Abstract
Current smoking is a risk factor for stroke. The aim of this study was to evaluate the effect of smoking one cigarette on cerebral hemodynamics. Using transcranial Doppler ultrasound, we studied the changes of flow velocity after hypercapnia in the middle cerebral arteries (MCAs) of 24 healthy young smokers and 24 healthy controls matched for age and sex. We obtained hypercapnia with breath-holding and evaluated cerebrovascular reactivity with the breath-holding index. In smokers, the evaluation was performed during basal condition, immediately after smoking one cigarette, and at 10-, 20-, and 30-min intervals thereafter. In controls, the evaluation was performed at corresponding time intervals. Breath-holding index (BHI) values at rest were similar for both controls and smokers. In the former, breath-holding index values remained constant for each of the different evaluations. On the contrary, in smokers, breath-holding index values were significantly lower immediately (p < 0.0001), at 10 min (p < 0.001), and at 20 min (p < 0.0001) after smoking with respect to baseline values. Smoking also caused more short-lasting changes, in this case increases in mean flow velocity (MFV), heart rate (HR), and mean blood pressure (MBP). These results suggest that a failure of cerebrovascular regulation occurs after smoking. This phenomenon might contribute to the increased risk of cerebrovascular disease in current smokers.
Smoking is a well-established risk factor for stroke (Molgaard et al., 1986; Colditz et al., 1988; Wolf et al., 1988). However, the cause-and-effect relationship between smoking and cerebrovascular disorders has not yet been clearly identified. Several mechanisms able to cause an increased risk of stroke among smokers have been proposed. These include vascular mechanisms, such as arterial vasoconstriction (Cruickshank et al., 1989), direct endothelial toxic action (Fitzgerald et al., 1988), promotion of atherosclerosis (Crouse et al., 1987), and changes of hematologic factors promoting thrombosis (Ring et al., 1983; Fitzgerald et al., 1988). Two interesting aspects of the association between smoking and cerebrovascular disorders are the decline in stroke risk with smoking cessation (Wolf et al., 1988; Robbins et al., 1994) and the fact that younger adults with the same number of pack-years as older adults seem to run a greater risk of cerebral infarction (Love et al., 1990). These aspects, taken together, might suggest that one of the mechanisms involved in the increased risk of stroke in smokers is related to an acute or short-term modification of cerebral hemodynamics.
In this study, we aimed to test the effect of cigarette smoking on cerebrovascular reactivity in young adult smokers.
SUBJECTS AND METHODS
After giving informed consent, 24 healthy smoker subjects (10 men, 14 women; mean age 32.3 years; range 26–39 years), with no clinical signs or history of cerebrovascular disease participated in the study. Daily cigarette consumption ranged from 15 to 20 cigarettes. The study was performed at 8 a.m. All subjects were drug-free and abstained from smoking, alcohol, and caffeine-containing beverages for at least 10 h prior to the study.
Right middle cerebral artery (MCA) mean flow velocity (MFV) was continuously monitored by means of a Multi-Dop X/TCD transcranial Doppler instrument (DWL Elektronische Systeme GmbH, Germany). One dual 2-MHz transducer fitted on a headband and placed on the temporal bone window was used to obtain continuous measurement. Vascular reactivity was examined by calculating the breath-holding index (BHI) (Markus and Harrison, 1992; Silvestrini et al., 1995). This index is obtained by dividing the percent increase in MFV occurring during breath-holding by the length of time (s) that subjects hold their breath after a normal inspiration.
The study was carried out in a quiet room, with subjects lying in a comfortable supine position without any visual and auditory stimulation. MFV at rest was obtained by the continuous recording of a 1-min period of normal room air breathing. After a breath-holding period of 30 s, the MFV over a 4-s interval was recorded. The efficacy of breath-holding was checked by means of a respiratory activity monitor (Normocapoxy, Datex, Italy). Mean blood pressure (MBP) and heart rate (HR) were continuously monitored by means of a blood pressure monitor (2300 Finapress, Ohmeda, CO, U.S.A.).
