Abstract
Stroke is a sudden and rapidly progressing ischemic or hemorrhagic cerebrovascular disease. When stroke damages the brain, the immune system becomes hyperactive, leading to systemic inflammatory response and immunomodulatory disorders, which could significantly impact brain damage, recovery, and prognosis of stroke. Emerging researches suggest that ischemic stroke-induced spleen contraction could activate a peripheral immune response, which may further aggravate brain injury. This review focuses on hemorrhagic strokes including intracerebral hemorrhage (ICH) and subarachnoid hemorrhage (SAH) and discusses the central nervous system-peripheral immune interactions after hemorrhagic stroke induction. First, inflammatory progression after ICH and SAH is investigated. As a part of this review, we summarize the various kinds of inflammatory cell infiltration to aggravate brain injury after blood-brain barrier interruption induced by hemorrhagic stroke. Then, we explore hemorrhagic stroke-induced systemic inflammatory response syndrome (SIRS) and discuss the interactions of CNS and peripheral inflammatory response. In addition, potential targets related to inflammatory response for ICH and SAH are discussed in this review, which may lead to novel therapeutic strategies for hemorrhagic stroke.
Keywords
Introduction
Many developed countries suffer from stroke as the third leading cause of morbidity and mortality, which can be either ischemic or hemorrhagic.1,2 A common type of stroke known as an ischemic stroke is caused by the loss of blood supply to an area of the brain. There are two types of hemorrhagic strokes: intracerebral hemorrhage (ICH) and subarachnoid hemorrhage (SAH). ICH is characterized by bleeding into the brain parenchyma, whereas SAH is characterized by bleeding into the subarachnoid space. 3
Hemorrhagic stroke is one of the most common critical diseases, and its high mortality and high disability rate greatly threaten human health worldwide.4–6 Although the situation has been alleviated with the continuous progress of the medical level in recent years, medical treatment for hemorrhagic stroke remains unsatisfactory.4,7 A series of pathophysiological reactions such as neuroinflammation, neuronal apoptosis, and cerebral edema after hemorrhagic stroke are associated with worse outcomes.8,9 Current studies show that neuroinflammation is closely related to neurological impairment in patients with stroke. 10 Acute brain injury can disrupt the blood-brain barrier (BBB) and increase inflammation, further intensifying brain damage following a hemorrhagic stroke.8,11,12 Therefore, it is urgent to explore the molecular mechanism of neuroinflammation after hemorrhage stroke and carry out a targeted intervention. As a physiological barrier, the BBB protects the central nervous system (CNS) from inflammatory mediators and immune effector cells in the peripheral circulation.13–15 Inflammatory processes caused by stroke, including innate and adaptive immune mechanisms, are triggered by bleeding into the brain.8,16 Stroke affects the CNS and the immune system in complex ways.17,18 They are also considered critical to recovery, as the brain repairs itself via inflammatory processes.19,20
It is increasingly evident that systemic immune changes also occur in peripheral organs and tissues in addition to the inflammatory response in the brain, such as in the spleen, lung, heart, and kidney after stroke.21–26 In addition, dysregulation of gut microbiota impacts SAH and ICH prognosis and inflammatory response.27–29 However, there are few reports on the CNS-peripheral immune interactions after hemorrhagic strokes. In this review, we examine the findings on the crosstalk between CNS and peripheral inflammatory response after ICH and SAH.
Inflammation in pathogenesis and progression of hemorrhagic stroke
ICH
ICH is caused by the rupture of the blood vessel and hematoma formation in the brain. The primary injury caused by ICH is mechanical damage due to the oppression of hematomas, such as intracranial pressure and cerebral hernia. The negative effect following the primary injury is known as secondary brain injury (SBI), including inflammation, brain edema, disruption of the BBB, and other pathological changes. 30 Inflammation cascade reaction is one of the earliest defense responses to brain injury after ICH, which will attack brain tissue and exacerbate brain damage. 8 CNS and peripheral immune cells contribute to inflammation around the hematoma. The complete physiological and pathological processes comprise inflammatory mediator release, cellular component activation, brain tissue breakdown, brain edema, and repair. 31
It is believed that hemoglobin and iron are released from the hematoma in the acute phase of ICH, causing microglial activation and inflammation. In the acute phase of ICH, microglia can be stimulated to an M1 phenotype to aggravate inflammation leading to pro-inflammatory cytokines release (e.g., interleukin-1β [IL-1β] and tumor necrosis factor-α [TNF-α]). These pro-inflammatory cytokines, chemokines, adhesion molecules, matrix metalloproteinases (MMPs), and cell death products break down the BBB and destroy surrounding tissues.5,32,33 With the inflammatory factors’ recruitment, A1 reactive astrocytes differentiation is induced, augmenting M1 polarization and potentiating inflammation. Other immune cells from peripheral blood (macrophages, neutrophils, lymphocytes, natural killer [NK] cells, and so on) are recruited to infiltrate inside hematoma, which will secrete molecular components to cause cytotoxicity and further strengthen inflammation and exacerbate brain injury. 34 Within seven days of ICH ictus, the M1 phenotype of microglia converts to an M2 phenotype, playing the role of hematoma removal, tissue healing, and anti-inflammation. 35 The secretion of anti-inflammatory cytokines (IL-4 and IL-10) can drive T helper (Th) 2 cells and T regulatory (Treg) cell differentiation. M2 polarization is maintained by the secretion of additional anti-inflammatory cytokines by the Treg and Th2 cells, suppressing M1 and Th1 phenotypes. With the reestablishment of the BBB, brain edema will be reduced. The mass effect will be mitigated, which breaks the cycle of inflammation increase and creates an environment to repair the brain injury. During this progress, microglia constantly survey the inflammatory process through the pattern recognition receptors (PRRs), including toll-like receptors (TLRs) and nucleotide-binding oligomerization domain-like receptors (NLRs), which could also regulate the microglia M1 polarization;36,37 meanwhile, IL-4/IL-10 could switch M1/M2 polarization by modulating JAK1/STAT6 pathway,35,38 whereas HDAC inhibition by JAK1/STAT1. 39 Therefore, inflammation is deemed to play an essential role in ICH-induced brain injury (Figure 1).
