Abstract
The purpose of this study was to identify the trigger mechanism activating the 5-lipoxygenase pathway during blood–brain cell contact and to estimate the contribution of blood and brain cells to the cysteinyl–leukotriene (LT) biosynthesis observed under these conditions. Incubation of dissociated rat brain cells in Krebs-Henseleit solution for up to 60 min did not stimulate any detectable cysteinyl–LT biosynthesis. Incubation of recalcified rat whole blood in vitro for up to 60 min led to release of only small amounts of cysteinyl–LT into the serum samples. However, coincubation of dissociated rat brain cells with physiologically recalcified autologous whole blood triggered a time-dependent release of large amounts of immunoreactive cysteinyl–LT into the serum samples. By reverse-phase HPLC, immunoreactive cysteinyl–LT was identified as a mixture of LTC4, LTD4, and LTE4. The extent of the 5-lipoxygenase stimulation depended on the amount of autologous blood coincubated with the dissociated brain cells. Activation of the 5-lipoxygenase pathway also occurred with coincubation of dissociated rat brain cells with recalcified autologous plasma. Stimulation of cysteinyl–LT biosynthesis during blood–brain cell contact remained unaffected by aprotinin, but concentration-dependent inhibition by the structurally and functionally unrelated thrombin inhibitors D-Phe-Pro-Arg-CH2Cl and recombinant hirudin was seen. Finally, when dissociated rat brain cells were incubated in Krebs-Henseleit solution in the presence of human α-thrombin, a concentration-dependent release of cysteinyl–LT into the buffer samples was observed. These data demonstrate that, in rats, during blood–brain cell contact, stimulation of the 5-lipoxygenase pathway in brain cells proceeds via α-thrombin as effector molecule.
Cysteinyl–leukotrienes (LTs) are generated via the 5-lipoxygenase pathway of arachidonic acid metabolism. In central nervous tissue their biosynthesis was first demonstrated in vitro in rat brain slices stimulated with the ionophore A23187 (Dembinska-Kiec et al., 1984; Lindgren et al., 1984). In addition, biosynthesis of cysteinyl–LT has been reported in central nervous tissue from various other species including gerbils (Moskowitz et al., 1984; Kiwak et al., 1985; Simmet et al., 1987, 1988b), guinea pigs (Shimizu et al., 1987), and humans (Simmet et al., 1988a). It has been suggested that cysteinyl–LT might play a role in neuroendocrine mechanisms (Hulting et al., 1985; Kiesel et al., 1991) and might act as a messenger or modulator of central nervous activity (Palmer et al., 1981; Schweitzer et al., 1990). On the other hand, several pathophysiological conditions, such as epileptic seizures (Simmet et al., 1988b; Simmet and Tippler 1990, 1991), ischemic insult (Kiwak et al., 1985; Simmet et al., 1987; Minamisawa et al., 1988; Ohtsuki et al., 1995), concussive injury (Kiwak et al., 1985), and subarachnoid hemorrhage (SAH) (Kiwak et al., 1985; Yokota et al., 1987; Paoletti et al., 1988; Shimizu et al., 1988; Watanabe et al., 1988; Gaetani et al., 1992), have been shown to trigger cerebral cysteinyl–L formation in vivo. With this information as background, it has been suggested that cysteinyl–LT, known to be a potent smooth muscle constrictors in peripheral tissues, might contribute to cerebral vasospasm during SAH (Kiwak et al., 1985; Yokota et al., 1987; Shimizu et al., 1988; Watanabe et al., 1988). In addition, it has been reported that patients with SAH having vasospasms had significantly higher CSF levels of cysteinyl–LT than did patients without symptomatic vasospasm (Paoletti et al., 1988; Rodriguez y Baena et al., 1987, 1988). This finding further supports the notion that cysteinyl–LT might play a pathophysiological role during SAH. So far, however, very little is known about the stimuli involved in the activation of the cerebral 5-lipoxygenase pathway under these pathophysiological conditions. We therefore sought to identify the stimulus triggering activation of cysteinyl–LT biosynthesis during blood–brain cell contact as it might occur during SAH. Moreover, we wanted to clarify whether cysteinyl–LT generated during blood–brain cell contact is primarily generated by cerebral tissues or by blood cells such as polymorphonuclear neutrophils. Similar to the experience of other authors (Gebicke-Haerter et al., 1991), in our hands astroglial cells kept under cell culture conditions lost their capability for cysteinyl–LT biosynthesis rather rapidly. Therefore, we decided to study the blood–brain cell interaction in vitro using freshly dissociated rat brain cells (Tildon and Roeder, 1988) coincubated with physiologically recalcified autologous whole blood.
METHODS
Animal preparation
Male Wistar rats weighing 400–600 g were briefly anesthetized with diethyl ether. The peritoneal cavity was opened and up to 5 ml of blood was collected from the abdominal aorta into 3.8% trisodium citrate (final concentration, 0.38%). The descending aorta was clamped, and a cannula was introduced into the ascending aorta via the left ventricle. The right ventricle was opened and brain tissue was flushed blood-free with 30 ml of ice-cold physiological saline. After decapitation, brain tissue was removed from the skull and immediately transferred into ice-cold saline.
