Abstract
Cerebral hypoxia-ischemia produces major alterations in energy metabolism and glucose utilization in brain. The facilitative glucose transporter proteins mediate the transport of glucose across the blood–brain barrier (BBB) (55 kDa GLUT1) and into the neurons and glia (GLUT3 and 45 kDa GLUT1). Glucose uptake and utilization are low in the immature rat brain, as are the levels of the glucose transporter proteins. This study investigated the effect of cerebral hypoxia-ischemia in a model of unilateral brain damage on the expression of GLUT 1 and GLUT3 in the ipsilateral (damaged, hypoxic-ischemic) and contralateral (undamaged, hypoxic) hemispheres of perinatal rat brain. Early in the recovery period, both hemispheres exhibited increased expression of BBB GLUT1 and GLUT3, consistent with increased glucose transport and utilization. Further into recovery, BBB GLUT1 increased and neuronal GLUT3 decreased in the damaged hemisphere only, commensurate with neuronal loss.
Cerebral hypoxia-ischemia involves a cascade of metabolic events that begin during insult and proceed well into the recovery period. These changes include major alterations in tissue glucose and energy metabolism, which reflect changes in the relationship between substrate supply and energy demand during the evolution of the hypoxic-ischemic brain damage (Siesjo, 1981; Raichle, 1983; Vannucci, 1990; Vannucci et al., 1994a). The initial step in cerebral glucose utilization involves transport of glucose from the blood across the endothelial cells of the blood–brain barrier (BBB) and then into the neurons and glia. These processes are mediated by the facilitative glucose transporter proteins, GLUT1 and GLUT3. A high molecular weight (55 kDA), more heavily glycosylated form of GLUT1 is concentrated in the endothelial cell membranes of the cerebral micro vasculature, whereas a 45 kDa GLUT1 is expressed in the parenchyma, (most likely in glia) and in choroid plexus and ependyma; GLUT3 is the neuronal glucose transporter (for review, see Maher et al., 1994). The levels of these proteins are quite low in the immature brain and are limiting to cerebral glucose transport and utilization (Vannucci, 1994; Vannucci et al., 1994c). We have previously reported that, in an immature rat model of unilateral hypoxic-ischemic brain damage (Rice et al., 1981), brain glucose concentrations, brain/plasma glucose ratios, and cerebral glucose utilization (CGU) are all significantly altered in both ipsilateral (damaged) and contralateral (undamaged) hemispheres at 4 and 24 h of recovery (Vannucci et al., 1994a). The purpose of this study was to examine the extent to which these observed alterations in cerebral glucose metabolism are related to hypoxia-ischemia-induced changes in the expression of the glucose transporter proteins, GLUT1 and GLUT3, and whether these changes persist.
MATERIALS AND METHODS
Pregnant Wistar rats (Charles River, Wilmington, DE, U.S.A.) were housed in individual cages and fed standard laboratory chow ad libitum. Offspring were delivered vaginally and litter size adjusted to 10 pups/litter on the day of delivery. Hypoxia–ischemia was induced in 7-day-old rat pups following ligation of the right common carotid artery, as previously described (Rice et al., 1981), with minor modifications. Prior to exposure to hypoxia (8% O2/92% N2), pups were placed in a 37°C water bath for 20 min followed by 2.5 h of hypoxic exposure. Preliminary studies demonstrated that this period of pre-warming produces a more uniform degree of damage in littermates but requires a reduction in the hypoxic exposure to insure survival. Control littermates were maintained in open jars at 37°C for the same interval, but were neither ligated nor exposed to hypoxia. Pups were returned to their dams for 4, 24, or 72 hours of recovery. At each interval, pups were killed by decapitation and the brain removed. A sample was taken from the middle of each hemisphere, in the distribution of the middle cerebral artery, for the preparation of total membranes, as previously described (Vannucci, 1994).
