Abstract
Perfusion and diffusion-weighted magnetic resonance imaging (MRI) can demonstrate, respectively, cerebral ischemia and ischemic brain injury in the first several hours after onset of symptoms, when proton density and T2-weighted MRI may appear normal. It is hypothesized that these techniques could distinguish regions destined for infarction from those that will not progress to infarction. We provide preliminary evidence from an analysis of 19 patients with severely disabling clinical deficits attributable to ischemia in at least an entire division of the middle cerebral artery, that initial perfusion and diffusion MRI were more accurate than conventional MRI in predicting no, partial or complete improvement—17 of 19 cases (p < 0.0001) versus 10 of 19 cases, respectively. In the subset of patients studied within 6 h of onset, diffusion/perfusion MRI was an even better predictor than conventional MRI—11 of 12 versus four of 12, respectively. In this small sample of patients with severe clinical deficits, perfusion and diffusion MRI were highly accurate in distinguishing those who would improve from those who would not. These results need to be confirmed in a larger prospective study, which may support a future role in the initial screening, selection, and evaluation of patients with stroke for acute pharmacologic interventions.
Keywords
Improved understanding of the pathophysiology of ischemic neuronal injury (Collins et al., 1989; Pulsinelli, 1992; Siesjo, 1992) has led to the development of pharmacological therapies that have proven effective in limiting the severity of stroke and clinical deficits in experimental models and preliminary clinical trials. As many of these potential therapies have been entered into clinical trials, the promise of effective interventions in the near future has given a greater importance to the issue of prognosis very early in the course of the disease. Since some drugs currently in clinical trial carry the risk of serious, life-threatening complications (Brott et al., 1992; Haley et al., 1992), the need to distinguish those patients who may potentially improve as result of such drug therapies from those who will not has taken on an urgency. Standard neurodiagnostic imaging techniques, i.e., computed tomography (CT) and T1-(T1WI) and T2-weighted (T2WI) magnetic resonance imaging (MRI), give high false negative rates during the first day after onset of ischemic symptoms (Bryan et al., 1991; Yuh et al., 1991) and, therefore, such negative scans are unreliable predictors of good outcome. The newer experimental interventions in stroke therapy are best initiated during the first several hours after onset, when the potential for neuroprotective effect is greatest, but also when diagnostic uncertainty is greatest.
Two functional MRI techniques—perfusion and diffusion imaging—are able to delineate, respectively, areas of hypoperfusion and ischemic neuronal injury within minutes after induction of cerebral ischemia in animal models (Back et al., 1994; Busza et al., 1992; Minematsu et al., 1992; Moseley et al., 1990b, c ; van der Toorn et al., 1994). Perfusion magnetic resonance (MR) with bolus injection of gadolinium-containing contrast media, which map relative cerebral blood volume, identified hypoperfused tissue; diffusion-weighted imaging (DWI) detects changes in the self diffusion of water molecules that are associated with early cytotoxic edema in ischemic stroke. We previously reported that these techniques may be applied clinically to detect very early abnormalities in patients with acute stroke—earlier than are conventional T2-weighted images (T2WI) (Warach et al., 1992a; Warach et al., 1995; Warach et al., 1992b). Since perfusion and diffusion MRI can identify very early ischemic lesions, we investigated whether, in the initial hours following onset of ischemic symptoms, they are more accurate than is conventional MRI in predicting outcome in stroke.
METHODS
Patients
We studied 19 patients who underwent perfusion and/or diffusion MRI at a time when they had acute clinical deficits attributable to ischemia in a vascular territory of an entire division of one middle cerebral artery or greater. Patients comprised a subset of cases previously studied (Warach et al., 1992a; Warach et al., 1995; Warach et al., 1992b). Patient characteristics are described in Table 1. Clinical criteria used for inclusion were presence, at the time of initial MRI study, of a moderate to severe contralateral hemiparesis (inability to maintain limb elevated, to use limb, and to overcome moderate resistance) in combination with one or more of the following: a severe aphasia, contralateral hemisensory neglect, or contralateral homonymous hemianopia. Deficits at the time of initial MRI study corresponded to National Institute of Health Stroke Scale score ≥10 and Modified Rankin Scale Score of 4 or 5 (0–5 scale). In some cases, scores were determined retrospectively from chart review. Patients receiving either urgent endarterectomy or experimental neuroprotective agents were not included, otherwise the sample was not controlled for use of anticoagulation or other therapies. Presumed etiology of stroke was classified using the TOAST criteria as large artery atherosclerotic, cardiogenic embolism, small vessel, other, or undetermined (Adams et al., 1993). Clinical follow-up was available in all cases at 1 week (±1 day), and this was used as the uniform follow-up time. Of the 12 patients with persistent deficits at 1 week, follow-up information at ≥30 days was also available in 10 cases, but did not change the classification of outcome. There were two deaths resulting from the stroke (cases 11 and 17), which were classified as no improvement.
