Abstract
The impetus to develop interventions aiming to prevent the onset of psychotic disorders or at least to minimize their impact is as strong today as it was when Harry Stack Sullivan first wrote about the possibility in 1927 [1]. The rationale for pursuing this aim is self-evident: psychotic disorders are frequently chronic and debilitating illnesses. The latest epidemiological research suggests that the lifetime prevalence of psychotic disorders might be as high as 3.48% [2] and these disorders are known to be associated with appreciable levels of burden as defined by the World Health Organization [3, 4]. Importantly, early treatment in the course of these disorders has been associated with better prognosis for the individual [5–8], particularly if there is minimal delay between the onset of illness and commencement of treatment [9, 10]. Earlier intervention prior to the onset of frank psychosis holds the promise of even better outcomes [11, 12].
PACE Clinic intake and full threshold psychosis criteria

PACE, Personal Assessment and Crisis Evaluation.
In an earlier study at PACE approximately 40% of young people who met UHR criteria developed a psychotic disorder within 12 months despite receiving supportive psychotherapy and any necessary antidepressant or anxiolytic medication [15]. This conversion rate was much higher than the incidence rate in the general population [3] or for individuals who had only a family history of psychosis [4, 16]. The UHR criteria were subsequently adopted by a number of other centres around the world and comparable transition rates were reported [17–20]. It is noted that the transition rate to psychosis in more recent UHR cohorts at various sites has decreased significantly from 40% to <10% in some settings. This may be due to sampling variation, earlier detection of UHR cases, improved efficacy of interventions provided and the possibility that treatment is more likely to be offered to this cohort within mental health treatment settings generally these days [12, 21, 22].
To date, three clinical trials have been published that have evaluated the efficacy of various treatments in reducing the transition rate to psychosis and minimizing functional decline. The first randomized trial, conducted at the PACE Clinic between 1996 and 1999, aimed to determine whether a combination of up to 2 mg risperidone plus cognitive therapy (CogTher; n = 31) was more effective than supportive counselling alone (n = 28) in preventing or delaying the onset of psychosis or in limiting the impact if a full-blown psychotic episode does develop [23]. Although significantly more people in the supportive counselling group developed a psychotic illness by the end of the 6 month treatment period (36% vs 9.7%, p = 0.03), this difference was no longer significant at the end of the 6 month no-treatment follow up (36% vs 19.4%, p = 0.24). Individuals in the combined treatment group who were fully compliant with antipsychotic medication during the treatment phase were significantly less likely to develop psychosis over the follow-up period than the supportive counselling group. Members of both groups reported symptom improvement at the end of the 6 month follow-up phase and this was maintained when they were followed up 3–4 years later [24]. A number of limitations of that study have been noted [23], including small sample size and lack of blind randomization to treatment group. In particular it was felt that treatment was not provided for long enough.
The Prevention through Risk Identification Management and Education (PRIME) North America study compared 12 months treatment with an antipsychotic (olanzapine, 5–15 mg daily, n = 31) with 12 months of placebo treatment (n = 29) [25]. At the end of the treatment phase, 16% of the olanzapine group developed a psychotic episode compared to 38% of the placebo group, a trend level difference (p = 0.08), and the olanzapine group reported greater improvement in level of positive psychotic symptoms than the placebo group [26]. After a second 12 month period during which neither group received treatment, the difference in the conversion-to-psychosis rate remained. The participants who took olanzapine reported significantly greater weight gain over the treatment phase than the placebo group (mean = 8.79 kg, SD = 9.05 compared to mean = 0.30 kg, SD = 4.24). This can be interpreted as skewing the risk–benefit ratio against the pharmacological intervention, at least for this medication at this phase of illness. McGlashan
Finally, a trial conducted at Early Detection and Intervention Evaluation (EDIE) in Manchester, UK reported that only 6% of UHR participants who received cognitive therapy (CogTher; n = 37) for 6 months developed a psychotic disorder compared to 22% of individuals who did not receive any active psychological or medical treatment (n = 23) [27]. Logistic regression analysis indicated that cognitive therapy was associated with significantly lower likelihood of developing psychosis than no treatment (p = 0.028). It was reported that there was a 96% reduction in the odds of developing psychosis in the cognitive therapy group than the monitoring group. This difference remained significant after another 6 months when no treatment was provided, suggesting that cognitive therapy significantly reduced the likelihood of developing psychosis over a reasonably short follow-up period. Over a 3 year period, cognitive therapy was associated with a significantly lower rate of transition to psychosis (when baseline cognitive factors were controlled for) and significantly reduced the likelihood of being prescribed antipsychotic medication [28]. There were a number of methodological limitations, however, in that study. First, the number of participants in the study was small and therefore the power of the analyses was low. The sample size was not assisted by the loss of a number of participants to follow up. The researchers also indicated that it was impossible to maintain blindness to treatment allocation when follow-up assessments were conducted. Finally, the randomization method resulted in uneven group sizes.