Cell preparation
Dissociated brain cells were prepared basically as described by Tildon and Roeder (1988). In brief, cerebellum, brain stem, and meninges were removed. Remaining tissue was weighed, moistened with ice-cold 0.9% NaCl, and minced on the precooled stage of a tissue chopper into 1-mm3 tissue pieces. The minced tissue was suspended in 30 ml of ice-cold 0.9% NaCl in a chilled 50-ml conical polypropylene centrifuge tube. This volume of 0.9% NaCl led to a yield of protein in the final cell suspension of 4–7 mg/ml as determined by the method of Bradford (1976) using bovine serum albumin as standard. The tissue was triturated until the suspension moved easily through the orifice of a 10-ml pipette. After vigorous vortexing for 1 min, the tissue suspension was filtered through nylon cloth (Nitex; pore size, 355 μm) and filtered again through a finer sieve (Nitex; pore size, 75 μm). The cells were washed twice in 50 ml of Krebs-Henseleit solution at 4°C and centrifuged at 355 g for 10 min. The cells were resuspended in 50 ml of Krebs–Henseleit solution and preincubated at 37°C for 30 min. After centrifugation the cell pellet was resuspended in ∼8 ml of Krebs–Henseleit solution and adjusted to a protein concentration of 5 mg/ml; this cell suspension was used for the experiments. As described by Tildon and Roeder (1988), viability of the cells was >80% as judged by trypan blue dye exclusion.
Recalcification of whole blood or plasma
Plasma was prepared by centrifugation of whole blood at 4°C and 2,500 g for 20 min. Until used, blood and plasma were stored in polypropylene tubes at 4°C. On incubation, whole blood or plasma was recalcified with 0.3 M CaCl2 solution, resulting in a final concentration of 1.1 mM of free Ca2+ ions as determined by a Ca2+-sensitive electrode (Orion Research, Cambridge, MA, U.S.A.) (Weide et al., 1994).
Experimental protocol
All incubations were performed in polypropylene tubes. Generally, 1 ml of brain cells adjusted to a protein concentration of 5 mg/ml in Krebs–Henseleit solution equilibrated with carbogen (O2/CO2, 95/5%) was coincubated for up to 60 min with either 0.5 ml of Krebs–Henseleit solution, autologous plasma, or blood. In experiments with plasma or blood, the material was physiologically recalcified as already described.
In experiments with varying amounts of blood, the brain cells (5 mg in terms of protein) were suspended in 0.5 ml of Krebs–Henseleit solution; Krebs–Henseleit solution was added to give a final incubation volume of 1.5 ml. When aprotinin, D-Phe-Pro-Arg-CH2Cl (PPACK), or the recombinant hirudin HBW 023 [(Leu1, Thr2)-63-desulphohirudin; Behringwerke AG/Hoechst AG] was used, the relevant compound was dissolved in 0.9% NaCl and added in a volume of 15 μl. Controls received the appropriate amounts of 0.9% NaCl.
Analytical procedure
Prior to radioimmunological determination of cysteinyl–LT, proteins contained in the samples were precipitated by addition of three volumes of ice-cold acetone. After storage at −20°C for 40 min, proteins were pelleted by centrifugation at 3000 g and 4°C for 20 min. After evaporation of the supernatants, the residues were resuspended in 10 mM of Tris–HCl, pH 7.4, for radioimmunological quantitation of cysteinyl–LT. Cysteinyl-LT were calculated in terms of LTC4-Hke material. All standard curves were run through the same procedure as the samples; therefore, the values presented are corrected for potential losses during the extraction procedure. The detection limit of the radioimmunoassay is 5.9 fmol per tube for LTC4. The antiplasma used recognizes mainly LTC4 but exhibits 78 and 49% relative cross-reaction with LTD4 and LTE4, respectively (Simmet and Tippler, 1990; Simmet et al., 1990).
For characterization of cysteinyl–LT by reverse-phase HPLC, the material was extracted by solid-phase extraction using C18–SEP PAK as previously described (Simmet et al., 1988a, 1988b, 1990). The residue was redissolved in 30% methanol (vol/vol) and injected onto a C18-Nucleosil column (250 × 4 mm; particle size, 5 μm; Macherey Nagel, Düren, Germany) protected by a pre-column using the solvent system methanol:water:acetic acid (65:35:0.1; vol/vol/vol; pH 5.6) containing ethylenediaminetetraacetic acid disodium (EDTA), 1 mM. UV absorbance was scanned at 280 nm, and all samples contained PGB2 as an internal standard. Eluted fractions collected in 1-min intervals were evaporated, resuspended, and finally tested in the radioimmunoassay for cysteinyl–LT (Simmet et al., 1988a, 1988b, 1990).
Statistics
Means ± SD were calculated and statistical analysis was performed by analysis of variance and the Newman–Keuls test. Differences were considered significant at p < 0.05.
RESULTS
Cysteinyl-LT generation in vitro
Dissociated rat brain cells equivalent to 5 mg of protein and incubated in a final volume of 1.5 ml of Krebs–Henseleit solution saturated with carbogen at 37°C for up to 60 min did not release detectable amounts of immunoreactive cysteinyl–LT (Fig. 1).
