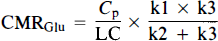Abstract
To investigate the influence of cholinergic projections from the basal forebrain on cerebral cortex metabolism, we evaluated the cerebral metabolic rate of glucose (CMRGlu) after selective inhibition of cholinergic neurons in the rat basal forebrain using the pyruvate dehydrogenase complex inhibitor 3-bromopyruvic acid (BPA), and compared the results with those obtained after lesioning the basal forebrain with ibotenic acid, as well as with those from a sham-operated control group. CMRGlu was measured using positron emission tomography (PET) with [18F]-2-fluoro-2-deoxy-D-glucose (FDG). Three days after surgery, CMRGlu and k3 (phosphorylation of FDG) were reduced similarly in the frontal cortex on the BPA-injected side and in the ibotenic acid-treated group, whereas K1 (transport rate of FDG from the plasma to brain) showed no marked changes. At 3 weeks postoperatively, the CMRGlu and k3 of the frontal cortex in both groups recovered to levels similar to those of the sham-operated group. The main difference between the BPA and ibotenic acid groups was that CMRGlu showed mild reduction on the side contralateral to the operation in the former, while such reduction was confined to the ipsilateral hemisphere in the latter. The present results indicate that the cholinergic system in the basal forebrain regulates cerebral cortex glucose metabolism through direct excitation of cortical neurons.
Keywords
Cholinergic projections from the basal forebrain to the cerebral cortex affect cortical metabolism, as demonstrated by the reduction in cerebral metabolic rate of glucose (CMRGlu) observed after lesioning of the basal forebrain (London et al., 1984; Orzi et al., 1988; Kiyosawa et al., 1989). In these studies, however, the cholinergic system could not be inhibited selectively, because the methods of lesioning the basal forebrain, including electrolytic coagulation or application of neurotoxins, damaged neuronal structures other than those of the cholinergic system (Pepeu et al., 1985). In the present study, we evaluated frontal cortical CMRGlu levels after selective inhibition of the cholinergic system in the basal forebrain of rats.
3-bromopyruvic acid (BPA) is an inhibitor of pyruvate dehydrogenase complex (PDHC) (Arendt et al., 1990), which has been reported to reduce the cholinergic neuronal activity of the cerebral cortex and cortical choline acetyltransferase activity. The procedure employing this inhibitor allows the selective cholinergic neuronal suppression of the cerebral cortex without concomitant basal forebrain destruction, and also excludes the possibility of the involvement of other ascending fiber systems such as monoaminergic or serotonergic projections (Dubois et al., 1985; Lerer et al., 1985). We also examined changes in CMRGlu levels with lesioning of the basal forebrain using ibotenic acid and compared the results with those of BPA administration to investigate the specificity and selectivity of the cholinergic suppressive effects of these procedures.
Serial CMRGlu measurement in the same rat was performed at 3 days and 3 weeks after operation using dynamic positron emission tomography (PET) with [18F]-2-fluoro-2-deoxy-D-glucose (FDG). This method allows monitoring of the time course of CMRGlu changes within the same animals as well as determination of glucose metabolic rate constants.
MATERIALS AND METHODS
Materials
Fifteen male Wistar rats weighing 250–350 g were examined. These rats were arbitrarily divided into the three groups: BPA-treated (n = 5), ibotenic acid-treated (n = 5), and sham-operated (n = 5) animals.
Surgical procedures. Rats were anesthetized with intraperitoneally injected sodium pentobarbital (20 mg/kg) and placed in a stereotaxic apparatus, the incisor bar of which was set 3.3 mm below the interaural line. The stereotaxic coordinates of the injection site were as follows: 1.4 mm posterior to the bregma, 2.8 mm right of the midline, and 6.9 mm below the surface of the brain, according to Paxinos and Watson's rat brain atlas (Paxinos et al., 1986).
Either a 0.1 μl volume of BPA solution consisting of 2 mM BPA and 0.2 mM thiamine pyrophosphate dissolved in 0.1 M phosphate buffered saline (PBS) (Arendt et al., 1990) or 0.1 μl of 2% ibotenic acid dissolved in 0.1 M phosphate buffer (pH = 7.3) was injected with a Hamilton microsyringe over a 5-min period at the right side of the nucleus basalis magnocellularis (NBM); the needle was removed over another 5-min period. Sham-operated rats were injected with 0.1 μl of 0.2 mM thiamine pyrophosphate dissolved in 0.1 M PBS in the same manner as the other injected groups.
PET imaging. PET examinations were performed in the same rats 3 days and 3 weeks after operation using a high-resolution PET scanner, designed for animal use, (SHR-2000, Hamamatsu Photonics K.K., Hamakita, Japan) equipped with four detector rings affording seven-slice imaging. The spatial resolution at the center was 3 mm in-plane at full width at half maximum and 4.8 mm axially. The averaged direct slice sensitivity was 2.3 kcps/μCi/ml and the cross-slice sensitivity was 3.8 kcps/μCi/ml. The total system sensitivity was 20.7 kcps/μCi/ml including the scatter component (Watanabe et al., 1992).
