Abstract
Keywords
For many years it has been recognized that people suffering from schizophrenia are sick for varying periods of time before they contact any psychiatric service and that the beginning of the first psychotic episode is not identical with the onset of the disorder. According to Häfner [1] there is, on average, a period of 4.5 years between the first signs of schizophrenia and the first hospital treatment. However, in a more recent publication the same author described a prodromal phase of 5 years and a 1-year accumulation of psychotic symptoms in threequarters of a sample of first episodes of schizophrenia [2]. Because of a lack of appropriate diagnostic instruments in the past, usually the time of the first hospital treatment or the beginning of the first psychotic episode have been used as the starting-point for further investigations into the course of the illness. However, this approach does not assess important aspects of the early course of schizophrenia. Consequently, Häfner et al. [3] developed an instrument within the ABC Schizophrenia Study to allow researchers to assess, retrospectively, the onset, development and course of psychotic, especially schizophrenic and paranoid, disorders; this instrument is called the Interview for the Retrospective Assessment of the Onset of Schizophrenia (IRAOS). The IRAOS tries to assess completely, in the sense of stocktaking, the early course of schizophrenia without anytheoretical presumptions and without considering first rank or subordinate symptoms.
The cooccurrence of psychotic disorders and substance abuse and dependence has been a subject of numerous scientific studies; this work has been summarized in several published work reviews [4], [5]. These studies have found that patients suffering from schizophrenia consume more alcohol, nicotine and illicit substances than people without a mental disorder. Furthermore, substance abuse among schizophrenic patients seems to lead to frequent relapses and rehospitalizations. In addition, according to Hoff and Rosenheck [6], dual diagnosis is associated with a significantly increased total cost of care.
In this paper, the term ‘comorbidity’ is used only when schizophrenia and drug or alcohol addiction are simultaneously present, but not as a general term for the presence of two or more defined psychiatric disorders.
This study is concerned with the following questions:
1 Does the pathological symptomatology as assessed by IRAOS influence the pattern of consumption of psychoactive substances? 2 Is this influence different in comorbid patients compared with drug-addicted patients who do not suffer from schizophrenia? 3 Is it therefore possible to identify a subgroup of non-psychotic-addicted patients who do not fulfil the criteria for schizophrenia, but display, as assessed by IRAOS, a similar symptomatology and similar changes in the pattern of substance abuse as found in patients with schizophrenia?
Method
The study was carried out in a rehabilitation hospital for young drug addicts between the ages of 16 and 35 years in a rural area of Germany. The hospital specializes in the treatment of ‘dual diagnosis patients’ who suffer, in addition to the addiction, at least one other defined psychiatric disorder. The rehabilitation of patients who suffer from a comorbid psychotic disorder is of central importance and these patients make up over 50% of all admitted patients. Most of the other patients have comorbid affective disorders, eating disorders, adjustment disorders and personality disorders (for more detailed information see Löhrer [7]). Patients suffering from a borderline personality disorder, who did not fulfil the criteria for schizophrenia, were included in the ‘addiction only group’.
Exclusion criteria for both groups were substance-induced psychotic disorders according to ICD-10. In addition, the ‘psychosis and addiction group’ did not include patients with a diagnosis of psychosis outside the ICD-10 category F20, such as schizoaffective disorders.
The hospital covers a large catchment area, because there are very few other hospitals that specialize in the treatment of dual diagnosis patients. Between 1 August 1997 and 31 July 1998 the average length of treatment for comorbid psychotic patients (the ‘psychosis and addiction group’) was 130 days, and for non-psychotic patients who may have had other psychiatric disorders comorbid with their addiction (the ‘addiction only group’) was 121 days. These figures include patients who terminated the treatment prematurely as well as patients who were discharged for disciplinary reasons. The average length of treatment for those patients who were discharged routinely at the end of their treatmentwas 218 days for the psychotic patients and 222 days for the addicted patients.