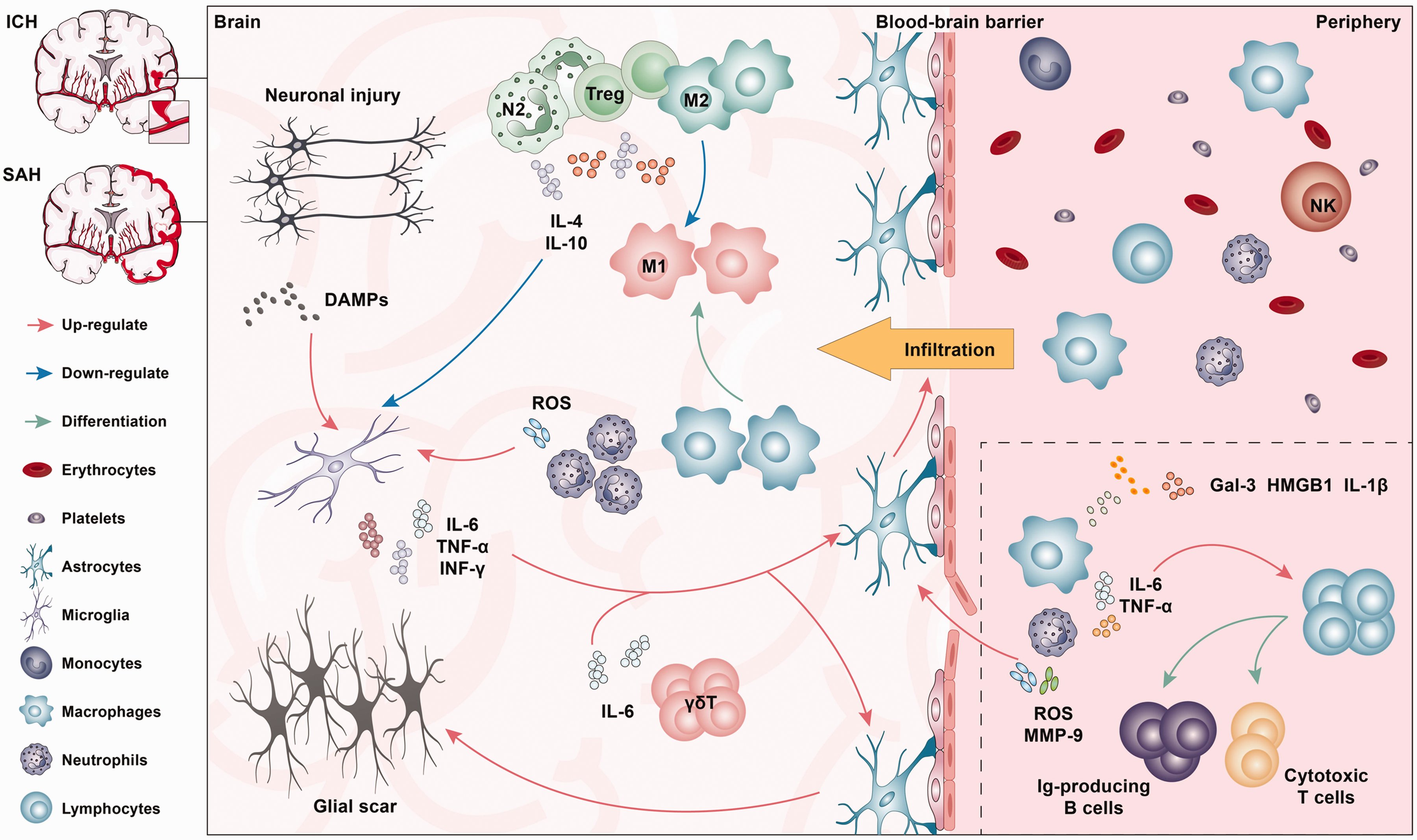
Inflammatory response after hemorrhagic stroke. In the brain parenchyma, neuronal injury secretes damage-associated molecular patterns (DAMPs), which could activate microglia. Microglia secrete pro-inflammatory cytokines and factors, activating astrocytes. Activation and dysfunction of astrocytes contribute to blood-brain barrier (BBB) disruption, causing infiltration of peripheral immune cells. Reactive astrocytes also form a glial scar. However, the “N2” neutrophils, Tregs, and “M2” Macrophages secrete anti-inflammatory cytokines and factors, playing a role in limiting neuroinflammation. In the periphery, Macrophages and Neutrophils secrete IL-6 and TNF-α, promoting the differentiation of lymphocytes into cytotoxic T cells and Ig-producing B cells. ROS and MMP-9 secreted by neutrophils also contribute to the dysfunction of the blood-brain barrier. ICH: intracerebral hemorrhage; SAH: subarachnoid hemorrhage; DAMPs: damage-associated molecular patterns; IL: interleukin; TNF: tumor necrosis factor; INF: interferon; ROS: reactive oxygen species; MMP: matrix metalloproteinases; Ig: immune globulin; Gal: galectin; HMGB: high-mobility group box.