Prior to imaging, rats were prepared by venous and arterial catheterization under sodium pentobarbital anesthesia and then placed in the prone position in the PET camera. The head of the rat was fixed horizontally with a molded plastic head-holder and positioned with the aid of a laser beam that allowed accurate repositioning for the second measurement 3 weeks later. A 20-min transmission scan using 68Ge/68Ga was performed for attenuation correction. Thirty min after anesthesia, 37 MBq (1 mCi) of FDG was injected intravenously and arterial blood collected at the following times: 10, 30, 50, and 90 s, and 2, 3, 5, 10, 20, 30, 40, 50, and 60 min. Serial dynamic PET data were also obtained simultaneously. Scans with a frame duration of 2 min were acquired successively; a total of 30 dynamic frames were obtained. Plasma glucose was determined twice, once prior to injection of FDG and once again upon cessation of scanning; the averaged value was used for CMRGlu calculation. During PET examination, systemic arterial blood pressure and arterial blood gases were monitored periodically and kept within physiological limits (blood pressure, 99.5 ± 6.1 mmHg, PaCO2: 39.1 ± 1.3 mmHg, PaO2: 93.7 ± 1.5 mmHg, and pH: 7.41 ± 0.02), and the rectal temperature maintained constant between 36 and 37°C with a heating pad.
Scanned data were reconstructed by Butterworth filter and Shepp-Logan filter as a 180 × 180 matrix image, with a pixel size of 1 × 1 mm.
PET data analysis. Each region of interest (ROI) were determined on the last 20 min of summed PET image data, and the same ROI were then transferred onto the dynamic scan data to obtain regional brain time activity curves. With reference to the magnetic resonance images (MRIs) of each animal [1 