Abstract
This study examined the kinematic differences of a bilateral transradial amputee using myoelectric and body-powered prostheses during select activities of daily living. First in harness suspended, body powered then self-suspended externally powered prostheses, the subject's shoulder and elbow joint movements were calculated and compared while completing an elbow range of motion test, simulated drinking from an empty cup, and opening a door. In this case, body-powered prostheses allowed for greater range of elbow flexion but required more shoulder flexion to complete the tasks that required continuous grasp. While using myoelectric prostheses, the user was able to compensate for limited elbow flexion by flexing the shoulder.
Introduction
Previous studies have compared function and use of myoelectric and body-powered prosthetic upper limbs among adults1-3 and prosthetic hands among children.4 These studies have been based on timed functional tests and surveys. Compensatory motion and kinematic differences of unilateral transradial amputees performing common tasks and during kayaking have been studied.5,6 These studies excluded bilateral amputees. The purpose of this study was to compare the shoulder and elbow movements of an amputee subject using bilateral myoelectric and body-powered prostheses while performing a common rehabilitative task in addition to daily tasks that were previously evaluated in healthy persons and myoelectric prosthesis users by the authors.
Case description
The subject was a 32-year-old male journeyman electrical worker. He required mid-length transradial amputations bilaterally secondary to an electrical injury. The subject's residual limbs were burned during the initial injury requiring skin grafting along the anteromedial aspect of both forearms. His limbs were moderately scarred with the left side having more graft coverage than the right side and both sides presented with distal invaginated scars along the surgical margins (Figure 1).
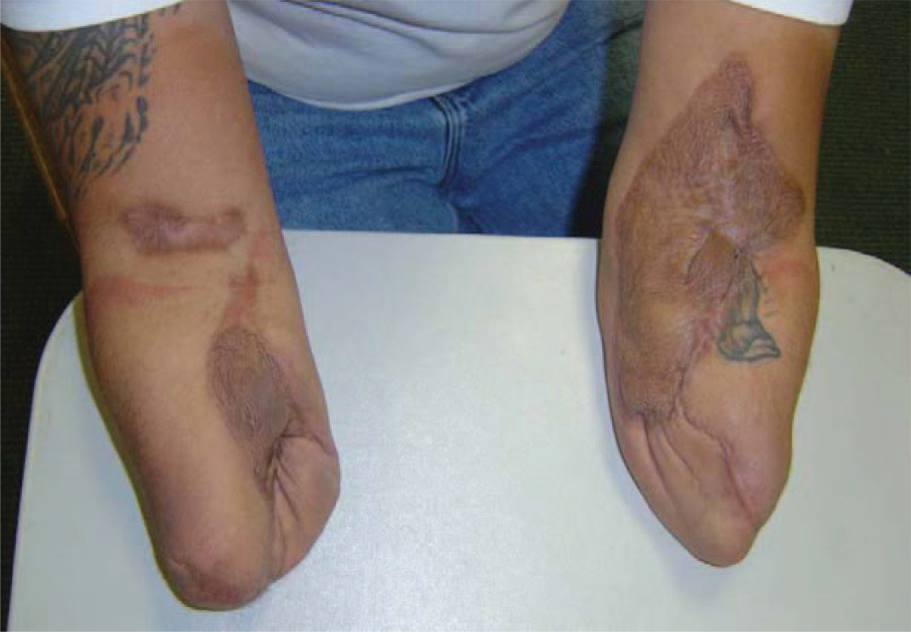
Anterior view of the subject's residual limbs. Graft sites are more extensive on the left side.
The subject's prosthetist reported that manual muscle tests and range of motion were within normal limits at the time he presented for this study. The subject reported that he lived alone and that he was independent with most activities of daily living. He further reported an active lifestyle via routine jogging, weight training, fishing and driving a motorcycle.