A questionnaire, specifically designed for this study, was completed by all patients treated in the hospital during a 3-month period (18 August 1998–18 November 1998). It included items on the date of first drug consumption as well as the type of substances used and the frequency of use. The substances were listed according to ICD-10 [8] namely ‘alcohol’, ‘cannabinoids’, ‘hallucinogens’, ‘cocaine’, ‘volatile solvents’, ‘opioids’, ‘sedatives or hypnotics’ and ‘other stimulants’. However, ‘tobacco’ and ‘caffeine’ were excluded, and a category, ‘biological drugs’, was added to cover biological hallucinogens such as mushrooms that are usually listed in ICD-10 category F16. Frequency of substance abuse was rated in a semiquantitative way according to the Schedules for Clinical Assessment in Neuropsychiatry (SCAN) [9]. For each substance category, the patients were asked to state on how many days per week, on average, or on how manydays overall, each substance was consumed. Possible scores ranged from ‘0’ (no use during the year) to ‘5’ (use on at least 4 days a week). The quantity consumed was not considered. Patients were asked to provide these data on substance abuse at 1-year intervals commencing 10 years before admission. Data covering the ICD-10 diagnostic criteria for addiction and schizophrenia were also collected. In addition, the 64 items of Part IV of the IRAOS were assessed using the ‘lifetime ever’ rating period. Part IV of the IRAOS, which is called Signs and Symptoms of Mental Disturbance, is mainly based on the Present State Examination(PSE) [10] and on the Disability Assessment Schedule (DAS) [11], with the addition of a number of other items.
The IRAOS items were aggregated into five different groups of symptoms, referred to as ‘syndromes’ in this paper, as well as an additional single item covering the patient's first recognition of subjective symptoms [12]. In order to include only syndromes of at least moderate severity, a cut-off point for every syndrome was determined and defined as the mean number of symptoms minus one standard deviation for all patients. The age at first appearance of each syndrome and the frequency of consumption of substances at onset were determined for each patient. PRE represents the onset of ‘non-specific and precursor symptoms’, DEP of ‘non-specific and depressive symptoms’, NEG of ‘negative symptoms’, POS of ‘positive symptoms’ and IMP represents the onset of ‘impaired social adjustment’. SUB is defined as the sub-ject's first recognition of ‘subjective symptoms’.
The data were analysed using a repeated measures anova in SPSS V 7.5.2. Although anova is relatively robust to the violation of some assumptions such as normal distribution, the more critical assumption of sphericity was not met for any of the repeated measures univariate analyses. Consequently, the Greenhouse–Geisser Epsilon was used to correct the degrees of freedom.
Results
During the 3 months covered by the study, 107 patients were treated in the hospital; of these 62 were included in the study with 30 in the ‘addiction only group’ and 32 in the ‘psychosis and addiction group’.
Forty-five patients were not included in the study. For 38 patients the data were incomplete because of lack of cooperation, early discharge, or severity of illness. The remaining seven patients were excluded because they did not meet the diagnostic criteria. The reasons for exclusion were not significantly different between the ‘psychosis and addiction group’ and the ‘addiction only group’. The patients with incomplete or missing data did not differ statistically from those included with regards to sex, age at admission and duration of addiction.
Of the patients participating in the study, 24 (80%) of the ‘addiction only group’ and 28 (87.5%) of the ‘psychosis and addiction group’ were male. The mean age on admission was 25.9 years (median 26 years) for the ‘addiction only group’ and 23.3 years (median 22 years) for the ‘psychosis and addiction group’, which was not significantly different. The mean duration of addiction was 6.6 years (median 6 years) in the ‘addiction only group’ and 5.3 years (median 4 years) in the ‘psychosis and addiction group’, which also was not significantly different. Within the ‘psychosis and addiction group’, 25 subjects fulfilled the ICD-10 criteria for ‘paranoid schizophrenia’, four for ‘hebephrenic (disorganized) schizophrenia’ and three for ‘undifferentiated schizophrenia’. Of all patients, 54 (87.1%) fulfilled the criteria for ‘dependence syndrome because of multiple drug use’, four for ‘cannabinoid dependence’ and one each for ‘alcohol dependence’, ‘opioid dependence’ and ‘sedativeor hypnotic-dependence’. Additional psychiatric diagnoses were not taken into consideration.
Table 1 describes the mean age at first consumption for the ‘addiction only’ and ‘psychosis and addiction’ groups. The age of first consumption of the different substance categories ranged between 13.8 and 19.8 years but, for each substance category, there was no significant difference between the ‘addiction only’ and ‘psychosis and addiction’ groups in age at first consumption.
Age at first consumption
There was a highly significant difference (t=−4.394, df=60, p=0.000) between groups in the mean number of positive IRAOS items with a mean of 25.9 (median 22) for the ‘addiction only group’ and a mean of 38.4 (median 40) for the ‘psychosis and addiction group’.