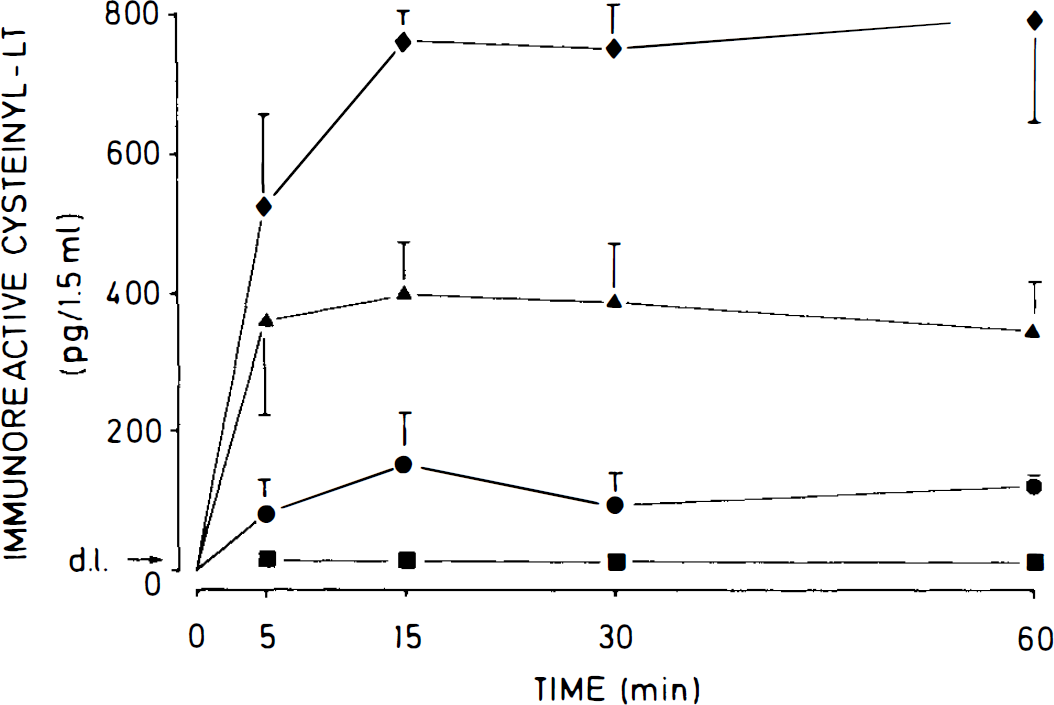
Time course of the release of immunoreactive cysteinyl–LT from 0.5 ml of recalcified whole rat blood coincubated with 1 ml of Krebs–Henseleit solution (circles) and from rat brain cell suspensions (1 ml) coincubated either with 0.5 ml of Krebs–Henseleit solution (squares), 0.5 ml of recalcified autologous whole blood (diamonds), or 0.5 ml of recalcified autologous plasma (triangles). The values are means ± SD of four independent experiments in each group; d.l., detection limit.
Incubation of a mixture of 0.5 ml of physiologically recalcified rat whole blood with 1 ml of Krebs–Henseleit solution led to a time-dependent release of small amounts of cysteinyl–LT; a maximum of 155 ± 74 pg/1.5 ml was reached at 15 min, and the amount decreased slightly to 125 ± 12 pg/1.5 ml at 60 min (n = 4 each; Fig. 1).
However, when 0.5 ml of physiologically recalcified rat whole blood was coincubated with dissociated autologous brain cells equivalent to 5 mg of protein in 1 ml of Krebs–Henseleit solution, a time-dependent release of large amounts of immunoreactive cysteinyl–LT was observed. Release of cysteinyl–LT reached a plateau phase at 15 min of 762 ± 44 pg/1.5 ml, which did not significantly change at 60 min at 795 ± 150 pg/1.5 ml (n = 4 each; Fig. 1).
Coincubation of 0.5 ml of physiologically recalcified rat plasma with 1 ml of dissociated autologous rat brain cells (5 mg of protein) similarly led to a time-dependent release of cysteinyl–LT, although to a diminished extent (Fig. 1). A plateau phase was reached at 15 min of 399 ± 76 pg/1.5 ml (n = 4), and a slight decrease of immunoreactive material was observed after 60 min of incubation (349 ± 74 pg/1.5 ml; n = 4; Fig. 1).
Characterization of immunoreactive cysteinyl–LT by reverse-phase HPLC
Because the anti-cysteinyl–LT antiplasma used cross-reacts with LTC4, LTD4, and LTE4 to a considerable extent, immunoreactive cysteinyl–LT were further characterized by reverse-phase HPLC. As shown in Fig. 2A, coincubation of recalcified rat whole blood with dissociated autologous brain cells for 15 min led to generation of immunoreactive cysteinyl–LT, which coeluted mainly with the retention times of synthetic LTC4 and LTE4. Smaller amounts of immunoreactivity comigrated with the retention time of LTD4.
