Abstract
To investigate the appearance of apoptosis in short-duration focal ischemia, the authors induced left middle cerebral artery (MCA) occlusion in male rats by insertion of an intraluminal suture. The total number of apoptotic cells was determined by hematoxylin–eosin staining and TUNEL labeling and confirmed by gel electrophoresis. The data indicate that the total number of apoptotic cells increased with ischemia duration (P = 0.0006), with most apoptotic cells located in the striatum of the ischemic hemisphere. As the duration of ischemia lengthened, necrosis became more prevalent and apoptosis receded to the periphery of the infarct. Using iodo[14C]-antipyrine to correlate the distribution of apoptosis to regional CBF (rCBF), the authors found that rCBF in the ischemic dorsolateral striatum was compatible with penumbra flow and significantly lower than the ventromedial striatum and frontoparietal cortex. This difference disappeared after 45 minutes of reperfusion. The authors conclude that focal ischemia of short duration results in changes compatible with apoptosis in regions of low rCBF, and this can occur without necrosis. This model is relevant to transient ischemic attack in the human and may suggest that, in addition to being a harbinger of stroke, transient ischemic attacks may cause histopathologic changes not yet clinically detectable.
A substantial body of scientific literature has addressed the role of apoptosis in cerebral ischemia in both global and focal models. The global ischemia model mimics cardiac arrest by temporarily restricting blood flow to the entire brain, whereas the focal model is reminiscent of a localized occlusion in the brain. Although apoptosis occurs in vulnerable areas of the brain, such as the CA1 region of the hippocampus (Pulisinelli and Buchan, 1988), in global ischemia this model does not allow for investigation of the role of apoptosis in the ischemic penumbra (Hakim, 1987). There is increasing molecular and biochemical evidence that supports the idea that apoptotic events may be occurring in focal ischemia models (MacManus et al., 1994; Li et al., 1995a,c,d; Krajewski et al., 1995; Renolleau et al., 1998).
Although many studies have examined apoptosis after relatively severe focal ischemia, where significant necrosis and infarction are also present, a significantly larger percentage of apoptotic cells are more likely to be found in regions exposed to a more moderate ischemic insult (MacManus et al., 1994; Li et al., 1995a, b ; Bonfoco et al., 1995; Linnik et al., 1995; States et al., 1996; Gillardon et al., 1996; Du et al., 1996). We wished to explore the occurrence of apoptosis in focal ischemia of moderate severity and correlate it with regional cerebral blood flow (rCBF). Our goal was to study apoptosis in a laboratory model that mimics transient cerebral ischemia in the human. A transient ischemic attack (TIA) is accepted as a harbinger for stroke because the stroke risk of patients with TIAs is increased 13- to 16-fold during the first year and approximately 7-fold during the subsequent 5 years after the TIA (American Heart Association, 1990). In general, however, the TIA itself is considered harmless, since by definition it leaves no clinical or radiologic sequelae, though CBF falls into the penumbral range in patients undergoing a TIA (Itoh et al., 1988; Powers et al., 1985). Showing apoptosis experimentally with focal ischemia durations comparable to TIAs may therefore raise interesting therapeutic dilemmas if an antiapoptotic treatment becomes available, particularly considering that apoptotic cell death develops slowly. In the Cooperative Study of Transient Ischemic Attacks the median duration of carotid distribution of TIAs was 14 minutes (Dyken et al., 1977). We therefore selected experimental ischemic durations of comparable magnitude.
MATERIALS AND METHODS
Experimental design
Studies were performed on adult male Sprague-Dawley rats, weighing between 260 and 330 g, obtained from Charles River Laboratories (St. Constant, Quebec, Canada). All animals exposed to ischemia underwent left middle cerebral artery (MCA) occlusion by insertion of an intraluminal suture. Five experimental groups of animals underwent the following ischemic and reperfusion periods: 5 minutes ischemia followed by 16 h (n = 8), 24 h (n = 8), or 48 h (n = 8) of reperfusion; 10 minutes of ischemia followed by 24 h of reperfusion (n = 14); and 15 minutes of ischemia followed by 24 h (n = 8) of reperfusion. Other rats were sham-operated for 5 minutes (n = 8), 10 minutes (n = 8), and 15 minutes (n = 8) followed by 24 h of reperfusion. Rats that served as sham-operated controls had a 10-mm-long nylon monofilament inserted into the internal carotid artery (ICA) for the indicated duration. This length of nylon monofilament is too short to occlude the MCA.