The mean age at first appearance of ‘non-specific and precursor symptoms’ for the two groups combined was 17.4 years and the time of first appearance was, on average, 1.5 years before the appearance of ‘non-specific and depressive symptoms’ (18.9 years); these appeared approximately 1year before ‘negative symptoms’ (19.6 years), ‘positive symptoms’ (19.7 years), ‘impaired social adjustment’ (19.8 years) and ‘subjective symptoms’ (20.1 years). The difference between the ‘addiction only’ and the ‘psychosis and addiction group’ for age at onset of the syndromes PRE, DEP, NEG, POS, IMP and SUB, respectively, was not significant. The ages at first appearance of some syndromes may be identical in a patient because most patients were very young and the 1-year assessment intervals were approximate only.
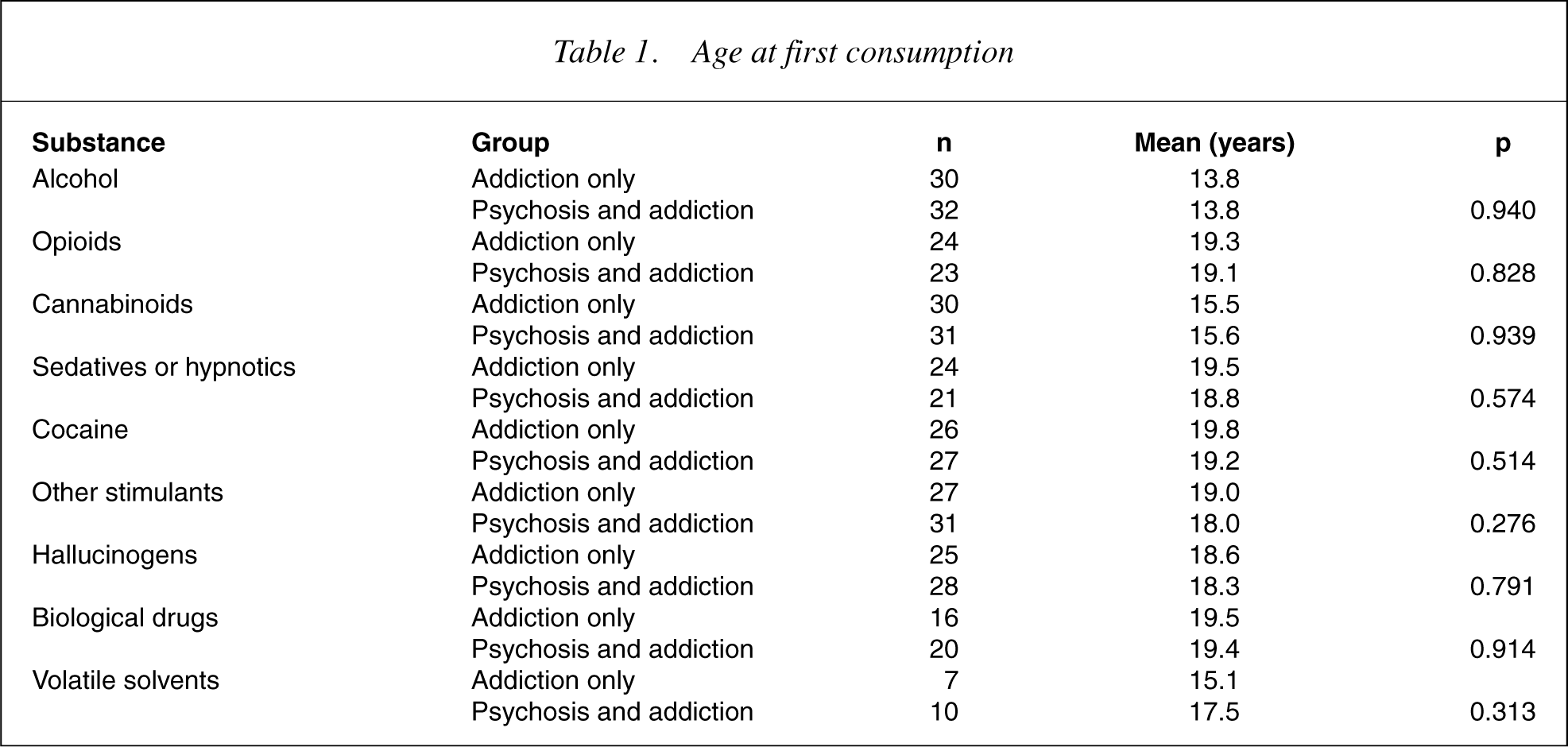
The distribution of heavy substance use at the time of onset of syndromes PRE, DEP, NEG, POS, IMP and SUB was calculated for both the ‘addiction only’ and the ‘psychosis and addiction’ groups. These descriptive data are graphically presented in Fig. 1. ‘Heavy use’ was defined according to ‘clinical reality’, as frequency rating ‘5’ only (use on at least 4 days a week) for the categories ‘alcohol’ and ‘cannabinoids’ and as frequency ratings ‘4’ and ‘5’ (approximately weekly up to 3 days a week, overall at least on 30 days per year) for all other categories.