mm slice thickness, 3 mm slice gap, obtained using a 1.5T imager (Signa Advantage, General Electric Co. Milwaukee, U.S.A.) with the spin echo technique; TR = 3000 ms, TE = 100 ms] and the standard rat brain atlas, each ROI was placed symmetrically and bilaterally in the frontal cortex and the basal forebrain (Fig. 1). We measured cortical CMRGlu in the frontal cortex, because this region receives projections from vast numbers of cholinergic afferents from the basal forebrain (Saper, 1984) and because measurements in the cortex would not be confounded by direct damage produced by the injection needle. ROIs in the basal forebrain were drawn over the affected NBM excluding the cerebral cortical region. Sizes of the ROIs ranged from 20 to 36 mm2.
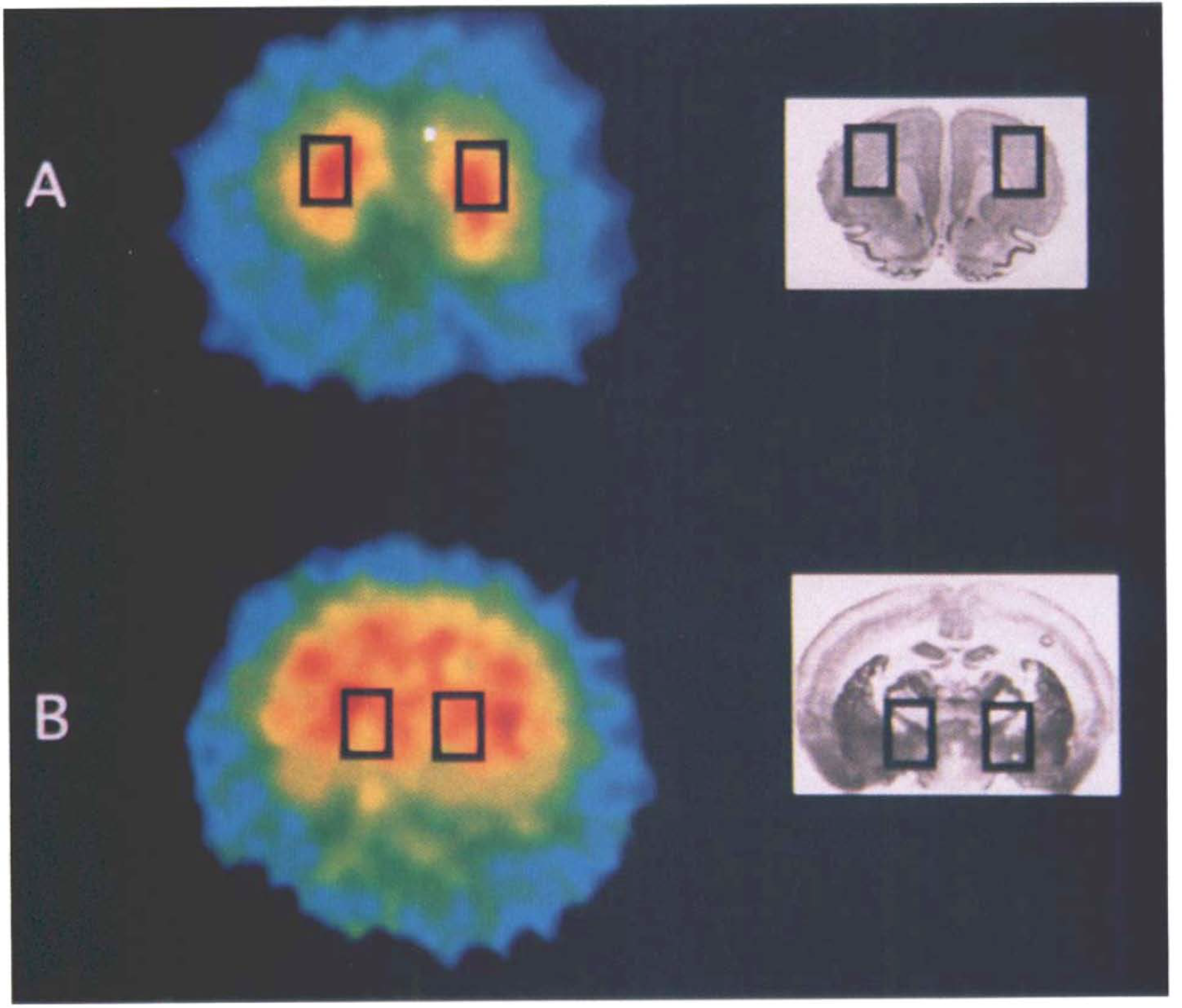
Regions of interest on the coronal sections of the rat brain. Rectangular regions of interest are drawn bilaterally covering the frontal cortex
Rate constants of glucose metabolism were determined by a nonlinear least squares algorithm fitted to the measured arterial and brain time activity curves (Fukuyama et al., 1989), in which the vascular space was fixed as 4% (Ingvar et al., 1991) of total brain activity. CMRGlu was calculated using the obtained rate constants by the following formula:
where Cp is the plasma glucose level, LC denotes the lumped constant, k1 is the rate constant for the FDG transport from the plasma to brain, k2 is the rate constant for the efflux from the brain to plasma compartment, and k3 is the rate constant for the phosphorylation of FDG. We used 0.58 as the value of LC (Lear and Ackerman, 1989).