SAH
SAH refers to a series of syndromes caused by blood flowing into the subarachnoid space after the rupture of an intracranial aneurysm. It has been reported that BBB disruption is a key mechanism of brain injury after SAH. 40 Oxidative stress, overproduction of free radicals, hemoglobin, and iron overload can all cause endothelial cell damage and downregulation of tight junctions, such as occludin, zonula occludens-1 (ZO-1), leading to the destruction of BBB, in the acute phase (30 min-3 h) after SAH.41,42 With the BBB disruption, inflammation and infiltration of immune cells further aggravate the damage after SAH.43,44 Microglia and astrocytes, as well as peripheral neutrophils and monocytes/macrophages, play important roles in this process. 45 Microglia, the resident macrophages of the CNS, take a crucial part in the immune response, which can show different phenotypes at different stages after SAH. 33 Similar with ICH, at the early stages of SAH, microglia exhibit pro-inflammatory M1 phenotypes, which are gradually replaced by anti-inflammatory M2 phenotypes at the chronic injury phase. Within 24 h of the hthe basal cistern, M1 phenotypes microglia are activated, which is more prevalent in brain regions adjacent to the location of the blood clot, leading to promote cell death, including necroptosis in neurons. 46 Neuronal necroptosis and necrosis lead to the further release of inflammatory molecules (alarmins) such as HMGB1, 47 which further activate microglia and neuronal loss. At the sub-acute and delayed injury phase after SAH, microglia activation could activate and recruit other inflammatory cells, including resident brain cells and peripheral immune cells. Astrocytes are abundant nerve cells in the brain, which play an essential role in keeping the integrity of the BBB, 48 which can be stimulated to differentiate into the A1 type and induce inflammation, causing brain damage after SAH.49,50 Due to BBB interruption and the chemokines released by local immune cells, a large number of neutrophils enter to the subarachnoid space from the peripheral circulation. Neutrophils transform to a pro-inflammatory phenotype, exacerbating neuroinflammation and leading to brain damage;51,52 Monocytes, which are produced in the bone marrow, infiltrate the bleeding site, transform into macrophages of different forms to participate in the inflammatory response and oxidative stress, and eventually cause brain damage.53–55 During the chronic impairment phase, the M2 phenotype of microglia is participated, exhibiting anti-inflammatory effects 56 (Figure 1). Although the pathological process is different, the signaling pathways related to neuroinflammation after SAH are similar to ICH.
Potential brain-peripheral immune communications in hemorrhagic stroke
Function of immune cells in hemorrhagic stroke: CNS and peripheral
The central immune system is usually thought to be isolated from the peripheral immune system through the BBB and composed of microglia, mast cells, and other glial cells. As introduced above, after a hemorrhagic stroke, some immune cells in the periphery infiltrate CNS with the BBB disruption. Here we describe the changes and activation of immune cells in CNS and peripheral after hemorrhagic stroke in detail.
Microglia and macrophages
As the largest immune cell population in the brain, microglia are essential in the inflammasome response to inflammation in the CNS. 57 Microglia and macrophage infiltrated from the peripheral, as vital immune cells, are involved in the development of neuroinflammation after stroke. As the first immune cell to be activated, microglia are activated by blood components (red blood cells, white blood cells, hemoglobin, Etc.) and undergo phenotypic and functional changes after hemorrhage stroke. 58 Activated microglia can produce a variety of deleterious cytokines, such as IL-1β, IL-6 and TNF-α, which may damage the BBB. Then, monocyte-derived macrophages infiltrate CNS and participate in neuroinflammatory responses with microglia after a hemorrhagic stroke, leading to brain injury aggravating. The prominent roles of activated microglia/macrophages are phagocytosis function, engulfing hematoma and damaged cell fragments, thereby maintaining the balance of the nervous system and promoting the recovery of neurological function.59,60 Activated microglia/macrophages can be polarized into two phenotypes, M1 and M2. M1 polarization state mainly plays a leading role in the brain injury region, aggravating brain injury by producing various pro-inflammatory mediators and chemokines. 61 M2 polarization state is usually interpreted as nerve repair cells, which promote the recovery of nerve function and maintain the balance of the nervous system by secreting anti-inflammatory factors and upregulation of neuroprotective factors. 62 Some studies have proved a mutual transformation relationship between the two phenotypes of microglia and dynamic change with time after stroke. 63 Moreover, microglia also play a critical role in activating other immune cells after hemorrhage stroke, such as astrocytes. 64
Astrocytes
Astrocytes play significant roles in the central nervous system, such as neurotrophic support, synapse-associated repair, and maintenance of the BBB. Astrocytes are one of the most responsive cell types after ICH. 65 When hemorrhage stroke occurs, astrocytes are triggered into a reactive state (reactive astrocytes), which has been suggested to be related to the induction of activated microglia. 66 Activated microglia secrete IL-1α, TNF, and Clq, and induce the increase of A1 reactive astrocytes after stroke, leading to up-regulation of pro-inflammatory cytokines, chemokines and growth factors. A1 reactive astrocytes could mediate abnormal synthesis and release of neurotransmitters, synaptic destruction, and loss of homeostasis in other balance functions, releasing neurotoxins, leading to apoptosis of neurons and surrounding oligodendrocytes. 67 The mechanisms of A1 reactive astrocytes that play a toxic role in hemorrhagic stroke have been elucidated. Hemoglobin from the hematoma is a decisive trigger factor of oxidative stress, which induces MMP 9 release by reactive astrocytes, BBB destruction, and cerebral edema after ICH. 68 In addition, thrombin, another component of hematoma, could induce the activation of PAR-1 and migration of astrocyte protoplasm, leading to cerebral edema. 69 In contrast, another type of reactive astrocyte, A2, showed more “protective” functions. Several studies have shown a positive role of A2 reactive astrocytes in recovery after hemorrhagic stroke. 70 It has been reported that A2 reactive astrocytes play an important role in axonal regeneration after hemorrhagic brain stroke. 71 Therefore, similar to microglia, astrocytes exert two phenotypes and perform different functions at different stages after stroke.