Evaluation of vascular reactivity was made during the basal condition, immediately after smoking a filtered cigarette (containing 1 mg of nicotine) for 5 min with a 5-s puff every 20 s, and then at 10, 20, and 30 min after smoking. Between baseline evaluation and smoking, no recording was made for a 5-min interval to ensure return of end-tidal CO2 values to the baseline condition (range 38–42 mm Hg). Twenty-four healthy nonsmoking subjects, matched for age and sex, comprised the control group. In these subjects, evaluation of vascular reactivity was carried out five times at 10-min intervals. All subjects involved in the study were right-handed. Handedness was established by means of the Edimburgh Inventory (Oldfield, 1971).
Data for analysis consisted of the five values of BHI calculated at 10-min intervals. Statistical analysis was performed by means of a two-way analysis of variance (ANOVA), with condition (i.e., smoking versus nonsmoking) as the “between” factor, and time (i.e., baseline, times-1, −2, −3, and −4) as the “within” factor. In smokers, time-1 corresponds to the immediate postsmoking period. The same analysis was performed on MFV, MBP, and HR recorded during the 1-min period preceding breath-holding.
The MFV, MBP, HR, and BHI values at baseline in smokers and controls were compared using a one-way ANOVA, with group (i.e., smokers and controls) as the between-subject factor.
RESULTS