Summary of patient data
study performed using echo planar MRI technique.
entire portion of MCA territory in that slice.
entire portion of ACA and MCA in that slice.
T2WI, T2 weighted images; DWI/PMR, diffusion weighted imaging/perfusion MR; Neg, no new stroke found; partial, lesion seen that partially but not fully explains clinical deficit or partial recovery from initial deficit; R, right; L, left; HP, hemiparesis; HH, homonymous hemianopia; F, frontal lobe; T, temporal lobe; P, parietal lobe; O, occipital lobe; WM, hemispheric white matter; BG, basal ganglia; IC, internal capsule; LAA, large artery atherosclerosis; CE, cardioembolic; ICA, internal carotid artery; NA, not available.
MRI studies
All studies were performed on a 1.5 Tesla MR whole-body system (Siemens AG, Erlangen, Germany). Fifteen subjects had both perfusion and diffusion imaging studies; technical factors including inadequate intravenous access, uncontrollable head motion, or severe agitation precluded both studies in four subjects. The first 13 subjects were studied with a standard clinical imager with conventional gradient sets. The next six patients were studied using an echo planar imaging (EPI) technique when the magnet was modified with EPI capable gradients. EPI minimized problems with agitation or head motion and permitted multislice acquisitions. Time to first MRI study was measured from the time (rounded to the nearest hour) that deficits were first noted. All studies were performed within the guidelines of the Committee on Clinical Investigations of Beth Israel Hospital.
Conventional imaging
T1WI proton-density-weighted and T2WI images were obtained using a spin-echo technique. For spin-echo imaging, scan parameters varied somewhat among patients; typical parameters were repetition time (TR)/echo time (TE) 600 ms/15 ms for T1WI, and TR/TE = 2400 ms/30 ms, 80 ms for T2WI double echo sequences. Section thickness was 5 mm, 20 slices, field-of-view = 23 × 17 cm, acquisition matrix 256 × 160, one or two excitations. Follow-up conventional MRI or CT scans were obtained on all patients except case 12.
Diffusion-weighted imaging
Principles and techniques for diffusion imaging in patients with acute stroke have been previously described (Warach et al., 1992a; Warach et al., 1995). In 13 subjects, a turboSTEAM technique was used giving a diffusion weighting (b-value) of ∼372 s/mm2 on a single 10 mm axial imaging slice through the area of brain suspected from the clinical deficit. In two patients, head movement created such severe artifact that DWI yielded no usable results; in these cases, perfusion imaging was successful. Six patients were studied using EPI and higher gradient strengths when these became available. This technique permitted greater diffusion sensitivity (up to b = 1271 s/mm2) using a single shot spin echo, echo planar technique), multislice contiguous imaging, and images free from motion artifact. Parameters used for EPI were TE = 100 ms, slice thickness = 5 or 7 mm, acquisition matrix = 128 × 128, field-of-view = 25–32 cm; readout period was ∼64 ms and acquisition time per image was ∼164 ms. Diffusion sensitivity, b-value, comprised two values (0 and maximum) or seven values by incrementing the diffusion gradient strength (see Warach et al., 1995 for details) up to a maximum b-value of 1205 or 1271 s/mm2. Either a single set of 20 heavily DWIs were acquired in 3 s or all seven diffusion sensitivities at all slices were acquired in a single measurement with a total acquisition time of 48 s.