Neutrophils
Neutrophils are recruited to the injury site and aggravate inflammation with the destruction of the BBB after microglia are activated by damage-associated molecular patterns (DAMPs) after hemorrhagic stroke. 72 It was found that neutrophil infiltration occurs in the hematoma and around it within a couple of days, peaks between 2 and 3 days, and then almost disappears within 3 to 7 days after ICH in mice. 73 Similarly, activation of microglia and infiltration of peripheral neutrophils were significantly increased at 24 h after SAH in rats. 74 Moreover, clinical studies have provided evidence supporting the role of neutrophils in ICH and SAH. In an autopsy study of patients who died within 5-8 h of onset of ICH, a small number of neutrophils were observed in the tissue surrounding the hematoma. 75 Also, it has been shown that depletion of neutrophils could downregulate inflammation after SAH. 76 As a result of these findings, infiltrating neutrophils may play a role in ICH and SAH-induced brain injury. However, the precise mechanisms remain a mystery.
NK cell
With the destruction of BBB after hemorrhagic stroke, NK cells are one of the earliest cells infiltrating into brain tissue; and they are the primary immune cell subgroup in the early stages of ICH and SAH. NK cells are immune cells with innate and adaptive immune properties, which can directly promote immune defense through releasing of cytokines and regulating the adaptive response antigen presenting cells and T cells. Single-cell transcriptional profiles showed two primary NK cell subgroups characterized by high cytotoxicity or strong chemokine production in the brain tissue after ICH, distinguishing them from periphery NK cells. 77 NK cells increase focal inflammation through cytotoxicity to brain endothelial cells and neutrophil recruitment, exacerbating BBB destruction and cerebral edema by cooperating with neutrophils after ICH. Consistent with these findings, previous studies have reported that NK cells promote focal inflammation and nerve damage in the SAH setting.78,79 However, the infiltration of NK cells in the brain is caused by a unique environment in the brain or a similar inflammatory environment and may also occur in other organs after nerve damage. Inhibition of NK cell infiltration into the brain may be an effective way to protect against brain injury after ICH and SAH. Future research on this aspect is essential to unravel the brain-specific characteristics of NK cells after nerve injury.
T lymphocytes cell
The role of T lymphocytes in ischemic stroke has been well studied, whereas their role in hemorrhage stroke is not well understood. The previous study demonstrated that CD4+ T lymphocytes were the predominant brain leukocyte population in murine models of ICH at ≥24 h. 80 Additionally, CD4+ Th cells were induced for 4 days after ICH; CD8+ cytotoxic T cells increased within 24 h of ICH, peaking 2–7 days after ICH.81,82 CD8+ and CD4+ cells may contribute to inflammatory damage after a hemorrhagic stroke, since lacking CD8+ or CD4+ cells have been reported to show protection against brain injury after ischemic stroke in mice. 83 However, emerging data indicate that the regulatory T cells (Treg) may also have neuroprotective effects in ICH and SAH. A significant increase of CD4+ Treg cells is observed in the hemorrhagic hemisphere, starting at 1 d, peaking at 4 d, and then decreasing at 14 d after ICH. 84 Stimulating of CD28 agonist antibodies or adoptive transfer of Tregs may improve ICH or SAH-induced inflammation. 85 Considering the delayed infiltration of helper T cells, they may influence the later stages of the immune response in the acute phase of hemorrhage stroke by shaping the inflammatory or protective microenvironment. Further studies are needed to understand T-cells’ mechanisms and roles and their pro-inflammatory and anti-inflammatory mediators after stroke. 8
Increasing evidence suggests that stroke is a systemic disease affecting more than just the brain, including neurogenic cardiac disorders, lung injury, and systemic immune dysfunction.21–25,86 Moreover, a significant role for peripheral immune responses in neuropathy is being recognized. 87 In ischemic stroke, brain-immune system interactions are extensively studied, and stroke can cause immune consequences such as local autoimmunity and peripheral immunosuppression. 88 The peripheral immune system and the crosstalk between the peripheral blood and the brain are thought to be involved in the immune response in hemorrhagic strokes, which may influence SBI and recovery from hemorrhagic stroke. 89
Brain-spleen crosstalk after hemorrhagic stroke
Spleen is the largest secondary lymphoid organ with extensive immune functions. As a result of its activation, inflammatory responses are altered, the immune system is dysregulated, neuroinflammation occurs, infection risks increase, and neurological recovery is impaired. 86 Multiple mechanisms of brain-spleen crosstalk following stroke have been previously reported, including sympathetic nervous system activation, chemokine production, and antigen presentation occurring in the damaged brain.90–93
Stroke-induced immunodepression syndrome (SIDS) is closely related to the activation of 3 systems: the sympathetic nervous system (SNS), 94 the parasympathetic nervous system (PNS), 95 and the hypothalamus-pituitary-adrenal (HPA) axis. 96 SIDS is characterized by profound atrophy of secondary lymphatic organs (spleen, thymus, and lymph node) that occur within the first few days after stroke. 97 There are several brain-spleen contacts after the occurrence of a hemorrhagic stroke. In response to SAH, the SNS/HPA should contract the spleen, which is thought to be the first link between the CNS and peripheral immune system after stroke. 98 Nevertheless, in the aftermath of stroke, it is unclear how the SNS/HPA axis regulates the splenic-mediated peripheral immune response. The sympathetic nervous system (PNS) is an important additional mediator of brain-pleural communication in the brain and may regulate the immune response of the spleen by reacting with the SNS rather than directly innervating the spleen. 99 In an animal model of hemorrhagic stroke, PNS activation reduces systemic inflammation and neuroinflammation and improves neurological function. 100 With the damage of BBB, CNS-specific antigen exposure and cytokines or chemokines-induced immune response can further exacerbate secondary brain injury, which is another crucial link to hemorrhagic stroke. In addition, lymphatic connections may also expose the peripheral immune system to CNS antigens. 101 Otherwise, splenic-mediated lymphocyte responses are recruited and infiltrated into the center to participate in the inflammatory response after a hemorrhagic stroke. Emerging research suggests that the crosstalk of the stroke-splenic-peripheral immune system exerts a significant role in the prognosis of stroke (Figure 2).