The subject was originally fitted bilaterally with body-powered prostheses one year prior to data collection. The prostheses contained hard sockets, flexible hinges and aluminum Hosmer hooks (Campbell, CA, USA), model #5XA bilaterally. The body-powered prostheses were joined together in a continuous figure-of-eight harness. Four months prior to data collection the subject was fitted with supracondylar, self-suspended myoelectric prostheses. The right (dominant) side was fitted with a Greifer terminal device and the left side was fitted with a Sensor Speed terminal device (Otto Bock Healthcare, Minneapolis, MN, USA). At the time of data collection, the subject reported preferential use of his body-powered prostheses for most self-care activities such as grooming, dressing and eating. The subject additionally reported proficient use of both sets of prostheses with no adverse affects. Figure 2 shows the subject with the body-powered (Figure 2a) and myoelectric (Figure 2b) prostheses.

Sagittal, right sided view of subject with prostheses. (a) Body-powered prosthesis; (b) flexible lined, myoeletric prosthesis with Griefer hand.
Methods
The study protocol was approved by the University of South Florida's Institutional Review Board. The subject gave informed consent to participate in the project. An eight-camera Vicon™ motion analysis system (Vicon, Oxford, UK) was used to collect and analyze movement data. Nineteen reflective markers were placed on bone landmarks of the subject's upper limbs, and torso (Table I, Figure 2). Prior to each data collection, the cameras were calibrated according to manufacturer guidelines. Anthropometric measurements of the subject such as height, weight, shoulder depth, and elbow, wrist and hand width were recorded and used to aid in computing joint centers and angles. Only the frontal and sagittal plane movements of the shoulder joint were considered. A static trial was collected to confirm visibility of the markers and to help in the computation of joints centers. Details regarding the kinematic model and analysis of the range of motions are described in previous work by the authors.5,6 The subject was asked to complete the following tasks: Elbow flexion task: With arms hanging comfortably at the sides, flex as far as possible and pause. From the flexed position, the subject was then asked to extend the elbows as far as possible. Simulated drinking from an empty cup: With the preferred (right) hand, the subject would hold an empty plastic cup and attempt to hold a starting position of 90° of elbow flexion and neutral pronation/supination and bring the cup to the mouth. The subject was asked to hold the position as if drinking from the cup then return to the start position. No further instructions or cues were provided in terms of how to approximate the cup to the mouth. Opening a door with a standard door knob: Using either hand the subject was asked to open the door sufficient to walk through it.
The subject completed these tasks first with his myoelectric prostheses and then with his body-powered prostheses.
Reflective marker placement.
Results
Table II shows the maximum, minimum and total range of elbow motion while wearing the body-powered and myoelectric prostheses in the range of motion task. The body-powered prostheses allow for a much greater range of motion of the elbow.
Maximum and minimum elbow flexion while wearing the two prostheses.

Figure 3 shows ranges of sagittal plane shoulder (Figure 3a) and elbow motion (Figure 3b) of the subject while simulating drinking from a cup using the two prostheses. With the body-powered prothesis the subject held the shoulder flexed near 90°and flexed the elbow through a range of 47–100°. The subject reported being ‘aware of the tension’ needed to grasp the compliant cup while using the body-powered prosthesis. Conversely, using the myoelectric prosthesis, the shoulder was flexed from approximately 0° through a 90° range of motion while maintaining 90° of elbow flexion. Though not calculated due to the choice of marker set, it was observed that the subject forward bent the cervical spine while drinking from a cup using the myoelectric prosthesis (Figure 4). The subject also tilted the cup in a way that may have spilled the liquid contained in it, which creates a potential limitation in properly simulating a drinking task.