Statistics. CMRGlu and the rate constants at 3 days postoperatively for the three groups were analyzed using a two-way analysis of variance (ANOVA) with respect to location, i.e., injected or noninjected side of the frontal cortex or basal forebrain, and procedure, i.e., sham-operated, ibotenic acid-treated, or BPA-treated groups. When the two-way ANOVA revealed cross reaction between two factors, a one-way ANOVA, including locations of ROIs and types of operation, was performed. When no cross reaction was observed, one-way ANOVA was performed comparing the operation effects in either hemisphere separately. Multiple comparisons among groups were performed with Scheffe's F-test. These parameters at 3 weeks postoperatively were also analyzed using the same test procedures. A p value of <0.05 was considered to be statistically significant.
Histology. The injection site was verified by administering 0.05 μl of wheat germ agglutinin (WGA)-horseradish peroxidase (HRP) conjugate at the site with the same coordinates as the BPA or ibotenic acid injection. For histochemical demonstration of WGA-HRP, the section was treated with tetramethylbenzidine (Mesulam, 1978) and then counterstained with 1% cresyl violet (Fig. 2A) to visualize the center of the injection site. The brain was removed from all rats after the 3-week PET examination to determine the injection site, as well as the spread of the necrotic area in the case of ibotenic acid treatment using cresyl violet staining (Figs. 2B and C).
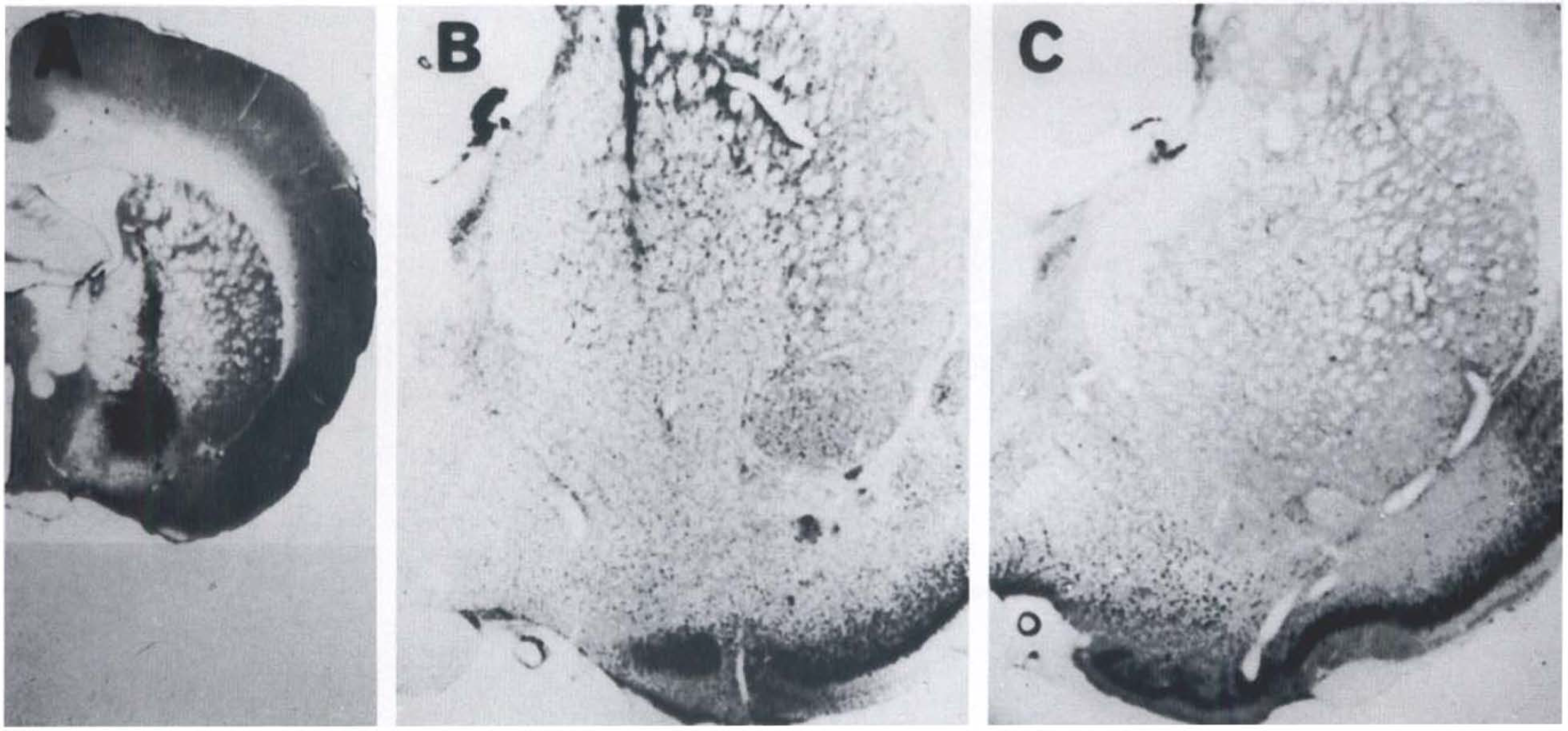
Site of wheat germ agglutinin-horseradish peroxidase conjugate (WGA-HRP) injection in the basal forebrain
RESULTS
Three-days postoperatively (Table 1)
CMRGlu and rate constants 3 days postoperatively
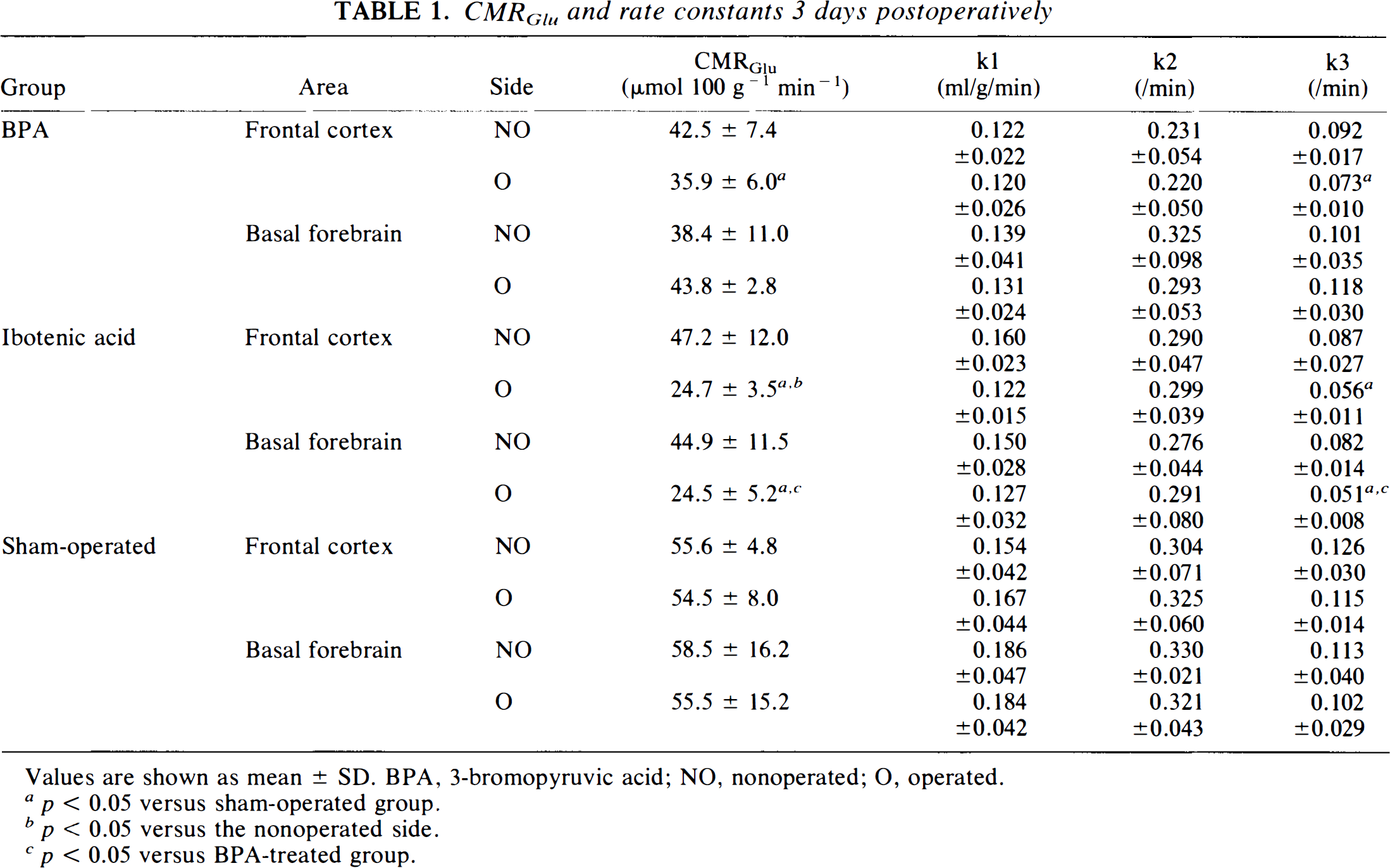
Values are shown as mean ± SD. BPA, 3-bromopyruvic acid; NO, nonoperated; O, operated.