The location of the apoptotic cells in the left MCA area was determined from frontal to occipital pole by examining 11 levels of brain (n = 5), defined using a rodent brain atlas (Paxinos and Watson, 1997). The number of cells displaying apoptotic bodies was counted within predefined regions.
Regional CBF was examined using iodo[14C]-antipyrine and autoradiography in experimental animals and sham controls immediately after 10 minutes of ischemia, after 45 minutes of reperfusion following 10 minutes of ischemia, and in sham-operated animals.
Surgical preparation
All procedures followed the guidelines established by the Canadian Council on Animal Care (Guide to the Care and the Use of Experimental Animals) and were approved by the University of Ottawa Animal Care Committee. All animals were given acetaminophen (Tylenol [McNeil Consumer Products Co., Ft. Washington, PA, U.S.A.) in their drinking water 2 days before surgery and underwent fast overnight immediately before surgery. Anesthesia was induced with 4.0% halothane in a mixture of 70% nitrous oxide and 30% oxygen. The surgery was performed in aseptic conditions. Rats were orally intubated and mechanically ventilated with 1% halothane in nitrous oxide and oxygen during the surgical procedure. The tail artery was cannulated for monitoring of arterial blood pressure, blood gases, and blood glucose. Blood samples were taken before occlusion, during ischemia, and 30 minutes after removal of the thread. Rectal temperature was maintained at 37°C by a rectal probe connected to a homeothermic feedback blanket (Ealing Scientific, Quebec, Canada). Cranial temperatures have been reported to remain within 1°C of rectal temperature (Minamisawa et al., 1990; Belayev et al., 1997; Zhao et al., 1997).
Rats were mounted in a stereotaxic frame, and a small burr hole was drilled over the left frontoparietal cortex (FPC; bregma 0.0 mm, lateral 4.0 mm, depth 2.0 mm). A 0.1-mm-diameter platinum electrode was implanted and fixed with dental cement in the left FPC for monitoring of rCBF via the hydrogen-clearance method. Intraoperative hydrogen-clearance rate was used to qualitatively monitor the success in producing cerebral ischemia during suture insertion and maintenance. A platinum wire reference electrode was inserted under the auricle of the left ear, and both electrodes were connected to a direct current amplifier and chart recorder (WindoGraf Recorder; Gould Instrument Systems, Inc., Cleveland, OH, U.S.A.).
The ischemia model
To produce ischemia in the area of the MCA, we used a previously described method (Matsushima et al., 1996), which is a modification of those of Koizumi et al. (1986), Zea Longa et al. (1989), and Nagasawa and Kogure (1989). Briefly, under an operating microscope, the left common (CCA), ICA, and external carotid arteries (ECA) were exposed through a midline neck incision and carefully dissected free. The proximal portion of the left ECA and ipsilateral CCA were ligated with 4–0 silk thread, the latter 5 mm proximal to the bifurcation of the ICA. A small incision was made with a 27-gauge needle 3 mm proximal to the bifurcation. Depending on the size of the rat, a 35-mm length of 4–0 or 3–0 nylon monofilament thread, its tip blunted by heat, was introduced into the CCA lumen through a small incision. The left CCA was ligated loosely with a 4–0 silk thread just distal to the point of the thread insertion to prevent bleeding. The nylon thread was gently advanced approximately 18 mm into the ICA lumen from the ICA and the ECA bifurcation. Occlusion was confirmed by rCBF measurement using the hydrogen-clearance technique. Rats whose rCBF during ischemia exceeded 20 mL · 100 g brain–1 · min–1 were eliminated from the study.
