Abstract
Using the microdialysis technique and laser-Doppler flowmetry, we performed simultaneous measurement of salicylate hydroxylation and glutamate release along with local CBF in the ischemic penumbral cortex of rat brain subjected to normothermic transient middle cerebral artery (MCA) occlusion. Cortical CBF fell to 24 ± 11% (mean ± SD) during ischemia and recovered to 84 ± 16% during reperfusion. Extracellular glutamate levels increased by 6.5-fold above baseline 10 min following MCA occlusion but subsequently returned to near baseline levels in spite of the persistent ischemia. Increase in 2,3- and 2,5-dihydroxybenzoic acid (DHBA) concentrations in the microdialysis perfusate was confirmed during both ischemia and reperfusion phase. Although the temporal profile and amount of salicylate hydroxylation were heterogeneous among individual animals, integrated 2,3-DHBA concentrations during reperfusion were correlated positively with integrated glutamate concentrations during ischemia and negatively with mean postischemic CBF. These relationships suggest a possible association of the enhanced production of 2,3-DHBA during reperfusion with larger amounts of intraischemic glutamate release and lower levels of postischemic CBF.
Oxygen free radical formation and subsequent lipid peroxidation have been postulated to be important steps in the pathogenesis of ischemic or hypoxic brain injury (Watson et al., 1984; Halliwell and Gutteridge, 1985; Braughler and Hall, 1989; Hall and Braughler, 1989). Involvement of free radicals in the production of ischemia/reperfusion injury is suggested by the beneficial effects of free radical scavenging drugs (Abe et al., 1988; Johshita et al., 1989; Oh and Betz, 1991; Watanabe et al., 1994) and superoxide dismutase (Liu et al., 1989; Kinouchi et al., 1991; Matsumiya et al., 1991).
There is increasing evidence that excitatory amino acids such as glutamate and aspartate play an important role in ischemic neuronal death. Large amounts of glutamate and/or aspartate were released into the extracellular space during both global and focal cerebral ischemia (Benveniste et al., 1984, 1989; Hagberg et al., 1985; Globus et al., 1988, 1991; Hillered et al., 1989; Butcher et al., 1990; Takagi et al., 1993). Deafferentation of glutamatergic afferent circuits protected hippocampal neurons from ischemic damage (Wieloch et al., 1985; Jørgensen et al., 1987; Benveniste et al., 1989). Glutamate receptor antagonists reduced infarction volume in experimental focal ischemia (Ozyurt et al., 1988; Gill et al., 1991; Buchan et al., 1993), and they also prevented selective hippocampal injury in global forebrain ischemia (Sheardown et al., 1990; Buchan et al., 1991; Nellgård and Wieloch, 1992; Li and Buchan, 1993).
Recent studies have begun to explore possible interactions between oxygen radicals and excitatory amino acids in ischemic neuronal injury. Using rat hippocampal slices subjected to simulated transient “ischemia” by oxygen and glucose deprivation, Pellegrini-Giampietro et al. (1990) reported possible induction of excitatory amino acid release by oxygen-derived free radicals. The authors hypothesized the existence of a vicious cycle in which free radical formation and excitatory amino acid release could affect each other in the generation and propagation of ischemic neuronal death (Pellegrini-Giampietro et al., 1990). Monyer et al. (1990) reported the ability of 21-aminosteroids to attenuate excitotoxicity in murine neuronal culture through inhibition of free radical-mediated lipid peroxidation. Glutamate agonists were reported to enhance oxygen free radical production both in vitro (Bondy and Lee, 1993; Lafon-Cazal et al., 1993) and in vivo (Hammer et al., 1993). In in vivo focal ischemia, lack of significant additive effect in combined treatment with dimethylthiourea and MK-801 suggested that oxygen free radicals and excitatory amino acids may damage the brain by a common pathway (Oh and Betz, 1991).
In the present study, we have attempted simultaneous measurement of salicylate hydroxylation and glutamate release in the penumbral cortex of rat brain subjected to transient middle cerebral artery (MCA) occlusion. Salicylate hydroxylation technique was reported as a potential measure of hydroxyl radical formation (Floyd et al., 1984, 1986; Grootveld and Halliwell, 1986). In this method, attack by hydroxyl radical upon salicylate yields free radical intermediates that undergo subsequent reactions to produce 2,3- and 2,5-dihydroxybenzoic acids (DHBAs). These hydroxylated products can be identified and quantified by high performance liquid chromatography (HPLC) with electrochemical detection.
Portions of this study have been reported in preliminary form (Ginsberg et al., 1994).
MATERIALS AND METHODS
Animal preparation
Male Sprague-Dawley rats (Charles River Laboratories, Wilmington, MA, U.S.A.) weighing 280–430 g were food-deprived overnight before surgery. Anesthesia was induced with 3% halothane and continued via a mask with 1.5% in 7:3 mixture of nitrous oxide/oxygen. Atropine sulfate (0.06 mg) was injected intraperitoneally. The left femoral artery and vein were catheterized with PE-50 polyethylene tubes for continuous blood pressure monitoring and intermittent arterial blood gas analysis and for the administration of drugs. Heparin (10 IU/ml) was injected into each line, the total amount of which was 30–50 IU. Rats were then intubated endotracheally with PE-240 polyethylene catheters, immobilized with intravenous administration of pancuronium bromide (initial dose 0.6 mg/kg; additional dose 0.2 mg/kg), and mechanically ventilated on 0.75% halothane in 7:3 mixture of nitrous oxide/ oxygen. The head was fixed in a stereotaxic frame (Stoelting, Wood Dale, IL, U.S.A.). Rectal temperature was measured continuously and maintained between 36.7 and 37.7°C throughout the experiment by means of a warming lamp placed above the body. Head temperature was monitored with a thermocouple probe (CN 9000; Omega, Stamford, CT, U.S.A.) inserted into the left temporalis muscle and maintained at 36.4–37.0°C throughout the experiment by means of a warming lamp placed above the head.
