Abstract
Cerebral infarction volume after occlusion of a short proximal segment of the middle cerebral artery (MCA) is reported to be different in Wistar compared to Fischer-344 (F344) rats, in both size and variability. Knowledge about the cause of these differences might enable us to explain and perhaps reduce the variation in infarct volume and create a reproducible model of focal cerebral ischemia in the rat. We investigated in Wistar and F344 rats both the effect of occlusion of a long proximal MCA segment on cerebral infarction volume, visualized by magnetic resonance imaging and histology, and the morphology of the major cerebral arteries. Occlusion of a long proximal MCA segment resulted in a striatal and a small cortical infarction in Wistar and a striatal and sizable cortical infarction in F344 rats (as is the case after occlusion of a short proximal MCA segment). In Wistar rats, however, occlusion of a long proximal MCA segment strongly reduced the variability in infarction volume in comparison to occlusion of a small proximal MCA segment. Analysis of the morphology of the major cerebral arteries showed a significantly higher number of proximal side branches of the long proximal MCA segment in Wistar rats than in F344 rats. We conclude that after short-segment proximal MCA occlusion, extreme variability in cerebral infarction volume in Wistar rats compared to F344 rats may be attributable to a significantly greater number of proximal MCA side branches in Wistar rats than F344 rats.
Rat models of focal cerebral ischemia show a substantial variability in infarct size after standardized middle cerebral artery (MCA) occlusion (Robinson et al., 1975; Albanese et al., 1980; Tamura et al., 1981; Coyle, 1982, 1986; Shigeno et al., 1985; Bederson et al., 1986; Chen et al., 1986; Duverger and MacKenzie, 1988). Duverger and MacKenzie (1988) found that there is interstrain variation in the volume of cerebral infarction after standardized MCA occlusion in rats. These authors found that, in general, occlusion of a short proximal segment of the MCA in Wistar rats resulted in a striatal infarction and a small cortical infarction, while in Fischer-344 (F344) rats both the striatum and a large part of the cortex were infarcted. The same authors also reported that the variability in infarction size in Wistar rats was extreme in comparison to that in Sprague–Dawley, F344, spontaneously hypertensive (SHR), and stroke prone-SHR (SHRSP) rats. Interstrain variability in cerebral infarction volume after MCA occlusion in rats might be due to differences in the anatomy of the cerebral arterial tree in the different rat strains. For example, variability in distal MCA branch patterns may contribute to variability in infarct size as was shown in Sprague–Dawley rats after distal occlusion of the MCA (Fox et al., 1993). We therefore studied the gross morphology of the anterior (ACA) and posterior (PCA) cerebral arteries and MCA in Wistar and F344 rats. Interstrain variability in cerebral infarction volume in rats might also be due to differences in MCA occlusion procedures. For example, the length and location of the occluded MCA segment determine the percentage of Sprague–Dawley rats that show cerebral infarction (Bederson et al., 1986). Thus we investigated in Wistar and F344 rats the effect on cerebral infarction volume after occlusion of a long proximal MCA segment.
It was hoped that information about mechanisms that might cause differences in the volume of cerebral infarction of Wistar and of F344 rats after proximal MCA occlusion might enable us eventually to design a rat model of focal cerebral ischemia with minimal variability.
MATERIALS AND METHODS
Animals
Male Wistar (outbred: WU:U) and F344 (inbred: Broekman-Iffa Credo, Someren, The Netherlands) rats (250–300 g) ∼3 months old were used. Rats had free access to standard laboratory chow and water both before and after surgical intervention.