Despite methodological limitations, these three studies provide preliminary evidence for the appropriateness of psychological and pharmacological approaches in the treatment of young people who meet UHR criteria. The PACE study suggested that the combined treatment was more effective in reducing transition rates to psychosis during the treatment phase. Over the longer term, however, there was no difference in transition to psychosis between the specific and non-specific treatment approaches unless participants were fully compliant with the antipsychotic medication. The PRIME study suggests that antipsychotic medication – in this case olanzapine – was no more effective than no treatment in reducing transition rates, while the EDIE study suggested that cognitive therapy alone (compared to no treatment) was beneficial in reducing transition rates. The design of those studies, however, did not enable direct comparison of each of the various treatment components to determine which component is most effective or whether a combination or sequence of approaches should be recommended. All of those studies have indicated that the beneficial effects of medication and cognitively oriented psychotherapy declined once the treatment was ceased, suggesting that extending the duration of specific treatments may be more effective in reducing incidence of psychotic disorders over a longer time frame.
In order to further investigate the benefits of providing a range of interventions for young people at incipient risk of developing psychosis, a second randomized intervention trial has been conducted at the PACE Clinic. This study was designed to determine whether pharmacological or psychological treatments alone or in combination were effective treatments for young people at heightened risk of psychosis by comparing the proportion of participants who develop psychosis between the various treatment groups. It also aimed to compare the change on dimensional measures of symptoms, functioning and quality of life during the 12 month treatment and 12 month follow-up phases between the treatment groups. It was hypothesized that the best outcomes (lowest transition rates and most symptomatic and functional improvement) would be achieved by participants who received a combination of antipsychotic medication and cognitive therapy compared to participants who received cognitive therapy and placebo, and participants who received supportive counselling and placebo. An antipsychotic medication/supportive counselling group was not included because this would have increased the number of participants required for the study to unachievable levels, given that the recruitment period was limited by the funding that was available for the study. A separate cohort of young people who met UHR criteria but did not agree to randomization in this trial were also included in the study. This group received standard clinical care from the staff of the PACE Clinic, including psychologically oriented case management and medical care but were not prescribed antipsychotic medication or placebo.
This paper describes the design of the study, baseline characteristics of the study cohort and issues involved with recruitment to the study.
Methods
Setting
This outpatient study was conducted at the PACE Clinic. The PACE Clinic is a subprogram of Orygen Youth Health, a mental health service for young people living in the western region of Melbourne, and the Orygen Youth Research Centre. Further information about the establishment of PACE and the rationale for the service has been previously provided [14, 29].
This study was approved by the North Western Mental Health Research and Ethics Committee. Informed consent was obtained from young people who met intake criteria and were judged fully competent to give consent. Informed consent was also obtained from parent/guardian for participants who were aged under 18 years. Participants were able to withdraw from the study at any time and this decision did not influence the possibility of receiving treatment at the PACE Clinic, other clinics within Orygen Youth Health, or other services provided by the Victorian public health system. This study was carried out in accordance with the Code of Ethics of the World Medical Association (Declaration of Helsinki) and was registered with the Australian Clinical Trials Registry (ACTRN012605000247673).
Participants
All participants were aged 14–30 years. Young people were referred to the PACE Clinic by community contacts including general practitioners, other mental health services, teachers and university health services, drug and alcohol services and other youth support services. An education campaign was conducted to inform potential referrers of the experiences of young people in the early phases of psychotic disorders and the support offered by PACE [14]. Referrals were also received from other programs within Orygen Youth Health.
A thorough clinical assessment was conducted by psychiatrists or psychologists at PACE to determine if entry criteria for the study were met. These criteria were (i) aged between 14 and 30 years; (ii) living in the Melbourne metropolitan area; and (iii) criteria for one or more of three UHR groups (Table 1). Criteria for the UHR groups are operationalized as scores on the first three subscales of the Comprehensive Assessment of At-Risk Mental States (CAARMS), a semi-structured interview and measure developed at the PACE Clinic to determine UHR status as well as to enable prospective assessment of pre-psychotic symptoms [30]. Criteria for psychosis used in the study are shown in Table 1 and were also operationalized using CAARMS scores [30]. Exclusion criteria were (i) known history of a previous psychotic or manic episode (treated or untreated); (ii) known history of a medical condition that may account for symptoms leading to initial referral (e.g., epilepsy); (iii) clinically relevant neurological, biochemical or haematological abnormalities; (iv) serious coexisting illnesses; (v) any previous use of neuroleptic medication (greater than the equivalent of 5 mg of haloperidol for 3 weeks or longer); (vi) any previous or current use of mood stabilizing medication; (vii) history of severe drug allergy; (viii) intellectual disability (IQ < 70); (ix) being pregnant or lactating; and (x) insufficient English language skills to participate in research interviews or psychological treatment without assistance from an interpreter.
Design
This was a single-site double-blind placebo-controlled randomized study with a 12 month treatment phase and a 12 month follow-up phase. Recruitment commenced in August 2000 and ceased in May 2006. The study design is illustrated in Figure 1. All young people who met PACE UHR criteria were given information about the trial by PACE clinicians and the opportunity to participate. Potential participants who declined involvement were asked if they would consent to involvement in an open-label trial of low-dose lithium as a neuroprotective agent (to be reported elsewhere) and/or regular follow up with a research interviewer over a 24 month period and standard clinical care including psychological treatment for presenting problems plus anxiolytic or antidepressant medication (if required) for 12 months at PACE. The majority of treatment sessions and research interviews were conducted on site at the PACE Clinic. In some cases treatment and research interviews were conducted at a setting of the participant's choice (usually home or school). Research interviewers and psychiatrists involved with the study were blind to treatment group. Psychologists were blind to treatment group as far as the design would allow (obviously to the pharmacological but not to the psychological intervention). Participants were blind to treatment they received in the randomized trial.