The right MCA was exposed by a subtemporal approach according to the method of Tamura et al. (1981a). A scalp incision was made between the eye and the ear to expose the cranial vault and the right lateral surface of the skull. The zygomatic arch was removed, and the temporalis muscle flap was retracted downward. A craniectomy rostral to the foramen ovale was made with a high-speed minidrill (Nihon Seimitsu Kikai Kogyo K.K., Japan), and the dura and the pia-arachnoid covering the MCA were incised with a 27-gauge needle.
A craniectomy for the microdialysis and laser-Doppler probes was made above the right frontoparietal cortex with a high-speed minidrill. The field was frequently irrigated with physiological saline to prevent thermal injury to the cortex. A small incision was made in the dura and pia-arachnoid with a 27-gauge needle to permit implantation of a microdialysis probe without possible compression injury to the cortex. The dura was left otherwise intact.
A microdialysis probe (2-mm dialysis membrane, CMA/12; Carnegie Medicin, Stockholm, Sweden) was stereotaxically implanted in the right dorsolateral parietal cortex (0.7 mm anterior and 4.0 mm lateral to the bregma and 2.5 mm ventral to the cortical surface). This probe position corresponded to the penumbral location used in previous studies in our laboratory (Nakayama et al., 1988; Takagi et al., 1993) and others (Tamura et al., 1981b; Tyson et al., 1984; Duverger and MacKenzie, 1988; Shiraishi et al., 1989) using the same proximal MCA occlusion model in Sprague-Dawley rats. The probe was continuously perfused with Ringer's solution (147 mM NaCl, 4 mM KCl, and 1.3 mM CaCl2) at a flow rate of 2 μl/min by means of a microinfusion pump (Carnegie Medicin). A 90-min stabilization period was permitted before initiating dialysate sampling.
Production of ischemia
In the MCA occlusion group (n = 9), the proximal portion of the right MCA was occluded with a small metal clip (Zen temporary clip, 15 g pressure; Ohwa Tsusho, Tokyo, Japan) 130 min after implantation of the microdialysis probe. Following 120 min of ischemia, the clip was released to allow reperfusion. Sham-operated nonischemic animals (n = 4), which underwent the same operative procedures including exposure of the MCA and implantation of the microdialysis probe without MCA occlusion, served as controls.
Laser-Doppler flowmetry
Local CBF was monitored continuously throughout the experiment by means of a laser-Doppler flow monitor (LaserFlo BPM 403 A; Vasamedics, St. Paul, MN, U.S.A.) with aprobe(P433-3; Vasamedics) placed on the dura mater ∼1 mm anterior to the microdialysis probe. Steady-state baseline values were recorded before MCA occlusion, and CBF was expressed as a percentage of the average of six baseline measurements taken every 5 min prior to MCA occlusion (Dirnagl et al., 1989). Because ambient light interferes with the flow reading, the heating lamp and microscope illumination were turned off at the time of each blood flow recording.
Microdialysis procedure
Following 90-min stabilization with Ringer's solution, the first 10-min sample of microdialysis perfusate, representing cortical extracellular fluid, was collected. The perfusion fluid was then switched to 5 mM sodium salicylate in Ringer's solution, and two 10-min samples of perfusate were collected prior to ischemia. After MCA occlusion, samples were collected at 10-min intervals during the 120-min ischemic period and the 120-min reperfusion period. In the sham-operated group, perfusate samples were collected at the same intervals as in the MCA occlusion group. All samples were collected on ice and stored at −20°C until analysis by HPLC.
Glutamate measurements
Analysis of glutamate concentration in the perfusate was performed by HPLC with electrochemical detection following precolumn derivatization. A two-step derivatization process was utilized (Allison et al., 1984). In the first step, a combination of o-phthalaldehyde and t-butylthiol was used for the formation of isoindole. The substitution of t-butylthiol for the more commonly used 2-mercaptoethanol improves the derivative's stability. To scavenge the leftover t-butylthiol, iodoacetamide was added in a second step. Both the derivatization and injection of the samples were performed under completely automatic control using the BAS/CMA 200 refrigerated microsampler (Bioanalytical Systems, West Lafayette, IN, U.S.A.). All work was done on a BAS 200 liquid Chromatograph equipped for gradient operation and amperometric detection (Bioanalytical Systems). Amino acids were separated on a microbore column C-18 SepStik with 3-μm particle size (Bioanalytical Systems). The glassy carbon electrode was maintained at +0.7 V vs. Ag/AgCl. The mobile phases consisted of dimethylacetamide/methanol/sodium acetate mixtures (solvent A, 7.0:24.0:69.0 by vol, pH 6.8; solvent B, 20:22.5:7.5 by vol, pH 8.0). The gradient profile for this method is based on simple linear changes in both solvents. This method provides good resolution of the common amino acids with a run time of roughly 30 min. A sharp solvent ramp was used to strip the column after the leucine peak. From peak heights, dialysate concentrations of glutamate were computed by the use of external standards. The sensitivity of the system allowed us to detect glutamate at the nanomolar level.
