Abstract
The role of endogenous endothelins in mediating postischaemic hypoperfusion after transient global ischaemia was investigated in halothane-anaesthetised rats. Pretreatment with the broad-spectrum (ETA and ETB) endothelin antagonist, Bosentan (17μmol/kg) had minimal effect on postischaemic hypoperfusion, measured by hydrogen clearance, in the caudate nucleus and the parietal cortex in the 3 h after bilateral common carotid artery occlusion with concomitant haemorrhagic hypotension (transient global ischaemia). In a separate series of rats with CBF measured by [14C]iodoantipyrine autoradiography at 90 min after carotid occlusion with concomitant haemorrhagic hypotension, Bosentan treatment failed to significantly alter CBF in any of the 35 brain regions examined. No significant alterations in CBF, measured by hydrogen clearance, were observed after transient bilateral common carotid artery occlusion. [14C]Iodoantipyrine autoradiography at 90 min after occlusion failed to demonstrate any significant increases in CBF after transient bilateral common carotid artery occlusion in any of the 35 brain regions examined in anaesthetised rats. The failure of the broad-spectrum endothelin antagonist Bosentan, at concentrations known to inhibit the cerebrovascular effects of exogenous ET-1, provide no support for the view that endothelins have a major role in mediating acute postischaemic hypoperfusion.
Keywords
Transient global cerebral ischaemia is characterised by a marked reduction in cerebral blood flow followed by transient hyperaemia and delayed hypoperfusion (Hossmann et al., 1973; Snyder et al., 1975; Miller et al., 1980; Pulsinelli et al., 1982; Kagstrom et al., 1983). The onset of postischaemic hypoperfusion is rapid after the transient hyperaemic response, and the mechanisms mediating postischaemic hypoperfusion, despite much speculation, remain unclear (Miller et al., 1980; Frerichs et al., 1992). An increase in vascular tone, obstruction of the cerebral microvasculature, as a result of blood aggregation or constriction of cerebral vessels due to tissue oedema or glial swelling, are but some of the mechanisms proposed (Wade et al., 1975; Grogaard et al., 1989; Moskowitz et al., 1990; Wahl and Schilling, 1993; del Zoppo, 1994; Dirnagl et al., 1994).
The endothelin isopeptides [endothelin-1 (ET-1), endothelin-2 (ET-2), endothelin-3 (ET-3)] are potent vasoconstrictors of vascular smooth muscle (Yanagisawa et al., 1988; Seo et al., 1994; Warner et al., 1993). The effects of the endothelins are mediated primarily via two receptor subtypes (Arai et al., 1990; Sakurai et al., 1990; Masaki et al., 1994). At the ETA receptor, ET-1 and ET-2 are more selective than ET-3, whereas the isopeptides are equipotent at the ETB receptor (Arai et al., 1990; Sakurai et al., 1990). The vasoconstrictor effects of the endothelins are mediated via ETA receptors, whereas the ETB receptor mediates the vasodilatory response (Masaki et al., 1994; Rubanyi and Polokoff, 1994). Recently the existence of ETB receptors mediating vasoconstriction have been demonstrated in some vascular beds (Clozel et al., 1992; Shetty et al., 1993). Endothelin-1 is a potent constrictor of major cerebral arteries, arterioles, and microvessels in vitro and in vivo in all species examined (Robinson and McCulloch, 1990; Feger et al., 1994; Sagher et al., 1994). The ability of exogenous ET-1 to reduce CBF to levels that induce neuronal damage has implicated these peptides in a variety of cerebrovascular pathologies (Robinson et al., 1990; Fuxe et al., 1992; Macrae et al., 1993). In the cerebrovasculature, topical or intraparenchymal application of ET-1 on to the middle cerebral artery in the rat results in marked decreases in CBF and neuronal damage (Robinson et al., 1990; Macrae et al., 1993; Sharkey et al., 1993). Intrastriatal injection of ET-1 results in marked neuronal damage within the striatum (Fuxe et al., 1992). Investigations have demonstrated elevations in tissue endothelin immunoreactivity after permanent and transient focal cerebral ischaemia (Duverger et al., 1992; Viossat et al., 1993; Barone et al., 1994). Increases in plasma and CSF endothelin levels after stroke and subarachnoid haemorrhage, as well as increases in tissue endothelin levels after focal and global ischaemia, have been reported by a number of groups (Suzuki et al., 1992; Ziv et al., 1992; Yamashita et al., 1993; Barone et al., 1994). The endothelins have also been implicated in experimental cardiac ischaemia and acute renal failure (Watanabe et al., 1990; Lopez-Farre et al., 1991; Brunner et al., 1992).
Investigations into the role of the endothelins in the cerebral circulation have been hampered by the absence of brain-penetrating endothelin-receptor antagonists. We recently demonstrated that the broad-spectrum (ETA/ETB) nonpeptide receptor antagonist Bosentan (Clozel et al., 1994) gains access to the adventitial surface of feline pial resistance arterioles, after systemic administration, and antagonises the vasoconstrictive effect of exogenous ET-1 (Patel et al., 1994). The role of endogenous endothelins in mediating postischaemic hypoperfusion was investigated by using the endothelin-receptor antagonist Bosentan. These investigations were carried out in halothane-anaesthetised rats by using models of severe global ischaemia (transient bilateral common carotid artery occlusion and concomitant haemorrhagic hypotension) and moderate global ischaemia (transient bilateral common carotid artery occlusion).
MATERIALS AND METHODS
Surgical Preparation
The experiments were carried out in 59 adult male Sprague–Dawley rats (303–439 g). The animals were initially anaesthetised with 4% halothane, tracheotomised, and mechanically ventilated with a gas mixture of 70% N2O/30% O2 containing halothane (0.75–1.5%). Both femoral arteries and veins were catheterised for the administration of drugs and fluids. The key physiologic variables (arterial blood pressure, arterial blood gas status, and rectal temperature) were continuously monitored for the duration of the experiment. Care was taken to prevent the direct heating of the head. The common carotid arteries were isolated from surrounding tissue by blunt dissection and loose loop ligatures (2–0 silk suture) placed around the arteries.
Hydrogen Electrode Placement
The animals were positioned in a stereotactic frame (Kopf instruments), and after a midline incision, the surface of the skull was exposed. Two burr holes were drilled to expose the surface of the dura. The dura was excised and Teflon-coated platinum electrodes (250μm outer diameter, Clark Electromedical) positioned in the left caudate nucleus (coordinates: anterior, 1 mm; medial, 3 mm; dorsoventral, 4 mm) and in the right parietal cortex (posterior, 2.4 mm; medial, 4 mm; dorsoventral, 1 mm). The coordinates were taken (relative to bregma) from the atlas of the rat brain of Paxinos and Watson (1986). The burr holes were sealed with cyanoacrylate glue, and the platinum electrodes were cemented in place with dental cement. A silver reference electrode was placed subcutaneously on the back of the animals.