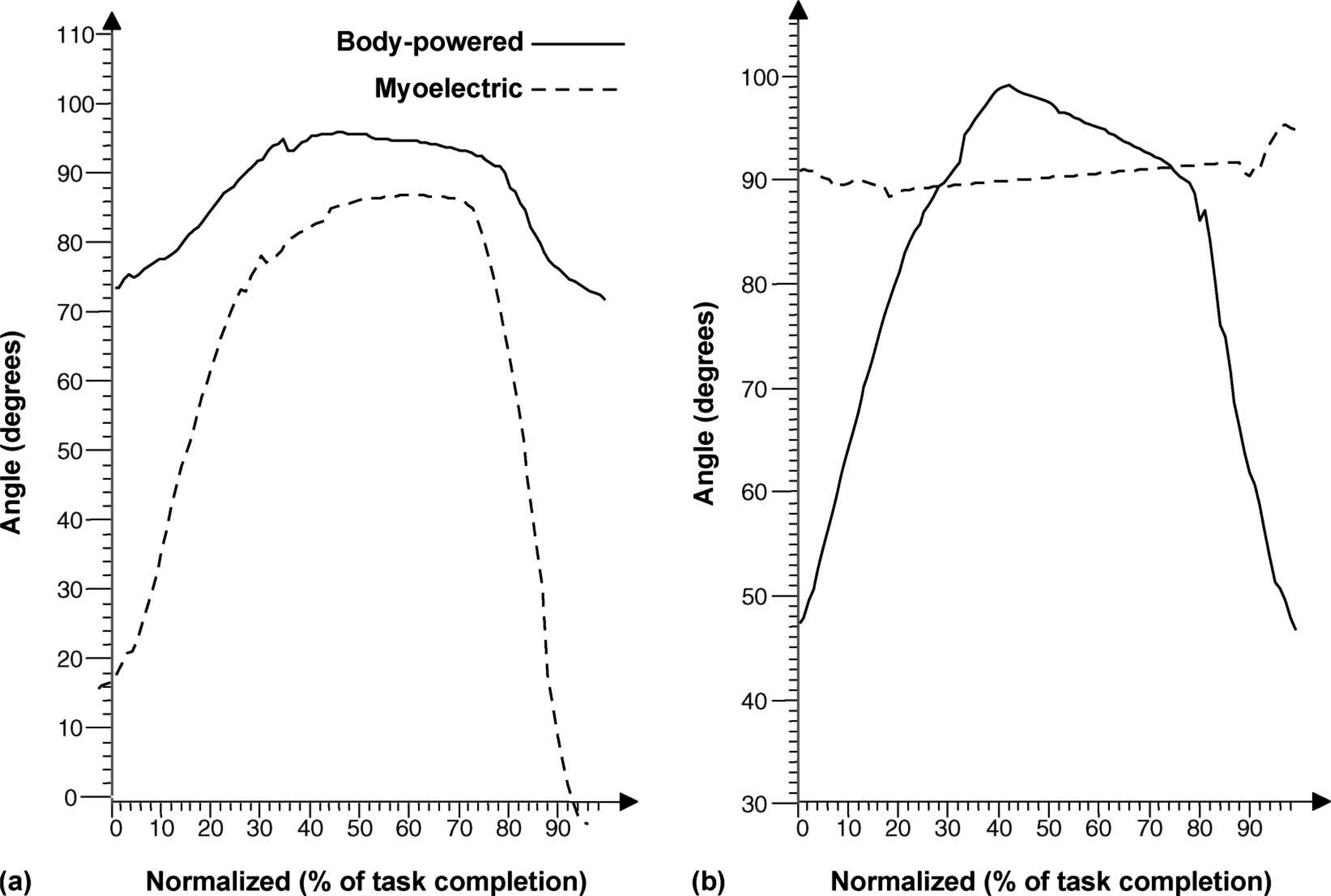
Simulating drinking from a cup (a) Range of motion at the shoulder joint in the sagittal plane; (b) range of motion at the elbow joint.