Measurement of 2,3- and 2,5-DHBA
The 2,3- and 2,5-DHBA adducts were analyzed in microdialysis samples by an HPLC system (Bioanalytical Systems) consisting of a solvent delivery module (BAS PM-80) customized with two amperometric detectors (BAS LC-4C), a dual glassy carbon electrode (BAS MF-100), and an Ag/AgCl reference electrode housed in a preheater module (BAS CC-5). The working electrodes were set at +0.75 V against the Ag/AgCl electrode. Each amperometric detector was set to a different gain to obtain a chromatogram with high sensitivity (for low sample concentrations) and low sensitivity (for high sample concentrations). The separation of different compounds was accomplished by an analytical microbore column C-18 SepStik (150 × 1 mm) with 5-μm particle size. The column was used at room temperature with a mobile phase of the following composition: 27 μM ethylenediaminetet-racetate, 14.7 mM NaH2PO4, 30 mM sodium citrate, 10 mM diethylamine HCl (pH 3.1), and 476 mg octane sulfonic acid to a total volume of 1 L. Thirty milliliters CH3CN and 15 ml THF were then added. The flow rate was 1 ml/min, and a back-pressure was created with an ODS 3-μm (100 × 3.2 mm) column. The system pressure was ∼ 3,000 psi. Calibration curves were run daily with 5 μl of four different concentrations of 2,3- and 2,5-DHBA standards. From peak heights of these external standards, dialysate concentrations of 2,3- and 2,5-DHBA were computed. The sensitivity of the system allowed us to detect 2,3- and 2,5-DHBA at the nanomolar level.
Statistical analysis
All data were expressed as means ± SD. DHBA and glutamate levels and CBF changes were assessed by the repeated measures analysis of variance and post hoc analysis (Bonferroni test) that adjusted the significance level for multiple comparisons. Differences were considered to be statistically significant at p < 0.05.
RESULTS
Physiological data
The body weights were 349 ± 48 g for the MCA occlusion group (n = 9) and 345 ± 6 g for the sham-operated group (n = 4). Table 1 summarizes the physiological variables, which were generally kept within normal limits during the experiment. The value of pH during ischemia was significantly lower in the sham-operated group than in the MCA occlusion group (p < 0.05).
Physiological variables
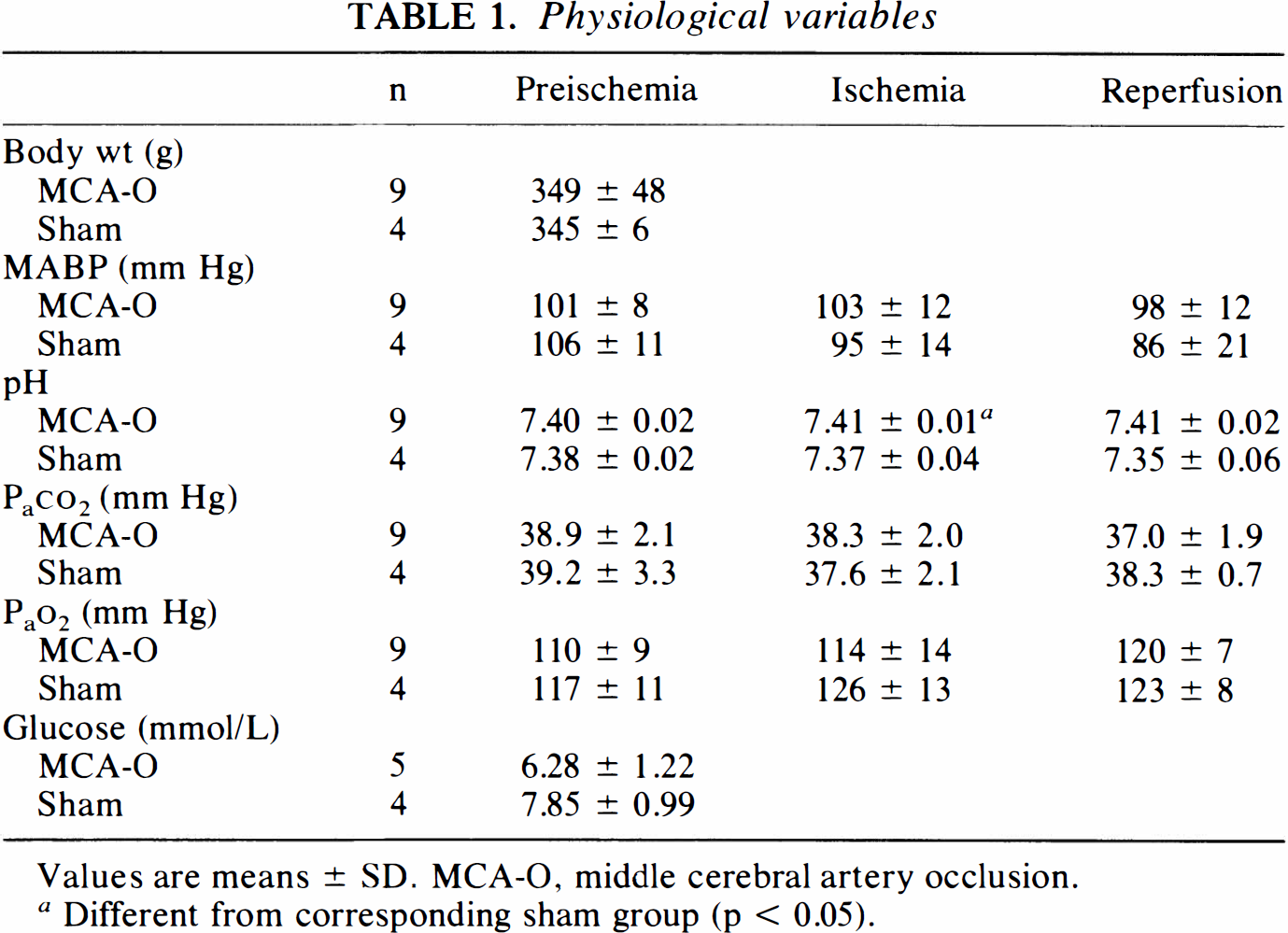
Values are means ± SD. MCA-O, middle cerebral artery occlusion.