Measurement of Cerebral Blood Flow
Hydrogen Clearance. After an equilibration period of ∼30 min, the platinum electrodes were connected to a reference circuit and polarised to −700 mV. A further period of 30 min was allowed for stabilisation after polarisation. Before the measurement of CBF, the inspired gas mixture was altered to 45% O2, 35% H2, and 20% N2O with 0.75%–1.5% halothane. A saturation period of 10 min was allowed, followed by 10 min for clearance. The CBF data were collected over a period of 1 min, 1 min after the H2 mixture had been switched off (to reduce artefacts due to recirculation of hydrogen). The data were fitted to an experimental curve, and blood flow was calculated by using the initial slope index technique (Young, 1980). Measurements of CBF were carried out before occlusion, at 7.5 min after the occlusion of the carotid arteries, at 5 min after the release of the carotid ligatures, and at 30-min intervals for 3 h during the reperfusion phase.
[14C]Iodoantipyrine Autoradiography. At 90 min after the release of the carotid ligatures, CBF was measured by using [14C]iodoantipyrine as the radiotracer. The method used was modified from the method of Sakurada et al., 1978. Briefly, 50 μCi of radioisotope in 1.5 ml saline was administered as a ramped intravenous infusion over a period of 30 s. During the infusion, 15–18 samples of arterial blood were collected on preweighed filter disks. At the end of the infusion of isotope, the animals were decapitated and the brains removed and frozen in isopentane at −42°C. The filter disks were reweighed, and a 30% solution of hydrogen peroxide was added to bleach the blood from the disks. After the addition of liquid scintillant, [14C] radioactivity was determined by liquid scintillation counting.
Twenty-micron sections of the frozen brains were serially cut in a cryostat at −20°C, and at 140-μm intervals, sections were collected onto glass coverslips and rapidly dried at 60°C. The coverslips were mounted onto cards and apposed to Kodak SB-5 film for 4 to 6 days. Precalibrated [14C] standards (concentration range, 44–2,500 nCi/g tissue equivalents) were placed in the film cassettes. The autoradiograms were analysed by using a computer-based image-analysis system (M4 MCID, Imaging Research, Inc.). Six bilateral readings were taken from sections where the structure could be identified from the rat brain atlas of Paxinos and Watson (1986).
The values of CBF were determined from the equation of Sakurada et al., 1978, with the knowledge of arterial blood history of [14C], tissue levels of [14C], and the tissue–blood partition coefficient (0.79).
Experimental Protocol
Bilateral Carotid Occlusion and Systemic Haemorrhagic Hypotension. Vehicle (0.9% saline) or Bosentan (17 μmol/kg) was administered as an intravenous bolus 10 min before the induction of ischaemia. The intravenous injections were carried out over a period of 3 min. The common carotid arteries were occluded, and MABP reduced to between 40 and 50 mm Hg by concomitant arterial haemorrhage for 15 min (Smith et al., 1984). The haemorrhaged blood was stored in heparinised syringes in a water bath at 37°C. CBF was restored by the removal of the carotid ligatures and the reinfusion of the blood via the femoral vein.
Bilateral Common Carotid Occlusion. The protocol for this group of animals was similar to that described previously. The main difference was that the common carotid arteries were occluded bilaterally without concomitant systemic hypotension.
Plasma glucose samples were taken before occlusion and at 30 min into the reperfusion phase. At each flow determination, a blood sample was taken to measure arterial blood gas status.
Statistical Analysis. The data between the two treatment groups, for the hydrogen clearance and [14C]iodoantipyrine studies, were analysed using a Student's unpaired two-tailed t test. The data are expressed as mean ± SD.
RESULTS
Bilateral Carotid Occlusion and Systemic Haemorrhagic Hypotension
Cardiovascular and Respiratory Variables. Hydrogen clearance. There were no significant differences in the cardiovascular and respiratory variables in either the drug- or vehicle-treated animals after bilateral common carotid artery occlusion and systemic hypotension. The MABP at the outset was 97 ± 3 mm Hg in vehicle-treated animals and 102 ± 4 mm Hg in the Bosentan-treated animals. Arterial Pco2 was 39.5 ± 2.2 mm Hg (vehicle group) and 39.2 ± 2.6 mm Hg (Bosentan group) at the outset of the experiment and did not vary significantly from these levels. Arterial oxygenation was maintained >100 mm Hg. The mean rectal temperature was 37.0 ± 0.3°C (vehicle group) and 36.9 ± 0.3°C (Bosentan group) and did not vary significantly from these levels. The MABP was maintained between 42 and 50 mm Hg during the period of occlusion (Fig. 1A). On the release of the ligatures around the carotid arteries, the blood pressure was restored to preocclusion levels by a slow intravenous infusion of blood. The administration of Bosentan (17 μmol/kg) had minimal effect on MABP.