The thread was withdrawn after the prescribed duration of ischemia. The suture over the ECA was untied and the neck incision was closed after topical application of bupivacaine hydrogen chloride jelly. Reperfusion was again confirmed using rCBF measurement.
Anesthesia was discontinued, and the rat was extubated when spontaneous breathing resumed. The rat was allowed to recover, with free access to food and water for the predetermined duration of reperfusion.
Histologic examination
At the appropriate time points, rats were anesthetized and then given an intraperitoneal injection of chloral hydrate–pentobarbital. The animals were transcardially perfused with heparinized saline followed by 4% paraformaldehyde. The brains were removed and postfixed in 10% formalin for a week before paraffin embedding. The entire brain was sectioned and 7-μm coronal sections were obtained from each rat and alternate sections were stained with hematoxylin and eosin (H&E) and a molecular biologic–histochemical system (In Situ Cell Death Detection Kit, POD; Boehringer Mannheim Canada, Laval, Quebec, Canada) for staining of DNA fragmentation and apoptotic bodies. The number of cells stained with apoptotic bodies was counted using a light microscope. Histologic features used to identify the apoptotic cells with light microscope analysis (Wyllie et al., 1980) included the recognition of rounded or oval bodies (apoptotic bodies) and typically intense dark purple–blue masses, variable in size, that frequently contain pyknotic chromatin in cells without inflammation. In this kit, terminal deoxynucleotidyl transferase is used to label DNA strand breaks. Incorporated fluorescein is detected by antifluorescein antibody Fab fragments from sheep, conjugated with horseradish peroxidase. After substrate reaction, visualization of labeled incorporated nucleotides was carried out with diaminobenzidine.
The adjacent coronal sections were also stained with cresyl violet to identify necrotic cells. Necrotic cells were identified by light microscopy as cells that did not contain the cresyl violet–stained Nissl bodies and nucleoli.
Gel electrophoresis
Rats were decapitated under halothane anesthesia, and the right cortex, the right striatum, the left cortex, and the left striatum were quickly removed from each animal and stored at −80°C. Five rats (10-minute ischemia followed by 24-h reperfusion) were examined separately. These samples were homogenized on ice and total DNA extracted from samples as previously described (MacManus et al., 1993). Gel electrophoresis for detection of DNA fragmentation was performed.
Cerebral blood flow measurement by autoradiography
Regional CBF of the dorsolateral striatum (DLS), ventromedial striatum (VMS), and FPC in separate groups of rats were measured by means of the iodo[14C]-antipyrine quantitative autoradiographic technique (Sakurada et al., 1978). Rats underwent the same surgery as described previously, and rCBF was measured at the end of 10 minutes of ischemia (n = 8), in rats sham-operated for 10 minutes (n = 5), rats exposed to 10 minutes of ischemia followed by 45 minutes of reperfusion (n = 6), and rats sham-operated for 10 minutes followed by 45 minutes of reperfusion (n = 6).
RESULTS
Physiologic variables
Mean arterial blood pressure, body temperature, arterial blood gases, and blood glucose were generally held within normal limits throughout the experiments (data not shown).
Histologic examination
Apoptosis.
Because the results from TUNEL staining and H&E histologic staining were not significantly different and showed similar trends, all of the following references and figures refer to cells that contain apoptotic bodies as determined by light microscopy after H&E staining.
Figure 1 shows the regional map we used to arbitrarily define the FPC, the DLS, and the VMS. The number of apoptotic cells in brain slices at this level compared with sham increased significantly with the duration of focal ischemia after 5, 10, and 15 minutes of ischemia followed by 24 h of reperfusion (Fig. 2A; P < 0.0001, by ANOVA). No statistically significant relation was observed between the total number of apoptotic cells and the duration of reperfusion (Fig. 2B). Figure 2C shows the DLS had the highest incidence of apoptotic cells (P < 0.0001, by repeated measures ANOVA).