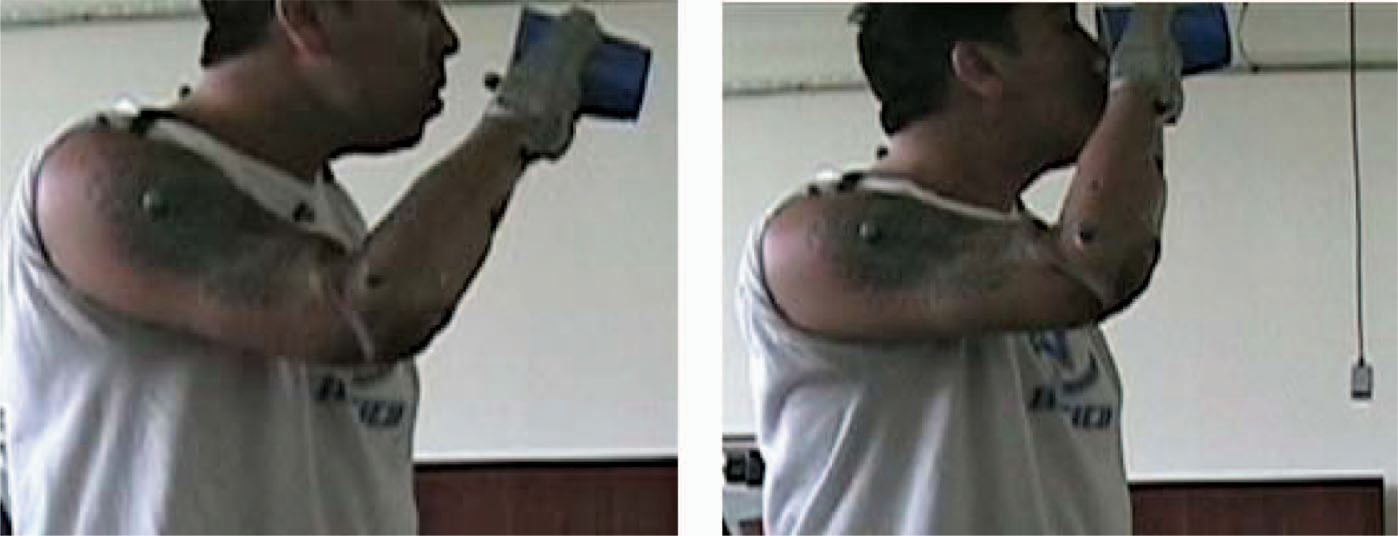
Subject drinking from a cup using a myoelectric prosthesis showing bending of cervical spine.
While using the body-powered prothesis, the subject utilized a greater range of shoulder flexion (24–88°) and elbow flexion (26–81°) when compared to using the myoelectric prosthesis during the door opening task. While using the myoelectric prosthesis, the subject used a shoulder range of flexion from 5–38° and a range of elbow flexion from 21–61° to open the door.
Discussion
Anatomic range of motion of the elbow joint is 0–140° (or 150°). According to Northwestern University,9 so long as the user's anatomic joint can produce the range of motion, a transradial prosthesis should permit 70° of elbow motion in the mid-range. The myoelectric prostheses in this case allowed for 67° on the left side and 41° on the right side. The body-powered prosthesis allowed for a much greater range of motion of the elbow (112° left, 119° right), which is closer to the anatomic range reported in classic goniometric tests.7,8
In order to eliminate a harness suspension system for a self-suspending transradial prosthesis, which arguably enhances cosmesis, higher, more proximal trimlines are required.9 The trimlines in this case were higher with the myoprostheses and incidently, elbow motion was limited relative to the body powered prostheses in all three tasks studied. Though difficult to measure, decreased motivation, a lack of understanding instructions and non-compliance could have also affected these measures.