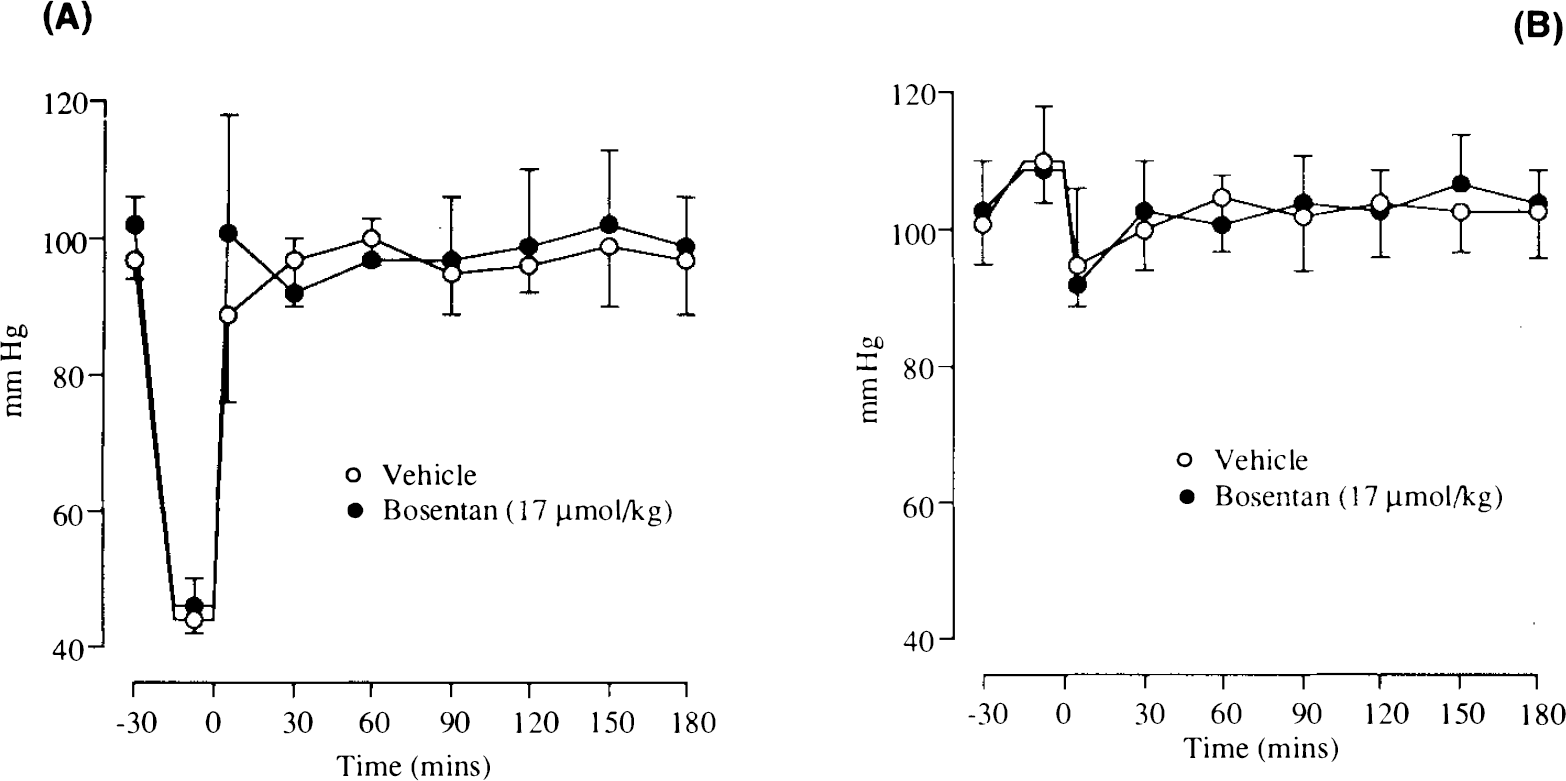
Autoradiography. There were no significant differences in the cardiovascular and respiratory variables in either the drug- or vehicle-treated animals subjected to bilateral common carotid artery occlusion and systemic hypotension. Before the start of the blood flow determination, MABP was 95 ± 9 mm Hg (Bosentan) and 105 ± 15 mm Hg (vehicle). The MABP was maintained at 43 ± 3.5 mm Hg (Bosentan) and 45 ± 4.3 mm Hg (vehicle) during the period of occlusion. Arterial Pco2 was 40.1 ± 2.6 mm Hg (Bosentan) and 39.8 ± 1.8 mm Hg (vehicle) before the start of the blood-flow determination (Table 1).
Physiologie variables: [14C]iodoantipyrine autoradiography study
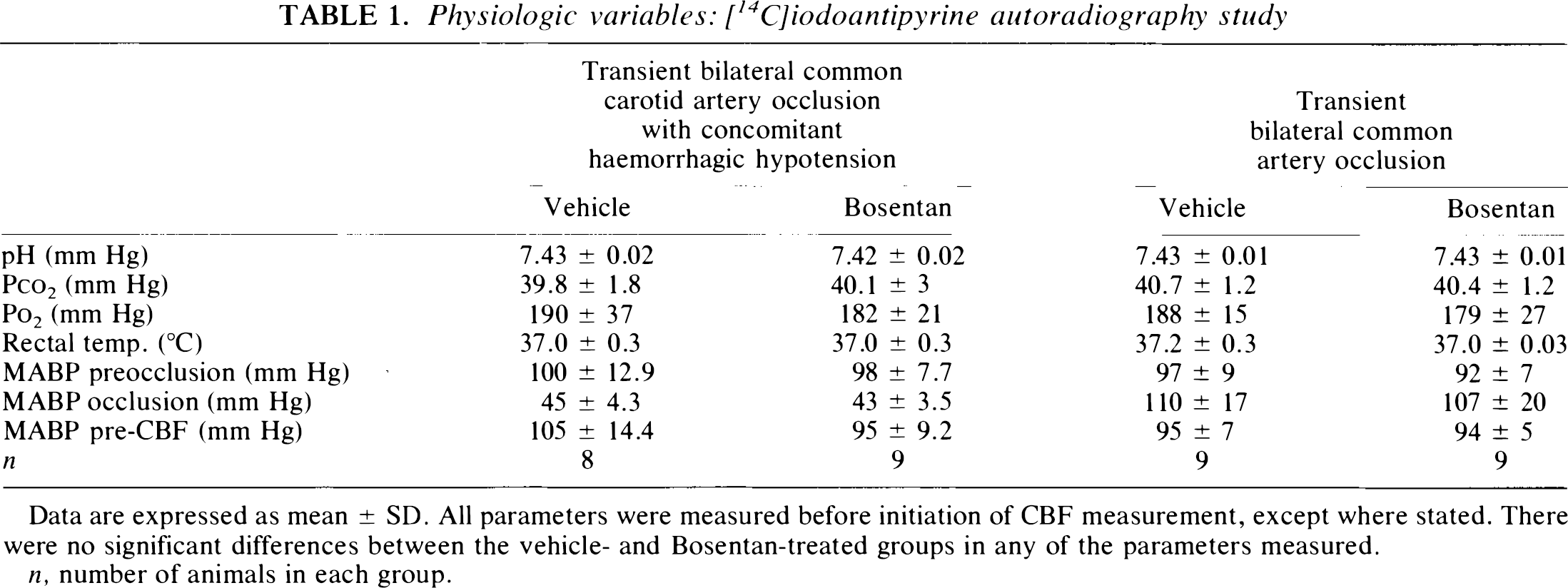
Data are expressed as mean ± SD. All parameters were measured before initiation of CBF measurement, except where stated. There were no significant differences between the vehicle- and Bosentan-treated groups in any of the parameters measured.
n, number of animals in each group.
Temporal Changes in CBF (Hydrogen clearance). In the caudate nucleus, CBF was reduced to 5 ± 4% (of preocclusion baseline) after the occlusion of the common carotid arteries and induction of hypotension. A similar decrease in CBF was obtained in the drug-treated animals (5 ± 6% of preocclusion baseline). The release of the carotid ligatures resulted in a marked hyperaemic response in both drug- and vehicle-treated group [160 ± 64% of baseline (Bosentan); 142 ± 72% of baseline (vehicle)]. The postischaemic hypoperfusion lasted from 30 to 180 min (end of experimental period). The hypoperfusion phase saw CBF reduced by ∼50% (of preocclusion baseline). Pre treatment with Bosentan did not alter the level of hypoperfusion (Fig. 2A).
