when the transmitted (vascular) radioactivity is ignored. The degree of overestimation depended on the length of data accumulation used to study the water exchange.
Gambhir et al. (1987) formulated a two-compartment model to describe the uptake of H215O in brain, assuming that arterial H215O fails to dissolve instantly and uniformly in brain tissue. With this model, estimates of clearance and distribution of H215O depended less on the duration of the study. This two-compartment model was based on the concept of an apparent vascular distribution volume, Vo, which holds the fraction of the tracer transmitted through the capillary bed at a concentration equal to the concentration in arterial blood.
The reformulated two-compartment model introduced in the present study yields both the unidirectional clearance of water from blood to brain, K1, and the apparent vascular distribution volume, Vo, of the tracer. It is, therefore, different from the two-compartment model of Gambhir et al. (1987). The introduction of Vo reveals that it is not the total vascular volume but a smaller fraction thereof that depends on the BBB permeability of the tracer. Brain maps of the water clearance, K1, and water's vascular distribution volume, Vo, were created by time-weighted integration (Alpert et al., 1984; Ohta et al., 1992). For comparison, we also generated maps with the one-compartment method (Alpert et al., 1984; Carson et al., 1986; Kanno et al., 1987; Raichle et al., 1983), which has no vascular volume. The precision of this new method's use of three-parameter analysis by weighted integration was tested by simulation.
EXPERIMENTAL DESIGN AND THEORY
The basic design of the study included derivation of novel equations describing water accumulation in brain tissue and comparison of the results of PET studies using these novel equations with results obtained by conventional computational strategies.
For the nonsteady-state, and with the assumption that larger vessels as well as exchange vessels may reside in the field of view of the tomograph, we derived the equation for the accumulation of inert tracer in a tissue (see Appendix for derivation of equations and Fig. 1 (left panel) and Table 1 for explanation of symbols):
Symbols and definitions

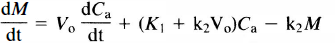
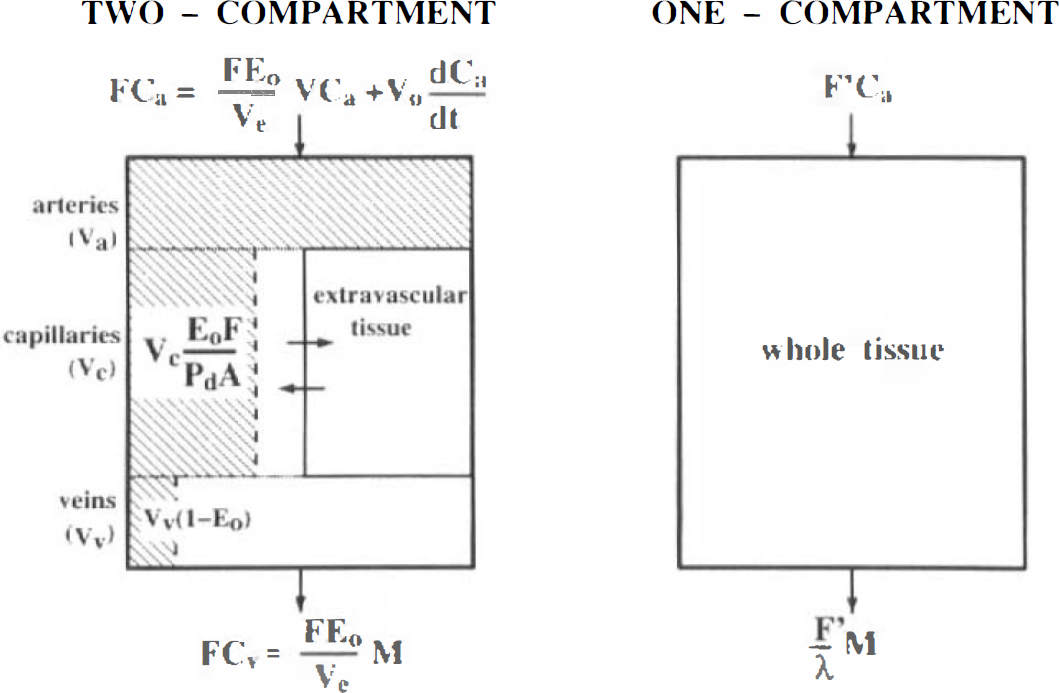
Two model configurations for the H215O PET studies. Two-compartment model for water clearance and apparent vascular distribution volume, Vo, (left, shaded area) and one-compartment model for blood flow (right). Note that for Eo→ 0, EoF→P and VcEoF/P→Vc, and Vv (1 — Eo)→Vv, such that Vo→Va +Vc + Vv. For Ea → 1,P >> F, therefore VcEoF/P→0, and Vv(1 — Eo) → 0, such that Vo → Va.
where M is the tracer quantity in brain and Ca the arterial concentration of the tracer. The three parameters, K1, k2, and Vo, were defined as follows: K1 is the unidirectional clearance of water and k2 is the rate constant of water removal from brain tissue. The term clearance is used with its conventional meaning of fraction of plasma water flow cleared of radioactive water, as derived below. Vo is a subvolume of the arterial (Va), capillary (Vc), and venous (Vv) vascular beds; its magnitude depends on the permeability of the tracer. Definitions are shown in Table 1. The two solutions of Eq. 1 used in this work are
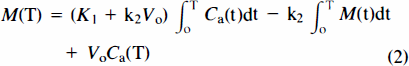
and
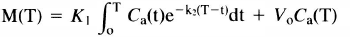
From these expressions, K1, k2, and Vo were estimated by multilinear regression (Eq. 2) or weighted integration (Eq. 3), as outlined in Table 2, which is a summary of computation strategies.
Summary of computational strategies

For comparison, we used the simplified bloodflow solutions derived in the theory section on one-compartment analysis to obtain single-compartment estimates of perfusion and partition coefficient
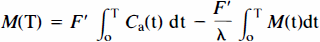
and
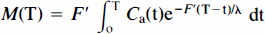
where F′ and λ are the single-compartment estimates of perfusion and partition coefficient. The comparison was used to reveal and interpret the discrepancy between K1 and F′.
The two compartments have three parameters (Fig. 1, left panel) that must be estimated by linear or nonlinear regression, or by weighted integration, as in the case of two parameters. The three parameters are either K1 [= k2(V — Vo], k2(= F Eo/Ve) and Vo (shaded area), or k2, V, and Vo. We expanded the two-weight integration to the estimation of three parameters, the three-weight integration, as described in the Appendix.
The single compartment has two parameters (Fig. 1, right panel), apparent blood flow, F′, and partition coefficient, λ. The two parameters were determined either simultaneously (two-parameter analysis) or apparent blood flow was calculated with an assumed fixed value of the partition coefficient (one-parameter analysis). The two-parameter analysis can proceed either by linear or nonlinear regression, or by weighted integration (Alpert et al., 1984; Carson et al., 1986; Koeppe et al., 1985). The one-parameter analysis is known as the ‘autoradiographic’ method because it mimics autoradiography in which a single measurement of the dependent variable is available (Kanno et al., 1987; Raichle et al., 1983).
METHODS
Simulated PET studies
We evaluated errors in the time-weighted integration caused by tissue heterogeneity, tracer delay and dispersion of the peripherally sampled arterial input function, Ca(t). For this purpose, a typical input function, Ca(t), was first generated from the sum of three time-weighted exponentials (Herscovitch et al., 1983b),
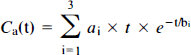
The effects of tissue heterogeneity, tracer dispersion and delay on the estimation of K1 and Vo by three-weight integration were evaluated as outlined by Herscovitch et al. (1983b), Iida et al. (1986), and Meyer (1989). Tissue heterogeneity was simulated using Eq 4 with gray-white matter K1 pairs of 0.4 and 0.2, 0.6 and 0.2, and 0.8 and 0.2 ml min−1 g−1 and Vo pairs of 0.010 and 0.005, 0.020 and 0.005, and 0.040 and 0.005 ml g−1. These values were assumed to cover the realistically expected range for the two parameters. The K1/k2 ratio was always fixed at 0.9 ml g−1. The accuracy of the three-weight integration was assessed by analysis of simulated data with superimposed random Poisson variability. First, uniform randomly distributed 5% Poisson noise was imposed on each point of the simulated input function. This variation was consistent with the error inherent in the manually sampled blood curves, including statistical variation and errors due to sample timing and weighing. Next, tissue time-activity curves were generated according to Eq. 3 with a 5-s sampling interval for a total length of 3 min, using the previously generated input function and selected values for K1, k2, and Vo. Uniform 5% Poisson distributed variability (PN), approximately corresponding to that observed on real time-activity curves for 2-cm diameter regions of interest (ROIs), was imposed on each point of a given tissue time-activity curve using the equation
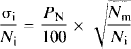
Here, σi is the standard deviation of the Poisson variability added to the ith scan, Ni the individual counts in the ith scan, and Nm the mean frame counts corresponding to a 4-cm2 region and 106 total counts/plane in 3 min (Evans et al., 1986). K1, k2, and Vo were then estimated by the three-weight integration from simulated variable tissue and arterial time-activity curves. The accuracy of each of the three parameter estimates was expressed as the fractional root-mean-squared error (ε) for N = 100 variable patterns of each simulation,
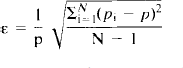
where pi is the parameter estimate and p its input value.