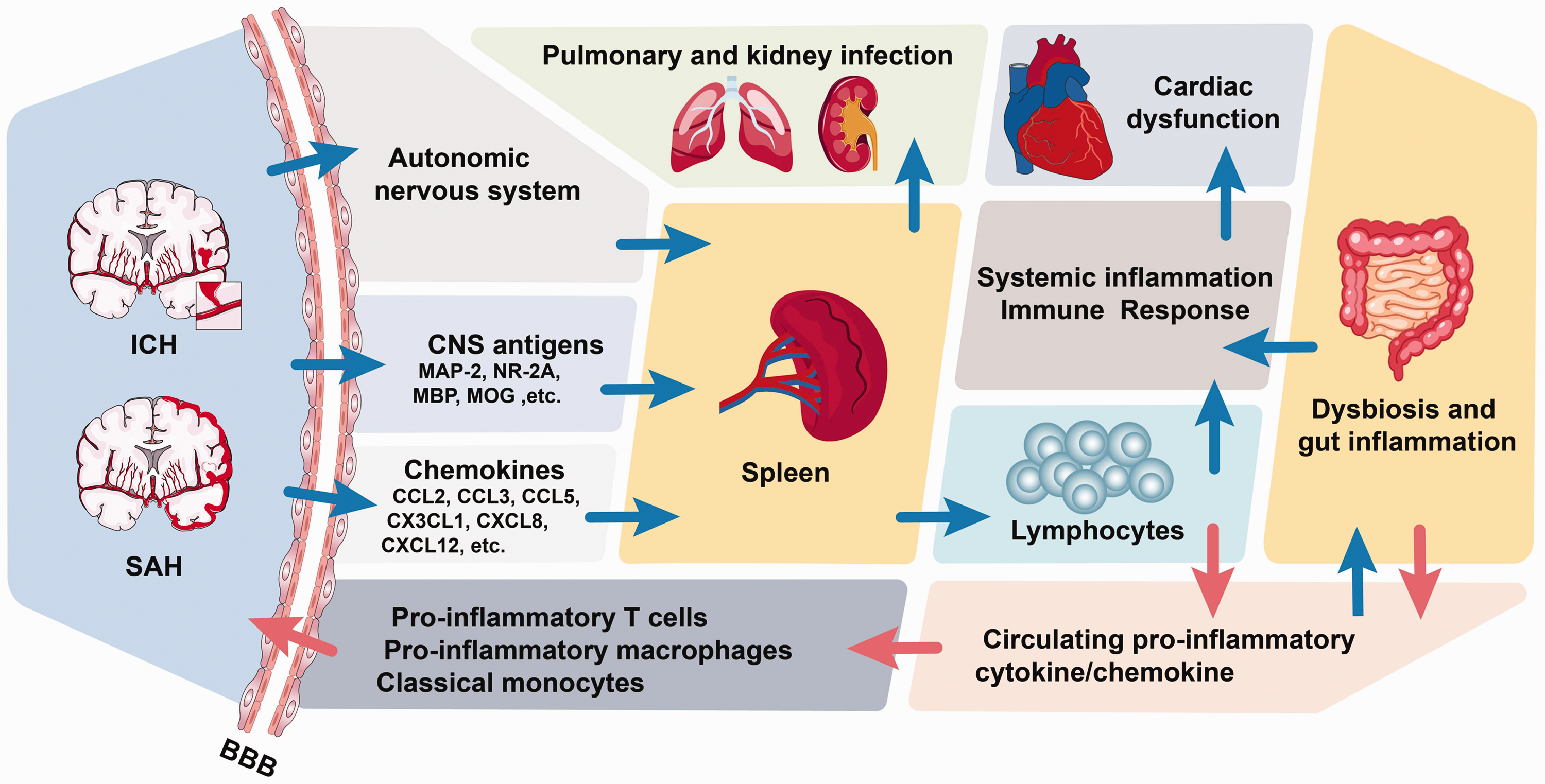
Hemorrhagic stroke-induced systemic inflammatory response syndrome. ICH and SAH brain promote the spleen to generate lymphocytes by the autonomic nervous system, CNS antigens, and chemokines, causing systemic inflammation and immune response. The systemic inflammatory response contributes to pulmonary infection, kidney infection, and cardiac dysfunction. Circulating pro-inflammatory cytokine/chemokine leads to dysbiosis and gut inflammation, while dysbiosis and intestinal inflammation also increase the associated cytokines/chemokines. All these circulating pro-inflammatory cytokines also increase the pro-inflammatory T cells, pro-inflammatory macrophages, and classical monocytes, which could access the brain parenchyma across the dysfunctional blood-brain barrier. ICH: intracerebral hemorrhage; SAH: subarachnoid hemorrhage; BBB: blood-brain barrier; MAP: microtubule-associated protein; NR: N-methyl-D-aspartate receptor; MBP: myelin basic protein; MOG: myelin oligodendrocyte glycoprotein.