Isolated microvessels were prepared from pooled samples of five rat brains, each divided into ipsilateral and contralateral hemispheres by a modification of the previously described method for immature rats (Betz and Goldstein, 1981; Vannucci, 1994). Briefly, pooled hemispheres, (∼0.5 g), were homogenized and the vascular fraction separated from the myelin by centrifugation through 17% dextran (3000 × g). Resuspended vessels were filtered through 125 μm nylon mesh directly onto 40 μm nylon mesh, and the microvessels recovered directly from the nylon mesh. Microvessels were sonicated and stored at −70°C. The vascular-free fraction was obtained by centrifugation of the original 1000 × g supernatant at 150,000 × g for 20 min, as described (Vannucci, 1994). Protein was determined on all samples with the bichinchonic acid reagent (Sigma Chemical Company, St. Louis, MO, U.S.A.).
Western blot analysis for GLUT 1 and GLUT3 proteins was accomplished with isoform-specific anti-C-terminal antisera (prepared by Hoffmann-La Roche, Nutley, NJ, U.S.A.), and 125I-protein A (New England Nuclear, Boston, MA, U.S.A.), as previously described (Maher et al., 1992, 1993). A monoclonal antibody, SP14, which detects SNAP-25 (Honer et al., 1993) was provided by Dr. Peter Davies, Albert Einstein College of Medicine, Bronx, NY, U.S.A. Autoradiograms were quantitated with the National Institutes of Health (NIH) Image Analysis System 1.45 for digital image processing and analysis, and experimental values were calculated relative to a uniform brain membrane “standard”, which was included on every blot (Vannucci, 1994).
Statistical analysis of the data included the paired Stu-dent's t-test and a one-way analysis of variance (ANOVA) with the Tukey test for multiple comparisons. Significance was assumed at p < 0.05.
RESULTS
Levels of expression of both forms of GLUT1 (i.e., the 55 kDa BBB form and the 45 kDa parenchymal form) and of neuronal GLUT3 were determined by Western blot analysis of membranes prepared from individual ipsilateral and contralateral hemispheres of hypoxic-ischemic rats following 4, 24, and 72 h of recovery and compared with litter-mate controls, as shown in the bottom panel of Fig. 1. Autoradiograms were quantitated and experimental values expressed relative to the brain membrane standard, S, in arbitrary standard units. Results were then calculated as the percentage of ipsilateral/contralateral for each experimental animal as shown in Fig. 1 (top), as well as relative to the age-matched control level, shown as the percentage of contralateral/control. At 4 h of recovery, there was no significant difference between the two hemispheres in the hypoxic-ischemic animals, but there was a 23% increase in the mean level of 55 kDa GLUT1 in both hemispheres, relative to control. Similarly, GLUT3 was increased by −20% in both hemispheres, relative to control. At this stage of recovery, brain glucose concentrations and brain/glucose plasma ratios in both hemispheres have been reported to be 130–145% of control, with the contralateral hemisphere exhibiting a higher value than the ipsilateral hemisphere (Vannucci et al., 1994a). At 24 h of recovery, the most dramatic effect of the hypoxia-ischemia was seen in the levels of 55 kDa GLUT1 in the ipsilateral hemisphere. These were 166% of the levels in the contralateral hemisphere, the latter having returned to control values by 24 h. GLUT3 in the ipsilateral hemisphere was 85% of the level measured in the contralateral hemisphere, the latter being 20% greater than the age-matched control value, although this difference was not significant (p < 0.055). By 72 h of recovery, i.e., P10, there were significant age-related increases in the levels of GLUT1 and GLUT3/mg membrane protein in control, animals (N, Fig. 1, Western blot), appropriate to normal development (Vannucci, 1994). At this time, there was no difference between the contralateral hemisphere and the age-matched control brain. Although the level of 55 kDa GLUT1 tended to be slightly reduced, there was no significant difference between hemispheres. GLUT3, however, was significantly reduced in the damaged, ipsilateral hemisphere.
