Abstract
Auditory hallucinations (AHs) of spoken speech occur in 60–80% of persons with schizophrenia and often produce high levels of distress, functional disability and behavioural dyscontrol. Insofar as AHs respond incompletely or not at all to treatment in approximately 25% of cases [1], this neuropsychiatric syndrome remains an important public health challenge.
Neurobiology of auditory hallucinations: an integrated picture
The biological basis of hallucinatory symptoms and thought disorder has been extensively examined. Although controversial, some interesting findings have been obtained. Neuroanatomical studies of AHs have not necessarily provided expected results, although several researchers have assumed that the temporal lobe is a possible locus associated with the production of AHs. Havermans et al. examined the auditory P300 and magnetic resonance imaging (MRI) volume measurements in schizophrenia patients with and without AHs [2]. In their study no evidence was found for volume reductions of temporal lobe structures. Upon re-examination, however, of the relationship between temporal lobe volume and AHs, Barta et al. stated that shrinkage of the left superior temporal gyrus (STG) strongly and selectively correlated with severity of AHs [3]. In contrast, DeLisi et al. reported that the volume of the anterior STG did not differ between first-episode schizophrenia patients and controls, nor the presence of formal thought disorder or AHs [4]. Functional neuroimaging studies (positron emission tomography (PET), single-photon emission computed tomography, functional MRI) of hallucinations have indicated a temporal lobe abnormality or a distributed network of cortical and subcortical areas and significantly lower metabolism in auditory and Wernicke's regions [5]. Suzuki et al. suggested that AHs may involve functional hyperactivity in the left STG [6]. In contrast, Silbersweig et al., using PET, reported that AHs were associated with activations in subcortical nuclei (thalamic and striatal nuclei), limbic structures (particularly the hippocampus), and paralimbic regions (parahippocampal and cingulate gyri, as well as orbitofrontal cortex) [7].