After a stroke, the spleen atrophies, cells change, and inflammatory cytokines are released, worsening the local inflammatory response. 23 Hemorrhagic and ischemic stroke models have shown that splenic constriction contributes to systemic inflammatory response syndrome (SIRS) by releasing leucocytes into the bloodstream and altering inflammation in the brain.102,103 It is found that more lymphocytes flow into the bloodstream with spleen contraction. 23 In an autologous ICH model, massive cerebral hemorrhage resulted in a significant decrease in leucocytes and lymphocytes and a significant increase in monocytes in the blood and spleen. 104 Moreover, splenic-derived T and B lymphocytes, Th cells, and cytotoxic T cells are significantly reduced in peripheral blood irrespective of the size of the cerebral hemorrhage. More severe splenic constriction, more risk of infection, and poorer prognosis are reported in patients with a larger area of cerebral hemorrhage.104,105 With splenic constriction, the cytokine levels, such as IFN-γ, IL-6, IL-10, IL-12, and IL-13, are all elevated after stroke patients.106,107 Moreover, previous studies have shown that splenectomy attenuates ICH-induced neurological and cognitive impairment and ICH-induced cardiac dysfunction. The results showed that splenectomy before ICH decreased inflammatory factor (such as ICF, MCSA, MCP-1, NOX2, and so on) expression and leukocyte and macrophage infiltration into the brain or heart, as well as decreased ICH- induced astrocyte and leukocyte activation, and increased M2 macrophage expression. 108 Therefore, Brain-spleen crosstalk is a critical point in CNS-peripheral immune dialogue after hemorrhagic stroke. Furthermore, a new therapeutic approach for hemorrhagic stroke may involve targeting the spleen's immune response.
Gut microbiota in hemorrhagic stroke
More and more evidence showed the vital role of the gut-brain axis during stroke.26,28,109 Human gut microbiota is involved in different stages of stroke, from risk factors and facilitation of stroke progression to stroke recurrence. The risk of stroke has been shown to be reduced by maintaining an average balance of commensal and pathogenic bacteria. 110 When ICH occurs, it can trigger an intense neuroinflammatory response and alter peripheral immunity, which mediates gut microbiota dysbiosis, leading to a marked reduction in species diversity, overgrowth of some flora, reduced intestinal dynamics, and increased intestinal permeability in mice. 111 Furthermore, recolonization with normal healthy flora is found to improve functional deficits and neuroinflammation after ICH. In addition, ICH-induced neurobehavioral deficits can be improved by modulating gut microbiota. 112 As the same, SAH could also induce alteration of intestinal flora. 113 After brain injury, Th1 cell differentiation promotes the secretion of inflammatory cytokines, enhances the inflammatory response, and aggravates ICH-induced neuroinflammation.34,114 In neurological disorders, the gut microbiota plays a crucial role in regulating T-cell homeostasis and neuroinflammation; gut microbiota instability can lead to changes in T-cell homeostasis, induce pro-inflammatory responses, and worsen outcomes after ICH. 111 Therefore, the gut-brain axis plays a dominant role in stroke, which could be a therapeutic target to mitigate this peripheral contribution to hemorrhagic stroke.
Peripheral organs and tissues infections were induced by hemorrhagic stroke
Growing evidence now suggests that systemic immune changes occur in peripheral organs and tissues after stroke, such as in the liver, lung, heart, and kidney.21,23–25,86,115,116 Pulmonary infections are a commonly seen complication in stroke patients. Previous studies have shown that the occurrence of pulmonary infection is associated with various risk factors, including disease history, hypoproteinemia, degree of impaired neurological function, stroke severity, stroke site, surgical approach, and medication. 117 In addition, L. Hosang et al. found a strong link between the lung microbiome and the immune response of the brain, showing that dysregulation of the lung microbiome significantly affects susceptibility to the development of CNS autoimmune diseases in rats. 25 Furthermore, here is evidence about the crosstalk between the brain and kidney after stroke. Stroke can lead to renal dysfunction, which hurts the patient's prognosis. During ICH and SAH, releasing inflammatory mediators from immune cells can increase systemic inflammatory and immune responses mediated by the spleen, which may play a central role in promoting renal dysfunction. 118 Animal studies also showed significant renoprotective effects could be achieved by activating cholinergic anti-inflammatory pathways in splenic immune cells. 119 Also, a positive correlation between cirrhosis with SAH and ICH is investigated by meta-regression analysis suggesting an association between brain and liver in hemorrhagic stroke, but the mechanism is unclear and requires further study in the future. 120
Moreover, cardiac injury is common and severe in patients with hemorrhagic stroke. 121 It usually occurs a few days after the onset of acute stroke or even death and includes abnormal heart rhythms such as heart attack, congestive heart failure, cardiac arrest, and atrial fibrillation. 121 Several mechanisms can contribute to cardiac dysfunction after strokes, such as hypothalamic-pituitary-adrenal (HPA) axis activation, sympathetic and parasympathetic nerve modulation, a surge of catecholamines, dysregulation of the gut microbiota, and immune response.24,116
Immune-based strategies for hemorrhagic stroke therapy
Effective therapeutic strategies for acute hemorrhagic stroke are still scarce, and many neuroprotective trials have failed. Based on the review above, the immune system plays a central role in affecting the progression, outcome, and recovery of stroke. Here, several strategies targeting different immune cells and organs are reviewed in ICH and SAH (Table 1).
Immune-based strategies for hemorrhagic stroke therapy.