Basal values of MFV, MBP, HR, and BHI were comparable in smokers and controls (Table 1). Regarding the BHI, the condition effect was significant (F = 14.44, p < 0.001 with 1 and 46 df) because, considering all five values, the mean BHI value in smokers was significantly lower than in controls (1.29 versus 1.50, respectively). The time effect was significant (F = 11.61, p < 0.0001 with 4 and 184 df). In fact, considering both conditions, the BHI significantly changed, passing from the baseline (1.47) to time-1 (1.31) to time-2 (1.36) to time-3 (1.35). Finally, the condition × time interaction was significant (F = 13.0, p < 0.0001 with 4 and 184 df). While, as shown in Table 1 and Fig. 1, the values of BHI in nonsmokers remained substantially unchanged, the BHI values in smokers after smoking initially decreased (time 1, time-2, and time-3) and then returned to values similar to those recorded during basal conditions (time-4). This observation was confirmed by a post-hoc analysis (Sheffe's test) performed to compare variations in BHI at different times in both smokers and controls. In smokers, a significant difference in BHI with respect to baseline was detected immediately after smoking (time 1: p < 0.0001), at 10 min (time-2: p < 0.001), and at 20 min (time-3: p < 0.0001) after smoking. No difference with baseline BHI was detected at time-4. On the contrary, in the control group, no comparisons were significant.