A previous study suggested that AHs may stem from abnormalities in brain areas that are involved in the perception of speech. Silbersweig et al. performed PET on six patients with schizophrenia who were hallucinating at the time and demonstrated increased blood flow in the left temporoparietal auditory linguistic association cortex [7]. They found activations in subcortical nuclei (thalamic, striatal), limbic structures and the paralimbic regions (parahippocampal and cingulate gyri as well as orbitofrontal cortex). Thus it was hypothesized that modulation of the orbitofrontal cortex with transcranial magnetic stimulation (TMS) may reduce AHs.
Repetitive transcranial magnetic stimulation in treatment of hallucinations
Hoffman et al [8] conducted the first study using low-frequency repetitive transcranial magnetic stimulation (rTMS; 1 Hz, 80% of MT, 2400 pulses per session) applied to the left temporoparietal cortex (TPC). All patients demonstrated greater improvement in AHs following active stimulation compared to sham stimulation. They concluded that extended duration, low-frequency rTMS applied to the brain region responsible for speech processing could reduce or interrupt AHs. In a recent double-blind sham controlled study Saba et al. Hoffman et al. used left TPC low-frequency rTMS in 18 schizophrenia patients and failed to show superiority over sham stimulation [9]. A review of studies on the effect of rTMS on AH by Saba et al. found rTMS to be active in reducing AH in schizophrenia patients even though negative results were found in a few controlled studies [10]. The two aspects of AH that showed significant improvement with active rTMS compared to sham stimulation, were frequency and attentional salience, whereas other AH parameters such as number of voices, loudness, duration of voices, and level of distress were not affected by any condition. Subsequently, several studies using different study designs showed improvement in AHs after rTMS treatment. Table 1 summarizes the studies to date of rTMS in AH in such patients:
Summary of studies on the effect of rTMS on auditory hallucinations
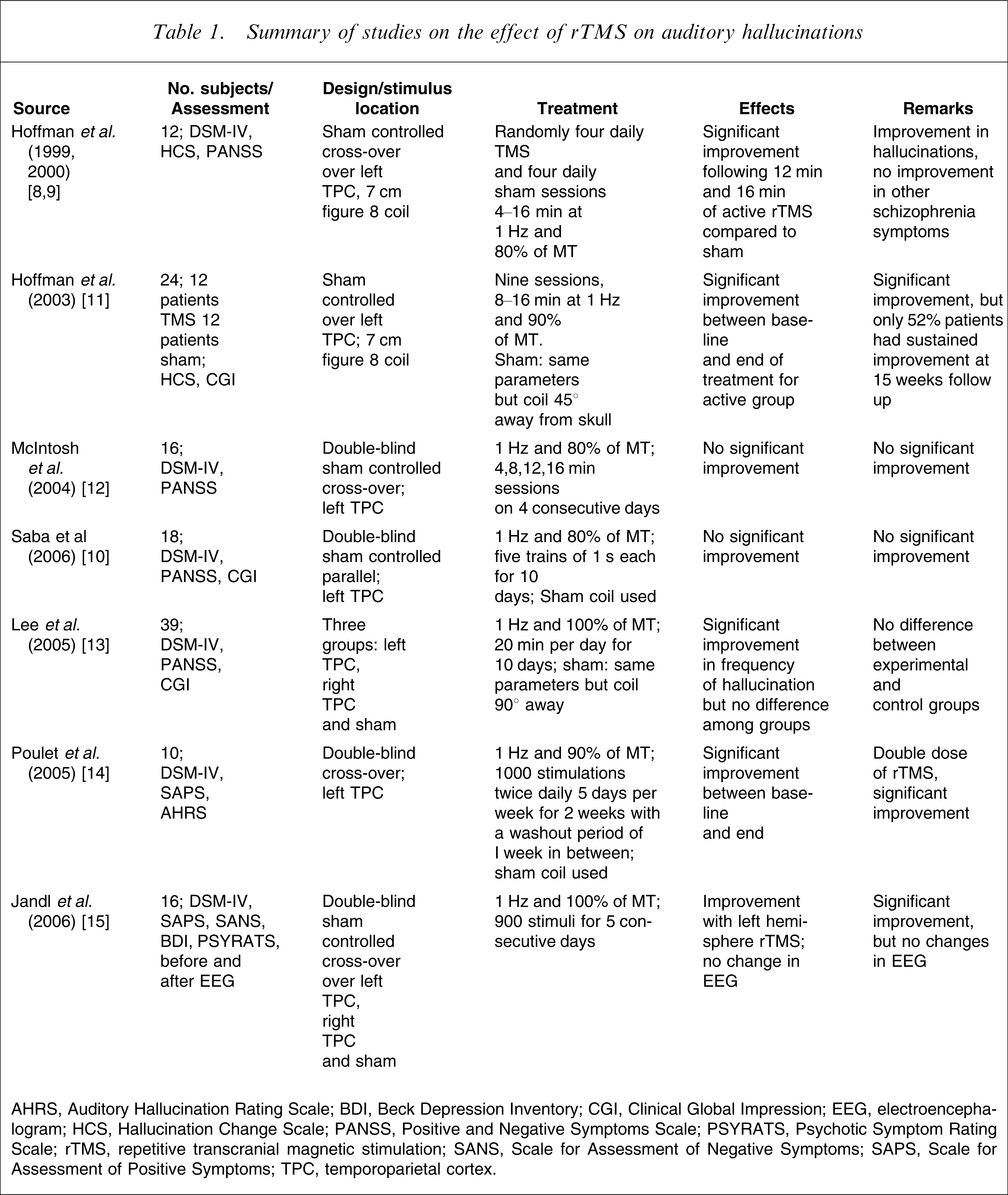
AHRS, Auditory Hallucination Rating Scale; BDI, Beck Depression Inventory; CGI, Clinical Global Impression; EEG, electroencephalogram; HCS, Hallucination Change Scale; PANSS, Positive and Negative Symptoms Scale; PSYRATS, Psychotic Symptom Rating Scale; rTMS, repetitive transcranial magnetic stimulation; SANS, Scale for Assessment of Negative Symptoms; SAPS, Scale for Assessment of Positive Symptoms; TPC, temporoparietal cortex.