Study design and flow. CogTher, cognitive therapy; PACE, Personal Assessment and Crisis Evaluation; Supp, supportive counselling.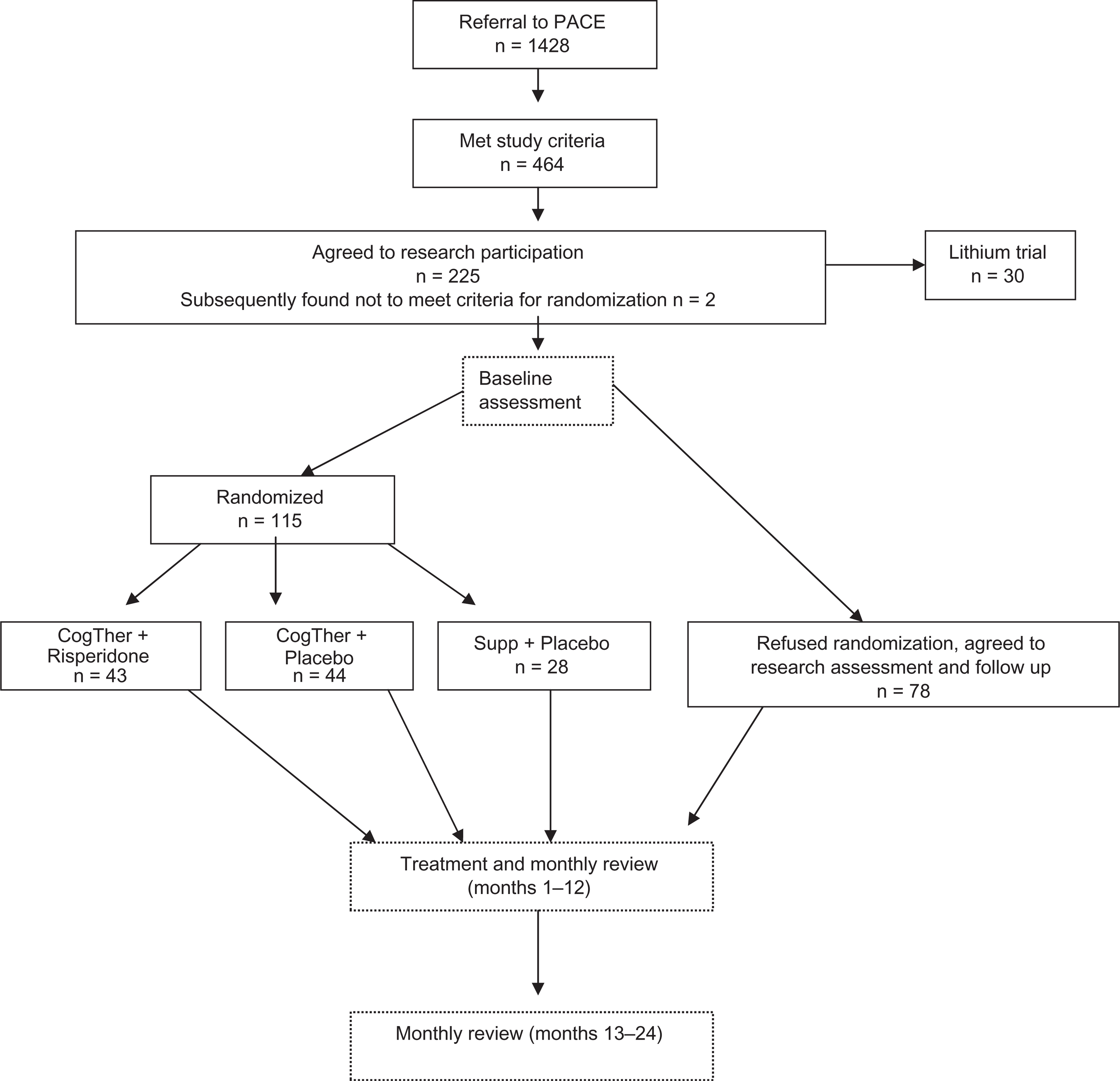
A comprehensive range of assessments was conducted at baseline and monthly for the duration of the treatment and follow-up periods by trained research interviewers. The baseline assessment was conducted prior to randomization and commencement of treatment; in most cases within a fortnight of enrolment in the study. Medical, neurocognitive, and neuroimaging assessments were also performed at baseline and at various points over the subsequent 23 months.
Interventions
The three randomized treatment groups were: cognitive therapy plus up to 2 mg risperidone (CogTher + Risp); cognitive therapy plus placebo (CogTher + Placebo); and supportive counselling + placebo (Supp + Placebo). Participants were randomized according to a 3:3:2 ratio (CogTher + Risp: CogTher + Placebo: Supp + Placebo) due to differences in predicted rates of transition to frank psychosis. The randomization sequence was determined prior to the commencement of the study by a statistician who was not involved in providing treatment or conducting research interviews. The cohort of young people who met intake criteria but did not agree to randomization but who did agree to monitoring and follow up were referred to as the ‘monitoring’ group.