Different from corresponding sham group (p < 0.05).
Local CBF changes
Figure 1 shows CBF changes in the focally ischemic cortex during the experiment. Following MCA occlusion, CBF fell to 27 ± 13% (range 12–46%) of the preischemic level and remained almost constant throughout the ischemic period. Average ischemic CBF was 24 ± 11% of the preischemic control. Following release of the clip, CBF increased sharply to 101 ± 52% (range 55–213%). After reduction to 65 ± 23% at 10 min following clip release, CBF recovered gradually and reached above 90% of the preischemic baseline. Hyperemic CBF was observed only transiently in one animal. Mean CBF averaged over the 120-min reperfusion period was 84 ± 16% of the preischemic control. In the sham-operated group, CBF showed no significant difference versus preischemic baseline.
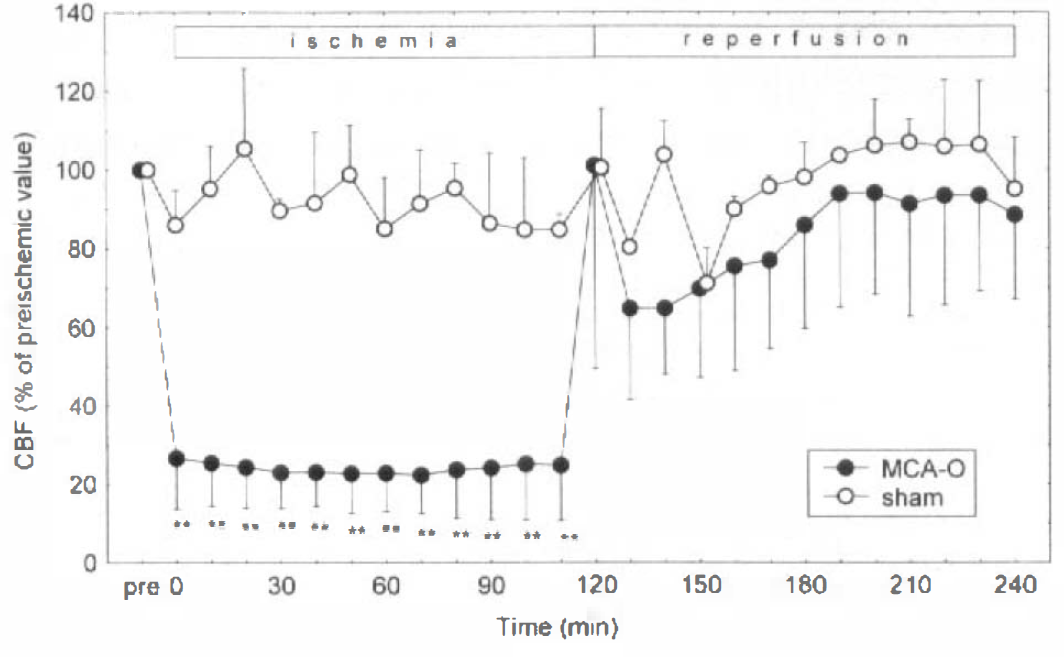
Change of CBF (mean ± SD) measured by laser-Doppler flowmetry in the ischemic cortex of rats subjected to 120-min middle cerebral artery occlusion (MCA-O) followed by 120-min reperfusion (filled circles) and in the corresponding cortex of sham-operated rats (open circles). In the MCA occlusion group, CBF during ischemia was significantly lower than preischemic control (**p < 0.01). Mean CBFs averaged over the ischemic and reperfusion periods were 24 ± 11 and 84 ± 16% of the preischemic control value, respectively. In the sham-operated group, CBF showed no significant difference versus preischemic baseline.
Extracellular 2,3- and 2,5-DHBA concentrations
Concentrations of 2,3- and 2,5-DHBA in the microdialysis perfusate taken from the focally ischemic cortex at 10-min intervals are depicted in Figs. 2 and 3, respectively. Significant increases in concentrations of both 2,3- and 2,5-DHBA occurred during MCA occlusion and subsequent reperfusion. A two-factor repeated measures analysis of variance on the 2,3-DHBA data (Fig. 2) showed a highly significant overall between-subject effect of MCA occlusion on DHBA levels compared with sham rats (p = 0.0033). Post hoc Bonferroni tests revealed significant elevations of 2,3-DHBA throughout the MCA occlusion period and at 20 min of reperfusion (p < 0.05; Fig. 2). A similar analysis carried out on the 2,5-DHBA data (Fig. 3) also showed a highly significant overall between-subject effect of ischemia (p = 0.0019), and Bonferroni testing revealed significant elevations at 20 and 60 min of ischemia and at various time points during the first 90 min of reperfusion (Fig. 3). For both 2,3-and 2,5-DHBA, patterns of elevation among individual animals were heterogeneous. In four of nine animals subjected to MCA occlusion, 2,3-DHBA levels increased during reperfusion. Another four animals showed elevated 2,3-DHBA levels during ischemia, which continued into the reperfusion phase in two rats. In one animal, only minor changes of 2,3-DHBA were observed. Changes of 2,5-DHBA in individual animals generally paralleled those of 2,3-DHBA.