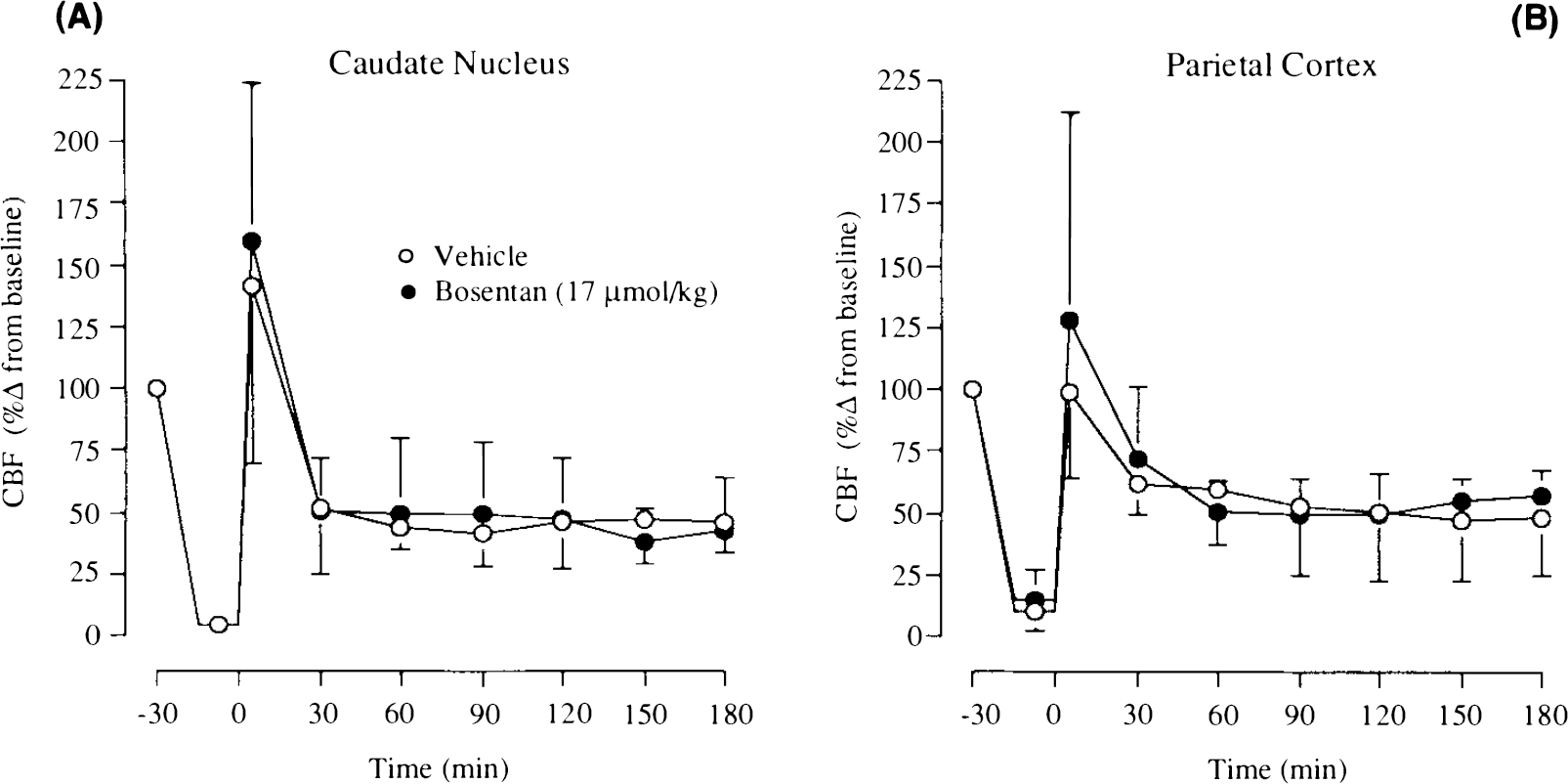
In the parietal cortex, the occlusion of the common carotid arteries and induction of haemorrhagic hypotension resulted in reductions in CBF in the cerebral cortex [15 ± 12% of preocclusion baseline (Besentan); 10 ± 8% of preocclusion baseline (vehicle)]. On the release of the carotid ligatures, a hyperaemic response was measured in animals pre-treated with Bosentan (128 ± 85% of baseline). However, no such response was measured in vehicle-treated animals. The reasons for the differences are unclear but could be attributed to the time of the blood flow measurement immediately after the release of the carotid ligatures. The level of hypoperfusion in the cerebral cortex stabilised after 60 min of reperfusion at ∼45% of preocclusion baseline (Fig. 2B). There was no alteration in the level of hypoperfusion after the pretreatment with Bosentan.
Spatial Distribution of CBF ([14C]Iodoantipyrine Autoradiography). There were no significant alterations in CBF between animals treated with vehicle or Bosentan in any of the 35 regions examined (Table 2; Fig. 3).