Human studies
Fifteen ‘dynamic’ (multiframe) H215O sessions were completed in nine young volunteers. Four subjects were studied both under normoventilation (average Paco2 39.6 mmHg) and under hyperventilation (average Paco2 23.0 mmHg). Six subjects were scanned with the Positome IIIp, a two-ring (BGO) Bi4Ge3O12 head tomograph with transverse and axial spatial resolutions of 11 and 16 mm full width at half maximum (FWHM), respectively, and an interplane separation of 18 mm (Thompson et al., 1986). Three subjects were scanned with the PC-2048 15B (Scanditronix), an eight-ring BGO head tomograph with transverse and axial resolutions of ∼6 mm FWHM and an interslice separation of 6.5 mm (Evans et al., 1991). Reconstruction software on the Positome IIIp included corrections for detector efficiency variations, random events, a deconvolution procedure to eliminate scattered events (Cooke et al., 1984), and attenuation correction with a projection contour finding method. On the PC-2048 15B, similar corrections were applied, except for attenuation correction, which was performed with a rotating Ge-68 transmission rod source. Radioactive water was prepared with a Japanese Steel Works (Muroran, Japan) Medical Cyclotron (BC-107).
After completion of a questionnaire designed to exclude volunteers with neurological deficits, informed consent was obtained from each participant. The protocol was approved by the Research and Ethics Committee of the Montreal Neurological Hospital and Institute. Subjects' heads were immobilized by means of a customized self-inflating foam headrest. A short indwelling catheter was placed into the left radial artery for blood sampling and blood gas determinations.
The tracer H215O was injected as a bolus into the brachial vein of the right arm (20 mCi for Positome IIIp, 40 mCi for PC-2048 15B). Arterial blood sampling and dynamic imaging began at injection time. The flow of blood from the freely-flowing arterial sampling catheter was kept ∼10–15 ml min−1 in order to minimize dispersion effects (Iida et al., 1986; Meyer, 1989). Dynamic scans with frame durations of 3 (Positome IIIp) or 5 s (PC–2048 15 B) were obtained over a period of 3 min and corrected for decay according to the method of Raichle et al. (1983). Blood samples were collected manually at ∼5-s intervals and assayed in a calibrated well-counter. As the sensitivity of K1 to tracer dispersion and delay was expected to be minimal, as revealed by simulations (Ohta et al., 1992), the difference in tracer arrival between the brain and the peripheral sampling site was corrected by the simple slope method (Herscovitch et al., 1983b; Meyer, 1989), and dispersion, already minimized by the relatively large flow rate in the arterial sampling catheter, was not accounted for. Maps of Vo were generated under the same conditions. However, it was expected that Vo, as any purely intravascular component, would be extremely sensitive to the latter two effects. This sensitivity of Vo to errors in tracer dispersion and delay was confirmed by means of simulations. In four studies, tracer dispersion and delay were, therefore, carefully corrected, as outlined by Iida et al. (Iida et al., 1989), and adapted to our model with inclusion of Vo (Fujita et al., 1992). From these four studies, Vo values were derived, although quantification of this parameter was not the primary objective of this study. A dispersion time constant of τ = 4 s was used. K1 and Vo values were determined for circular ROIs, each with an area of 3.7 cm2, which were evenly placed over two brain slices (20 ROIs per slice) situated ∼40 mm above the orbitomeatal (OM)-line. Care was taken to avoid ventricular space as well as larger vessels.
[C15]O and [15O]O2 were used in one subject to compare the CO-derived steady-state vascular volume (reflecting the total vascular volume) and the O2-derived apparent vascular volume (expected to reflect mainly the arterial plus part of the venous volume) with the H2O-derived apparent vascular volume (expected to reflect mainly the arterial volume), in order to test the validity of the numerical values provided by the corresponding Vo maps.
[15O]O2 was administered by bolus inhalation and [C15]O by continuous inhalation for 2 min (Grubb et al., 1978; Meyer et al., 1987).
Each subject underwent a magnetic resonance (MR) scan (Phillips Gyroscan 1.5 Tesla superconducting magnet system; Best, Holland). MR images (MRIs) were coregistered with PET images to corresponding imaging planes by means of a procedure developed by Evans et al. (1989).
Data analysis
From the dynamic PET data, which had a reconstructed spatial resolution of 10 mm FWHM, we generated 388 tissue time-activity records from circular ROIs, each with an area of 3.7 cm2, uniformly distributed over the entire area of the selected brain slices. From 15 bolus H215O studies, 289 curves were obtained from normocapnic studies and 99 from hypocapnic studies. Blood flow was calculated using one-compartment analyses, which included two-parameter regression, two-weight integration, look-up table, and polynominal approximation methods. For the latter two methods, a tissue-blood partition coefficient of 0.9 ml g−1 and 60 s of data were used.
The clearance of H215O, K1, and the apparent vascular distribution volume, Vo, were calculated by three-parameter regression and three-weight integration, both based on two-compartment analysis. The three-weighted integration is computationally more demanding than is the two-weight integration, but yields 128 × 128 pixel maps of K1, k2, and Vo in 1.5 min with a Microvax III (Ohta et al., 1992).
For all regression analyses, a 3-min data segment was used. Functional images were processed with a nine-point smoothing filter. Despite the relatively large ROI size, tissue-heterogeneity effects were not expected to significantly affect K1 estimates, as suggested by simulation (Ohta et al., 1992). Images of Vo were merged with corresponding MRIs for ease of analysis and interpretation.
RESULTS
Simulated PET studies
Simulated PET studies confirmed that the three-parameter analysis by weighted integration, in theory, yields precise estimates of apparent water clearance, K1 (Fig. 2A). The fractional root-mean-squared (RMS) error inherent in the k2 and Vo estimates by three-weight integration, with 5% Poisson variability added to both arterial and tissue radioactivity, increased markedly for small values of k2 and Vo (Fig. 2 B,C). However, the error in K1 was never >5%. K1 was also relatively insensitive to dispersion effects of the arterial input function (Fig. 3A), arrival time difference of tracer between sampled site and brain (Fig. 3B), and tissue heterogeneity (Fig. 3C) (Herscovitch et al., 1983a; Iida et al., 1986; Kanno et al., 1987; Meyer, 1989). Vo, on the other hand, was extremely sensitive to systematic errors of tracer dispersion and arrival time estimates (Fig. 4A,B), while being moderately affected by tissue heterogeneity (Fig. 4C). Underestimation of the magnitude of any one of these three factors tended to elevate the estimate of Vo, to account for the presence of otherwise unexplained radioactivity in the tissue.
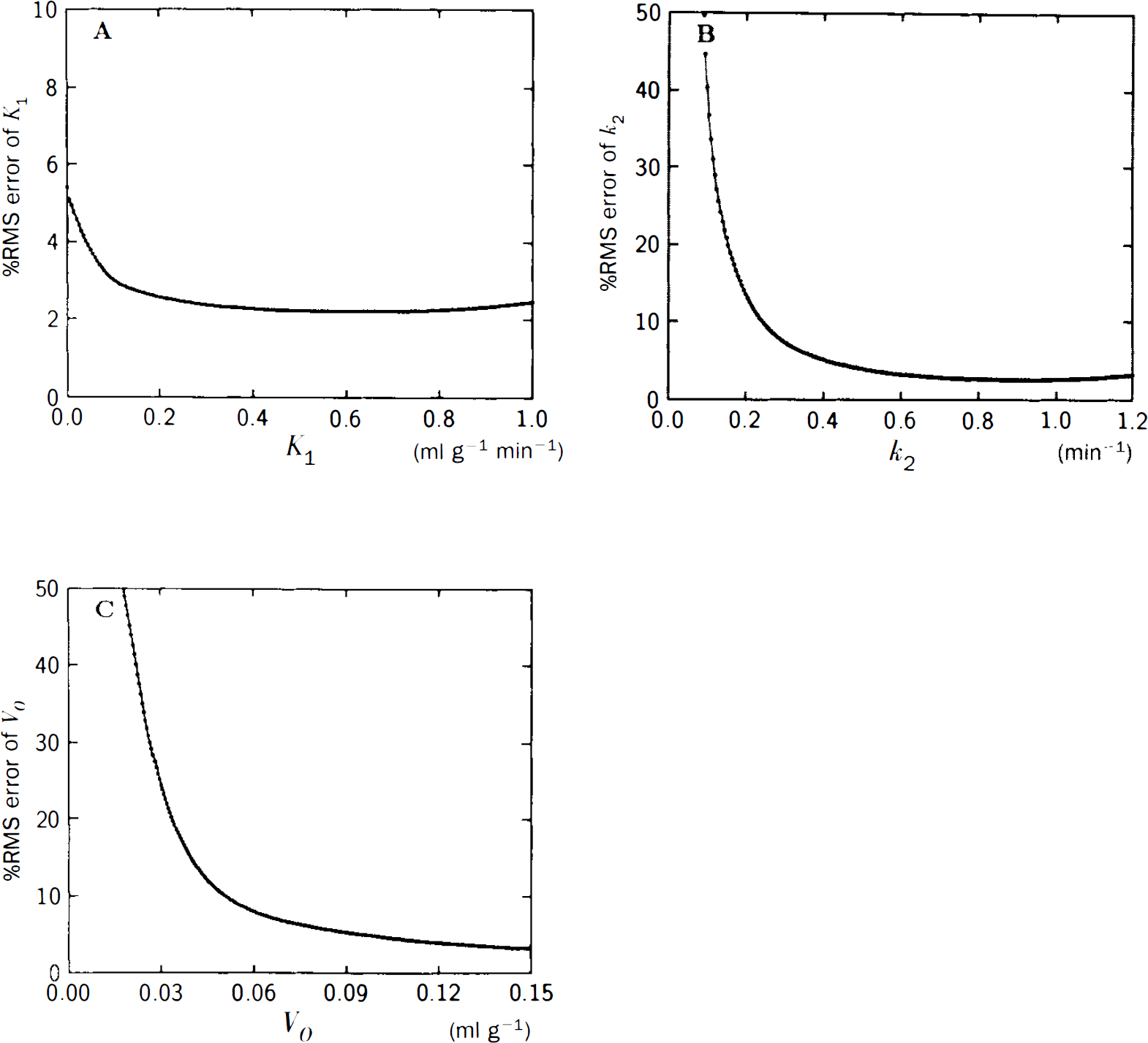
Root-mean-squared (RMS) errors of K1, k2, and Vo estimates determined by the three-weight integration after superposition of 5% statistical variability onto the arterial and tissue records.