Percentage of heavy users of substances at onset of syndromes by addiction/psychosis group; separate graph for each substance. PRE: onset of non-specific and precursor symptoms; DEP: onset of non-specific and depressive symptoms; NEG: onset of negative symptoms; POS: onset of positive symptoms; IMP: onset of impaired social adjustment; SUB: subject first recognizes subjective symptoms.
A repeated measures anova was performed in order to test whether the level of substance consumption changed in relation to the onset of the syndromes PRE, DEP, NEG, POS, IMP and SUB and whether these results differed for the comorbid sample compared to the addiction sample. The dependent variable (the within-subjects factor) was the levelofsubstance use at the onset of each of the six syndromes and has been called ‘pathology’, and the independent variable (the betweensubjects factor) was ‘group’, namely, ‘addiction only’ or ‘psychosis and addiction’ (Table 2). In order for a patient to be included in the analysis, data were required for all six items. This led to a very small sample in the ‘addiction only group’ (n=4comparedton=16 for the ‘psychosis and addiction group’). To increase the sample size, single missing values were imputed for 13 patients by examining the pattern of substance abuse for each individual. For example, if a patient did not fulfil the criteria for a particular syndrome, then the missing value for level of consumption was imputed from values available for the other syndromes. Imputation increased the sample size for the ‘addiction only group’ to between 12 and 17, depending on the substance; the sample size for the ‘psychosis and addiction group’ remained the same at 16.
Repeated measures anova: influence of pathology and group affiliation on level of consumption of psychotropic substances
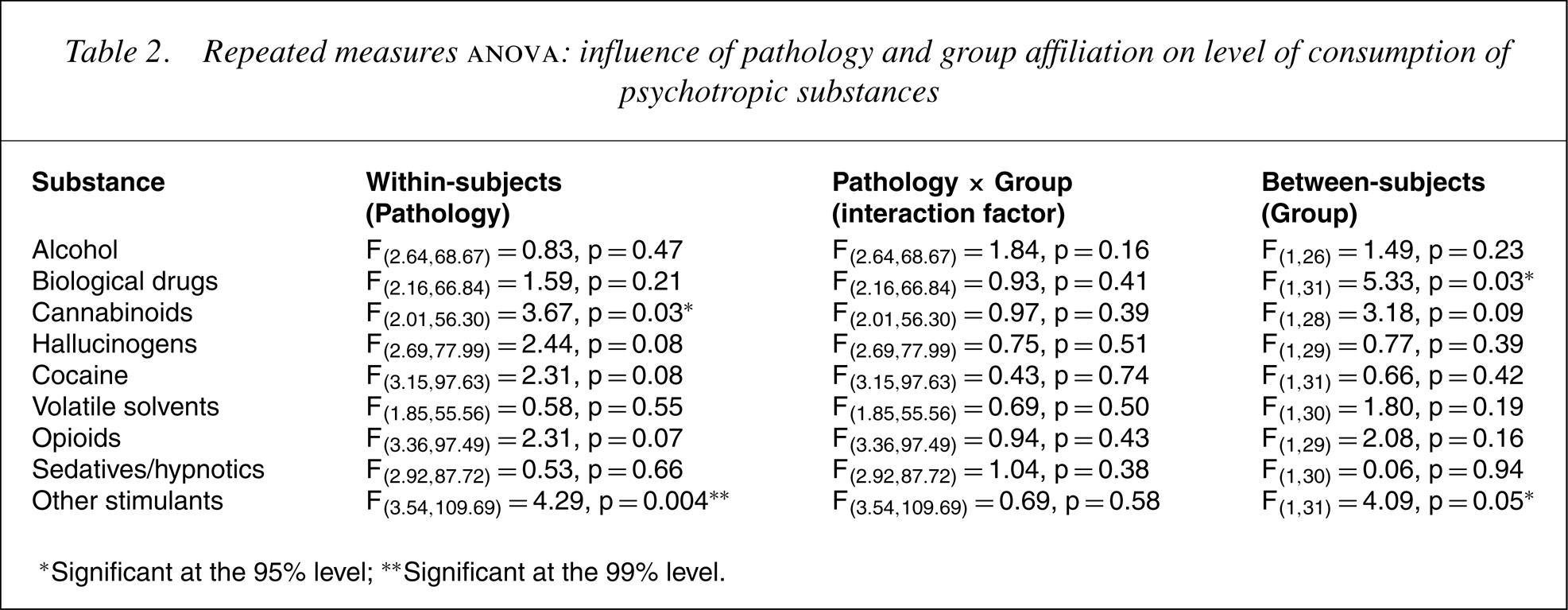
∗Significant at the 95% level; ∗∗Significant at the 99% level.