Analysis of cerebral arterial morphology
For analysis of the gross morphology of the six major cerebral arteries (ACA, MCA, and PCA bilaterally) casts were made of the cerebral arterial tree of Wistar (n = 15) and F344 (n = 10) rats by injection of the plastic Araldite F (Ciba-Geigy, Arnhem, The Netherlands), as described by Van der Zwan and Hillen (1990). After injection with 500 IU of heparin in 1 ml of saline to prevent blood clotting, the rats were deeply anesthetized with 90 mg/kg i.p. pentobarbital. A blunt metal 14-G cannula was placed in the ascending aorta via the left ventricle and secured with a ligature. After flushing with 400 ml of saline to remove all blood, the descending aorta was clamped at the level of the diaphragm. The cannula was connected to a bottle containing 0.2% glutaraldehyde in 0.1333 M phosphate buffer with 1% glucose (pH 7.38, 360 mOsm) according to Hanstede and Gerrits (1982). The fixative was perfused by connecting the bottle with an electrical air pump with a needle valve for fine regulation. Excess fixative was removed by flushing with saline again, and the colored Araldite F mixture was perfused. After decapitation, all extracranial soft tissues of the head were removed and the skull was suspended for 1 week in a 50% potassium hydroxide solution at room temperature. Thereafter the cast was thoroughly rinsed to remove adhering brain tissue. Scanning electron microscopy had shown (Van der Zwan and Hillen, 1990) that, using this method of Araldite F injection, even capillaries with a diameter between 5 and 10 μm show filling with the plastic. Hence, with this method effects of vessel smooth muscle contraction that could produce variability in luminal width or prevent vessels from filling are not likely to cause or obscure interstrain differences in cerebral arterial morphology, so that no dilating agents were needed. A macroscope was used to measure diameters of the main trunks of AC A, MCA, and PCA in both hemispheres (magnification, 85×); also, possible interstrain variations in branching pattern were checked for (magnification, 5.8×). The diameters of ACA, MCA, and PCA were measured just distal from their origin in a segment (length, ∼250–500 μm) in which the vessel diameter reached a constant maximum value. The resolution of the method used for measurements of vessel diameter was ±12 μm. Proximal (small-caliber) side branches of the MCA were counted in the segment from the origin of the MCA to the first bifurcation of the main trunk of the MCA. This was done by focusing along the lumen of the MCA so no hidden side branches could be missed. The length of this long proximal MCA segment was also measured. The first bifurcation of the MCA was just distal to the place where the MCA crosses the inferior cerebral vein. This means that these proximal small-caliber side branches of the MCA include the (lenticulo) striate arteries.
MCA occlusion
For the MCA occlusion experiments (Wistar, n = 6; F344, n = 7) anesthesia was induced with 3% isoflurane in a mixture of 70% N2O and 30% O2. Following intubation the rats were mechanically ventilated (Harvard Rodent Ventilator, Model 683) with 1.5% isoflurane in 70% N2O/30% O2. The Paco2 and Pao2 (Ciba-Corning 288 Blood Gas System) were kept within the range of 32–40 and 95–150 mm Hg, respectively. Body temperature was measured with a rectal probe and kept at 37 ± 0.5°C with a negative feedback system (Harvard Homeothermic Blanket Control Unit). The MCA was occluded by a modification of the technique of Tamura et al. (1981). With the animal placed in the lateral position in a stereotaxic apparatus, a standardized craniotomy was made (displaying the long proximal MCA segment) and the dura was opened. Bipolar thermocoagulation was used to occlude a long proximal segment of the MCA, starting at the place where the MCA originates from the internal carotid artery and ending at the place where the MCA crosses the inferior cerebral vein. The anatomical location of this crossing appears not to vary for either strain, with no obvious interstrain difference. After occlusion, the main trunk of the MCA was severed. Blood pressure was not monitored because wounds were sutured quickly directly after MCA occlusion and the animal recovered from anesthesia shortly thereafter. Parallel experiments performed in our laboratory under identical experimental conditions showed no significant difference in blood pressure between Wistar and F344 rats.
Assessment of infarction size
Between 48 and 72 h after MCA occlusion (no rats died during or after surgery) the rats were anesthetized with intraperitoneally injected 0.25 mg/100 g diazepam and intramuscularly injected 0.05 ml/100 g of 10 mg fluanisone and 0.315 mg fentanyl citrate/ml. T2-weighted magnetic resonance imaging (MRI) of the brain was then performed. This schedule was chosen because the increase in T2 relaxation of ischemic tissue is maximal and nearly constant between 24 and 72 h after the onset of cerebral ischemia (Knight et al., 1991; Verheul et al., 1992), and the correlation with the volume of cerebral infarction is high (Berry et al., 1988; Barone et al., 1991). Spin-echo images were acquired on a SISCO 200/400 spectrometer equipped with a 4.7-T magnet and Oxford actively shielded gradients (18 mT/m). A sagittal scout image was used for positioning of the transverse multislice images. Images were obtained using an Alderman–Grant type RF coil (φ, 5 cm). T2-weighted images were acquired with a repetition time of 2.7 s and an echo delay of 60 ms. Two excitations per phase-encoding step were averaged. The field of view was 5 × 5 cm and the matrix size 256 × 128, yielding an in-plane resolution of 0.08 mm2. Slice thickness was 1.1 mm, and 14 adjacent slices were made to obtain images from the entire forebrain. The size of the lesion was determined from the multislice images using NMRi image analysis software (New Methods Research Inc., Syracuse, NY, USA) on the basis of increased signal intensity (hyperintensity). The threshold of segmentation was set manually at a level at which no hyperintense pixels above the threshold were found contralaterally. The total volume of the lesion in one rat brain could be calculated by multiplying the area of hyperintensity in the slices by the slice thickness.