The PACE CogTher intervention has been described previously [14, 31]. Essentially this treatment draws strongly from the stress–vulnerability model of psychosis [32] and emphasizes the development of strategies to cope with pre-psychotic symptomatology and life stressors. This treatment targets the unique concerns, experiences and skills of each individual and relates to a case formulation that is developed collaboratively at the start of treatment by the psychologist and patient [33]. The treatment incorporates specific cognitive behavioural strategies that have been developed to target positive psychotic symptoms [34–38].
Supportive counselling is an active psychological treatment directly assisting individuals to cope with current problems. Cognitive behavioural techniques were not incorporated in this treatment but some psychoeducational information about psychosis and stress management was.
Young people in the monitoring group received standard treatment at the PACE Clinic in response to their presenting issues. This treatment included psychologically oriented case management (including in some cases cognitive therapy) and medical care but did not include antipsychotic medication or placebo.
In addition to providing either the cognitive therapy or supportive counselling, psychologists also provided case management where necessary (assisting patients to address practical issues such as finding housing, arranging social security payments, enrolling in school, applying for employment and so forth) and also monitored level of risk and assisted in providing risk management and crisis intervention as needed. Family education and support was offered to all participants if deemed necessary, regardless of treatment group.
Clinical psychologists who were fully registered with the Psychologists Registration Board of Victoria, were eligible for membership with the Australian Psychological Society College of Clinical Psychologists and had completed a Master of Psychology provided the psychological interventions. Additionally, all had been trained in the provision of cognitively oriented psychotherapy. Each psychologist provided treatment to members of the three treatment groups.
Clearly the psychologists were not blind to the treatment they were providing and hence were aware of which participants were randomized to the Supp + Placebo group. This information was not shared with other members of the PACE research team. Psychological treatment was provided on a weekly to monthly basis depending on level of symptoms, functioning and risk. All psychology treatment sessions were 50–60 min in duration. The number of psychology sessions was not predetermined but psychologists aimed to schedule sessions weekly for the first 6 months of treatment, then fortnightly and finally monthly for the final 3 months. Therefore approximately 35 sessions was aimed at. It was recognized, however, that this schedule could not always be adhered to if the patient had high case-management needs, if symptoms resolved or if patients were unmotivated or non-compliant. Patients could not be compelled to attend sessions unless there was concern about a high risk of harm to self or someone else. A comparison of the number of psychological treatment sessions received by participants in the different treatment groups will be reported in detail in a later paper along with other information about the treatment that participants received.
Weekly supervision meetings were held with psychologists to discuss treatment fidelity. Psychological treatment sessions were audiotaped and reviewed by a psychologist who did not provide treatment within the trial but who was trained in cognitive behavioural therapy and was blind to group allocation. The Cognitive Therapy Scale-Revised [39] and a checklist aimed at differentiating between the cognitive and supportive treatment that was developed by the researchers was completed.
The medication was prescribed and monitored by consultant and registrar psychiatrists. The dose commenced at 0.5 mg and increased by 0.5 mg steps over a 4 week period if no or minimal side-effects were reported. The Udvalg for Kliniske Undersogelser (UKU) side-effect checklist [40] was administered weekly over the first 4 weeks and then monthly from months 2 to 12. Participants were instructed to return all medication packs so that compliance could be monitored.
A research interviewer independently assessed any participant who was thought to have progressed to psychosis by the treating psychologist or psychiatrist and the operationally defined criteria for psychosis threshold was applied. The outcome of this assessment was reviewed by a panel of PACE psychologists and psychiatrists. Treatment was ceased if the psychosis threshold was reached and open-label antipsychotic medication was commenced. The trial treatment was not unblinded under these circumstances but an alternative to risperidone was prescribed because the individuals may have transitioned to psychosis despite taking risperidone. Treatment was also ceased if participants reported unacceptable levels of adverse effects such as weight gain, extra-pyramidal symptoms, lactation and so forth. Treatment was unblinded only in extreme circumstances, for example if a participant overdosed on trial medication.
Measures
In addition to gathering background demographic information, and history of hospitalization for mental health difficulties over the lifetime, the following measures were administered at baseline: Structured Clinical Interview for DSM-IV-TR (SCID-IV) [41], Global Assessment of Functioning (GAF) from the DSM-IV-TR [42], Quality of Life Scale (QLS) [43], CAARMS [30], Brief Psychiatric Rating Scale (BPRS) [44], Scale for the Assessment of Negative Symptoms (SANS) [45], Substance Use Questionnaire (SUQ) developed at PACE based on DSM-IV-TR [42], and the Hamilton Rating Scale for Depression [46]. All of these measures apart from the SCID, GAF and QLS were administered at each follow-up point. The SCID, GAF and QLS were re-administered at months 6, 12 and 24. Information regarding any concomitant medication use was also collected over the duration of involvement in the treatment and follow-up phases.