Mean values of the hemodinamic variables recorded at 10-min intervals in smoking and nonsmoking subjects. In smokers time I corresponds to the immediately after smoking period
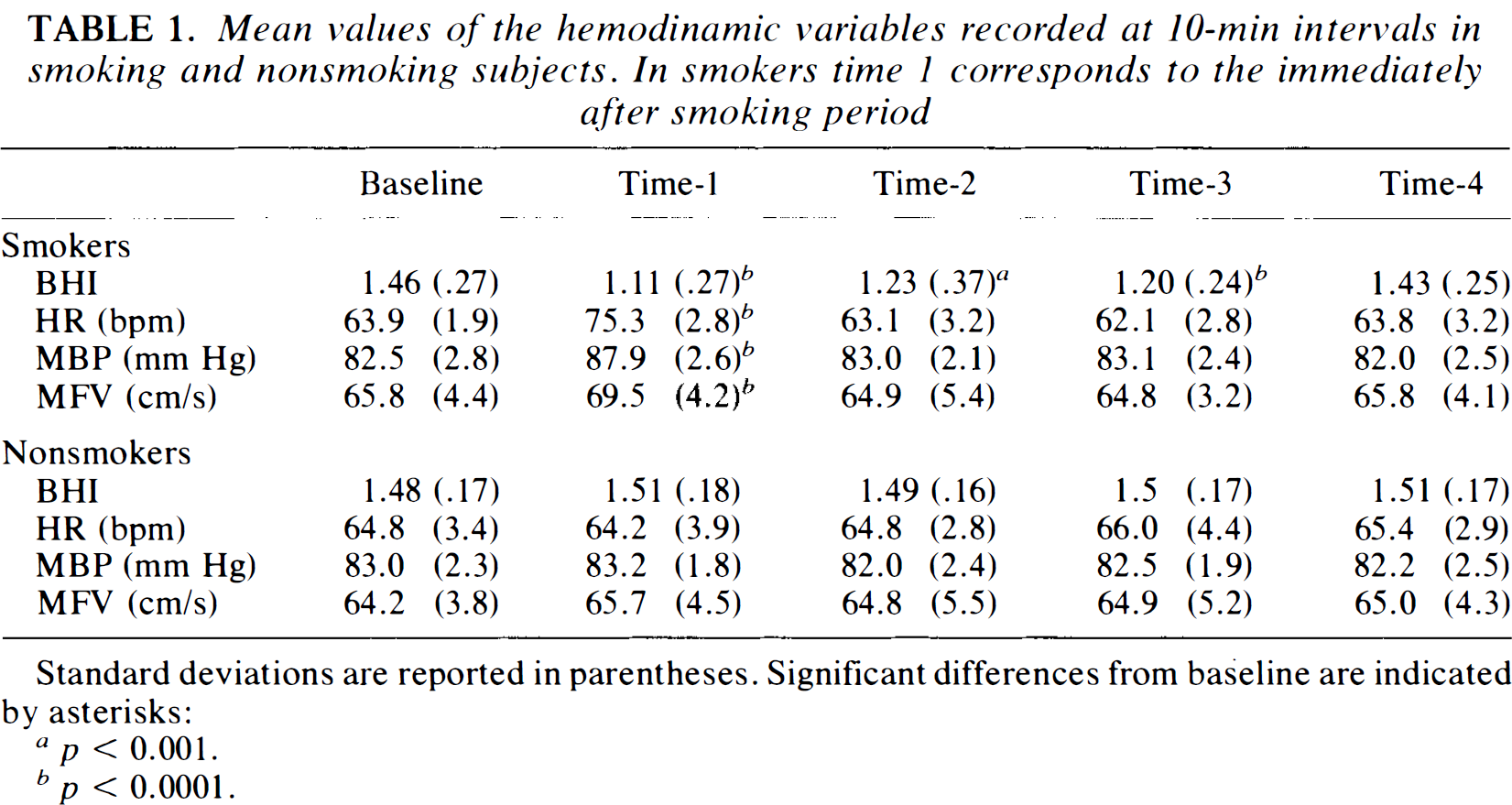
Standard deviations are reported in parentheses. Significant differences from baseline are indicated by asterisks:
p < 0.001.
p < 0.0001.
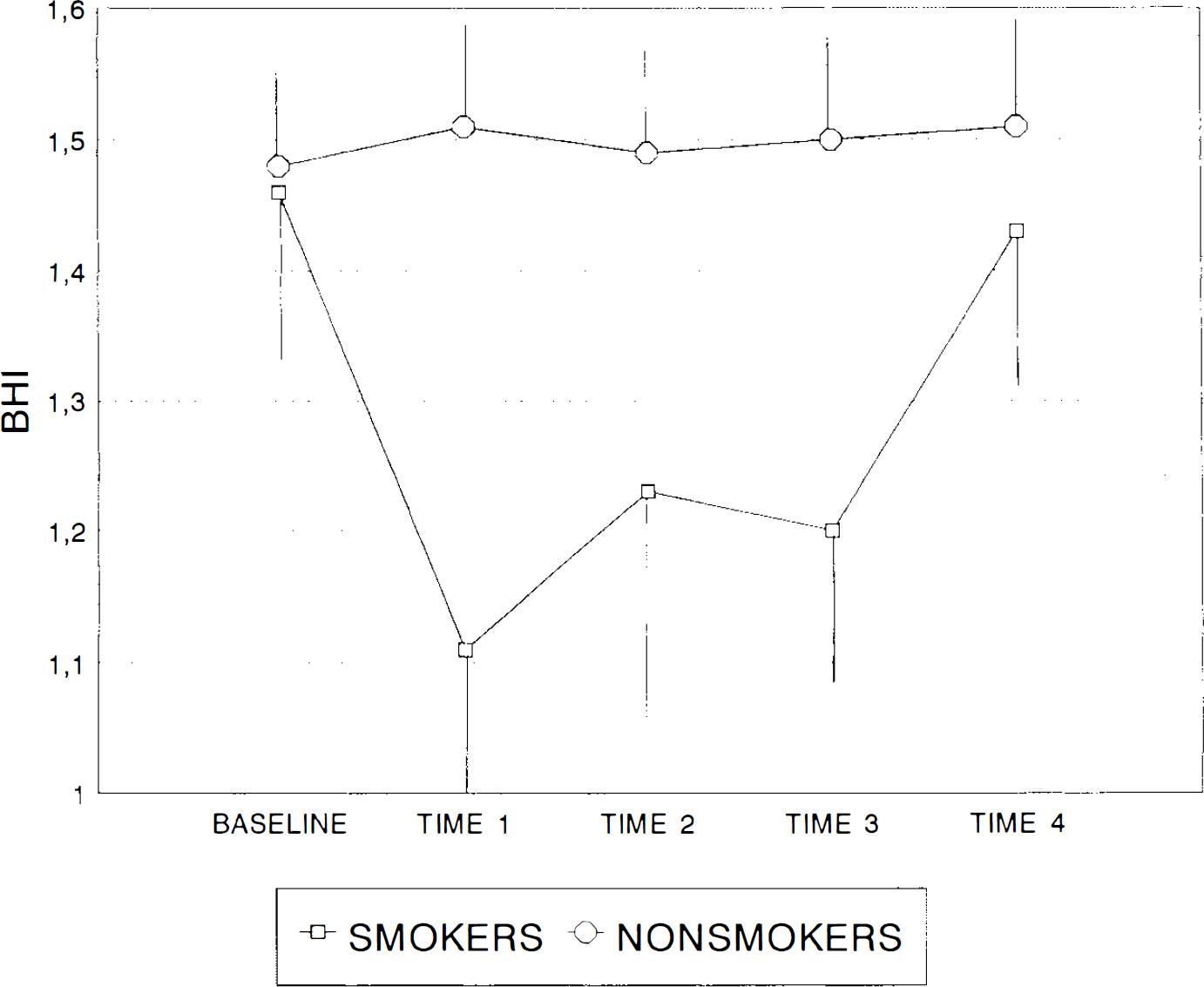
Mean values of BHI in basal conditions and at times-1, −2, −3, and −4 in smoking and nonsmoking subjects. In smokers, time-1 corresponds to the immediately-after smoking period. In smokers, values of BHI are significantly lower at times-1, −2, and −3 with respect to the basal condition. In nonsmokers, BHI remained stable throughout the period of the study. Error bars represent standard deviations.
Regarding the other variables considered, smoking produced very short-term changes in MFV, HR, and MBP (Table 1). In particular, while the three variables remained stable in controls in each of the different evaluations, in smokers, there was a significant increase at time-1 (immediately after smoking) in all three variables, with a subsequent return to values similar to those of basal conditions (times −2, −3, and −4). The post-hoc Sheffe's test performed to compare variations in MFV, HR, and MBP with respect to baseline values showed a significant difference in smokers only at time-1 (MFV: p < 0.0001; HR: p < 0.0001; MBP: p < 0.0001). All other comparisons in both smokers and nonsmokers were insignificant.