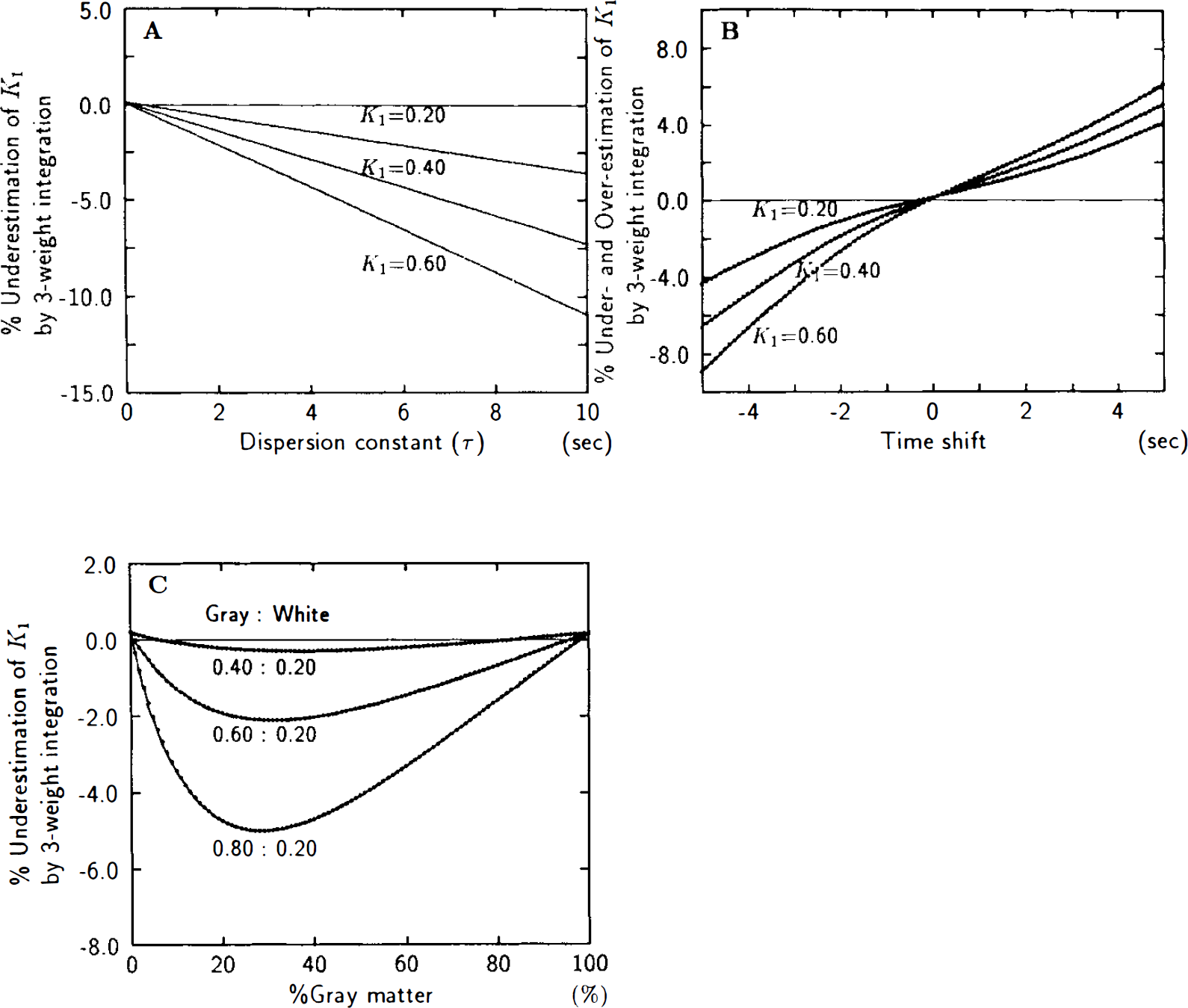
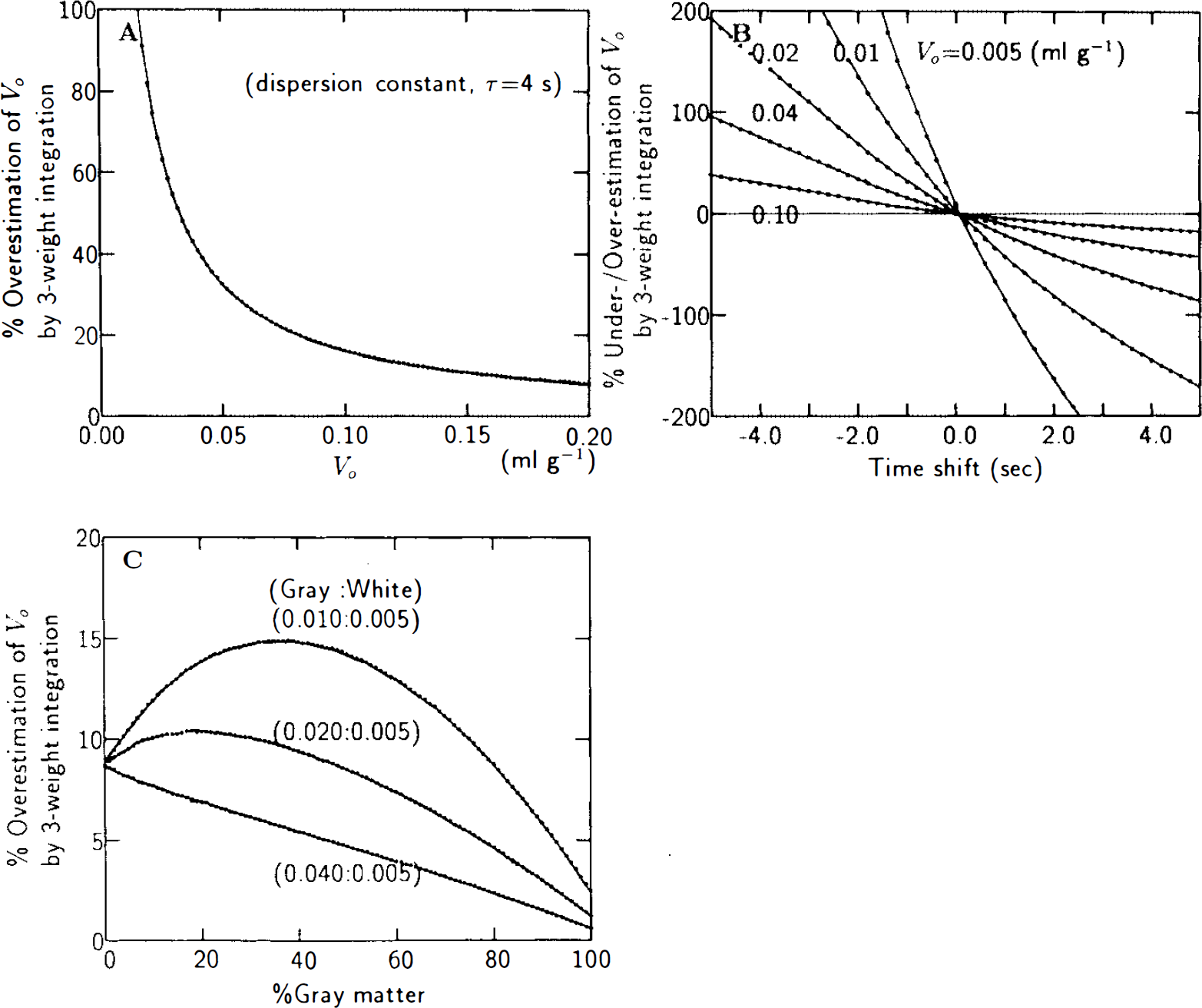
Human studies
The real PET studies showed that F′ estimates always exceed K1 estimates. By F′, we denote the value of K1 estimated by means of the simplified single compartment model. Values of F′ (”blood flow”) calculated on the basis of the one-compartment analyses [two-parameter regression (Fig. 5A), two-weight integration (Fig. 5B), look-up table (Fig. 5C), and polynominal approximation (Fig. 5D)] were compared with values of K1 obtained by the two-compartment model. Calculations were performed for a total of 388 regions in nine subjects. As expected, the discrepancy between F′ and K1 markedly increased for larger values of the water clearance, K1. This overestimation of K1 by the single-compartment approach increased when Vo increased (Fig. 6). The degree of overestimation was positively correlated to the size of Vo.