Perfusion imaging
Principles and techniques for perfusion imaging in patients with acute stroke have been previously described (Belliveau et al., 1990; Edelman et al., 1990; Rosen et al., 1990; Warach et al., 1992b). During repetitive scanning with a susceptibility-weighted (T2-weighted) pulse sequence, a bolus injection of 20 cc of gadopentatate-dimeglumine was administered intravenously via an 18-gauge catheter placed in an antecubital vein by manual injection for ∼5 s followed by a saline flush. For 12 patients, T2-weighting was obtained on a single slice using a spoiled gradient-echo pulse sequence, FLASH, with TR/TE/flip angle/number of excitations = 35 ms/25 ms/10°/L, slice thickness = 8 mm, acquisition matrix = 256 × 80, and rectangular field-of-view = 23 cm × 17 cm. Scan time per image was ∼.8 s with no interscan delay; 25 images were acquired. Sequential T2-weighted MR images were displayed in a cine loop, which shows the transient drop in signal intensity as the bolus passes through, and the area of ischemia was clearly delineated when present. When EPI became available on our imaging system, five patients had multislice perfusion studies using a TE = 60 ms, slice thickness = 7 or 8 mm, acquisition matrix = 128 × 128, and field-of-view = 25 to 32 cm. A delay of 1.5–2 s occurred between sequential acquisitions at each slice position. Two patients had inadequate intravenous access for the study.
Data analysis
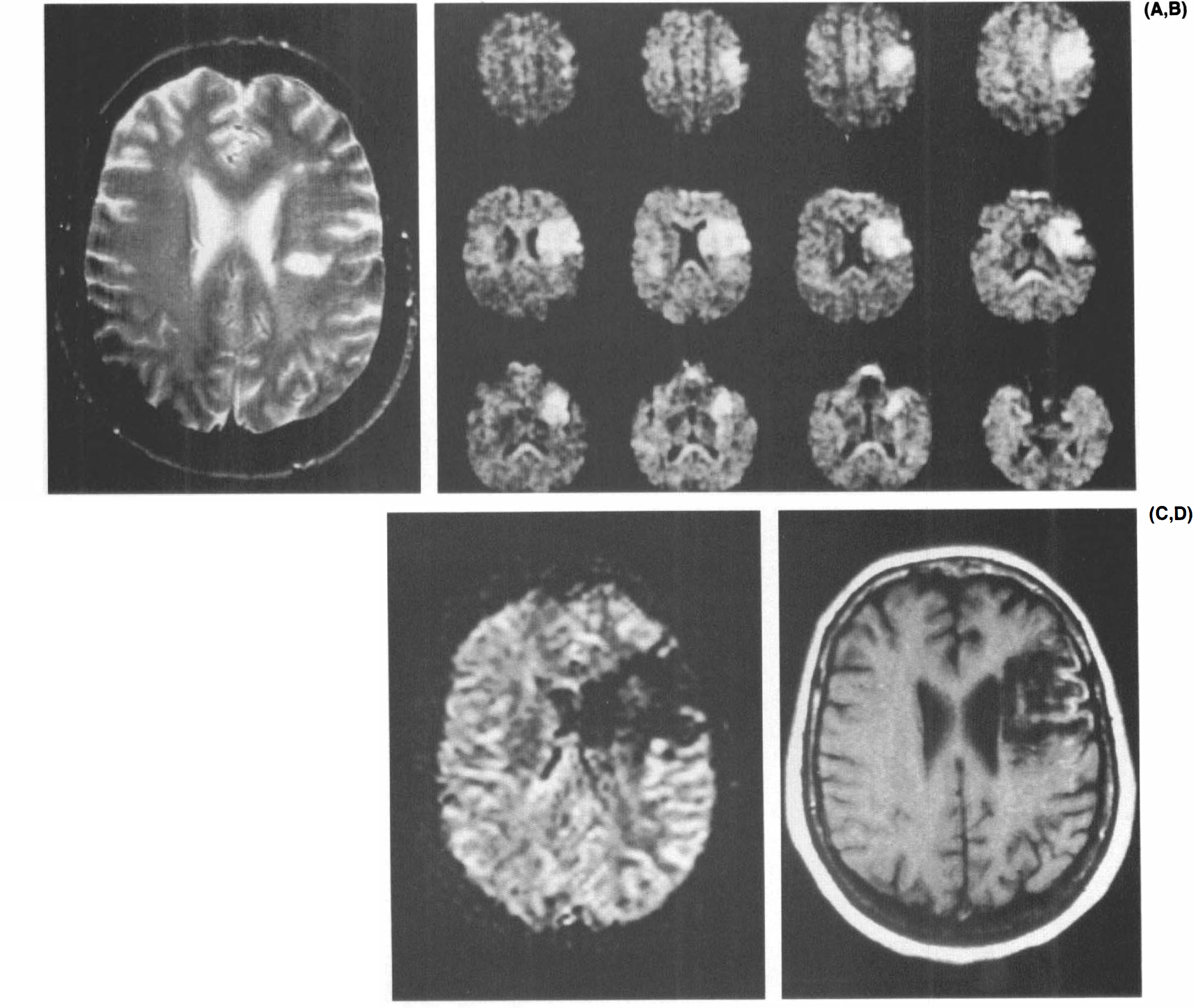
MRI scans acquired in the acute phase were analyzed without knowledge of the patients' clinical outcomes. Lesions were identified as one or more of the following: areas of abnormal hyperintensity in T2WI, areas of hyperintensity in DWI, and well-demarcated areas of reduced perfusion on dynamic blood volume imaging. Examples of the lesions are depicted in Fig. 1. A case was judged to have lesions