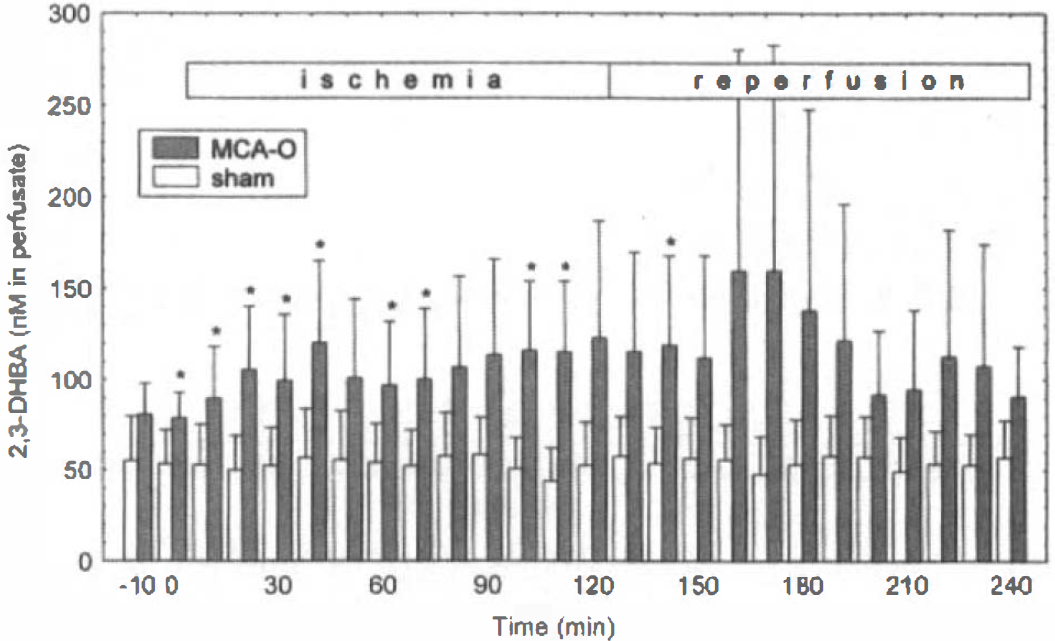
Levels of 2,3-dihydroxybenzoic acid (2,3-DHBA) (mean ± SD) in dialysis perfusate collected from the ischemic cortex of rats subjected to 120-min middle cerebral artery occlusion (MCA-O) followed by 120-min reperfusion (hatched bars) and from the corresponding cortex of sham-operated rats (open bars). Asterisks denote significant differences from sham-operated rats (p < 0.05) by repeated measures analysis of variance followed by the Bonferroni test.
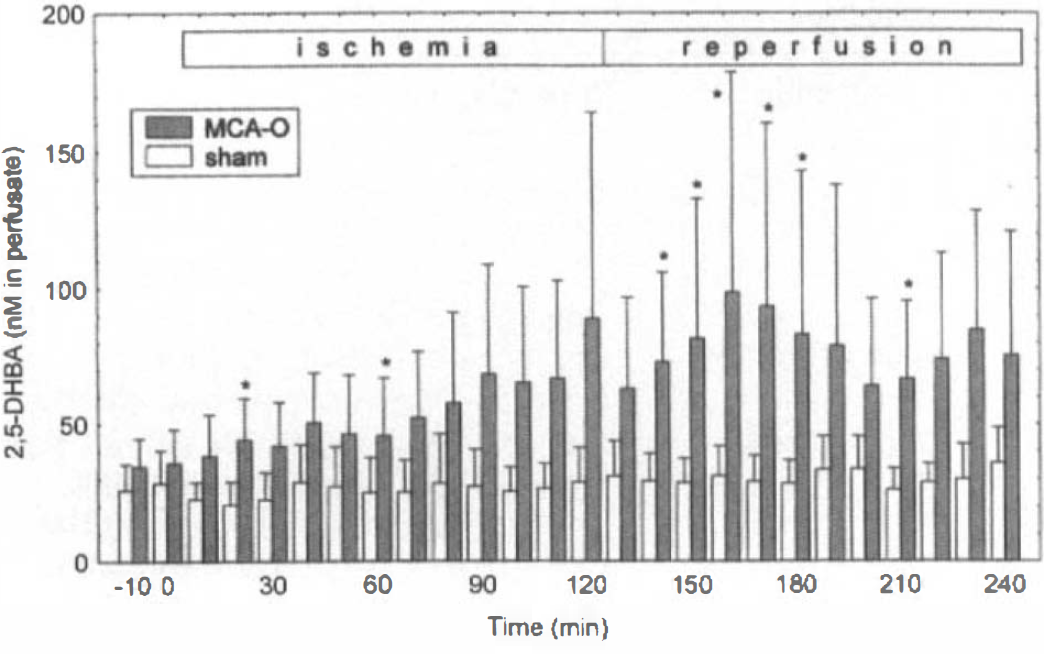
Levels of 2,5-dihydroxybenzoic acid (2,5-DHBA) (mean ± SD) in dialysis perfusate collected from the ischemic cortex of rats subjected to 120-min middle cerebral artery occlusion (MCA-O) followed by 120-min reperfusion (hatched bars) and from the corresponding cortex of sham-operated rats (open bars). Asterisks denote significant differences from sham-operated rats (p < 0.05) by repeated measures analysis of variance followed by the Bonferroni test.