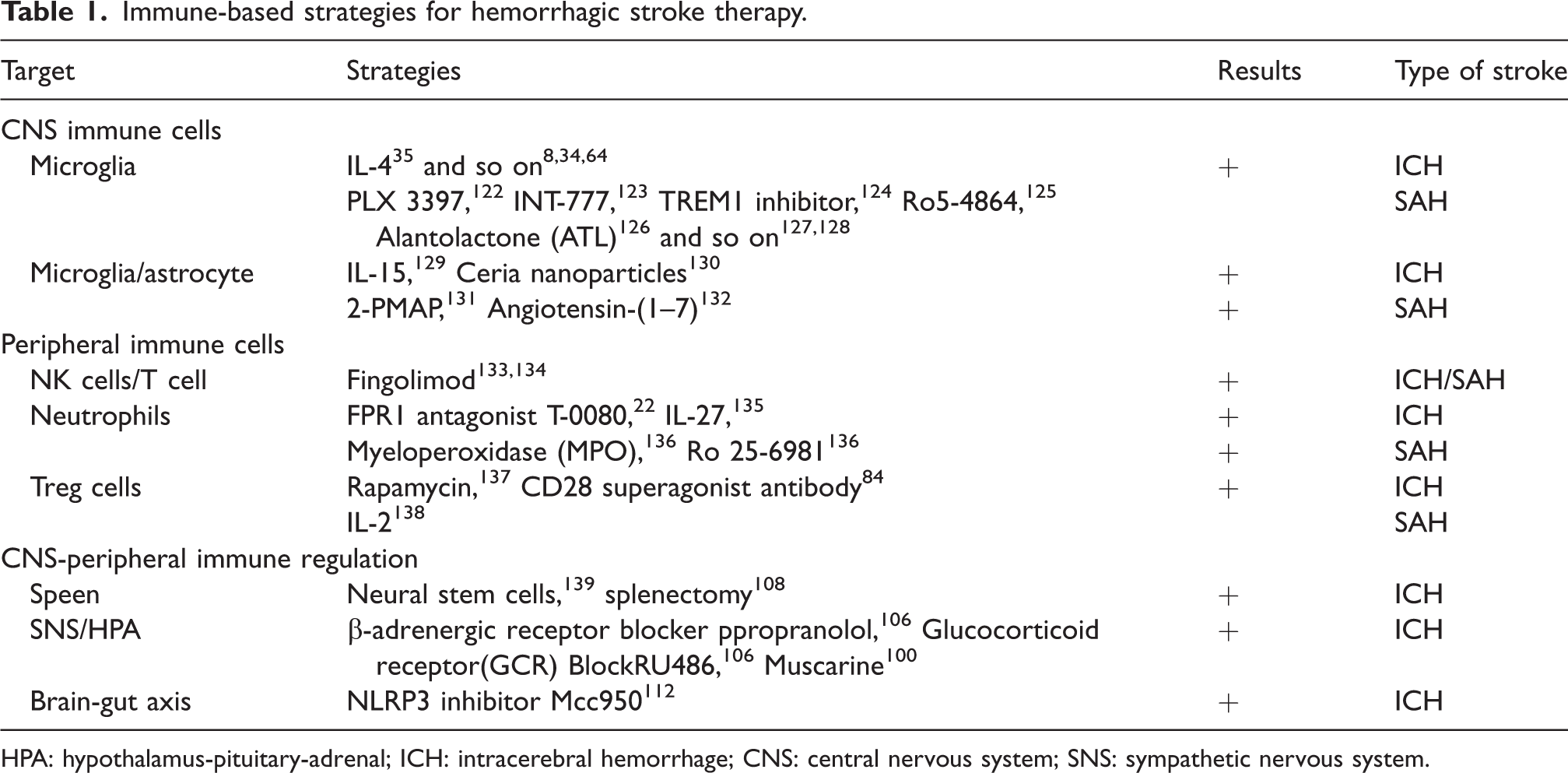
HPA: hypothalamus-pituitary-adrenal; ICH: intracerebral hemorrhage; CNS: central nervous system; SNS: sympathetic nervous system.