Methods
The patients were recruited from both psychiatric wards and on an outpatient basis. Inclusion criteria were ICD-10 diagnosis of schizophrenia and Auditory Hallucinations Rating Scale (AHRS) score >20, right-handedness and normotension. Exclusion criteria included substance dependence except nicotine and caffeine, history of epilepsy, presence of pacemakers or metal parts in the body, and history of electroconvulsive therapy in the past 6 months. Mean duration of illness in the active group was 5.36 years and in the control group, 4.35 years. Written informed consent was obtained after explaining the procedure in detail. To ensure patients were right-handed, the Handedness Preference Schedule was applied. A detailed physical examination was done to rule out any neurological disease. Schizophrenia patients were randomly assigned to the active group (rTMS treatment with antipsychotic medications), and to the control group (antipsychotic treatment only). Prior to the randomization, no change in the medication or the dose of the medication was made in the preceding weeks. Most of the patients were receiving medications for the first time. This allocation was done by the coin-toss method. Overall there were three patients with a diagnosis of treatment-resistant depression (fulfilling the modified Kane criteria). After randomization, two of these patients fell in the experimental group and one in the control group. Table 2 summarizes the sociodemographic characteristics of the patients recruited for the study.
Subject characteristics
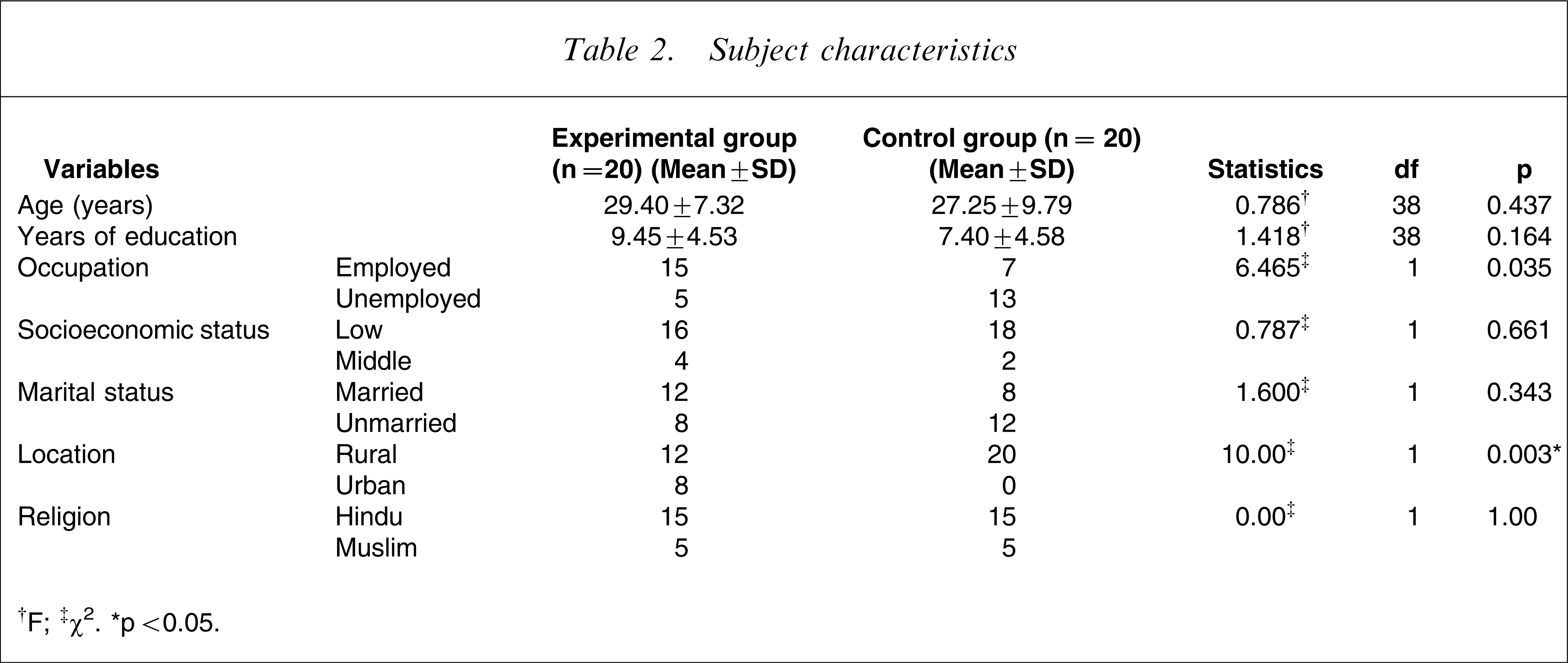
†F; ‡χ2. ∗p < 0.05.