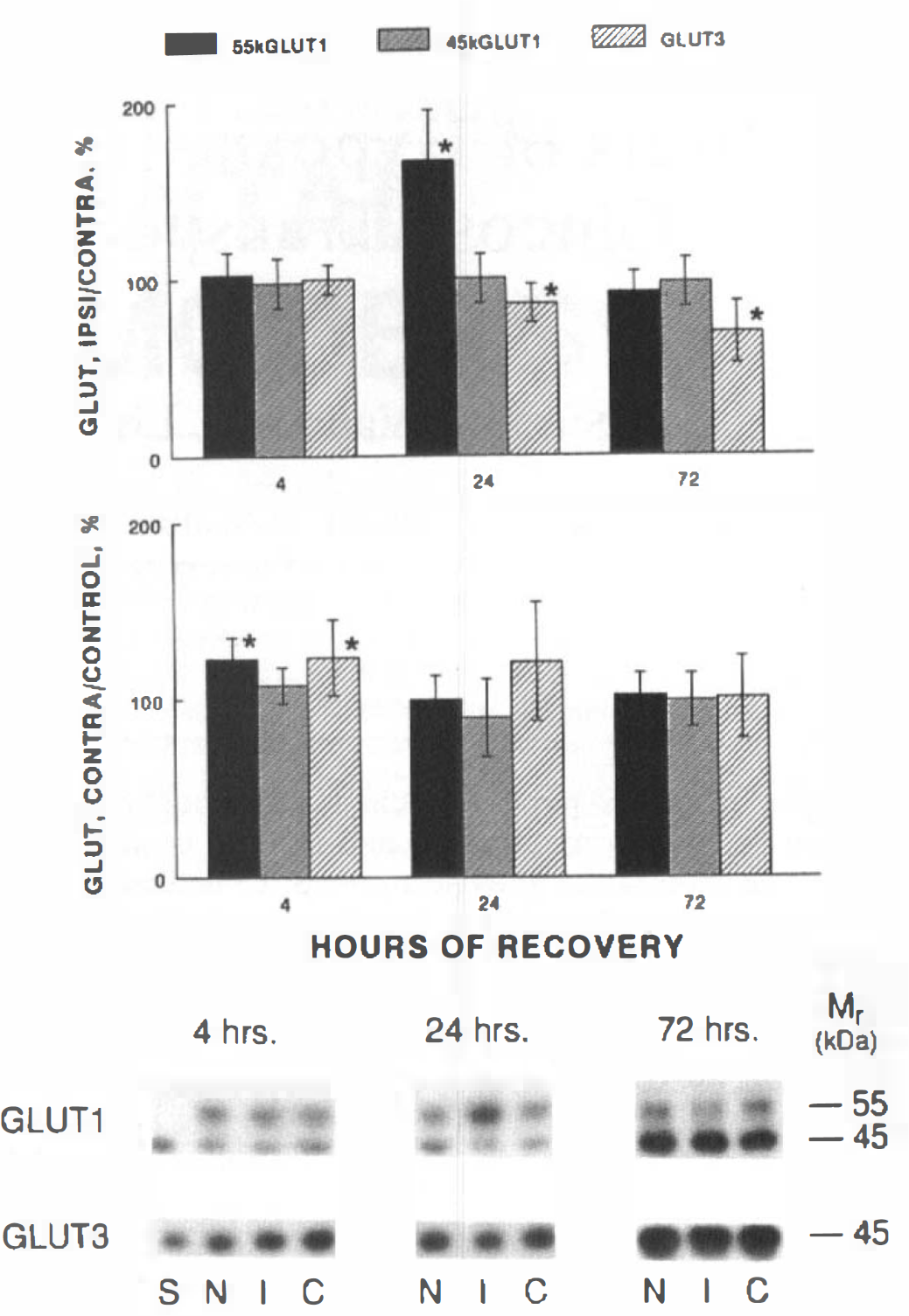
Western blot analysis of GLUT1 and GLUT3 in immature rat brain: effect of hypoxia-ischemia. Total membranes were prepared from ipsilateral and contralateral hemispheres of immature rat brains at 4, 24, and 72 h of recovery and analyzed for GLUT1 and GLUT3 content. The lower panel is a composite autoradiogram of representative samples of paired ipsilateral (I) and contralateral (C) hemispheres from animals at each point of recovery compared with untouched, age-matched control littermates (N), 15 g membrane protein per lane; and S, a brain microsomal membrane standard, 5 μg. All samples (n = 12 at each time point) were quantitated relative to the brain membrane standard, S, in arbitrary standard units and then calculated as the percentage of ipsilateral/contralateral and contralateral/control, expressed as mean ± SD. * denotes significant interhemispheric differences, as determined by the paired Student's t test, p < 0.05, or intergroup differences, as determined by ANOVA.