Extracellular glutamate concentrations
Concentrations of glutamate in microdialysis perfusate collected from the focally ischemic cortex are shown in Fig. 4. Focal ischemia due to MCA occlusion was associated with glutamate release into the extracellular space in one-half of the animals, whereas in the other one-half, glutamate levels remained almost stable. Ten minutes following MCA occlusion, extracellular glutamate concentrations increased by 6.5-fold above baseline to a mean concentration of 5.2 ± 6.7 μM. The glutamate levels began to decline at 20 min of ischemia and returned to near baseline by ∼90 min following occlusion. Glutamate levels at 0–10 and 10–20 min were significantly elevated when compared with preischemic control (p < 0.05).
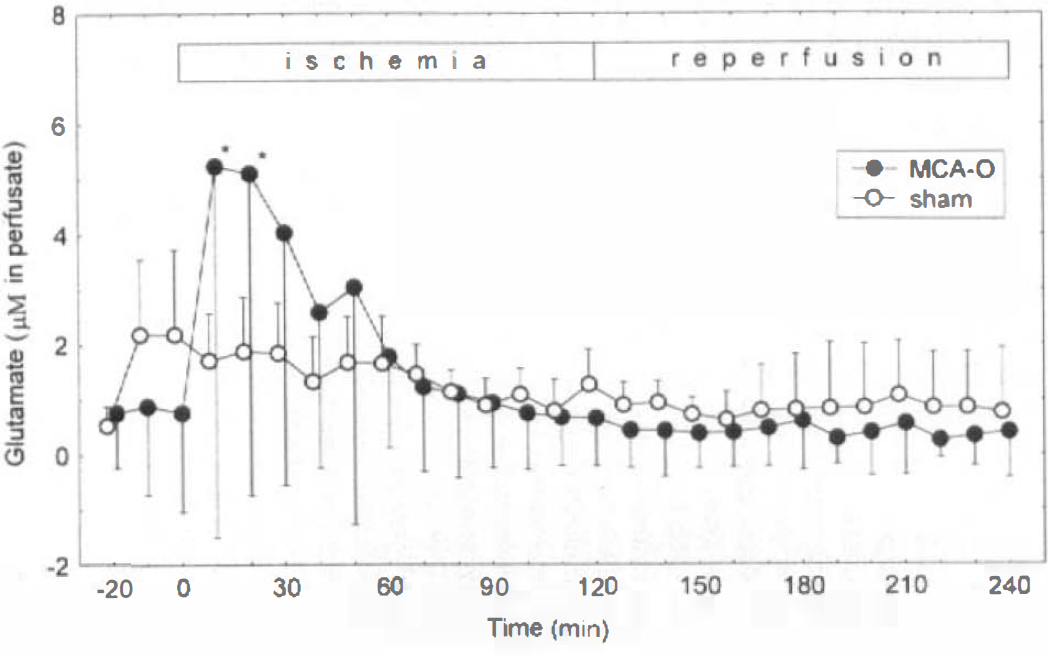
Levels of glutamate (mean ± SD) in dialysis perfusate collected from the ischemic cortex of rats subjected to 120-min middle cerebral artery occlusion (MCA-O) followed by 120-min reperfusion (filled circles) and from the corresponding cortex of sham-operated rats (open circles). Ten minutes following MCA-O, extracellular glutamate concentrations increased by 6.5-fold above baseline. Glutamate levels began to decline at 20 min of ischemia and returned to near baseline by ∼90 min following occlusion. Glutamate was significantly elevated at 0–10 and 10–20 min following MCA-O (*p < 0.05 vs. preischemic value).
Relationships among CBF, salicylate hydroxylation, and glutamate release
Linear regression analysis of DHBA versus glutamate levels revealed a significant positive correlation between integrated 2,3-DHBA levels (corrected for baseline) during reperfusion and integrated glutamate concentrations (corrected for baseline) during ischemia: 2,3-DHBA = 1.35 × 103 + 16.8 × glutamate (R = 0.89, p = 5.1 × 10−5) (Fig. 5). A significant inverse correlation was noted between integrated 2,3-DHBA levels during reperfusion and mean postischemic CBF: 2,3-DHBA = 2.75 × 104 − 261 × CBF (R = −0.81, p = 0.028) (Fig. 6). No significant correlation was found between 2,3-DHBA concentrations during ischemia and CBF or glutamate concentrations, nor was any significant correlation noted between 2,5-DHBA levels and CBF or glutamate.