explaining the clinical deficits if the lesion found on MRI scans was in an anatomical region that could account for the clinical deficits at the time of the scan. Improvement on follow-up examination was graded as none (little or no improvement, or worse), partial (substantial improvement in strength or ability to perform tasks, but still with a disabling deficit requiring assistance corresponding to improvement in Modified Rankin Scale score from 5 to 2, 3 or 4; or 4 to 2 or 3), or complete (complete or near complete resolution of deficits, slight deficits, or disabilities present but patient able to resume baseline level of function, Modified Rankin Scale Score 0–1). Diagnosis of clinical improvement was agreed upon by two neurologists. MRI scans were considered to accurately predict outcome if a match occurred between an explanatory lesion on acute MRI and no improvement, a partially explanatory lesion and partial improvement, or no explanatory lesion and complete recovery. Because of the categorical nature of the data, statistical analyses were performed using a χ2 test for the three by three contingency table of Fisher's exact tests for the dichotomous categories. Significance level was set at p < 0.05.
MRI results from partially explanatory lesion.
RESULTS AND DISCUSSION
Data from the 19 patients studied are summarized in Table 1. Results for proton-density weighted images and T2WI were equivalent; only results for T2WI are reported. Table 2 describes the relative accuracy of T2WIs and diffusion/perfusion MRI in predicting clinical outcome for all cases and for only those cases studied within 6 h from onset of symptoms.
Predicting improvement by MRI
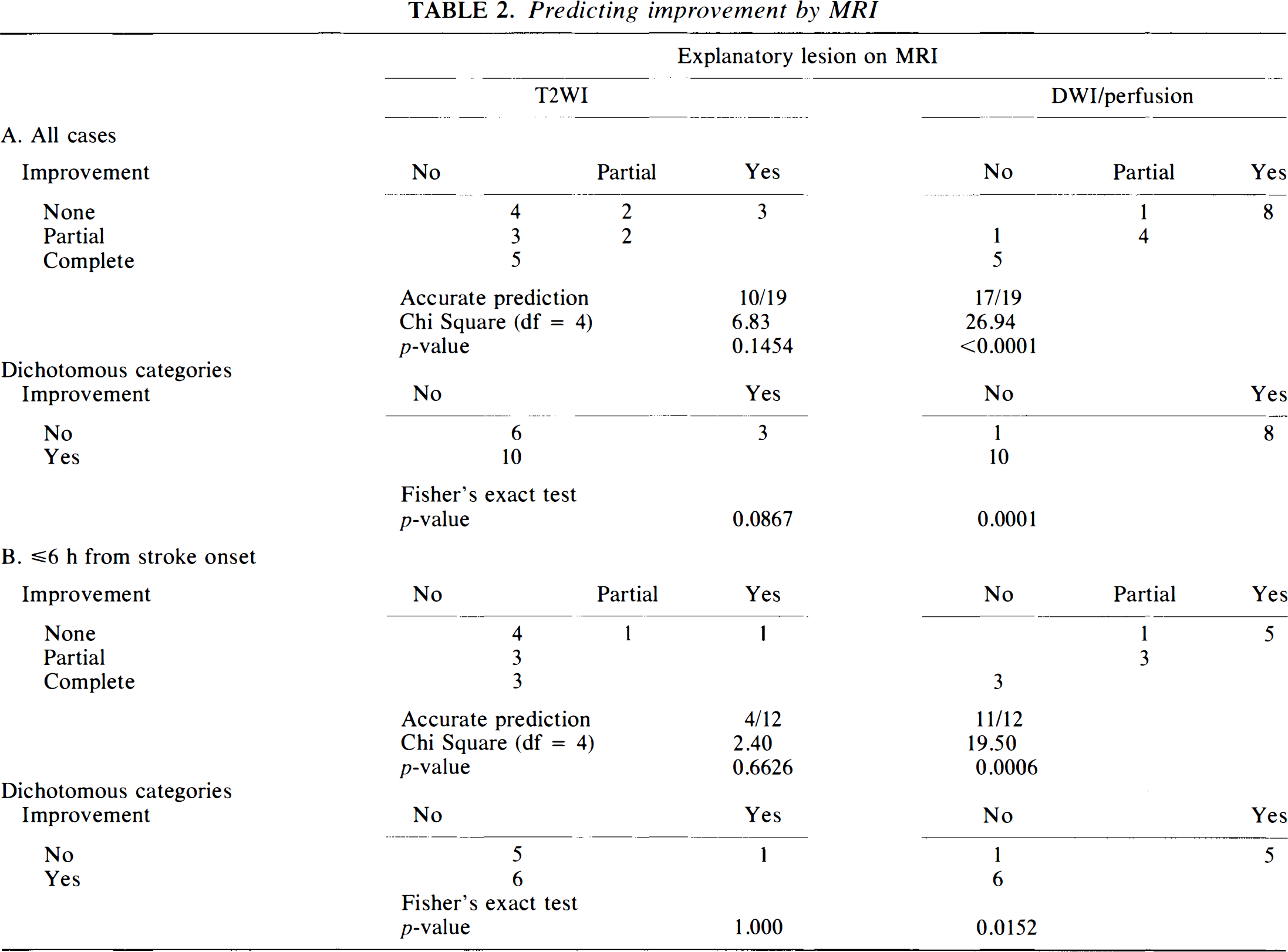