Transient bilateral common carotid artery occlusion
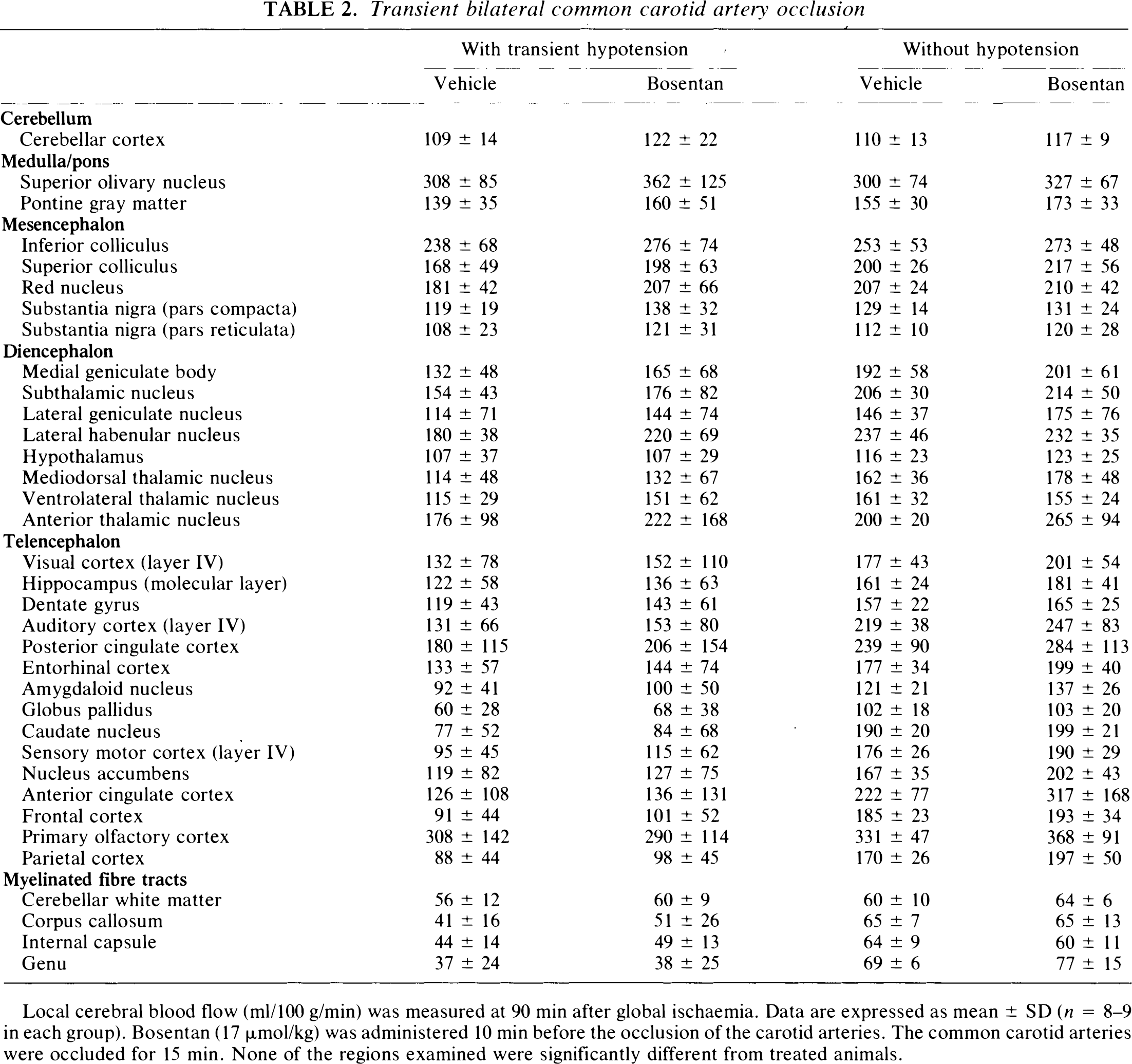
Local cerebral blood flow (ml/100 g/min) was measured at 90 min after global ischaemia. Data are expressed as mean ± SD (n = 8–9 in each group). Bosentan (17 μmol/kg) was administered 10 min before the occlusion of the carotid arteries. The common carotid arteries were occluded for 15 min. None of the regions examined were significantly different from treated animals.
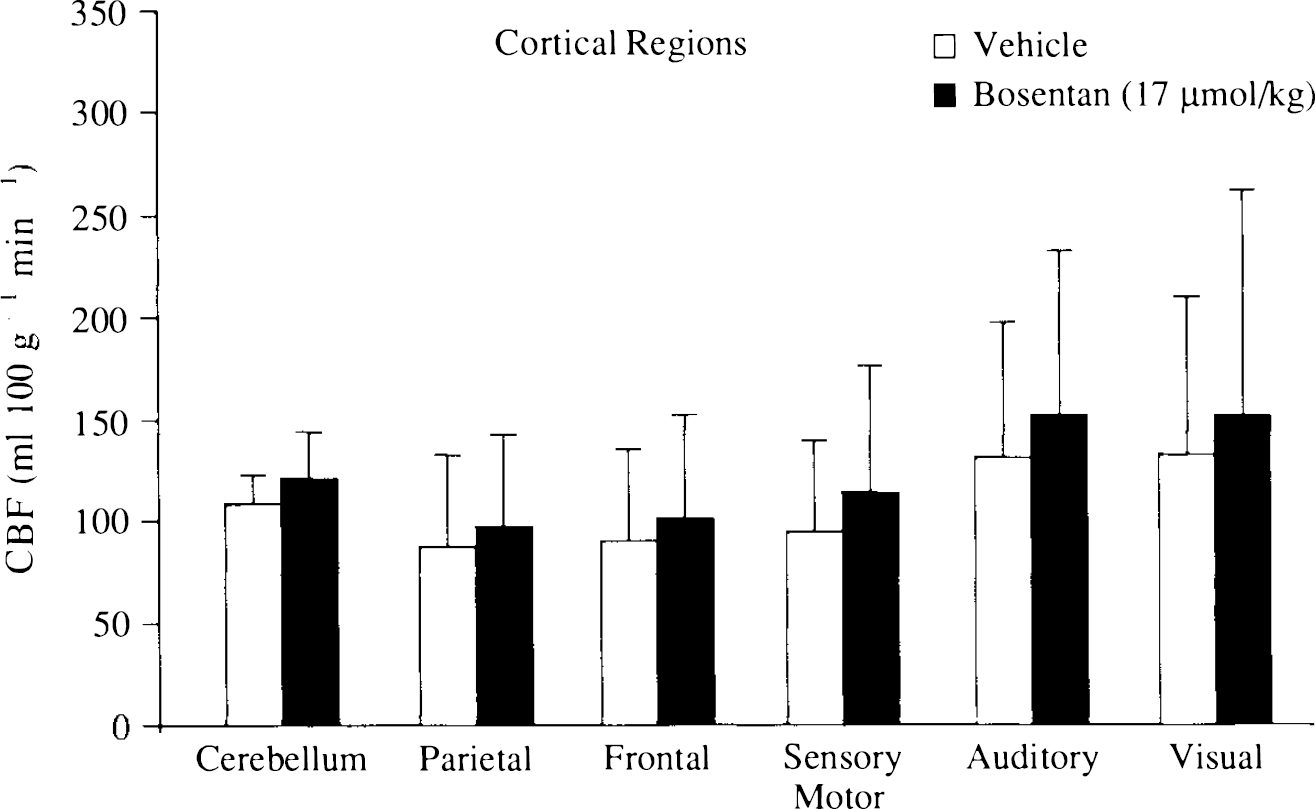
Effect of Bosentan (17 μmol/kg) on cerebral blood flow in the cerebral cortex of animals subjected to transient bilateral common carotid occlusion and systemic haemorrhagic hypotension. CBF was measured using [14C]iodoantipyrine autoradiography at 90 min after the end of ischaemia. Bosentan was administered as an intravenous bolus 10 min before the induction of ischaemia. Results are expressed as mean ± SD (ml 100 −1 min−1); n = 8 (vehicle); n = 9 (Bosentan).