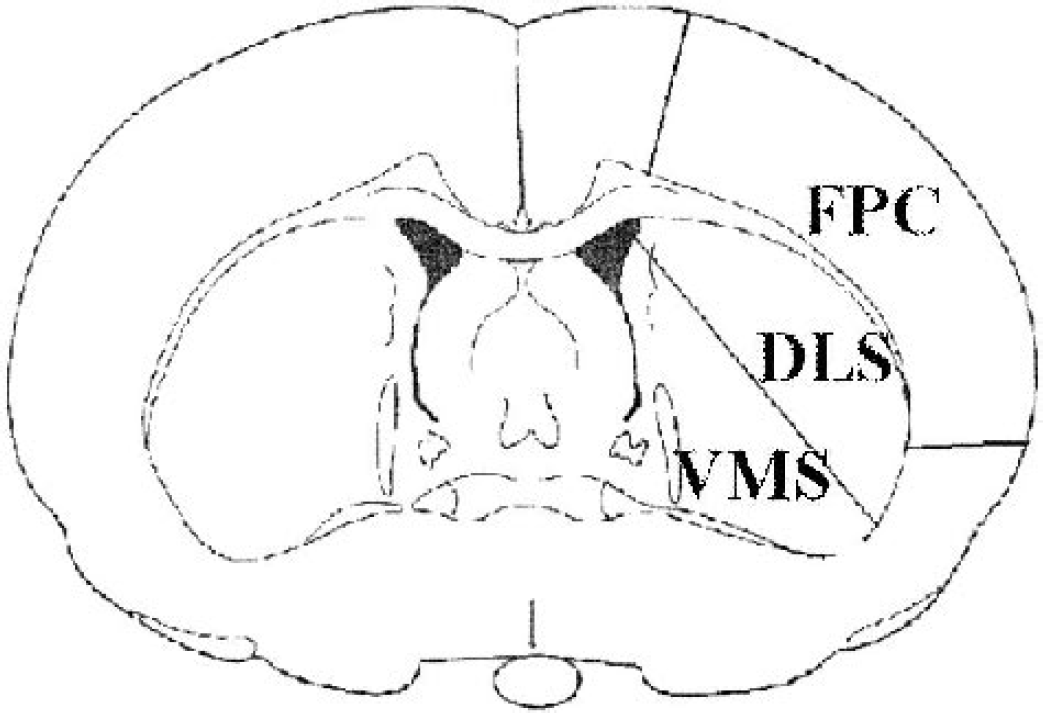
Arbitrarily defined anatomic regions that were used as a template for histopathologic and regional CBF analysis. FPC, frontoparietal cortex; DLS, dorsolateral striatum; VMS, ventromedial striatum.
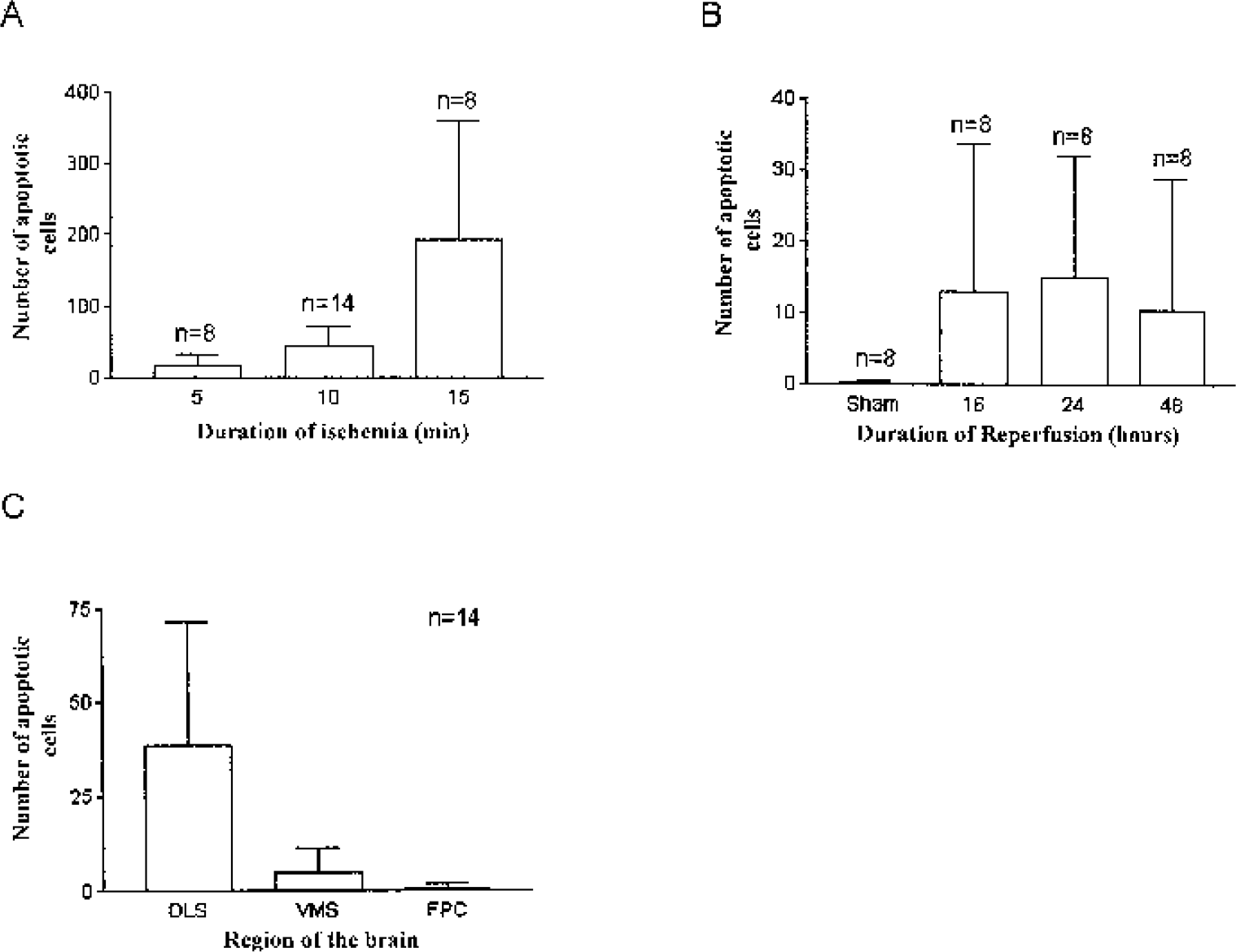
Apoptosis in short-duration focal ischemia.
Figure 3 shows the quantitative distribution of the total number of apoptotic cells in brain slices spanning the length of the hemisphere in 5 of the 14 animals exposed to 10 minutes of ischemia and 24 h of reperfusion. The anatomic levels of the slices are shown below where, in addition, in that particular animal, the distribution of the apoptotic cells (in red) and the gray zones of necrotic changes are also shown.