Overall, diffusion and perfusion MRI correctly predicted outcome category in 17 of 19 (89%) cases (χ2 = 26.938, p < 0.0001), whereas conventional MRI correctly predicted outcome in 10 of 19 (53%) cases (not significant). In the subset of patients studied at ≤6 h, the difference was greater; diffusion/perfusion MRI was an accurate predictor in 11 of 12 (92%) cases (χ2 = 19.50, p = 0.0006) and conventional MRI was an accurate predictor in 4 of 12 cases (33%; not significant). In no case did conventional or diffusion/perfusion MRI overestimate clinical outcome, i.e., predict worse outcome than actually occurred. One false negative result for the diffusion/perfusion MRI involved a hemispheric white matter lacune that was not in the single imaging slice selected for study. In the other erroneous prediction (case no. 6), a prediction of partial improvement was made following a deep hemispheric lesion. The clinical correlation predicted from the lesion in this latter case was incorrect as the patient did not improve.
Table 2 also summarizes results in a dichotomous fashion, i.e., whether the scans fully explained the deficits and whether any substantial improvement (partial or complete) occurred. When acute diffusion or perfusion MRI did not fully account for the deficits, significant improvement occurred in 10 of 11 (91%) patients, whereas when conventional T2WI did not fully account for the deficits, the improvement was noted in 11 of 18 (61%) patients. Cases of complete improvement had no lesions in five of five (100%) cases. For cases with <6 h of ischemic symptoms, conventional MRI correctly identified only one of six patients who would not improve, whereas diffusion/perfusion MRI identified five of six. Figure 1 illustrates the imaging results for a patient in whom no acute stroke was seen in T2WI but in whom a lesion smaller than expected from clinical examination was found in diffusion and perfusion images. The patient improved from severe disability with need for constant nursing care to moderate disability.