Analysis
This paper reports baseline characteristics of participants and compares baseline characteristics between participants who agreed to randomization and those who did not (the monitoring group). In line with recommendations from the CONSORT group (http://www.consort-statement.org/index.aspx?o=1088), comparison of baseline characteristics between the treatment groups was not conducted. The statistical software SPSS for Windows 14.0 (SPSS, Chicago, IL, USA) was used and all comparisons were by intention to treat. T-tests and chi-square tests were used to compare measures between the groups. All tests were two-tailed and significance was set at 0.05. Accordingly the effect size of the comparisons was medium (n = 0.6) and the power was 0.98.
Results
Study sample
Of the 1428 young people referred to PACE over the recruitment period, 464 (32.5%) met study criteria and of them 193 (41.6%) agreed to involvement in the randomized trial or to be included in the monitoring group. No difference was found in age (t(488) = −0.75, p = 0.455) and gender distribution (χ2(1) = 0.14, p = 0.707) between young people who met intake criteria and agreed to some research involvement and those who met intake criteria but did not agree to any research involvement at all. A total of 115 young people agreed to randomization (CogTher + Risp, n = 43; CogTher + Placebo; n = 44; Supp + Placebo; n = 28) and another 78 agreed to monthly monitoring (Figure 1).
Participant baseline characteristics
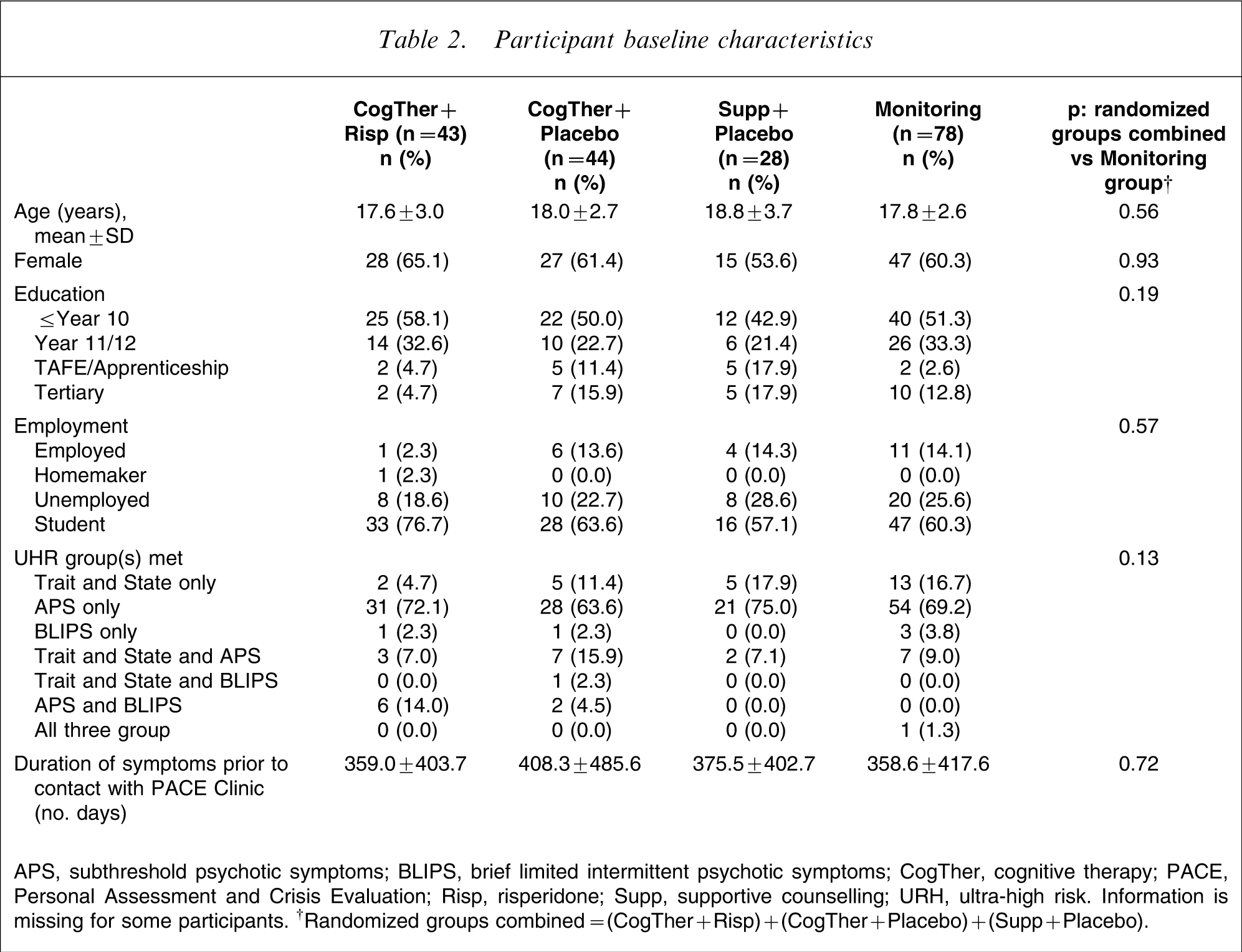
APS, subthreshold psychotic symptoms; BLIPS, brief limited intermittent psychotic symptoms; CogTher, cognitive therapy; PACE, Personal Assessment and Crisis Evaluation; Risp, risperidone; Supp, supportive counselling; URH, ultra-high risk. Information is missing for some participants. †Randomized groups combined=(CogTher + Risp)+(CogTher + Placebo)+(Supp + Placebo).