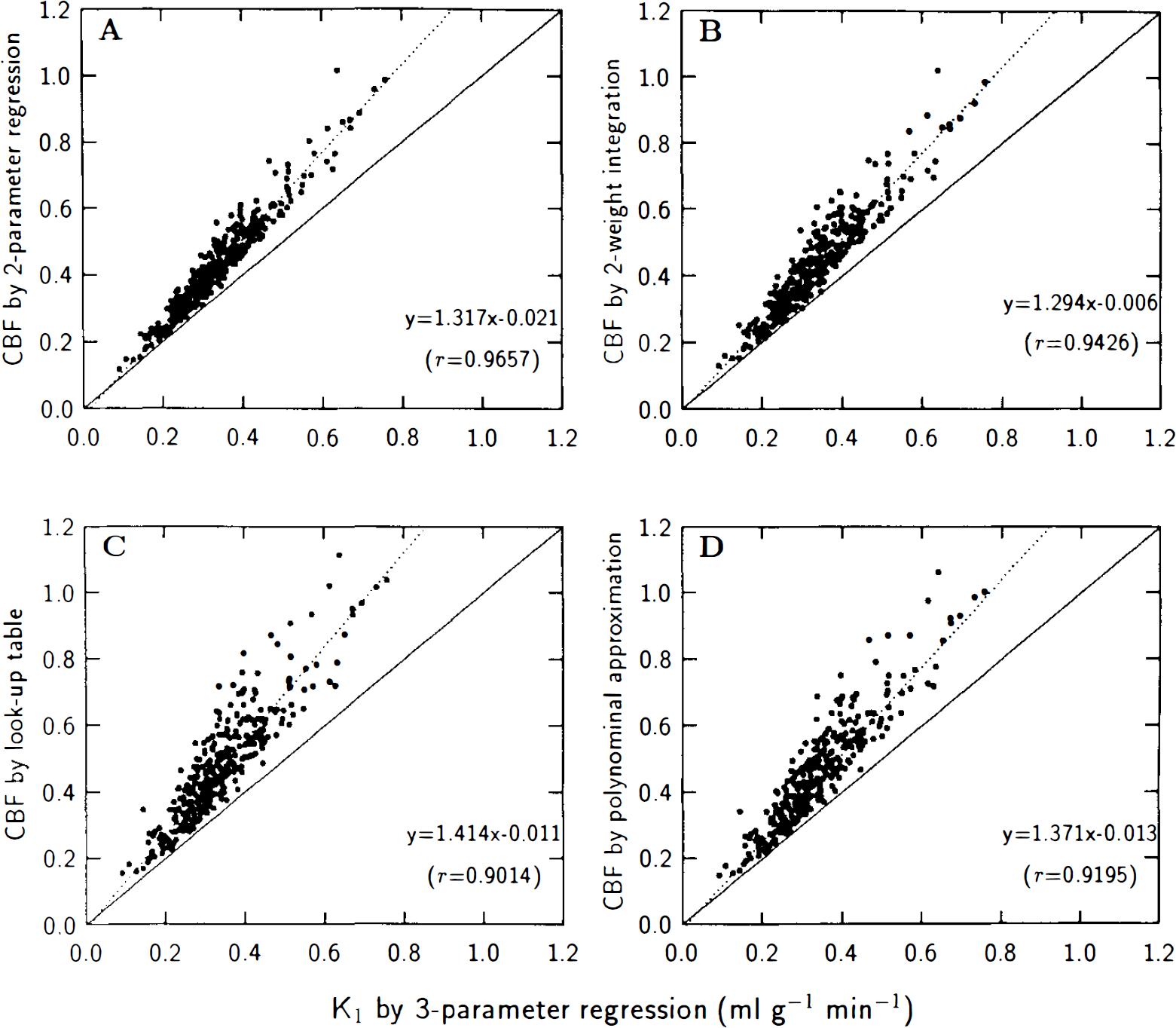
Water clearance, K1, calculated by three-parameter regression, compared with F′ calculated according to one-compartment model, using in turn two-parameter regression, two-weight integration, look-up table, and polynominal approximation methods. The slopes of regression lines significantly exceeded unity (n = 388, p < 0.001).
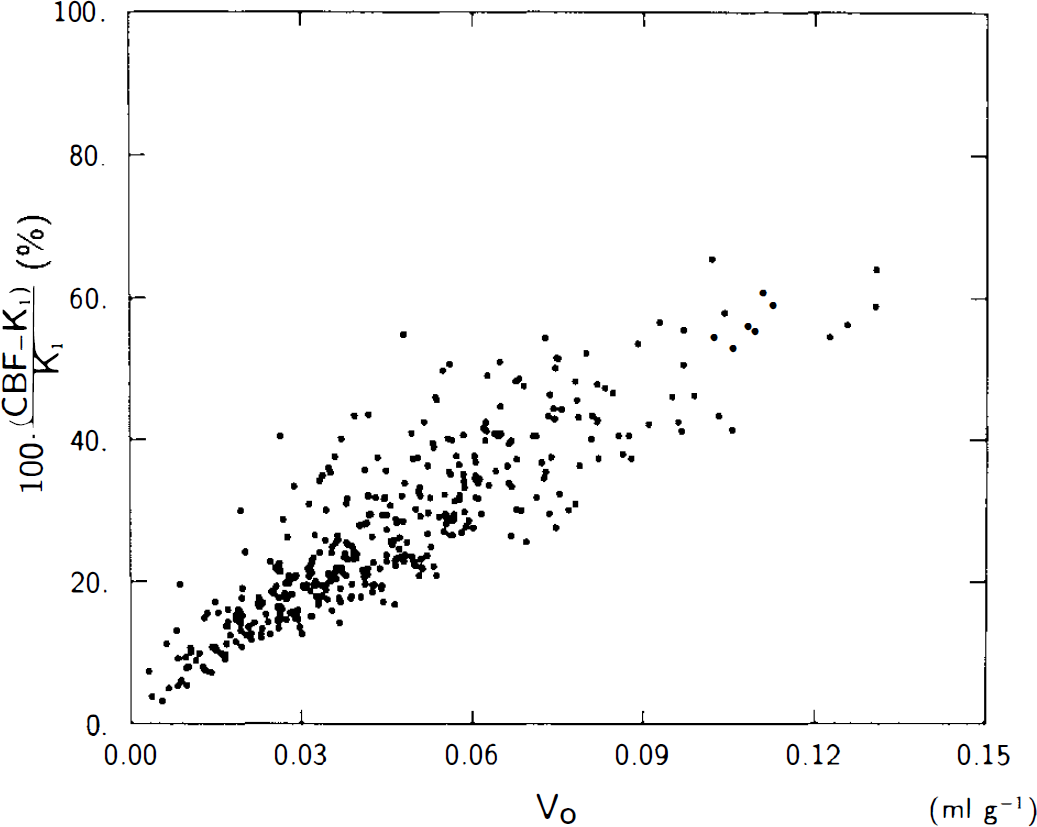
Percentage overestimation of F′ calculated by two-parameter regression, relative to water clearance, K1, calculated by three-parameter regression, versus apparent vascular distribution volume, Vo, obtained by three-parameter regression. Note significant linear relationship between variables (n = 388, y = 454.43x + 6.7, r = 0.8552, p < 0.005).
Estimates of K1 were considerably less time-dependent than those of F′ as evidenced by the calculated mean values and standard deviations of F′ and K1 for the 289 normocapnic ROIs as a function of integration time (Fig. 7). Figure 8 illustrates the tissue time-activity curves predicted by the one-and two-compartment models. The two-compartment model (three-weight integration) described the observed data (black dots) well over the entire observation interval, in contrast to the one-compartment analyses (autoradiography and two-weight integration), which did not. Since tissue heterogeneity effects are not only minimal but very similar for the one- and two-compartment models (Fig. 3C and Fig. 4 in Ohta et al., 1992), we have used tissue data from an entire brain slice, which presents a lower noise level and, therefore, gives a clearer picture of the goodness of the various fitted curves. The result strongly validates the two-compartment model and its K1 estimates.
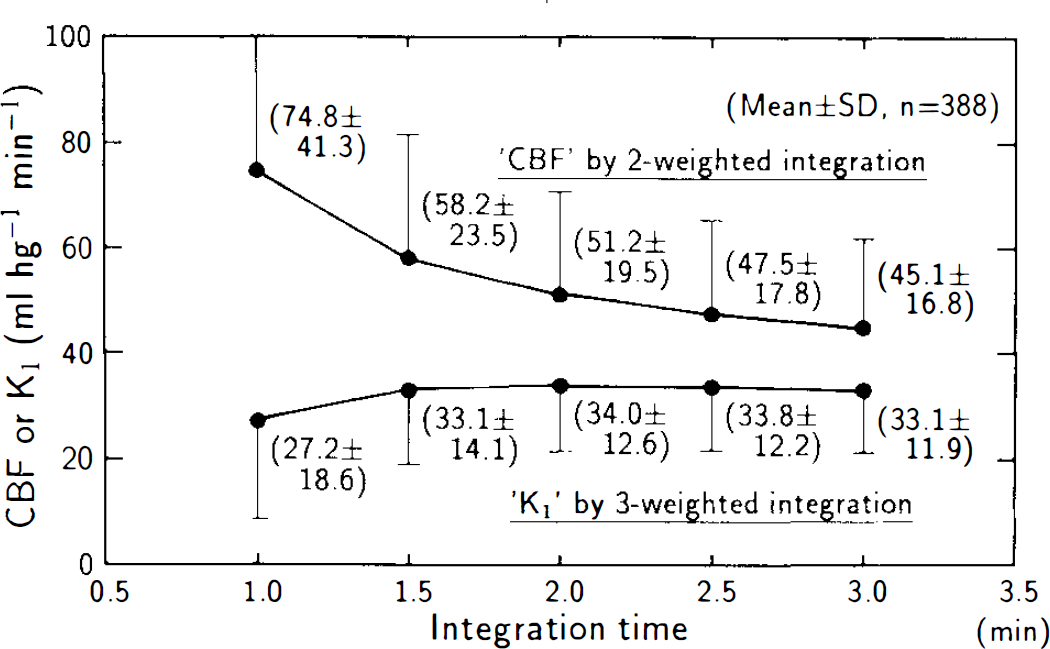
Mean and standard deviation of F′, for all regions at normocapnia, calculated by two-weight integration, and K1 calculated by three-weight integration, as function of data collection (integration) time. Dots and vertical bars represent mean value and standard deviation, respectively.