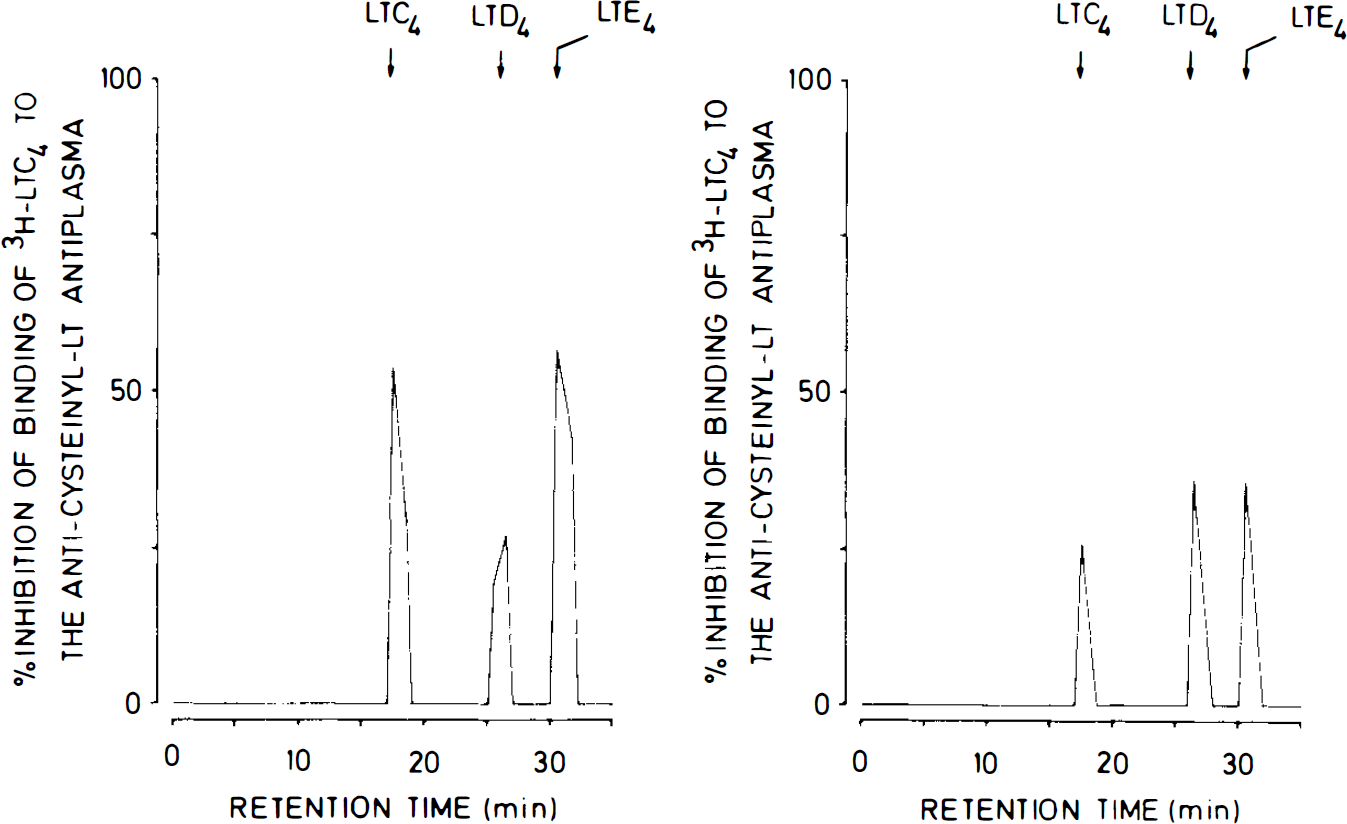
Reverse-phase HPLC profiles of immunoreactive cysteinyl–LT from rat brain cell suspensions coincubated with recalcified autologous whole blood
When dissociated rat brain cells were coincubated with recalcified autologous plasma for 15 min, the largest portions of immunoreactivity consisted of LTD4 and LTE4, but only a slightly smaller amount of immunoreactive material comigrated with LTC4 (Fig. 2B).
Dependence of cysteinyl–LT formation on the relative concentration of blood
Since the data obtained indicated that blood contains a trigger mechanism activating the 5-lipoxygenase pathway during blood–brain cell contact in brain cells, we tested the effects of various amounts of recalcified autologous blood on cysteinyl–LT generation. As Fig. 3 demonstrates, incubation of dissociated rat brain cells (5 mg in terms of protein) with increasing amounts of recalcified autologous blood for 15 min led to a concentration-dependent increase in cysteinyl–LT release, which released 882 ± 157 pg/1.5 ml (n = 3) when brain cells in a volume of 0.5 ml were coincubated with 1 ml of recalcified blood.