Carey et al.5 previously reported the kinematics of 10 non-amputee subjects completing many of these same tasks and reported a maximum shoulder flexion of 70° (range of 69°) and maximum elbow flexion of 123° (range of 31°) while simulating drinking from an empty cup. Carey et al. further reported that myoelectric transradial prosthesis users had a maximum of 112° of elbow flexion and range of elbow flexion of 40° during this drinking task. The subject of this study, while using his myoelectric prosthesis to simulate drinking from a cup, held his elbow at approximately 90° and used a very limited range of motion to complete the task. These limitations were compensated for by increasing the range of shoulder flexion and although not measured, bending of the cervical spine to simulate drinking from a cup. The subject in this case utilized his shoulder and elbow joints somewhat inversely between prostheses. Additionally, his completion of the drinking task differed from those reported by Carey et al. in that he utilized less range of motion. By self-report, the subject indicated equal wearing time and equal efficiency in both systems but also indicated a preference for his body-powered systems for most common tasks. It is possible that certain subjective aspects such as preconceived ideas about the limitations of his myoprostheses, comfort/discomfort, being self-conscious about abnormal movements necessary for the completion of functional tasks, all difficult to measure, played roles in the differing kinematic profiles in completing this task.
The greater range of elbow flexion allowed by the body-powered prosthesis in this case was present during both the drinking task and the door-opening task. The subject also utilized more shoulder flexion to open the door with his body-powered prothesis then he did while using the myoprosthesis. In the drinking from a cup task, it seemed that a certain range of motion in the sagittal plane was required to complete the task and that if the shoulder was less mobile then the elbow's motion increased, making up the difference necessary to complete the task. This was not a recurring finding with the door task. Although this greater shoulder flexion cannot be totally explained, it is possibe that the subject was reluctant to move his shoulder harness in such a way as to open the terminal device causing a loss of grip on the door to open. It is also possible that the terminal device was slipping on the door knob requiring increased body motion to capture the required knob rotation that would result in door opening. It may be a benefit in the future to investigate opening a door in two phases: (i) Turning the knob to release the bolt that involves dexterity of the terminal device, and (ii) pulling the door open. It may additionally be beneficial to instrument subjects' harnesses to determine if shoulder elevation is affected by harness tension or vice versa.
A case study does not allow for generalizations to be made regarding the use of body-powered and myoelectric prostheses. Further limitations of this study include the use of a laboratory setting that only allow for the simulation of actual tasks. The cup was empty and compliant and the subject may not have successfully been able to drink from the cup without spilling liquid. The door was free standing. The marker set was limited to the upper limbs and torso. This did not allow measurement of other compensatory motions that may have occurred, such as the bending of the cervical spine or movement of the lower limbs. No measurements were taken during passive range of the elbow while the subject was both wearing and not wearing his prostheses. This limited comparisons of differences between active and passive range of motion which did not allow for the study of internal (e.g., anatomic, psychologic) factors. Future work should also consider the times to complete tasks when comparing the types of prostheses since length of time is an important factor to users when determining prosthetic use.
Conclusion
In this case the subject's myoelectric prostheses limited his range of elbow motion bilaterally. The subject was able to compensate for the decreased elbow motion and complete the tasks by increasing shoulder flexion or bending the cervical spine. While using his body-powered prostheses, a greater range of elbow motion was used in all three tasks studied. Greater maximal shoulder flexion was also utilized with the body-powered prostheses. In the drinking from a cup task this was due to the need for scapular/harness tension needed to maintain constant grip force on the compliant cup and to provide proprioceptive information about the object during manipulation. In the door-opening task, greater shoulder range was possibly utilized to prevent terminal device opening and subsequent loss of grip or because the prehensor was already slipping on the knob requiring increased body motion to actually cause the door to open. Future studies should consider instrumenting the body-powered harness to determine if harness tension has an influential role on range of motion, more detailed study of the door opening task in phases and a comparison of muscle force necesssary to complete tasks using a body-powered versus a myoelectric prosthesis.
Footnotes
Acknowledgements
This project was funded in part by the U.S. Department of Education, Rehabilitation Services Administration Award # H235J050020 “Demonstration Project on Prosthetics and Orthotics”. The authors of this study would like to acknowledge Westcoast Brace & Limb of Tampa, FL, for subject recruitment and Sam Phillips, CP, PhD, Dean of St Petersburg College of Orthotics and Prosthetics, for methodologic insight.