Directly after T2-weighted brain images were made, rats were injected with 90 mg/kg pentobarbital i.p. Following thoracotomy, a blunt metal cannula was placed in the ascending aorta via the left ventricle and secured. The right atrium was incised, the abdominal aorta was clamped, and the animal was perfused with 100–150 ml of saline and then with 200 ml of fixative (4% formalin in a 0.1 M phosphate buffer). The animals were decapitated, and their brains were placed in the same fixative for at least 1 week. The entire brain was embedded in paraffin and coronal sections, 10 μm thick, were cut throughout the rostrocaudal extent of the brain. Every 25th coronal section was selected, stained with a conventional hematoxylin/eosin staining method, and mounted on slides under coverslips. A computerized Image Processing System, IBAS (Kontron, Munich, Germany), was used to measure the area of infarction in all sections, both in absolute numbers and as a percentage of the total slice area. The total volume of infarction was determined by integration of the infarction areas of the sections and the distance between them (250 μm).
Statistics and calculations
Left-right differences in the diameter of corresponding cerebral arteries within both rat strains were compared by analysis of variance (ANOVA). Interstrain differences in the diameter of the ACA, MCA, or PCA, the length of the long proximal MCA segment, and the volume of cerebral infarction, as measured by both planimetry and the volume of hyperintensity in T2-weighted MRI, were also compared by ANOVA. Statistical analysis of interstrain differences in the number of proximal side branches of the MCA was done with the Mann–Whitney U test. Differences were considered significant atp < 0.05.
RESULTS
Analysis of cerebral arterial morphology
Figure 1 shows examples of an Araldite F cast of the cerebral arterial tree in a Wistar and a F344 rat. No significant left–right differences in the diameters of ACA, MCA, and PCA were found in either Wistar (n = 15) or F344 (n = 9) rats (data not shown). Also, no significant differences were found between the two strains for the diameters of corresponding cerebral arteries. Vessel diameters (represented as mean ± SD) were as follows: ACA, 0.29 ± 0.03 mm in Wistar and 0.30 ± 0.02 mm in F344; MCA, 0.32 ± 0.02 mm in Wistar and 0.31 ± 0.01 mm in F344; and PCA, 0.29 ± 0.03 mm in Wistar and 0.28 ± 0.03 mm in F344 (Fig. 2). The mean ratio of the diameter of the MCA, divided by the sum of the diameters of the MCA, PCA, and ACA, also did not differ significantly between the two strains, namely, 0.37 ± 0.02 mm in Wistar rats and 0.36 ± 0.01 mm in F344 rats. In the long proximal MCA traject (from the origin out of the internal carotid artery to the first diverging branching of the MCA), we found, however, a significantly (p = 0.004; Mann–Whitney U test) higher number of (small-caliber) side branches in the Wistar rats than in the F344 rats, namely, 6.3 ± 1.7 (mean ± SD) in Wistar and 3.7 ± 1.9 in F344 rats (Fig. 2). The length of this long proximal MCA segment was not significantly different between strains, namely, 9.3 ± 1.1 mm in Wistar rats and 9.0 ± 1.0 mm in F344 rats.