DISCUSSION
Cigarette smoking affects cerebral circulation negatively and increases the risk of cerebrovascular events (Molgaard et al., 1986; Colditz et al., 1988; Wolf et al., 1988; Cruickshank et al., 1989). Some studies have postulated that the increased risk of stroke in smokers is associated with accelerated atherosclerosis of cervical and cerebral vessels (Rogers et al., 1984; Whisnant et al., 1990). In this respect, duration of cigarette smoking has been found to be a strong predictor of both carotid artery atherosclerosis (Whisnant et al., 1990) and cardiovascular mortality (Kannel and Higgins, 1990). However, the effect of smoking on cardiovascular disease seems to be reversible and, while all studies have demonstrated dose dependency for smoking and vascular risk, the risk does not appear obviously related to the duration of the smoking habit (Benowitz, 1988). The above findings, along with the fact that cigarette smoking can be considered an important and independent risk factor for all subtypes of cerebral infarction (not only atherosclerotic infarction) in young adults (Love et al., 1990), suggest that other effects on cerebral vasculature, besides atherosclerosis, may be implied in the increased occurrence of stroke in smokers.
In the present study, we investigated the short-term effects of cigarette smoking on cerebral hemodynamic and other cardiovascular variables. Similar to previously reported results (Cruickshank et al., 1989), we found that smoking one cigarette causes an increase in HR, systemic blood pressure, and flow velocity in the MCA. Even if short-lasting and fully reversible within 10 min after smoking, these effects deserve consideration. In particular, the increase of MFV, probably sustained by vasoconstriction of the insonated vessel (Cruickshank et al., 1989; Dorrance and Dwyer, 1989), could result in a decrease of cerebral blood flow. The most relevant finding of our study is the reduced cerebrovascular reactivity to hypercapnia after smoking. This effect was found immediately after and at least 20 min after smoking. In this study, we used a simple method, not requiring CO2 administration, to produce hypercapnia. While the advantages of this kind of approach are evident, there is insufficient evidence in the literature for it to be considered reliable and reproducible enough for studying vascular reactivity. However, we believe that the constant values of the BHI found in the normal volunteers for each of the different evaluations and the fact that, in our study population, index values returned to baseline 30 min after smoking constitutes validation of this method. For the same reasons, we can also exclude the idea that reduction of vascular reactivity after smoking is due to chance. Cerebrovascular reactivity to hypercapnia is considered an index of the capability of cerebral vessels to respond to the metabolic request of the brain. Reduction of this property could be interpreted as increased susceptibility to ischemic problems (Powers, 1991). One possible explanation of the reduced cerebrovascular reactivity may be that smoking decreases vascular resistance in cerebral arteries. In this respect, Kodaira et al. (1993) found that the increased MFV after smoking is associated with a reduction of the pulsatility index, so suggesting a dilatation of the small resistance vessels. However, other studies, in which the effects of smoking on cerebral hemodynamics were investigated using TCD and the 133Xe inhalation technique, did not confirm this effect (Cruickshank et al., 1989; Dorrance and Dwyer, 1989). In fact, the increased flow velocity in the cerebral arteries after smoking was associated with a reduction of cerebral blood flow. This finding suggests that smoking induces vasoconstriction in the large basal cerebral arteries. The hypothesis that smoking takes effect at the arterial level is also supported by a recent study on the short-term effects of smoking on arterial wall properties, showing that arterial wall stiffness increases after smoking one cigarette (Kool et al., 1993). All these effects on cerebral arteries could account for our finding of a reduction of vascular responsiveness to hypercapnia as well as for the increased risk of cerebrovascular disease related to current smoking. Moreover, the potential importance of short-term changes induced by smoking in the pathophysiology of cerebrovascular problems is supported by studies showing neither functional nor structural differences in vessel wall properties between smokers and nonsmokers (Jacobs et al., 1991; Gasser, 1991). In the present study, we did not find any differences between controls and smokers in MFV, HR, MBP, and cerebrovascular reactivity to hypercapnia in the basal condition. These observations further strengthen the above-mentioned hypothesis, even if one cannot exclude that hemodynamic changes in basal conditions could not yet be detected in the group of young smokers in the present study.
In conclusion, results of our study suggest that smoking can cause acute negative effects on cerebral hemodynamics. These effects could contribute, at least in part, to the increased risk of cerebral infarction in current smokers. Further studies should be performed to investigate other factors, such as the cumulative effect of repeated cigarette smoking, on cerebral circulation.