The experimental group consisted of 20 patients with a mean age of 29.40 years (SD = 7.32) and the control group consisted of 20 patients with a mean age of 27.25 years (SD = 9.79). Thirty-six patients were male and four were female with at least 8 years of education in both groups (Table 2). The majority of patients belonged to low socioeconomic status, and significantly more patients in the control group had a rural background (p < 0.05). There were also more unemployed patients in the control group (total of 13) as compared to the experimental group (total of seven). There was no significant difference in the other variables. Chlorpromazine equivalent between experimental and control group did not differ significantly. Most of the patients in the experimental and control groups were on haloperidol and risperidone. Table 3 lists the drugs on which the patients were maintained:
Drugs prescribed
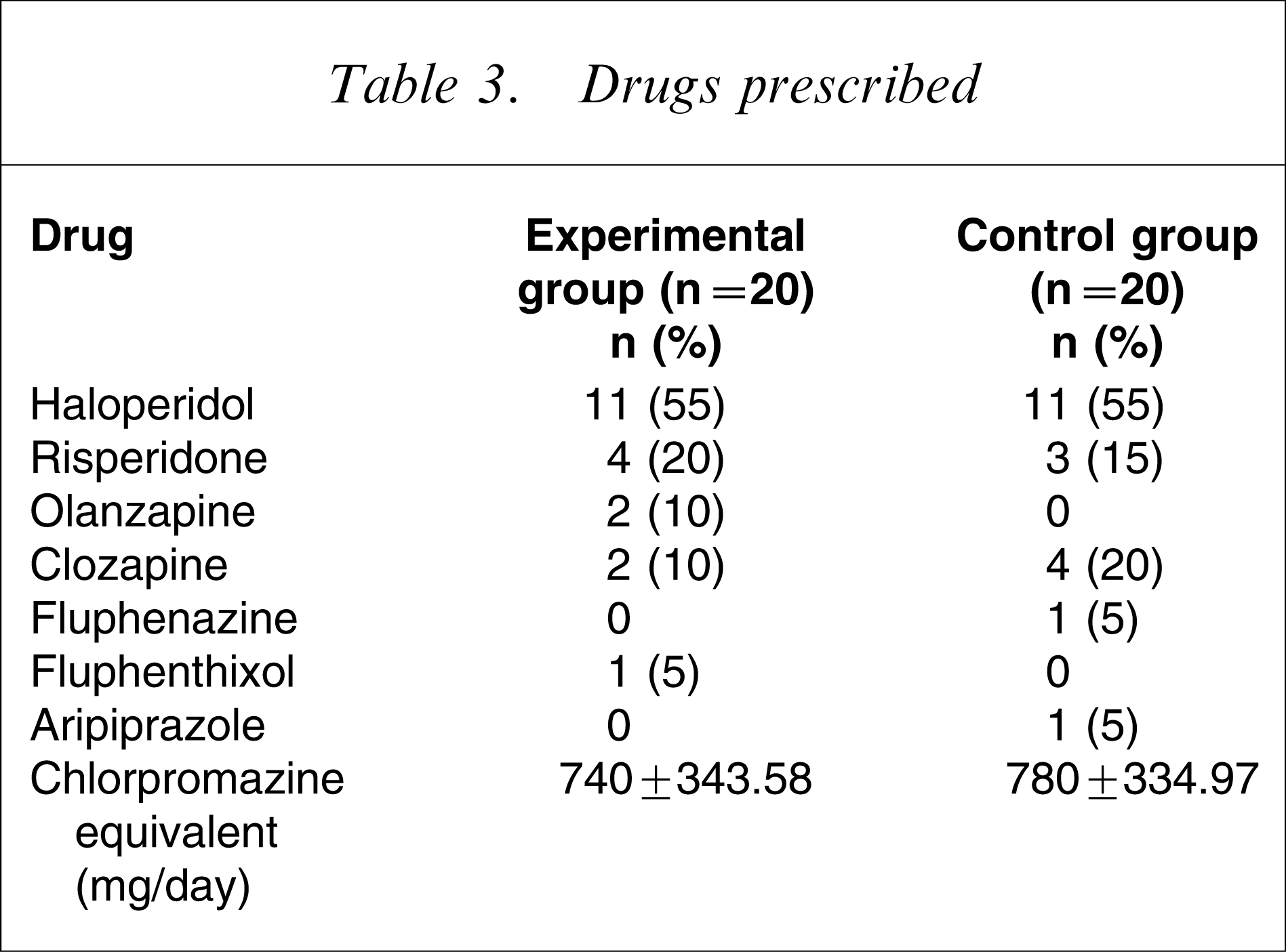
The medications were not changed for the patients for either experimental group or the control group (Table 3), but dosage titration was adjusted as per improvement of psychopathological symptoms.
Patients were allocated after rTMS protocol had been explained to them and written informed consent was obtained. The protocol was approved by institute ethics committee. Complete physical evaluation of the patients was done before enrolment. All the patients were simultaneously receiving antipsychotic also and dosage of antipsychotic were kept the same during the procedure.