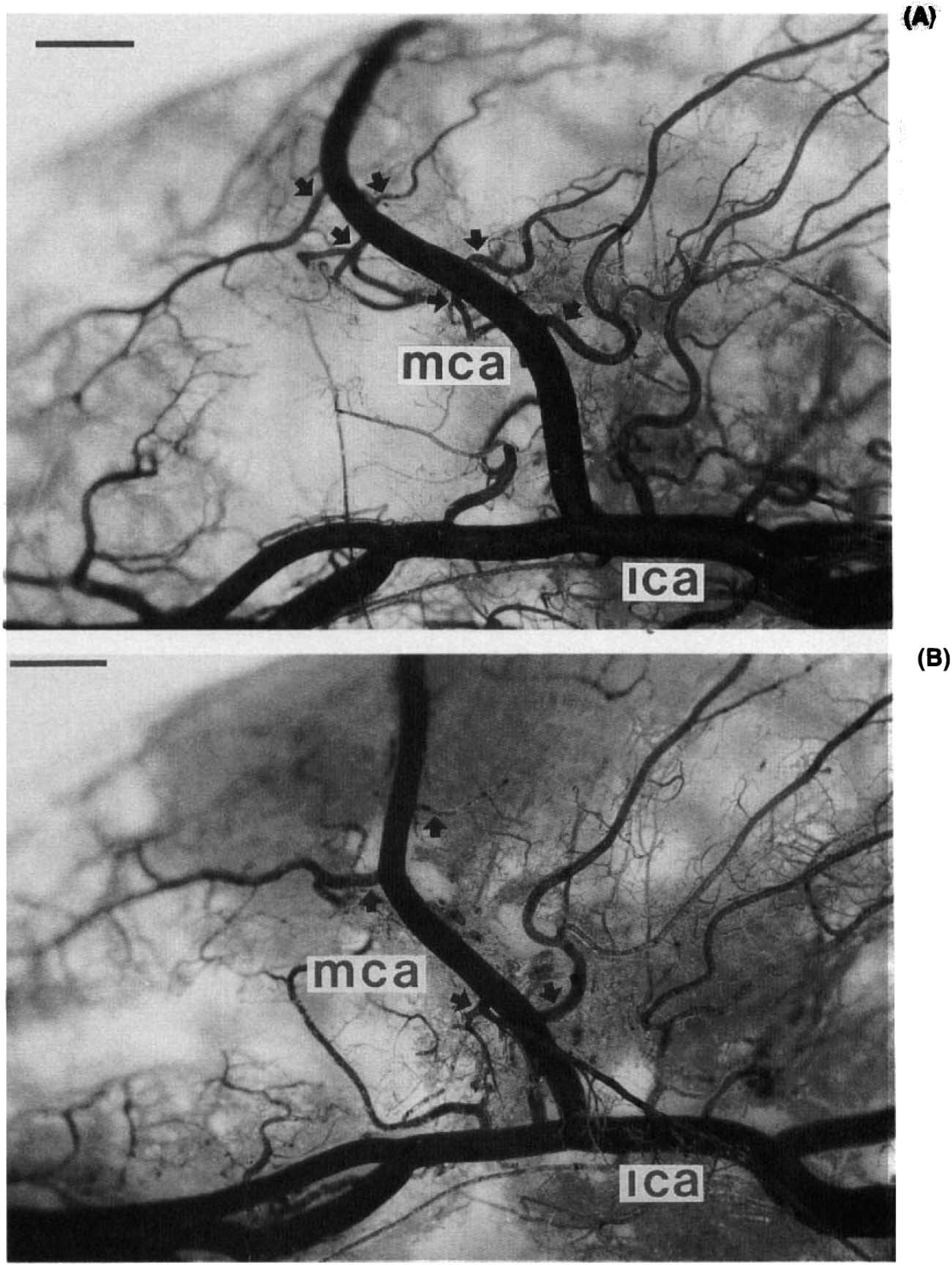
Representative casts of the arterial cerebral circulation (ventral view) of
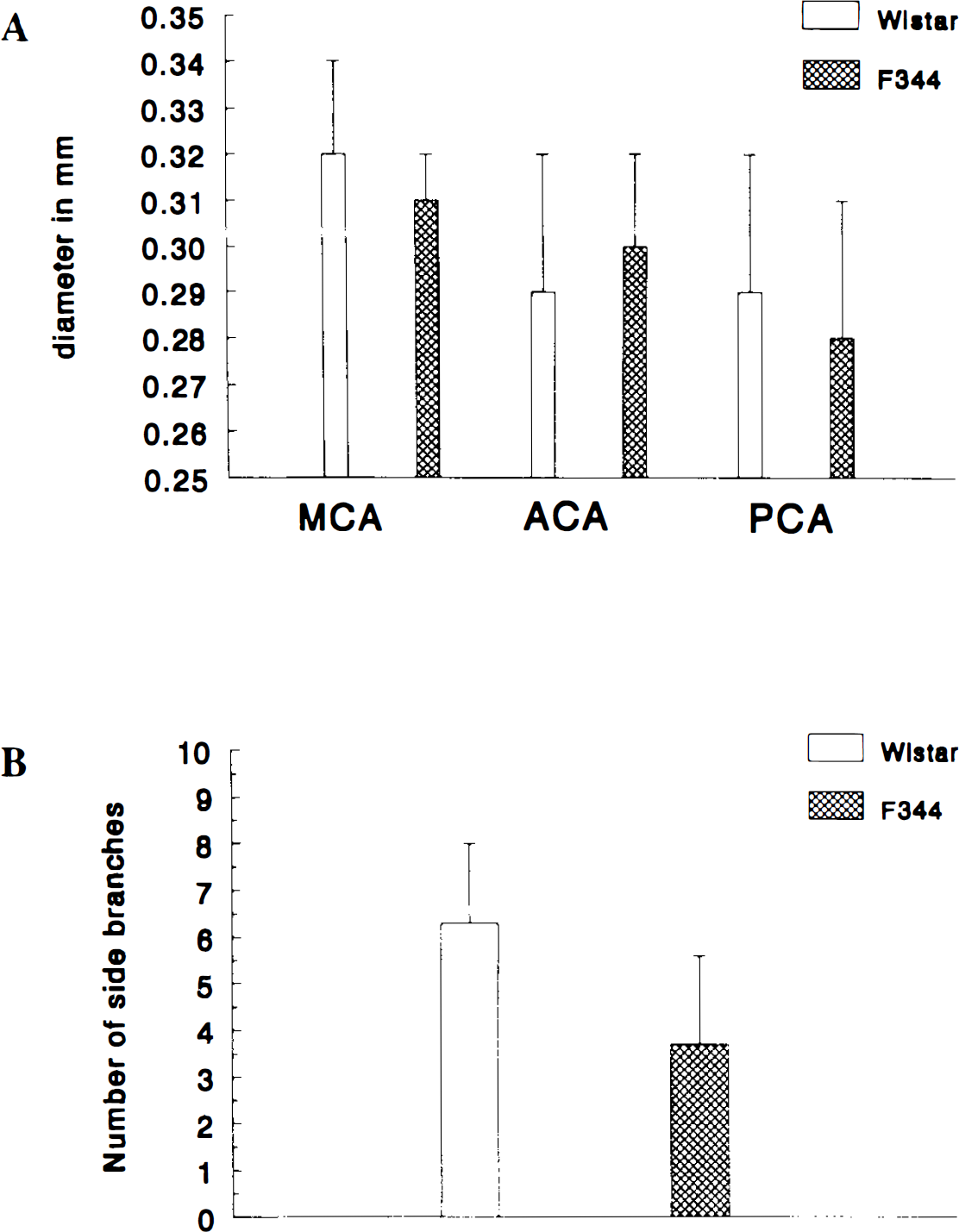
Quantification of infarction size
The anatomical location of the brain lesion measured as hyperintensity in T2-weighted imaging differed between the two strains: in Wistar rats the striatum was invariably involved, but only one rat also had a sizable cortical component of hyperintensity. In F344 rats not only the striatum but also a large part of the cortex invariably showed hyperintensity (Fig. 3). The mean