Baseline levels of symptoms and functioning
Baseline scores on measures of symptoms and functioning
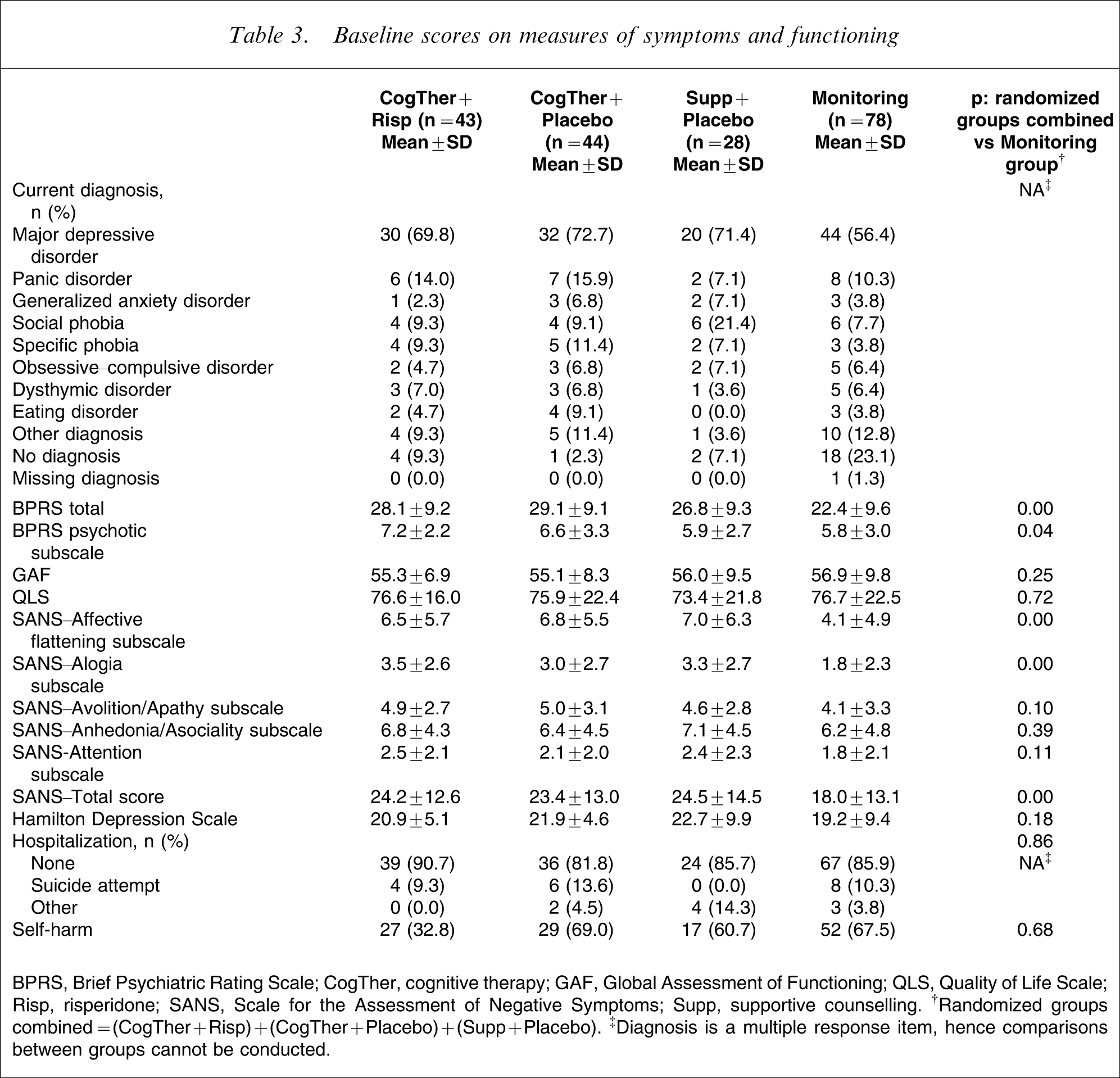
BPRS, Brief Psychiatric Rating Scale; CogTher, cognitive therapy; GAF, Global Assessment of Functioning; QLS, Quality of Life Scale; Risp, risperidone; SANS, Scale for the Assessment of Negative Symptoms; Supp, supportive counselling. †Randomized groups combined=(CogTher + Risp)+(CogTher + Placebo)+(Supp + Placebo). ‡Diagnosis is a multiple response item, hence comparisons between groups cannot be conducted.