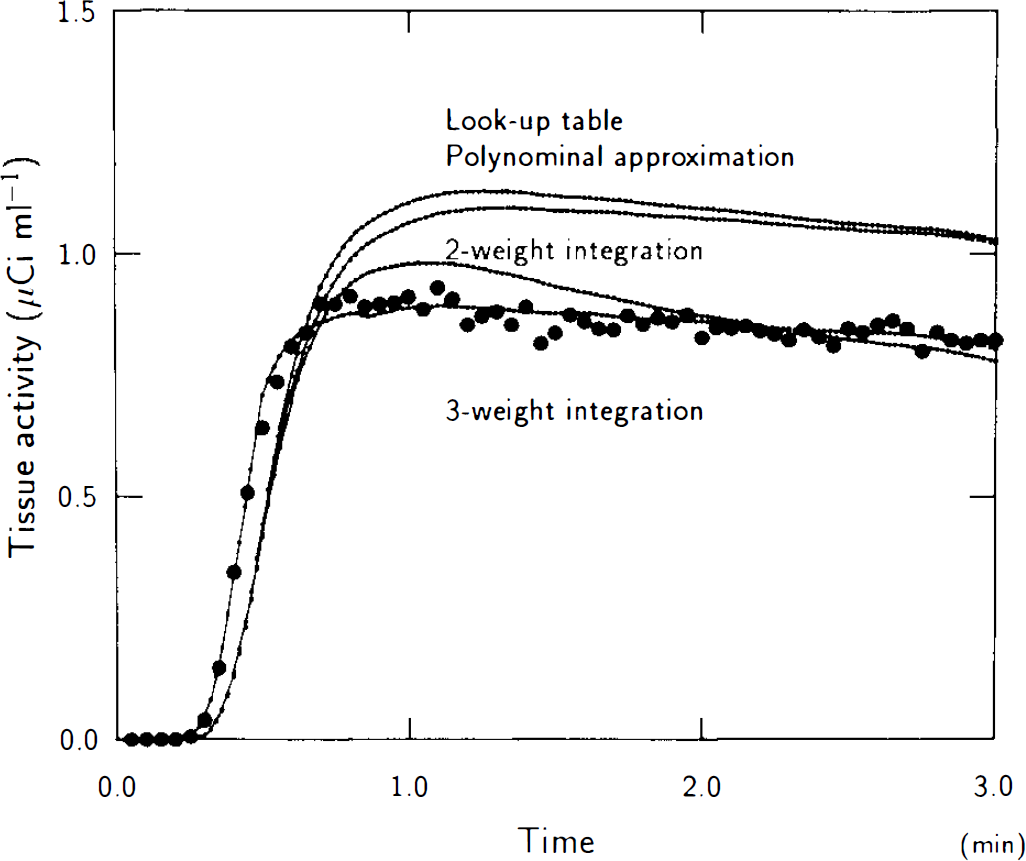
Single-plane H215O tissue time-activity record (dots), compared with record predicted by methods based on one-or two-compartment models. Note excellent fit obtained with three-weight integration (two-compartment) method.
Mean values and standard deviations of F′ and K1 are summarized in Table 3. F′ estimates were close to accepted average CBF values (Lassen, 1985), but significantly larger than estimates of K1 by two-compartment analysis (p < 0.001). Table 4 lists values of F′/λ and k2, the values from the one-compartment analysis again being larger than those from the two-compartment analysis.
F′/λ and K2
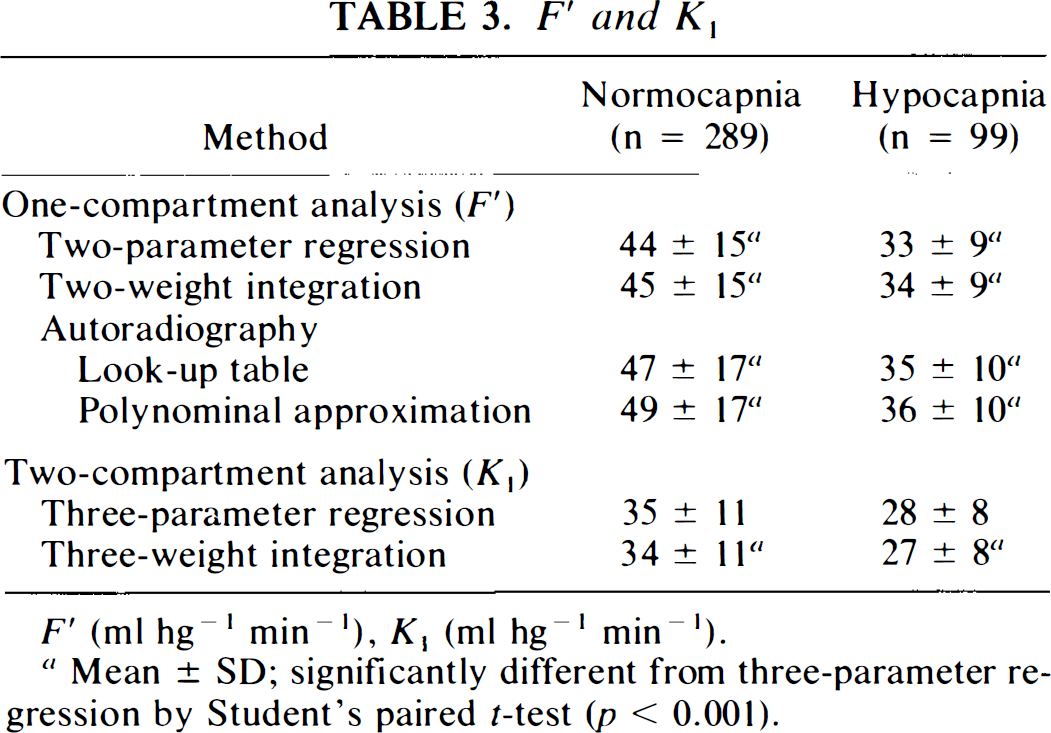
F′ (ml hg−1 min−1), K1 (ml hg−1 min−1).
Mean ± SD; significantly different from three-parameter regression by Student's paired Mest (p < 0.001).
F′/λ and k2
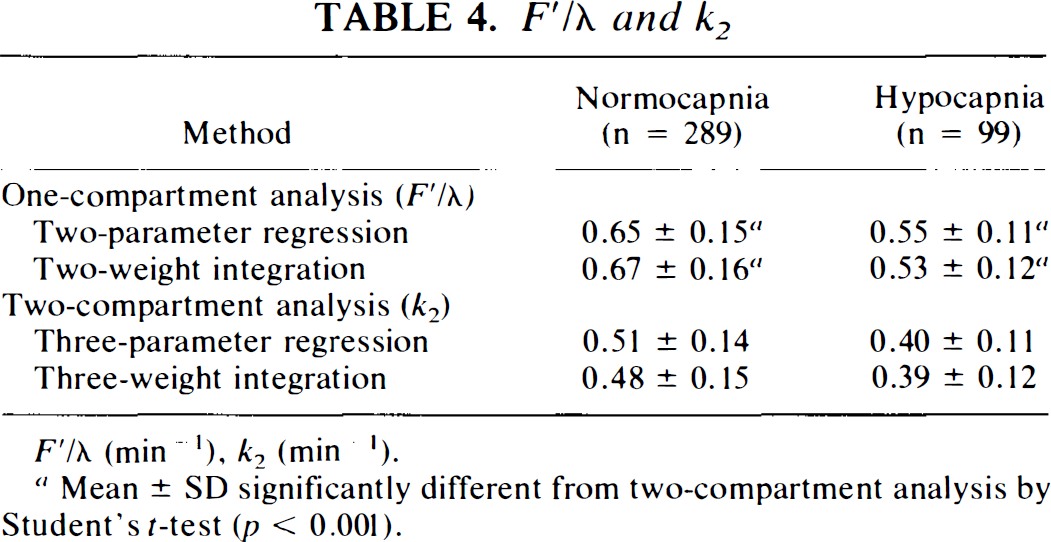
F′/λ (min−1), k2 (min−1).
Mean ± SD significantly different from two-compartment analysis by Student's t-test (p < 0.001).
K1 values ± SD determined in four studies without dispersion correction (τ = 0) and using the slope method for tracer delay estimation were 42.10 ± 4.10, 38.38 ± 3.60, 41.50 ± 3.20, and 39.55 ± 5.21 ml hg−1 min−1 while, with dispersion correction (τ = 4 s) and estimation of tracer delay using a global fitting approach (Iida et al., 1989), the corresponding values were 43.43 ± 4.10,39.86 ± 3.93, 41.66 ± 3.20, and 39.34 ± 5.32 ml hg− min−1. As predicted by the simulations, the difference between the two data sets was not statistically significant (p < 0.6).