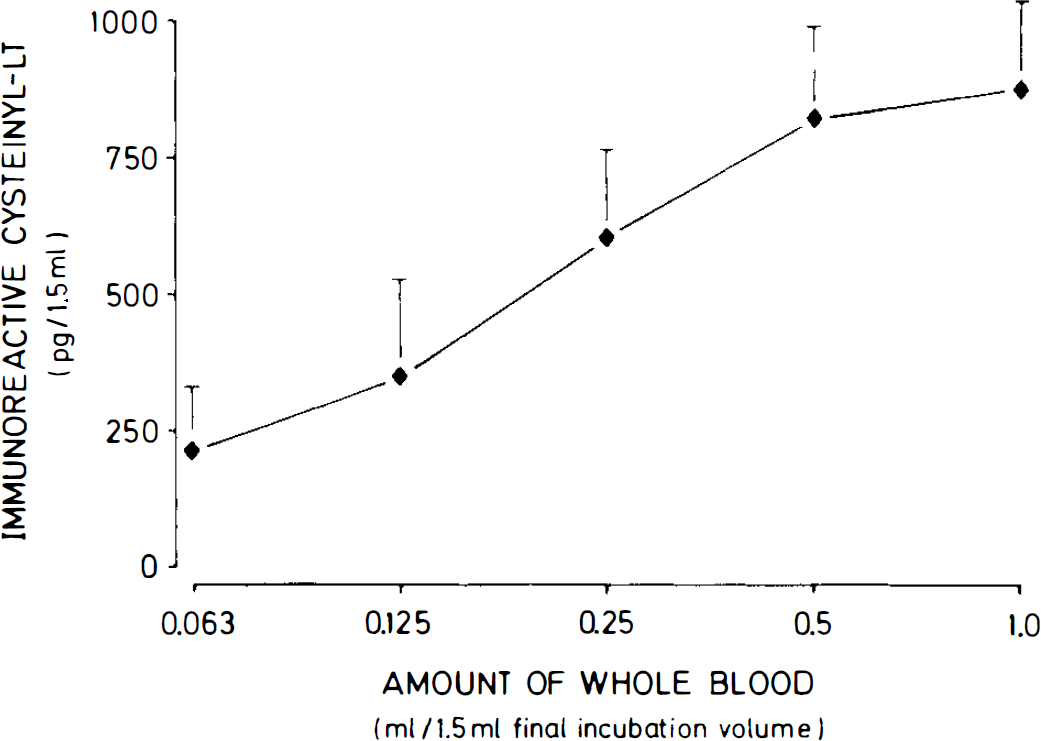
Effects of various amounts of recalcified autologous whole blood coincubated with dissociated rat brain cells for 15 min on the release of immunoreactive cysteinyl–LT. Dissociated brain cells were added in a constant volume of 0.5 ml, and recalcified whole blood was added in the volumes indicated. The 1.5-ml final volume was compensated for with an appropriate amount of Krebs–Henseleit solution. The values are mean ± SD of three independent experiments.
Effects of compounds inhibiting serine proteases on the release of immunoreactive cysteinyl–LT
Because previous studies had shown that plasmin generated in the course of blood coagulation might stimulate the 5-lipoxygenase pathway in human monocytes (Weide et al., 1994), we tested the potential effects of aprotinin, a potent inhibitor of plasmin (Stephens et al., 1989). However, this compound, at amounts from 1 to 1000 KIU/ml, had no effect on cysteinyl–LT formation during blood–brain cell contact (data not shown), precluding participation of plasmin or other aprotinin-sensitive proteases in the trigger mechanism involved.
Stimulation of the 5-lipoxygenase pathway by clotting plasma clearly implicates a link to the coagulation process. In further experiments, we therefore tested the effects of compounds inhibiting the activity of thrombin. As shown in Fig. 4A, the chloromethylketone PPACK, as a highly specific thrombin inhibitor, inhibited the cysteinyl–LT release in a concentration-dependent manner when 0.5 ml of recalcified whole blood was coincubated with 1 ml of autologous dissociated rat brain cells (5 mg of protein) for 15 min. Compared with controls, PPACK (0.3 μM) significantly reduced immunoreactive cysteinyl–LT by 41.1 ± 15.1% (n = 5; p < 0.01), while PPACK (10 μM) led to an inhibition of 75.0 ± 7.9% (n = 5; p < 0.01; Fig. 4A).
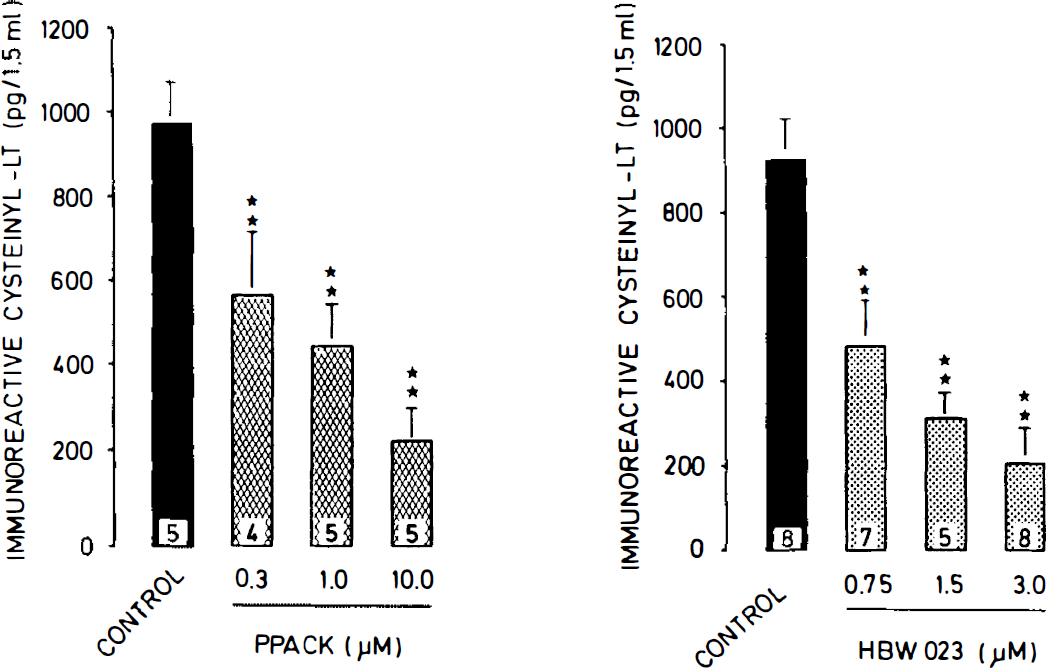
Concentration-dependent effects of the thrombin inhibitors PPACK
Additional experiments with the highly specific thrombin inhibitor recombinant hirudin (HBW 023) revealed similar concentration-dependent inhibitory effects as seen with PPACK (Fig. 4B). Compared with controls, HBW 023 (0.75 μM) reduced cysteinyl–LT release by 47.8 ± 11.9% (n = 7; p < 0.01) when 0.5 ml of recalcified whole blood was coincubated with 1 ml of autologous dissociated brain cells (5 mg of protein) for 15 min. At 3.0 μM, HBW 023 cysteinyl–LT release was reduced by 77.9 ± 9.3% (n = 9; p < 0.01; Fig. 4B).