p < 0.05 versus sham-operated group.
p < 0.05 versus the nonoperated side.
p < 0.05 versus BPA-treated group.
Two-way ANOVA revealed significant alterations in the CMRGlu for the experimental procedures (p < 0.0001) and the locations of ROI (injected side versus noninjected side) (p < 0.01) with cross-reactivity (p < 0.05). Thus, we also performed a one-way ANOVA including the locations of ROI and the differences in procedures.
This one-way ANOVA revealed that the CMRGlu of the frontal cortex ipsilateral to the operated side was significantly lower than that in the sham-operated group in both BPA and ibotenic acid-treated groups. The injected and noninjected sides showed a marked difference in the ibotenic acid-treated, but not in the BPA-injected, group (Fig. 3). The CMRGlu in the basal forebrain ipsilateral to the operated side was significantly reduced in the ibotenic acid-treated group compared to the sham-operated and BPA-injected groups.
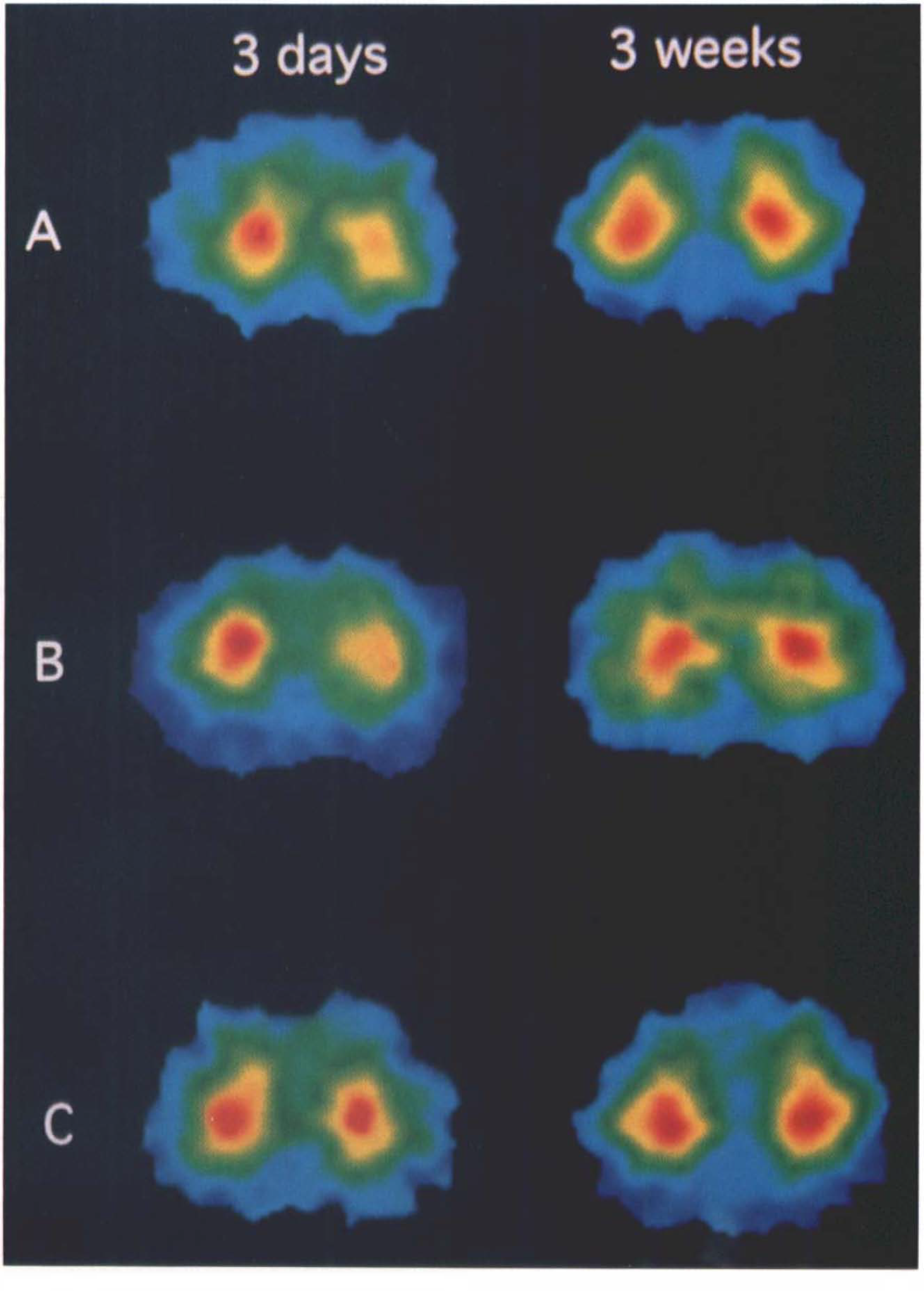
PET images of FDG uptake in the frontal cortex 3 days and 3 weeks after operation.