volume (±SD) of hyperintensity in T2-weighted imaging in F344 rats was significantly (ANOVA, p = 0.002) greater (268.8 ± 85.5 mm3; n = 7) than the volume in Wistar rats (123.1 ± 15.6 mm3; n = 6) (Table 1).
Hemispheric brain lesion volume in Wistar and F344 rats: T2-weighted MRI and histology
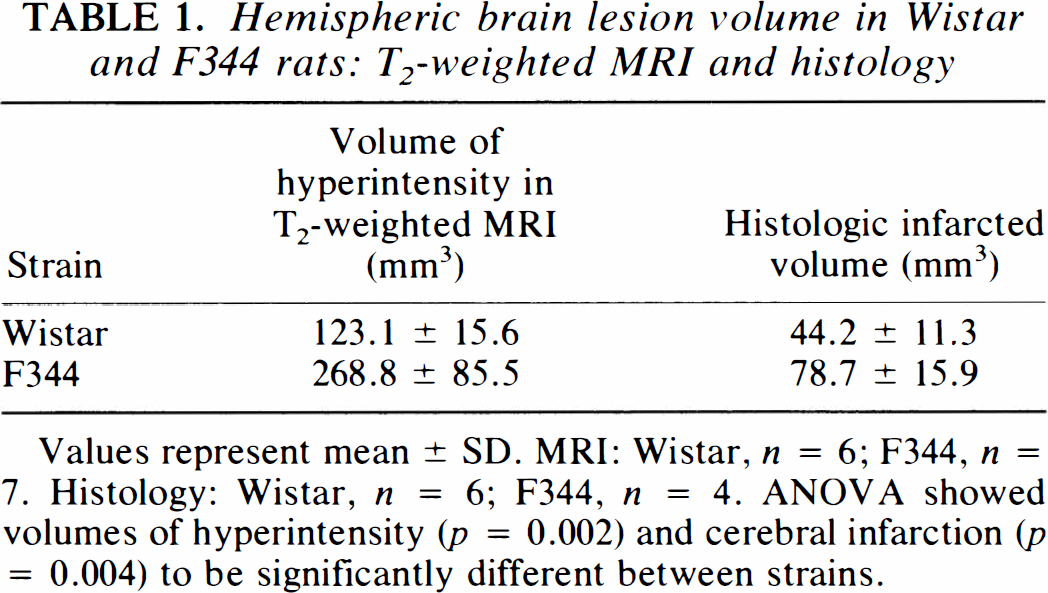
Values represent mean ± SD. MRI: Wistar, n = 6; F344, n = 7. Histology: Wistar, n = 6; F344, n = 4. ANOVA showed volumes of hyperintensity (p = 0.002) and cerebral infarction (p = 0.004) to be significantly different between strains.