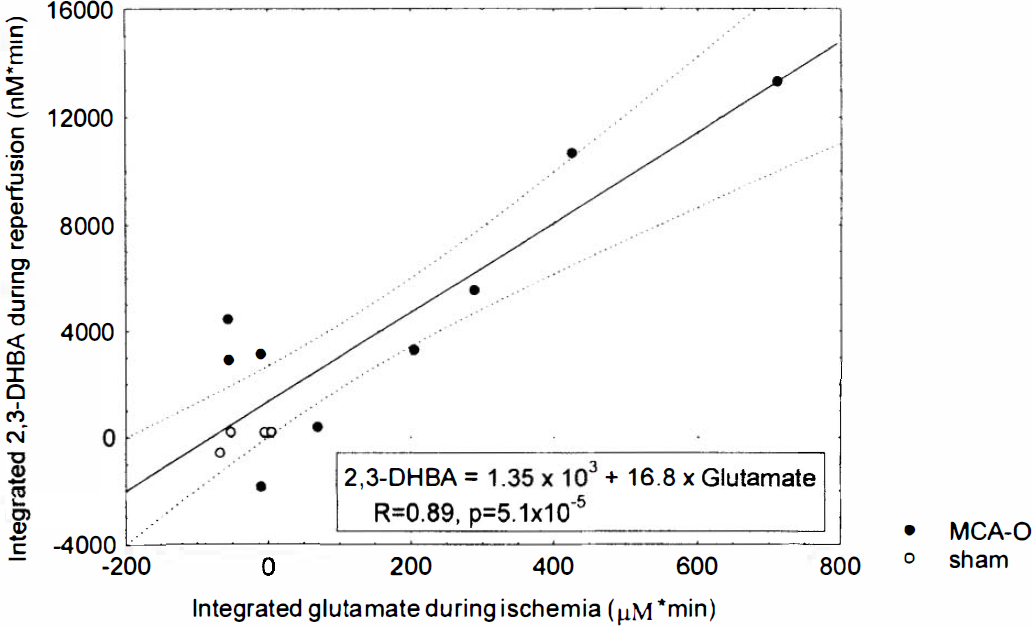
Linear regression of integrated glutamate levels during ischemia and integrated 2,3-dihydroxybenzoic acid (2,3-DHBA) levels during reperfusion. Increased 2,3-DHBA production during reperfusion was associated with higher levels of glutamate release during ischemia (R = 0.89, p = 5.1 × 10−5). MCA-O, middle cerebral artery occlusion.
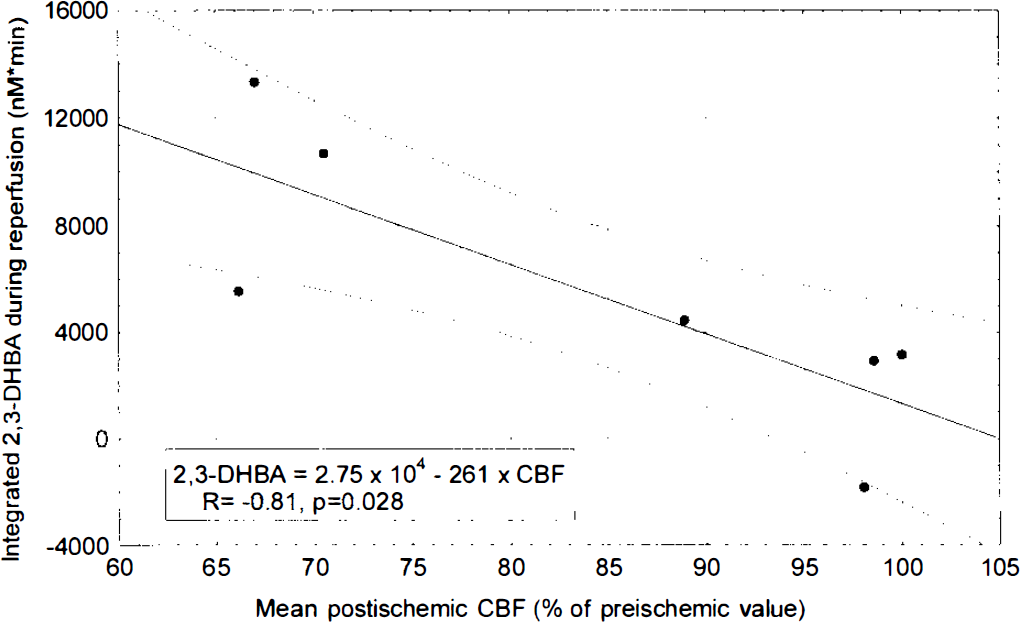
Linear regression of mean cortical CBF during reperfusion and integrated 2,3-dihydroxybenzoic acid (2,3-DHBA) levels during reperfusion. Increased 2,3-DHBA production during reperfusion was associated with lower levels of postishemic CBF (R = −0.81, p = 0.028).
DISCUSSION
In the present study, we performed simultaneous measurement of salicylate hydroxylation and glutamate release in the ischemic penumbral cortex of rats subjected to transient MCA occlusion, and we detected an increase in 2,3- and 2,5-DHBA during both the ischemic and the reperfusion phases. The temporal profile and amount of 2,3- and 2,5-DHBA in the ischemic cortex were highly heterogeneous among individual animals. As a result of linear regression analysis among CBF and DHBA and glutamate concentrations, we demonstrated that integrated 2,3-DHBA concentrations during reperfusion were correlated positively with integrated glutamate concentrations during ischemia (Fig. 5) and negatively with mean postischemic CBF (Fig. 6). These relationships suggest a possible association of the enhanced production of 2,3-DHBA during reperfusion with larger amounts of intraischemic glutamate release and lower levels of postischemic CBF. Although glutamate release may predispose to oxygen radical production, we cannot deduce exact cause-and-effect relationships from these correlations only. It is beyond the scope of this article to infer the downstream mechanisms that can explain the time lag between glutamate release and salicylate hydroxylation.
Although we used the fixed probe position, which corresponded to that used in a previous study in our laboratory (Takagi et al., 1993), the relative position within the ischemic tissue is not constant among individual animals because of variations in infarction volume. Therefore, the probe position in some animals may have been more proximate to the penumbra than in others. These variations are a possible source of heterogeneity in the temporal profile of 2,3- and 2,5-DHBA and glutamate in the microdialysis perfusate.