In order to ascertain the extent to which the observed changes in GLUT3 were simply due to neuronal loss following the hypoxic-ischemic insult, samples were subjected to immunoblot analysis with the monoclonal antibody SP-14, which detects the synaptic vesicle protein SNAP-25 (Honer et al., 1993), as shown in Fig. 2 (bottom). Autoradiograms were quantitated relative to the brain membrane standard and ratios of GLUT3/SNAP-25 calculated for each sample and expressed as a percentage (Fig. 2, top). During the period between 7 and 10 postnatal days, levels of SNAP-25 and GLUT3 proteins increased similarly such that the ratios obtained in normal rat pups at each time point remained unchanged and are shown combined as the control group, C. At 4 h of recovery, the ratio was significantly increased in both hemispheres, reflective of an absolute increase in the GLUT3 protein and, as seen in Fig. 1, with no interhemispheric difference. By 24 h of recovery, the contralateral GLUT3 level and the GLUT3/SNAP-25 ratio had returned to near control levels. Observation of a still elevated ratio in the ipsilateral hemisphere relates to a more rapid loss of SNAP-25 protein than GLUT3 in the damaged hemisphere, although whether this GLUT3 is still functional is impossible to detect by these methods. By 72 h of recovery, however, GLUT3 levels were reduced to the same extent as were SNAP-25, and the ratio was not different from that of control.
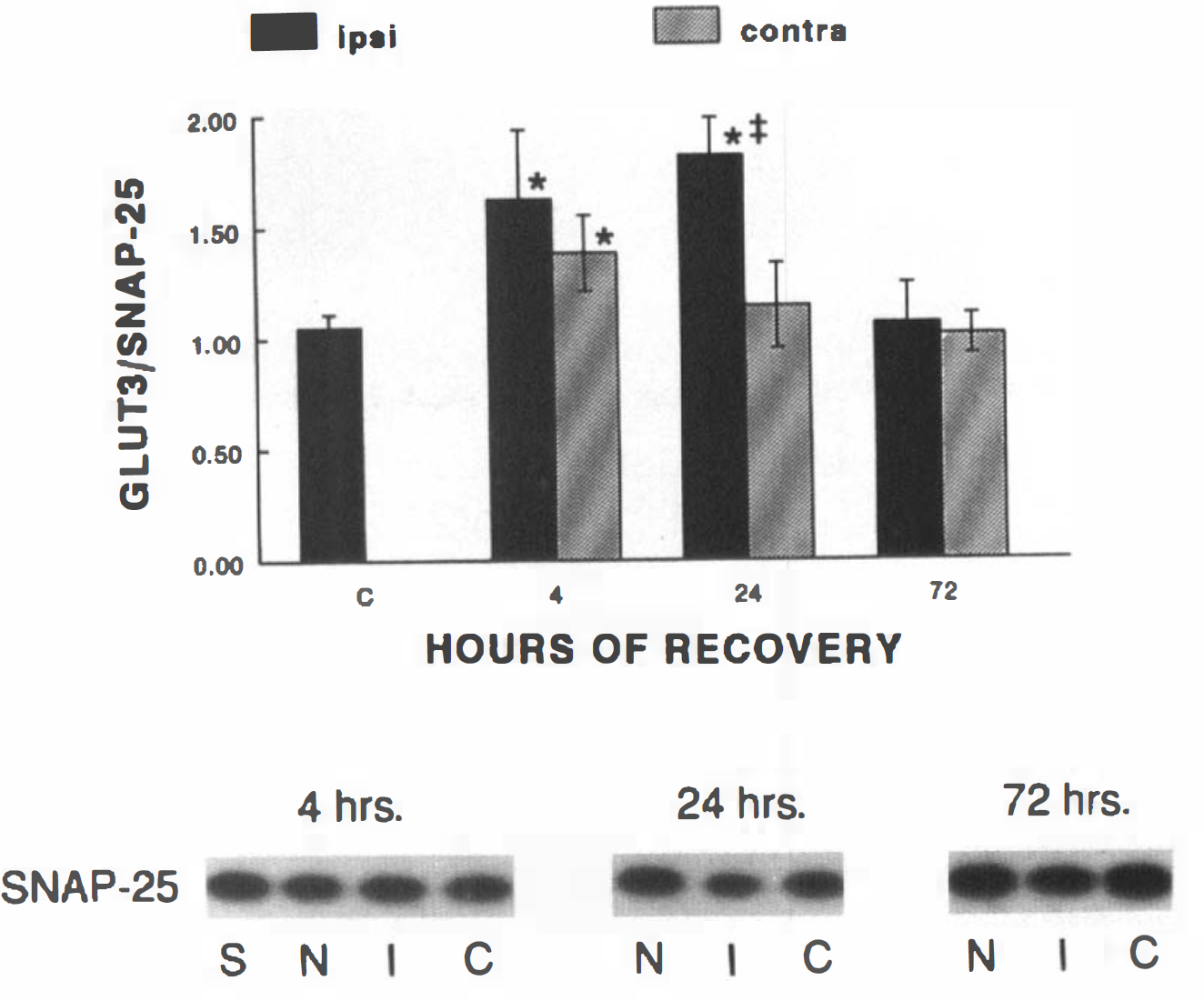
Ratio of GLUT3/SNAP-25 proteins in immature rat brain: effect of hypoxia-ischemia. Total membranes, prepared from ipsilateral and contralateral hemispheres of immature rat brains at 4, 24, and 72 h of recovery, were analyzed for the synaptic vesicle protein, SNAP-25. The lower panel is a composite autoradiogram of representative samples of paired ipsilateral (I) and contralateral (C) hemispheres from animals at each point of recovery compared with untouched, age-matched control littermates (N), 15 μg membrane protein per lane; and S, a brain microsomal membrane standard, 5 