In this small sample of patients with clinical deficits diagnostic of massive hemispheric ischemia, we have found that early diffusion and perfusion MRIs are accurate in predicting clinical outcome. This advantage of diffusion and perfusion MRIs versus conventional MRI or clinical examination is greatest during the first 6 h after onset, the period considered most critical for acute therapeutic intervention (Fisher and Bogousslavsky, 1993; Le vine and Brott, 1992). Although animal studies have suggested that some early, less severe diffusion abnormalities are potentially reversible (Kucharczyk et al., 1991; Lo et al., 1994; Minematsu et al., 1993a, b ). In the present clinical study, the presence of a diffusion abnormality invariably led to permanent neurological deficits even if T2WI was normal initially.
This study was limited to patients with severe deficits suggestive of large areas of cerebral ischemia. For the purpose of this study, only these unambiguous substantial clinical deficits and broad categories of clinical improvement were considered in order to maximize the probability of finding a large hemispheric lesion and to minimize the chance of bias or disagreement between examiners. We selected large clinical deficits for the initial evaluation of these techniques, since if diffusion/perfusion MRI techniques are not accurate predictors of outcome for large deficits, they are unlikely to be so for smaller ones. Prior to the availability of EPI, a large hemispheric lesion was also preferred to minimize the chance of false negative diffusion/perfusion MRI due to inaccurate selection of imaging slice in the single slice technique. Results for smaller infarcts may be more variable.
Three cases studied within 6 h had normal diffusion and perfusion studies and complete recovery. These patients had atrial fibrillation and probable cardiogenic emboli. We presume that by the time of scanning, spontaneous lysis and reperfusion had occurred prior to permanent ischemic injury, with a latency of several hours before complete clinical recovery. Subtle, potentially reversible diffusion abnormalities could have been missed by the single slice moderate diffusion weighting of the turboSTEAM cases.
The false negative diffusion and perfusion case in this study occurred because a subtotal lesion was present outside the single imaging slice selected for study. In >80 cases of stroke studied with DWI (unpublished observations), false negatives (true lesions not found in DWIs) occurred only due to incorrect imaging slice selection for those studies for which only a single slice could be studied. Since implementation of multislice EPI on our system, diffusion and perfusion imaging are no longer limited to a single slice. We have begun to screen the entire brain, and lesions as small as 4 mm can be detected. Echo planar DWI of the entire brain can be completed in 3 s, and perfusion imaging of the entire brain in <1 min; thus their use as screening tests would not unduly delay urgent therapies. In this series, no errors in prediction occurred for the six cases acquired with EPI.
The use of rapid diffusion and perfusion MRI techniques in the evaluation of early stroke promises to give more accurate localization and better prognosis than are possible by conventional imaging methods alone. They will also provide quantitative measures of lesion size and, possibly, severity and viability. Thus, these techniques will act to interactively guide the development and implementation of future pharmacologic therapies. Very early perfusion and diffusion MRIs could, potentially, provide the information necessary to assess the neuroprotective effects of drugs, identify areas of persistent ischemia that indicate areas of brain at risk of infarction, and differentiate patients who may spontaneously improve from those who will not. For example, diffusion/perfusion MRI techniques might be used to avoid high risk interventions such as arteriography or thrombolytic therapy in patients likely to improve spontaneously. In addition, the combination of DWI and perfusion MRI may be able to identify the ischemic penumbra and identify regions at risk for evolution into infarction (Moseley et al., 1990a). Thus, these techniques may prove to be useful for screening and monitoring patients for pharmacologic interventions with neuroprotective drugs. It is encouraging that combined perfusion and metabolic studies with positron emission tomography in stroke patients within the first 24 h are predictive of functional outcome, distinguishing patients with no spontaneous recovery from those with complete spontaneous recovery or with variable outcome (Marchal et al., 1993). Whether diffusion and perfusion MRI techniques can live up to the promise of early prediction of outcome requires a larger scale prospective study of the sensitivity and specificity of these tests than reported on here.
Footnotes
Acknowledgment:
Supported by grants from the National Institute of Neurological Diseases and Stroke (NS01634), the Harcourt General Charitable Foundation, and the Friends of Beth Israel Hospital. We thank Dr. Wei Li for assistance.