The patients receiving rTMS were not blind to the procedure. rTMS was given over the left temporoparietal region at the centre of T3–T4 according to the 10–20 electroencephalography system at 1 Hz and 90% motor threshold. rTMS as adjuvant to conventional pharmacotherapy including the antipsychotics and other drugs was given for 5 days per week for 2 weeks. rTMS was administered using a Magstim Rapid R device (Magstim, Wales, UK) with internal cooling system with figure of eight coil, which has been used worldwide for investigational and therapeutic processes in psychiatry. Because TMS using these conventional coils must be administered by an individual who is aware of the treatment conditions, double blinding could not be done [15]. Hence the possibility of examiner bias cannot be ruled out. The stimulation sessions were performed as an add on, that is, parallel to conventional antipsychotic treatment. The patients were prescribed other medications in a naturalistic manner. Patients underwent rTMS on 10 consecutive days and were rated after 2 weeks.
Ratings
For assessment of AH, the AH subscale of Psychotic Symptom Rating Scale (PSYRATS) developed by Haddock et al. [16], Positive and Negative Symptoms Scale, and Clinical Global Impression were used before and after 2 weeks. The rater was blind to the procedure. The ratings and the rTMS application were done by different individuals so as to prevent the bias.
For rTMS the Magstim Rapid R device (Magstim) was used. It consists of a main unit, control unit, figure of eight stimulating coil and four booster modules. This is a fast repetitive phase device and it is biphasic, that is, the magnetic pulse occurs first in one polarity and then reverses. The main unit of the machine has a charging system with four high-voltage capacitors. During the discharge energy initially stored in the capacitor in the form of electrostatic charge is converted into magnetic energy in the stimulating coil.