g. All samples (n = 12 at each time point) were quantitated relative to the standard, S, in arbitrary standard units and then used to normalize the GLUT3 values depicted in Fig. 1, i.e., the percentage of GLUT3/SNAP-25 expressed as mean ± SD. * denotes significant difference from control and 72 h as determined by ANOVA and Tukey statistics, p < 0.05; # denotes significant interhemispheric difference, as determined by the paired Student's t test, p < 0.05.
The Western blot analysis depicted in Fig. 1 was performed on whole (i.e., unfractionated) membrane samples. In normal brain, the 55 kDa GLUT1 is confined to the cerebral microvasculature of the BBB and can be separated from the 45 kDa parenchymal GLUT1 by preparing isolated microvessels and vascular-free membranes (Maher et al., 1993; Vannucci, 1994). However, the possibility exists that another cell type might express the higher molecular weight form in response to hypoxic-ischemic injury. To examine this possibility, isolated microvessels were prepared from ipsilateral and contralateral hemispheres of experimental animals 24 h following the hypoxic-ischemic insult, as this was the point at which the increased expression of the 55 kDa GLUT1 was most pronounced. Previous preparations of isolated microvessels from immature rat brains were prepared from 3 g of brain tissue/preparation. We have modified the procedure such that 0.5 g (equivalent to five single hemispheres) was used for each preparation, as described in the Methods section. Western blot analysis for GLUT1 of ipsilateral and contralateral microvessels from two groups of hypoxic-ischemic animals compared with an equivalent amount of microvessel protein from an adult rat is shown in Fig. 3. It is evident from this figure that the concentration of GLUT1/mg microvessel protein was dramatically increased in the ipsilateral hemisphere compared to the contralateral hemisphere. Only 45 kDa GLUT1 was detected in the vascular-free membranes (data not shown), in agreement with previous findings (Maher et al., 1993; Vannucci, 1994).