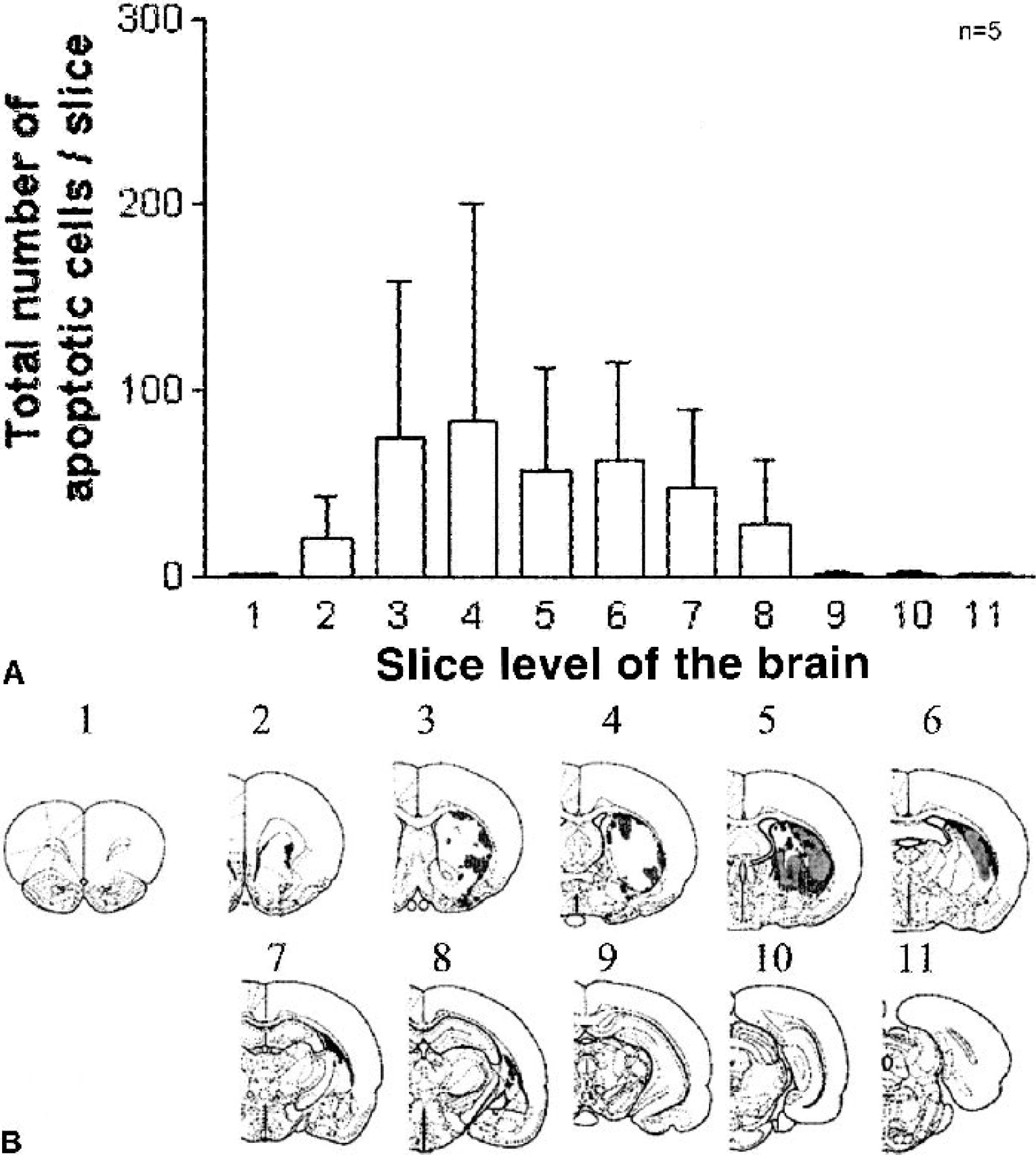
Necrosis.
After 5 minutes of MCA occlusion, when all reperfusion durations were included (n = 24), necrosis was evident in only 2 rats (8.3%) with a mean area of necrosis of 0.57 ± 2.7 mm2. After 10 minutes of focal ischemia followed by 24 h of reperfusion, necrosis was detected in 6 of 14 rats (43%) with an area of necrosis of 3.35 ± 4.99 mm2. Fifteen minutes of focal ischemia always produced injury that included both apoptotic and necrotic cells (16 of 16 rats, 100%) with a mean necrosis area of 7.77 ± 3.81 mm2. Necrosis was always located primarily in the striatum of the ischemic hemisphere.
Gel electrophoresis
The left (ischemic hemisphere) striatum showed DNA laddering indicative of apoptosis, whereas no DNA laddering was noted in the left cortex or any structure on the contralateral hemisphere (data not shown).
Regional cerebral blood flow measurement by autoradiography
Results are shown in Table 1. After 10 minutes of ischemia, the rCBF value of the ischemic DLS (27.37 ± 7.5) was significantly lower than that of the VMS and the FPC in the same hemisphere (P < 0.01, Friedman test). After 45 minutes of reperfusion, the rCBF value of the DLS corrected substantially, and there was no significant difference between the rCBF value of the DLS and the VMS. At the end of 10 minutes of sham surgery, there were no statistically significant differences among the rCBF values of the DLS, the VMS, and the FPC.