In the same four studies, Vo values obtained with careful delay and dispersion correction were 1.14 ± 1.18, 0.81 ± 0.71, 1.04 ± 0.93, and 1.22 ± 1.04 ml hg−1, with a global mean of 1.05 ml hg−1, which represents about one-fifty to one-quarter of the average cerebral blood volume (Gjedde, 1988). The large standard deviations were predicted by our simulations (Fig. 2C0.
Hypocapnia caused a reduction of 20–25% for all parameters, regardless of the method of analysis.
Maps
The parameter maps visualize the results obtained, namely that the true water clearance, K1, is consistently lower than the conventionally derived “blood flow” value, F′, and that this discrepancy is a function, in part, of the apparent vascular distribution, Vo (Figs. 9, 10). The distribution of the Vo maps are consistent with the distribution of known vascular beds. Figures 9 and 10 were derived from a selected water bolus study completed in a single subject using the PC–2048 15B (Scanditronix) tomograph. They enable the comparison of values of F′, K1, and Vo for various, more or less vascularized, cortical and subcortical brain regions. The two transaxial planes represent (a) an F′ map generated by the look-up table, (b) an F′ map generated by the two-weight integration, (c) a K1 map generated by the three-weight integration, (d) a Vo map generated by the three-weight integration, and (e) a merger of Vo with the corresponding MR map. The plane shown in Fig. 9 is 6.5 mm above the OM line. The merged maps (Fig. 9E) identify the circle of Willis in the basal cisterna, the middle cerebral arteries in the Sylvian stem, and the posterior cerebral arteries in the ambient cisternae. The F′ maps (Fig. 9A,B) show erroneously high blood flow in the area of the basal cisterna, in which there is no tissue and blood flow, therefore, should be zero. This error does not appear in the K1 map (Fig. 9C).
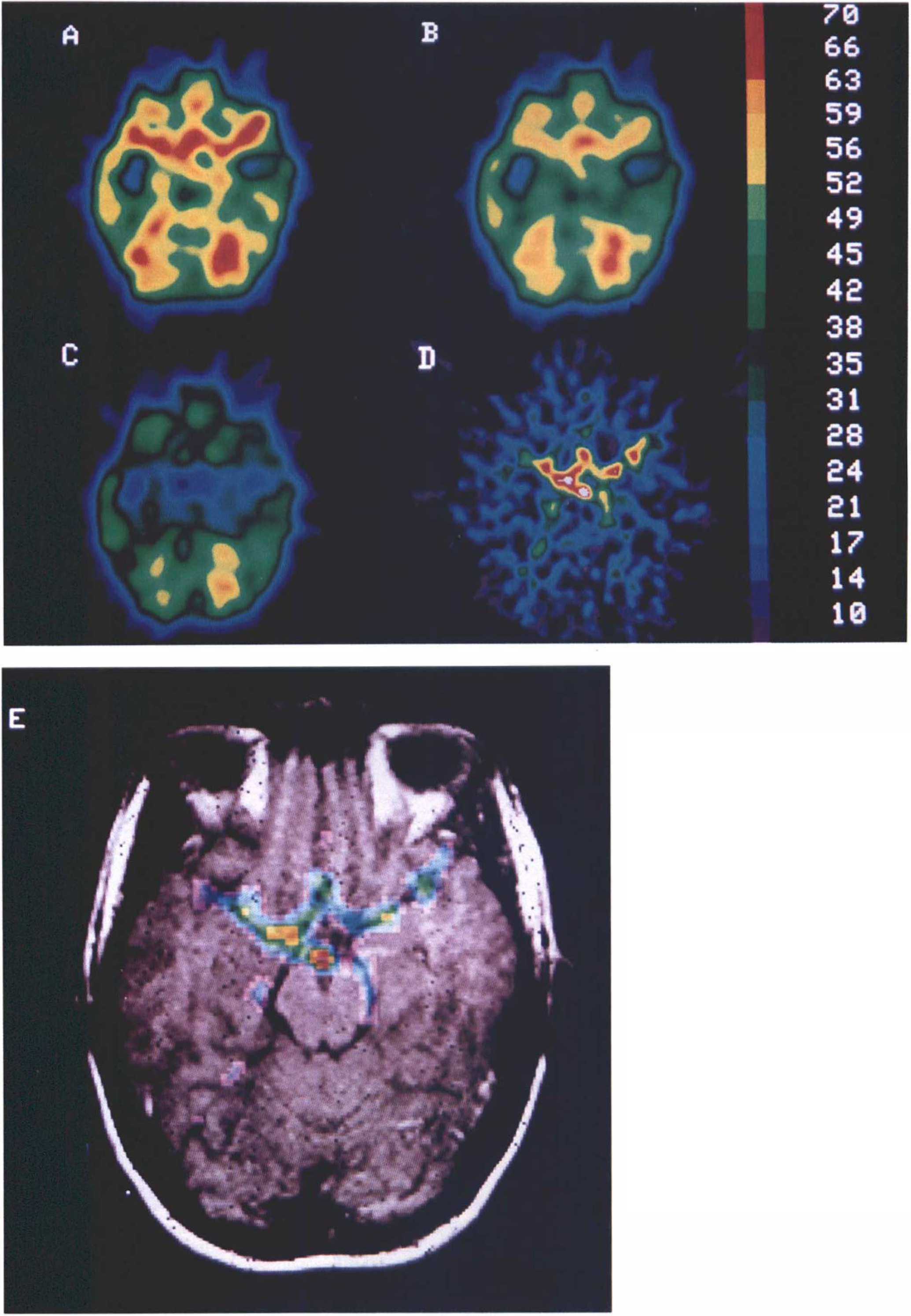
Maps generated from H215O study with the PC–2048 15B tomograph at 6.5 mm above OM line.
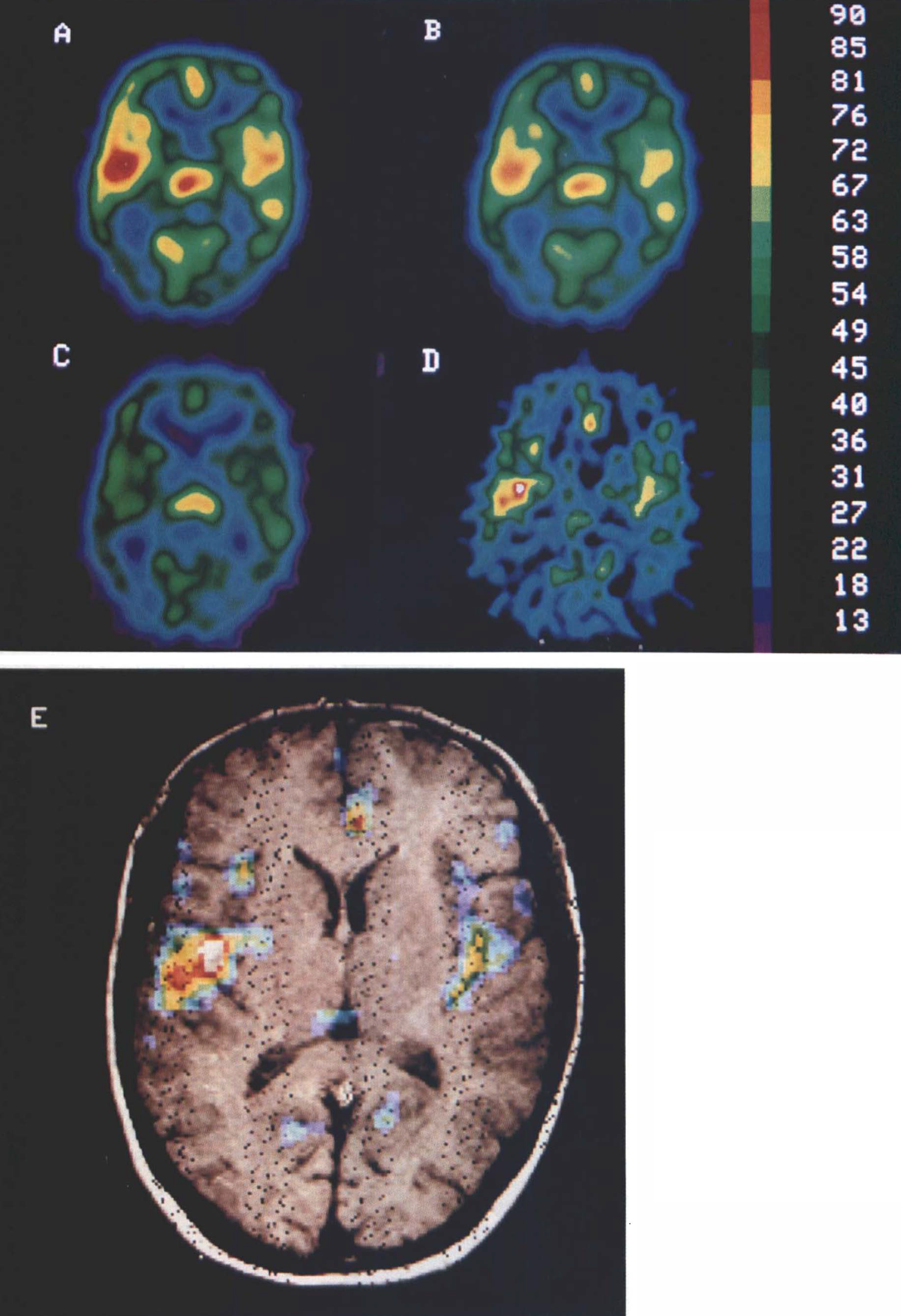
Maps generated from H215O study as in Fig. 9, but 39 mm above OM line. Maps
At 39 mm above the OM line (Fig. 10), high intensities are seen (Fig. 10A,B) bilaterally in the insular cisternae (Fig. 10E), which gives the erroneous impression of high blood flow rates in the poles of the temporal lobes and insular gyri. The merged maps (Fig. 10E) demonstrate that the error arises from the middle cerebral arteries in the Sylvian fissure. As evident from the K1 map (Fig. 10C), the water clearance is no larger in these regions than in other areas of the cortex. The pericallosal artery in the interhemispheric fissure and the medial occipital arteries in the parieto-occipital sulci also are clearly shown in the superposition maps (Fig. 10E) and affect the flow maps (Fig. 10A,B) in the manner just described.