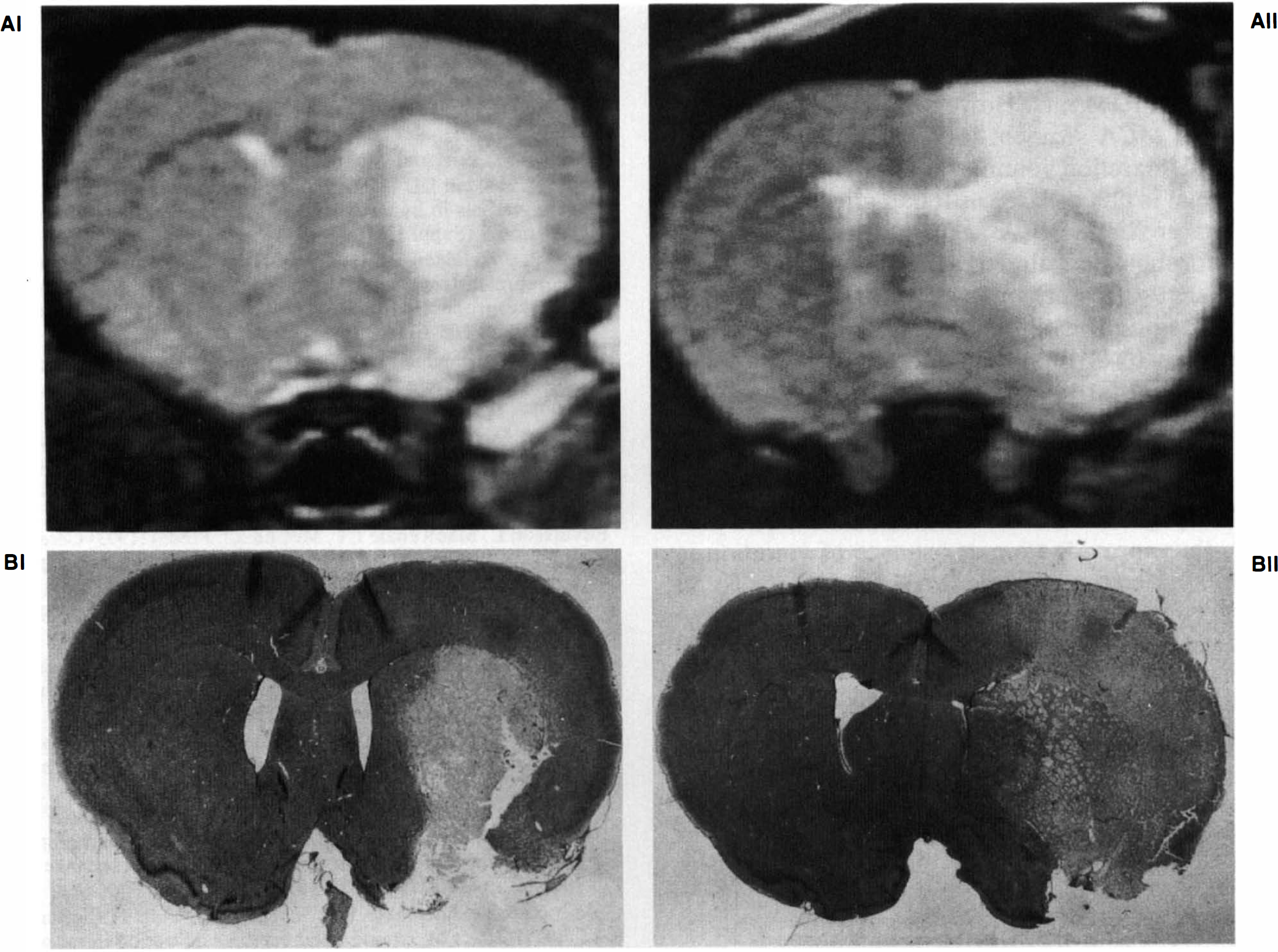
We also measured the volume of cerebral infarction in both rat strains by planimetry on brain sections stained with hematoxylin and eosin. The borders of the infarction were sharply delineated. The anatomical boundaries of the infarctions in the Wistar and the F344 rats were roughly the same as the location of the T2-weighted hyperintensity in the two strains (Fig. 3). Cerebral infarction volumes were significantly different (ANOVA, p = 0.004) between strains: 44.2 ± 11.3 mm3 in Wistar and 78.7 ± 15.9 mm3 in F344 rats (mean ± SD; see Tables 1 and 2).