Both PPACK and HBW 023 at the highest concentrations used, i.e., 10 and 3 μM, respectively, had no effect on the cysteinyl–LT formation triggered by stimulation of dissociated brain cells with the Ca2+ ionophore A23187 (10 μM; data not shown).
Effects of human α-thrombin on cysteinyl–LT formation by dissociated rat brain cells
Since the experiments with the thrombin inhibitors suggested involvement of thrombin in the trigger mechanism activated during blood–brain cell contact, we investigated the effects of various concentrations of human α-thrombin on cysteinyl–LT release from dissociated rat brain cells (5 mg of protein) incubated in 1.5 ml of Krebs–Henseleit solution at 37°C for 15 min. Human α-thrombin triggered a concentration-dependent stimulation of the 5-lipoxygenase pathway in dissociated rat brain cells (Fig. 5). A maximum cysteinyl–LT release of 912 ± 89 pg/1.5 ml was observed at an α-thrombin concentration of 3 U/ml (n = 5). At 6 U/ml of α-thrombin, cysteinyl–LT release decreased to 719 ± 233 pg/ml (n = 5; Fig. 5).
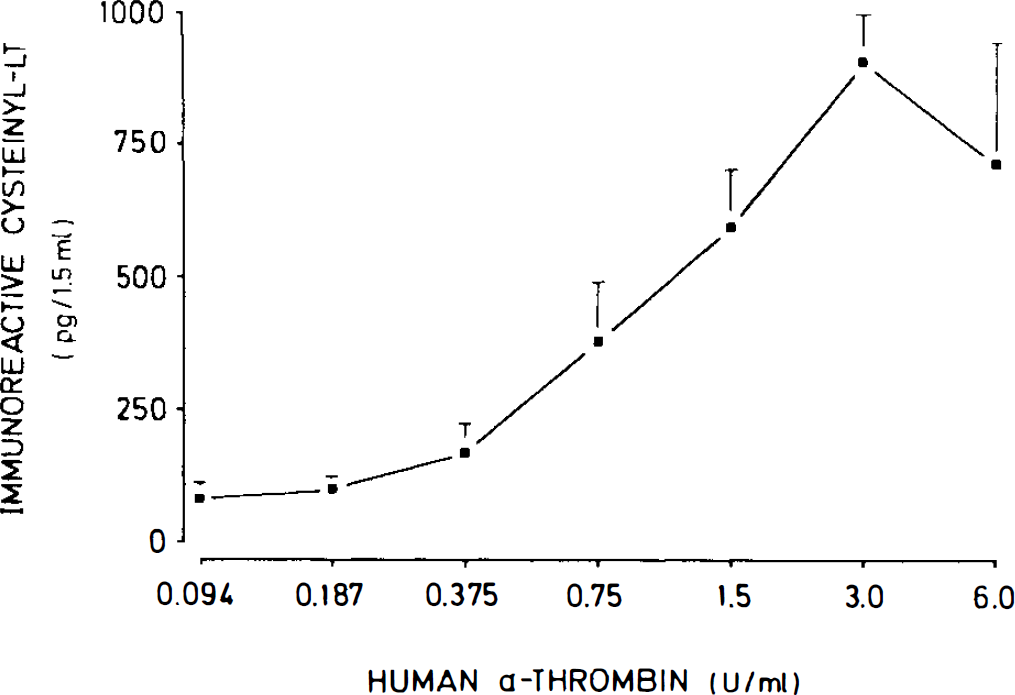
Concentration-dependent effects of human α-thrombin on immunoreactive cysteinyl–LT release from dissociated rat brain cells incubated in Krebs–Henseleit solution at 37°C for 15 min. The values are means ± SD of five experiments.
DISCUSSION
Cerebral vasospasm following SAH due to rupture of an intracranial aneurysm may trigger severe cerebral ischemia and infarction. Despite intensive research, the pathogenesis of arterial vasospasm and neuronal damage is poorly understood. Substantial evidence suggests it to be multifactorial, and numerous compounds in circulating blood have been implicated in the induction of early and late vasospasm following SAH (Findlay et al., 1991). Among the potential vasoconstrictors, hemoglobin, endothelin, and the 5-lipoxygenase-derived cysteinyl–LT have gained considerable interest. Elevated levels of cysteinyl–LT in brain tissue and CSF have been described in association with SAH in a variety of species, including humans (Rodriguez y Baena et al., 1987).
Cysteinyl and LT are vasospastic agents in peripheral tissues, but the results regarding their activity on cerebral tissues have not been unequivocal (Simmet and Peskar, 1990). Nevertheless, the majority of studies suggest a vasoconstrictor activity of cysteinyl–LT in cerebral blood vessels (Simmet and Peskar, 1990).