Analysis
Analysis of the data was done using SPSS version 13 (SPSS, Chicago, IL, USA). For the purpose of analysis, repeated measures of analysis of variance (ANOVA) was used. The group interaction of psychopathology was also measured with repeated measures (ANOVA). The results of statistical analysis are shown in Tables 4–6.
Results
The AHRS, mentioned earlier in this paper, is the AH subscale of PSYRATS and was the primary outcome measure. The recordings on AHRS was done at baseline and after 2 weeks of rTMS treatment.
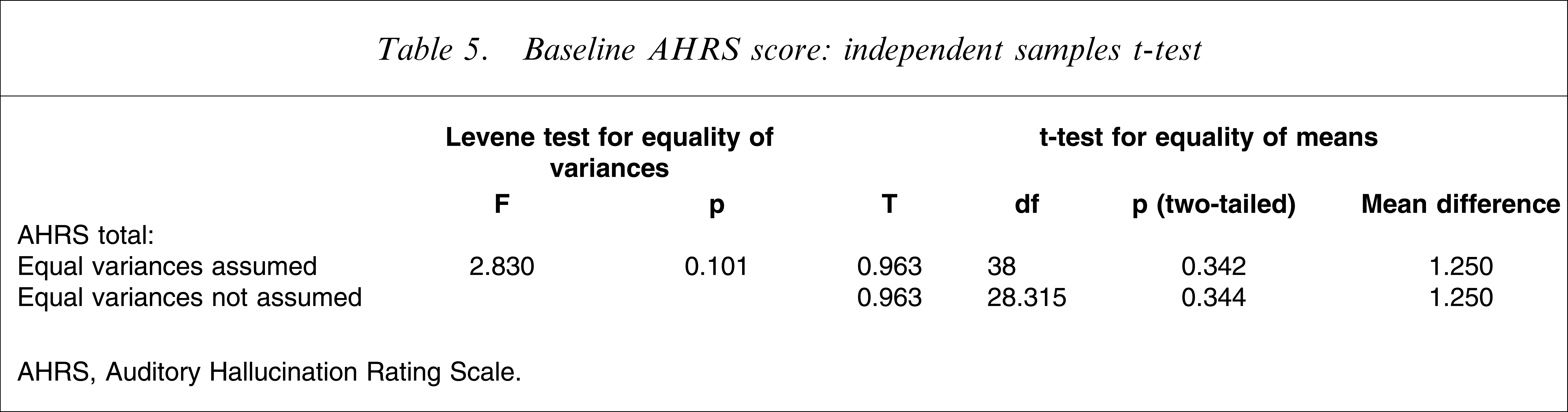
The mean AHRS score was 35.5 for the experimental group whereas the control group mean was 34.25 (Table 4). The mean scores were not significantly different in the two groups at baseline (including the scores of treatment-resistant AHs in both groups).
Auditory hallucination scores at baseline

No significant differences were seen in the AHs scores at baseline in cases of both equal variances and unequal variances (Table 5).
Baseline AHRS score: independent samples t-test
AHRS, Auditory Hallucination Rating Scale.
Effect of treatment was measured using AHRS to quantify the psychopathological domains of patients before and after treatment (repeated measure ANOVA; Table 6). Both groups received similar doses of antipsychotic to control for the effect of antipsychotic agent on change in psychopathological variables. Improvement in AH score was obtained (p < 0.00).
Effect of treatment over time: AHRS scores (repeated-measures ANOVA)

AHRS, Auditory Hallucination Rating Scale; rTMS, repetitive transcranial magnetic stimulation.
∗p < 0.05.