For the within-subjects analysis, there was no interaction between ‘pathology’ and ‘group’ for any of the substances. The main effect, pathology, was significant at the 95% level for ‘cannabinoids’ and at the 99% level for ‘other stimulants’ and approached significance for ‘hallucinogens’, ‘cocaine’ and ‘opioids’.
The main effect for the between-subjects factor ‘group’ was significant for ‘biological drugs’ and for ‘other stimulants’, with mean levels of consumption higher for the ‘psychosis and addiction group’. There was a similar trend for ‘cannabinoids’ which was significant at the 90% level.
The data were reanalysed using non-parametrical statistical methods (Wilcoxon test, Mann–WhitneyU-test) to test the impact of group status on level of consumption of substances. This additional analysis of the data indicated an increase in the consumption of ‘biological drugs’ in the ‘psychosis and addiction group’ between the times of onset of DEP, NEG and IMP, as well as significant differences in consumption regarding the substances ‘cannabinoids’, ‘opiods’ and ‘other stimulants’ between the two groups. Both these results are supporting the results of the anova.
Discussion
The central result of the study is that pathological symptomatology as assessed by the IRAOS significantly influences the pattern of substance abuse in addicted patients, and that this influence is relatively independent of whether the symptomatology is an expression of the onset of schizophrenia or not. In other words, a similar symptomatology in addicted patients, with and without schizophrenia, influences the pattern of substance abuse of psychoactive drugs in a similar way. This result does not refer to a single pathological symptom but to the syndromes ‘non-specific and precursor symptoms’, ‘nonspecific and depressive symptoms’, ‘negative symptoms’, ‘positive symptoms’ and ‘impaired social adjustment’ concurrently. However, it is possible that the pattern of consumption itself may also influence the pathological symptomatology interactively. In such a case, changes in the pattern of substance abuse are not a result of changing pathological symptomatology but because of changing habits of consumption caused, for example, by changes in the patients' social environment; these changes would themselves influence pathological symptomatology.
Mueser et al. point out that identifying subgroups of comorbid patients has implications for pharmacological and non-pharmacological treatment [13]. Dixon et al., like other authors, state that people with schizophrenia seem to use substances for reasons similar to those of those without schizophrenia [14], [15]. Dixon also points out that: ‘For the most part, research has failed to find patterns of drug choice in persons with schizophrenia that differ from those of other groups of patients with major mental disorders’ [16], although no clear distinction was made with regard to the pattern of symptoms.
One of the limitations of this study is the strong bias towards the selection of patients with a comorbid diagnosis at the specialized institution in which this study was conducted. Among the patients with schizophrenia in the hospital, there were many more with multiple substance abuse than there are found in acute psychiatric wards, in day clinics or outpatient clinics [17], [18]. Among the non-psychotic patients there was still a large number suffering from a second psychiatric disorder, especially from borderline and narcissistic personality disorder, and also from affective disorders and eating disorders. Therefore, this is not a representative sample of comorbid schizophrenic patients or of patients suffering from drug addiction with patients having multiple diagnoses and difficult courses over-represented.
The reliability of the information collected from patients and significant others relating to the onset of symptoms decreases significantly after approximately 5 years between event and inquiry [19]. The situation is probably similar for information about the amount of substance abuse of psychoactive drugs. Hambrecht and Häfner, however, have found a high concordance between patients' and relatives' reports on the onset of substance abuse [20].
It is also important to consider the possibility of a difference in the awareness of illness between the two compared groups. There are indications that the deficit in the awareness of illness in patients with schizophrenia is of significant diagnostic relevance [21].
In the anova, the assumption of normality was violated and the dependent variable, level of substance use at onset of syndromes, was treated as an interval-level measure although, strictly speaking, it was ordinal. Therefore, although anova is robust to departures from normality, the results still need to be interpreted with caution. However, non-parametric methods also supported these findings.
Conclusions
The results of this study suggest that there is a subgroup of non-psychotic addicted patients whose pattern of psychoactive substance abuse is similar to that found in addicted patients suffering from schizophrenia. It may be helpful to systematically identify this subgroup with regard to possible therapeutic implications, particularly with regard to possible pharmacological treatment options.