Two-way ANOVA indicated significant changes in k3 for the experimental procedures (p < 0.0001) and the locations of ROI (p < 0.01), but since there was no cross reaction between these two factors, we compared them separately. The rate constant k3 was significantly lower in the ipsilateral frontal cortex in BPA- and ibotenic acid-treated rats than in the sham-operated animals. In the basal forebrain, k3 was decreased significantly in the ibotenic acid-treated group compared to either the BPA-treated or sham-operated group.
Two-way ANOVA indicated significant differences in k1 and k2 for the experimental procedures (k1, p < 0.05; k2, p < 0.001). However, a post-hoc Scheffe's f-test could not detect any significant differences among the kinds of operations.
Three-weeks postoperatively (Table 2)
CMRGlu and rate constants 3 weeks postoperatively
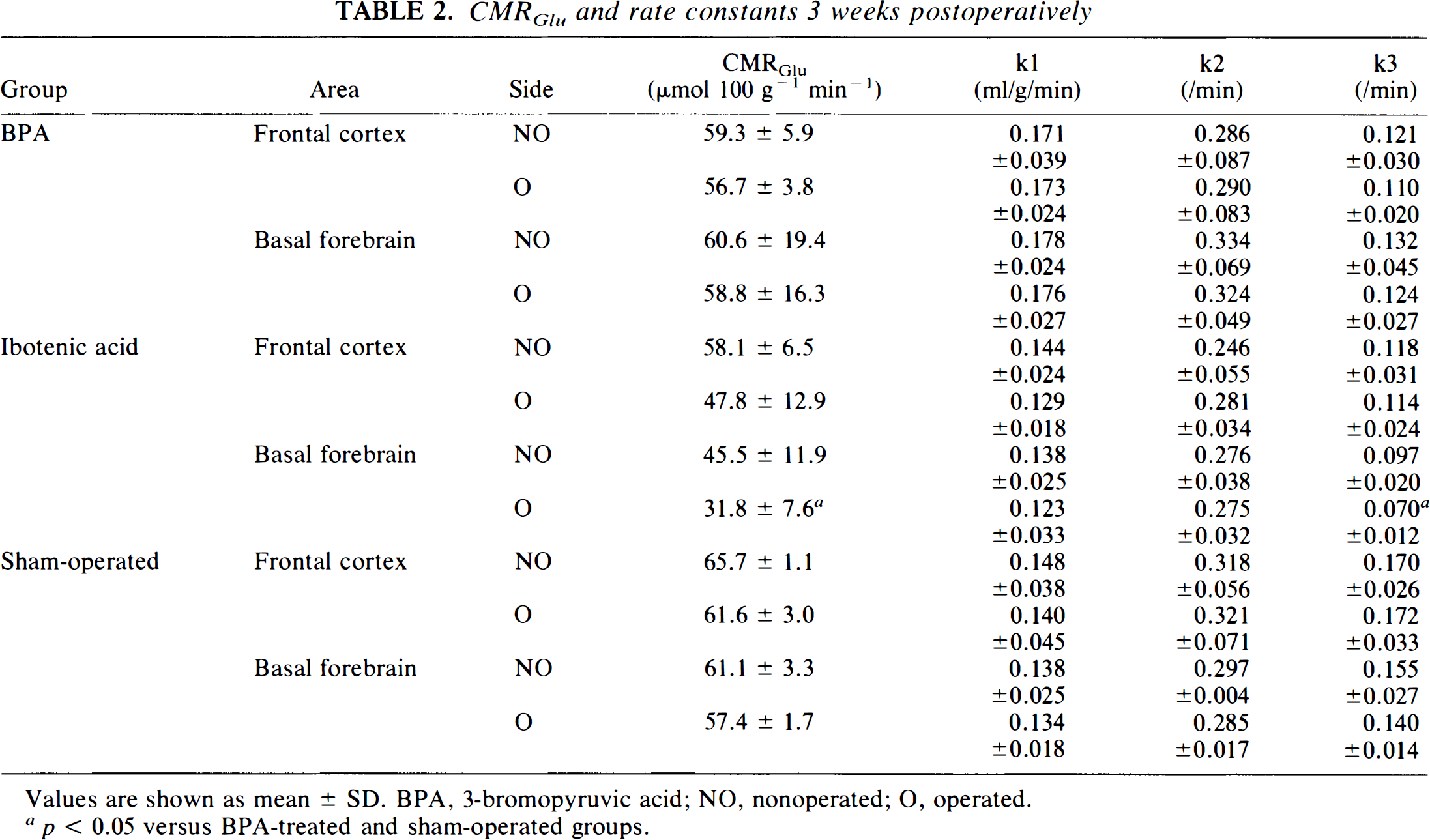
Values are shown as mean ± SD. BPA, 3-bromopyruvic acid; NO, nonoperated; O, operated.