Targeting on CNS immune cells
Due to the BBB, the inflammatory system of the brain is different from that of other tissues. Microglia, the tissue-resident inflammatory cells of the brain, could be a key target for hemorrhagic stroke. Numerous therapeutic strategies targeting om microglia activation and polarization have been summarized and shown to be effective in ICH treatment.8,34,64,140 There is an ongoing clinical trial (NCT03737344) studying immune therapies targeting IL-1, and IL-4 has been reported to facilitate hematoma resolution after ICH. 35 In addition, two microglia-focused treatment strategies have been reported, one of which is inflammatory preconditioning by LPS; the other is pharmacological deactivation with PLX3397, which significantly reduces brain injury. 122 Also, many compounds or drugs have been reported to exert an inhibition effect on microglia activation, such as NLRP3 inhibitor INT-777 and TREM1 inhibitor.123,124 Moreover, modulating microglia polarization could attenuate brain injuries after SAH, such as LRP1 activation, TSPO ligand Ro5-4864, Alantolactone (ATL), and sirt1.125–128
Astrocytes is also an important member of the CNS immune system. There are several strategies targeting astrocytes. It has been reported that IL-15 can bridge the crosstalk of astrocytes and microglia after ICH, which would exacerbate the brain injury, indicating that IL-15 modifying could be a therapeutic strategy for ICH. 129 Ceria nanoparticles have been reported to ameliorate white brain injury after ICH by targeting microglia-astrocytes interactions. 130 2-PMAP can significantly reduce apoptotic endothelial cells after SAH, inhibit vasospasm, and improve microglia and astrocyte activation as well as neuroinflammation and oxidative stress, leading to reduce neuronal apoptosis in the cerebral cortex. 131 Ang-(1-7)/MASR has neuroprotective and modulatory properties that can reduce brain injury through effects on neuroinflammation, vascular contraction, and antithrombosis. 132
Targeting peripheral immune cells
Strategies targeting peripheral immune cells have also been tried after hemorrhagic stroke. In recent clinical trials, oral fingolimod for ICH proved effective at reducing SBI caused by the initial hematoma. Essentially, it prevents lymphocytes from migrating from lymph nodes to circulating pools, which prevents T cell or NK cell infiltration into the central nervous system. 133 In addition, Fingolimod shows improvement in neurological outcomes and significantly reduced intravascular adhesion of leukocytes to pial venules after SAH in rats. 134
Neutrophil infiltration into the CNS is linked to neurobehavior deficits after SAH and ICH. Neutrophils depletion has been researched to reduce the BBB breakdown and neuroinflammation after ICH 141 and improve memory via NMDA receptors after SAH, showing that Ro 25-6981 is a specific NR2B inhibitor that ameliorates vasospasm after SAH. 142 The formyl peptide receptor 1 (FPR1) is one of the earliest expressed genes after ICH, which is a DAMP receptor and participates in microglia activation. Activated microglia attract neutrophils to the surrounding tissues of hematoma through FPR1-mediated IL-1. 22 FPR1 antagonist T-0080 can attenuate brain edema and improve neurological outcomes after ICH. 22 Also, MPO (myeloperoxidase), a neutrophil enzyme, has been reported to play important roles in the activation of mediate microglia and astrocyte as well as the death of neuronal cells. 136 Indeed, many compounds, and interleukin factors have been reported to inhibit neutrophils and improve hemorrhagic stroke.
At the ICH and SAH recovery stage, Treg cells increase and secrete immunosuppressive cytokines such as IL-10, which can be important for ICH and SAH recovery.34,85,143 Previous studies have shown that Transferring Treg cells in the ICH model exert inhibit effect on microglia activation and neuroinflammation after ICH. 144 Enhancement of Tregs with CD28 superagonist antibodies reduces inflammatory damage, and rapamycin significantly improved neurobehavioral deficits after cerebral hemorrhage by inhibiting mTOR activation, increasing peripheral blood and, brain Treg counts, increasing IL-10 and transforming growth factor-β levels, and decreasing interferon-gamma levels.84,137 And low doses of IL-2 can elevate the number of Tregs, significantly suppress brain pro-inflammatory factors and peripheral neutrophils, and reduce neuronal damage after SAH. 138
Targeting on CNS-peripheral immune regulation
As a result of hemorrhagic stroke, systemic oxidative stress and peripheral immune responses are activated, which contribute to SIDS and SRIS development. Therefore, targeting on CNS-peripheral immune regulation can find some effective strategies for hemorrhagic therapy.
Firstly, spleen plays an integral part in SIDS and SRIS. In recent studies, splenectomy has been shown to ameliorated ICH-induced cognitive and neurological impairments. 108 Studies have shown that early intravenous neural stem cells (NSC) injection can reduce TNF-α and IL-6 levels and promote neuroprotection, mainly by NSC accumulating in the spleen and interrupting splenic inflammatory responses after ICH. 139 In addition, propranolol (β-adrenergic receptor blocker) and RU486 (glucocorticoid receptor (GCR) blocker) could partially inhibit spleen shrinkage induced by ICH and improve the outcome of ICH. 106 For the cholinergic anti-inflammatory pathway (CHAIP) is a physiological mechanism for the central nervous system regulating immune response, and targeting the cholinergic anti-inflammatory pathway is a promising way for immunomodulation. 100 It has been reported that muscarine improved neurologic outcomes in ICH rats, showing that reduction of brain water content and levels of inflammatory mediators decrease in both brain and spleen. However, muscarine is reported to be ineffective for ICH rats with spleen, suggesting that the effect of muscarine is mediated by the vagus nerve-spleen pathway rather than direct interaction with the brain. 100
In addition, the Gut-Brain axis also participates in ICH, exhibiting that gut bacteria could produce neuroactive compounds and modulate neuronal functions. Thereby, gut microbiota can be targeted for ICH therapy. It has been found that ICH can cause intestinal microbiome dysregulation, and the selective NLRP3 inflammasome inhibitor MCC950 was administered to regulate intestinal microbiome dysregulation and improve neurological deficits. 112
Perspective and conclusion
In the past 20 years, nearly all clinical trials of neuroprotective drugs for hemorrhagic stroke have failed, making the study of stroke enter embarrassing situation. It is imperative to reexamine the pathological mechanisms of hemorrhagic stroke. In this review, we systematically revealed the roles of immunity and inflammation in stroke pathology, laying a foundation for stroke immune intervention.
During a hemorrhagic stroke, the harmful signals released by damaged neuronal cells can activate the immune response, including glia activation, peripheral immune cell infiltration, and production of cytokines. Neuroinflammation after stroke accelerates the destruction of the BBB, aggravates brain edema and oxidative stress, and leads to microcirculation disorders, resulting in secondary brain injury. In the subacute phase of stroke, SIRS occurs, manifested by splenic contraction, imbalance of gut microbiota, lung and kidney infections, and induction of cardiac disease. Therefore, understanding the interaction between the central and peripheral immune systems after hemorrhagic stroke helps provide new strategies for early prevention and intervention of hemorrhagic stroke. Furthermore, it will be of great scientific to explore the molecular mechanisms of interaction between CNS and peripheral immune cells and seek possible targets for splenic contraction and modulation of gut microbiota, which will improve the prognosis of patients and reduce the risk of death after a hemorrhagic stroke.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (No. 81830036).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