Common Carotid Artery Occlusion
Cardiovascular and Respiratory Variables. Hydrogen clearance. There were no significant differences in the cardiovascular and respiratory variables in either the Bosentan- or vehicle-treated animals subjected to bilateral common carotid artery occlusion. The MABP at the outset was 101 ± 6 mm Hg in vehicle-treated animals and 103 ± 2 mm Hg in the Bosentan-treated animals. Arterial Pco2 was 39.3 ± 2.2 mm Hg (vehicle) and 39.4 ± 2.2 mm Hg (Bosentan) at the outset of the experiment and did not vary significantly from these levels. Arterial Po2 was maintained at >100 mm Hg. The mean rectal temperature was 37.1 ± 0.2°C (vehicle) and 36.8 ± 0.3°C (Bosentan) and did not vary significantly from these levels. There was a slight increase in the MABP in animals after bilateral common carotid artery occlusion. On the release of the ligatures, the blood pressure returned to preocclusion baseline levels (Fig. 1B). Intravenous administration of Bosentan (17 μmol/kg) had minimal effect on MABP.
Autoradiography. There were no significant differences in the cardiovascular and respiratory variables in either the Bosentan- or vehicle-treated animals subjected to bilateral common carotid artery occlusion. Before the start of the blood-flow determination, MABP was 92 ± 6 mm Hg (Bosentan) and 97 ± 9 mm Hg (vehicle). The MABP was 107 ± 20 mm Hg (Bosentan) and 110 ± 17 mm Hg (vehicle) during the period of occlusion. Arterial Pco2 was 40.7 ± 1.2 mm Hg (Bosentan) and 40.4 ± 1.2 mm Hg (vehicle) before the start of the blood-flow determination (Table 1).
Temporal Changes in CBF (Hydrogen Clearance). In the caudate nucleus, occlusion of the common carotid arteries substantially reduced CBF to 42 ± 21% of preocclusion baseline in Bosentan-treated animals and 50 ± 17% of preocclusion baseline in vehicle-treated animals. The release of the carotid ligatures was followed by a small transient hyperaemic response in only the drug-treated animals. CBF, in Bosentan-treated animals, was elevated by between 111 and 125% of preocclusion baseline during the first 2 h of reperfusion (Fig. 4A). This increase in blood flow was not statistically significant at any of the time points examined (p > 0.06, unpaired Student's t test).
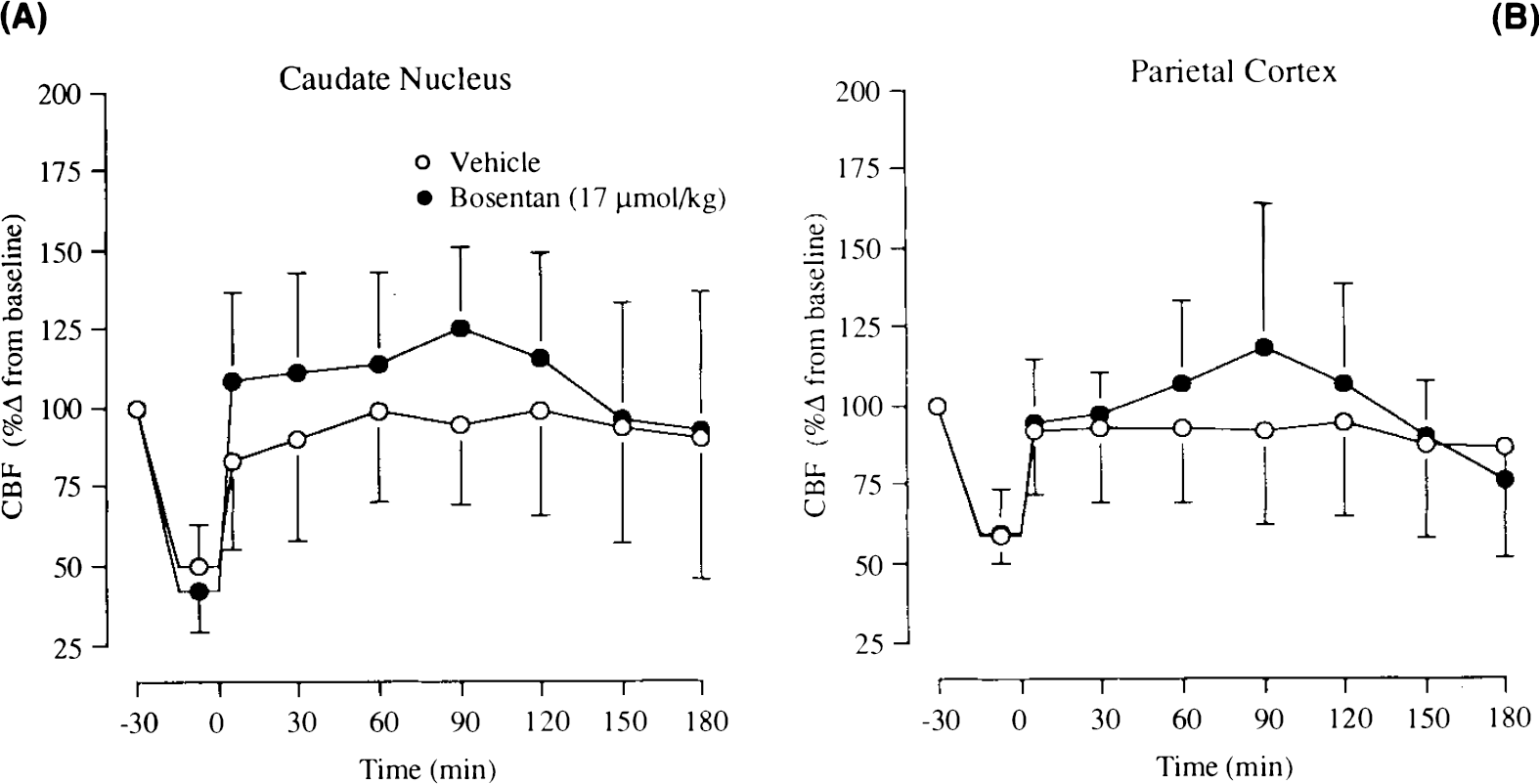
In the parietal cortex, occlusion of the common carotid arteries resulted in a reduction of CBF [60 ± 14% of preocclusion baseline (Bosentan); 59 ± 9% of preocclusion baseline (vehicle)]. CBF was elevated in Bosentan-treated animals (for the 2 h after occlusion), whereas it was reduced by 5 to 13% of preocclusion baseline in the vehicle-treated animals (Fig. 4B). The small increases in CBF were not statistically significant (p > 0.32, unpaired Student's t test).