Substance use at baseline of 115 participants randomized to the study and 78 who refused randomization
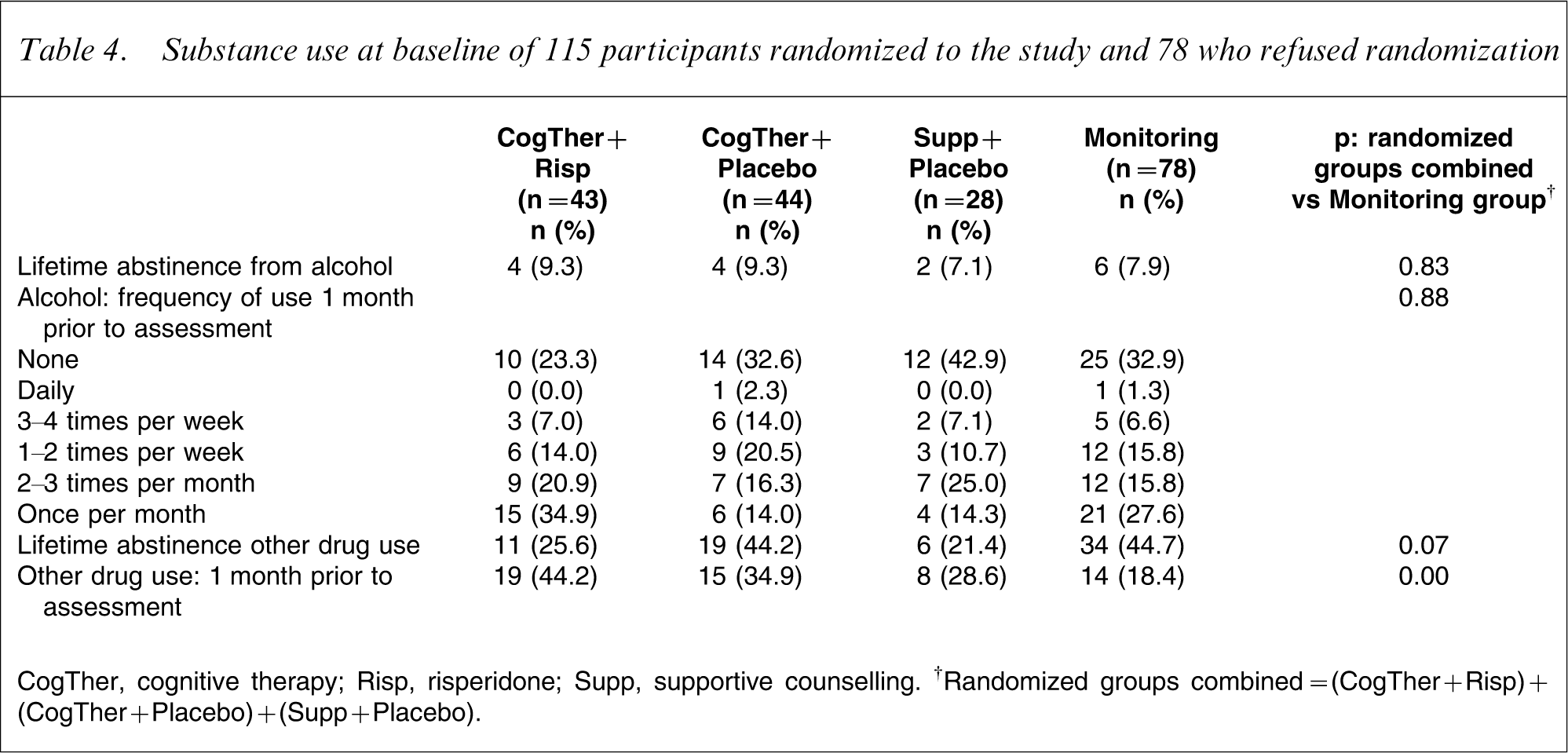
CogTher, cognitive therapy; Risp, risperidone; Supp, supportive counselling. †Randomized groups combined=(CogTher + Risp)+(CogTher + Placebo)+(Supp + Placebo).
Discussion
This is the first double-blind randomized treatment trial with young people who are identified as being at incipient risk of developing a psychotic disorder that has included both psychological treatment and antipsychotic medication. This is a strength of the study over previous intervention studies with UHR cohorts, which have either compared one treatment modality with no treatment or placebo [26, 27], or have not been blinded [23]. Another strength is that treatment was provided for a 12 month period (followed by a second 12 month non-treatment follow-up period). This was in response to the first PACE trial, which concluded that treatment needed to be provided for longer than 6 months [23, 24].
Of the 1428 young people referred to PACE over the recruitment period, 464 (32.5%) met study criteria. Slightly less than half of all eligible young people agreed to involvement in the randomized trial or to be included in the monitoring group (n = 193; 41.6% of all who met entry criteria). The education campaign that was conducted about PACE to support treatment was deliberately broad: referrers were encouraged to refer young people who could possibly meet UHR criteria and were informed that a thorough assessment would be conducted at the Clinic to determine if criteria were met. As previously indicated, many of the young people who were referred to PACE but did not meet UHR criteria were experiencing other clinically relevant difficulties such as major depressive disorder or even full-threshold psychosis and were able to be referred to an appropriate clinical service for treatment either elsewhere in Orygen Youth Health or to other services [14]. The fact that just over one-third of all eligible young people agreed to involvement in research was disappointing, and explanations for such a low rate can only be speculated upon because reasons for refusal were not assessed. Fortunately most people who agreed to involvement in research also agreed to enrolment in the randomized trial. It is thought that participants who agreed to research follow up but did not agree to being included in the randomized trial may have felt uncomfortable not knowing what treatment they were receiving, even though they were aware that being in the trial may have meant receiving more structured and intensive psychological treatment and antipsychotic medication.