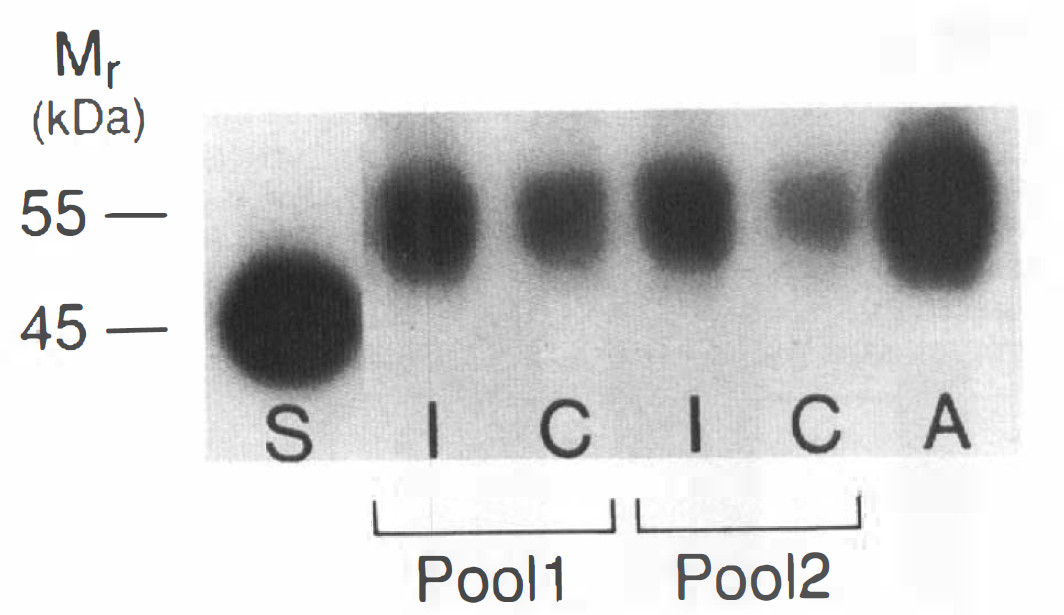
Western blot analysis of GLUT1 in microvessels isolated from ipsilateral and contralateral hemispheres of hypoxic-ischemic rats at 24 h of recovery. Isolated microvessels were prepared from pooled cortical shells of brains of five rats, separated into ipsilateral (I) and contralateral (C) hemispheres, at 24 h of recovery, equivalent to 0.5 g tissue/sample. Shown are two pools analyzed for GLUT1 content, compared with adult microvessels (A) and brain standard (S), 10 μg protein/lane. Only 55 kDa GLUT1 is detected in microvessels compared to only 45 kDa GLUT1 in the vascular-free standard.