Discussion
Recently, efforts have been made to investigate the effect of rTMS on AHs (Table 1). The present study, however, involved patients with both resistant and non-resistant AHs, which is unique in this context. The number of treatment-resistant patients was higher in the experimental group as compared to the control group (two as compared to one). This further strengthened the study because the effect of rTMS on treatment-resistant AHs could be included in the overall improvement.
Low-frequency (<1 Hz), extended duration (>15 min), rTMS reduces activation in the brain areas directly stimulated as well as in other functionally connected brain areas [7]. The physiological basis of this effect is not well understood but may reflect reduced neuronal excitation or neuroplastic changes associated with long-term depression. The present results raise questions regarding the pathophysiological basis of AHs. The present findings are consistent with studies demonstrating a statistical association between left temporoparietal activation and occurrence of AH [7, 17].
Considered together these findings suggest that low-frequency rTMS delivered to the left TPC would curtail AH by reducing excitation of disturbed neurocircuits that produce these experiences. The present aim was to confirm previous results suggesting the potential utility of low-frequency rTMS administered to the left TPC in reducing both resistant and non-resistant AH in patients with schizophrenia. Although most of the studies to date have used DSM-IV criteria for diagnosis of schizophrenia, we preferred to used ICD-10-DCR for this purpose because of its highly operationalized criteria.
Forty patients with a diagnosis of schizophrenia who were included in the study were randomly allotted to the experimental and control groups, by the coin-toss method. This is one of the few studies to include a sample size of 40 [18]. In the study conducted by Hoffman et al. 50 patients were enrolled and double blinding was done [9], which was not done in the present study. Separate clinicians, however, were involved in the process of rating and rTMS use. In addition, a third clinician was involved in allocating the patients to the rater and to the therapist. This provided a single-blinded procedure, in which the rater did not know the therapeutic status of the patient, thereby minimizing the rater-related bias. The patients, however, were not blind to the intervention procedure, which is the major limitation of the study. This could have produced huge placebo effects, resulting in the differences in results. The possibility of this, however, is rare given the large level of significance (p = 0.005) between the improvement scores for AH on AHRS. Mean patient age in the active group was 29.40±7.32 years, and that in the control group was 27.25±9.79 years. This is consistent with the average age of presentation of the illness at a tertiary centre, reflecting delay due to lack of early identification and assessment of illness [10]. The two groups did not differ in education and socioeconomic status, but did differ in background, with the majority of the patients in the control group being from a rural background (p < 0.05). The two groups did not differ in mean duration of illness: the active group had a mean duration of illness of 5.36±4.30 years, while the control group had a mean of 4.57±4.35 years, which is similar to that seen in other studies [10]. Most of the patients had a diagnosis of paranoid schizophrenia, followed by undifferentiated schizophrenia, which is the usual prevalence of subtypes. Most of the patients in both the active and control group were on haloperidol; the second most common drug was risperidone. The chlorpromazine equivalent of the experimental and control groups was 740±343.58 mg day−1 and 780±334.97 mg day−1, with no significant difference between the two groups, as seen in other trials
Effect of treatment was measured over time, with treatment as a between-group factor; this showed that the rTMS group had a statistically highly significant improvement in AH, which was similar to other studies [18].
The present study was an augmentation study, which involved addition of rTMS to ongoing antipsychotics. Significant reductions of AH parameters of AHRS scores were observed in both groups. In the rTMS group the improvement of AH can be attributed to the use of antipsychotics alone but, given that several patient variables were controlled for (i.e. age, gender, antipsychotics use etc.), and also the randomization of the patients and the single-blinded model of ratings, it cannot be denied that the improvement of the experimental group above the control group must have a component derived from rTMS. In addition, the level of significance of the difference between the groups was very high (p < 0.005), which again supports the validity of the improving effect of rTMS on AH. Finally, the difference in AHs from baseline in both groups was again significant, with the experimental group having a bigger difference. This again adds strength to the evidence.
Footnotes
Acknowledgements
We thank Professor Paul B. Fitzerald for his help with this paper.