Most of the young people who met UHR criteria and agreed to some level of research involvement over the recruitment phase of the study reported significant symptomatic and functional impairment as indicated by scores on the BPRS, SANS, QLS and GAF. Additionally, 87% met diagnostic criteria for at least one DSM Axis I condition, with major depressive disorder being most common, and almost two-thirds of the cohort reported at least one previous occurrence of self-harm. Therefore, treatment of the UHR population is appropriate both due to existing symptomatic and functional impairment and as indicated prevention [47]. This high level of symptomatology and functional impairment also indicates the appropriateness of providing a broad intervention for UHR young people and not just focusing on low-grade psychotic symptoms.
Participants who agreed to randomization were well matched with those in the monitoring group on demographic variables or level of functioning. The monitoring group, however, reported significantly lower levels of global psychopathology, positive psychotic symptoms and negative psychotic symptoms than the randomized participants. It is also noted that 23% of monitoring group members were not diagnosed with a current Axis I disorder at baseline, compared to 6.1% of the randomized cohort. Members of the monitoring group were also significantly less likely to have used drugs other than alcohol in the month prior to baseline assessment and to have been abstinent from drugs other than alcohol during their entire lifetime. Thus, there were significant differences in a number of key areas between members of the monitoring group and those who agreed to randomization that may have influenced the decision not to agree to randomization: young people who were less symptomatic (and potentially less distressed by their symptoms although this was not assessed) may have been wary of receiving what might be seen as more serious treatment (i.e. medication). Alternatively, young people who meet UHR criteria and are more symptomatic may be more willing to try a more targeted treatment approach. It is also possible that clinicians involved in recruitment to the trial were inadvertently more encouraging of randomization when working with a young person who was describing more symptoms, poorer functioning and/or greater levels of distress.
Study limitations
The design of this study had a number of shortcomings. First, the three-group design did not allow for an antipsychotic plus supportive counselling group to assess the effect of all possible combinations of treatment. The reason for this was largely pragmatic: inclusion of another treatment cell would have increased the required participant number substantially and it was unlikely that it would have been possible to recruit such a large number of participants within the funding period. Even so, it took over 5 years to recruit to the study with the current design. Results from other studies (published after the commencement of this trial) suggest that antipsychotic medication is no more effective in reducing transition rates over the medium term than providing no treatment at all [24, 26], and that cognitive therapy alone is more effective than no treatment in reducing transition rates [27, 28]. The ideal study design would enable comparison of the effectiveness of all of the individual potential treatment approaches for the UHR population, but a large-scale multicentre study may be required to recruit sufficient numbers of participants to power such a study.
The supportive treatment that was provided to the Supp + Placebo and monitoring groups was initially intended as a placebo psychological treatment. This did not eventuate because the psychologists, as the primary case workers, had a duty of care to regularly assess the mental state and degree of risk of participants and felt ethically bound to provide some level of active treatment given the level of distress and symptoms reported by this cohort. The clinicians were provided with regular supervision and monitoring to ensure that cognitive behavioural strategies were not included in the treatment, but active problem-solving was permitted. When designing the trial, befriending as described by Sensky
Naturally the psychologists were not blind to the treatment they provided and due to the three-group design were able to identify which participants were in the Supp + Placebo group. The psychologists did not label the treatment they provided to participants as either cognitive therapy or supportive counselling but it is possible that some participants were able to make the distinction. Although the psychologists did not disclose participant group allocation to the psychiatrists or research staff involved in the trial, in some situations this information may have become evident: for example in case review meetings or clinical records.
Summary
This study encompasses several design elements that have not been included in previous treatment studies with UHR cohorts. In particular the design aimed to directly compare the effectiveness of cognitive therapy either alone or in conjunction with antipsychotic medication in reducing the transition rate to psychosis in a UHR group as well as in improving psychological health and quality of life. Although the recruitment rate to this study was slower than anticipated, many young people who meet UHR criteria seek treatment and are willing to enrol in a randomized clinical trial to assist in determining the most appropriate treatment approach for this population. High levels of symptoms and functional impairment were reported by the participants, suggesting that treatment is definitely warranted. There were some significant differences, however, in level of symptomatology, diagnosis and substance use history between young people who met UHR criteria and agreed to randomization to one of the treatment groups and those who did not.
Footnotes
Acknowledgements
We gratefully acknowledge the young people and their families who agreed to involvement in this trial as well as the numerous clinicians and research staff who assisted in conducting this research. We gratefully acknowledge the financial and additional support of Janssen Cilag in the conduct of this study. In particular we thank Dr Ivo Caers for his personal advocacy for this project and Claire Methven for her constant and good-humoured support over several years. Funding was provided through a major investigator-initiated grant from Janssen-Cilag Pharmaceuticals (RIS-AUS-9). Janssen-Cilag had no further role in study design, in the collection, analysis and interpretation of data, in the writing of this report and in the decision to submit this paper for publication. Janssen-Cilag did provide assistance with monitoring the trial and assuring quality of data collection and recording over the treatment phase. Alison Yung, Lisa Phillips and Patrick McGorry have received investigator-initiated funding from Janssen Pharmaceuticals; Patrick McGorry has received investigator-initiated funding from Astra-Zeneca.