In order to further illustrate the concept of the apparent vascular distribution volume of a tracer, we created three different Vo maps in the plane of the glabellainion line from three studies completed in a single subject using three different tracers (Fig. 11). Figure 11A is a Vo map from a study with H215O, which has a relatively high unidirectional extraction. Figure 11B is a Vo map, obtained as described by Ohta et al., 1992, from a study with [15O]O2, which has a relatively low unidirectional extraction. Figure 11C is a Vo map, obtained as described by Ohta et al., 1992 from a study with [C15]O, which has a negligible unidirectional extraction.
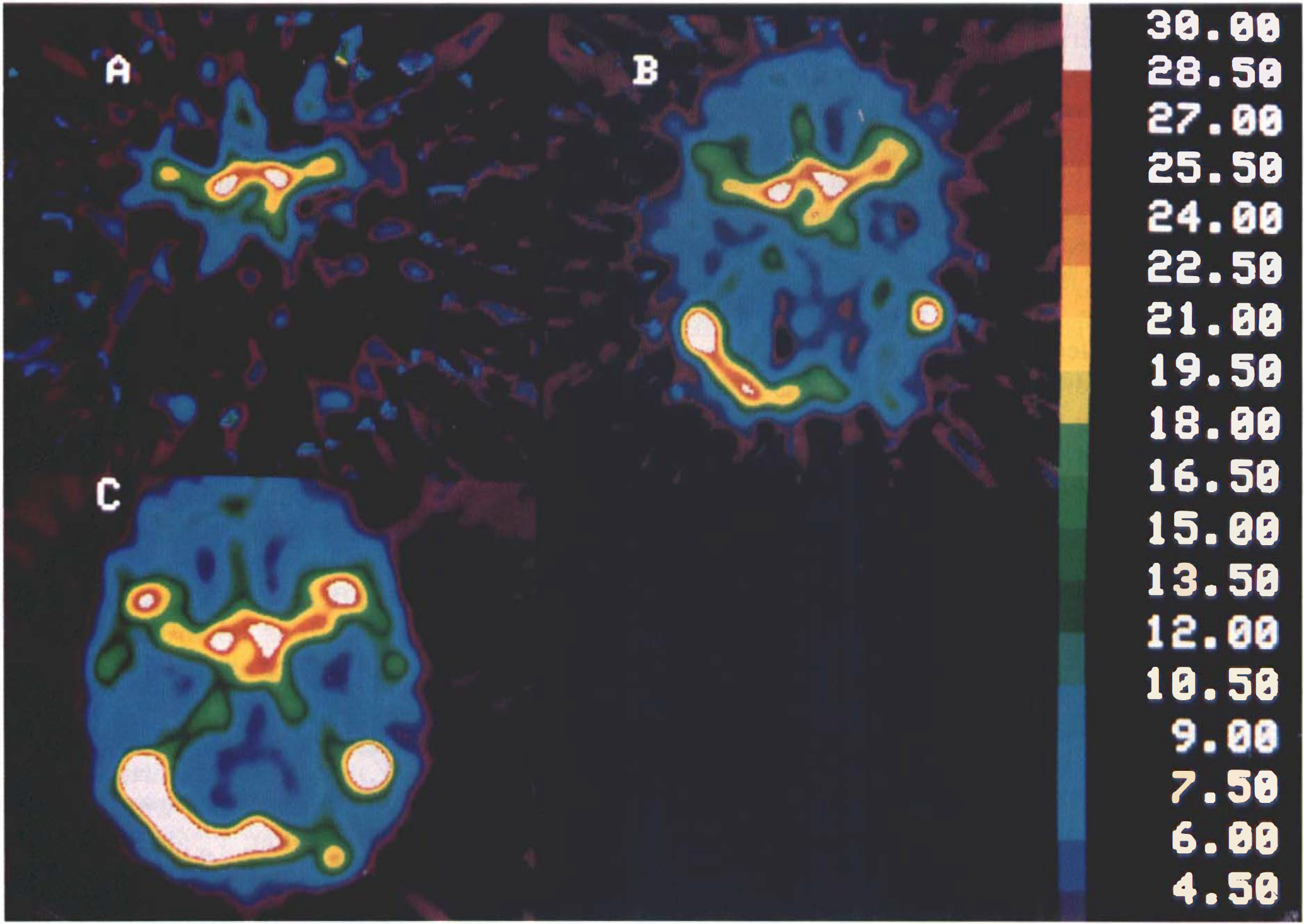
Maps of apparent vascular distribution volume, Vo, obtained at level of glabellainion line from three different studies in same subject.
Comparison of the Vo maps supports the following interpretations: The map derived from the [C15]O study represents the whole blood volume, while the Vo map, calculated from the H215O distribution, represents mainly the arterial volume such as that in the middle cerebral arteries in the Sylvian stem. Finally, the [15O]O2-derived Vo map includes part of the venous volume, such as that in the transverse sinus in addition to the middle cerebral arteries, as predicted by Eq. 31.
DISCUSSION
In this report, we show that the single-compartment analysis of brain water uptake significantly overestimates cerebral water clearance. This finding, based on the model proposed here, is not consistent with the claim that flow rates measured with water are too low. Despite this flaw, H215O is widely used for the measurement of CBF by PET. The most common implementations are the single-image continuous inhalation (Frackowiak et al., 1980) and bolus injection (Herscovitch et al., 1983b; Kanno et al., 1987; Raichle et al., 1983) methods. Kinetic (multiframe) methods also have been proposed (Carson et al., 1986), but have not reached the same popularity. Both implementations are based on Kety's single-compartment model (Kety, 1951), which assumes that the tracer is perfectly diffusible, i.e., that there is no diffusion barrier between blood and brain. However, the accuracy of blood flow estimates based on the single-compartment analysis of water uptake has been questioned often, and it has been shown that the diffusion limitation may result in significant underestimation of blood flow (Herscovitch et al., 1987; Raichle et al., 1983). The assumption of an instantaneous equilibrium between blood and brain parenchyma is also an oversimplification, since venous radioactivity concentration is higher than that of brain tissue when complete equilibrium between postarterial blood and tissue is not achieved in a single capillary transit (Koeppe et al., 1987).
Two-compartment and distributed-parameter models have recently been proposed to provide a more adequate description of dynamic blood flow tracer data (Gambhir et al., 1987; Larson et al., 1987; Sawada et al., 1987). These models require at least three parameters, rendering the generation of pixel-by-pixel maps of blood flow from PET data impractical with current algorithms.
In addition to the residual radioactivity in the vascular bed and the limited clearance of the tracer across the capillary endothelium, analysis of water clearance by brain is also affected by the delayed arrival of tracer and its dispersion at the peripheral arterial sampling site. The latter two factors tend to affect estimates of clearance, partition volume, and residual vascular radioactivity, since excess radioactivity in brain may mean either a vascular residuum, early arrival of the bolus, or both. Thus, the corrections for these factors tend to be inversely correlated (Lammertsma et al., 1990).
The present study examines the effect of the delayed equilibration of H215O with brain tissue to answer the question whether reliable clearance rates can be obtained by a one-compartment analysis of cerebral H215O uptake. For this purpose, we studied a two-compartment model consisting of extravascular brain tissue, i.e., brain parenchyma with activity, Me(t), and a vascular component characterized by the apparent vascular distribution volume, Vo (Evans et al., 1986; Gjedde, 1981; Ohta et al., 1992).
Since a major advantage of PET is its ability to generate functional maps of the brain, visual comparison of images of the same parameter derived from different models provides a qualitative evaluation of the various approaches. Comparison of F′ maps obtained with the one-compartment model (Figs. 9A and 10A, “standard method”) and K1 maps obtained with the two-compartment model (Figs. 9C and 10C, note that these maps look like blood flow maps, except for a possibly nonlinear scaling function, since K1 = E0F for Vc << Ve) revealed striking regional differences, indicating that F′ estimates based on the single-compartment approach must grossly overestimate true water clearance in regions with high, predominantly arterial, vascularity. In some cases, the errors caused by ignoring the vascular radioactivity component were >25%. Other errors, such as tracer arrival delay and dispersion of the arterial input function, tissue heterogeneity and incorrect choice of partition coefficient, were rarely >15%. In other words, the difference between F′ and K1 cannot be fully eliminated by manipulation of the arterial input function (Iida et al., 1986; Lammertsma et al., 1990).