Salicylate, a relatively nontoxic but highly effective hydroxyl radical trap, forms the stable adducts 2,3- and 2,5-DHBA upon scavenging hydroxyl radicals (Floyd et al., 1984, 1986). In an in vitro study using liver microsomal fractions, 2,3-DHBA was regarded as being rather specific for hydroxyl radical production, although 2,5-DHBA was generated via the cytochrome P-450 system (Halliwell et al., 1991; Ingelman-Sundberg et al., 1991). In an in vivo study, however, hydroxyl radical reaction is to be considered one of many possible reactions that generate salicylate hydroxylated species.
Hall et al. (1993) reported that posttraumatic increase in 2,5-DHBA was eliminated by perfusing the brains with saline, whereas perfusion did not affect the baseline levels in uninjured animals. They considered that the injury-induced increase in 2,5-DHBA might be generated in the microvascular wall. It is possible that the correlation between glutamate and salicylate hydroxylation in the present study may be due to subtle changes in the blood-brain barrier evoked by ischemia.
Production of free radicals during hypoxic or ischemic conditions is widely accepted (Braughler and Hall, 1989; Hall and Braughler, 1989). In global ischemia, radicals are formed predominantly during the period of postischemic reperfusion. Cao et al. (1988) reported a significant increase in brain DHBA in gerbils subjected to bilateral carotid occlusion for at least 5 min, followed by 5 or 15 min of reperfusion. Without reperfusion or with only 2 min of ischemia followed by reperfusion, the production of DHBA was not increased. Althaus et al. (1993) showed increased hydroxyl radical formation in the gerbil bilateral carotid occlusion model with 1 min of reperfusion; in contrast to the former report, 5 min of reperfusion abolished the enhanced production of free radicals. By means of the spin-trapping method, free radicals were detected in lipid extracts of cortical homogenates in the gerbil or rat forebrain ischemia model with reperfusion (Oliver et al., 1990; Sakamoto et al., 1991). However, free radical production may also occur during ischemia. With the use of the microdialysis or cortical cup techniques and a spin-trapping agent, the production of strong oxidants was confirmed during ischemia and the initial phase of reperfusion in the rat global ischemia model (Zini et al., 1992; Phillis and Sen, 1993; Sen and Phillis, 1993).
In focal ischemia, the participation of free radicals in the production of ischemia/reperfusion injury was suggested by the effectiveness of free radical-scavenging drugs (Abe et al., 1988; Johshita et al., 1989; Oh and Betz, 1991; Watanabe et al., 1994) and superoxide dismutase (Liu et al., 1989; Kinouchi et al., 1991; Matsumiya et al., 1991). However, direct measurement of free radical production was not performed in those studies. In the present study, we measured 2,3- and 2,5-DHBA in vivo in the ischemic penumbral cortex following proximal MCA occlusion in the rat and confirmed an increase in their concentrations during both the ischemia and the reperfusion phases. As shown by laser-Doppler flowmetry, local CBF during ischemia at a site adjacent to the microdialysis probe was almost constant at 24 ± 11% of the preischemic baseline. The percentage CBF relative to preischemic control was reported to correlate well with that obtained by autoradiography (Dirnagl et al., 1989). This residual flow was apparently able to deliver enough oxygen to the ischemic tissues to produce free radicals. The possibility of free radical formation even when tissues contain only 5% of normal oxygen levels has been reported (Demopoulos et al., 1980).
Extracellular glutamate levels increased rapidly in response to the ischemic insult but subsequently returned to baseline levels in spite of the persistent ischemia. These results confirm previous studies from our laboratory (Takagi et al., 1993) and those of other workers (Butcher et al., 1990). Others have reported sustained elevations of excitatory amino acids following MCA occlusion in rat striatum (Hillered et al., 1989) and cat cortex (Matsumoto et al., 1992, 1993). This difference may be, at least in part, due to the position of the microdialysis probe. In the ischemic core, extracellular glutamate concentrations showed sustained elevations (Hillered et al., 1989; Butcher et al., 1990), whereas at peripheral sites, they showed a transient rise and then a decline in spite of ongoing ischemia (Butcher et al., 1990; Takagi et al., 1993).
Recent reports emphasize possible interactions between oxygen radicals and excitatory amino acids in ischemic neuronal injury. Pellegrini-Giampietro et al. (1990) reported the possibility that oxygen-derived free radicals could induce excitatory amino acid release in rat hippocampal slices. They hypothesized the existence of a vicious cycle in which free radical formation and excitatory amino acid release could affect each other in the generation and propagation of ischemic neuronal injury. Inhibition of free radical-mediated lipid peroxidation by 21-aminosteroids was effective in protecting cultured neurons against excitotoxic injury (Monyer et al., 1990). Oh and Betz (1991) considered that excitatory amino acid and oxygen free radicals might damage the brain by a common pathway because combined treatment with dimethylthiourea and MK-801 lacked a synergistic effect. Several reports demonstrated that glutamate agonists were able to enhance free radical generation both in vitro (Bondy and Lee, 1993; Lafon-Cazal et al., 1993) and in vivo (Hammer et al., 1993). Our results, which suggest a possible association between larger amounts of glutamate release during ischemia and the enhanced production of 2,3-DHBA during reperfusion, support the idea that oxygen free radicals and excitatory amino acids may interact with each other in the formation of ischemic neuronal injury.
Footnotes
Acknowledgment:
This study was supported by U.S. Public Health Service grant NS 05820.