DISCUSSION
This study demonstrates that cerebral hypoxia-ischemia in the immature rat produces significant alterations in both BBB GLUT1 and neuronal GLUT3 proteins in both ipsilateral and contralateral hemispheres that begin soon after the insult and proceed well into the recovery period. The magnitude and time course of these changes differed both with respect to the transporter isoform as well as the hemisphere involved. These findings are best interpreted in the context of the alterations in cerebral glucose metabolism that occur during this interval of recovery. Previous studies from our laboratory (Vannucci et al., 1989; Palmer et al., 1990; Vannucci et al., 1994a) have demonstrated that at the end of the hypoxic period, brain glucose decreased to 0.15–0.3 mM, despite the persistence of ∼3 mM plasma glucose, reflecting the inability of the BBB glucose transport system, (i.e., GLUT1) to provide sufficient substrate to meet the increased demands of anaerobic metabolism. With recovery, there was a rapid rise in brain glucose concentrations in both hemispheres such that at 4 h of recovery, brain glucose and brain/plasma ratios were ∼160% of control. These findings suggest increased glucose transport into brain, which is likely being mediated by the increased GLUT1 in the cerebral microvessels. The early increase in 55 kDa GLUT1 was evident in both hemispheres and was, presumably, an acute response to hypoxia. By 24 h of recovery, the level of BBB GLUT1 had normalized in the contralateral hemisphere (hypoxia alone) but was increased even further in the hypoxic-ischemic, ipsilateral hemisphere. That these changes were confined to the BBB was confirmed in the isolated microvessels. (This study is the first demonstration of the feasibility of preparing microvessels from small amounts of brain, making such analysis possible in fewer experimental animals.)
There was also an acute response of neuronal GLUT3 which, at 4 h of recovery, was equivalent in both hemispheres, relative to control. This early increase in GLUT3 levels nearly resolved by 24 h in the contralateral hemisphere and was actually further reduced in the ipsilateral hemisphere with the onset of neuronal necrosis. It would appear that SNAP-25 is a more sensitive indicator of necrosis, than is GLUT1, since the loss of this protein precedes that of GLUT3, evident in the increased ratio shown in Fig. 2. Glucose utilization, as well as GLUT3 levels, are somewhat increased in the contralateral hemisphere at 24 h of recovery, presumably reflecting an increased demand for glucose disposal, possibly coupled with repair, occurring at this time. By 72 h of recovery, brain glucose levels have normalized in both hemispheres (Palmer et al., 1990) and the sole enduring difference in the expression of the glucose transporter proteins is a reduction in the concentration of GLUT3 in the area of the infarct, commensurate with neuronal loss.
An enhancement of GLUT1 expression in response to hypoxia has been reported for adult rat brain (Harik et al., 1994) in vivo and in endothelial cells in vitro (Loike et al., 1992), and it is well known that GLUT1 responds acutely to several stresses, much like an inducible heat shock protein (Wertheimer et al., 1991; Murakami et al., 1992). However, the present finding is the first demonstration of this effect in immature brain. The effect observed in the contralateral, purely hypoxic hemisphere was transient, whereas the combination of hypoxia and ischemia (ipsilateral hemisphere) produced a more profound effect. In both hemispheres, the effect on GLUT1 expression was confined to the BBB. It is important to note, however, that the lack of an overall change in the level of the 45 kDa GLUT1 does not necessarily indicate that there are no specific alterations in this transporter isoform. As 45 kDa GLUT1 is primarily expressed by nonvascular epithelial cells, i.e., choroid plexus and ependyma, and glia, it is likely that with the evolution of brain damage there are cell-specific increases and decreases in GLUT1 levels. However, such changes are not detected easily by analysis of total brain membranes, as employed in this study. Regulation of neuronal GLUT3 has been less extensively studied. During normal development, GLUT3 levels in rat brain increase in concert with synaptogenesis and functional maturity (Vannucci, 1994). Increased GLUT3 has also been observed with chronic neuronal excitation/depolarization and metabolic activity both in vitro (Maher and Simpson, 1994) and in vivo (Vannucci et al., 1994b). Thus, the increased GLUT3 levels reported here may relate to increased metabolic demand, which is reflected in increased glucose utilization, occurring early during recovery from hypoxia-ischemia.
Footnotes
Acknowledgment:
This work was supported by Grants R29HD31521 (S.J.V.) and P01HD30704 (R.C.V.). We would also like to thank Drs. Peter Davies for providing the SP-14 antibody and Ian A. Simpson for provision of the GLUT1 and GLUT3 antisera as well as for helpful comments and critical review of the manuscript.