Compared with K1 images, the many artifacts in the F′ images are easily recognized, particularly the erroneously high blood flow values in the regions of the major arteries or in the cisternae. As an example, blood flow appeared to be high in the temporal lobes (Fig. 10A,B) due to the proximity of the middle cerebral arteries on the surface of the insular gyrus. This means that the presently accepted values of blood flow for the temporal cortex may be overestimations, since K1 values appeared not to be higher in the temporal lobe than in other areas of the cortex (Fig. 10C). When compared to corresponding K1 values, the artificially high conventional F′ values, particularly in highly vascularized regions, indicate that instantaneous equilibrium of H215O between brain tissue and blood is not achieved, contrary to the fundamental assumption underlying the one-compartment model. In other words, blood flow values derived from the one-compartment analysis have limited physiological meaning.
With regard to Vo, which is displayed in Fig. 11 (as derived from studies performed on the same subject with three different tracers, namely H215O, [15O]O2, and [C15]O2) it is apparent that, as expected, its magnitude decreases as the capillary permeability of the tracer increases (see also Eq. 25). Derived from H215O with its relatively high unidirectional extraction, Vo represents the arterial volume, while when derived from [15O]O2 with a relatively low unidirectional extraction, Vo also includes part of the venous volume. Finally, Vo obtained from [C15]O with its negligible unidirectional extraction reveals the entire arterial and venous circulation. These observations are in line with blood volume estimates derived from 18F-labeled 2-fluoro-2-deoxy-D-glucose (18FDG) studies (unidirectional extraction fraction of ∼0.2) by Evans et al. (1986).
Values of blood volume and Vo can be read from Fig. 11, noting that Vo, unlike K1, is very sensitive to dispersion of the tracer in the radial artery and errors in the time delay determination (Fig. 4A,B). For the generation of K1 and Vo maps, dispersion in the radial artery is accounted for in a subsequent article, rather than in the present. For K1 estimates, the consequent error was <5% for a typical dispersion time constant of τ = 4 s (Fig. 3A). On the other hand, Vo is so sensitive to image noise (Fig. 2C) as well as to the effects of dispersion and tracer arrival delay (Fig. 4) that its exact magnitude is highly uncertain. In particular, ignoring dispersion always results in an overestimation of Vo. Our attempt to quantify Vo in four cases in which dispersion and tracer arrival delay had been carefully corrected yielded an average of Vo = 1.05 ml hg−1, which amounts to 22% of an accepted literature value for the entire average cerebral blood volume of 4.8 ml hg−1 (Gjedde, 1988). This result is consistent with Eq. 25 and our observation (Figs. 9–11) that Vo derived from H215O is weighted towards the arterial component of the cerebral vascular volume. When mapped on a pixel-by-pixel basis, Vo estimates accurately outline major vessels in many areas (Fujita et al., 1996). We regard this as sufficient assurance that two compartments are more appropriate than one compartment for providing accurate estimations of CBF.
Our measurements on human subjects, analyzed with a more realistic two-compartment model of transport and clearance at the capillary endothelium, suggest that the widely used one-compartment model systematically overestimates local CBF. We note that, even when an infinitely diffusible tracer is used, a correction for the vascular component may, therefore, be necessary to account for tracer not exchanged in the arterial vasculature under physiological conditions.
Footnotes
Acknowledgment:
This work was supported by MRC (Canada) grants PG-41 and SP-30, the Isaac Walton Killam Fellowship Fund of the Montreal Neurological Institute, and by the Quebec Heart Foundation. We thank the technical staff of the McConnell Brain Imaging Centre, and the Neurophotography Department for their assistance. Special thanks are due Mr. Peter Neelin for providing the registered MR images. The helpful comments of Dr. Hiroto Kuwabara as well as the contribution of Professor Ludvik Bass, Department of Biomathematics, University of Brisbane, Brisbane, Australia, are particularly appreciated.
APPENDIX
Two-compartment analysis. Capillary permeability. Assume that local microvascular properties (F, PdA, Vd) are uniform within a brain ROI in which Pd is the apparent permeability of the endothelium to a tracer passing from the blood to the tissue, A is the capillary surface area, F is the blood flow, Vc is the volume of the capillary bed, Cc is the concentration of the tracer in this volume, Vd is the volume of the extravascular space, Ce is the uniform tracer concentration in this space, α1 is the solubility coefficient of the capillary fluid, and α2 is the solubility coefficient of the extravascular fluid. Initially, we assume that the region includes no arterial or venous volume. Below, these will be added to the region (see Arterial and venous volumes). The compartmental assumption that Ce is uniform means that the incremental addition made to Ce during a single passage of a single capillary is negligible. It also means that discontinuities must exist at both ends of the capillary bed, such that the concentration is Ca at the single inlet and, on the average, C̅c in the capillary bed between the single inlet and single outlet. Also, it is assumed that brain capillaries do not run in parallel; rather, they are believed to form intricate convolutions that contribute to the uniformity of Ce.
The solubility coefficients are defined as the ratios α1 ≡ Vc/Vωc and α2 ≡ Vd/Vωd where Vcω and Vdω are defined as the water volumes of Vc and Vd, respectively. Assuming uniform concentration within an elemental capillary crossection, tracer transport across the entire wall of the capillary bed is then described by the equation
Derivation of Mc. The symbol Mc represents the quantity of tracer in the capillary bed, equal to the product of Vc and C̅c. To determine C̅c, let Cc(τ) be the capillary tracer concentration in a small volume of plasma, ΔVc, at the time τ after its entry into a capillary
where Me is the tracer product VdCe, which is not assumed to change appreciably during a single capillary transit. The average of all transit times, , equals Vc/F. As ΔA/ΔVc = A/Vc and Cc(0) = Ca, integration from τ = 0 to τ = u for any single transit yields
Here, we define P = PdA/α1 and Ve = Vd/α1. The term Ve represents the tissue distribution volume, corrected for any difference between the solubility coefficients of the capillary bed and the brain tissue proper. The capillary bed can now be simulated by a single imaginary vessel with a volume of Vc, surface area A, and flow of F, with sufficient cross-sectional involutions to render cross-sectional gradients nonexistent. The mean tracer concentration in this vessel is then the average of all concentrations of this capillary
where 1 — e−P/F is revealed as the extraction fraction, Eo, for unidirectional transfer of tracer into the tissue (Crone, 1963), i.e., for Me = 0
The concentration C̅c varies both with Ca and with the tissue concentration of tracer, which, in turn varies with Ca. Generally speaking, therefore, C̅c is lowest initially when Ce ∼ 0 and highest at the steady-state level Ca.
where Vo is the apparent vascular volume of distribution. Since we have not yet introduced arterial and venous volumes, Vo is temporarily defined as the portion of the true vascular volume of distribution, Vc, that equals the quotient VcEoF/P. Below, Vo and V will be expanded with arterial and venous volumes. Equation 11 indicates that the capillary tracer content, Mc, changes as a function of Me. This result shows that the tracer in the capillary bed has two apparent states, one at the arterial concentration in the volume Vo, and one at the tissue concentration (corrected for a difference in solubility) in the remaining volume of the capillary bed.
in which Me is the product Vd × Ce. When k2 is the rate constant of tracer washout from the compartment Me
Eq. 15 reduces to
The solution to Eq. 17 is
Derivation of M. For a two-compartment system (Fig. 1, left panel), having the two compartments Mc and Me, according to Eq. 14, the total amount of tracer in the RO1 is
where V ≡ Vc + Ve.
where K1, the water blood-brain clearance, is defined by the relationship
The term clearance is adopted from kidney physiology where it refers to the removal of substances from the circulation. Equations 20 is the solution to the equation
From the definition of Vo, it follows that Vo approaches the capillary volume Vc and V — Vo the partition volume Ve, only for low values of P relative to EoF. For large values of P, Vo includes no fraction of the capillary vascular bed (Vo approaches Va, see below for explanation), and V — Vo approaches Ve + Vc.
By integration of Eq. 22, we obtain the integral solution
Arterial and venous volumes. The above derivation was based on tissue with exchange vessels and extravascular tissue, i.e., tissue without appreciable arterial or venous components. For tissue that also includes arteries and arterioles, and venules, and veins, represented by the volume Va and Vv, respectively, the terms V and Vo in Eq. 19, 21, and 23 must be expanded as follows:
and
Thus, when P is large relative to F, Eo approaches unity and Vo reduces to Va.
One-compartment analysis. When H215O is used to measure CBF by a one-compartment analysis, the tacit assumption is made that H215O is inert and perfectly diffusible, i.e., faces no diffusion barriers between the vascular space and brain tissue, and that tracer delivery to the brain is only limited by the blood flow, F (Fig. 1, right panel). Thus, in the conventional one-compartment approach, the tracer in brain tissue, including its vascular bed, occupies a single compartment. This model ignores the result of the above derivation, which indicates that Vo is at least equal to Va. Hence, we must evaluate the assumption of zero Vo as if it were appropriate for a tracer of infinitely high permeability. When P >> F, Eo approaches unity, and Vo declines to Va. These changes cause Eq. 22 to reduce to
To obtain the commonly used form, we must assume that Va is zero
where F′ equals FV/Ve and equals V. F′ is the resulting estimate of “blood flow” and λ is the corresponding estimate of the volume V. Therefore, F′ equals the parameter F and λ equals the parameter Ve, only when the vascular volumes Va + Vc + Vv are negligible compared to Ve.