Spatial Distribution of CBF ([14C]Iodoantipyrine Autoradiography). There was no significant alteration in blood flow in either the vehicle- or the Bosentan-treated animals. There were no obvious increases in blood flow in any of the 35 regions examined (Table 2; Fig. 5).
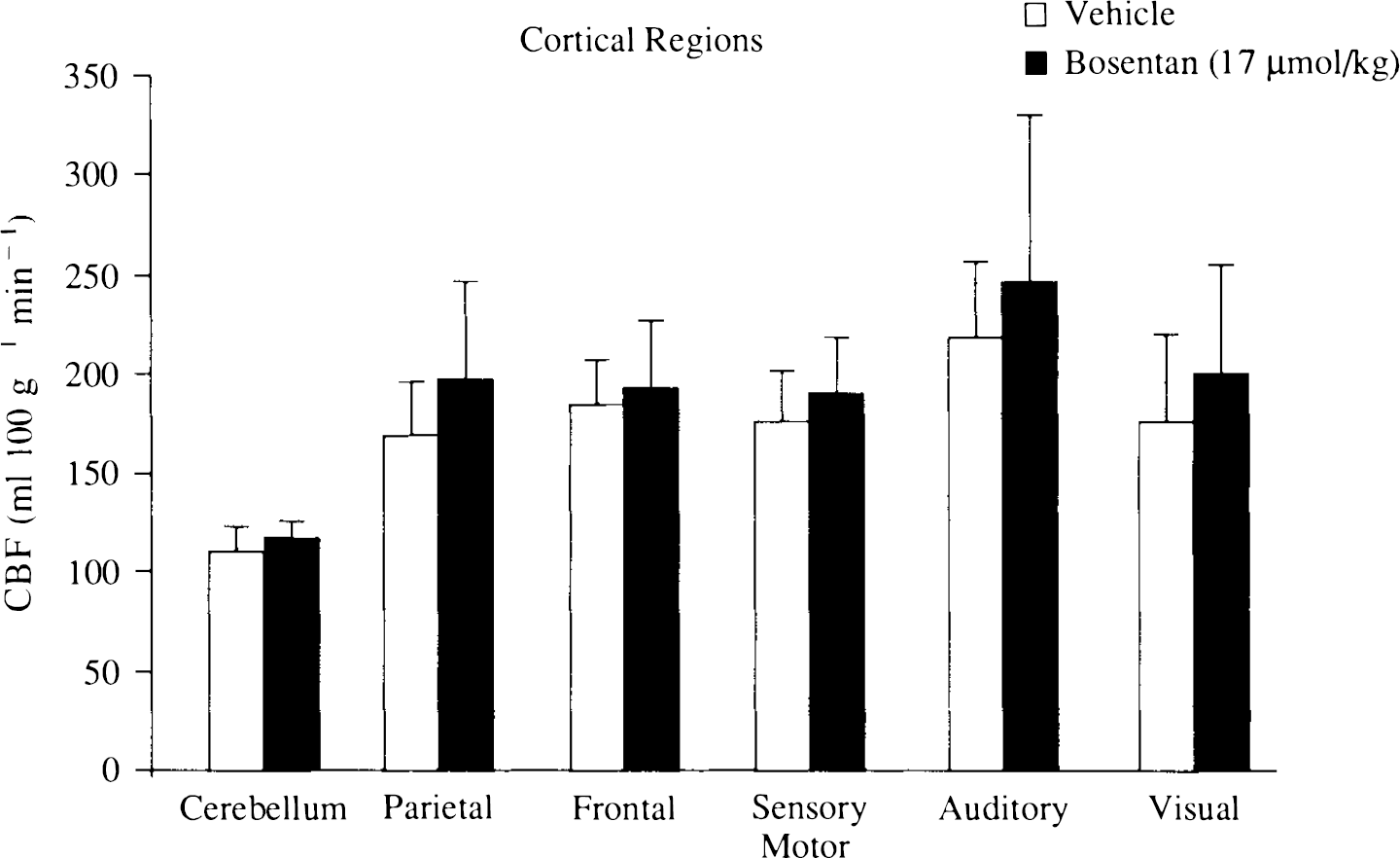
Effect of Bosentan (17 μmol/kg) on cerebral blood flow in the cerebral cortex of animals subjected to transient bilateral common carotid occlusion. CBF was measured using [14C]iodoantipyrine autoradiography at 90 min after the release of the carotid ligatures. Bosentan was administered as an intravenous bolus 10 min before the ligation of the common carotid arteries. Results are expressed as mean ± SD (ml 100 g−1 min−1); n = 9 in each group).
DISCUSSION
The phenomenon of postischaemic hypoperfusion has been investigated after transient focal and global ischaemia in the rat and the cat (Hossmann et al., 1973; Snyder et al., 1975; Miller et al., 1980; Pulsinelli et al., 1982; Kagstrom et al., 1983). A number of factors can be involved in the maintenance of hypoperfusion, such as increased vascular tone or microvascular plugging (Wade et al., 1975; Grogaard et al., 1989; Moskowitz et al., 1990; Wahl and Schilling, 1993; Dirnagl et al., 1994). This investigation failed to demonstrate any increases in the endothelin-mediated vascular tone during postischaemic hypoperfusion. The vascular mechanisms involved in postischaemic hypoperfusion have been investigated by using the calcium channel antagonist nimodipine (Kazda et al., 1982; Steen et al., 1984). The ability of nimodipine to alleviate hypoperfusion indicates an increase in vascular tone of cerebral vessels during the postischaemic period (Kazda et al., 1982). This attenuation of postischaemic hypoperfusion has been associated with increases in neuronal survival and neurologic outcome in experimental models of global ischaemia (Steen et al., 1984).
The endothelins have been implicated in cerebrovascular pathology (Rubanyi and Polokoff, 1994). Increases in tissue endothelin immunoreactivity have been measured after global and focal ischaemia (Duverger et al., 1992; Viossat et al., 1993; Barone et al., 1994). In focal cerebral ischaemia, tissue endothelin immunoreactivity was markedly increased from 24 to 72 h (Duverger et al., 1992; Viossat et al., 1993). Increases in plasma and CSF endothelin levels have been reported in subarachnoid haemorrhage, stroke, and global ischaemia at shorter time points (Giuffrida et al., 1990; Giuffrida and Malatino, 1992; Suzuki et al., 1992; Willette et al., 1992; Ziv et al., 1992; Barone et al., 1994). This investigation sought to demonstrate the role of endothelins in the acute phase after transient global ischaemia; the effect of endothelin antagonists in the rat model of global ischaemia at later time points is unknown. The endothelin ETA-receptor antagonists BQ123 and SB209670 have demonstrated a decrease in neuronal damage in hippocampal CA1 neurones in a gerbil model of global ischaemia (Feuerstein et al., 1994; Ohlstein et al., 1994). The mechanism underlying this process is unclear, but an increase in cerebral blood flow and direct neuronal effects of the endothelin-receptor antagonists are possibilities. This investigation did not attempt to investigate the effect of Bosentan on neuronal damage after transient global ischaemia in the acute phase. Recently Yamashita and colleagues (1994) demonstrated an increase in endothelin ETB receptors in CA1 pyramidal neurones after transient fore-brain ischaemia. The increase in endothelin receptors is associated with microglia, and the role of endothelins in the progression of neuronal cell death after forebrain ischaemia is unclear.