Regional cerebral blood flow
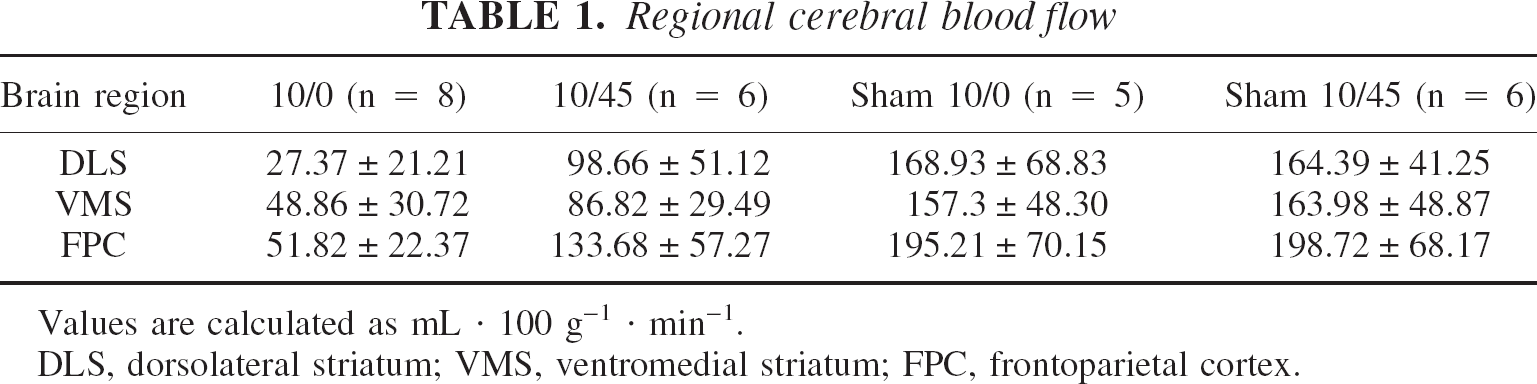
Values are calculated as mL · 100 g–1 · min–1.
DLS, dorsolateral striatum; VMS, ventromedial striatum; FPC, frontoparietal cortex.
DISCUSSION
These studies were performed to answer three questions: 1. Does short-term focal ischemia, in the temporal range of human transient ischemic attacks, result in programmed cell death? 2. At what ischemic intensity does necrosis appear versus apoptosis? and 3. Are there thresholds of focal CBF that correlate with the appearance of apoptotic cell death? To answer these questions, we studied durations of focal ischemia ranging from 5 to 15 minutes and with varying reperfusion times. We identified apoptosis in several different ways including H&E staining, TUNEL labeling, and gel electrophoresis. Necrosis was identified in adjacent brain sections by cresyl violet staining. Finally, under identical experimental conditions, rCBF was measured using traditional autoradiographic methods.
We investigated the appearance of apoptosis in a focal ischemia model and its correlation with rCBF. Our results indicate that focal ischemia as brief as 5 minutes may result in histologic change compatible with apoptosis. This is a shorter duration than has been previously studied and raises the possibility that even very short durations of ischemia result in focal cellular brain damage evident only when apoptosis is specifically searched for. We have previously reported that 10 minutes of forebrain ischemia by four-vessel occlusion resulted in scattered cell damage in the striatum suggesting the occurrence of an apoptotic process (Schmidt-Kastner et al., 1997). Others using the same intraluminal MCA occlusion technique as used here have shown that neuronal damage in small- to medium-sized neurons in the dorsolateral striatum develop slow progressive damage in rats subjected to 15 minutes of MCA occlusion (Nakano et al., 1990). Li et al. (1995c) have shown a hierarchy of anatomic vulnerability to ischemic damage after 10 minutes of ischemia followed by 48 h of reperfusion, with few nuclear pyknotic and eosinophillic neurons in the margin of the caudate putamen near the subcortical white matter. They have also shown that after 10 to 20 minutes of MCA occlusion, apoptotic cells exhibiting DNA fragmentation were present in the regions of selective neuronal necrosis in the preoptic area and in the striatum. Thus, our data extend these observations to very short durations of focal ischemia.