Since cysteinyl–LT supposedly acts as a mediator of vasospasm following SAH, it is obviously important to gain some insight into the activation mechanism involved in the 5-lipoxygenase stimulation under those circumstances. To the best of our knowledge, a specific trigger mechanism leading to activation of cysteinyl–LT biosynthesis during blood–brain cell contact has not yet been identified. To investigate the mechanism activating the 5-lipoxygenase pathway during SAH, we reduced the complex in vivo models of SAH to a simple blood–brain cell contact model in vitro. This approach permits pharmacological modulation of processes potentially involved in 5-lipoxygenase activation.
Our results clearly demonstrate that coincubation of physiologically recalcified whole blood with autologous dissociated rat brain cells triggers a time-dependent biosynthesis of cysteinyl–LT. In contrast, incubation for up to 60 min of dissociated rat brain cells alone was not accompanied by any detectable release of cysteinyl–LT, while incubation of physiologically recalcified whole blood led to a time-dependent generation of only small amounts of cysteinyl–LT. These in vitro findings are in line with previous data showing that cysteinyl–LT levels in clotted gerbil blood or ex vivo brain tissue were rather low, while intracerebroventricular injection of freshly clotted blood led to a large increase in cerebral cysteinyl–LT contents (Kiwak et al., 1985). Moreover, in experiments ex vivo with rat brain tissue slices incubated in vitro subsequent to intracisternal injection of autologous blood, increased biosynthesis of cysteinyl–LT was also reported (Rodriguez y Baena et al., 1989; Gaetani et al., 1990).
Numerous investigators have used the two-hemorrhage model in dogs (Yokota et al., 1987; Shimizu et al., 1988; Watanabe et al., 1988). In this model, where 5-lipoxygenase activity seems to be induced in basilar blood vessels by the injection of blood, 5-lipoxygenase inhibitors such as AA-861 and l,2-bis(nicotinamido)propane or a cysteinyl–LT antagonist, ONO-1078, were found to have therapeutic effects on the delayed cerebral vasospasm, providing strong evidence for a pathophysiological role of cysteinyl–LT under these conditions (Yokota et al., 1987; Shimizu et al., 1988; Watanabe et al., 1988; Kobayashi et al., 1992).
The data presented here extend the findings from those in vivo models by showing that there exists no absolute requirement for contact of whole blood with brain cells for stimulation of cysteinyl–LT biosynthesis. Instead, physiologically recalcified plasma is apparently sufficient for activation of 5-lipoxygenase activity in dissociated brain cells. This finding clearly shows that, first, the dissociated brain cells probably represent a major source of cysteinyl–LT during short-term blood–brain cell contact. A similar conclusion was drawn from experiments in gerbils subjected to experimental SAH, in which brain cells were suggested to be the major source of cysteinyl–LT (Kiwak et al., 1985). Second, the activation mechanism ought to be present in recalcified plasma. Interestingly, in dogs intracisternal injection of plasma was found to elicit early vasospasm only (Nozaki et al., 1990). The time course of cerebral cysteinyl–LT levels after SAH in gerbils is also in favor of a role of cysteinyl–LT during early vasospasm (Kiwak et al., 1985). An early acute cerebral vasospasm after SAH is known to occur in both humans (Taneda et al., 1990) and rats (Delgado et al., 1985). The rapid generation of cysteinyl–LT, as seen in our in vitro experiments, makes these compounds probable mediators during the acute phase of cerebral vasospasm, as has been suggested for the gerbil model (Kiwak et al., 1985). However, after experimental SAH, rat brain tissue slices were reported to exhibit enhanced cysteinyl–LT formation for up to 2 days after injection (Rodriguez y Baena et al., 1989; Gaetani et al., 1990). Independent from an initial activation of cysteinyl–LT biosynthesis, such prolonged generation of cysteinyl–LT could additionally be due to induction of the 5-lipoxygenase enzyme, as reported for the dog model (Watanabe et al., 1988).
The fact that recalcified plasma stimulates activation of the 5-lipoxygenase pathway led us to assume that the trigger mechanism might be related to activation of the coagulation cascade during plasma-brain cell contact. We have previously shown that the process of blood coagulation can stimulate monocyte 5-lipoxygenase via plasmin (Weide et al., 1994). However, aprotinin, which is known to inhibit plasma even in its membrane-bound form (Stephens et al., 1989), was unable to affect cysteinyl–LT formation during blood–brain cell contact, indicating that plasmin was not involved in the trigger mechanism.
By contrast, when we coincubated whole blood and brain cells in the presence of PPACK, a highly selective chloromethylketone substrate that covalently reacts with the catalytic center of α-thrombin (Kettner and Shaw, 1979; Bode et al., 1989), a significant and concentration-dependent inhibition of cysteinyl–LT formation was observed, suggesting that thrombin might be involved in the activation of the 5-lipoxygenase pathway. Moreover, coincubation of whole blood and brain cells in the presence of recombinant hirudin, a highly specific polypeptide inhibitor of thrombin originally derived from the European medicinal leech Hirudo medicinalis (Rydel et al., 1990), also yielded a concentration-dependent and significant inhibition of cysteinyl–LT formation during blood–brain cell contact. Hirudin is known to form stable complexes with thrombin by binding to the catalytic and anion binding sites of the thrombin molecule (Rydel et al., 1990). Similar to findings in previous experiments with human blood cells (Simmet and Weide, 1991), we did not observe any effects of the thrombin inhibitors on the ionophore A23187-induced cysteinyl–L formation, thereby excluding a nonspecific interaction of those compounds with 5-lipoxygenase activity.