The vasoconstrictor actions of endothelins are mediated primarily via endothelin ETA receptors, although constrictor ETB receptors have been reported (Rubanyi and Polokoff, 1994; Seo et al., 1994). Recent data have indicated the presence of ETB receptors in the cerebral resistance arterioles that mediate vasodilatation (Kobari et al., 1994; Patel et al., unpublished observations). Bosentan is a combined ETA/ETB-receptor antagonist, and the absence of any increases in CBF during postischaemic hypoperfusion could be the result of the inhibition of the dilator ETB receptor and the constrictor ETA receptor (Clozel et al., 1994). The increases in endothelin-mediated tone in cerebral resistance arterioles after focal cerebral ischaemia in the cat indicates a regulatory role for the endothelin peptides during the postischaemic phase (Patel et al., 1995). However, Bosentan failed to increase CBF or reduce the volume of damaged tissue in focal ischaemia in the rat (McAuley et al., 1994). Similarly, the inability of Bosentan to increase CBF after transient global ischaemia indicates that the contribution of endothelins in mediating postischaemic vascular tone may be less pronounced in the rat. The reasons for such differences are not immediately clear, but differences in distribution or density of endothelin-receptor subtypes in the rat and cat are possibilities (Feger et al., 1994; Kobari et al., 1994). No clear evidence suggests that differences in endothelin receptors may exist between these two species. Other possible explanations include the differences in the cerebrovascular anatomy of the rat and cat (Coyle and Jokelainen, 1982; Mchedlishvili and Kuridze, 1984). The lower density of connections between collateral vessels in the cerebral cortex of the rat is one possible explanation for the failure of Bosentan to increase CBF after transient global ischaemia (Coyle and Jokelainen, 1982; Mchedlishvili and Kuridze, 1984).
The source of endothelins after cerebral ischaemia is unclear. Endothelin-receptor binding sites and messenger RNA (mRNA) have been described on neuronal and glial cells (MacCumber et al., 1990; Ehrenreich et al., 1991). There are a number of possible triggers for the synthesis of endothelin peptides [e.g., tissue hypoxia and thrombin (Kurihara et al., 1989)]. Tissue hypoxia during the period of global ischaemia and the pooling of blood in the microvasculature could be triggers for the release or synthesis of endothelins. The role of vascular elements, such as leucocytes and neutrophils, in microvascular obstruction has been investigated (Wahl and Schilling, 1993; Dirnagl et al., 1994). The haemorrhage and the subsequent reinfusion of blood could cause trauma to the cellular contents and increase the ability of platelets to aggregate and increase leukocyte adhesion to vascular endothelium (Grogaard et al., 1989; Dirnagl et al., 1994). The increased trauma to blood cells such as the platelets can result in the release of vasoconstrictor factors or an increase in ET-1 mRNA and thus increase vascular tone (Kurihara et al., 1989). The haemorrhagic hypotension employed to reduce CBF could by itself result in the increase in systemic endothelin release (Zimmerman et al., 1994). An increase in the shear stress on endothelial cells has been shown to stimulate increases in endothelin release (Malek and Izumo, 1992). The transient hyperaemic response immediately after the restoration of CBF may be one of the triggers for increased endothelin release or production. The transition from transient hyperaemia to delayed hypoperfusion is a rapid event (∼5 min). The mechanism mediating the rapid transition is unclear but Moskowitz and colleagues (1990) suggested that there is a loss of dilator vascular tone. Pial cerebral arterioles have perivascular nerves containing dilator peptides, such as calcitonin gene-related peptide (CGRP), and sudden loss of tone leads to the constriction of vessels and reduction in CBF. We have demonstrated an increase in endothelin-mediated vascular tone in vessels after middle cerebral artery occlusion in the cat (Patel et al., 1994). However, the inability of Bosentan to alleviate the hypoperfusion after transient global ischaemia indicates that the endothelins may play a minor role in the maintenance of hypoperfusion.
In a pharmacologic investigation that fails to reveal a significant effect, the issue of adequacy of experimental design is crucial to the interpretation of the results. The dose of Bosentan (17 μmol/kg) used in our investigation has been demonstrated significantly to reduce the pressor effect of exogenous ET-1 in the rat by ∼40 mm Hg (50% effective dose; Clozel et al., 1994). Our own investigations in feline pial arterioles demonstrated that this dose of Bosentan completely inhibits the vasoconstrictor response after the perivascular application of ET-1 (10 nM) (Patel et al., 1994). However, differences in the reactivity of feline pial arterioles and rat intracerebral microvessels to endothelins or endothelin-receptor antagonists cannot be excluded absolutely (Robinson and McCulloch, 1990; Feger et al., 1994; Sagher et al., 1994). The plasma half-life of Bosentan is ∼3 h (Clozel et al., 1994); thus the absence of an effect may not be due to rapid clearance of the drug from the circulation. The issue of the adequacy of experimental design was addressed by post hoc statistical power analysis. In the iodoantipyrine autoradiography investigations, our studies would permit the significant detection of a 40% increase in CBF after treatment with Bosentan. In the hydrogen-clearance studies, a 30% increase in CBF would be detected significantly with our experimental design. The studies were carried out by using two different techniques for measuring CBF, one with excellent spatial resolution ([14C]iodoantipyrine autoradiography) and the other with good temporal resolution (hydrogen clearance), and employing two different models of ischaemia, one severe, associated with marked hypoperfusion (carotid artery occlusion with hypotension), and the other with moderate severity with minimal hypoperfusion (common carotid artery occlusion). The failure of Bosentan to alter acutely the postischaemic hypoperfusion does not rule out the possibility of the endothelins playing a role in global cerebral ischaemia.
Footnotes
Acknowledgment:
The authors thank Dr. M. Clozel (F. Hoffmann-LaRoche, Switzerland) for the gift of Bosentan. T.R.P. is supported by an MRC/CASE studentship.