Our data show that the total number of apoptotic cells is positively correlated with the duration of ischemia, whereas no relation is seen between the total number of apoptotic cells and the duration of reperfusion. Similarly, Li et al. (1995a) reported that, after 2 h of ischemia, there was no difference in the number of apoptotic cells between the 24- and 48-h reperfusion groups. They also showed that, after 2 h of focal ischemia, apoptosis appeared within 30 minutes of reperfusion and persisted at least 4 weeks, whereas Chen et al. (1997) showed that single-stranded DNA breaks were evident within 1 minute of reperfusion. Our data show that necrotic changes become more evident as the duration of focal ischemia lengthens. Li et al. (1995a) have shown that, with prolonged ischemic durations, apoptosis appears only in the periphery of necrotic damage, with necrosis becoming the dominant ischemic outcome. These data support the current therapeutic emphasis on interrupting the ischemic cascade as soon as possible.
The intraoperative measurement of CBF by the hydrogen-clearance method in the FPC had to be 20 mL · 100 g–1 · min–1 or less for us to proceed with that particular experiment, but the data reported in Table 1 shows CBF in the FPC to be 51.82 ± 22.37 mL · 100 g–1 · min–1. This discrepancy is due to the fact that, whereas hydrogen clearance samples CBF qualitatively along the platinum electrode, IAP allows quantitative and regional determination of CBF. In addition, the two measurements were obtained at different occlusion durations. Finally, the hydrogen electrode was located quite posteriorly in the FPC. Thus, it is not surprising that the CBF measurements reported for the FPC differ between the two techniques.
We observed that the dorsolateral striatum is the brain region most vulnerable to ischemic insult as a result of MCA occlusion. This is supported by our observation that CBF in this region is the lowest during MCA occlusion and is consistent with the levels reported in the literature (Belayev et al., 1997; Nagasawa and Kogure, 1989; MacManus et al., 1993; Tsuchidate et al., 1997). Our data show that apoptosis appears predominantly where rCBF may be in the penumbral range, though reperfusion restores rCBF to near normal values. In rabbits that have undergone focal ischemia, the penumbral region is seen to have decreased 23Na signal intensity measured with magnetic resonance imaging, as compared with the increased 23Na intensity that is observed in infarcted regions (Bartha et al., 2001), and the examination of areas with a decreased 23Na signaling show an increase in cleaved caspase-3, suggesting, using different techniques and a different animal model, that apoptotic events are initiated in the penumbra zone (Bartha et al., 2001). Thus, we propose that apoptotic mechanisms are triggered in regions of moderate ischemic severity. The CBF value of 27.37 ± 21.21 we report in the dorsolateral striatum is higher than the strict definition of “penumbra” and higher than flow measurements we have previously reported in this region (Hakim et al., 1992). We suspect that this discrepancy is due to the use of a “thread” to occlude the MCA rather than coagulating the vessel as we had done in the earlier study. Nonetheless, the preferential appearance of apoptotic cell death in this region suggests a strong correlation with blood flows in the penumbra range.
There are several important clinical considerations arising from this work. In patients who undergo a TIA, rCBF to the affected brain regions falls to penumbra levels and durations comparable with those used in our studies (Powers et al., 1985). Although, by definition, patients who have undergone a TIA have no clinical or radiologic sequelae, we must entertain the possibility that this may be owing to our inability to detect apoptotic cell death clinically. It is possible that the brain responds to any ischemia by evident or subtle forms of cell death depending on the severity or duration of the episode. Further evidence supporting this work may lead to the recognition that TIAs are harmful events in and by themselves, in addition to being harbingers of stroke. As such, TIA patients may potentially be aided by an antiapoptotic therapy that becomes available.
Footnotes
Acknowledgments:
The authors would like to thank the Canadian Stroke Network, for its financial support; Dr. John P. MacManus, for allowing use of his facilities for gel electrophoresis; Mr. Robert Cadonic, for his careful technical work; and Dr. Charlie Thompson, for his able assistance. They also thank Ava K. Chow, for her helpful suggestions during manuscript proofreading.