p < 0.05 versus BPA-treated and sham-operated groups.
Neither CMRGlu nor k3 was reduced in the frontal cortex in any of the three groups (Fig. 3). However, CMRGlu and k3 in the ipsilateral basal forebrain in the ibotenic acid-treated group remained low.
DISCUSSION
This experimental protocol is subject to several limitations, the first of which is the possibility of an anesthetic effect in the rats examined during PET scanning. The anesthesia level was, however, not so deep that the CMRGlu in this study was higher than previously reported values obtained under deep pentobarbital anesthesia (Otsuka, 1991), but the influence of anesthesia cannot be excluded completely. Therefore, we compared experimental CMRGlu with that measured in a sham-operated group under similar anesthesia conditions. The second limitation is that the sizes of the ROIs covered only a small portion of the rat brain; thus, data obtained for areas less than twice the spatial resolution of the PET scanner may have been underestimated (Baron and Miyazawa, 1991). A final limitation is that, because we compared two different scanning data sets, we paid careful attention when positioning the head in the second scan—to fix it in the same position as in the first scan using the head holder; nevertheless, there might have been a small positioning error. Therefore, the present PET data were subject to the effects of anesthesia, partial volume effect, and positioning error, and these factors might have caused slight deviation of the observed data from actual values. However, there was no marked difference from the reported value for any of the metabolic rate constants obtained under the control conditions (Redies et al., 1987; Ingvar et al., 1991). Therefore, the present experimental procedure seems to be valid.
We found that inhibition of the cholinergic system in the basal forebrain reduced cortical CMRGlu to a degree comparable to that obtained by lesioning the basal forebrain by ibotenic acid administration. Our findings indicate that the cholinergic system in the basal forebrain affects cortical neuronal activity and metabolism. No reduction was apparent in k1, but k3 was significantly decreased, which suggests that the observed reduction in cortical glucose metabolism was due to diminished glucose phosphorylation and that the glucose transport rate was not significantly affected by cholinergic denervation.
In the ibotenic acid-injected group, the CMRGlu in the ipsilateral basal forebrain was decreased. This reduction reflected the damage of both cholinergic and noncholinergic neurons in the basal forebrain as a result of the neurotoxicity of ibotenic acid. The reduced CMRGlu in the cerebral cortex after ibotenic acid lesioning would include the observed damage to these two types of neurons originating from the basal forebrain, and, therefore, it cannot necessarily be concluded from such experiments that only cholinergic input from the basal forebrain affects cortical glucose metabolism. We applied selective inhibition, however, of the cortical cholinergic system with BPA. BPA irreversibly inactivates PDHC, which has a regulatory function in carbohydrate and acetylcholine metabolism in brain tissue (Maldonado et al., 1972; Lowe and Perham, 1984) and catalyzes the flux-generating step of the tricarboxylic acid cycle. Since <1% of pyruvate comprises the substrate for acetylcholine synthesis (Tucek and Cheng, 1974; Gibson and Blass, 1983), acetylcholine synthesis may be more affected by PDHC inhibition than is carbohydrate metabolism. Our study disclosed no significant reduction in CMRGlu in the BPA-injected basal forebrain, indicating that BPA administration did not substantially alter glucose metabolism via its inhibitory action on PDHC. Thus, we can exclude the possibility that the inhibited PDHC transported to the cortex from the basal forebrain was related to the observed CMRGlu reduction in the cortex.