Histologically assessed hemispheric volume of infarction in Wistar and F344 rats

Values represent mean ± SD. Wistar, n = 6; F344, n = 4. ANOVA showed volumes of cerebral infarction to be significantly different (p = 0.004) between strains.
DISCUSSION
The study aimed to gain information about probable causes of differences between Wistar and F344 rats regarding size and variability of cerebral infarction volume after proximal MCA occlusion. Knowledge of these mechanisms might enable us to reduce the variation in infarct size and create a reproducible rat model of focal cerebral ischemia. Knowledge of the cerebrovascular anatomy in the species under study is essential so that only appropriate methods will be used to study cerebral ischemia. Methodological problems may have been the reason for the contradictory and discrepant conclusions reached in a number of studies with experimental animals (Edvinsson et al., 1993).
We investigated the effect on cerebral infarction volume after occlusion of a long proximal MCA traject in Wistar rats and F344 rats. We hypothesized that more branches would be prevented from retrograde filling through leptomeningeal collaterals in a long MCA segment occlusion than in a short MCA segment occlusion. Thus, branches distal to the short proximal segment occluded by Duverger and MacKenzie (1988) but proximal to where the MCA crosses the inferior cerebral vein, the end point of our long segment occlusion, would be prevented from filling and perfusing the distal MCA stump (Symon, 1960; Coyle, 1993). This hypothesis would explain why occlusion of a small proximal MCA segment (as is frequently used in models of focal cerebral ischemia) rarely, if ever, results in the development of a cortical infarct in Wistar rats. In F344, occlusion of a short proximal MCA segment is sufficient to produce a sizable and consistent infarction that involves the striatum and a large part of the cerebral cortex (Duverger and MacKenzie, 1988). It was of interest, however, to find whether occlusion of a long proximal MCA segment would further decrease variability in infarction volume in the F344 rats. Comparing the cerebral infarction volume in Wistar and F344 rats in our experiments with that found by Duverger and MacKenzie (1988), the percentage of histologically assessed cerebral infarction of total hemispheric volume for Wistar and F344 was almost the same after both types of MCA occlusion [15% in Wistar and 26% in F344 (Duverger and MacKenzie, 1988), versus 15% in Wistar and 25% in F344 (our experiments)]. The absolute volumes of cerebral infarction and of total hemispheric volume, however, differ between the two models [volume of cerebral infarction: 62.2 ± 10.9 mm3 in Wistar and 106.2 ± 7.4 mm3 in F344 (mean ± SEM) (Duverger and MacKenzie, 1988) versus 44.2 ± 11.3 mm3 in Wistar and 78.7 ± 15.9 mm3 in F344 (mean ± SD) (our experiments)]. Because the anatomical boundaries of the average cerebral infarction volume in both strains were comparable in the two studies (as were sex, age, and weight of the rats), these differences are most likely caused by differences in shrinkage during the different methods of histologic fixation of the brains. Differences in the experimental conditions used by Duverger and MacKenzie (1988) and those in our experiments, e.g., genetic composition of the rats, anesthetic used (halothane versus isoflurane), and the fact that the craniotomy we made was somewhat larger, could also affect cerebral infarction volume. The volumes of hyperintensity in the T2-weighted images were much greater than the volumes of infarction measured by planimetry, first, because of shrinkage of the brain during fixation; second, because hyperintensity in T2-weighted imaging is produced not only by infarcted tissue but also by edema; and third, because an overlap between adjacent slices leads to overestimation of the volume of hyperintensity in T2-weighted MRI. It is noteworthy that we found that the coefficient of variation (%) of cerebral infarction volume in Wistar rats after occlusion of a long proximal MCA segment was almost half as large as the coefficient of variation previously reported for this strain (Coyle et al., 1984; Duverger and MacKenzie, 1988). The coefficient of variation in cerebral infarction in F344 rats in our experiments and those of Duverger and MacKenzie (1988) is comparable.