In our last approach to demonstrating the significance of thrombin as a stimulus for the 5-lipoxygenase pathway during blood–brain cell contact, we used human α-thrombin to stimulate the dissociated rat brain cells. Human α-thrombin exhibits a high degree of sequence homology with rat α-thrombin (Banfield and MacGillivray, 1992) and is known to interact with thrombin receptors in rat tissues (Antonaccio et al., 1993). When dissociated rat brain cells were incubated in Krebs–Henseleit solution in the presence of human α-thrombin, a concentration-dependent stimulation of cysteinyl–LT biosynthesis was observed, which reached a maximum at ∼3 U/ml. Indeed, the amounts of cysteinyl–LT released by this concentration of α-thrombin were comparable with those detected after coincubation of recalcified whole blood with dissociated rat brain cells. Beyond this optimal concentration of human α-thrombin, release of cysteinyl–LT tended to decrease again. The reason for the apparently reduced efficacy of thrombin at the highest concentration used is unknown. However, such a concentration-response curve is not uncommon for thrombin, as it has also been observed in a mouse macrophage cell line, in which thrombin exerts mitogenic effects (Bar-Shavit et al., 1986), and in human monocytes, in which thrombin was found to trigger a chemotactic response (Bar-Shavit et al., 1983).
It is well-known that cerebral tissues are a rich source of tissue factor, the primary cellular initiator of blood coagulation (Drake et al., 1989; Nemerson, 1992). In the CNS of the baboon, tissue factor antigen and related procoagulant activity are confined primarily to gray matter parenchyma and to the noncapillary micro vasculature (del Zoppo et al., 1992). In addition, mouse, rat, and human astrocyte cell lines constitutively express tissue factor mRNA as well as functional protein (Eddleston et al., 1993). Therefore, it is reasonable to assume that under our in vitro conditions, tissue factor generated by dissociated brain cells might trigger activation of the coagulation cascade. However, the precise role of tissue factor in our model will have to be elaborated in further experiments, perhaps with monoclonal antitissue factor antibodies.
Thrombin receptor mRNA is broadly distributed across the neuroaxis (Weinstein et al., 1995). Although white matter was found to be negative, patterns of hybridization included neuronal, glial, and ependymal cells. Since prothrombin mRNA was found in codistribution with thrombin receptor mRNA in rat brain, it has been suggested that the thrombin-thrombin receptor cell signaling system might have multiple physiologic, and possibly pathophysiologic, functions in the CNS (Weinstein et al., 1995). From our data, we cannot conclude which cell type is activated by thrombin during blood–brain cell contact. We and others have shown that rat astroglia might serve as a source of cerebral cysteinyl–LT formation (Simmet and Peskar, 1990), and astroglial cells are known to respond to thrombin. Indeed, thrombin can induce expression of c-fos and c-jun mRNA in astroglial cells (Trejo et al., 1992) and the synthesis and secretion of nerve growth factor (Neveu et al., 1993) and endothelial (Ehrenreich et al., 1993), which has also been suggested to play a pathophysiological role during delayed vasospasm (Findlay et al., 1991).
The fact that thrombin is the stimulus involved in activation of the cerebral 5-lipoxygenase pathway during blood–brain cell contact may at least partially explain why dissociated brain cells incubated in the presence of recalcified plasma generate less cysteinyl–LT than do brain cells coincubated with recalcified whole blood. Platelets, which were not present in the plasma preparation but were in the whole blood samples, of course are important for activation of the coagulation cascade. They provide an appropriate “surface membrane” for the efficient assembly of the prothrombinase complexes and thereby enhance the amount of thrombin generated (Tracy et al., 1985). While this might lead to further enhancement of the thrombin-stimulated 5-lipoxygenase pathway, platelets could also contribute to an enhanced cysteinyl–LT biosynthesis by converting the instable intermediate LTA4 released by brain cells to LTC4, as has been observed in coincubation of monocytes with platelets (Weide et al., 1994). By the same mechanism, polymorphonuclear leukocytes could contribute to cysteinyl–LT formation. Only recently, it has been reported that cysteinyl–LT biosynthesis by neutrophils seems to require uptake of exogenous LTA4 (Palmentier et al., 1995).
In conclusion, our data demonstrate that thrombin generated during blood–brain cell contact is the stimulus triggering activation of the 5-lipoxygenase pathway of arachidonic acid metabolism in brain cells. Thrombin-mediated activation of brain cells may be important for our understanding of the pathophysiology of SAH since thrombin stimulates cerebral release of cysteinyl–LT and endothelin (Ehrenreich et al., 1993), compounds purportedly involved in the development of vasospasm. To our knowledge, this is the first report of thrombin-mediated stimulation of the 5-lipoxygenase pathway of arachidonic acid metabolism. The significance of the thrombin-mediated 5-lipoxygenase stimulation will be further investigated in experiments in vivo by the combined intracerebroventricular application of autologous blood and thrombin inhibitors such as hirudin.
Footnotes
Acknowledgment:
Human α-thrombin and recombinant hirudin (HBW 023) were a generous gift from Dr. J Römisch, Behringwerke AG, Marburg, Germany. Aprotinin was kindly provided by the Bayer AG, Wuppertal, Germany. The technical assistance of Mr. M. Riese is gratefully acknowledged. This work was supported by the Deutsche Forschungsgesellschaft.