The dose of BPA used in the present study was sufficient to inhibit PDHC activity by ∼90% at maximum in vitro and ∼40–80% in vivo (Arendt et al., 1990). The degree of CMRGlu reduction in the BPA-treated group was slightly less marked than that in the ibotenic acid-lesioned group. Although the difference was not statistically significant, cortical glucose metabolism was not completely suppressed by BPA, in contrast to the effect of ibotenic acid, and a proportion of PDHC was speculated to function in acetylcholine production in the cortex. The reduction of cortical CMRGlu in the BPA-treated group was not significantly different interhemispherically, in contrast to the significant difference observed between hemispheres in ibotenic acid-treated rats. The reason for this difference is unclear at present, but we speculate that the injected BPA may have spread and reached to contralateral side of the basal forebrain, resulting in equivalent suppression of PDHC in the cholinergic neurons of the contralateral basal forebrain. Thus, when localized and complete unilateral cholinergic suppression is required, ibotenic acid should be injected. On the other hand, diffuse but specific suppression of cholinergic systems in the cerebral cortex could be expected when BPA is injected.
The cholinergic system in the basal forebrain is related to vasodilatation and excitation of cortical neurons (Casamenti et al., 1986; Dauphin et al., 1994). Both the increase in cerebral blood flow (CBF) reported after basal forebrain stimulation (Biesold et al., 1989; Lacombe et al., 1989; Adachi et al., 1990; Linville and Arneric, 1991;) and the decrease in CBF described after lesioning of the basal forebrain (Gomi et al., 1991) support the concept that the activity of the basal forebrain cholinergic system is related to cortical vasodilatation. It was also reported that CBF does not change after lesioning of the basal forebrain (Scremin et al., 1991; Namba et al., 1991). One biochemical study has suggested that the perimicrovascular cholinergic terminals in the cerebral cortex do not originate from the basal forebrain (Galea et al., 1991). It is reasonable to speculate that the vasodilative action of the cholinergic projections originated from the basal forebrain may be mediated through cholinergic neuronal activation (Lamour et al., 1982a). The present results indicated that there were no marked alterations in the value of k1 in any experimental group. The constant k1 represents glucose influx from blood vessels into the brain parenchyma and is the product of CBF and the glucose extraction fraction by the brain (Crane et al., 1983). Thus, glucose transport from blood vessels into the brain was not influenced by cholinergic input suppression. The absence of marked deviations in k1 and in the calculated distribution volume, Vd, of FDG (BPA: Vd = 0.409 ± 0.049; ibotenic acid: Vd = 0.349 ± 0.064; sham-operation: Vd = 0.377 ± 0.072) suggests the possibility that basal forebrain cholinergic projections may not directly regulate cerebral cortical vessel reactivities.
In a previous study in baboons (Kiyosawa et al., 1989), a tendency toward reduction in rate constants k1 and k3 was observed, but neither of these decreases was statistically significant. Our results indicate a reduced value of k3 on the injected side of the frontal cortex in both the BPA- and ibotenic acid-treated groups. Methodological differences, including experimental lesioning technique, tomographic sensitivity, size of ROI, rate constant calculation algorithm, and interspecies biological metabolic differences might have contributed to these differences in results for k3. Of course, inhibition of the cholinergic system in the basal forebrain would not lead directly to a reduction in k3. In vitro, acetylcholine can regulate neuronal activities through intracellular signaling processes such as those mediated by adenylate cyclase, phosphoinositide, or phospholipase C (Brown and Brown, 1984; Ashkenazi et al., 1987; Dauphin et al., 1994). It has been shown pharmacologically that acetylcholine effectively excites cortical neurons (Dam et al., 1982; Sato et al., 1987). Thus, neuronal control by acetylcholine via these mechanisms presumably increased the neuronal activity and energy demands of neurons, resulting in increased glucose consumption, which was reflected as the increase in both CMRGlu and hexokinase activity, i.e., k3.
The initially reduced frontal cortex CMRGlu recovered 3 weeks after operation in both the BPA-and ibotenic acid-treated groups. In a study of lesioning by ibotenic acid injection, basal forebrain neurons were considered to be necrosed, but recovery of cortical neuronal activity was observed after 3 weeks (London et al., 1984). The compensatory sprouting of cortical intrinsic cholinergic neurons (Lamour et al., 1982b; Sconcrant et al., 1992; Cossette et al., 1993) may be responsible for the recovery. Our results corroborate this possibility, as 3 weeks after operation there was still a low CMRGlu in the basal forebrain, in which populations of cholinergic neurons would have remained damaged. Recovery after BPA treatment has been speculated to be due to gradual replacement of PDHC inhibited by BPA via intact enzyme transport from basal forebrain cholinergic neurons, thus clearing the inhibition from the cortical area (Weiss, 1969). Therefore, neither of these two experimental procedures mimics the state of permanent suppression of cortical cholinergic neurons as seen in Alzheimer's disease, which is typified by defective cholinergic neuronal activities in the basal forebrain (Whitehouse et al., 1982) as well as in the cortical intrinsic cholinergic neurons due to pathological processes.
Our findings indicate that the cholinergic projection system from the basal forebrain plays an important role in regulating cerebral cortical metabolism through stimulating cortical neuronal activities.
Footnotes
Acknowledgment:
This work was supported by Scientific Research grants (C-05670556, A-04404043) from the Japanese Ministry of Education, Science, and Culture. We thank Dr. S. Matsuzaki for his special support.