Another explanation for interstrain variability in cerebral infarction volume after MCA occlusion in Wistar and F344 rats could be differences in the anatomy of the cerebral arterial tree in the different rat strains. It has been reported that in humans the variability of the diameters of the major cerebral arteries is strongly correlated with the variability in the territorial distribution of these arteries (Van der Zwan et al., 1993). Extrapolation of these results in humans to the rat might be justified because the territories of distribution of the ACA, MCA, and PCA in the rat brain are remarkably similar to those in the human brain (Coyle, 1975, 1978; Yamori et al., 1976). Gross morphology of the ACA, MCA, and PCA in Wistar and F344 rats in our experiments revealed no significant differences in the diameters of corresponding cerebral arteries between the two strains. Extrapolation of the results for humans (Van der Zwan et al., 1993) to the results of our morphological analysis suggests that a comparable variability of the diameters of the major cerebral arteries in the two rat strains would imply that there would also be no obvious difference in territorial flow pattern in this cerebral arterial network between Wistar and F344 that could explain the interstrain difference in cerebral infarction volume after MCA occlusion.
Another explanation for the existence of interstrain differences in cerebral infarction volume after MCA occlusion in rats was brought to our notice by Jacewicz (1992). He suggested that the occurrence of large infarcts after MCA occlusion in a minority of normotensive rat strains could be explained by inadequate (genetically encoded) vascular collateral flow. In his opinion, the gene (or set of genes) governing infarct susceptibility might presumably occur at a variable frequency in normotensive populations of rats (as may also be the case in the outbred Wistar and the inbred F344 rats we used). Coyle and co-workers found that the mean diameter of anastomoses between branches of the MCA, ACA, and PCA was significantly smaller in SHRSP than in Wistar Kyoto (WKY) rats, thereby affecting the adequacy of this dorsal collateral circulation after MCA occlusion and thus causing the large difference in cerebral infarction pattern between the strains (Coyle, 1982, 1986, 1987; Coyle and Jokelainen, 1983; Coyle and Heistad, 1986). Because our analysis of the gross morphology of the major cerebral arteries in Wistar and F344 rats did not include the leptomeningeal anastomoses, we cannot exclude the possibility that differences in mean internal diameter of the MCA-ACA anastomoses or the number of these anastomoses in these strains may determine, through a difference in adequacy of collateral blood flow after MCA occlusion, the differential outcome in cerebral infarction volume.
It can be concluded from our results that extension of the proximal segment of the MCA that is occluded has no effect on the interstrain difference in size of cerebral infarction volume between Wistar and F344 rats. However, occlusion of a long proximal MCA segment in Wistar rats results in a cerebral infarction volume with a coefficient of variation that is half as large as found after occlusion of a short proximal MCA segment. We suggest that our morphological finding, that Wistar rats had a significantly greater number of proximal (small caliber) side branches in the long proximal MCA segment than F344 rats, may be responsible for this diminished variability in infarction volume in Wistar rats. The chance that one or more proximal small-caliber side branches remain patent if only part of this long proximal MCA segment is occluded is clearly greater in Wistar than in F344 rats and may, thus, provide an extra source of variation in cerebral infarction volume in Wistar rats.
Footnotes
Acknowledgment:
We are grateful to Mrs. Ria Matt hijssen of the Department of Functional Anatomy for assistance with the histological preparations and to Mr. J. H. G. Hoekman of the Laboratory of Pathology, National Institute of Public Health and Environmental Protection, Bilthoven, The Netherlands, for assistance with the IBAS system. Part of this research was carried out at the Netherlands in vivo NMR facility at the Bijvoet Center for Biomolecular Research, which is supported by the Netherlands Organization for Scientific Research (NWO).
